

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (2): 333.doi: 10.7503/cjcu20200663
詹舒辉1,2,赵亚松1,杨乃亮1,2,王丹1,2
收稿日期:2020-09-08
出版日期:2021-02-10
发布日期:2020-12-30
作者简介:王 丹, 男, 博士, 研究员, 主要从事无机多功能结构体系的合成化学研究. E-mail: 基金资助:
ZHAN Shuhui1,2, ZHAO Yasong1, YANG Nailiang1,2( ), WANG Dan1,2(
), WANG Dan1,2( )
)
Received:2020-09-08
Online:2021-02-10
Published:2020-12-30
Contact:
YANG Nailiang
E-mail:nlyang@ipe.ac.cn;danwang@ipe.ac.cn
Supported by:摘要:
石墨炔作为一种新兴碳材料, 由于具有特殊的电子特性、 丰富的纳米级孔隙以及能在相对低温下合成的特点, 在催化、 能源及生物等领域受到广泛关注. 石墨炔自下而上的合成特点使其在结构上具有可设计性, 而其显著特征在于具有拓扑有序的规则孔道结构. 在过去的10年间, 研究人员在石墨炔孔结构设计方面进行了大量的实验和理论研究. 孔结构设计所带来的独特性能为其提供了良好的应用前景. 本文从石墨炔的合成特点出发, 总结了单体、 催化剂、 模板及溶液4个因素对石墨炔结构的影响, 并从孔结构出发讨论了其应用. 为研究者以应用为导向, 设计合成具有特殊孔结构的石墨炔提供了思路. 最后还探讨了孔结构设计给石墨炔带来的机遇与挑战, 并对三维多孔结构石墨炔的设计进行了展望.
中图分类号:
TrendMD:
詹舒辉, 赵亚松, 杨乃亮, 王丹. 石墨炔孔结构: 设计、 合成和应用. 高等学校化学学报, 2021, 42(2): 333.
ZHAN Shuhui, ZHAO Yasong, YANG Nailiang, WANG Dan. Pore Structure of Graphdiyne: Design, Synthesis and Application. Chem. J. Chinese Universities, 2021, 42(2): 333.

Fig.1 Classification of GDY’s pore structuresMolecular pore: (A) fluorine-substituted GDY[11], Copyright 2018, RSC Publishing; (B) GDY; piled pore: (C) GDY nanowall[13], Copyright 2015, ACS Publishing; (D) GDY nanotube[14], Copyright 2011, ACS Publishing; (E) 3D hierarchical structured GDY[15], Copyright 2017, ACS Publishing; (F) 3D diatomite shaped GDY[12], Copyright 2018, Wiley Publishing.
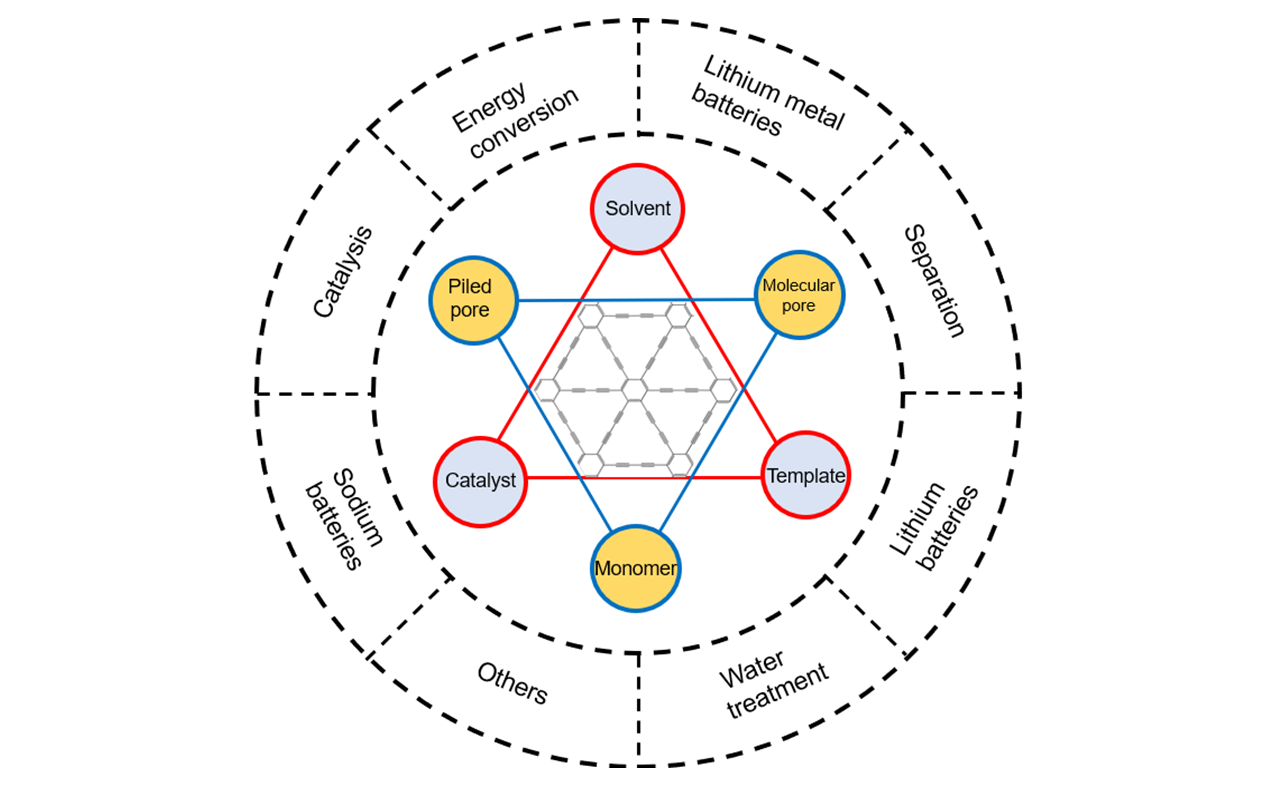
Fig.2 Schematics of GDY’s pore structures designThe structures can be manipulated by adjusting monomers, catalysts, templates and solvent for target-applications such as water treatment, lithium-ion batteries, catalysis, etc.
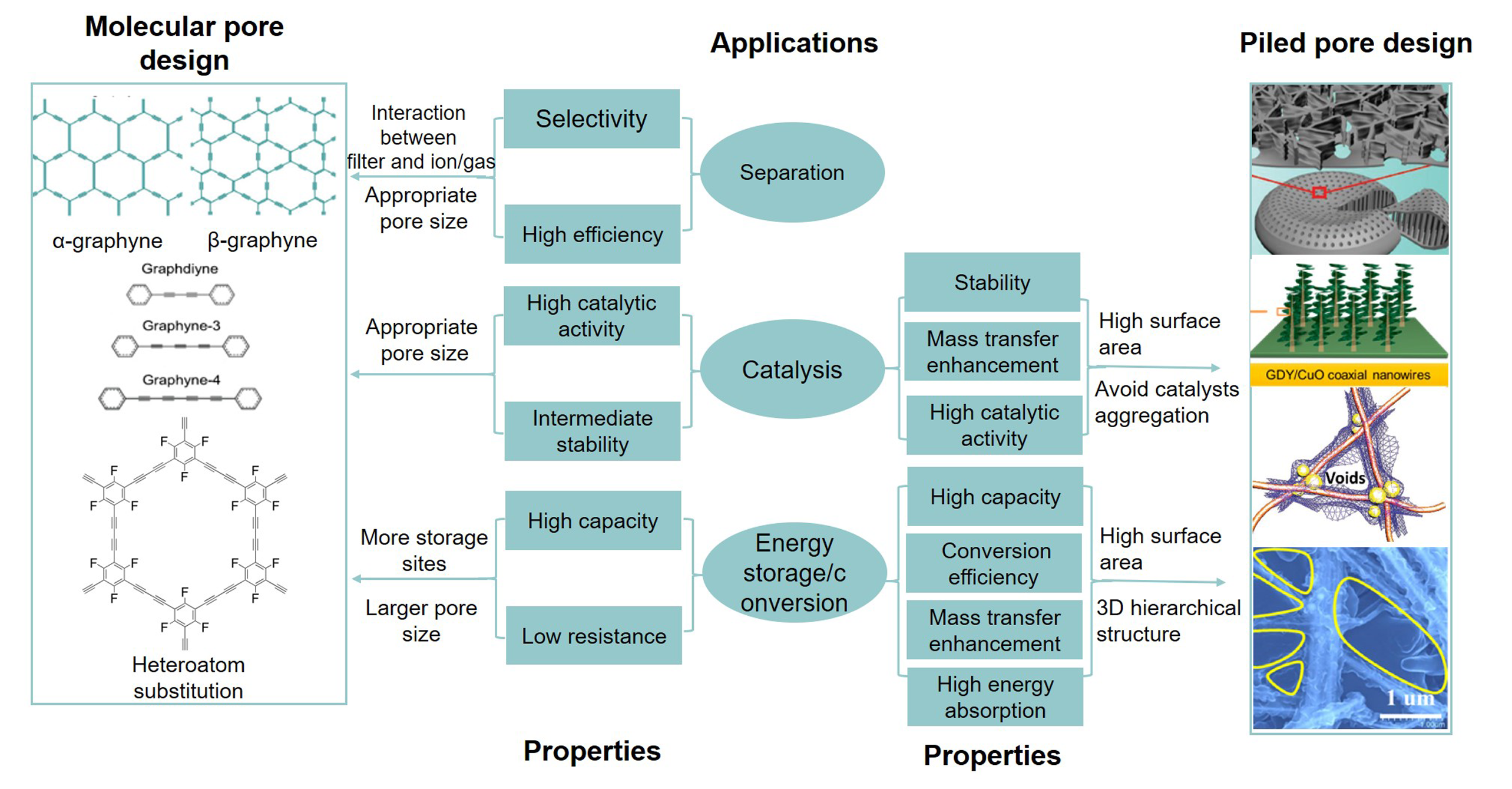
Fig.3 Application?oriented design of GDYPerformance required in the application and its corresponding structure design strategies[12,15,22—24]. Copyright 2018, Wiley Publishing; Copyright 2017, ACS Publishing; Copyright 2013, IOP Publishing; Copyright 2018, Wiley Publishing; Copyright 2019, ACS Publishing.
| Monomer | Structure | Size(diameter)/nm | Condition | Ref. |
|---|---|---|---|---|
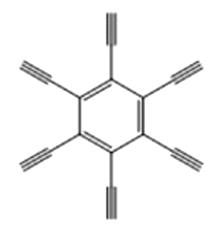 |  | 0.54 | Copper foil, pyridine | [ |
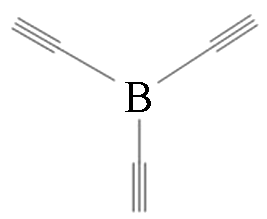 |  | 0.59 | Copper foil, pyridine | [ |
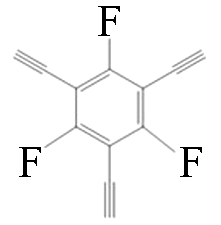 |  | 1.3 | Copper foil, pyridine | [ |
| Monomer | Structure | Size(diameter)/nm | Conditions | Ref. |
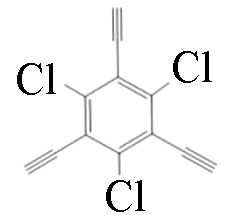 |  | 1.6 | Copper foil, pyridine | [ |
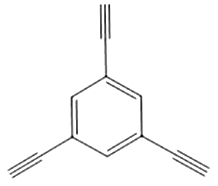 |  | 1.63 | Copper foil, pyridine | [ |
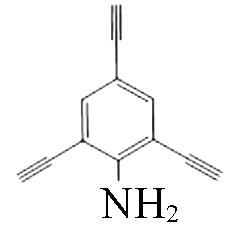 |  | 1.66 | Copper foil, CH2Cl2, TMEDA | [ |
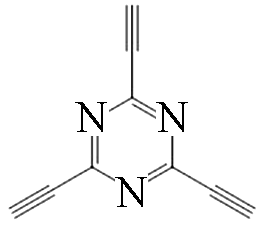 |  | 1.5 | Copper foil, pyridine | [ |
Table 1 Molecular pore structures and sizes of GDY prepared by different monomers
| Monomer | Structure | Size(diameter)/nm | Condition | Ref. |
|---|---|---|---|---|
 |  | 0.54 | Copper foil, pyridine | [ |
 |  | 0.59 | Copper foil, pyridine | [ |
 |  | 1.3 | Copper foil, pyridine | [ |
| Monomer | Structure | Size(diameter)/nm | Conditions | Ref. |
 |  | 1.6 | Copper foil, pyridine | [ |
 |  | 1.63 | Copper foil, pyridine | [ |
 |  | 1.66 | Copper foil, CH2Cl2, TMEDA | [ |
 |  | 1.5 | Copper foil, pyridine | [ |
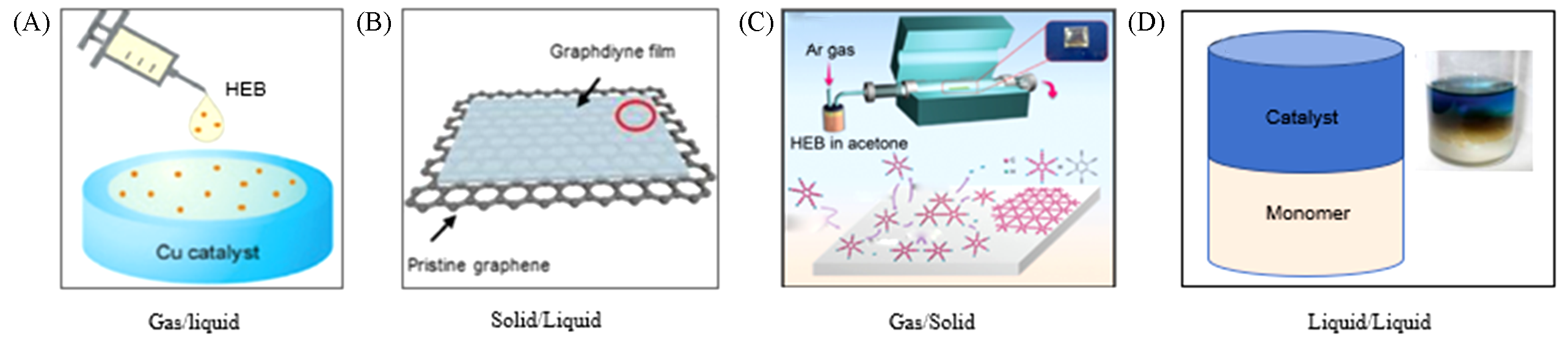
Fig.5 Growth of GDY on gas/liquid interface(A)[40], solid(graphene)/liquid(solvent) interface(B)[41], silver through CVD method(C)[42] and liquid/liquid interface(D)[40](A, D) Copyright 2017, ACS Publishing; (B) Copyright 2018, American Association for the Advancement of Science; (C) Copyright 2017,Wiley Publishing.
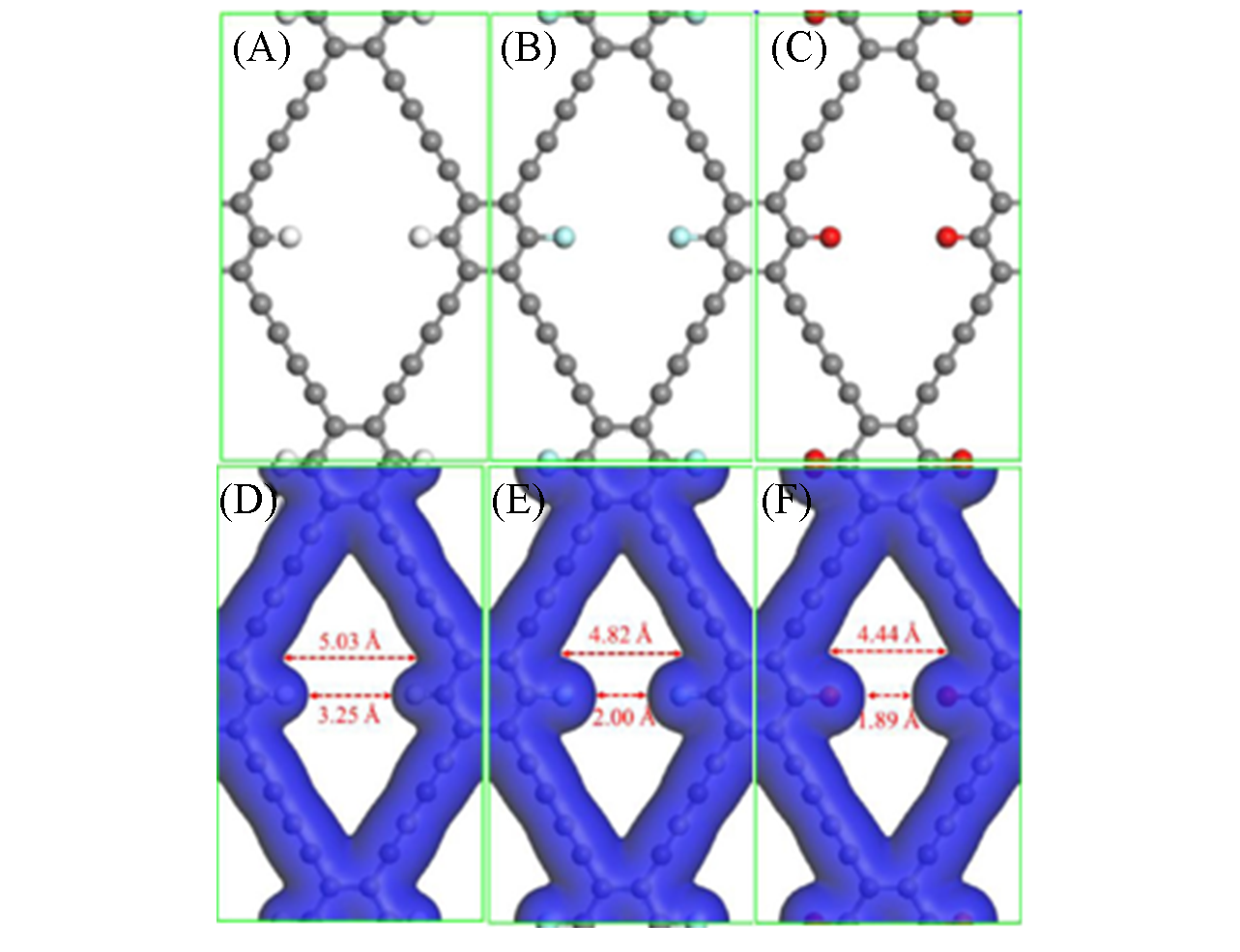
Fig.6 Structures(A—C) and pore electron density isosurfaces(D—F) of GDY?H(A, D), GDY?F(B, E) and GDY?O(C, F) monolayers[54]Gray sphere: C; white sphere: H; blue sphere: F; red sphere: O. Copyright 2017, Elsevier Publishing.
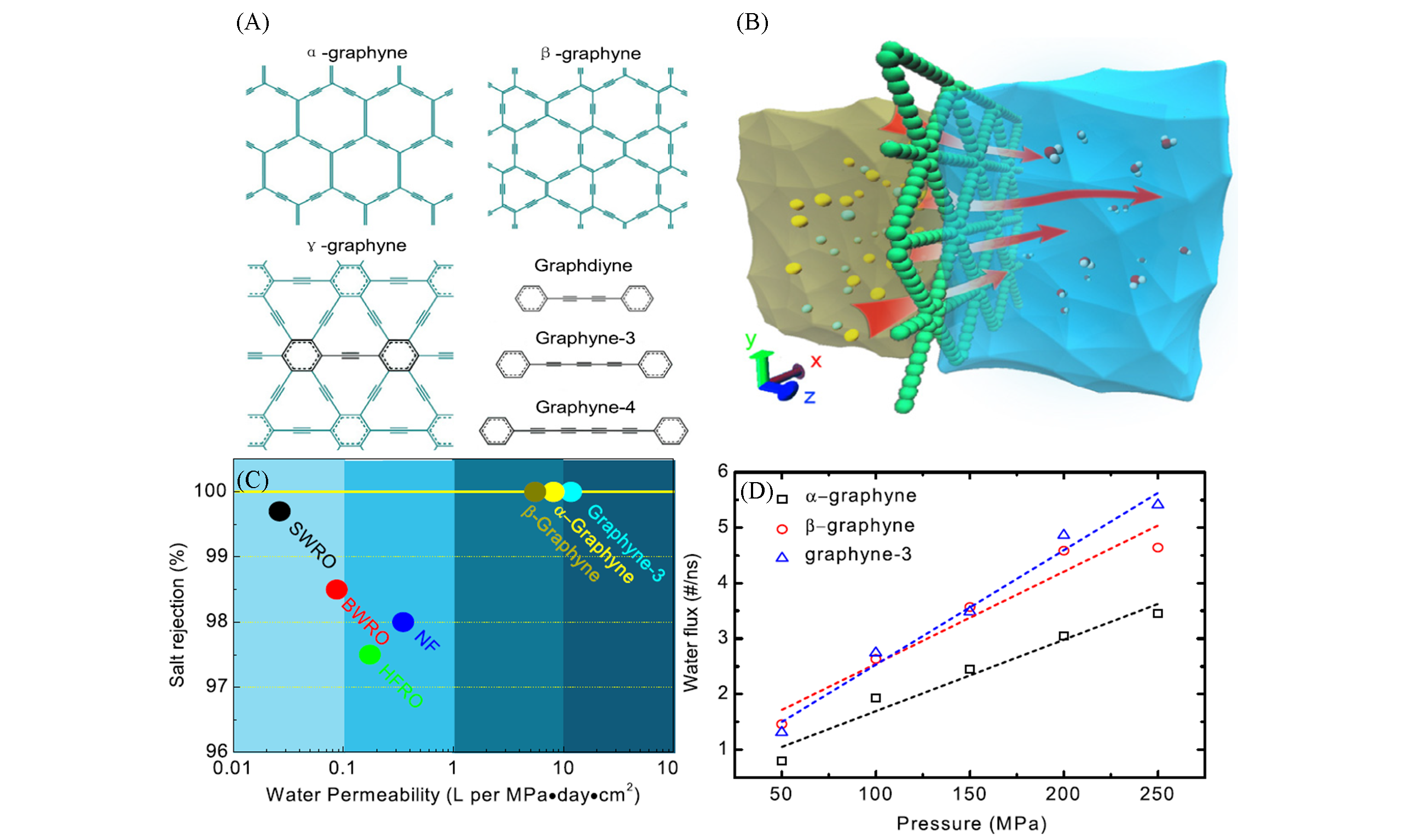
Fig.7 Structures of α?garphyne, β?garphyne and γ?garphyne(A), schematic diagram of graphyne used for seawater desalination(B), performance comparison between graphyne and commercial reverse osmosis membranes(C) and single?pore flow rates of α?garphyne, β?garphyne and graphyne?3 at different pressures(D)[22]Copyright 2013, IOP Publishing.
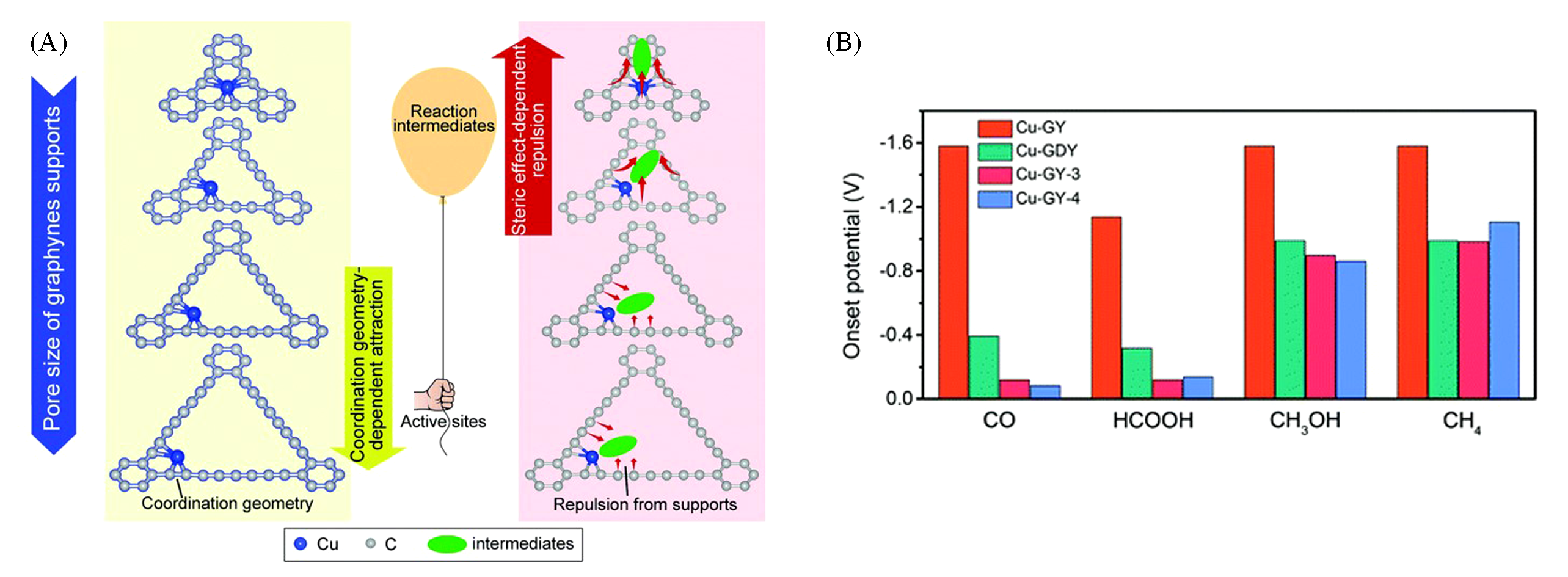
Fig.8 Coordination number of Cu atoms and the positions of intermediates vary with molecular different pore sizes(A) and onset potentials of the products of CO2 reduction on Cu?Graphynes(B)[60]Copyright 2020, RSC Publishing.

Fig.9 Storage sites of lithium ions in Cl?substituted GDY(gray sphere: C, green sphere: Cl, orange sphere: Li)(A), the cycling performance of Cl?substituted GDY in lithium batteries(B)[26], storage sites of lithium ion in F?substituted GDY(gray sphere: C, yellow sphere: F, purple sphere: Li)(C) and cycling performance of F?substituted GDY in lithium batteries(D)[11](A, B) Copyright 2017, Wiley Publishing; (C, D) Copyright 2018, RSC Publishing.
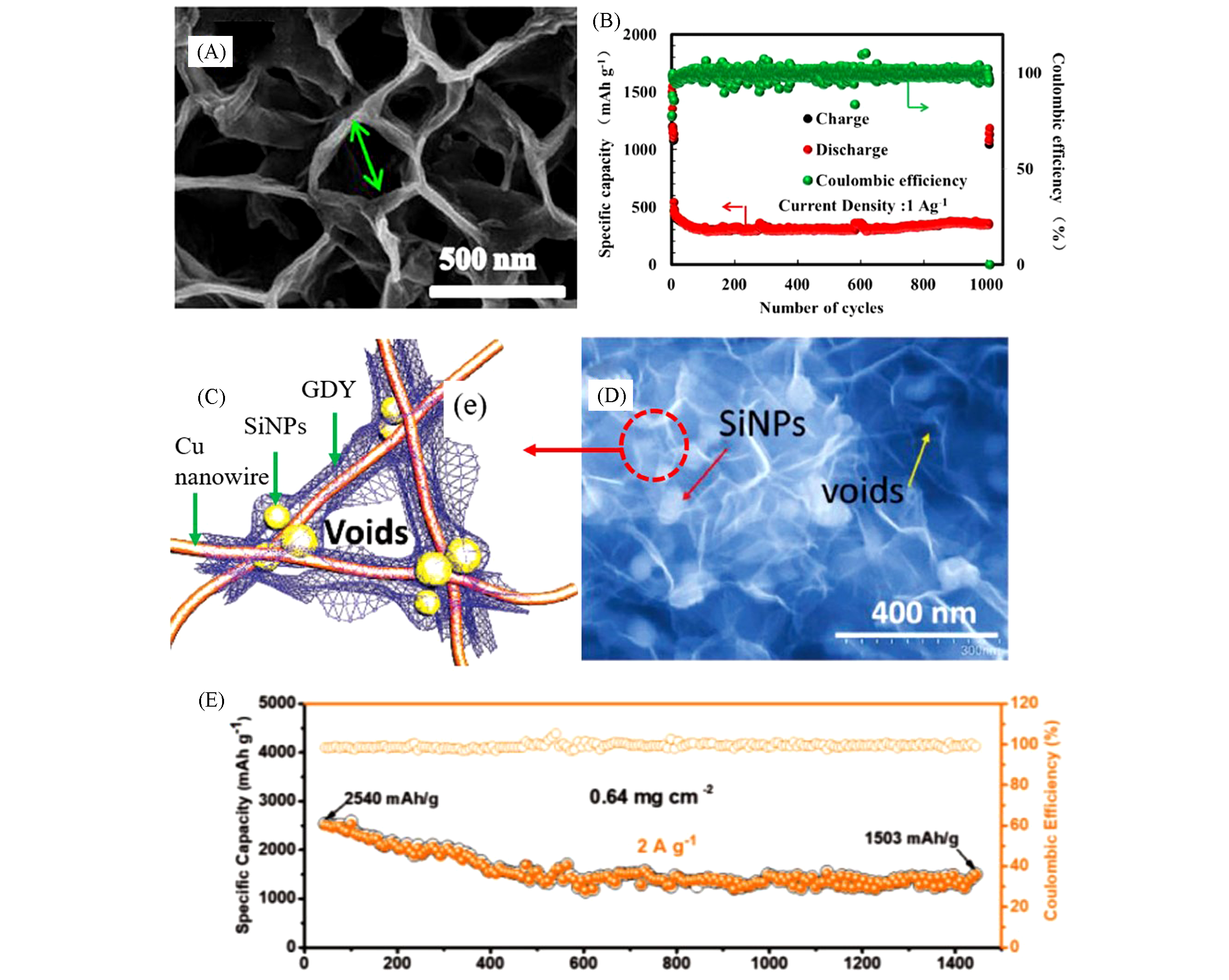
Fig.10 Three?dimensional GDY nanosheets on copper foil substrates prepared by a modified Glaser?Hay coupling reaction(A), the cycle performance of sodium batteries based on 3D GDY nanosheets at the current density of 1 A/g(B)[69], schematic for weaving the network of ultrathin GDY nanosheets on the Si anode(C), SEM images of ultrathin GDY nanosheets on the Si anode(D) and the performance of the as‐ prepared sample at a high current density of 2 A/g(E)[23](A, B) Copyright 2017, ACS Publishing; (C—E) Copyright 2018, Wiley Publishing.
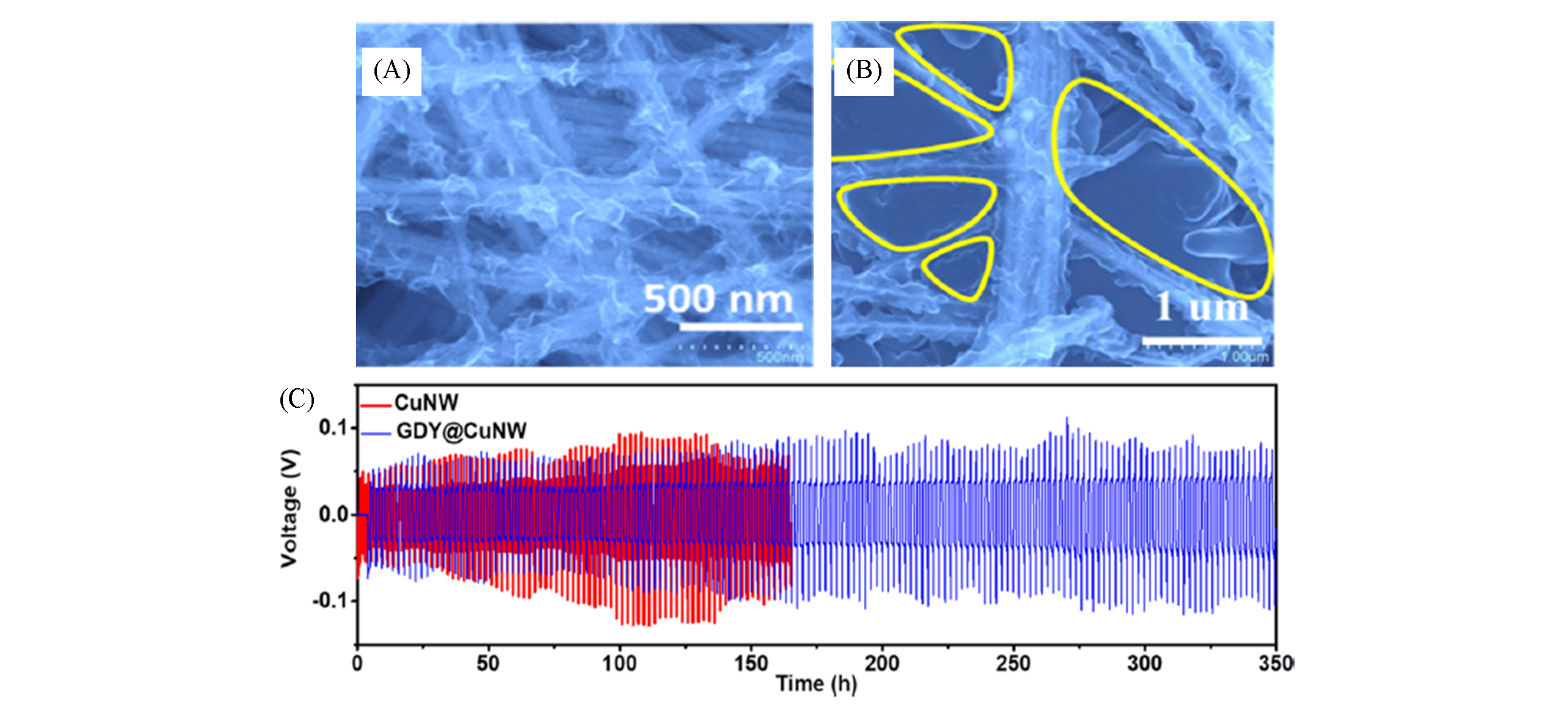
Fig.11 3D copper nanowires@GDY(A), lithium deposited in the pores of 3D copper nanowires@GDY(B) and stability of lithium metal batteries based on 3D copper nanowires@GDY(C)[24]Copyright 2019, ACS Publishing.
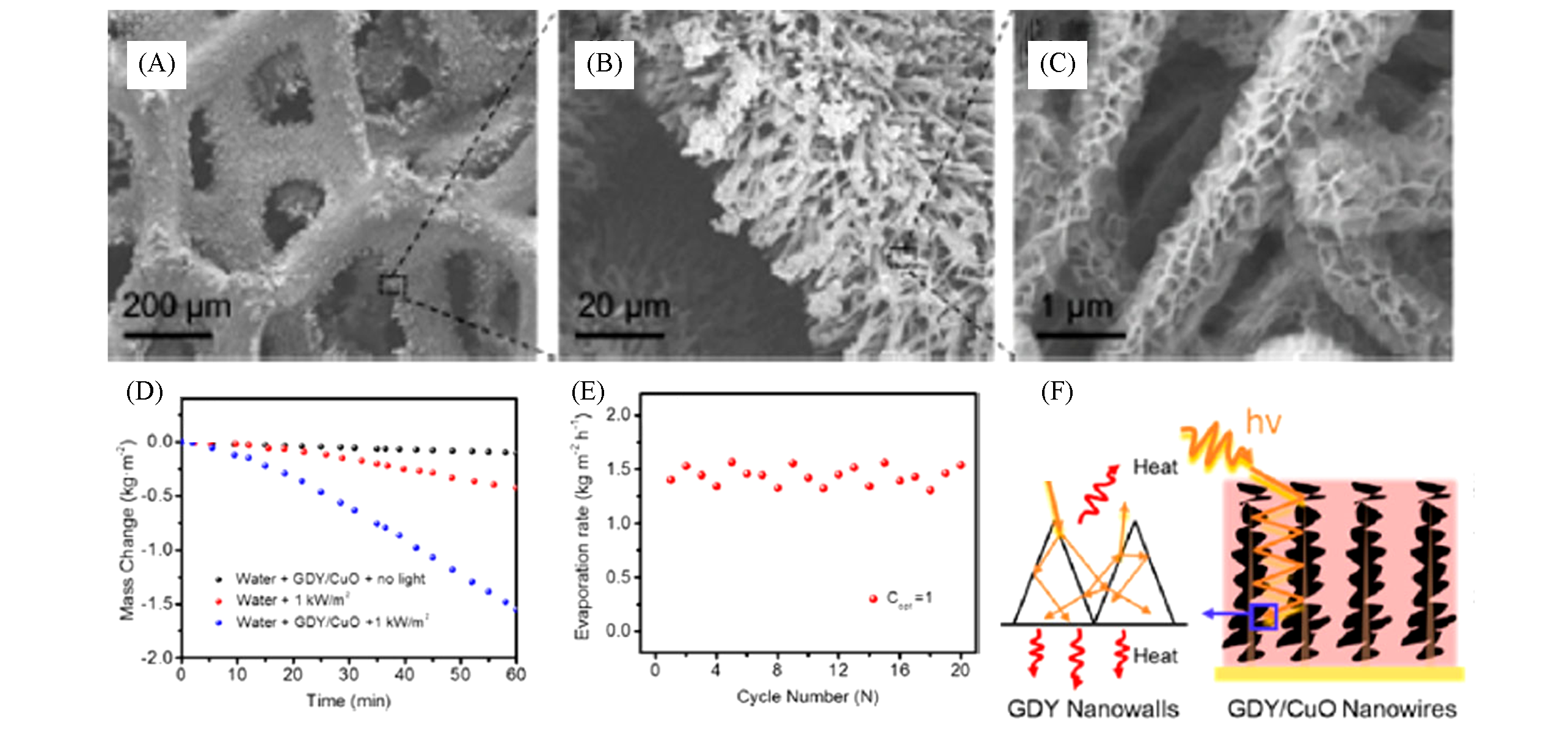
Fig.12 SEM images of 3D GDY at different scales(A—C), water evaporation rate(D), evaporation stability(E) and schematic diagram of promoting light absorption(F)[15]Copyright 2017, ACS Publishing.
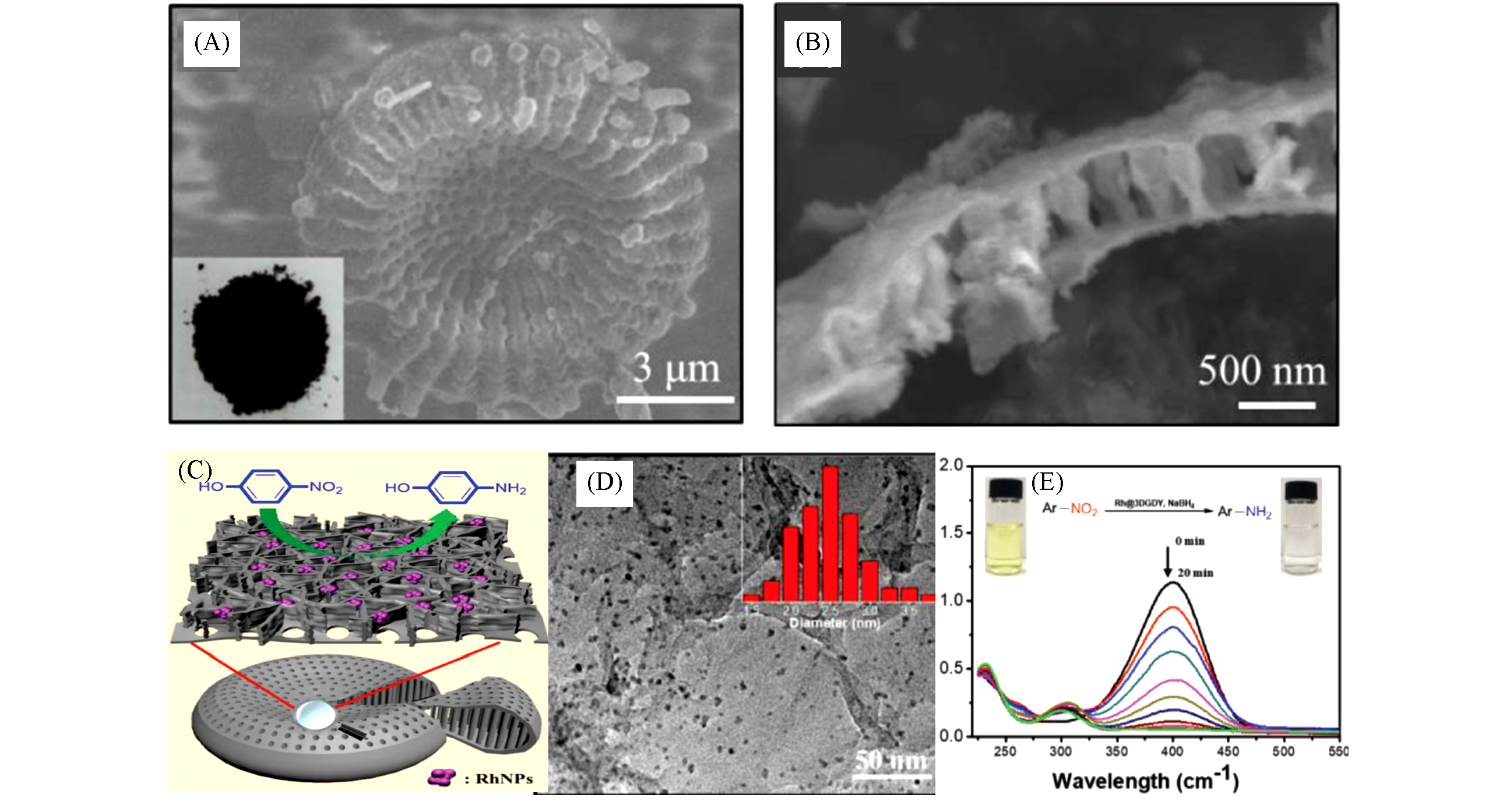
Fig.13 SEM images of 3D diatomite shaped GDY(A, B), schematic illustration of 4?nitrophenol reduction catalyzed by Rh@3DGDY(C), TEM image of Rh@3DGDY(D) and time?dependent UV?Vis absorption spectra recorded during the 4?nitrophenol reduction catalyzed by Rh@3DGDY(E)[12]Copyright 2018, Wiley Publishing.
| 1 | Zhou J. Y., Zhang J., Liu Z. F., Acta Phys.⁃Chim. Sin., 2018, 34(9), 977—991 |
| 2 | Chen M. Q., Guan R. N., Yang S. F., Adv. Sci., 2019, 6, 1800941 |
| 3 | Liu L. Q., Ma W. J., Zhang Z., Small, 2011, 7(11), 1504—1520 |
| 4 | Zeng M., Wang W. L., Bai X. D., Chin. Phys. B, 2013, 098105 |
| 5 | Li G. X., Li Y. L., Liu H. B., Guo Y. B., Li Y. J., Zhu D. B., Chem. Commun., 2010, 46, 3256—3258 |
| 6 | Liu M. H., Li Y. L., Acta Phys.⁃Chim. Sin., 2018, 34(9), 959—960 |
| 7 | Gao X., Liu H. B., Wang D., Zhang J., Chem. Soc. Rev., 2019, 48, 908—936 |
| 8 | Janas D., Liszka B., Mater. Chem. Front., 2018, 2(1), 22—35 |
| 9 | Dai L. M., Mau A. W. H., Adv. Mater., 2001, 13(12/13), 899—913 |
| 10 | Yang N. L., Zhai J., Wang D., Chen Y. S., Jiang L., ACS Nano, 2010, 4, 887—894 |
| 11 | He J. J., Wang N., Yang Z., Shen X. Y., Wang K., Huang C. S., Yi Y. P., Tu Z. Y., Li Y. L., Energy Environ. Sci., 2018, 11, 2893—2903 |
| 12 | Li J. Q., Xu J., Xie Z. Q., Gao X., Zhou J. Y., Xiong Y., Chen C. G., Zhang J., Liu Z. F., Adv. Mater., 2018, 30, 1800548 |
| 13 | Zhou J. Y., Gao X., Liu R., Xie Z. Q., Yang J., Zhang S. Q., Zhang G. M., Liu H. B., Li Y. L., Zhang J., Liu Z. F., J. Am. Chem. Soc., 2015, 137, 7596—7599 |
| 14 | Li G. X., Li Y. L., Qian X. M., Liu H. B., Lin H. W., Chen N., Li Y. J., J. Phys. Chem. C, 2011, 115, 2611—2615 |
| 15 | Gao X., Ren H. Y., Zhou J. Y., Du R., Yin C., Liu R., Peng H. L., Tong L. M., Liu Z. F., Zhang J., Chem. Mater., 2017, 29, 5777—5781 |
| 16 | Jang B., Koo J., Park M., Lee H., Nam J., Kwon Y., Lee H., Appl. Phys. Lett., 2013, 103, 263904 |
| 17 | Wang F., Zuo Z. C., Li L., Li K., He F., Jiang Z. Q., Li Y. L., Angew. Chem. Int. Ed., 2019, 58, 15010—15015 |
| 18 | Zhang L. Y., Wu C., Ding X. D., Fang Y., Sun J., Phys. Chem. Chem. Phys., 2018, 20, 18192—18199 |
| 19 | Bartolomei M., Carmona⁃Novill E., Hernandez M. I., Campos⁃Martinez J., Pirani F., Giorgi G., J. Phys. Chem. C, 2014, 118, 29966—29972 |
| 20 | He J. J., Wang N., Cui Z. L., Du H. P., Fu L., Huang C. S., Yang Z., Shen X. Y., Yi Y. P., Tu Z. Y., Li Y. L., Nat. Commun., 2017, 8, 1172 |
| 21 | Yu H. D., Xue Y. R., Hui L., Zhang C., Li Y. J., Zuo Z. C., Zhao Y. J., Li Z. B., Li Y. L., Adv. Mater., 2018, 1707082 |
| 22 | Xue M. M., Qiu H., Guo W. L., Nanotechnology, 2013, 24, 505720 |
| 23 | Shang H., Zuo Z. C., Yu L., Wang F., He F., Li Y. L., Adv. Mater., 2018, 30, 1801459 |
| 24 | Shang H., Zuo Z. C., Li Y. L., ACS Appl. Mater. Interfaces, 2019, 11, 17678—17685 |
| 25 | Wang N., Li X. D., Tu Z. Y., Zhao F. H., He J. J., Guan Z. Y., Huang C. S., Yi Y. P., Li Y. L., Angew. Chem. Int. Ed., 2018, 57, 3968—3973 |
| 26 | Wang N., He J. J., Tu Z. Y., Yang Z., Zhao F. H., Li X. D., Huang C. S., Wang K., Jiu T. G., Yi Y. P., Li Y. L., Angew. Chem. Int. Ed., 2017, 56, 10740—10745 |
| 27 | Shang H., Zuo Z. C., Zheng H. Y., Li K., Tu Z. Y., Yi Y. P., Liu H. B., Li Y. J., Li Y. L., Nano Energy, 2017, 44, 144—154 |
| 28 | Sonoda M., Inaba A., Itahashi K., Tobe Y., Org. Lett., 2001, 3, 2419 |
| 29 | Anthony J. E., Khan S. I., Rubin Y., Tetrahedron Lett., 1997, 38, 3499—3502 |
| 30 | Shi W., Lei A.W., Tetrahedron Lett., 2014, 55, 17, 2763—2772 |
| 31 | Sindhu K. S., Anilkumar G., RSC Adv., 2014, 4, 27867—27887 |
| 32 | Si H. Y., Deng Q. X., Chen L. C., Wang L., Liu X. Y., Wu W. S., Zhang Y. H., Zhou J. M., Zhang H. L., J. Alloys Compd., 2019, 794, 261—267 |
| 33 | Glaser C., Ber. Dtsch. Chem. Ges., 1869, 2, 422—424 |
| 34 | Eglinton G., Galbralth A. R., Soc. Chemical Industry, 1956, 28, 737—738 |
| 35 | Hay A. S., J. Org. Chem., 1962, 27(9), 3320—3321 |
| 36 | Vilhelmsen M. H., Jensen J., Tortzen C. G., Nielsen M. B., Eur. J. Org. Chem., 2013, 701—711 |
| 37 | Tang J. Y., Jiang H. F., Deng G. H., Chin. J. Org. Chem., 2005, 25, 1503 |
| 38 | Gao X., Li J., Du R., Zhou J. Y., Huang M. Y., Liu R., Li J., Xie Z. Q., Wu L. Z., Liu Z. F., Zhang J., Adv. Mater., 2017, 1605308 |
| 39 | Zhang S. C., Chen Y., Kang Z., Wu P. W., Wu J., Zhang Z., Liao Q. L., Zhang J., Zhang Y., ACS Appl. Mater. Interfaces, 2019, 11, 2745—2749 |
| 40 | Matsuoka R., Sakamoto R., Hoshiko K., Sasaki S., Masunaga H., Nagashio K., Nishihara H., J. Am. Chem. Soc., 2017, 139, 3145—3152 |
| 41 | Gao X., Zhu Y. H., Yi D., Zhou J. Y., Zhang S. S., Yin C., Ding F., Zhang S. Q., Yi X. H., Wang J. Z., Tong L. M., Han Y., Liu Z. F., Zhang J., Sci. Adv., 2018, 4(7), eaat6378 |
| 42 | Liu R., Gao X., Zhou J. Y., Xu H., Li Z. Z., Zhang S. Q., Xie Z. Q., Zhang J., Liu Z. F., Adv. Mater., 2017, 29, 1604665 |
| 43 | Bieri M., Nguyen M. T., Groning O., Cai J. M., Treier M., Ait⁃Mansour K., Ruffieux P., Pignedoli C. A., Passerone D., Kastler M., Mullen K., Fasel R., J. Am. Chem. Soc., 2010, 132, 16669 |
| 44 | Yuan Q. H., Ding D., Nanoscale, 2014, 21, 12727—12731 |
| 45 | Huang C. S., Li Y. J., Wang N., Xue Y. R., Zuo Z. C., Liu H. B., Li Y. L., Chem. Rev., 2018, 118(16), 7744—7803 |
| 46 | Zhang T., Hou Y., Dzhagan V., Liao Z. Q., Chai G. L., Loffler M., Olianas D., Milani A., Xu S. Q., Tommasini M., Zahn D. R. T., Zheng Z. K., Zschech E., Jordan R., Feng X. L., Nat. Commun., 2018, 9, 1140 |
| 47 | van Gelderen L., Rothenberg G., Calderone V. R., Wilson K., Shiju N. R., Appl.Organometal. Chem., 2013, 27, 23—27 |
| 48 | Zhao Y. S., Wan J. W., Yao H. Y., Zhang L. J., Lin K. F., Wang L., Yang N. L., Liu D. B., Song L., Zhu J., Gu L., Liu L., Zhao H. J., Li Y. L., Wang D., Nat. Chem., 2018, 10(9), 924—931 |
| 49 | Domyati D., Latifi R., Tahsini L., J. Organomet. Chem., 2018, 860, 98—105 |
| 50 | Reichardt C., Solvents and Solvent Effects in Organic Chemistry, Third Edition, Wiley⁃VCH Verlag GmbH & Co. KGaA, Veinheim, 2003 |
| 51 | Peng J. B., Wu X. F., Angew. Chem. Int. Ed., 2018, 57, 1152—1160 |
| 52 | Kim S., Gamallo P., Vines F., Lee J. Y., Appl. Surf. Sci., 2020, 499, 143927 |
| 53 | Cranford S. W., Buehler M. J., Nanoscale, 2012, 4, 4587—4593 |
| 54 | Zhao L. M., Sang P. P., Guo S., Liu X. P., Li J., Zhu H. Y., Guo W. Y., Appl. Surf. Sci., 2017, 405, 455—464 |
| 55 | Xue Y. R., Huang B. L., Yi Y. P., Guo Y., Zuo Z. C., Li Y. J., Jia Z. Y., Liu H. B., Li Y. L., Nat. Commun., 2018, 9, 1460 |
| 56 | Hui L., Xue Y. R., Yu H. D., Zhang C., Huang B. L., Li Y. L., ChemPhysChem, 2020, 21, 1—6 |
| 57 | Zhai X. W., Yan H. X., Ge G. X., Yang J. M., Chen F., Liu X. Y., Yang D. Z., Li L. F., Zhang J. L., Appl. Surf. Sci., 2020, 506, 144941 |
| 58 | He T. W., Matta S. K., Will G., Du A. J., Small Methods, 2019, 9, 1800419 |
| 59 | Xu H. X., Cheng D. J., Cao D. P., Zeng X. C., Nat. Catal., 2018, 1, 632—632 |
| 60 | Ni Y. X., Miao L. C., Wang J. Q., Liu J. X., Yuan M. J., Chen J., Phys. Chem. Chem. Phys., 2020, 22, 1181—1186 |
| 61 | Xu Z. M., Lv X., Li J., Chen J. G., Liu Q. S., RSC Adv., 2016, 6, 25594—25600 |
| 62 | Shekar S. C., Swathi R. S., J. Phys. Chem. A, 2013, 117, 8632—8641 |
| 63 | Zhang S. L., He J. J., Zheng J., Huang C. S., Lv Q., Wang K., Wang N., Lan Z. G., J. Mater. Chem. A, 2017, 5, 2045—2051 |
| 64 | Fu G. T., Yan X. X., Chen Y. F., Xu L., Sun D. M., Lee J. M., Tang Y. W., Adv. Mater., 2018, 30, 1704609 |
| 65 | Zhao M. Q., Xie X. Q., Ren C. E., Makaryan T., Anasori B., Wang G. X., Gogotsi Y., Adv. Mater., 2017, 29, 1702410 |
| 66 | Song H. Q., Zhu Q., Zheng X. J., Chen X. G., J. Mater. Chem. A, 2015, 3, 10368—10377 |
| 67 | Qiu H. J., Du P., Hu K. L., Gao J. J., Li H. L., Liu P., Ina T., Ohara K., Ito Y., Chen M. W., Adv. Mater., 2019, 31, 1900843 |
| 68 | Qin J., Wang T. S., Liu D. Y., Liu E. Z., Zhao N. Q., Shi C. S., He F., Ma L. Y., He C. N., Adv. Mater., 2018, 30, 1704670 |
| 69 | Wang K., Wang N., He J. J., Yang Z., Shen X. Y., Huang C. S., ACS Appl. Mater. Interfaces, 2017, 9, 40604—40613 |
| 70 | Wei Y. Z., Wang J. Y., Yu R. B., Wan J. W., Wang D., Angew. Chem. Int. Ed.,2019, 58, 1422—1426 |
| 71 | Hu X. Z., Xu W. C., Zhou L., Tan Y. L., Wang Y., Zhu S. N., Zhu J., Adv. Mater., 2017, 29, 1604031 |
| 72 | Qiu B. C., Xing M. Y., Zhang J. L., Chem. Soc. Rev., 2018, 47, 2165—2216 |
| 73 | Gao X., Zhou J. Y., Du R., Xie Z. Q., Deng S. B., Liu R., Liu Z. F., Zhang J., Adv. Mater., 2016, 28, 168—173 |
| 74 | Liu R., Zhou J. Y., Li J. Q., Xie Z. Q., Li Z. Z., Zhang S. Q., Tong L. M., Zhang J., Liu Z. F., Adv. Electron. Mater., 2017, 3, 1700122 |
| 75 | Yin C., Li J. Q., Li T. R., Yu Y., Kong Y., Gao P., Peng H. L., Tong L. M., Zhang J., Adv. Funct. Mater., 2020, 2001396 |
| 76 | Yan H. L., Yu P., Han G. C., Zhang Q. H., Gu L., Yi Y. P., Liu H. B., Li Y. L., Mao L. Q., Angew. Chem. Int. Ed., 2018, 57, 1—6 |
| [1] | 曹舒杰, 李泓君, 管文丽, 任梦田, 周传政. 硫代磷酸酯寡聚核苷酸的立体控制合成研究进展[J]. 高等学校化学学报, 2022, 43(Album-4): 20220304. |
| [2] | 姚青, 俞志勇, 黄小青. 单原子催化剂的合成及其能源电催化应用的研究进展[J]. 高等学校化学学报, 2022, 43(9): 20220323. |
| [3] | 王茹玥, 魏呵呵, 黄凯, 伍晖. 单原子材料的冷冻合成[J]. 高等学校化学学报, 2022, 43(9): 20220428. |
| [4] | 王新天, 李攀, 曹越, 洪文浩, 耿忠璇, 安志洋, 王昊宇, 王桦, 孙斌, 朱文磊, 周旸. 单原子材料在二氧化碳催化中的技术经济分析与产业化应用前景[J]. 高等学校化学学报, 2022, 43(9): 20220347. |
| [5] | 王学斌, 薛源, 茆华女, 项艳鑫, 包春燕. 光/还原双重响应水凝胶微球的制备及在细胞三维(3D)培养中的应用[J]. 高等学校化学学报, 2022, 43(8): 20220116. |
| [6] | 金睿明, 穆晓清, 徐岩. 生物-化学法合成黑色素前体5, 6-二羟基吲哚[J]. 高等学校化学学报, 2022, 43(8): 20220134. |
| [7] | 韦春洪, 蒋倩, 王盼盼, 江成发, 刘岳峰. 贵金属Pt促进Co基费托合成催化剂的原子尺度结构分析[J]. 高等学校化学学报, 2022, 43(8): 20220074. |
| [8] | 张昕昕, 许狄, 王艳秋, 洪昕林, 刘国亮, 杨恒权. CO2加氢制低碳醇CuFe基催化剂中的Mn助剂效应[J]. 高等学校化学学报, 2022, 43(7): 20220187. |
| [9] | 周紫璇, 杨海艳, 孙予罕, 高鹏. 二氧化碳加氢制甲醇多相催化剂研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220235. |
| [10] | 李祎頔, 田晓春, 李俊鹏, 陈立香, 赵峰. 半导体-微生物界面电子传递及其在环境领域的应用[J]. 高等学校化学学报, 2022, 43(6): 20220089. |
| [11] | 鲁聪, 李振华, 刘金露, 华佳, 李光华, 施展, 冯守华. 一种新的镧系金属有机骨架材料的合成、 结构及荧光检测性质[J]. 高等学校化学学报, 2022, 43(6): 20220037. |
| [12] | 黄汉浩, 卢湫阳, 孙明子, 黄勃龙. 石墨炔原子催化剂的崭新道路:基于自验证机器学习方法的筛选策略[J]. 高等学校化学学报, 2022, 43(5): 20220042. |
| [13] | 沙蒙, 许维庆, 吴志超, 顾文玲, 朱成周. 单原子材料类酶催化及生物医学应用研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220077. |
| [14] | 陈朝阳, 薛玉瑞, 李玉良. 石墨炔零价金属原子催化剂的合成及应用[J]. 高等学校化学学报, 2022, 43(5): 20220063. |
| [15] | 樊小勇, 朱永强, 毋妍, 张帅, 许磊, 苟蕾, 李东林. 三维多孔Sn-Zn合金电极助力Zn的均匀沉积/剥离[J]. 高等学校化学学报, 2022, 43(4): 20210861. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||