

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (11): 2340.doi: 10.7503/cjcu20190308
唐佳易1,姚俊杰1,张晓君2,马梁2,张婷妹3,牛峥1,陈祥祯1,赵亮1,江林3,孙迎辉1,*( )
)
收稿日期:2019-05-28
出版日期:2019-11-10
发布日期:2019-08-20
通讯作者:
孙迎辉
E-mail:yinghuisun@suda.edu.cn
基金资助:
TANG Jiayi1,YAO Junjie1,ZHANG Xiaojun2,MA Liang2,ZHANG Tingmei3,NIU Zheng1,CHEN Xiangzhen1,ZHAO Liang1,JIANG Lin3,SUN Yinghui1,*( )
)
Received:2019-05-28
Online:2019-11-10
Published:2019-08-20
Contact:
SUN Yinghui
E-mail:yinghuisun@suda.edu.cn
Supported by:摘要:
设计合成了一种多壳层中空多孔结构的磷化铁(FeP)微球, 通过扫描电子显微镜(SEM)、 透射电子显微镜(TEM)、 X射线衍射(XRD)和X射线光电子能谱(XPS)对微球的表面形貌和物相组成等进行表征, 并通过电化学工作站测试了材料的析氢性能. 结果表明, FeP微球和掺杂导电剂碳纳米管(CNT)后的FeP/CNT复合电催化剂在宽pH范围的电解液中均展现出了优异的电催化活性, 在酸性、 碱性和中性条件下析氢反应过程中的塔菲尔斜率分别为55.0, 64.9, 163.2 mV/dec, 当电流密度达到10 mA/cm 2时, 过电势仅分别为97, 169, 495 mV(vs. RHE), 且表现出了超长的循环稳定性.
中图分类号:
TrendMD:
唐佳易,姚俊杰,张晓君,马梁,张婷妹,牛峥,陈祥祯,赵亮,江林,孙迎辉. 多壳层中空FeP微球的制备及全pH范围的电催化产氢性能. 高等学校化学学报, 2019, 40(11): 2340.
TANG Jiayi,YAO Junjie,ZHANG Xiaojun,MA Liang,ZHANG Tingmei,NIU Zheng,CHEN Xiangzhen,ZHAO Liang,JIANG Lin,SUN Yinghui. Multi-shell Hollow FeP Microspheres as Efficient Electrocatalyst for Hydrogen Evolution at All pH Values †. Chem. J. Chinese Universities, 2019, 40(11): 2340.
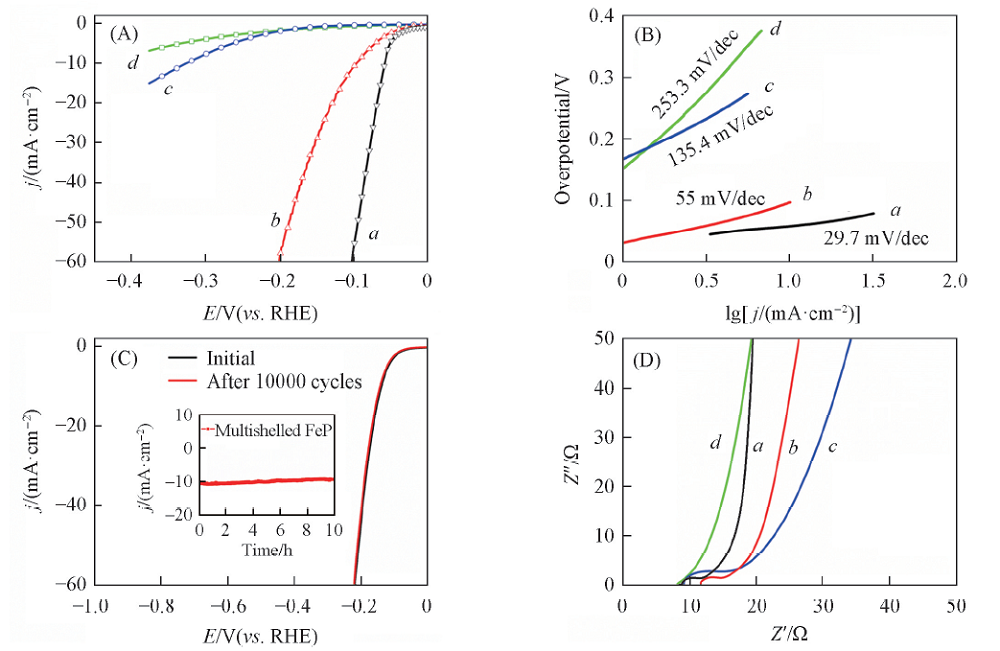
Fig.5 HER performances in 0.5 mol/L H2SO4 solution (A) Polarization curves(scan rate: 10 mV/s); (B) Tafel plots; (C) polarization stability plot of FeP/CNT(scan rate 500 mV/s); (D) impedance plots. a. Pt/C; b. FeP/CNT; c. FeP; d. CNT. Inset is current density curves for 10 h under constant overpotential of 97 mV.
| Electrolyte | a/mV | b /(mV·dec-1) | |
|---|---|---|---|
| 0.5 mol/L H2SO4 | 220 | 55.0 | 0.266 |
| 1.0 mol/L KOH | 320 | 64.9 | 0.021 |
| 1.0 mol/L Na2SO4 | 920 | 163.2 | 0.012 |
Table 1 Tafel kinetic parameters for the HER on FeP/CNT/GC electrode in different electrolytes
| Electrolyte | a/mV | b /(mV·dec-1) | |
|---|---|---|---|
| 0.5 mol/L H2SO4 | 220 | 55.0 | 0.266 |
| 1.0 mol/L KOH | 320 | 64.9 | 0.021 |
| 1.0 mol/L Na2SO4 | 920 | 163.2 | 0.012 |
| Catalyst | Electrolyte | η10/mA | Tafel slope/(mV·dec-1) | Ref. |
|---|---|---|---|---|
| FeP/CNT | 0.5 mol/L H2SO4 | 97 | 55 | This work |
| 1.0 mol/L KOH | 169 | 64.9 | This work | |
| 1.0 mol/L Na2SO4 | 495 | 163.2 | This work | |
| FeP powder/carbon | 0.5 mol/L H2SO4 | 110 | 57 | [ |
| 1.0 mol/L KOH | 185 | 93 | [ | |
| FeP bond carbon nanowires networks | 0.5 mol/L H2SO4 | 256 | 75.8 | [ |
| Nanoporous FeP nanosheets | 0.5 mol/L H2SO4 | 230 | 67 | [ |
| FeP nanowires | 0.5 mol/L H2SO4 | 96 | 37 | [ |
| 1.0 mol/L KOH | 194 | 67 | [ | |
| FeP NPs@NPC | 0.5 mol/L H2SO4 | 130 | 67 | [ |
| 1.0 mol/L KOH | 214 | 82 | [ | |
| 1.0 mol/L PBS | 386 | 136 | [ | |
| FeP NPs @hollow carbon nanobox | 0.5 mol/L H2SO4 | 88 | 49 | [ |
| 1.0 mol/L KOH | 180 | 71 | [ | |
| Ni-doped FeP/C hollow nanorods | 0.5 mol/L H2SO4 | 72 | 54 | [ |
| 1.0 mol/L KOH | 95 | 72 | [ | |
| 1.0 mol/L PBS | 117 | 70 | [ | |
| FeP NCs@NCNs | 0.5 mol/L H2SO4 | 114 | 84 | [ |
| 1.0 mol/L KOH | 205 | 70 | [ | |
| 1.0 mol/L PBS | 409 | 92 | [ | |
| CoP/Co2P NPs@NC | 0.5 mol/L H2SO4 | 126 | 79 | [ |
| 1.0 mol/L KOH | 198 | 82 | [ | |
| 1.0 mol/L PBS | 459 | —— | [ | |
| NiP NPs | 0.5 mol/L H2SO4 | 115 | 46 | [ |
Table 2 Comparison of the electrocatalytic activities of FeP/CNT with representative HER electrocatalysts in electrolytes
| Catalyst | Electrolyte | η10/mA | Tafel slope/(mV·dec-1) | Ref. |
|---|---|---|---|---|
| FeP/CNT | 0.5 mol/L H2SO4 | 97 | 55 | This work |
| 1.0 mol/L KOH | 169 | 64.9 | This work | |
| 1.0 mol/L Na2SO4 | 495 | 163.2 | This work | |
| FeP powder/carbon | 0.5 mol/L H2SO4 | 110 | 57 | [ |
| 1.0 mol/L KOH | 185 | 93 | [ | |
| FeP bond carbon nanowires networks | 0.5 mol/L H2SO4 | 256 | 75.8 | [ |
| Nanoporous FeP nanosheets | 0.5 mol/L H2SO4 | 230 | 67 | [ |
| FeP nanowires | 0.5 mol/L H2SO4 | 96 | 37 | [ |
| 1.0 mol/L KOH | 194 | 67 | [ | |
| FeP NPs@NPC | 0.5 mol/L H2SO4 | 130 | 67 | [ |
| 1.0 mol/L KOH | 214 | 82 | [ | |
| 1.0 mol/L PBS | 386 | 136 | [ | |
| FeP NPs @hollow carbon nanobox | 0.5 mol/L H2SO4 | 88 | 49 | [ |
| 1.0 mol/L KOH | 180 | 71 | [ | |
| Ni-doped FeP/C hollow nanorods | 0.5 mol/L H2SO4 | 72 | 54 | [ |
| 1.0 mol/L KOH | 95 | 72 | [ | |
| 1.0 mol/L PBS | 117 | 70 | [ | |
| FeP NCs@NCNs | 0.5 mol/L H2SO4 | 114 | 84 | [ |
| 1.0 mol/L KOH | 205 | 70 | [ | |
| 1.0 mol/L PBS | 409 | 92 | [ | |
| CoP/Co2P NPs@NC | 0.5 mol/L H2SO4 | 126 | 79 | [ |
| 1.0 mol/L KOH | 198 | 82 | [ | |
| 1.0 mol/L PBS | 459 | —— | [ | |
| NiP NPs | 0.5 mol/L H2SO4 | 115 | 46 | [ |
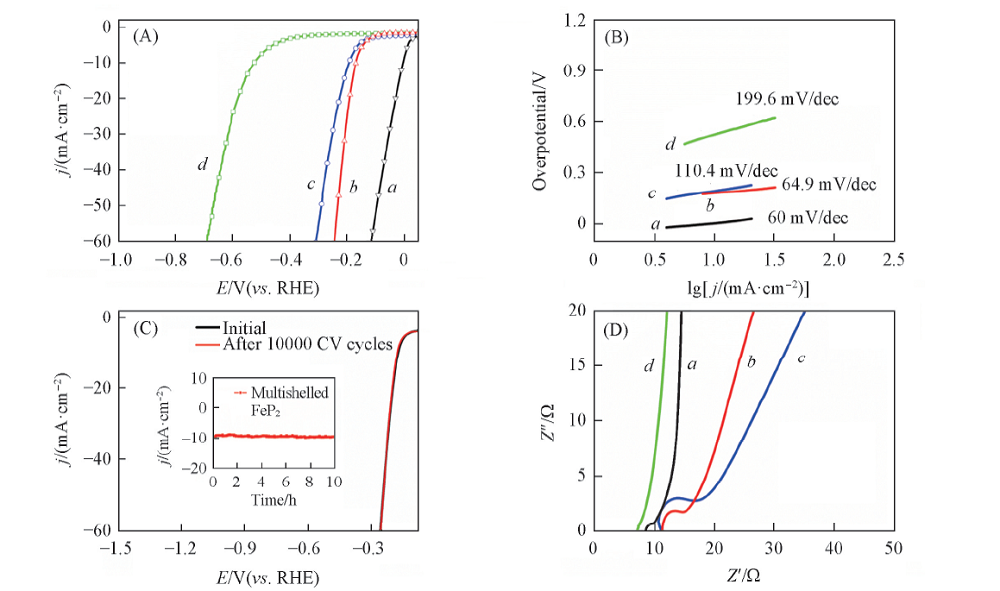
Fig.6 HER performances in 1.0 mol/L KOH solution (A) Polarization curves(scan rate: 10 mV/s); (B) Tafel plot; (C) polarization stability plot of FeP(scan rate 500 mV/s); (D) impedance plots. a. Pt/C; b. FeP/CNT; c. FeP; d. CNT. The inset is current density curves for 10 h under constant overpotential of 169 mV.
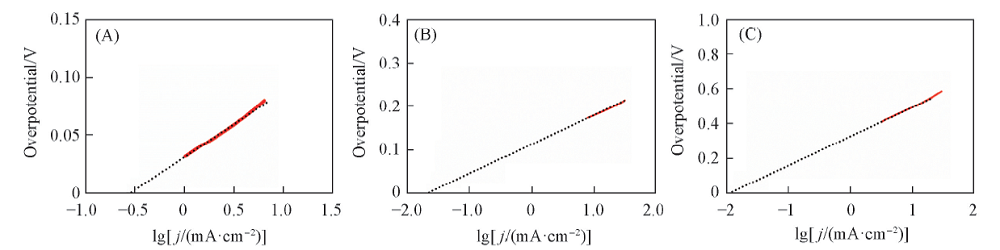
Fig.8 Exchange current densities of FeP/CNT electrocatalyst in different electrolytes calculated by the extrapolation of Tafel curves Electrolyte: (A) 0.5 mol/L H2SO4; (B) 1.0 mol/L KOH; (C) 1.0 mol/L Na2SO4 solution.
| [1] | Feng X. L., Qu Z. K., Chen J., Wang D. D., Chen X., Yang W. S., Chem. J. Chinese Universities, 2017,38(11), 1999— 2005 |
| ( 冯晓磊, 曲宗凯, 陈俊, 王登登, 陈旭, 杨文胜 . 高等学校化学学报, 2017,38(11), 1999— 2005) | |
| [2] | Jian J., Yuan L., Li H., Liu H. H., Zhang X. J., Sun X. J., Yuan H. M., Feng S. H., Chem. Res. Chinese Universities, 2019,35(2), 179— 185 |
| [3] | Pu Z., Amiinu I. S., Kou Z., Li W., Mu S ., Angew. Chem. Int. Ed., 2017,56(38), 11559— 11564 |
| [4] | Zheng Y., Jiao Y., Zhu Y., Li L. H., Han Y., Chen Y., Jaroniec M., Qiao S. Z., J. Am. Chem. Soc., 2016,138(49), 16174— 16181 |
| [5] | Han L., Dong S. J., Wang E. K ., Adv. Mater., 2016,28(42), 9266— 9291 |
| [6] | Tang C., Gan L. F., Zhang R., Lu W. B., Jiang X. E., Asiri A. M., Sun X. P., Wang J., Chen L ., Nano Lett., 2016,16(10), 6617— 6621 |
| [7] | Li D., Baydoun H., Kulikowski B., Brock S. L ., Chem. Mater., 2017,29(7), 3048— 3054 |
| [8] | Zhang T. Q., Liu J., Huang L. B., Zhang X. D., Sun Y. G., Liu X. C., Bin D. S., Chen X., Cao A. M., Hu J. S., Wan L. J., J. Am. Chem. Soc., 2017,139(32), 11248— 11253 |
| [9] | Jiang P., Liu Q., Liang Y. H., Tian J. Q., Asiri A. M., Sun X. P ., Angew. Chem. Int. Ed., 2014,53(47), 12855— 12859 |
| [10] | Lv C. C., Peng Z., Zhao Y. X., Huang Z. P., Zhang C., J. Mater. Chem. A, 2016,4(4), 1454— 1460 |
| [11] | Yan Y., Zhao B., Yi S. C., Wang X., J. Mater. Chem. A, 2016,4(33), 13005— 13010 |
| [12] | Popczun E. J., Read C. G., Roske C. W., Lewis N. S., Schaak R. E ., Angew. Chem. Int. Ed., 2014,53(21), 5427— 5430 |
| [13] | Chung D. Y., Jun S. W., Yoon G., Kim H., Yoo J. M., Lee K. S., Kim T., Shin H., Sinha A. K., Kwon S. G., Kang K., Hyeon T., Sung Y. E., J. Am. Chem. Soc., 2017,139(19), 6669— 6674 |
| [14] | Ran J. R., Ma T. Y., Gao G. P., Du X. W., Qiao S. Z ., Energy Environ. Sci., 2015,8(12), 3708— 3717 |
| [15] | Lu Q. P., Yu Y. F., Ma Q. L., Chen B., Zhang H ., Adv. Mater., 2016,28(10), 1917— 1933 |
| [16] | Sun H. M., Xu X. B., Yan Z. H., Chen X., Cheng F. Y., Weiss P. S., Chen J ., Chem. Mater., 2017,29(19), 8539— 8547 |
| [17] | Gao W., Yan M., Cheung H. Y., Xia Z. M., Zhou X. M., Qin Y. B., Wong C. Y., Ho J. C., Chang C. R., Qu Y. Q., Nano Energy, 2017,38, 290— 296 |
| [18] | Xiao P., Alam Sk M., Thia L., Ge X. M., Lim R. J., Wang J. Y., Lim K. H., Wang X ., Energy Environ. Sci., 2014,7(8), 2624— 2629 |
| [19] | Yan Y., Shi X. R., Miao M., He T., Dong Z. H., Zhan K., Yang J. H., Zhao B., Xia B. Y ., Nano Res., 2018,11(7), 3537— 3547 |
| [20] | Wang F. L., Yang X. D., Dong B. X., Yu X., Xue H. G., Feng L. G ., Electrochem. Commun., 2018,92, 33— 38 |
| [21] | Lu X. F., Yu L., Lou X. W., Sci. Adv., 2019, 5(2), eaav6009 |
| [22] | Wang X. H., Feng J., Bai Y. C., Zhang Q., Yin Y. D ., Chem. Rev., 2016,116(18), 10983— 11060 |
| [23] | Zeng Y., Luo J. Z., Wang Y. Z., Qiao L., Zou B., Zheng W. T., Nanoscale, 2017,9(44), 17576— 17584 |
| [24] | Zhang T. M., Zheng J. Z., Liang Z. Q., Zhao B., Zeng H. J., Guo W., Zhao L., Sun Y. H., Abdulhalim I., Jiang L., Electrochim. Acta, 2019,306, 151— 158 |
| [25] | Grosvenor A. P., Wik S. D., Cavell R. G., Mar A ., Inorg. Chem., 2005,44(24), 8988— 8998 |
| [26] | Tian J. Q., Liu Q., Liang Y. H., Xing Z. C., Asiri A. M., Sun X. P., ACS Appl. Mater. Interfaces, 2014,6(23), 20579— 20584 |
| [27] | Jiang P., Liu Q., Sun X. P., Nanoscale, 2014,6(22), 13440— 13445 |
| [28] | Li J. S., Wang Y., Liu C. H., Li S. L., Wang Y. G., Dong L. Z., Dai Z. H., Li Y. F., Lan Y. Q ., Nat Commun., 2016,7, 11204— 11211 |
| [29] | Li F., Zhao X. L., Mahmood J., Okyay M. S., Jung S. M., Ahmad I., Kim S. J., Han G. F., Park N., Baek J. B., ACS Nano, 2017,11(7), 7527— 7533 |
| [30] | Li M., Liu T. T., Bo X. J., Zhou M., Guo L. P., Guo S. J., Nano Energy, 2017,33, 221— 228 |
| [31] | Xu Y., Wu R., Zhang J. F., Shi Y. M., Zhang B ., Chem. Commun., 2013,49(59), 6656— 6658 |
| [32] | Son C. Y., Kwak I. H., Lim Y. R., Park J., Chem. Commun., 2016,52(13), 2819— 2822 |
| [33] | Pu Z. H., Amiinu I. S., Zhang C. T., Wang M., Kou Z. K., Mu S. C., Nanoscale, 2017,9(10), 3555— 3560 |
| [34] | Peng Z., Qiu X. Y., Yu Y., Jiang D., Wang H. T., Cai G. Y., Zhang X. X., Dong Z. H., Carbon, 2019,152, 16— 23 |
| [35] | Yu Y., Peng Z., Asif M., Wang H. T., Wang W., Wu Z. X., Wang Z. Y., Qiu X. Y., Tan H., Liu H. F ., ACS Sustainable Chem. Eng., 2018,6(9), 11587— 11594 |
| [36] | Lv X. W., Ren J. T., Wang Y. S., Liu Y. P., Yuan Z. Y., ACS Sustainable Chem. Eng., 2019,7(9), 8993— 9001 |
| [37] | Popczun E. J., McKone J. R., Read C. G., Biacchi A. J., Wiltrout A. M., Lewis N. S., Schaak R. E., J. Am. Chem. Soc., 2013,135(25), 9267— 9270 |
| [38] | Kucernak A. R., Zalitis C., J. Phys. Chem.C, 2016,120, 10721— 10745 |
| [39] | Watzele S., Fichtner J., Garlyyev B., Schwämmlein J. N., Bandarenka A. S., ACS Catal., 2018,8, 9456— 9462 |
| [1] | 范建玲, 唐灏, 秦凤娟, 许文静, 谷鸿飞, 裴加景, 陈文星. 氮掺杂超薄碳纳米片复合铂钌单原子合金催化剂的电化学析氢性能[J]. 高等学校化学学报, 2022, 43(9): 20220366. |
| [2] | 林高鑫, 王家成. 单原子掺杂二硫化钼析氢催化的进展和展望[J]. 高等学校化学学报, 2022, 43(9): 20220321. |
| [3] | 汪思聪, 庞贝贝, 刘潇康, 丁韬, 姚涛. XAFS技术在单原子电催化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220487. |
| [4] | 杨丽君, 于洋, 张蕾. 双功能2D/3D杂化结构Co2P-CeO x 异质结一体化电极的构筑及电催化尿素氧化辅助制氢性能[J]. 高等学校化学学报, 2022, 43(6): 20220082. |
| [5] | 黄汉浩, 卢湫阳, 孙明子, 黄勃龙. 石墨炔原子催化剂的崭新道路:基于自验证机器学习方法的筛选策略[J]. 高等学校化学学报, 2022, 43(5): 20220042. |
| [6] | 张小玉, 薛冬萍, 杜宇, 蒋粟, 魏一帆, 闫文付, 夏会聪, 张佳楠. MOF衍生碳基电催化剂限域催化O2还原和CO2还原反应[J]. 高等学校化学学报, 2022, 43(3): 20210689. |
| [7] | 丁钦, 张梓轩, 徐培程, 李晓宇, 段莉梅, 王寅, 刘景海. Cu, Ni, Co掺杂对Fe碳纳米管的结构及电催化性能的影响[J]. 高等学校化学学报, 2022, 43(11): 20220421. |
| [8] | 何宇婧, 李佳乐, 王东洋, 王福玲, 肖作旭, 陈艳丽. 锌活化Fe/Co/N掺杂的生物质碳基高效氧还原催化剂[J]. 高等学校化学学报, 2022, 43(11): 20220475. |
| [9] | 王祖民, 孟程, 于然波. 过渡金属磷化物析氢催化剂的掺杂调控[J]. 高等学校化学学报, 2022, 43(11): 20220544. |
| [10] | 武亚强, 刘思明, 金顺敬, 严永情, 王朝, 陈丽华, 苏宝连. 锌掺杂NiCoP多孔双层阵列电极材料的制备及电催化产氢性能[J]. 高等学校化学学报, 2021, 42(8): 2483. |
| [11] | 季小好, 王祖民, 陈晓煜, 于然波. 过渡金属磷化物的制备及电催化析氢性能提升策略[J]. 高等学校化学学报, 2021, 42(5): 1377. |
| [12] | 杨涛, 姚会影, 李青, 郝伟, 迟力峰, 朱嘉. 高催化活性M-BHT(M=Co, Cu)电催化还原CO2为CH4的密度泛函理论研究[J]. 高等学校化学学报, 2021, 42(4): 1268. |
| [13] | 张楠, 韩阔, 李悦, 王春茹, 赵凤, 韩冬雪, 牛利. 尖晶石型过渡金属硫化物CuCo2S4与MoS2复合材料的制备及电催化析氢性能[J]. 高等学校化学学报, 2021, 42(4): 1307. |
| [14] | 何倩倩, 王哲, 孟令佳, 陈乾, 宫勇吉. 基于过渡金属二硫族化物析氢催化的研究进展[J]. 高等学校化学学报, 2021, 42(2): 523. |
| [15] | 史江维, 孟楠楠, 郭亚梅, 于一夫, 张兵 . 二维材料用于电催化析氢的研究进展[J]. 高等学校化学学报, 2021, 42(2): 492. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||