

高等学校化学学报 ›› 2022, Vol. 43 ›› Issue (7): 20220248.doi: 10.7503/cjcu20220248
收稿日期:2022-04-14
出版日期:2022-07-10
发布日期:2022-05-10
通讯作者:
宁汇
E-mail:ninghui@upc.edu.cn
基金资助:
SONG Dewen, WANG Mingwang, WANG Yani, JIAO Zhenmei, NING Hui( ), WU Mingbo
), WU Mingbo
Received:2022-04-14
Online:2022-07-10
Published:2022-05-10
Contact:
NING Hui
E-mail:ninghui@upc.edu.cn
Supported by:摘要:
近年来, 二氧化碳过量排放所引发的全球变暖等气候问题引起了全世界的广泛关注, 碳减排已成为人类社会可持续发展面临的共同挑战. 利用电化学方法将二氧化碳转化为高附加值化学品是实现碳减排和二氧化碳高附加值利用的理想途径之一, 但仍面临能耗高、 二氧化碳转化率低、 产物选择性差和难分离等问题. 本文以电还原二氧化碳制草酸为例, 从反应机理、 催化剂、 电解液、 催化电极及反应器等方面介绍该反应的研究进展, 对当前二氧化碳电还原制草酸存在的关键问题进行了分析, 并对其未来研究方向进行了展望.
中图分类号:
TrendMD:
宋德文, 汪明旺, 王亚旎, 焦振梅, 宁汇, 吴明铂. 二氧化碳电还原制草酸研究进展. 高等学校化学学报, 2022, 43(7): 20220248.
SONG Dewen, WANG Mingwang, WANG Yani, JIAO Zhenmei, NING Hui, WU Mingbo. Progress of CO2 Electroreduction to Oxalic Acid. Chem. J. Chinese Universities, 2022, 43(7): 20220248.
| Reaction | Eθ/V(vs. SHE) |
|---|---|
| CO2(g)+e-→CO2- | -1.900 |
| CO2(g)+2H++2e-→CO(g)+H2O(l) | -0.530 |
| CO2(g)+H2O(l)+2e-→CO(g)+2OH-(aq) | -1.347 |
| CO2(g)+2H+(aq)+2e-→HCOOH(l) | -0.610 |
| CO2(g)+H2O(l)+2e-→HCOO-(aq)+OH-(aq) | -1.491 |
| CO2(g)+4H+(aq)+2e-→HCHO(l)+H2O(l) | -0.480 |
| CO2(g)+3H2O(l)+4e-→HCHO(l)+4OH-(aq) | -1.311 |
| CO2(g)+6H+(l)+6e-→CH3OH(l)+H2O(l) | -0.380 |
| CO2(g)+5H2O(l)+6e-→CH3OH(l)+6OH- | -1.225 |
| CO2(g)+8H+(aq)+8e-→CH4(g)+2H2O(l) | -0.240 |
| CO2(g)+6H2O(l)+8e-→CH4(g)+8OH-(aq) | -1.072 |
| 2CO2(g)+12H+(aq)+12e-→C2H4(g)+4H2O(l) | -0.349 |
| 2CO2(g)+8H2O(l)+12e-→C2H4(g)+12OH- | -1.177 |
| 2CO2(g)+12H+(aq)+12e-→CH3CH2OH(l)+3H2O(l) | -0.329 |
| 2CO2(g)+9H2O(l)+12e-→CH3CH2OH(l)+12OH-(aq) | -1.157 |
| 3CO2+18H+(aq)+18e-→CH3CH2CH2OH(l)+5H2O(l) | -0.320 |
| 2CO2(g)+2H+(aq)+2e-→H2C2O4(l) | -0.913 |
| 2CO2(g)+2e-→C2O | -1.003 |
| 2H+(aq)+2e-→H2(g) | -0.420 |
Table 1 Standard electrode potential for electrocatalytic reduction of CO2 to different products[8,9](pH=7)
| Reaction | Eθ/V(vs. SHE) |
|---|---|
| CO2(g)+e-→CO2- | -1.900 |
| CO2(g)+2H++2e-→CO(g)+H2O(l) | -0.530 |
| CO2(g)+H2O(l)+2e-→CO(g)+2OH-(aq) | -1.347 |
| CO2(g)+2H+(aq)+2e-→HCOOH(l) | -0.610 |
| CO2(g)+H2O(l)+2e-→HCOO-(aq)+OH-(aq) | -1.491 |
| CO2(g)+4H+(aq)+2e-→HCHO(l)+H2O(l) | -0.480 |
| CO2(g)+3H2O(l)+4e-→HCHO(l)+4OH-(aq) | -1.311 |
| CO2(g)+6H+(l)+6e-→CH3OH(l)+H2O(l) | -0.380 |
| CO2(g)+5H2O(l)+6e-→CH3OH(l)+6OH- | -1.225 |
| CO2(g)+8H+(aq)+8e-→CH4(g)+2H2O(l) | -0.240 |
| CO2(g)+6H2O(l)+8e-→CH4(g)+8OH-(aq) | -1.072 |
| 2CO2(g)+12H+(aq)+12e-→C2H4(g)+4H2O(l) | -0.349 |
| 2CO2(g)+8H2O(l)+12e-→C2H4(g)+12OH- | -1.177 |
| 2CO2(g)+12H+(aq)+12e-→CH3CH2OH(l)+3H2O(l) | -0.329 |
| 2CO2(g)+9H2O(l)+12e-→CH3CH2OH(l)+12OH-(aq) | -1.157 |
| 3CO2+18H+(aq)+18e-→CH3CH2CH2OH(l)+5H2O(l) | -0.320 |
| 2CO2(g)+2H+(aq)+2e-→H2C2O4(l) | -0.913 |
| 2CO2(g)+2e-→C2O | -1.003 |
| 2H+(aq)+2e-→H2(g) | -0.420 |
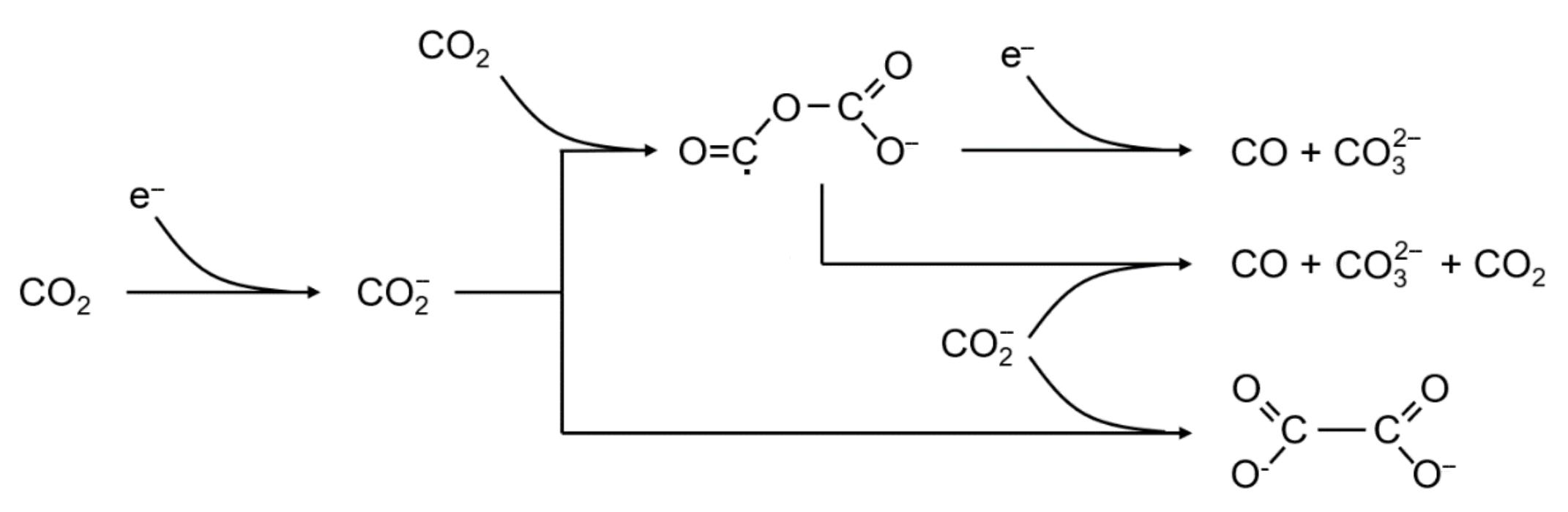
Fig.1 Suggested reaction mechanisms for the disproportionation to carbonate and carbon monoxide (above, middle) and the dimerization to oxalate(below) in aprotic media
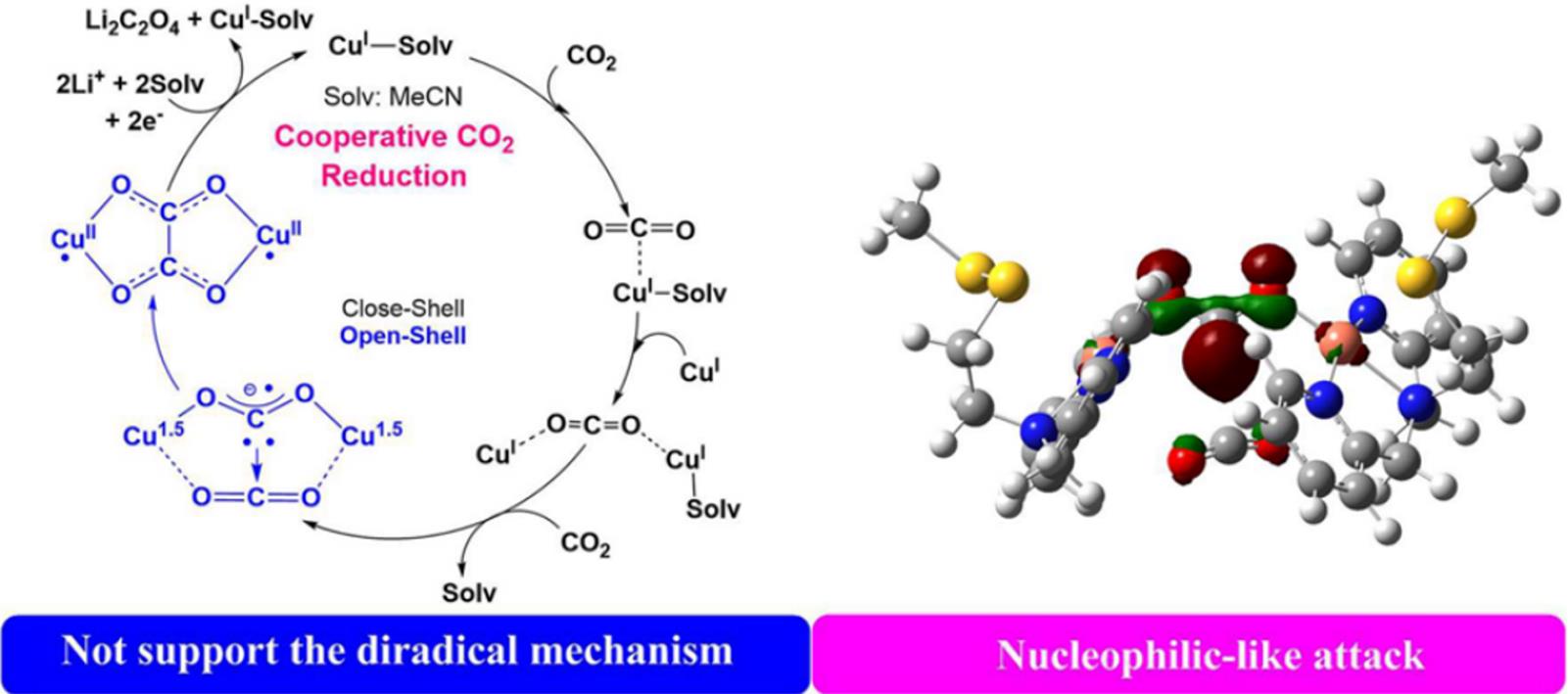
Fig.4 Schematic representation of the mechanism of selective oxalate formation mediated by two cationic copper(Ⅰ) complexes[33]Copyright 2017, American Chemical Society.
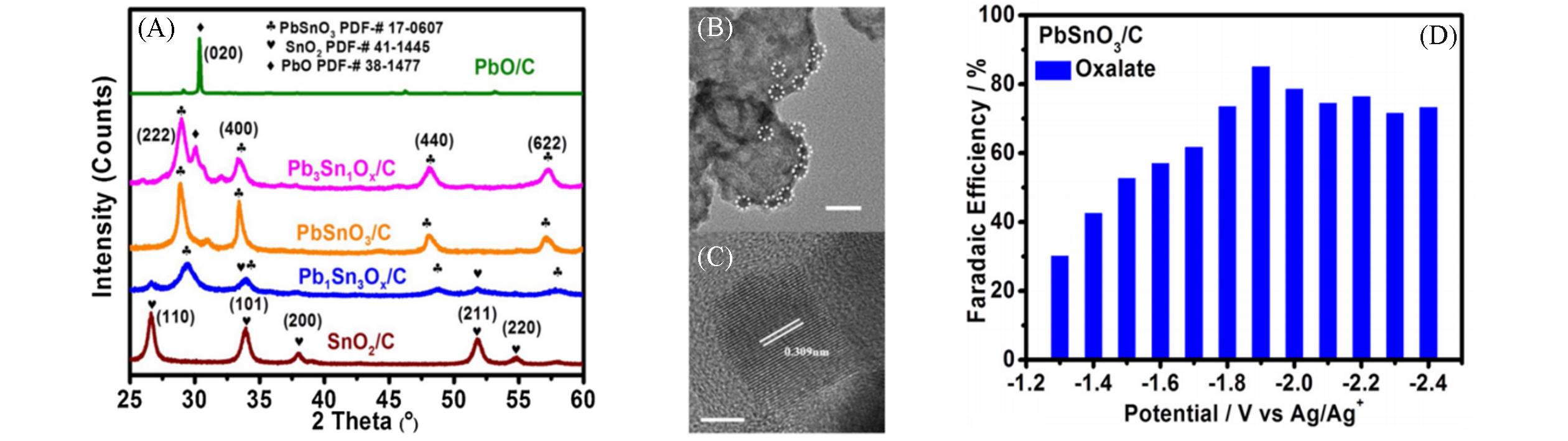
Fig.5 XRD patterns of PbO/C, Pb3Sn1O x /C, PbSnO3/C, Pb1Sn3O x /C, SnO2/C(A), TEM(B) and HRTEM(C) images of PbSnO3/C and Faradaic efficiencies for oxalate of PbSnO3/C(D)[34]Copyright 2020, Elsevier.
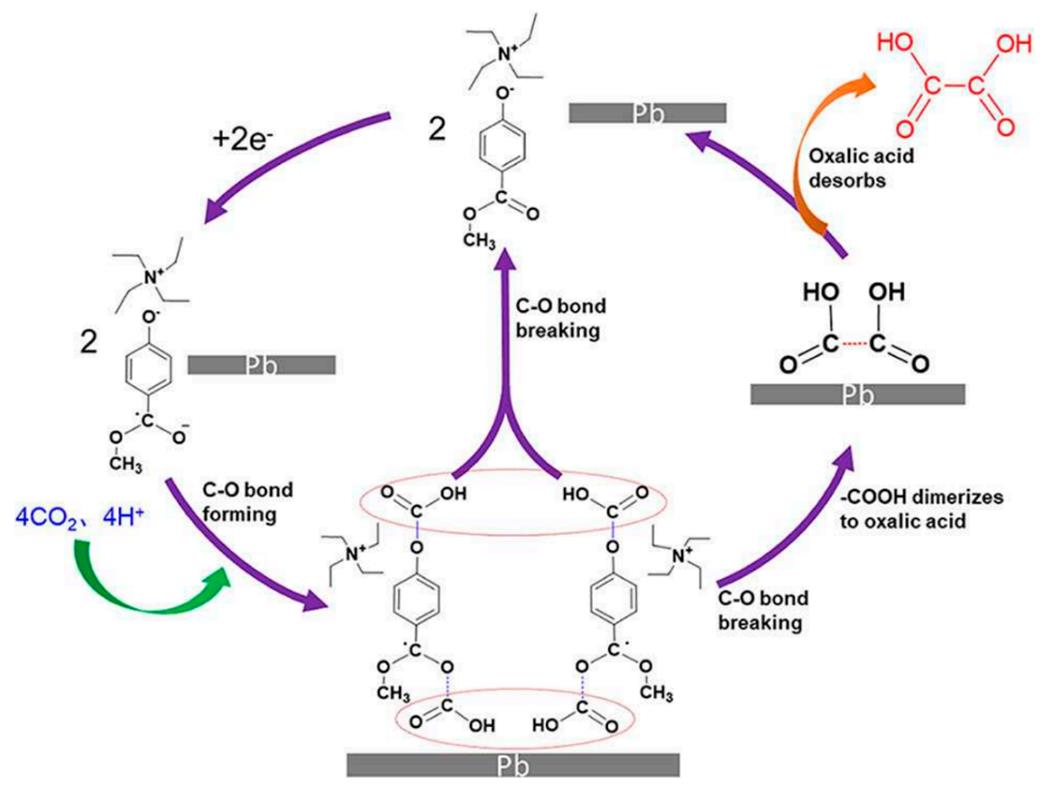
Fig.6 Mechanism of CO2 electroreduction to oxa?lic acid on Pb electrode in the [TEA][4?MF?PhO](0.9 mol/L)?AcN electrolyte[50]Copyright 2020, Wiley?VCH.
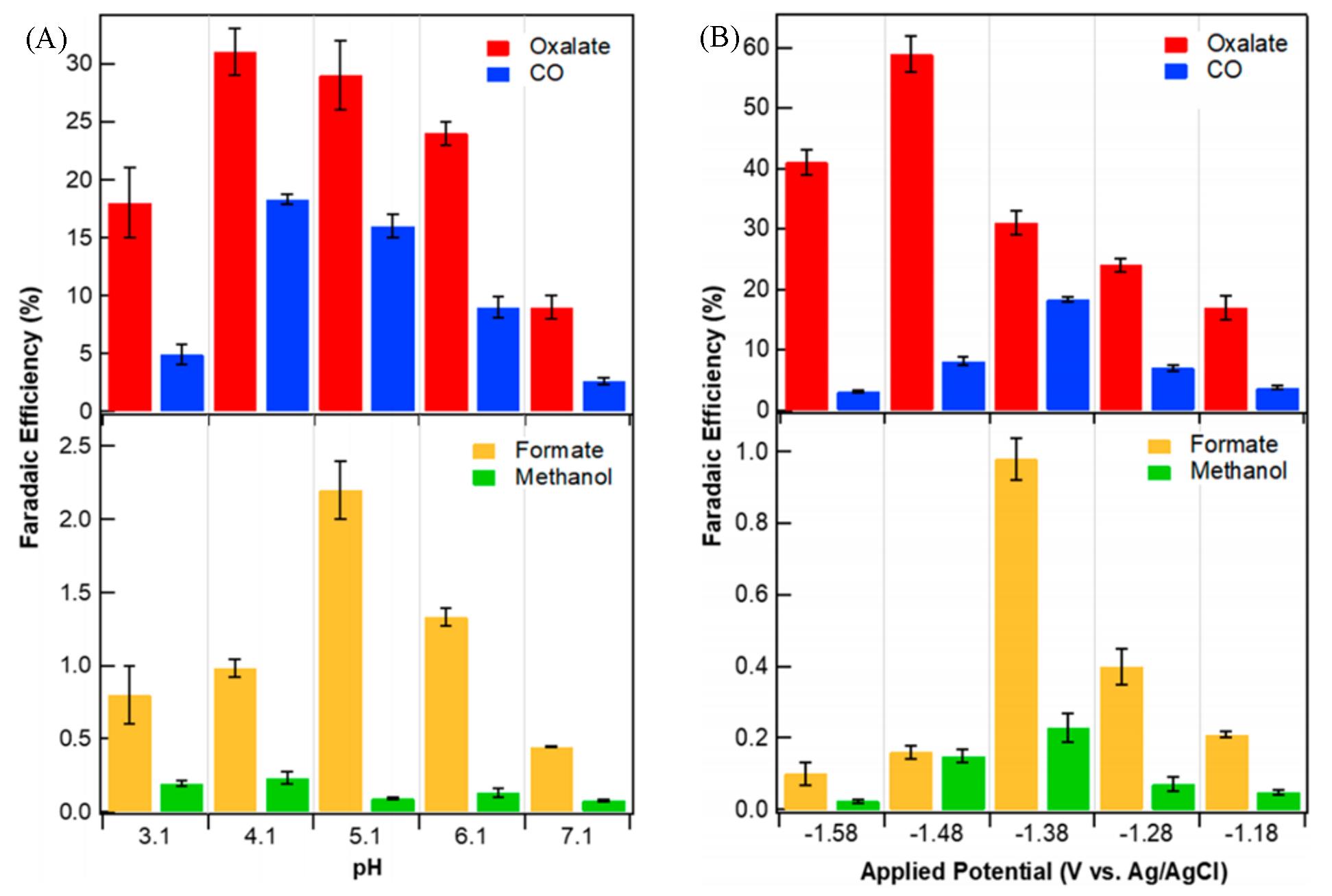
Fig.7 pH dependence(A) and potential dependence(B) of Faradaic efficiencies for carbon?containing products[27](A) Experiments were performed at -1.38 V(vs. Ag/Ag+) using CO2?saturated 0.1 mol/L KCl buffered with KHCO3(pH>4) or adjusted with HCl(pH<4); (B) pH=4.1, with 0.1 mol/L KCl as electrolyte.Copyright 2019, American Chemical Society.
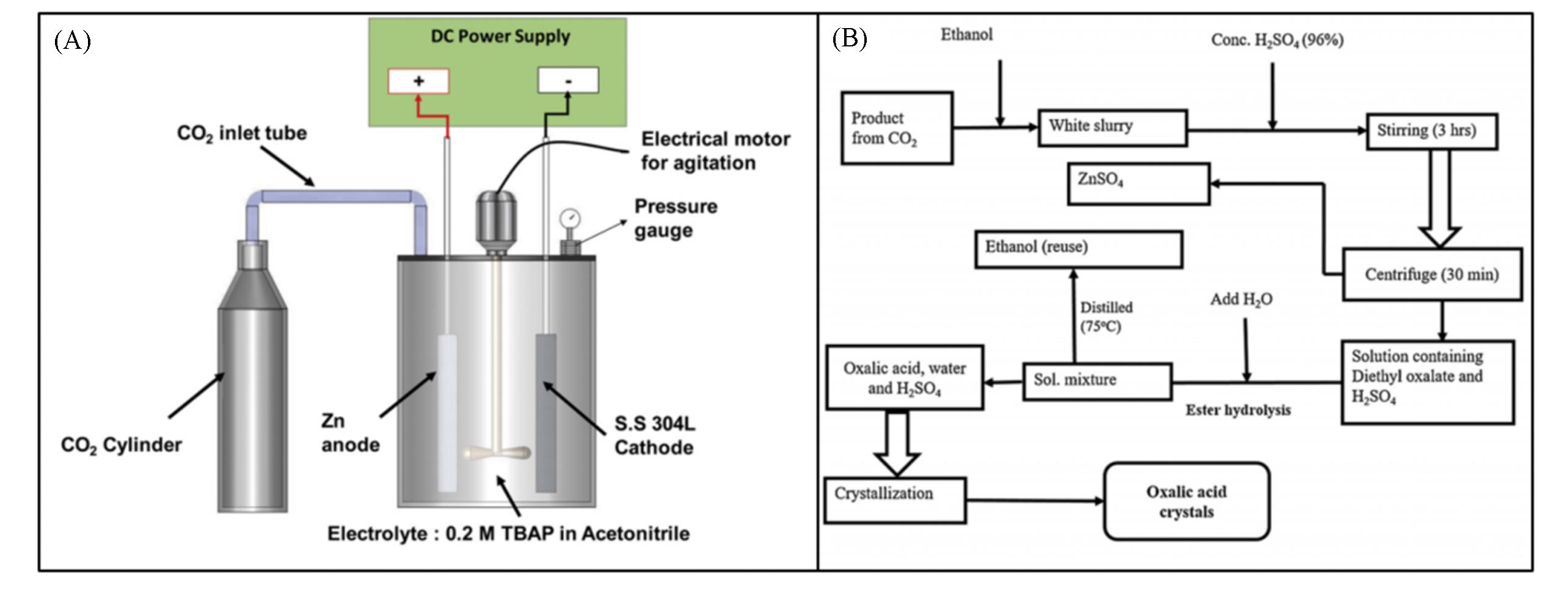
Fig.8 Electrochemical setup for the conversion of CO2 to oxalate in a batch reactor at 2 bar pressure(A) and flowchart of ester hydrolysis process used for the extraction of oxalic acid from zinc oxalate(B)[43]1 bar=100 kPa. Copyright 2019, Elsevier.
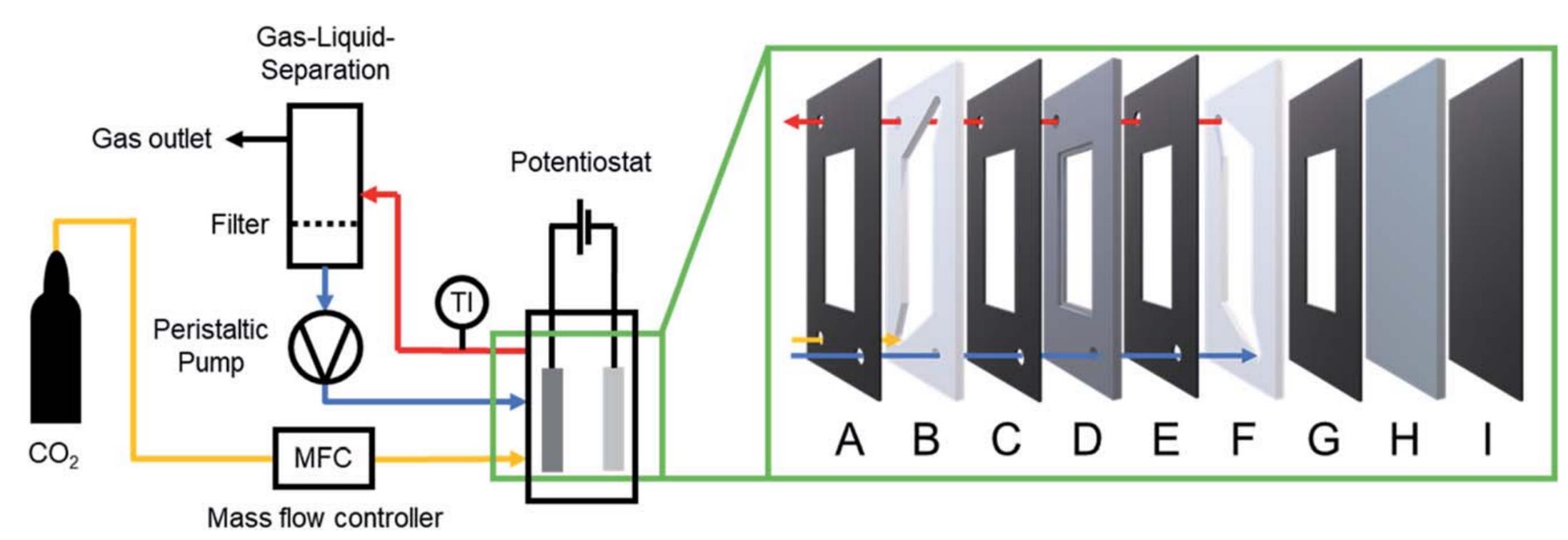
Fig.9 Semi?batch flow cell setup for the electrochemical reduction of CO2 to oxalate using a GDE[51](A, C, E, G, I) Viton? gaskets; (B) PTFE frame gas chamber; (D) stainless steel GDE support frame; (F) PTFE frame electrolyte chamber; (H) zinc sacrificial anode. The electrolyte is circulated between the electrochemical cell and an electrolyte reservoir (filtering ZnC2O4 and separating the liquid and gas phases) using a peristaltic pump. CO2 is supplied through the GDL at the back of the GDE into the electrolyte chamber. Copyright 2021, the Royal Society of Chemistry.
| 1 | Mariani G., Cheung W. W. L., Lyet A., Sala E., Mayorga J., Velez L., Gaines S. D., Dejean T., Troussellier M., Mouillot D., Sci. Adv., 2020, 6(44), eabb4848 |
| 2 | Pan F. P., Yang Y., Energy & Environ. Sci., 2020, 13(8), 2275—2309 |
| 3 | Chen C., Khosrowabadi K. J. F., Sheehan S. W., Chem., 2018, 4(11), 2571—2586 |
| 4 | Yaashikaa P. R., Senthil K. P., Varjani S. J., Saravanan A., J. CO2 Util., 2019, 33, 131—147 |
| 5 | De Luna P., Hahn C., Higgins D., Jaffer S. A., Jaramillo T. F., Sargent E. H., Sci., 2019, 364, eaav3506 |
| 6 | Chen C., Khosrowabadi K. J. F., Sheehan S. W., Chem., 2018, 4, 2571—2586 |
| 7 | Saha P., Amanullah S., Dey A., Acc. Chem. Res., 2022, 55, 134—144 |
| 8 | Sun Z. Y., Ma T., Tao H. C., Fan Q., Han B. X., Chem., 2017, 3, 560—587 |
| 9 | Wang Y. F., Han P., Lv X. M., Zhang L. J., Zheng G. F., Joule, 2018, 2(12), 2551—2582 |
| 10 | Gao F. Y., Bao R. C., Gao M. R., Yu S. H., J. Mater. Chem. A, 2020, 8(31), 15458—15478 |
| 11 | Jin S., Hao Z. M., Zhang K., Yan Z. H., Chen J., Angew. Chem. Int. Ed., 2021, 60, 20627—20648 |
| 12 | Zhao R. B., Ding P., Wei P. P., Zhang L. C., Liu Q., Luo Y. L., Li T. S., Lu S. Y., Shi X. F., Gao S. Y., Asiri A. M., Wang Z. M., Sun X. P., Adv. Funct. Mater., 2021, 31, 2009449 |
| 13 | Duan C. C., Nat. Catal., 2021, 4, 264—265 |
| 14 | Ilic S., Gesiorski J. L., Weerasooriya R. B., Glusac K. D., Acc. Chem. Res., 2022, 55, 844—856 |
| 15 | Roy A., Jadhav H. S., Park S. J., Seo J. G., J. Alloy. Compd., 2021, 887, 161449 |
| 16 | Zhang Z. Y., Bian L., Tian H., Liu Y., Bando Y., Yamauchi Y., Wang Z. L., Small, 2022, 18, 2107450 |
| 17 | Zhu S. Q., Delmo E. P., Li T. H., Qin X. P., Tian J., Zhang L. L., Shao M. H., Adv. Mater., 2021, 33, 2005484 |
| 18 | Karapinar D., Creissen C. E., De La Cruz J. G. R., Schreiber M. W., Fontecave M., ACS Energy Lett., 2021, 6, 694—706 |
| 19 | Nguyen T. N., Guo J. X., Sachindran A., Li F. W., Seifitokaldani A., Dinh C. T., J. Mater. Chem. A, 2021, 9, 12474—12494 |
| 20 | Senthilkumar P., Mohapatra M., Basu S., RSC Adv., 2022, 12, 1287—1309 |
| 21 | Van Daele K., De Mot B., Pupo M., Daems N., Pant D., Kortlever R., Breugelmans T., ACS Energy Lett., 2021, 6, 4317—4327 |
| 22 | Wang X. S., Wang W. H., Zhang J. Q., Wang H. Z., Yang Z. X., Ning H., Zhu J. X., Zhang Y. L., Guan L., Teng X. L., Zhao Q. S., Wu M. B., Chem. Eng. J., 2021, 426, 131867 |
| 23 | Yang Z. X., Wang H. Z., Fei X., Wang W. H., Zhao Y. Z., Wang X. S., Tan X. J., Zhao Q. S., Wang H. P., Zhu J. X., Zhou L., Ning H., Wu M. B., Appl. Catal. B⁃Environ., 2021, 298, 120571 |
| 24 | Yue H. R., Zhao Y. J., Ma X. B., Gong J. L., Chem. Soc. Rev., 2012, 41(11), 4218—4244 |
| 25 | Li A. M., Li Y. B., Geng S. Y., Song P. Z., Coal Chemical Industry, 2006, 10(5), 58—60 |
| 李安民, 李一兵, 耿书元, 宋培中. 煤化工, 2006, 10(5), 58—60 | |
| 26 | Murcia Valderrama M. A., Van Putten R. J., Gruter G. J. M., Eur. Polym. J., 2019, 119, 445—468 |
| 27 | Paris A. R., Bocarsly A. B., ACS Catal., 2019, 9(3), 2324—2333 |
| 28 | Twardowski Z., Cole E. B., Kaczur J. J., Teamey K., Keets K. A., Parajuli R., Bauer A., Sivasankar N., Leonard G., Kramer T. J., Method and System for Production of Oxalic Acid and Oxalic Acid Reduction Products, PCT/US2013/077610, 2014⁃06⁃26 |
| 29 | Chernyshova I. V., Somasundaran P., Ponnurangam S., Proc. Nati. Acad. Sci. USA, 2018, 115(40), E9261—E9270 |
| 30 | Gennaro A., Bhugun I., Savéant J. M., J. Chem. Soc., Faraday Trans., 1996, 92, 3963—3968 |
| 31 | Rudolph M., Dautz S., Jäger E. G., J. Am. Chem. Soc., 2000, 122, 10821—10830 |
| 32 | Angamuthu R., Byers P., Lutz M., Spek A. L., Bouwman E., Sci., 2010, 327, 313—315 |
| 33 | Lan J. L., Liao T., Zhang T. H., Chung L. W., ACS Inorg. Chem., 2017, 56, 6809—6819 |
| 34 | Cheng Y. Y., Hou P. F., Pan H., Shi H., Kang P., Appl. Catal. B⁃Environ., 2020, 272, 118954 |
| 35 | Senthil K. R., Senthil K. S., Anbu K. M., Electrochem. Commun., 2012, 25, 70—73 |
| 36 | Martindale B. C. M., Compton R. G., Chem. Commun., 2012, 48, 6487—6489 |
| 37 | Eneau⁃Innocent B., Pasquier D., Ropital F., Léger J. M., Kokoh K. B., Appl. Catal. B⁃Environ., 2010, 98, 65—71 |
| 38 | Kaname I., Shoichiro I., Nobuhiro Y., Takaya L., Takehiko T., Bull. Chem. Soc. Jpn., 1985, 58, 3027—3028 |
| 39 | Amatore C., Saveant J. M., J. Am. Chem. Soc., 1981, 103, 5021—5023 |
| 40 | Gennaro A., Isse A. A., Severin M. G., Vianello E., Bhugun I., Savéant J. M., J. Chem. Soc., Faraday Trans., 1996, 92(20), 3963—3968 |
| 41 | Fischer J., Lehmann T., Heitz E., J. Appl. Electrochem., 1981, 11, 743—750 |
| 42 | Haynes L. V., Sawyer D. T., Anal. Chem., 1967, 39(3), 332—338 |
| 43 | Subramanian S., Athira K. R., Anbu K. M., Senthil K. S., Barik R. C., J. CO₂ Util., 2020, 36, 105—115 |
| 44 | Lv W. X., Zhang R., Gao P. R., Gong C. X., Lei L. X., J. Solid State Electrochem., 2013, 17, 2789—2794 |
| 45 | Oh Y., Vrubel H., Guidoux S., Hu X. L., Chem. Commun., 2014, 50, 3878—3881 |
| 46 | Goodridge F., Presland G., J. Appl. Electrochem., 1984, 14, 791—796 |
| 47 | Shoichiro I., Takehiko T., Kaname I., Bull. Chem. Soc. Jpn., 1987, 60, 2517—1987 |
| 48 | Lv W. X., Zhang R., Gao P. R., Gong C. X., Lei L. X., Adv. Mat. Res., 2013, 807—809, 1322—1325 |
| 49 | Yang D. W., Li Q. Y., Shen F. X., Wang Q., Li L., Song N., Dai Y. N., Shi J., Electrochim. Acta, 2016, 189, 32—37 |
| 50 | Yang Y. L., Gao H. S., Feng J. Q., Zeng S. J., Liu L., Liu L. C., Ren B. Z., Li T., Zhang S. J., Zhang X. P., ChemSusChem, 2020, 13(18), 4900—4905 |
| 51 | König M., Lin S. H., Vaes J., Pant D., Klemm E., Faraday Discuss., 2021, 230, 360 |
| [1] | 秦永吉, 罗俊. 单原子催化剂在CO2转化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220300. |
| [2] | 吴玉, 李轩, 杨恒攀, 何传新. 钴单原子的双重限域制备策略及高效CO2电还原性能[J]. 高等学校化学学报, 2022, 43(9): 20220343. |
| [3] | 王新天, 李攀, 曹越, 洪文浩, 耿忠璇, 安志洋, 王昊宇, 王桦, 孙斌, 朱文磊, 周旸. 单原子材料在二氧化碳催化中的技术经济分析与产业化应用前景[J]. 高等学校化学学报, 2022, 43(9): 20220347. |
| [4] | 何鸿锐, 夏文生, 张庆红, 万惠霖. 羟基氧化铟团簇与二氧化碳和甲烷作用的密度泛函理论研究[J]. 高等学校化学学报, 2022, 43(8): 20220196. |
| [5] | 崔伟, 赵德银, 白文轩, 张晓东, 余江. CO2在非质子溶剂与铁基离子液体复合体系中的吸收[J]. 高等学校化学学报, 2022, 43(8): 20220120. |
| [6] | 郭志强, 杨博如, 席婵娟. 硼氢化试剂在二氧化碳还原官能化反应中的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220199. |
| [7] | 张昕昕, 许狄, 王艳秋, 洪昕林, 刘国亮, 杨恒权. CO2加氢制低碳醇CuFe基催化剂中的Mn助剂效应[J]. 高等学校化学学报, 2022, 43(7): 20220187. |
| [8] | 周紫璇, 杨海艳, 孙予罕, 高鹏. 二氧化碳加氢制甲醇多相催化剂研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220235. |
| [9] | 杨丹, 刘旭, 戴翼虎, 祝艳, 杨艳辉. 金团簇电催化二氧化碳还原反应的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220198. |
| [10] | 王征文, 高凤翔, 曹瀚, 刘顺杰, 王献红, 王佛松. 基于二氧化碳共聚物的紫外光固化高分子材料的制备与性能[J]. 高等学校化学学报, 2022, 43(7): 20220236. |
| [11] | 黄孝舜, 马海英, 柳淑娟, 王斌, 王红利, 钱波, 崔新江, 石峰. 二氧化碳间接转化制化学品的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220222. |
| [12] | 赵润瑶, 纪桂鹏, 刘志敏. 吡咯氮配位单原子铜催化剂的电催化二氧化碳还原性能[J]. 高等学校化学学报, 2022, 43(7): 20220272. |
| [13] | 彭奎霖, 李桂林, 江重阳, 曾少娟, 张香平. 电解液调控CO2电催化还原性能微观机制的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220238. |
| [14] | 张振, 邓煜, 张琴芳, 余达刚. 可见光促进二氧化碳参与的羧基化反应[J]. 高等学校化学学报, 2022, 43(7): 20220255. |
| [15] | 王丽君, 李欣, 洪崧, 詹新雨, 王迪, 郝磊端, 孙振宇. 调节氧化镉-炭黑界面高效电催化CO2还原生成CO[J]. 高等学校化学学报, 2022, 43(7): 20220317. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||