

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (5): 1377.doi: 10.7503/cjcu20200658
收稿日期:2020-09-07
出版日期:2021-05-10
发布日期:2020-12-08
通讯作者:
于然波
E-mail:ranboyu@ustb.edu.cn
基金资助:
JI Xiaohao1, WANG Zumin2, CHEN Xiaoyu1, YU Ranbo1( )
)
Received:2020-09-07
Online:2021-05-10
Published:2020-12-08
Contact:
YU Ranbo
E-mail:ranboyu@ustb.edu.cn
Supported by:摘要:
氢能是一种绿色、 高效的二次能源, 在廉价的非贵金属催化剂的辅助下, 电解水制氢以其低成本和高效率受到广泛关注. 过渡金属磷化物因其独特近似球形三角棱柱单元结构能够暴露出更多配位不饱和表面原子, 因此在电解水制氢中表现出优异的催化活性和强耐腐蚀性. 本文综述了过渡金属磷化物的制备方法和在电催化析氢中的应用和性能的改善策略. 最后讨论了过渡金属磷化物催化剂存在的一些亟待解决的问题, 并展望了其未来的发展方向.
中图分类号:
TrendMD:
季小好, 王祖民, 陈晓煜, 于然波. 过渡金属磷化物的制备及电催化析氢性能提升策略. 高等学校化学学报, 2021, 42(5): 1377.
JI Xiaohao, WANG Zumin, CHEN Xiaoyu, YU Ranbo. Overview of Transition Metal Phosphide Catalysts and Hydrogen Production by Electrolyzed Water. Chem. J. Chinese Universities, 2021, 42(5): 1377.
Transition metal compound | Typical material | Conductivity | Acid?base resistance | Synthesis procedure |
|---|---|---|---|---|
| TMDs | MoS2, WS2 | Good along the lamellar, poor in vertical direction | Only active in acid solution | Dangerous or complex exfoliation required |
| TMOs | MoO2, Co3O4, WO3-x | Poor | Poor | Low temperature, low cost |
| TMCs/TMNs | Mo2C, Mo2N | Good | Good | High temperature, incomplete reaction |
| TMPs | Ni2P, CoP | Good | Good | Low temperature, special attention to air impermeability |
| Alloy | NiMo | Good | Poor to medium | Always high temperature required |
Table 1 Comparison of common hydrogen evolution transition metal compounds
Transition metal compound | Typical material | Conductivity | Acid?base resistance | Synthesis procedure |
|---|---|---|---|---|
| TMDs | MoS2, WS2 | Good along the lamellar, poor in vertical direction | Only active in acid solution | Dangerous or complex exfoliation required |
| TMOs | MoO2, Co3O4, WO3-x | Poor | Poor | Low temperature, low cost |
| TMCs/TMNs | Mo2C, Mo2N | Good | Good | High temperature, incomplete reaction |
| TMPs | Ni2P, CoP | Good | Good | Low temperature, special attention to air impermeability |
| Alloy | NiMo | Good | Poor to medium | Always high temperature required |
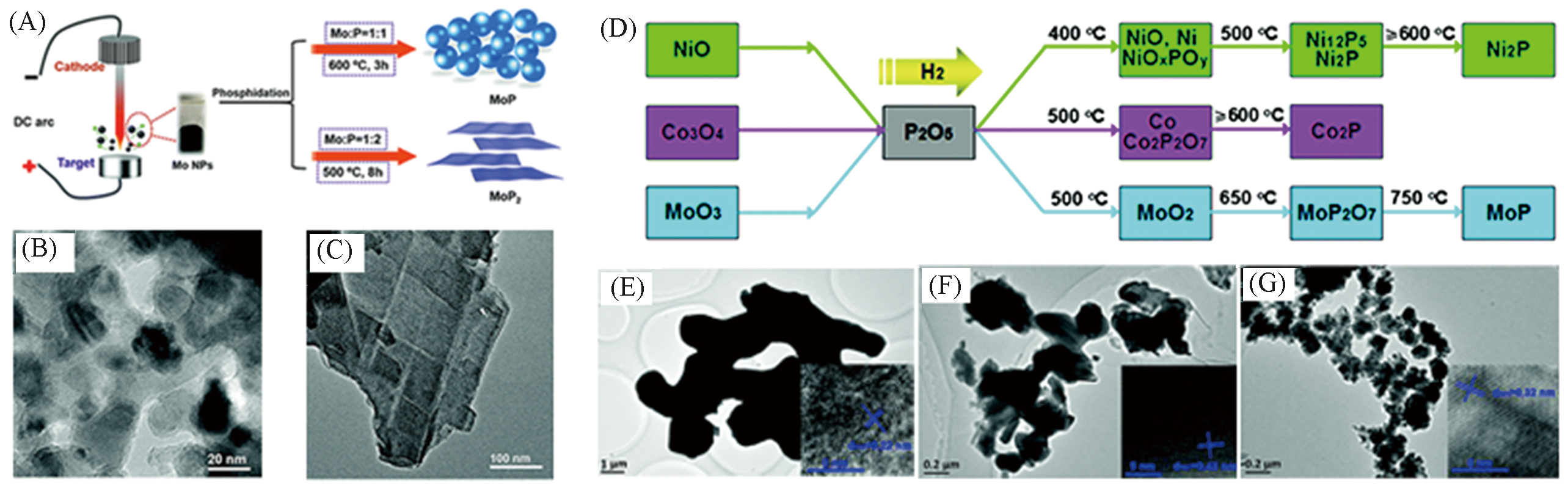
Fig.2 Schematic diagram of the preparation process of MoP nanoparticles and MoP2 nanosheets(A), TEM images of MoP nanoparticles(B) and MoP2 nanosheets(C)[37], schematic diagram of the synthesis of Ni, Co, Mo oxide and P2O5 co?reduction to prepare the corresponding phosphide(D), TEM images of S?Ni2P(E), S?Co2P(F) and S?MoP(G)[38](A―C) Copyright 2016, Royal Society of Chemistry; (D―G) Copyright 2020, Royal Society of Chemistry.

Fig.4 Schematic diagram of NixPy nanoparticle synthesis(A), TEM images of crystalline NPs of nickel NPs(B,C), hollow NixPy NPs(D,E), solid NixPy NPs(F,G)[44], the deposition apparatus(H), SEM images of FeP(I), Fe2P(J), and Fe3P(K) on FTO[45](B, D, F) 50 nm; (C, E, G) 5 nm. (A―G) Copyright 2009, American Chemical Society; (H―K) Copyright 2018, American Chemical Society.
| Synthesis system | Subdivision | Advantage | Disadvantage |
|---|---|---|---|
| Solid phase synthesis | High temperature element direct reaction method, solid phase exchange reaction, metal oxide and phosphorus oxide co?reduction reaction | Professional experimental equipment is not demanding and no additional preparation process is required | The required temperature is relatively high(600―800 ℃); the product size is large, the reaction time is long, and strict sealing is required |
| Vapor?solid reaction | Low temperature(300 ℃), simple equipment | The morphology of the product is limited by the morphology of the precursor, and most of the phosphorus source gas reactants are toxic and flammable | |
Vapor phase synthesis | Metal organic precursor pyrolysis method, chemical vapor deposition method | Temperature and reaction time are lower than solid phase synthesis | Released gases are toxic, and strict sealing is required |
Liquid phase synthesis | Liquid reflux method, solvothermal method, hydrothermal method | The reaction temperature is low (<400 ℃), no complicated experimental equipment is needed, and the product size, shape, composition and structure are controllable | More toxic and harmful organic solvents are used to release toxic gases and produce more waste liquid |
Other synthetic methods | Electrochemical method, microwave assisted method | Low temperature, fast reaction speed, less harmful products | The size, morphology and structure of the obtained product cannot be finely controlled |
Table 2 Advantages and disadvantages of common synthesis methods for transition metal phosphides
| Synthesis system | Subdivision | Advantage | Disadvantage |
|---|---|---|---|
| Solid phase synthesis | High temperature element direct reaction method, solid phase exchange reaction, metal oxide and phosphorus oxide co?reduction reaction | Professional experimental equipment is not demanding and no additional preparation process is required | The required temperature is relatively high(600―800 ℃); the product size is large, the reaction time is long, and strict sealing is required |
| Vapor?solid reaction | Low temperature(300 ℃), simple equipment | The morphology of the product is limited by the morphology of the precursor, and most of the phosphorus source gas reactants are toxic and flammable | |
Vapor phase synthesis | Metal organic precursor pyrolysis method, chemical vapor deposition method | Temperature and reaction time are lower than solid phase synthesis | Released gases are toxic, and strict sealing is required |
Liquid phase synthesis | Liquid reflux method, solvothermal method, hydrothermal method | The reaction temperature is low (<400 ℃), no complicated experimental equipment is needed, and the product size, shape, composition and structure are controllable | More toxic and harmful organic solvents are used to release toxic gases and produce more waste liquid |
Other synthetic methods | Electrochemical method, microwave assisted method | Low temperature, fast reaction speed, less harmful products | The size, morphology and structure of the obtained product cannot be finely controlled |
| Catalyst | Precursor | Phosphorus source/ atmosphere | Temperature/℃ | Time/h | Tafel slope/ (mV·dec-1) | η10/mV | Ref. |
|---|---|---|---|---|---|---|---|
| Ni12P5 | Nickel(II) 2,4?pentanedionate | TPP/N2 | 300 | 0.5 | 63 | 110 | [ |
| Ni2P | Ni(AC)2 | P(SiMe3)3/N2 | 180 | 1 | 41.4 | 78 | [ |
| Ni2P | Ni | TOP | 320 | 2 | 68 | ― | [ |
| Co2P | Co?tri?n?octylphosphine | TPP/N2 | 350 | 0.25 | 45 | 95 | [ |
| Co2P | Co | TOP/Ar | 290 | 0.67 | 45 | 95 | [ |
| Co | 330 | 1 | 50 | 75 | |||
| Ni2P | Ni | P | 200 | 48 | 106.1 | 131 | [ |
| MoP | Mo(CO)6 | TOP/Ar | 320 | 2 | 45 | 90 | [ |
| Co2P | (CH3COO)2Co·4H2O | TPP/N2 | 370 | 0.17 | 51.7 | η20 =167 | [ |
| CoP | Co(acac)2 | TOPO/Ar | 355―360 | 3 | 48 | 100 | [ |
Table 3 Synthesis conditions and hydrogen evolution performance in 0.5 mol/L H2SO4 of various transition metal phosphides prepared by liquid phase method
| Catalyst | Precursor | Phosphorus source/ atmosphere | Temperature/℃ | Time/h | Tafel slope/ (mV·dec-1) | η10/mV | Ref. |
|---|---|---|---|---|---|---|---|
| Ni12P5 | Nickel(II) 2,4?pentanedionate | TPP/N2 | 300 | 0.5 | 63 | 110 | [ |
| Ni2P | Ni(AC)2 | P(SiMe3)3/N2 | 180 | 1 | 41.4 | 78 | [ |
| Ni2P | Ni | TOP | 320 | 2 | 68 | ― | [ |
| Co2P | Co?tri?n?octylphosphine | TPP/N2 | 350 | 0.25 | 45 | 95 | [ |
| Co2P | Co | TOP/Ar | 290 | 0.67 | 45 | 95 | [ |
| Co | 330 | 1 | 50 | 75 | |||
| Ni2P | Ni | P | 200 | 48 | 106.1 | 131 | [ |
| MoP | Mo(CO)6 | TOP/Ar | 320 | 2 | 45 | 90 | [ |
| Co2P | (CH3COO)2Co·4H2O | TPP/N2 | 370 | 0.17 | 51.7 | η20 =167 | [ |
| CoP | Co(acac)2 | TOPO/Ar | 355―360 | 3 | 48 | 100 | [ |
| Catalyst | Precursor | Phosphorus source/ atmosphere | Temperature/℃ | Time/h | Electrolyte | Tafel slope/ (mV·dec-1) | η10/mV | Ref. |
|---|---|---|---|---|---|---|---|---|
| C@NiPx | Ni/carbon fiber | NaH2PO2/Ar | 300 | 2 | 1.0 mol/L KOH | 48.3 | 118 | [ |
| MoP2 | MoS2/carbon cloth | NaH2PO2/Ar | 700 | 1.07 | 0.5 mol/L H2SO4 | 63.6 | 58 | [ |
| 1.0 mol/L KOH | 70 | 67 | ||||||
| 1.0 mol/L PBS | 98.3 | 85 | ||||||
| MoP2/Mo | MoOx/Mo | NaH2PO2/N2 | 750 | 2 | 0.5 mol/L H2SO4 | 57 | 143 | [ |
| 1.0 mol/L KOH | 80 | 194 | ||||||
| 1.0 mol/L PBS | 81 | 211 | ||||||
| MoP2 | MoO3, red phosphorus | Red phosphorus/ vacuum | 850 | 5 | 0.5 mol/L H2SO4 | 52 | 121 | [ |
| CoP/C | Co3O4 | NaH2PO2·H2O/N2 | 300 | 2 | 1.0 mol/L KOH | 85.9 | 111 | [ |
| CoP/Ti | Co3O4 | NaH2PO2/Ar | 300 | 2 | 0.5 mol/L H2SO4 | 40 | 203 | [ |
| 1.0 mol/L KOH | 115 | ― | ||||||
| CoP/Ti | Co(OH)2/Ti | NaH2PO2/Ar | 300 | 1 | 0.5 mol/L H2SO4 | 43 | 90 | [ |
| CoP/Ti | CoP precursor | NaH2PO2 | 300 | 2 | 0.5 mol/L H2SO4 | 49.3 | 45 | [ |
| 1.0 mol/L KOH | 49.1 | 60 | ||||||
| CoP/C | Co/C | NaH2PO2 | 300 | ― | 0.5 mol/L H2SO4 | 30.1 | 49 | [ |
| 1.0 mol/L KOH | 42.6 | 48 | ||||||
| CoP/C | Co(OH)F | NaH2PO2/Ar | 300 | 1 | 0.5 mol/L H2SO4 | 51 | 67 | [ |
| 1.0 mol/L KOH | 129 | 209 | ||||||
| CoP/C | Co(CO3)0.5(OH)·0.11H2O/C | NaH2PO2 | 300 | 1 | 1.0 mol/L KOH | 60 | 95 | [ |
| FeP/Ti | β?FeOOH | NaH2PO2/Ar | 300 | 2 | 0.5 mol/L H2SO4 | 38 | 55 | [ |
| FeP/C | Fe2O3 | NaH2PO2/Ar | 300 | 2 | 0.5 mol/L H2SO4 | 45 | 58 | [ |
| FeP/C | Fe3O4 | NaH2PO2/Ar | 300 | 2 | 0.5 mol/L H2SO4 | 32 | η20=54 | [ |
Table 5 Synthesis conditions and hydrogen evolution performance of various transition metal phosphides prepared by gas-solid method
| Catalyst | Precursor | Phosphorus source/ atmosphere | Temperature/℃ | Time/h | Electrolyte | Tafel slope/ (mV·dec-1) | η10/mV | Ref. |
|---|---|---|---|---|---|---|---|---|
| C@NiPx | Ni/carbon fiber | NaH2PO2/Ar | 300 | 2 | 1.0 mol/L KOH | 48.3 | 118 | [ |
| MoP2 | MoS2/carbon cloth | NaH2PO2/Ar | 700 | 1.07 | 0.5 mol/L H2SO4 | 63.6 | 58 | [ |
| 1.0 mol/L KOH | 70 | 67 | ||||||
| 1.0 mol/L PBS | 98.3 | 85 | ||||||
| MoP2/Mo | MoOx/Mo | NaH2PO2/N2 | 750 | 2 | 0.5 mol/L H2SO4 | 57 | 143 | [ |
| 1.0 mol/L KOH | 80 | 194 | ||||||
| 1.0 mol/L PBS | 81 | 211 | ||||||
| MoP2 | MoO3, red phosphorus | Red phosphorus/ vacuum | 850 | 5 | 0.5 mol/L H2SO4 | 52 | 121 | [ |
| CoP/C | Co3O4 | NaH2PO2·H2O/N2 | 300 | 2 | 1.0 mol/L KOH | 85.9 | 111 | [ |
| CoP/Ti | Co3O4 | NaH2PO2/Ar | 300 | 2 | 0.5 mol/L H2SO4 | 40 | 203 | [ |
| 1.0 mol/L KOH | 115 | ― | ||||||
| CoP/Ti | Co(OH)2/Ti | NaH2PO2/Ar | 300 | 1 | 0.5 mol/L H2SO4 | 43 | 90 | [ |
| CoP/Ti | CoP precursor | NaH2PO2 | 300 | 2 | 0.5 mol/L H2SO4 | 49.3 | 45 | [ |
| 1.0 mol/L KOH | 49.1 | 60 | ||||||
| CoP/C | Co/C | NaH2PO2 | 300 | ― | 0.5 mol/L H2SO4 | 30.1 | 49 | [ |
| 1.0 mol/L KOH | 42.6 | 48 | ||||||
| CoP/C | Co(OH)F | NaH2PO2/Ar | 300 | 1 | 0.5 mol/L H2SO4 | 51 | 67 | [ |
| 1.0 mol/L KOH | 129 | 209 | ||||||
| CoP/C | Co(CO3)0.5(OH)·0.11H2O/C | NaH2PO2 | 300 | 1 | 1.0 mol/L KOH | 60 | 95 | [ |
| FeP/Ti | β?FeOOH | NaH2PO2/Ar | 300 | 2 | 0.5 mol/L H2SO4 | 38 | 55 | [ |
| FeP/C | Fe2O3 | NaH2PO2/Ar | 300 | 2 | 0.5 mol/L H2SO4 | 45 | 58 | [ |
| FeP/C | Fe3O4 | NaH2PO2/Ar | 300 | 2 | 0.5 mol/L H2SO4 | 32 | η20=54 | [ |
| Catalyst | Precursor | Phosphorus source/ atmosphere | Time/h | Electrolyte | Tafel slope/ (mV·dec-1) | η10/mV | Ref. |
|---|---|---|---|---|---|---|---|
| Ni?P | NiSO4·7H2O | NaH2PO2 | 2 | 1.0 mol/L KOH | 50 | 80 | [ |
| CoP | CoCl2 | NaH2PO2 | 0.25 | 0.5 mol/L H2SO4 | 49.6 | 85 | [ |
| Co?P/FTO | CoCl2·6H2O | NaH2PO4 | ― | 1.0 mol/L KOH | 54 | 125 | [ |
Table 4 Summary of the synthesis conditions and hydrogen evolution properties of various transition metal phosphides prepared by electrochemical methods at room temperature
| Catalyst | Precursor | Phosphorus source/ atmosphere | Time/h | Electrolyte | Tafel slope/ (mV·dec-1) | η10/mV | Ref. |
|---|---|---|---|---|---|---|---|
| Ni?P | NiSO4·7H2O | NaH2PO2 | 2 | 1.0 mol/L KOH | 50 | 80 | [ |
| CoP | CoCl2 | NaH2PO2 | 0.25 | 0.5 mol/L H2SO4 | 49.6 | 85 | [ |
| Co?P/FTO | CoCl2·6H2O | NaH2PO4 | ― | 1.0 mol/L KOH | 54 | 125 | [ |
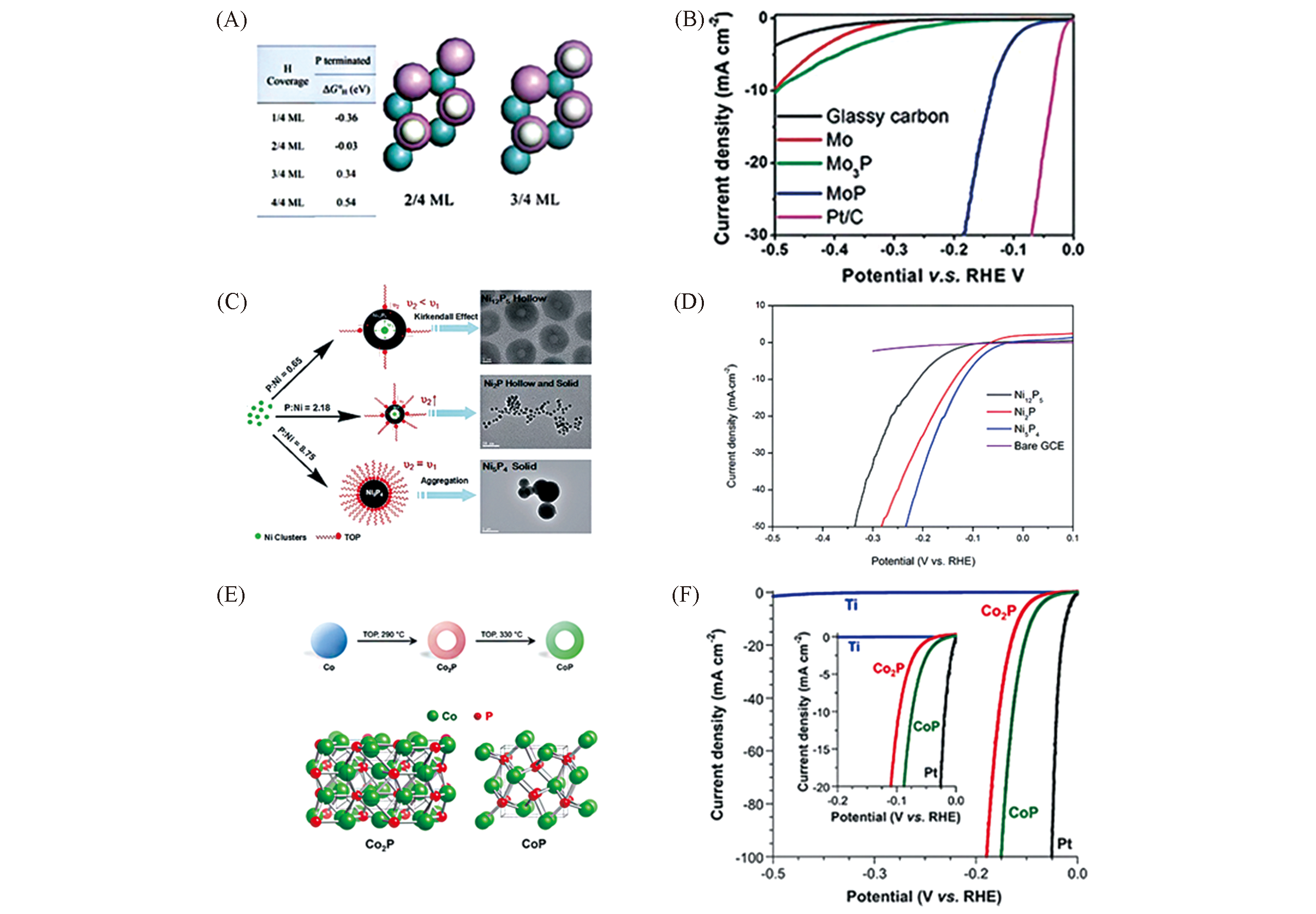
Fig.6 Gibbs free energy of H adsorption on Mo(110) and MoP(001)(A), polarization curves of MoP(B)[77], possible formation mechanism of the as?synthesized nickel phosphide NCs(C), LSV curves of bare Ni5P4 NCs(D)[78], schematic showing the multistep reaction pathway for Co2P and CoP and crystal structures(E), polarization data for Co2P/Ti and CoP/Ti(F)[59](A, B) Copyright 2014, Royal Society of Chemistry; (C, D) Copyright 2015, Royal Society of Chemistry; (E, F) Copyright 2015, American Chemical Society.
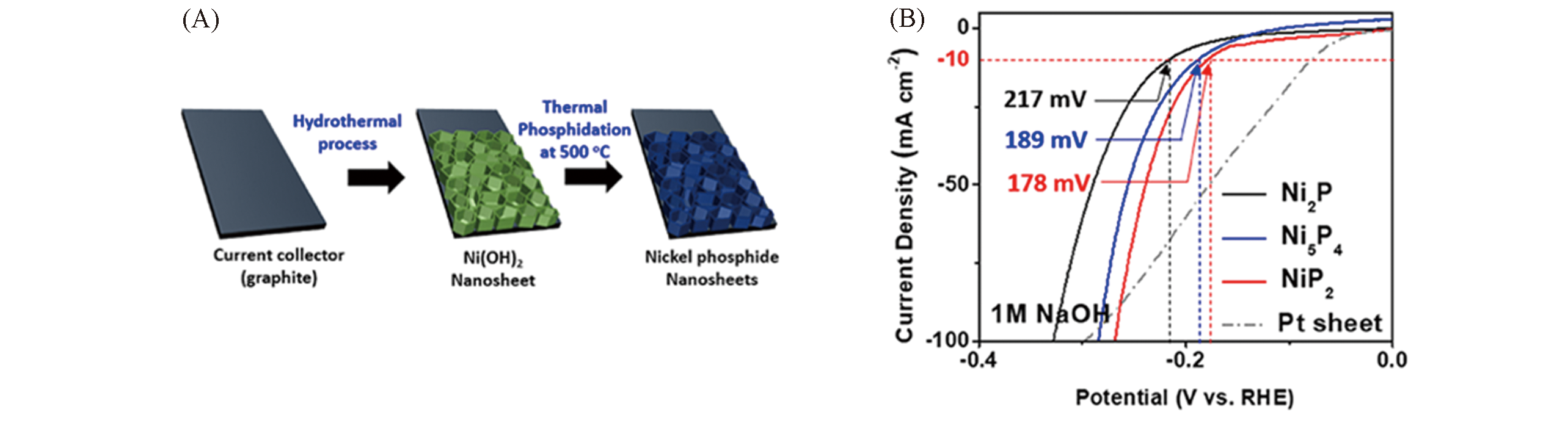
Fig.7 Schematic for the preparation of nickel phosphides(A), the polarization curves of nickel phosphide nanosheets with different phases in 1 mol/L NaOH(B)[34]Copyright 2020, Elsevier.
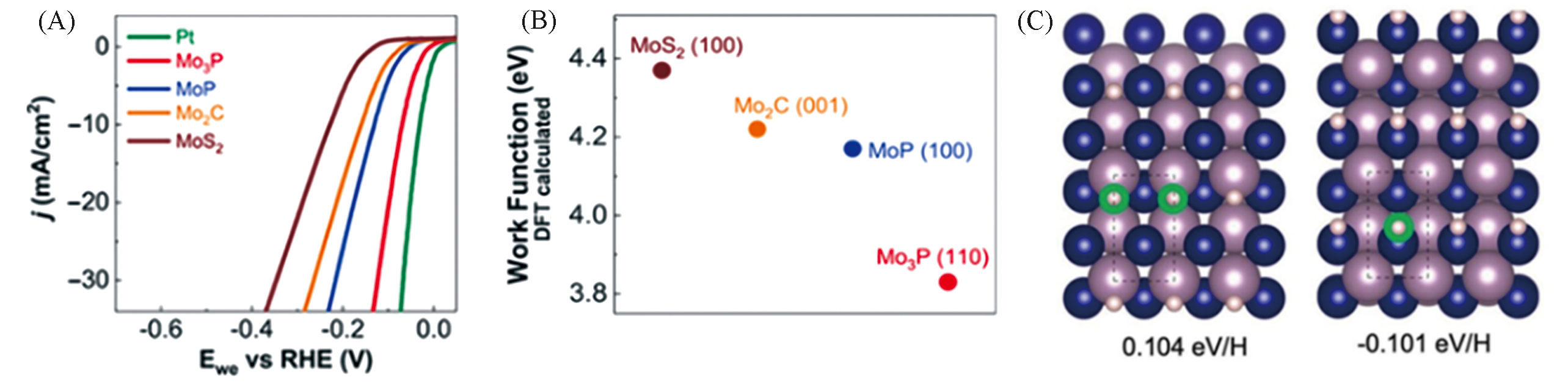
Fig.8 Polarization data in 0.5 mol/L H2SO4 for Mo3P, MoP, Mo2C and MoS2(A), comparison of absolute work function for each catalyst(B), top view of hydrogen adsorption on MoP(100) corresponded to two different adsorption sites of Mo sites and P sites(large purple atom: Mo; blue atom: P; small purple atom: H)(C)[15]Copyright 2019, Wiley?VCH.
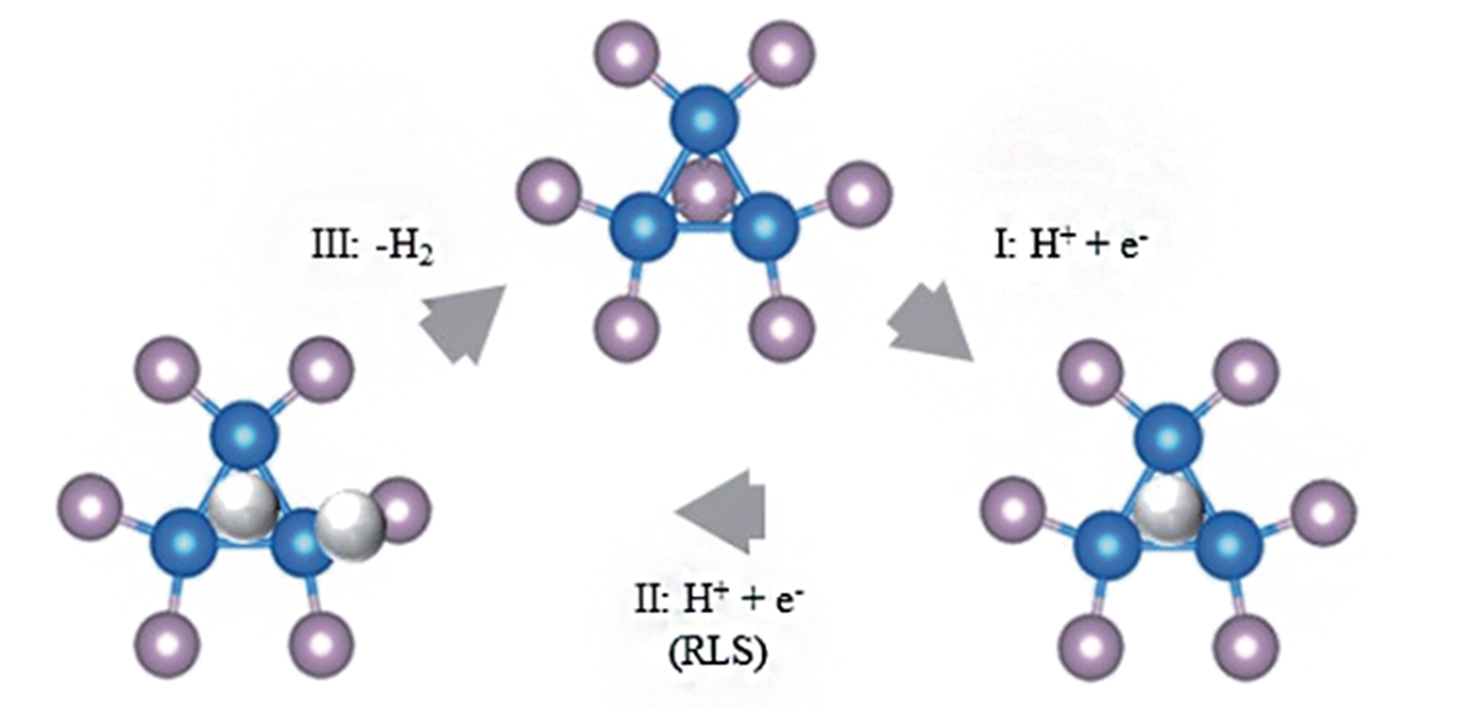
Fig.9 Schematic representation of the reaction mechanism suggested by Liu and Rodriguez[28](blue: Ni; purple: P; white: H)[79]Copyright 2015, Royal Society of Chemistry.
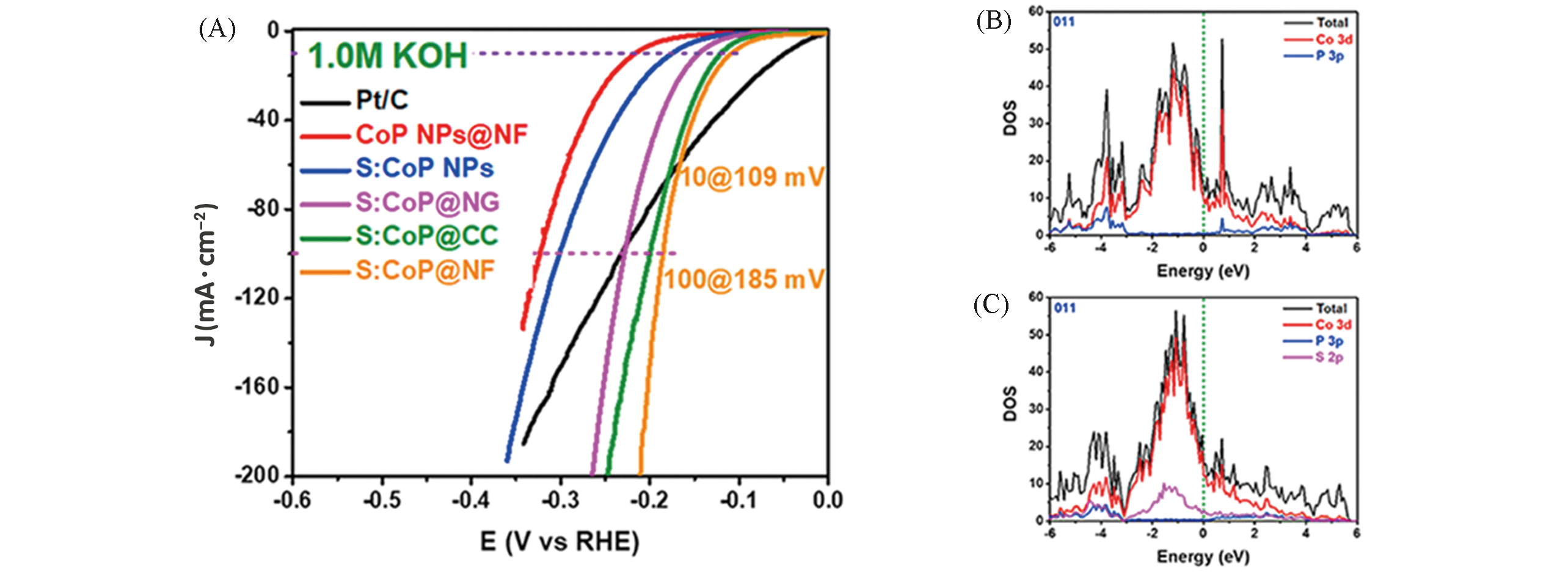
Fig.10 iR?corrected polarization curves during HER in 1.0?mol/L KOH for S:CoP NPs(A), total density of states(TDOS) and projected density of states(PDOS) for CoP(011) surface(B, C)[84]Copyright 2018, Elsevier.
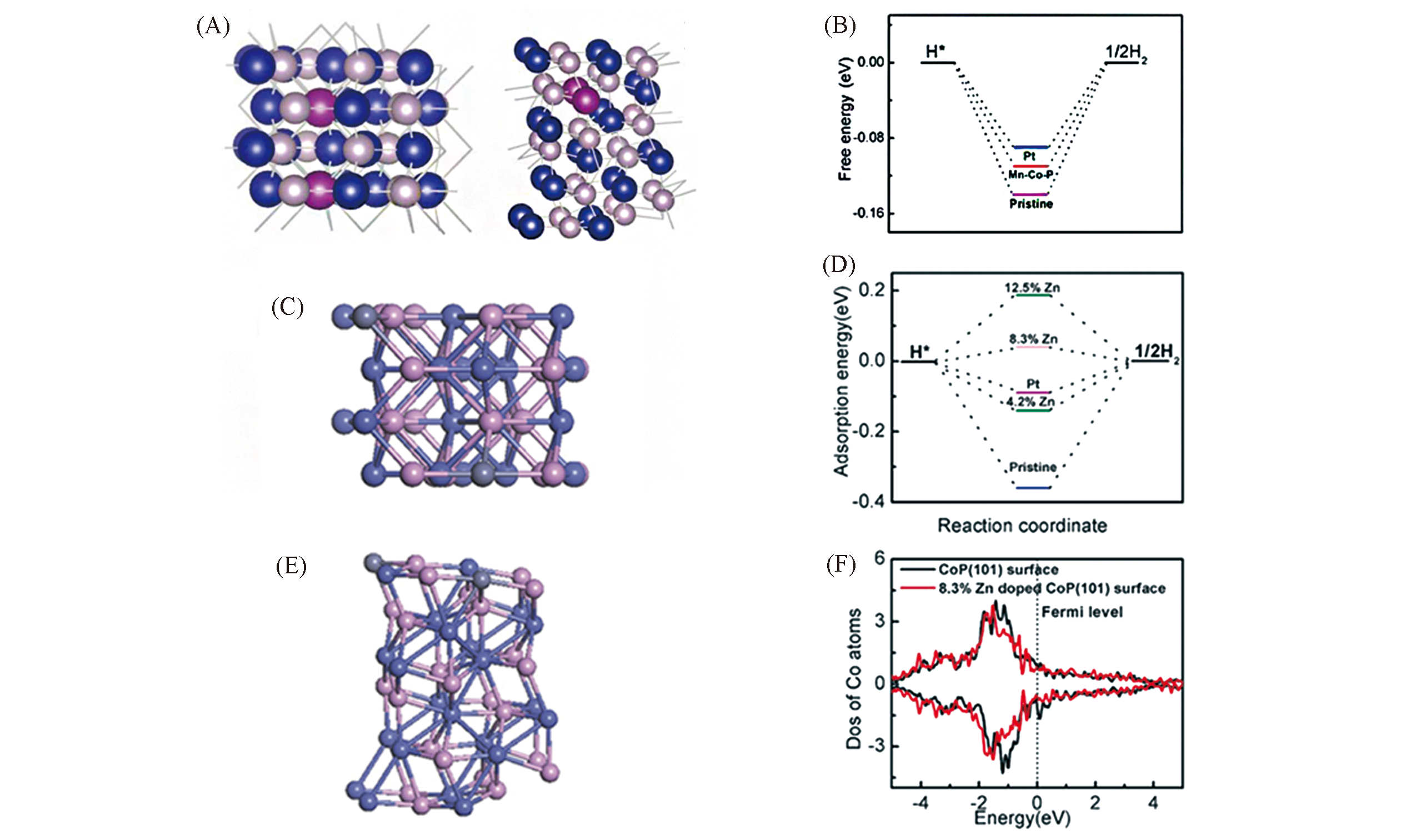
Fig.11 Top and side view of Mn?Co?P(101) with two Mn atoms replacing the subsurface Co atoms(A), free?energy diagram for HER on Mn?Co?P(101) with 8.3%(atom fraction) Mn doping(B)[85], top view(C) and side view(E) of 8.3% Zn‐doped CoP, free energy diagram for HER on pristine CoP(101), CoP(101) with 4.2%, 8.3% and 12.5% Zn doping(D), calculated DOS of surface Co atoms on the pristine CoP(101) and CoP(101) surface with 8.3% Zn(F)[86](A, B) Copyright 2016, American Chemical Society; (C―F) Copyright 2017, Wiley?VCH.
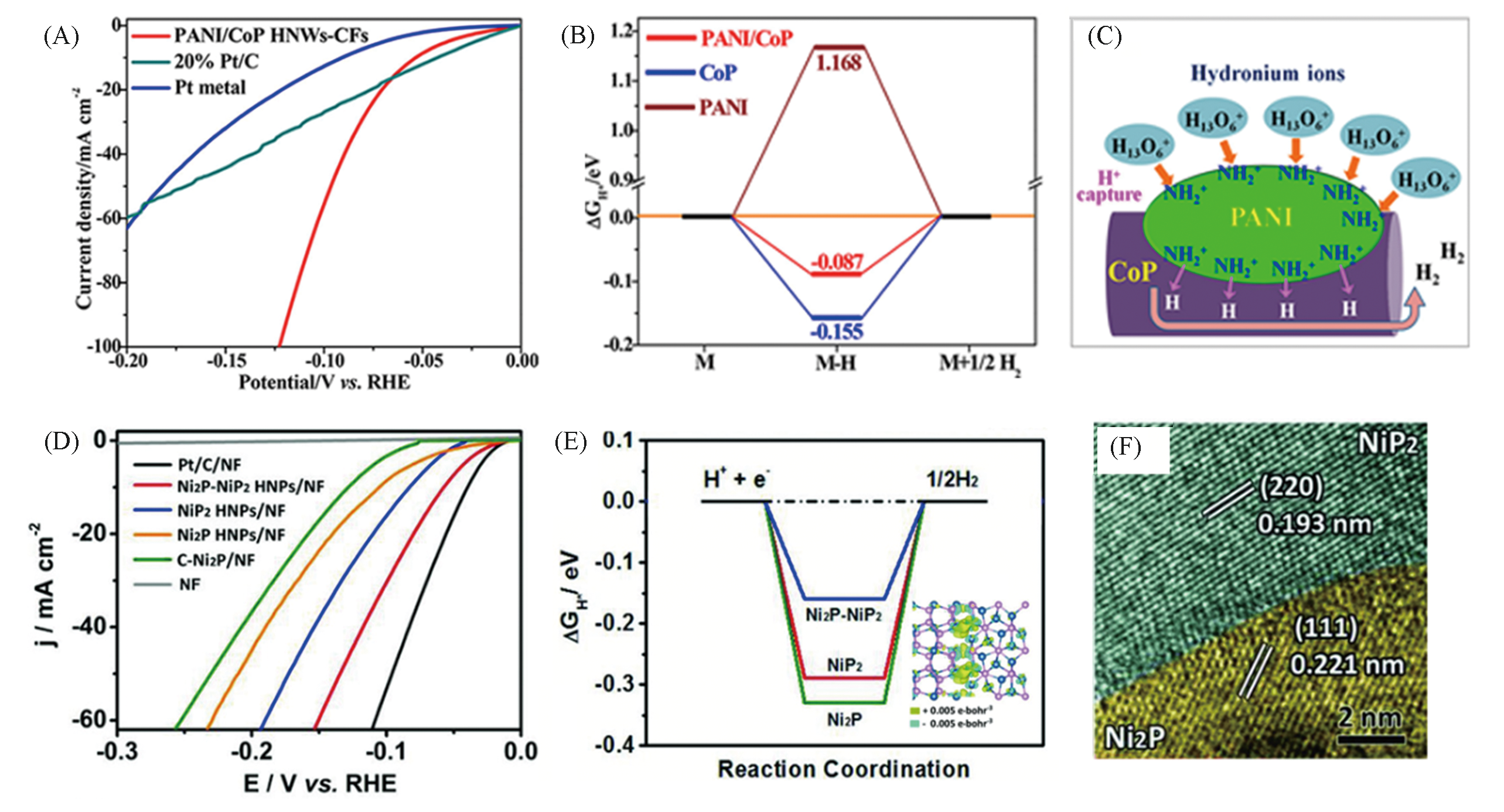
Fig.12 Polarization curves of PANI/CoP HNWs?CFs(A), free energy profile of H adsorption on the different catalysts(B), scheme of HER improvement by capturing the H+ in hydronium ions to form the protonated amine groups on PANI/CoP HNWs(C)[88], HER polarization curves of the Ni2P?NiP2 HNPs/NF(D), HER free‐energy diagram calculated at the equilibrium potential for the Ni2P?NiP2 polymorph and charge density difference plot at the Ni2P?NiP2 interface(E), HRTEM image of the Ni2P?NiP2 HNPs(F)[89](A―C) Copyright 2018, American Chemical Society; (D―F) Copyright 2018, Wiley?VCH.
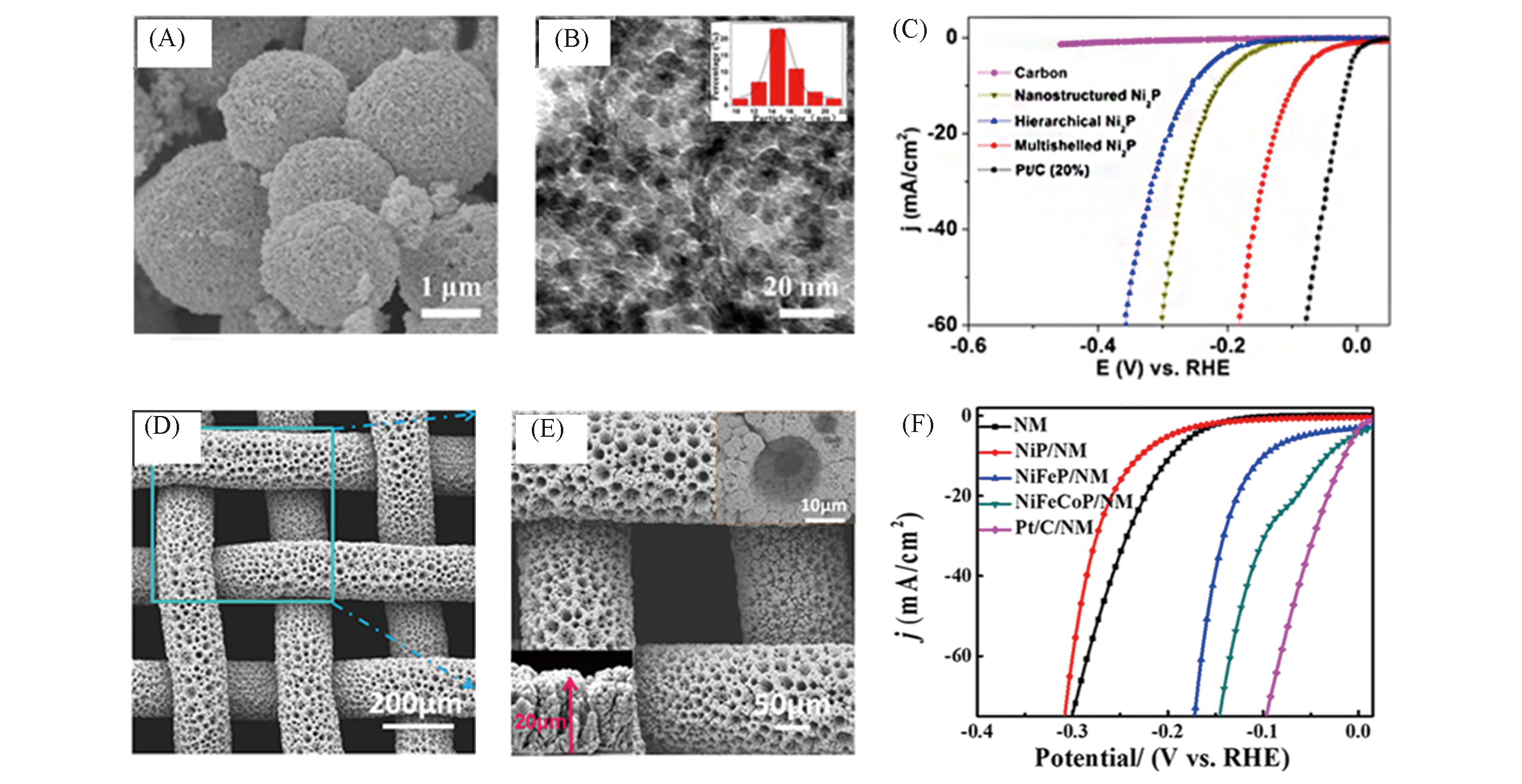
Fig.13 Scanning electron micrograph of the Ni2P particles(A) and high?magnification TEM image of the shell of a sphere(B), linear sweep voltammetry(LSV) polarization curves of multishelled Ni2P(C)[91], SEM images of the NiFeCoP/NM with the low(D) and the high magnification(E), iR?corrected LSV curves for NiFeCoP/NM(F)[93](A―C) Copyright 2017, American Chemical Society; (D―F) Copyright 2019, Elsevier.
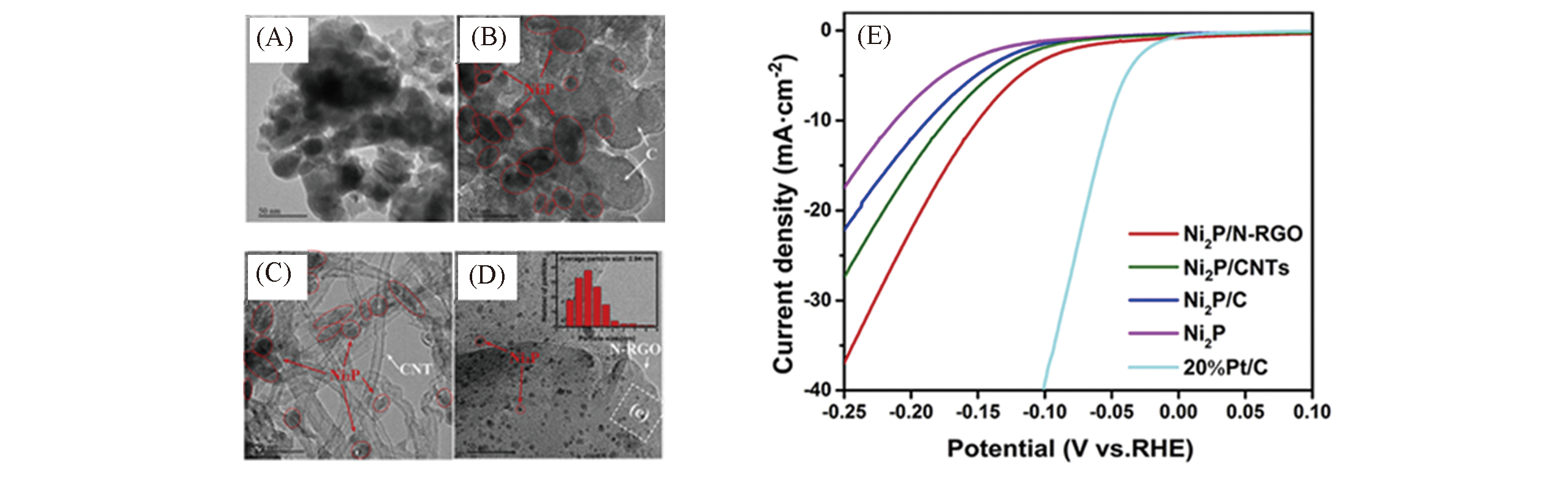
Fig.14 TEM images of Ni2P(A), Ni2P/C(B), Ni2P/CNTs(C) and Ni2P/N?RGO(D), LSV curves of Ni2P, Ni2P/C, Ni2P/CNTs and Ni2P/N?RGO(E)[94]Copyright 2019, Elsevier.
| 1 | Wang J., Xu F., Jin H., Chen Y., Wang Y., Adv. Mater., 2017, 29(14), 1605838 |
| 2 | Lewis N. S., Nocera D. G., Proc. Natl. Acad. Sci. USA, 2006, 103(43), 15729—15735 |
| 3 | Song C. S., Catal. Today, 2006, 115(1—4), 2—32 |
| 4 | Dinca M., Surendranath Y., Nocera D. G., Proc. Natl. Acad. Sci. USA, 2010, 107(23), 10337—10341 |
| 5 | Conte M., Di Mario F., Iacobazzi A., Mattucci A., Moreno A., Ronchetti M., Energies, 2009, 2(1), 150—179 |
| 6 | Wang M. Y., Wang Z., Gong X. Z., Guo Z. C., Renew. Sust. Energ. Rev., 2014, 29, 573—588 |
| 7 | Lu F., Zhou M., Zhou Y., Zeng X., Small, 2017, 13(45), 1701931 |
| 8 | Gong M., Li Y., Wang H., Liang Y., Wu J. Z., Zhou J., Wang J., Regier T., Wei F., Dai H., J. Am. Chem. Soc., 2013, 135(23), 8452—8455 |
| 9 | Li Y., Wang H., Xie L., Liang Y., Hong G., Dai H., J. Am. Chem. Soc., 2011, 133(19), 7296—7299 |
| 10 | Sabatier P., Ber. Dtsch. Chem. Ges., 1911, 44(3), 1984—2001 |
| 11 | Jaramillo T. F., Jorgensen K. P., Bonde J., Nielsen J. H., Horch S., Chorkendorff I., Science, 2007, 317(5834), 100—102 |
| 12 | Greeley J., Jaramillo T. F., Bonde J., Chorkendorff I. B., Norskov J. K., Nat. Mater., 2006, 5(11), 909—913 |
| 13 | Parsons R., Trans. Faraday Soc., 1958, 54, 1053—1063 |
| 14 | Skúlason E., Tripkovic V., Björketun M. E., Gudmundsdóttir S., Karlberg G., Rossmeisl J., Bligaard T., Jónsson H., Nørskov J. K., J. Phys. Chem. C, 2010, 114(42), 18182—18197 |
| 15 | Kondori A., Esmaeilirad M., Baskin A., Song B., Wei J., Chen W., Segre C. U., Shahbazian‐Yassar R., Prendergast D., Asadi M., Adv. Energy Mater., 2019, 9(22), 1900516 |
| 16 | Zang Y., Niu S., Wu Y., Zheng X., Cai J., Ye J., Xie Y., Liu Y., Zhou J., Zhu J., Liu X., Wang G., Qian Y., Nat. Commun., 2019, 10(1), 1217 |
| 17 | Zhu K. L., Li C. Y., Jing Z. H., Liu X. C., He Y. C., Lv X. X., Wang Y., Liu K., FlatChem, 2019, 18, 100140 |
| 18 | Voiry D., Yamaguchi H., Li J., Silva R., Alves D. C., Fujita T., Chen M., Asefa T., Shenoy V. B., Eda G., Chhowalla M., Nat. Mater., 2013, 12(9), 850—855 |
| 19 | Ye G., Gong Y., Lin J., Li B., He Y., Pantelides S. T., Zhou W., Vajtai R., Ajayan P. M., Nano Lett., 2016, 16(2), 1097—1103 |
| 20 | Wang Y., Li J., Wei Z. D., J. Mater. Chem. A, 2018, 6(18), 8194—8209 |
| 21 | Zhong Y., Xia X., Shi F., Zhan J., Tu J., Fan H. J., Adv. Sci., 2016, 3(5), 1500286 |
| 22 | Balogun M. S., Huang Y. C., Qiu W. T., Yang H., Ji H. B., Tong Y. X., Mater. Today, 2017, 20(8), 425—451 |
| 23 | Wang Y., Kong B., Zhao D. Y., Wang H. T., Selomulya C., Nano Today, 2017, 15, 26—55 |
| 24 | Shi Y., Zhang B., Chem. Soc. Rev., 2016, 45(6), 1529—1541 |
| 25 | Paseka I., Electrochim. Acta, 1995, 40(11), 1633—1640 |
| 26 | Paseka I., Velicka J., Electrochim. Acta, 1997, 42(2), 237—242 |
| 27 | Burchardt T., Int. J. Hydrog. Energy, 2000, 25(7), 627—634 |
| 28 | Liu P., Rodriguez J. A., J. Am. Chem. Soc., 2005, 127(42), 14871—14878 |
| 29 | Popczun E. J., Mckone J. R., Read C. G., Biacchi A. J., Wiltrout A. M., Lewis N. S., Schaak R. E., J. Am. Chem. Soc., 2013, 135(25), 9267—9270 |
| 30 | Popczun E. J., Roske C. W., Read C. G., Crompton J. C., Mcenaney J. M., Callejas J. F., Lewis N. S., Schaak R. E., J. Mater. Chem. A, 2015, 3(10), 5420—5425 |
| 31 | Wang T. Y., Du K. Z., Liu W. L., Zhu Z. W., Shao Y. H., Li M. X., J. Mater. Chem. A, 2015, 3(8), 4368—4373 |
| 32 | Tian J., Liu Q., Liang Y., Xing Z., Asiri A. M., Sun X., ACS Appl. Mater. Interfaces, 2014, 6(23), 20579—20584 |
| 33 | Heine V., Phys. Rev., 1967, 153(3), 673—682 |
| 34 | Kim W. S., Cho D. K., Lim H. W., Kim K. H., Hong S. H., Kim J. Y., Electrochim. Acta, 2020, 344, 136116 |
| 35 | Oyama S. T., J. Catal., 2003, 216(1/2), 343—352 |
| 36 | Yu P., Wang F. M., Shifa T. A., Zhan X. Y., Lou X. D., Xia F., He J., Nano Energy, 2019, 58, 244—276 |
| 37 | Jin X., Tian R., Wu A., Xiao Y., Dong X., Hu F., Huang H., Dalton Trans., 2020, 49(7), 2225—2233 |
| 38 | Guo L., Zhao Y., Yao Z., Dalton Trans., 2016, 45(3), 1225—1232 |
| 39 | Stein B. F., Walmsley R. H., Phys. Rev., 1966, 148(2), 933—939 |
| 40 | Liu Q., Tian J., Cui W., Jiang P., Cheng N., Asiri A. M., Sun X., Angew. Chem. Int. Ed., 2014, 53(26), 6710—6714 |
| 41 | Tian J., Liu Q., Asiri A. M., Sun X., J. Am. Chem. Soc., 2014, 136(21), 7587—7590 |
| 42 | Tang C., Zhang R., Lu W., He L., Jiang X., Asiri A. M., Sun X., Adv. Mater., 2017, 29(2), 1602441 |
| 43 | Liu X. H., Wei B., Su R., Zhao C. G., Dai D. M., Ma X., Xu L. L., Energy Technol., 2019, 7(6), 1900021 |
| 44 | Wang J. W., Johnston⁃Peck A. C., Tracy J. B., Chem. Mat., 2009, 21(19), 4462—4467 |
| 45 | Schipper D. E., Zhao Z. H., Thirumalai H., Leitner A. P., Donaldson S. L., Kumar A., Qin F., Wang Z. M., Grabow L. C., Bao J. M., Whitmire K. H., Chem. Mat., 2018, 30(10), 3588—3598 |
| 46 | Yu Y. D., Song Z. L., Ge H. L., Wei G. Y., Prog. Nat. Sci., 2014, 24(3), 232—238 |
| 47 | Kurowski A., Schultze J. W., Staikov G., Electrochem. Commun., 2002, 4(7), 565—569 |
| 48 | Jiang N., You B., Sheng M., Sun Y., Angew. Chem., Int. Ed., 2015, 54(21), 6251—6254 |
| 49 | Mabayoje O., Dunning S. G., Kawashima K., Wygant B. R., Ciufo R. A., Humphrey S. M., Mullins C. B., ACS Appl. Energy Mater., 2019, 3(1), 176—183 |
| 50 | Liu T., Li P., Yao N., Cheng G., Chen S., Luo W., Yin Y., Angew. Chem. Int. Ed., 2019, 58(14), 4679—4684 |
| 51 | Bu F. X., Chen W. S., Aboud M. F. A., Shakir I., Gu J. J., Xu Y. X., J. Mater. Chem. A, 2019, 7(24), 14526—14535 |
| 52 | Zhang C., Xin B., Xi Z., Zhang B., Li Z., Zhang H., Li Z., Hao J., ACS Sustain. Chem. Eng., 2017, 6(1), 1468—1477 |
| 53 | Zhang T. Q., Liu J., Huang L. B., Zhang X. D., Sun Y. G., Liu X. C., Bin D. S., Chen X., Cao A. M., Hu J. S., Wan L. J., J. Am. Chem. Soc., 2017, 139(32), 11248—11253 |
| 54 | Zhang G., Wang G., Liu Y., Liu H., Qu J., Li J., J. Am. Chem. Soc., 2016, 138(44), 14686—14693 |
| 55 | Huang Z., Chen Z., Chen Z., Lv C., Meng H., Zhang C., ACS Nano, 2014, 8(8), 8121—8129 |
| 56 | Zhou M., Kang Y. Y., Huang K. K., Shi Z., Xie R. G., Yang W. S., RSC Adv., 2016, 6(78), 74895—74902 |
| 57 | Shi Y., Xu Y., Zhuo S., Zhang J., Zhang B., ACS Appl. Mater. Interfaces, 2015, 7(4), 2376—2384 |
| 58 | Jin Z. Y., Li P. P., Xiao D., Green Chem., 2016, 18(6), 1459—1464 |
| 59 | Callejas J. F., Read C. G., Popczun E. J., Mcenaney J. M., Schaak R. E., Chem. Mat., 2015, 27(10), 3769—3774 |
| 60 | Wang X., Kolen'ko Y. V., Liu L., Chem. Commun., 2015, 51(31), 6738—6741 |
| 61 | Mcenaney J. M., Crompton J. C., Callejas J. F., Popczun E. J., Biacchi A. J., Lewis N. S., Schaak R. E., Chem. Mat., 2014, 26(16), 4826—4831 |
| 62 | Huang Z. P., Chen Z. Z., Chen Z. B., Lv C. C., Humphrey M. G., Zhang C., Nano Energy, 2014, 9, 373—382 |
| 63 | Tang C., Asiri A. M., Luo Y. L., Sun X. P., ChemNanoMat, 2015, 1(8), 558—561 |
| 64 | Saadi F. H., Carim A. I., Verlage E., Hemminger J. C., Lewis N. S., Soriaga M. P., J. Phys. Chem. C, 2014, 118(50), 29294—29300 |
| 65 | Han G. Q., Li X., Liu Y. R., Dong B., Hu W. H., Shang X., Zhao X., Chai Y. M., Liu Y. Q., Liu C. G., RSC Adv., 2016, 6(58), 52761—52771 |
| 66 | Zhang Z. Y., Liu S. S., Xiao J., Wang S., J. Mater. Chem. A, 2016, 4(24), 9691—9699 |
| 67 | Zhu W. X., Tang C., Liu D. N., Wang J. L., Asiri A. M., Sun X. P., J. Mater. Chem. A, 2016, 4(19), 7169—7173 |
| 68 | Pu Z., Saana Amiinu I., Wang M., Yang Y., Mu S., Nanoscale, 2016, 8(16), 8500—8504 |
| 69 | Wu T. L., Pi M. Y., Zhang D. K., Chen S. J., J. Power Sources, 2016, 328, 551—557 |
| 70 | Chang J. F., Liang L., Li C. Y., Wang M. L., Ge J. J., Liu C. P., Xing W., Green Chem., 2016, 18(8), 2287—2295 |
| 71 | Niu Z. G., Jiang J., Ai L. H., Electrochem. Commun., 2015, 56, 56—60 |
| 72 | Pu Z. H., Liu Q., Jiang P., Asiri A. M., Obaid A. Y., Sun X. P., Chem. Mat., 2014, 26(15), 4326—4329 |
| 73 | Wang P., Song F., Amal R., Ng Y. H., Hu X., ChemSusChem, 2016, 9(5), 472—477 |
| 74 | Jiang P., Liu Q., Liang Y., Tian J., Asiri A. M., Sun X., Angew. Chem. Int. Ed., 2014, 53(47), 12855—12859 |
| 75 | Liang Y. H., Liu Q., Asiri A. M., Sun X. P., Luo Y. L., ACS Catal., 2014, 4(11), 4065—4069 |
| 76 | Kibsgaard J., Tsai C., Chan K., Benck J. D., Norskov J. K., Abild⁃Pedersen F., Jaramillo T. F., Energy Environ. Sci., 2015, 8(10), 3022—3029 |
| 77 | Xiao P., Sk M. A., Thia L., Ge X. M., Lim R. J., Wang J. Y., Lim K. H., Wang X., Energy Environ. Sci., 2014, 7(8), 2624—2629 |
| 78 | Pan Y., Liu Y. R., Zhao J. C., Yang K., Liang J. L., Liu D. D., Hu W. H., Liu D. P., Liu Y. Q., Liu C. G., J. Mater. Chem. A, 2015, 3(4), 1656—1665 |
| 79 | Laursen A. B., Patraju K. R., Whitaker M. J., Retuerto M., Sarkar T., Yao N., Ramanujachary K. V., Greenblatt M., Dismukes G. C., Energy Environ. Sci., 2015, 8(3), 1027—1034 |
| 80 | Wang B., Tang C., Wang H. F., Chen X., Cao R., Zhang Q., Adv. Mater., 2019, 31(4), e1805658 |
| 81 | Xiao P., Chen W., Wang X., Adv. Energy Mater., 2015, 5(24), 1500985 |
| 82 | Zheng Y., Jiao Y., Jaroniec M., Qiao S. Z., Angew. Chem. Int. Ed., 2015, 54(1), 52—65 |
| 83 | Kibsgaard J., Jaramillo T. F., Angew. Chem. Int. Ed., 2014, 53(52), 14433—14437 |
| 84 | Anjum M. A. R., Okyay M. S., Kim M., Lee M. H., Park N., Lee J. S., Nano Energy, 2018, 53, 286—295 |
| 85 | Liu T., Ma X., Liu D., Hao S., Du G., Ma Y., Asiri A. M., Sun X., Chen L., ACS Catal., 2016, 7(1), 98—102 |
| 86 | Liu T. T., Liu D. N., Qu F. L., Wang D. X., Zhang L., Ge R. X., Hao S., Ma Y. J., Du G., Asiri A. M., Chen L., Sun X. P., Adv. Energy Mater., 2017, 7(15), 1700020 |
| 87 | Chen P., Tong Y., Wu C., Xie Y., Acc. Chem. Res., 2018, 51(11), 2857—2866 |
| 88 | Feng J. X., Tong S. Y., Tong Y. X., Li G. R., J. Am. Chem. Soc., 2018, 140(15), 5118—5126 |
| 89 | Liu T., Li A., Wang C., Zhou W., Liu S., Guo L., Adv. Mater., 2018, 30(46), e1803590 |
| 90 | Yu F., Zhou H., Huang Y., Sun J., Qin F., Bao J., Goddard Ⅲ W. A., Chen S., Ren Z., Nat. Commun., 2018, 9(1), 2551 |
| 91 | Sun H. M., Xu X. B., Yan Z. H., Chen X., Cheng F. Y., Weiss P. S., Chen J., Chem. Mat., 2017, 29(19), 8539—8547 |
| 92 | Mat M. D., Aldas K., Ilegbusi O. J., Int. J. Hydrog. Energy, 2004, 29(10), 1015—1023 |
| 93 | Zhou M., Sun Q. Q., Shen Y. Q., Ma Y., Wang Z. L., Zhao C., Electrochim. Acta, 2019, 306, 651—659 |
| 94 | Ma D., Meng K., Ma J. Z., Jia Z. J., Wang Y., Liu L. J., Zhu G. M., Qi T., Int. J. Hydrog. Energy, 2019, 44(60), 31960—31968 |
| [1] | 秦永吉, 罗俊. 单原子催化剂在CO2转化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220300. |
| [2] | 姚青, 俞志勇, 黄小青. 单原子催化剂的合成及其能源电催化应用的研究进展[J]. 高等学校化学学报, 2022, 43(9): 20220323. |
| [3] | 范建玲, 唐灏, 秦凤娟, 许文静, 谷鸿飞, 裴加景, 陈文星. 氮掺杂超薄碳纳米片复合铂钌单原子合金催化剂的电化学析氢性能[J]. 高等学校化学学报, 2022, 43(9): 20220366. |
| [4] | 唐全骏, 刘颖馨, 孟蓉炜, 张若天, 凌国维, 张辰. 单原子催化在海洋能源领域的应用[J]. 高等学校化学学报, 2022, 43(9): 20220324. |
| [5] | 林高鑫, 王家成. 单原子掺杂二硫化钼析氢催化的进展和展望[J]. 高等学校化学学报, 2022, 43(9): 20220321. |
| [6] | 汪思聪, 庞贝贝, 刘潇康, 丁韬, 姚涛. XAFS技术在单原子电催化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220487. |
| [7] | 韩付超, 李福进, 陈良, 贺磊义, 姜玉南, 徐守冬, 张鼎, 其鲁. CoSe2/C复合电催化材料修饰隔膜对高载量锂硫电池性能的影响[J]. 高等学校化学学报, 2022, 43(8): 20220163. |
| [8] | 赵润瑶, 纪桂鹏, 刘志敏. 吡咯氮配位单原子铜催化剂的电催化二氧化碳还原性能[J]. 高等学校化学学报, 2022, 43(7): 20220272. |
| [9] | 王茹涵, 贾顺涵, 吴丽敏, 孙晓甫, 韩布兴. CO2参与电化学构筑C—N键制备重要化学品[J]. 高等学校化学学报, 2022, 43(7): 20220395. |
| [10] | 彭奎霖, 李桂林, 江重阳, 曾少娟, 张香平. 电解液调控CO2电催化还原性能微观机制的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220238. |
| [11] | 王丽君, 李欣, 洪崧, 詹新雨, 王迪, 郝磊端, 孙振宇. 调节氧化镉-炭黑界面高效电催化CO2还原生成CO[J]. 高等学校化学学报, 2022, 43(7): 20220317. |
| [12] | 龚妍熹, 王建兵, 柴歩瑜, 韩元春, 马云飞, 贾超敏. 钾掺杂g-C3N4薄膜光阳极的制备及光电催化氧化降解水中双氯芬酸钠性能[J]. 高等学校化学学报, 2022, 43(6): 20220005. |
| [13] | 杨丽君, 于洋, 张蕾. 双功能2D/3D杂化结构Co2P-CeO x 异质结一体化电极的构筑及电催化尿素氧化辅助制氢性能[J]. 高等学校化学学报, 2022, 43(6): 20220082. |
| [14] | 夏天, 万家炜, 于然波. 异原子配位结构碳基单原子电催化剂结构与性能相关性的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220162. |
| [15] | 黄汉浩, 卢湫阳, 孙明子, 黄勃龙. 石墨炔原子催化剂的崭新道路:基于自验证机器学习方法的筛选策略[J]. 高等学校化学学报, 2022, 43(5): 20220042. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||