

高等学校化学学报 ›› 2014, Vol. 35 ›› Issue (4): 665.doi: 10.7503/cjcu20131066
收稿日期:2013-11-04
出版日期:2014-04-10
发布日期:2013-12-30
作者简介:联系人简介: 何圣贵, 男, 博士, 研究员, 主要从事团簇物理化学研究. E-mail: 基金资助:
LIU Qingyu1,2, HE Shenggui1,*( )
)
Received:2013-11-04
Online:2014-04-10
Published:2013-12-30
Contact:
HE Shenggui
E-mail:shengguihe@iccas.ac.cn
Supported by:摘要:
研究气态条件下原子团簇在完全可控、 可重复条件下与CO的反应, 可以在分子水平上理解CO的氧化, 本综述对这些团簇研究工作进行了一些分析和总结, 讨论了团簇研究方法的特点、 CO氧化的新见解以及有待解决的问题.
中图分类号:
TrendMD:
刘清宇, 何圣贵. 原子团簇上一氧化碳的氧化. 高等学校化学学报, 2014, 35(4): 665.
LIU Qingyu, HE Shenggui. Oxidation of Carbon Monoxide on Atomic Clusters†. Chem. J. Chinese Universities, 2014, 35(4): 665.
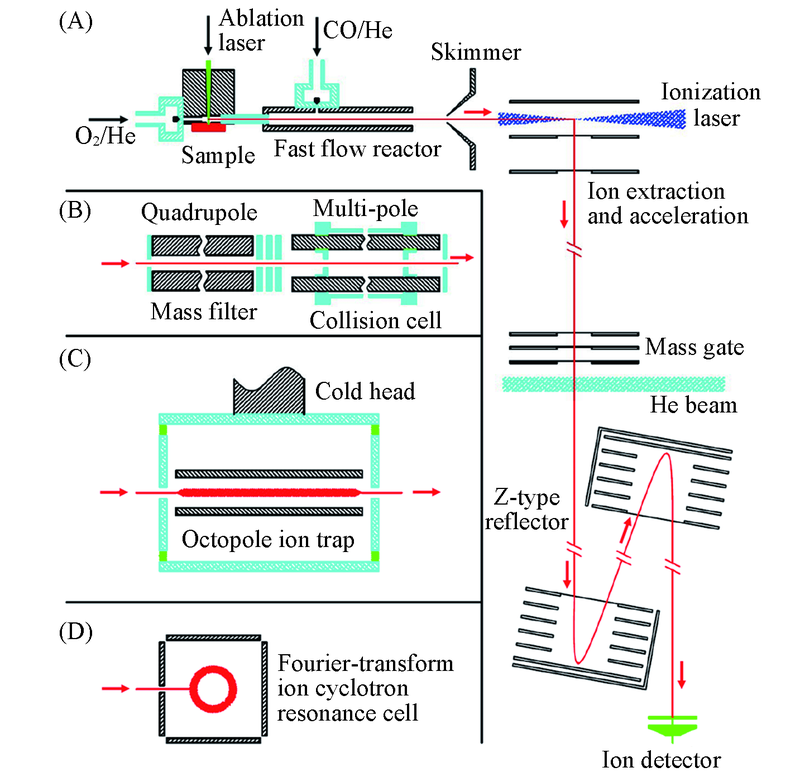
Fig.1 Experimental setups used for studying the reactions between CO and atomic clustersPart (A) shows that a fast flow reactor is coupled with a laser ablation cluster source and a time-of-flight mass spectrometer in which a laser ionizes the neutral clusters, a mass gate selects cluster ions of interest, and a helium beam collides with the selected ions(adapted from refs.[33,42,45]). The reactor in (A) can be replaced by a quadrupole mass filter and a multi-pole(hexapole or octopole) collision cell(B, adapted from refs.[32,46]). The collision cell in (B) can be further replaced by an octopole ion trap(C, adapted from ref.[47]) and a Fourier-transform ion cyclotron resonance cell(D, adapted from refs.[49]). For (B) and (C), the ions coming out of the cell or trap are detected by a quadrupole[46] or a time-of-flight mass spectrometer[33].
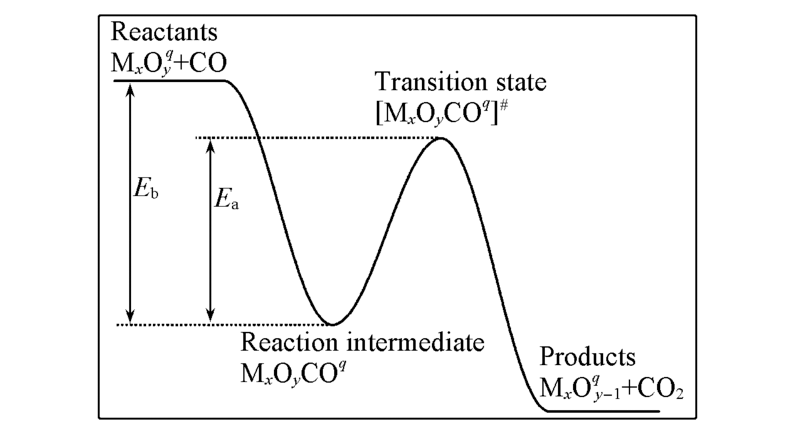
Fig.2 Typical potential energy profile for CO oxidation on the MxOyq clusterThe reaction barrier and the binding energy between CO and MxOyq are indicated by variables Ea and Eb, respectively.
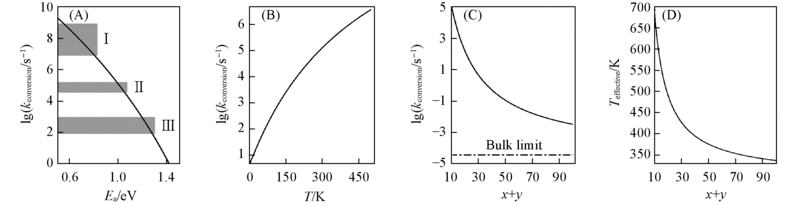
Fig.3 Calculated rates(A—C) and effective temperatures(D) for conversions of MxOyCOq into MxOy-1q+CO2(see Fig.2) The conversion rates with respect to the reaction barrier, the cluster vibrational temperature, and the number of atoms in the cluster are given in (A), (B) and (C), respectively. Eq.(2) in the text is used and the ν, Eb, and Ek are fixed at 3×1012 s-1, 1.5 eV, and 0.14 eV(a center-of-mass velocity of 1 km/s and a reduced mass of 28 amu), respectively. Other parameters fixed: T=298 K and x+y=10 for (A); Ea=1.0 eV and x+y=10 for (B); and Ea=1.0 eV and T=298 K for (C), in which the value denoted as bulk limit is calculated with Eq.(3) in the text. In (A), the ranges labeled as Ⅰ, Ⅱ, and Ⅲ represent the rates of energy dissipation(collision or infrared radiation) in the fast flow reactor[Fig.1(A)], in the collision cell and ion trap[Fig.1(B) and 1(C)], and in the FT-ICR cell[Fig.1(D)]. (D) shows the effective temperate(Teffective) at which the conversion rate of bulk limit(Eq.(3) with T=Teffective) equals to that of the cluster system in (C).
| Species | Cluster | Product | k1 | Reactor | Remark | Year | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Main | Al2 | Al2 | [1.4×10-11] | B | 2008[ | |||||||||||
| group | Al2 | Al2 | [ca.10-14] | B | 2008[ | |||||||||||
| Al2 | Al2 | [4.6×10-13] | B | 2008[ | ||||||||||||
| CaO+ | Ca+ | 2.5×10-10 | A | 2005[ | ||||||||||||
| CaO+ | Ca+ | (2.8±1.5)×10-10 | A | 2008[ | ||||||||||||
| GeO+ | Ge+ | 2.1×10-10 | A | 2005[ | ||||||||||||
| SrO+ | Sr+ | 1.0×10-10 | A | 2005[ | ||||||||||||
| BaO+ | Ba+ | 2.3×10-11 | A | 2005[ | ||||||||||||
| d-Block | Sc2 | Sc2 | 2.0×10-10 | A | Theory | 2012[ | ||||||||||
| Ti2 | Ti2 | [4.5×10-12] | B | Theory | 2011[ | |||||||||||
| (TiO2)nO- | (TiO2 | ca.10-11 | A | n=3—25, theory(n=3—8) | 2013[ | |||||||||||
| V4 | V4 | 4.4×10-11 | B | Theory | 2013[ | |||||||||||
| VO3 | VO2 | A | Theory | 2013[ | ||||||||||||
| Mn2O4 | Mn2O3 | 6.5×10-13 | A | Theory | 2013[ | |||||||||||
| Mn3O7 | Mn3O6 | 1.9×10-13 | A | Theory | 2013[ | |||||||||||
| FeO+ | Fe+ | 9×10-10 | D | 1981[ | ||||||||||||
| FeO+ | Fe+ | 1.8×10-10 | A | Theory | 2005[ | |||||||||||
| FeO(N2O | Fe+ | ca.10-10 | A | n=0—3 | 1995[ | |||||||||||
| Fe | FeO+, FeCO+ | [5.3×10-12] | B | Theory | 2007[ | |||||||||||
| Fe | FeO+, FeOCO+, Fe+ | B | 2007[ | |||||||||||||
| Fe | Fe | [1.4×10-12] | B | 2007[ | ||||||||||||
| Fe | FeO2CO+, Fe(CO | [5.5×10-12] | B | Loss of molecular O2, theory | 2007[ | |||||||||||
| Fe | Fe | B | 2007[ | |||||||||||||
| Fe2O+ | F | B | Theory | 2007[ | ||||||||||||
| Fe2 | Fe2O+, F | [8.8×10-13] | B | Theory | 2007[ | |||||||||||
| Fe2 | Fe2 | [ca.10-13] | B | 2007[ | ||||||||||||
| Fe2 | Fe2O2CO+, Fe2 | B | Theory | 2007[ | ||||||||||||
| Fe2 | Fe2 | B | Theory | 2007[ | ||||||||||||
| Fe2 | ||||||||||||||||
| FeO2 | FeO | 5.3×10-12 | A | Theory | 2008[ | |||||||||||
| FeO3 | FeO2 | 1.3×10-12 | A | Theory | 2008[ | |||||||||||
| Fe | FeO- | B | 2007[ | |||||||||||||
| Fe | FeO- | 1.9×10-12 | B | Theory | 2010[ | |||||||||||
| Fe | Fe | [ca.10-14] | B | 2007[ | ||||||||||||
| Fe | Fe | B | 2007[ | |||||||||||||
| Fe | Fe | [2.9×10-13] | B | Loss of molecular O2 and | 2007[ | |||||||||||
| CO oxidation | ||||||||||||||||
| Species | Cluster | Product | k1 | Reactor | Remark | Year | ||||||||||
| d-Block | Fe2 | Fe2 | B | 2007[ | ||||||||||||
| Fe2 | Fe2 | 1.8×10-12 | B | 2010[ | ||||||||||||
| Fe2 | Fe2 | B | 2007[ | |||||||||||||
| Fe2 | Fe2 | B | 2007[ | |||||||||||||
| Fe2 | Fe2 | [4.5×10-13] | B | Loss of molecular O2 and | 2007[ | |||||||||||
| CO oxidation | ||||||||||||||||
| CoO+ | Co+ | B | 2008[ | |||||||||||||
| Co | Co+, CoCO+ | [9.3×10-12] | B | Loss of molecular O2,theory | 2008[ | |||||||||||
| Co | CoO+, CoOCO+, Co+ | [7.2×10-12] | B | Loss of molecular O2 | 2008[ | |||||||||||
| Co | Co | B | Loss of molecular O2 | 2008[ | ||||||||||||
| Co2 | CoO2CO+ , Co(CO | [1.3×10-12] | B | Loss of molecular O2, | 2008[ | |||||||||||
| cluster fragmentation | ||||||||||||||||
| Co2 | Co2 | [4.7×10-12] | B | Loss of molecular O2 | 2008[ | |||||||||||
| CoO2CO+, Co(CO | ||||||||||||||||
| Co2 | Co2 | B | Loss of molecular O2 | 2008[ | ||||||||||||
| ConOm | Con | 10-13—10-12 | A | n=3—9, m=3—13, | 2010[ | |||||||||||
| theory(Co3O4) | ||||||||||||||||
| Co | CoO- | B | 2008[ | |||||||||||||
| Co | CoO- | 0.3×10-12 | B | 2010[ | ||||||||||||
| Co | Co | B | 2008[ | |||||||||||||
| Co2 | Co2 | B | 2008[ | |||||||||||||
| Co2 | Co2 | 1.6×10-12 | B | 2010[ | ||||||||||||
| Co2 | Co2 | [4.8×10-14] | B | 2008[ | ||||||||||||
| Co3 | Co3 | [1.4×10-13] | B | 2008[ | ||||||||||||
| Co3 | Co3 | [1.0×10-13] | B | 2008[ | ||||||||||||
| Co2 | ||||||||||||||||
| Ni6 | Ni6 | 2.2×10-13 | A | 2013[ | ||||||||||||
| Ni8 | Ni8 | 1.2×10-13 | A | 2013[ | ||||||||||||
| Ni | NiO- | (9.2±0.5)×10-14 | B | 2009[ | ||||||||||||
| Ni | NiO- | 0.4×10-12 | B | 2010[ | ||||||||||||
| Ni | Ni | B | 2009[ | |||||||||||||
| Ni2 | Ni2 | (1.9±0.1)×10-13 | B | 2009[ | ||||||||||||
| Ni2 | Ni2 | 1.0×10-12 | B | 2010[ | ||||||||||||
| Ni3 | Ni3 | (9.6±0.8)×10-14 | B | 2009[ | ||||||||||||
| Ni4 | Ni4 | (3.0±0.2)×10-13 | B | 2009[ | ||||||||||||
| Cu | CuO- | 0.7×10-12 | B | 2010[ | ||||||||||||
| Cu2 | Cu2 | 1.5×10-12 | B | 2010[ | ||||||||||||
| Cu5 | Cu5O- | B | 2013[ | |||||||||||||
| Cu9 | Cu9O- | B | 2013[ | |||||||||||||
| Zr2 | Zr2 | 1.8×10-10 | A | 2010[ | ||||||||||||
| (ZrO2 | Zrn | 2.4×10-12, 1.6×10-12, | B | n=1—5, k1 for | 2008[ | |||||||||||
| 1.2×10-12, 6.3×10-13 | n=2—5,theory | |||||||||||||||
| (ZrO2)nO- | (ZrO2 | 2.1×10-12, 2.2×10-13, | B | n=1—4, theory | 2009[ | |||||||||||
| 3.4×10-13, 1.8×10-13 | ||||||||||||||||
| (ZrO2)nO- | (ZrO2)nC | ca.10-11 | A | n=3—25, Oxidative adsorption | 2013[ | |||||||||||
| Mo | Mo(CO | A | n=1—3 | 2006[ | ||||||||||||
| C6 | 2007[ | |||||||||||||||
| RhnOm | Rhn | ca.10-10 | A | n=10—28, m=1—5, | 2012[ | |||||||||||
| k1 for m=4, 5 | ||||||||||||||||
| Pd | PdO+, OPdCO+, PdCO+ | B | Loss of molecular O2 and | 2011[ | ||||||||||||
| CO oxidation, theory | ||||||||||||||||
| Pd | Pd | B | Loss of molecular O2 and | 2011[ | ||||||||||||
| OPdCO+ | CO oxidation, theory | |||||||||||||||
| PdnO+ | Pdn(CO | C | n=2—7 | 2012[ | ||||||||||||
| Pdn | Pdn(CO | C | n=4—6 | 2012[ | ||||||||||||
| Species | Cluster | Product | k1 | Reactor | Remark | Year | ||||||||||
| d-Block | Pd6 | Pd6 | C | Theory | 2012[ | |||||||||||
| Pd6 | Pd6O5CO+ | C | Eliminate CO2 and form | 2012[ | ||||||||||||
| Pd6 | ||||||||||||||||
| Ag3 | Ag3On(OCO | C | n=1—3, with N2O | 2011[ | ||||||||||||
| Agn | A | C | n=7, 9, 11 | 2004[ | ||||||||||||
| Ag5 | Ag4 | C | Cluster fragmentation | 2005[ | ||||||||||||
| Ag3C | ||||||||||||||||
| AgnNx | Ag3NO-, AgN2 | C | n=2—5, y>x, cluster | 2007[ | ||||||||||||
| fragmentation | ||||||||||||||||
| La2 | La2O3+e- | 1.9×10-10 | A | Electron detachment, theory | 2012[ | |||||||||||
| Wn | Wn | 3.0×10-12, 3.6×10-13, | B | n=1—3 | 2008[ | |||||||||||
| 5.9×10-13 | ||||||||||||||||
| Re | Re | 3.3×10-10 | D | 2001[ | ||||||||||||
| Re | Re | 2.4×10-10 | D | 2001[ | ||||||||||||
| Os | Os | ≥4.0×10-13 | A | n=1—4, k1 for n=1 | 2005[ | |||||||||||
| Ir | Ir | 3.5×10-10 | A | n=1—3, k1 for n=1 | 2005[ | |||||||||||
| Pt | PtO+, Pt+ | 6.6×10-10 | D | 2001[ | ||||||||||||
| Pt | Pt | 2.3×10-10 | A | n=1—3, k1 for n=1 | 2005[ | |||||||||||
| Pt7 | Pt7 | ca.10-9 | D | n=1—2 | 2004[ | |||||||||||
| Pt4 | Pt4 | 10-11—10-9 | D | n=1—3, 4(slow) | 2007[ | |||||||||||
| Ptn | Ptn | B | n=3—6, m=1—2 | 1998[ | ||||||||||||
| AuO+ | AuCO+ | B | Theory | 2008[ | ||||||||||||
| AuO+ | AuCO+ | 7.93×10-12 | B | 2008[ | ||||||||||||
| Au2O+ | Au2CO+ | B | Theory | 2008[ | ||||||||||||
| Au2O+ | Au2CO+ | 2.42×10-12 | B | 2008[ | ||||||||||||
| Au3O+ | Au3CO+ | 4.98×10-12 | B | 2008[ | ||||||||||||
| Au3 | Au3(CO2 | C | 2011[ | |||||||||||||
| Aun | AunOx(CO | A | n=1—2, m=1—5, | 2004[ | ||||||||||||
| x=0—3, y=0—2 | ||||||||||||||||
| AuO- | Au- | ca.10-11 | A | Theory | 2004[ | |||||||||||
| Au | AuO- | A | Very slow, theory | 2004[ | ||||||||||||
| Au | Au- | ca.10-12 | A | Slow, loss of molecular O2, | 2004[ | |||||||||||
| theory | ||||||||||||||||
| Au2O- | A | ca.10-13 | A | Theory | 2006[ | |||||||||||
| Au2 | A | B | 2003[ | |||||||||||||
| Au2 | A | C | 2005[ | |||||||||||||
| Au2 | A | A | Theory | 2006[ | ||||||||||||
| Au2 | Au2 | ca.10-13 | A | Theory | 2006[ | |||||||||||
| Au2 | Au2 | ca.10-13 | A | Theory | 2006[ | |||||||||||
| Au6 | Au6O-, A | A | CO and O2 co-adsorption | 2002[ | ||||||||||||
| Aun | AuxOy(CO | A | n ≥ 4, Oxidation, replacement | 2006[ | ||||||||||||
| and association | ||||||||||||||||
| f-Block | Cen | Cen | 1.9×10-12 | A | n=2—6, k1 for Ce2 | 2010[ | ||||||||||
| theory(n =2—5) | ||||||||||||||||
| Cen | Cen | 8×10-11 | A | n=4—21, k1 for Ce4 | 2011[ | |||||||||||
| theory(n =1—6) | ||||||||||||||||
| EuO+ | Eu+ | ≥3.3×10-12 | A | 2005[ | ||||||||||||
| YbO+ | Yb+ | 1.2×10-11 | A | 2005[ | ||||||||||||
| Hetero- | AlV | AlV | 4.1×10-10 | D | Theory | 2011[ | ||||||||||
| nuclear | VCoO4 | VCoO3 | ca.10-11 | A | Theory | 2012[ | ||||||||||
| YAl | YAl | D | Theory | 2013[ | ||||||||||||
| AuTi3 | AuTi3O6CO+ | A | Molecular association | 2011[ | ||||||||||||
Table 1 Experimentally studied reactions between the oxide clusters MxOyq(q=0, ±1) and CO*
| Species | Cluster | Product | k1 | Reactor | Remark | Year | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Main | Al2 | Al2 | [1.4×10-11] | B | 2008[ | |||||||||||
| group | Al2 | Al2 | [ca.10-14] | B | 2008[ | |||||||||||
| Al2 | Al2 | [4.6×10-13] | B | 2008[ | ||||||||||||
| CaO+ | Ca+ | 2.5×10-10 | A | 2005[ | ||||||||||||
| CaO+ | Ca+ | (2.8±1.5)×10-10 | A | 2008[ | ||||||||||||
| GeO+ | Ge+ | 2.1×10-10 | A | 2005[ | ||||||||||||
| SrO+ | Sr+ | 1.0×10-10 | A | 2005[ | ||||||||||||
| BaO+ | Ba+ | 2.3×10-11 | A | 2005[ | ||||||||||||
| d-Block | Sc2 | Sc2 | 2.0×10-10 | A | Theory | 2012[ | ||||||||||
| Ti2 | Ti2 | [4.5×10-12] | B | Theory | 2011[ | |||||||||||
| (TiO2)nO- | (TiO2 | ca.10-11 | A | n=3—25, theory(n=3—8) | 2013[ | |||||||||||
| V4 | V4 | 4.4×10-11 | B | Theory | 2013[ | |||||||||||
| VO3 | VO2 | A | Theory | 2013[ | ||||||||||||
| Mn2O4 | Mn2O3 | 6.5×10-13 | A | Theory | 2013[ | |||||||||||
| Mn3O7 | Mn3O6 | 1.9×10-13 | A | Theory | 2013[ | |||||||||||
| FeO+ | Fe+ | 9×10-10 | D | 1981[ | ||||||||||||
| FeO+ | Fe+ | 1.8×10-10 | A | Theory | 2005[ | |||||||||||
| FeO(N2O | Fe+ | ca.10-10 | A | n=0—3 | 1995[ | |||||||||||
| Fe | FeO+, FeCO+ | [5.3×10-12] | B | Theory | 2007[ | |||||||||||
| Fe | FeO+, FeOCO+, Fe+ | B | 2007[ | |||||||||||||
| Fe | Fe | [1.4×10-12] | B | 2007[ | ||||||||||||
| Fe | FeO2CO+, Fe(CO | [5.5×10-12] | B | Loss of molecular O2, theory | 2007[ | |||||||||||
| Fe | Fe | B | 2007[ | |||||||||||||
| Fe2O+ | F | B | Theory | 2007[ | ||||||||||||
| Fe2 | Fe2O+, F | [8.8×10-13] | B | Theory | 2007[ | |||||||||||
| Fe2 | Fe2 | [ca.10-13] | B | 2007[ | ||||||||||||
| Fe2 | Fe2O2CO+, Fe2 | B | Theory | 2007[ | ||||||||||||
| Fe2 | Fe2 | B | Theory | 2007[ | ||||||||||||
| Fe2 | ||||||||||||||||
| FeO2 | FeO | 5.3×10-12 | A | Theory | 2008[ | |||||||||||
| FeO3 | FeO2 | 1.3×10-12 | A | Theory | 2008[ | |||||||||||
| Fe | FeO- | B | 2007[ | |||||||||||||
| Fe | FeO- | 1.9×10-12 | B | Theory | 2010[ | |||||||||||
| Fe | Fe | [ca.10-14] | B | 2007[ | ||||||||||||
| Fe | Fe | B | 2007[ | |||||||||||||
| Fe | Fe | [2.9×10-13] | B | Loss of molecular O2 and | 2007[ | |||||||||||
| CO oxidation | ||||||||||||||||
| Species | Cluster | Product | k1 | Reactor | Remark | Year | ||||||||||
| d-Block | Fe2 | Fe2 | B | 2007[ | ||||||||||||
| Fe2 | Fe2 | 1.8×10-12 | B | 2010[ | ||||||||||||
| Fe2 | Fe2 | B | 2007[ | |||||||||||||
| Fe2 | Fe2 | B | 2007[ | |||||||||||||
| Fe2 | Fe2 | [4.5×10-13] | B | Loss of molecular O2 and | 2007[ | |||||||||||
| CO oxidation | ||||||||||||||||
| CoO+ | Co+ | B | 2008[ | |||||||||||||
| Co | Co+, CoCO+ | [9.3×10-12] | B | Loss of molecular O2,theory | 2008[ | |||||||||||
| Co | CoO+, CoOCO+, Co+ | [7.2×10-12] | B | Loss of molecular O2 | 2008[ | |||||||||||
| Co | Co | B | Loss of molecular O2 | 2008[ | ||||||||||||
| Co2 | CoO2CO+ , Co(CO | [1.3×10-12] | B | Loss of molecular O2, | 2008[ | |||||||||||
| cluster fragmentation | ||||||||||||||||
| Co2 | Co2 | [4.7×10-12] | B | Loss of molecular O2 | 2008[ | |||||||||||
| CoO2CO+, Co(CO | ||||||||||||||||
| Co2 | Co2 | B | Loss of molecular O2 | 2008[ | ||||||||||||
| ConOm | Con | 10-13—10-12 | A | n=3—9, m=3—13, | 2010[ | |||||||||||
| theory(Co3O4) | ||||||||||||||||
| Co | CoO- | B | 2008[ | |||||||||||||
| Co | CoO- | 0.3×10-12 | B | 2010[ | ||||||||||||
| Co | Co | B | 2008[ | |||||||||||||
| Co2 | Co2 | B | 2008[ | |||||||||||||
| Co2 | Co2 | 1.6×10-12 | B | 2010[ | ||||||||||||
| Co2 | Co2 | [4.8×10-14] | B | 2008[ | ||||||||||||
| Co3 | Co3 | [1.4×10-13] | B | 2008[ | ||||||||||||
| Co3 | Co3 | [1.0×10-13] | B | 2008[ | ||||||||||||
| Co2 | ||||||||||||||||
| Ni6 | Ni6 | 2.2×10-13 | A | 2013[ | ||||||||||||
| Ni8 | Ni8 | 1.2×10-13 | A | 2013[ | ||||||||||||
| Ni | NiO- | (9.2±0.5)×10-14 | B | 2009[ | ||||||||||||
| Ni | NiO- | 0.4×10-12 | B | 2010[ | ||||||||||||
| Ni | Ni | B | 2009[ | |||||||||||||
| Ni2 | Ni2 | (1.9±0.1)×10-13 | B | 2009[ | ||||||||||||
| Ni2 | Ni2 | 1.0×10-12 | B | 2010[ | ||||||||||||
| Ni3 | Ni3 | (9.6±0.8)×10-14 | B | 2009[ | ||||||||||||
| Ni4 | Ni4 | (3.0±0.2)×10-13 | B | 2009[ | ||||||||||||
| Cu | CuO- | 0.7×10-12 | B | 2010[ | ||||||||||||
| Cu2 | Cu2 | 1.5×10-12 | B | 2010[ | ||||||||||||
| Cu5 | Cu5O- | B | 2013[ | |||||||||||||
| Cu9 | Cu9O- | B | 2013[ | |||||||||||||
| Zr2 | Zr2 | 1.8×10-10 | A | 2010[ | ||||||||||||
| (ZrO2 | Zrn | 2.4×10-12, 1.6×10-12, | B | n=1—5, k1 for | 2008[ | |||||||||||
| 1.2×10-12, 6.3×10-13 | n=2—5,theory | |||||||||||||||
| (ZrO2)nO- | (ZrO2 | 2.1×10-12, 2.2×10-13, | B | n=1—4, theory | 2009[ | |||||||||||
| 3.4×10-13, 1.8×10-13 | ||||||||||||||||
| (ZrO2)nO- | (ZrO2)nC | ca.10-11 | A | n=3—25, Oxidative adsorption | 2013[ | |||||||||||
| Mo | Mo(CO | A | n=1—3 | 2006[ | ||||||||||||
| C6 | 2007[ | |||||||||||||||
| RhnOm | Rhn | ca.10-10 | A | n=10—28, m=1—5, | 2012[ | |||||||||||
| k1 for m=4, 5 | ||||||||||||||||
| Pd | PdO+, OPdCO+, PdCO+ | B | Loss of molecular O2 and | 2011[ | ||||||||||||
| CO oxidation, theory | ||||||||||||||||
| Pd | Pd | B | Loss of molecular O2 and | 2011[ | ||||||||||||
| OPdCO+ | CO oxidation, theory | |||||||||||||||
| PdnO+ | Pdn(CO | C | n=2—7 | 2012[ | ||||||||||||
| Pdn | Pdn(CO | C | n=4—6 | 2012[ | ||||||||||||
| Species | Cluster | Product | k1 | Reactor | Remark | Year | ||||||||||
| d-Block | Pd6 | Pd6 | C | Theory | 2012[ | |||||||||||
| Pd6 | Pd6O5CO+ | C | Eliminate CO2 and form | 2012[ | ||||||||||||
| Pd6 | ||||||||||||||||
| Ag3 | Ag3On(OCO | C | n=1—3, with N2O | 2011[ | ||||||||||||
| Agn | A | C | n=7, 9, 11 | 2004[ | ||||||||||||
| Ag5 | Ag4 | C | Cluster fragmentation | 2005[ | ||||||||||||
| Ag3C | ||||||||||||||||
| AgnNx | Ag3NO-, AgN2 | C | n=2—5, y>x, cluster | 2007[ | ||||||||||||
| fragmentation | ||||||||||||||||
| La2 | La2O3+e- | 1.9×10-10 | A | Electron detachment, theory | 2012[ | |||||||||||
| Wn | Wn | 3.0×10-12, 3.6×10-13, | B | n=1—3 | 2008[ | |||||||||||
| 5.9×10-13 | ||||||||||||||||
| Re | Re | 3.3×10-10 | D | 2001[ | ||||||||||||
| Re | Re | 2.4×10-10 | D | 2001[ | ||||||||||||
| Os | Os | ≥4.0×10-13 | A | n=1—4, k1 for n=1 | 2005[ | |||||||||||
| Ir | Ir | 3.5×10-10 | A | n=1—3, k1 for n=1 | 2005[ | |||||||||||
| Pt | PtO+, Pt+ | 6.6×10-10 | D | 2001[ | ||||||||||||
| Pt | Pt | 2.3×10-10 | A | n=1—3, k1 for n=1 | 2005[ | |||||||||||
| Pt7 | Pt7 | ca.10-9 | D | n=1—2 | 2004[ | |||||||||||
| Pt4 | Pt4 | 10-11—10-9 | D | n=1—3, 4(slow) | 2007[ | |||||||||||
| Ptn | Ptn | B | n=3—6, m=1—2 | 1998[ | ||||||||||||
| AuO+ | AuCO+ | B | Theory | 2008[ | ||||||||||||
| AuO+ | AuCO+ | 7.93×10-12 | B | 2008[ | ||||||||||||
| Au2O+ | Au2CO+ | B | Theory | 2008[ | ||||||||||||
| Au2O+ | Au2CO+ | 2.42×10-12 | B | 2008[ | ||||||||||||
| Au3O+ | Au3CO+ | 4.98×10-12 | B | 2008[ | ||||||||||||
| Au3 | Au3(CO2 | C | 2011[ | |||||||||||||
| Aun | AunOx(CO | A | n=1—2, m=1—5, | 2004[ | ||||||||||||
| x=0—3, y=0—2 | ||||||||||||||||
| AuO- | Au- | ca.10-11 | A | Theory | 2004[ | |||||||||||
| Au | AuO- | A | Very slow, theory | 2004[ | ||||||||||||
| Au | Au- | ca.10-12 | A | Slow, loss of molecular O2, | 2004[ | |||||||||||
| theory | ||||||||||||||||
| Au2O- | A | ca.10-13 | A | Theory | 2006[ | |||||||||||
| Au2 | A | B | 2003[ | |||||||||||||
| Au2 | A | C | 2005[ | |||||||||||||
| Au2 | A | A | Theory | 2006[ | ||||||||||||
| Au2 | Au2 | ca.10-13 | A | Theory | 2006[ | |||||||||||
| Au2 | Au2 | ca.10-13 | A | Theory | 2006[ | |||||||||||
| Au6 | Au6O-, A | A | CO and O2 co-adsorption | 2002[ | ||||||||||||
| Aun | AuxOy(CO | A | n ≥ 4, Oxidation, replacement | 2006[ | ||||||||||||
| and association | ||||||||||||||||
| f-Block | Cen | Cen | 1.9×10-12 | A | n=2—6, k1 for Ce2 | 2010[ | ||||||||||
| theory(n =2—5) | ||||||||||||||||
| Cen | Cen | 8×10-11 | A | n=4—21, k1 for Ce4 | 2011[ | |||||||||||
| theory(n =1—6) | ||||||||||||||||
| EuO+ | Eu+ | ≥3.3×10-12 | A | 2005[ | ||||||||||||
| YbO+ | Yb+ | 1.2×10-11 | A | 2005[ | ||||||||||||
| Hetero- | AlV | AlV | 4.1×10-10 | D | Theory | 2011[ | ||||||||||
| nuclear | VCoO4 | VCoO3 | ca.10-11 | A | Theory | 2012[ | ||||||||||
| YAl | YAl | D | Theory | 2013[ | ||||||||||||
| AuTi3 | AuTi3O6CO+ | A | Molecular association | 2011[ | ||||||||||||
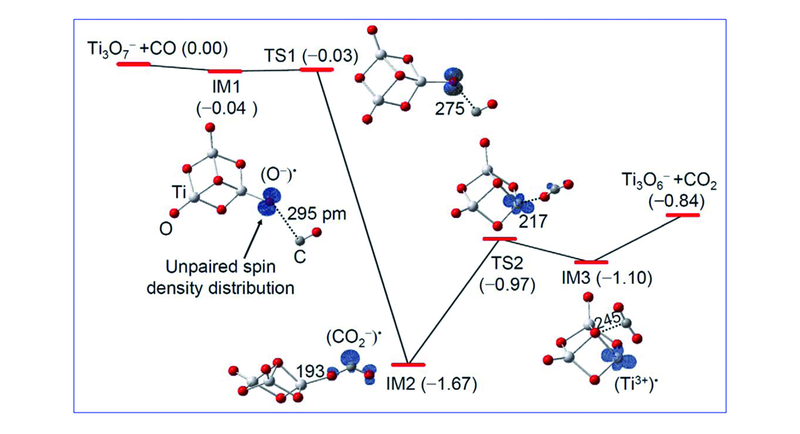
Fig.4 DFT calculated reaction pathway for Ti3O7-+CO→Ti3O6-+CO2[33]The zero-point vibration corrected energies(DH0K/eV) of the reaction intermediates(IM1—IM3), transition states(TS1 and TS2), and products(Ti3O6-+CO2) with respect to the separated reactants(Ti3O7-+CO) are given. Bond lengths in pm are shown.
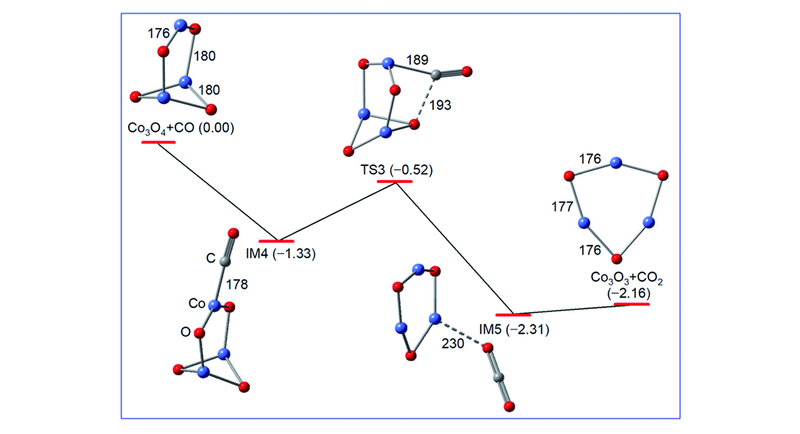
Fig.5 DFT calculated reaction pathway for Co3O4+CO→Co3O3+CO2[65]The zero-point vibration corrected energies(DH0 K/eV) of the reaction intermediates(IM4 and IM5), transition state(TS3), and products(Co3O3+CO2) with respect to the separated reactants(Co3O4+CO) are given. Bond lengths in pm are shown.
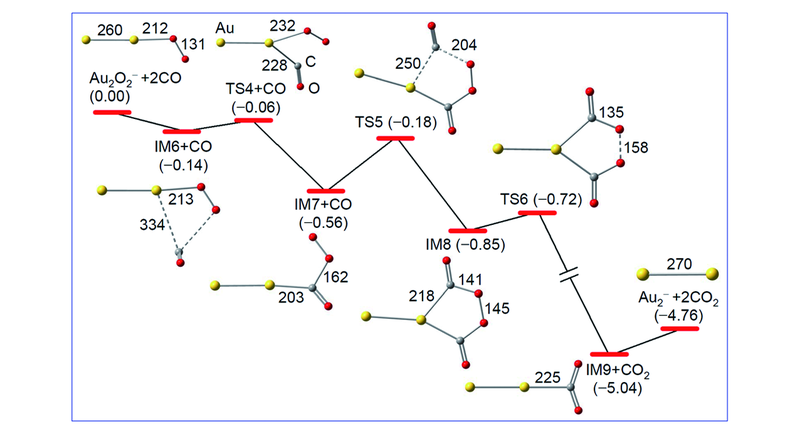
Fig.6 DFT calculated reaction pathway for Au2O2-+2CO→Au2-+2CO2[84] The zero-point vibration corrected energies(DH0 K/eV) of the reaction intermediates(IM6—IM9), transition states(TS4—TS6), and products(Au2-+2CO2) with respect to the separated reactants(Au2O2-+2CO) are given. Bond lengths in pm are shown.
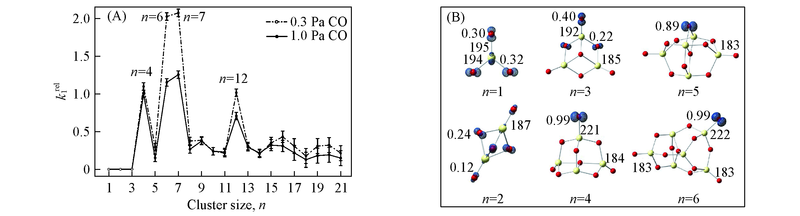
Fig.7 Experimentally determined relative pseudo-first order rate constants(k1) for(CeO2)nO-+CO→ (CeO2)n-+CO2(n=4—21)(A) and DFT calculated structures and profiles of unpaired spin density distributions for(CeO2)nO-(n=1—6) clusters(B)[28] The experiment identified no evidence of reaction between(CeO2)nO-(n=1—3) and CO, so the rate constants are simply set to be zero. The absolute values of k1(Ce4O9-+CO) under the conditions of 0.3 and 1.0 Pa CO in the fast flow reactor are 8.6×10-11 and 7.8×10-11 cm3·molecule-1·s-1, respectively. Abundance of un-reactive cluster isomers can lead to the result that the rate constants under the condition of 0.3 Pa are generally larger than those under 1.0 Pa. The spin density values over oxygen atoms in μB and some Ce—O bond lengths in pm are shown.
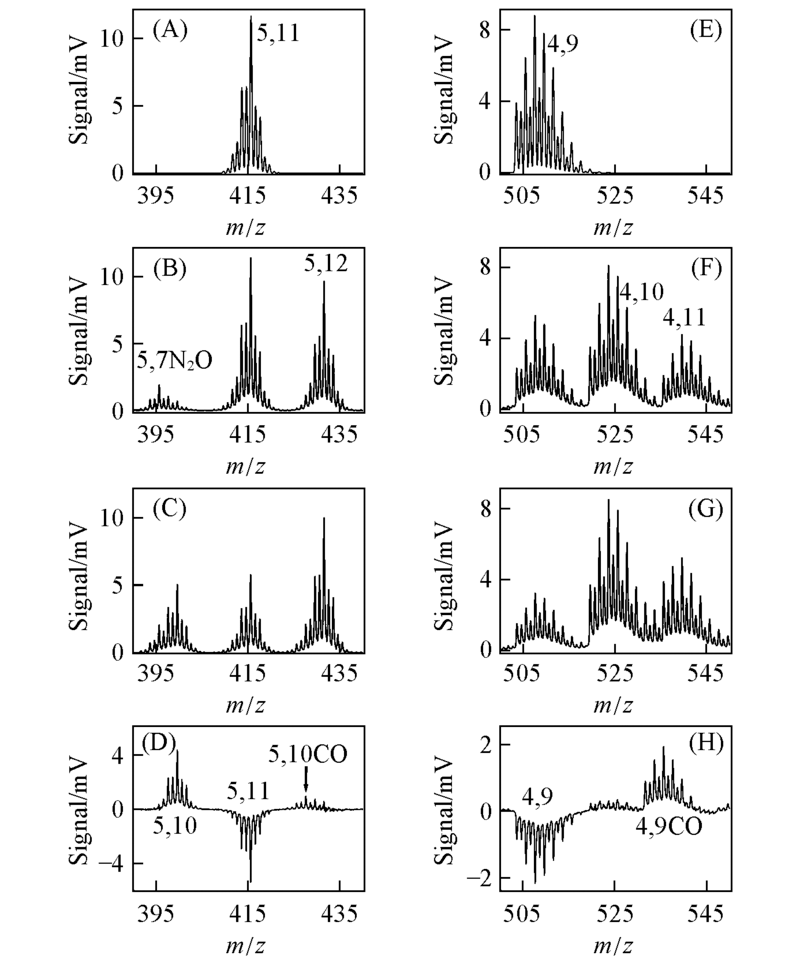
Fig.8 TOF mass spectra for reactions of Ti5Oy-(A—D) and Zr4Oy-(E—H) with CO in the fast flow reactor[33]The numbers x, y denote MxO-y in which M=Ti or Zr. The simulated Ti5O11- and Zr4O9- isotopomers are given in (A) and (E). The reference spectra with N2 in the reactor are given in (B) and(F). The spectra with 1.4 and 0.6 Pa CO in the reactor are given in (C) and (G), respectively. The difference spectra[(D)=(C)-(B) and (H)=(G)-(F)] are also shown.
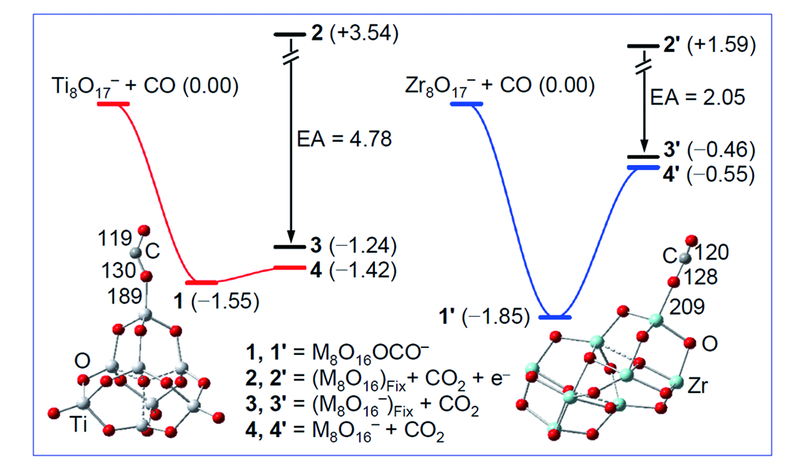
Fig.9 Simplified potential energy profiles for oxidative CO absorption and CO2 desorption in the reactions of CO with M8O17-(M=Ti, Zr) calculated by the DFT[33]The zero-point vibration corrected energies(DH0 K/eV) of the reaction intermediates(1 and 1') and products(4 and 4') with respect to the separated reactants are given. The energies of 2/3 and 2'/3' are calculated with the geometric parameters of M8O16 being fixed at the values of the M8O16 moiety in 1 and 1', respectively(EA denotes electron affinity in eV). Bond lengths in pm are shown.
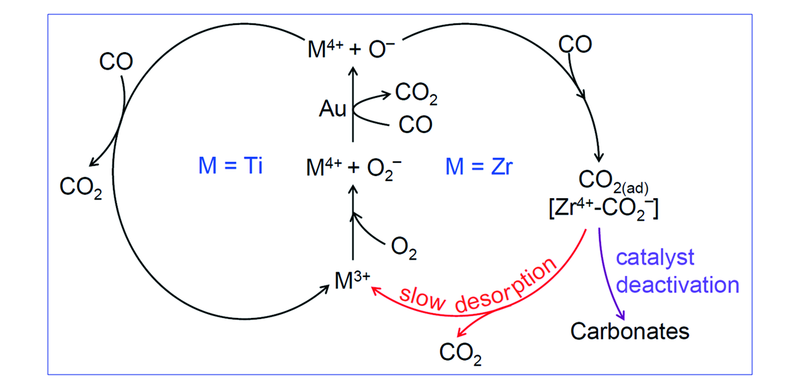
Fig.10 Proposed catalytic cycles involving O2- and O- radicals for low-temperature CO oxidation over titania and zirconia supported gold[33] Gold may participate in the O—O bond activation because it was identified that O2- is unable to react with CO in the absence of gold at low-temperature[103,136,137].
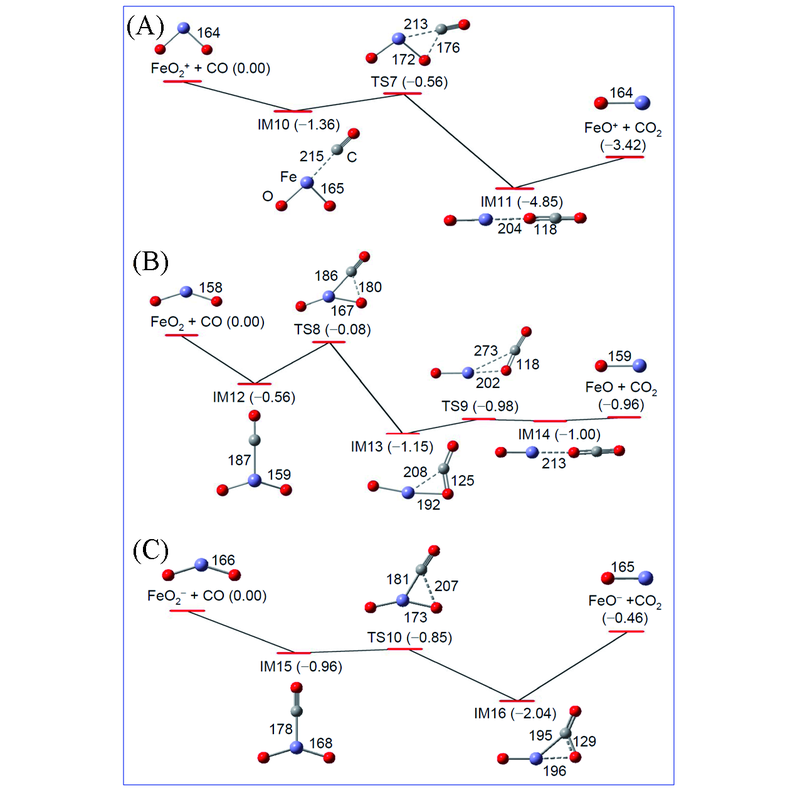
Fig.11 B3LYP/6-311+G* calculated reaction pathways for FeO2q+CO→FeOq+CO2 in which q=+1(A), 0(B) and -1(C)The zero-point vibration corrected energies(ΔH0 K/eV) of the reaction intermediates(IM10—IM16), transition states(TS7—TS10), and products(FeOq+CO2) with respect to the separated reactants(FeO2q+CO) are given. Bond lengths in pm are shown. The structures and mechanisms of FeO2+, FeO2, and FeO2- reaction systems are adapted from refs.[61], [43], and [63], respectively.
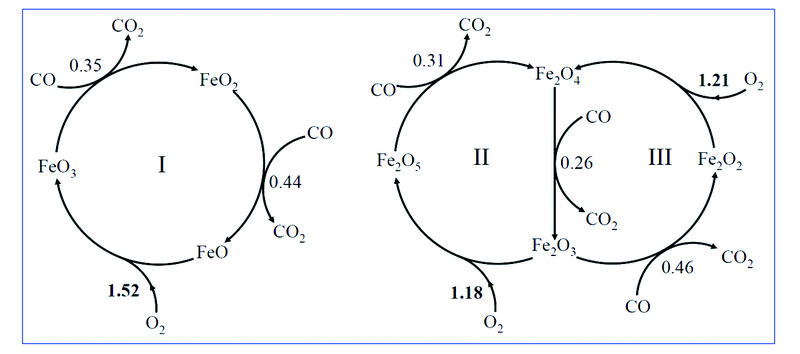
Fig.12 Three model catalytic cycles Ⅰ—Ⅲ for CO oxidation by O2 over FeO1—3 and Fe2O2—5 clusters[43] The free energy barrier(eV) of the rate-limiting step in each elementary reaction is given.
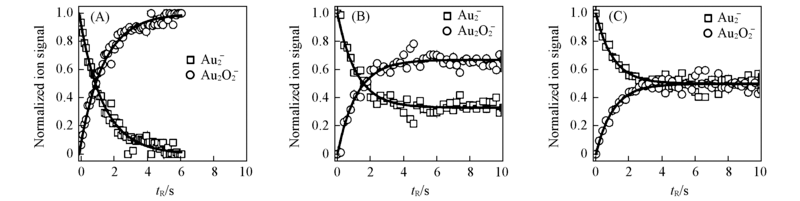
Fig.13 Concentration of all product ions observed during the reactions of mass selected Au2- with CO and O2 inside the octopole ion trap as a function of the reaction time(tR) for different CO partial pressures[87] (A) p(CO)=0 and p(O2)=0.12 Pa, (B) p(CO)=p(O2)=0.12 Pa, (C) p(CO)=2p(O2)=0.24 Pa. The reaction temperature and bath gas(He) pressure are 300 K and 1.2 Pa, respectively.
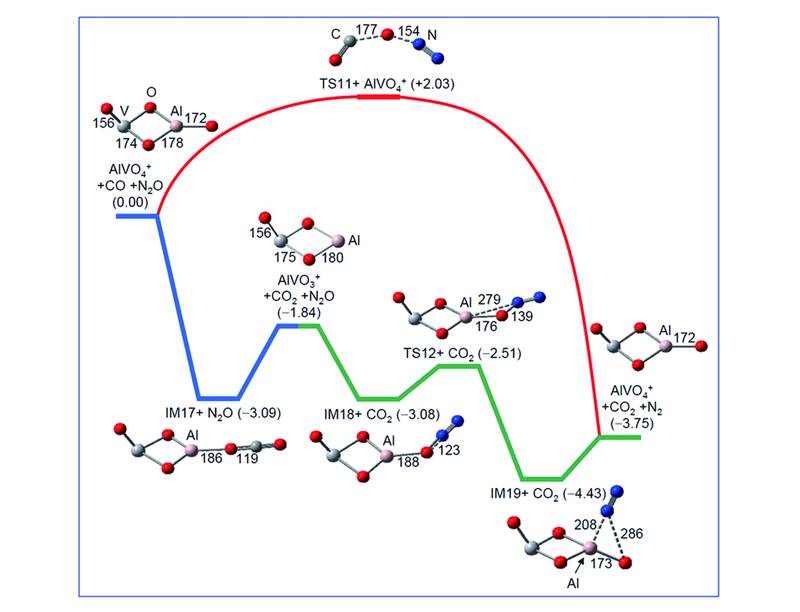
Fig.14 DFT calculated potential energy profile for direct and catalytic oxidations of CO by N2O[91]Catalysts=AlVO3+ and AlVO4+. The energies of the intermediates(IM17—IM19), transition states(TS11 and TS12), and products with respect to the separated reactants are expressed in eV. Bond lengths in pm are shown.
| Catalyst | Oxidant | Reactor | Remark | Year |
|---|---|---|---|---|
| Al2 | N2O | B | 2008[ | |
| Al2 | N2O | B | 2008[ | |
| Ca+, CaO+ | N2O | A | 2005[ | |
| Ti2 | N2O | B | Theory | 2011[ |
| VO2, VO3 | N2O | A | Theory | 2013[ |
| Mn2O4, Mn2O5 | NO2 | A | Theory | 2013[ |
| Fe+, FeO+ | N2O | D | 1981[ | |
| Fe+, FeO+ | N2O | A | Theory | 2005[ |
| Ge+, GeO+ | N2O | A | 2005[ | |
| Sr+, SrO+ | N2O | A | 2005[ | |
| Zrn | N2O | B | n=1—4, theory | 2008[ |
| RhnOm | N2O | A | n=10—28, m=0—5 | 2011[ |
| Pd6 | O2 | C | Theory | 2012[ |
| A | O2 | C | n=7, 9, 11 | 2004[ |
| Ba+, BaO+ | N2O | A | 2005[ | |
| Eu+, EuO+ | N2O | A | 2005[ | |
| Yb+, YbO+ | N2O | A | 2005[ | |
| Os | N2O | A | 2005[ | |
| Ir | N2O | A | 2005[ | |
| Pt | N2O | A | 2005[ | |
| Pt+, Pt | N2O | D | 2001[ | |
| Pt7 | N2O | D | 2004[ | |
| Pt4 | N2O | D | 2007[ | |
| Ptn | O2/N2O | B | n=3—6, m=0—2 | 1998[ |
| A | O2 | C | 2003[ | |
| Au3(CO)2—5 | O2 | A | Theory | 2011[ |
| A | O2 | A | 2002[ | |
| AlV | N2O | D | Theory | 2011[ |
| YAl | N2O | D | Theory | 2013[ |
Table 2 Experimentally studied catalytic oxidations of CO on atomic cluster catalysts
| Catalyst | Oxidant | Reactor | Remark | Year |
|---|---|---|---|---|
| Al2 | N2O | B | 2008[ | |
| Al2 | N2O | B | 2008[ | |
| Ca+, CaO+ | N2O | A | 2005[ | |
| Ti2 | N2O | B | Theory | 2011[ |
| VO2, VO3 | N2O | A | Theory | 2013[ |
| Mn2O4, Mn2O5 | NO2 | A | Theory | 2013[ |
| Fe+, FeO+ | N2O | D | 1981[ | |
| Fe+, FeO+ | N2O | A | Theory | 2005[ |
| Ge+, GeO+ | N2O | A | 2005[ | |
| Sr+, SrO+ | N2O | A | 2005[ | |
| Zrn | N2O | B | n=1—4, theory | 2008[ |
| RhnOm | N2O | A | n=10—28, m=0—5 | 2011[ |
| Pd6 | O2 | C | Theory | 2012[ |
| A | O2 | C | n=7, 9, 11 | 2004[ |
| Ba+, BaO+ | N2O | A | 2005[ | |
| Eu+, EuO+ | N2O | A | 2005[ | |
| Yb+, YbO+ | N2O | A | 2005[ | |
| Os | N2O | A | 2005[ | |
| Ir | N2O | A | 2005[ | |
| Pt | N2O | A | 2005[ | |
| Pt+, Pt | N2O | D | 2001[ | |
| Pt7 | N2O | D | 2004[ | |
| Pt4 | N2O | D | 2007[ | |
| Ptn | O2/N2O | B | n=3—6, m=0—2 | 1998[ |
| A | O2 | C | 2003[ | |
| Au3(CO)2—5 | O2 | A | Theory | 2011[ |
| A | O2 | A | 2002[ | |
| AlV | N2O | D | Theory | 2011[ |
| YAl | N2O | D | Theory | 2013[ |
| [1] | Lide D.R., CRC Handbook of Chemistry and Physics, 87th ed.: Physical Constants of Inorganic Compounds, CRC Taylor & Francis Group, Boca Raton, 2006, 4—56 |
| [2] | Jones R. A., Strickland J. A., Stunkard J. A., Siegel J., Toxicol. Appl. Pharmacol., 1971, 19, 46 |
| [3] | Royer S., Duprez D., Chem. Cat. Chem., 2011, 3, 24—65 |
| [4] | Freund H. J., Meijer G., Scheffler M., Schlogl R., Wolf M., Angew. Chem. Int. Ed., 2011, 50, 10064—10094 |
| [5] | Cotton F.A., Wilkinson G., Advanced Inorganic Chemistry, A Comprehensive Text, 4th ed., John Wiley & Sons, New York, 1980, 1049—1079 |
| [6] | Zhou M.F., Andrews L., Bauschlicher C. W. Jr., Chem. Rev., 2001, 101, 1931—1961 |
| [7] | Roithova J. , Schröder D., Chem. Rev., 2010, 110, 1170—1211 |
| [8] | Zhai H. J., Wang L. S., Chem. Phys. Lett., 2010, 500, 185—195 |
| [9] | Castleman A. W. Jr., Catal. Lett., 2011, 141, 1243—1253 |
| [10] | Yin S., Bernstein E. R., Int. J. Mass Spectrom., 2012, 321/322, 49—65 |
| [11] | Lang S. M., Bernhardt T. M., Phys. Chem. Chem. Phys., 2012, 14, 9255—9269 |
| [12] | Asmis K. R., Phys. Chem. Chem. Phys., 2012, 14, 9270—9281 |
| [13] | Ding X. L., Wu X. N., Zhao Y. X., He S. G., Acc. Chem. Res., 2012, 45, 382—390 |
| [14] | Schlangen M., Schwarz H., Catal. Lett., 2012, 142, 1265—1278 |
| [15] | Qiao B. T., Wang A. Q., Yang X. F., Allard L. F., Jiang Z., Cui Y. T., Liu J. Y., Li J., Zhang T., Nat. Chem., 2011, 3, 634—641 |
| [16] | Yoon B., Häkkinen H., Landman U., Worz A. S., Antonietti J. M., Abbet S., Judai K., Heiz U., Science,2005, 307, 403—407 |
| [17] | Valden M., Lai X., Goodman D. W., Science,1998, 281, 1647—1650 |
| [18] | Min B. K., Friend C. M., Chem. Rev., 2007, 107, 2709—2724 |
| [19] | Fierro-Gonzalez J.C., Gates B. C., Chem. Soc. Rev., 2008, 37, 2127—2134 |
| [20] | O'Hair R. A. J., Khairallah G. N., J. Cluster Sci., 2004, 15, 331—363 |
| [21] | Böhme D. K., Schwarz H., Angew. Chem. Int. Ed., 2005, 44, 2336—2354 |
| [22] | Johnson G. E., Mitrić R., BonaČić-Koutecky V., Castleman A. W. Jr., Chem. Phys. Lett., 2009, 475, 1—9 |
| [23] | Bernhardt T. M., Int. J. Mass Spectrom., 2005, 243, 1—29 |
| [24] | Zhao Y.X., Wu X. N., Ma J. B., He S. G., Ding X. L., Phys. Chem. Chem. Phys., 2011, 13, 1925—1938 |
| [25] | Lang S. M., Fleischer I., Bernhardt T. M., Barnett R. N., Landman U., J. Am. Chem. Soc., 2012, 134, 20654—20659 |
| [26] | Lang S. M., Schnabel T., Bernhardt T. M., Phys. Chem. Chem. Phys., 2012, 14, 9364—9370 |
| [27] | Wu X. N., Zhao Y. X., Xue W., Wang Z. C., He S. G., Ding X. L., Phys. Chem. Chem. Phys., 2010, 12, 3984—3997 |
| [28] | Wu X. N., Ding X. L., Bai S. M., Xu B., He S. G., Shi Q., J. Phys. Chem. C,2011, 115, 13329—13337 |
| [29] | Wang Z. C., Yin S., Bernstein E. R., J. Phys. Chem. Lett., 2012, 3, 2415—2419 |
| [30] | Xu B., Zhao Y. X., Ding X. L., He S. G., Int. J. Mass Spectrom., 2013, 334, 1—7 |
| [31] | Ma J. B., Wang Z. C., Schlangen M., He S. G., Schwarz H., Angew. Chem. Int. Ed., 2013, 52, 1226—1230 |
| [32] | Yuan Z., Zhao Y. X., Li X. N., He S. G., Int. J. Mass Spectrom., 2013, 354/355, 105—112 |
| [33] | Ma J. B., Xu B., Meng J. H., Wu X. N., Ding X. L., Li X. N., He S. G., J. Am. Chem. Soc., 2013, 135, 2991—2998 |
| [34] | Wang Z. C., Yin S., Bernstein E. R., Phys.Chem. Chem. Phys., 2013, 15, 10429—10434 |
| [35] | Yamada A., Miyajima K., Mafuné F., Phys. Chem. Chem. Phys., 2012, 14, 4188—4195 |
| [36] | Hirabayashi S., Kawazoe Y., Ichihashi M., Eur. Phys. J. D,2013, 67, 1—6 |
| [37] | Sakuma K., Miyajima K., Mafuné F., J. Phys. Chem. A,2013, 117, 3260—3265 |
| [38] | Yin S., Wang Z. C., Bernstein E. R., J. Chem Phys., 2013, 139, 084307(9) |
| [39] | Dietz T. G., Duncan M. A., Powers D. E., Smalley R. E., J. Chem. Phys., 1981, 74, 6511—6512 |
| [40] | Himeno H., Miyajima K., Yasuike T., Mafuné F., J. Phys. Chem. A,2011, 115, 11479—11485 |
| [41] | Blagojevic V., Orlova G., Bohme D. K., J. Am. Chem. Soc., 2005, 127, 3545—3555 |
| [42] | He S. G., Xie Y., Dong F., Heinbuch S., Jakubikova E., Rocca J. J., Bernstein E. R., J. Phys. Chem. A,2008, 112, 11067—11077 |
| [43] | Xue W., Wang Z. C., He S. G., Xie Y., Bernstein E. R., J. Am. Chem. Soc., 2008, 130, 15879—15888 |
| [44] | Geusic M.E., Morse M. D., O’brien S. C., Smalley R. E., Rev. Sci. Instrum., 1985, 56, 2123—2130 |
| [45] | Wu X. N., Ma J. B., Xu B., Zhao Y. X., Ding X. L., He S. G., J. Phys. Chem. A,2011, 115, 5238—5246 |
| [46] | Bell R. C., Zemski K. A., Justes D. R., Castleman A. W. Jr., J. Chem. Phys., 2001, 114, 798—811 |
| [47] | Socaciu L. D., Hagen J., Heiz U., Bernhardt T. M., Leisner T., Wöste L., Chem. Phys. Lett., 2001, 340, 282—288 |
| [48] | Eller K., Schwarz H., Int. J. Mass Spectrom. Ion Processes,1989, 93, 243—257 |
| [49] | Berg C., Schindler T., Niednerschatteburg G., Bondybey V. E., J. Chem. Phys., 1995, 102, 4870—4884 |
| [50] | Wallace W. T., Whetten R. L., J. Am. Chem. Soc., 2002, 124, 7499—7505 |
| [51] | Bondybey V. E., Beyer M. K., J. Phys. Chem. A,2001, 105, 951—960 |
| [52] | Cramer C. J., Truhlar D. G., Phys. Chem. Chem. Phys., 2009, 11, 10757—10816 |
| [53] | Burke K., J. Chem. Phys., 2012, 136, 150901-1—150901-9 |
| [54] | Steinfeld J.I., Francisco J. S., Hase W. L., Chemical Kinetics and Dynamics, Prentice Hall, Upper Saddle River, NJ, 1999, 340—345 |
| [55] | Beyer T., Swinehart D. R., Commun. ACM,1973, 16, 379—379 |
| [56] | Baxter R. J., Hu P., J. Chem. Phys., 2002, 116, 4379—4381 |
| [57] | Johnson G. E., Tyo E. C., Castleman. A. W. Jr., J. Phys. Chem. A,2008, 112, 4732—4735 |
| [58] | Broadley S., Vondrak T., Wright T. G., Plane J. M. C., Phys. Chem. Chem. Phys., 2008, 10, 5287—5298 |
| [59] | Tyo E. C., Nöβler M., Mitrić R., BonaČić-Koutecky V., Castleman A. W. Jr., Phys. Chem. Chem. Phys., 2011, 13, 4243—4249 |
| [60] | Kappes M. M., Staley R. H., J. Am. Chem. Soc., 1981, 103, 1286—1287 |
| [61] | Reilly N. M., Reveles J. U., Johnson G. E., Del Campo J. M., Khanna S. N., Köster A. M., Castleman. A. W. Jr., J. Phys. Chem. C,2007, 111, 19086—19097 |
| [62] | Reilly N. M., Reveles J. U., Johnson G. E., Khanna S. N., Castleman A. W. Jr., Chem. Phys. Lett., 2007, 435, 295—300 |
| [63] | Reilly N. M., Reveles J. U., Johnson G. E., Khanna S. N., Castleman A. W. Jr., J. Phys. Chem. A,2007, 111, 4158—4166 |
| [64] | Reveles J. U., Johnson G. E., Khanna S. N., Castleman. A. W. Jr., J. Phys. Chem. C,2010, 114, 5438—5446 |
| [65] | Xie Y., Dong F., Heinbuch S., Rocca J. J., Bernstein E. R., Phys. Chem. Chem. Phys., 2010, 12, 947—959 |
| [66] | Johnson G. E., Reveles J. U., Reilly N. M., Tyo E. C., Khanna S. N., Castleman A. W. Jr., J. Phys. Chem. A,2008, 112, 11330—11340 |
| [67] | Johnson G. E., Reilly N. M., Castleman A. W. Jr., Int. J. Mass Spectrom., 2009, 280, 93—100 |
| [68] | Johnson G. E., Mitrić R., Tyo E. C., BonaČić-Koutecky V., Castleman A. W. Jr., J. Am. Chem. Soc., 2008, 130, 13912—13920 |
| [69] | Johnson G. E., Mitrić R., Nössler M., Tyo E. C., BonaČić-Koutecky V., Castleman A. W. Jr., J. Am. Chem. Soc,2009, 131, 5460—5470 |
| [70] | Wyrwas R. B., Jarrold C. C., J. Am. Chem. Soc., 2006, 128, 13688—13689 |
| [71] | Wyrwas R. B., Robertson E. M., Jarrold C. C., J. Chem. Phys., 2007, 126, 214309-1—214309-8 |
| [72] | Reber A. C., Khanna S. N., Tyo E. C., Harmon C. L., Castleman A. W. Jr., J. Chem. Phys., 2011, 135, 234303-1—234303-7 |
| [73] | Popolan D. M., Bernhardt T. M., J. Chem. Phys., 2011, 134, 091102-1—091102-3 |
| [74] | Socaciu L.D., Hagen J., Le Roux J., Popolan D., Bernhardt T. M., Wöste L., Vajda S., J. Chem. Phys., 2004, 120, 2078—2081 |
| [75] | Bernhardt T. M., Socaciu-Siebert L. D., Hagen J., Wöste L., Appl. Catal. A-Gen., 2005, 291, 170—178 |
| [76] | Hagen J., Socaciu-Siebert L. D., Le Roux J., Popolan D., Vajda S., Bernhardt T. M., Wöste L., Int. J. Mass Spectrom., 2007, 261, 152—158 |
| [77] | Johnson G. E., Tyo E. C., Castleman A. W. Jr., Proc. Natl. Acad. Sci. USA,2008, 105, 18108—18113 |
| [78] | Beyer M. K., Berg C. B., Bondybey V. E., Phys. Chem. Chem. Phys., 2001, 3, 1840—1847 |
| [79] | Brönstrup M., Schröder D., Kretzschmar I., Schwarz H., Harvey J. N., J. Am. Chem. Soc., 2001, 123, 142—147 |
| [80] | Balaj O. P., Balteanu I., Rossteuscher T. T. J., Beyer M. K., Bondybey V. E., Angew. Chem. Int. Ed., 2004, 43, 6519—6522 |
| [81] | Siu C. K., Reitmeier S. J., Balteanu I., Bondybey V. E., Beyer M. K., Eur. Phys. J. D,2007, 43, 189—192 |
| [82] | Shi Y., Ervin K. M., J. Chem. Phys., 1998, 108, 1757—1760 |
| [83] | Kimble M. L., Castleman A. W. Jr., Mitrić R., Bürgel C., BonaČić-Koutecky V., J. Am. Chem. Soc., 2004, 126, 2526—2535 |
| [84] | Kimble M. L., Moore N. A., Johnson G. E., Castleman A. W. Jr., Burgel C., Mitrić R., BonaČić-Koutecky V., J. Chem. Phys., 2006, 125, 204311-1—204311-14 |
| [85] | Kimble M. L., Castleman A. W. Jr., Bürgel C., BonaČić-Koutecky V., Int. J. Mass Spectrom., 2006, 254, 163—167 |
| [86] | Kimble M. L., Moore N. A., Castleman A. W. Jr., Bürgel C., Mitrić R., BonaČić-Koutecky V., Eur. Phys. J. D,2007, 43, 205—208 |
| [87] | Socaciu L. D., Hagen J., Bernhardt T. M., Wöste L., Heiz U., Hakkinen H., Landman U., J. Am. Chem. Soc., 2003, 125, 10437—10445 |
| [88] | Kimble M. L., Castleman A. W. Jr., Int. J. Mass Spectrom., 2004, 233, 99—101 |
| [89] | Bürgel C., Reilly N. M., Johnson G. E., MitrićR., Kimble M. L., Castleman A. W. Jr., BonaČić-Koutecky V., J. Am. Chem. Soc., 2008, 130, 1694—1698 |
| [90] | Johnson G. E., Reilly N. M., Tyo E. C., Castleman A. W. Jr., J. Phys. Chem. C,2008, 112, 9730—9736 |
| [91] | Wang Z. C., Dietl N., Kretschmer R., Weiske T., Schlangen M., Schwarz H., Angew. Chem. Int. Ed., 2011, 50, 12351—12354 |
| [92] | Baranov V., Javahery G., Hopkinson A. C., Bohme D. K., J. Am. Chem. Soc., 1995, 117, 12801—12809 |
| [93] | Che M., Tench A. J., Adv. Catal., 1982, 31, 77—133 |
| [94] | Lee J., Grabowski J. J., Chem. Rev., 1992, 92, 1611—1647 |
| [95] | Tang D. Y., Hu J. P., Zhang Y. Q., Hu C. W., Acta Chim. Sin., 2009, 67, 1859—1864 |
| [96] | Tang D. Y., Hu J. P., Zhang Y. Q., Hu C. W., Acta Chim. Sin., 2010, 68, 1379—1384 |
| [97] | Grigorieva A. V., Goodilin E. A., Dubova K. L., Anufrieva T. A., Derlyukova L. E., Vyacheslavov A. S., Tretyakov Y. D., Solid State Sci., 2010, 12, 1024—1028 |
| [98] | Liu X. W., Zhou K. B., Wang L., Wang B. Y., Li Y. D., J. Am. Chem. Soc., 2009, 131, 3140—3141 |
| [99] | Gonzalez-Rovira L., Sanchez-Amaya J. M., Lopez-Haro M., del Rio E., Hungria A. B., Midgley P., Calvino J. J., Bernal S., Botana F. J., Nano Lett., 2009, 9, 1395—1400 |
| [100] | Haruta M., Kobayashi T., Sano H., Yamada N., Chem. Lett., 1987, 2, 405—408 |
| [101] | Konova P., Naydenov A., Venkov C., Mehandjiev D., Andreeva D., Tabakova T., J. Mol. Catal. A: Chem., 2004, 213, 235—240 |
| [102] | Konova P., Naydenov A., Tabakova T., Mehandjiev D., Catal. Commun., 2004, 5, 537—542 |
| [103] | Carrettin S., Concepción P., Corma A., Nieto J. M. L., Puntes V. F., Angew. Chem. Int. Ed., 2004, 43, 2538—2540 |
| [104] | Widmann D., Behm R. J., Angew. Chem. Int. Ed., 2011, 50, 10241—10245 |
| [105] | Kotobuki M., Leppelt R., Hansgen D. A., Widmann D., Behm R. J., J. Catal., 2009, 264, 67—76 |
| [106] | Widmann D., Liu Y., Schüth F., Behm R. J., J. Catal., 2010, 276, 292—305 |
| [107] | Zhao Y. X., Ding X. L., Ma Y. P., Wang Z. C., He S. G., Theor. Chem. Acc., 2010, 127, 449—465 |
| [108] | Wu X. N., Xu B., Meng J. H., He S. G., Int. J. Mass Spectrom., 2012, 310, 57—64 |
| [109] | Yin S., Ma Y. P., Du L., He S. G., Ge M. F., Chin. Sci. Bull., 2008, 53, 3829—3838 |
| [110] | Ma Y. P., Zhao Y. X., Li Z. Y., Ding X. L., He S. G., Chin. J. Chem. Phys., 2011, 24, 586—596 |
| [111] | Tian L. H., Zhao Y. X., Wu X. N., Ding X. L., He S. G., Ma T. M., Chem. Phys. Chem., 2012, 13, 1282—1288 |
| [112] | Panov G. I., Dubkov K. A., Starokon E. V., Catal. Today,2006, 117, 148—155 |
| [113] | Zhao C., Wachs I. E., Catal. Today,2006, 118, 332—343 |
| [114] | Feyel S., Döbler J., Schröder D., Sauer J., Schwarz H., Angew. Chem. Int. Ed., 2006, 45, 4681—4685 |
| [115] | Feyel S., Döbler J., Höckendorf R., Beyer M.K., Sauer J., Schwarz H., Angew. Chem. Int. Ed., 2008, 47, 1946—1950 |
| [116] | Schlangen M., Schwarz H., Dalton Trans., 2009, 46, 10155—10165 |
| [117] | Dietl N., Höckendorf R. F., Schlangen M., Lerch M., Beyer M. K., Schwarz H., Angew. Chem. Int. Ed., 2011, 50, 1430—1434 |
| [118] | Zhao Y. X., Wu X. N., Wang Z. C., He S. G., Ding X. L., Chem. Commun., 2010, 46, 1736—1738 |
| [119] | Ding X. L., Zhao Y. X., Wu X. N., Wang Z. C., Ma J. B., He S. G., Chem.-Eur. J., 2010, 16, 11463—11470 |
| [120] | Wang Z. C., Wu X. N., Zhao Y. X., Ma J. B., Ding X. L., He S. G., Chem. Phys. Lett., 2010, 489, 25—29 |
| [121] | Ma J. B., Wu X. N., Zhao X. X., Ding X. L., He S. G., Phys. Chem. Chem. Phys., 2010, 12, 12223—12228 |
| [122] | Ma J. B., Wu X. N., Zhao Y. X., Ding X. L., He S. G., J. Phys. Chem. A,2010, 114, 10024—10027 |
| [123] | Zhao Y. X., Wu X. N., Ma J. B., He S. G., Ding X. L., J. Phys. Chem. C,2010, 114, 12271—12279 |
| [124] | Ma J. B., Wu X. N., Zhao Y. X., Ding X. L., He S. G., Chin. J. Chem. Phys., 2010, 23, 133—137 |
| [125] | Zhao Y. X., Yuan J. Y., Ding X. L., He S. G., Zheng W. J., Phys. Chem. Chem. Phys., 2011, 13, 10084—10090 |
| [126] | Wang Z. C., Wu X. N., Zhao Y. X., Ma J. B., Ding X. L., He S. G., Chem.-Eur. J., 2011, 17, 3449—3457 |
| [127] | Meng J. H., Zhao Y. X., He S. G., J. Phys. Chem. C,2013, 117, 17548—17556 |
| [128] | Dietl N., Schlangen M., Schwarz H., Angew. Chem. Int. Ed., 2012, 51, 5544—5555 |
| [129] | Dong F., Heinbuch S., He S. G., Xie Y., Rocca J. J., Bernstein E. R., J. Chem. Phys., 2006, 125, 164318—164318 |
| [130] | Wang W. G., Wang Z. C., Yin S., He S. G., Ge M. F., Chin. J. Chem. Phys., 2007, 20, 412—418 |
| [131] | Dong F., Heinbuch S., Xie Y., Rocca J.J., Bernstein E. R., Wang Z. C., Deng K., He S. G., J. Am. Chem. Soc., 2008, 130, 1932—1943 |
| [132] | Yin S., Xue W., Ding X. L., Wang W. G., He S. G., Ge M. F., Int. J. Mass Spectrom., 2009, 281, 72—78 |
| [133] | Li X. N., Xu B., Ding X. L., He S. G., Dalton Trans., 2012, 41, 5562—5570 |
| [134] | Ma J.B., Zhao Y. X., He S. G., Ding X. L., J. Phys. Chem. A, 2012, 116, 2049—2054 |
| [135] | Manzoli M., Boccuzzi F., Chiorino A., Vindigni F., Deng W., Flytzani-Stephanopoulos M., J. Catal., 2007, 245, 308—315 |
| [136] | Liu H., Kozlov A. I., Kozlova A. P., Shido T., Asakura K., Iwasawa Y., J. Catal., 1999, 185, 252—264 |
| [137] | Guzman J., Carrettin S., Fierro-Gonzalez J. C., Hao Y. L., Gates B. C., Corma A., Angew. Chem. Int. Ed., 2005, 44, 4778—4781 |
| [138] | Walker J. S., Straguzzi G. I., Manogue W. H., Schuit G. C. A., J. Catal., 1988, 110, 298—309 |
| [139] | Uddin M. A., Komatsu T., Yashima T., J. Catal., 1994, 146, 468—475 |
| [140] | Li P., Miser D. E., Rabiei S., Yadav R. T., Hajaligol M. R., Appl. Catal. B: Environ., 2003, 43, 151—162 |
| [141] | Xiong Y. J., Li Z. Q., Li X. X., Hu B., Xie Y., Inorg. Chem., 2004, 43, 6540—6542 |
| [142] | Lin H. Y., Chen Y. W., Wang W. J., J. Nanopart. Res., 2005, 7, 249—263 |
| [143] | Szegedi A., Hegedüs M., Margitfalvi J.L., Kiricsi I.,Chem. Commun., 2005, (11), 1441—1443 |
| [144] | Khedr M. H., Halim K. S. A., Nasr M. I., El-Mansy A. M., Mater. Sci. Eng. A-Struct. Mater. Prop. Microstruct. Process., 2006, 430, 40—45 |
| [145] | Hu C. Q., Gao Z. H., Yang X. R., Chem. Lett., 2006, 35, 1288—1289 |
| [146] | Zheng Y. H., Cheng Y., Wang Y. S., Bao F., Zhou L. H., Wei X. F., Zhang Y. Y., Zheng Q., J. Phys. Chem. B,2006, 110, 3093—3097 |
| [147] | Foster J. P., Weinhold F., J. Am. Chem. Soc., 1980, 102, 7211—7218 |
| [148] | Reed A. E., Curtiss L. A., Weinhold F., Chem. Rev., 1988, 88, 899—926 |
| [149] | Li J., Li X., Zhai H. J., Wang L. S., Science,2003, 299, 864—867 |
| [150] | Wang L. S., Phys. Chem. Chem. Phys., 2010, 12, 8694—8705 |
| [151] | Woodham A. P., Meijer G., Fielicke A., Angew. Chem. Int. Ed., 2012, 51, 4444—4447 |
| [152] | Häkkinen H., Landman U., J. Am. Chem. Soc., 2001, 123, 9704—9705 |
| [153] | Hagen J., Socaciu L. D., Elijazyfer M., Heiz U., Bernhardt T. M., Wöste L., Phys. Chem. Chem. Phys., 2002, 4, 1707—1709 |
| [154] | Tang D. Y., Zhang Y. Q., Hu C. W., Acta Chim. Sin., 2008, 66, 1501—1507 |
| [155] | Xie Y., Dong F., Bernstein E. R., Catal. Today,2011, 177, 64—71 |
| [156] | Lira E., Hansen J.Ø., Huo P., Bechstein R., Galliker P., Lægsgaard E., Hammer B., Wendt S., Besenbacher F., Surf. Sci., 2010, 604, 1945—1960 |
| [157] | Blagojevic V., Flaim E., Jarvis M. J. V., Koyanagi G. K., Bohme D. K., J. Phys. Chem. A,2005, 109, 11224—11235 |
| [158] | Blagojevic V., Bohme D. K., Int. J. Mass Spectrom., 2006, 254, 152—154 |
| [159] | DašićA., Zhao X., Bohme D. K., Int. J. Mass Spectrom., 2006, 254, 155—162 |
| [160] | Blagojevic V., BožovićA., Orlova G., Bohme D. K., J. Phys. Chem. A,2008, 112, 10141—10146 |
| [161] | Tian L. H., Ma T. M., Li X. N., He S. G., Dalton Trans., 2013, 42, 11205—11211 |
| [162] | Coquet R., Howard K.L., Willock D. J., Chem. Soc. Rev., 2008, 37, 2046—2076 |
| [1] | 滕镇远, 张启涛, 苏陈良. 聚合物单原子光催化剂的载流子分离和表面反应机制[J]. 高等学校化学学报, 2022, 43(9): 20220325. |
| [2] | 杨静怡, 施思齐, 彭怀涛, 杨其浩, 陈亮. Ga-C3N4单原子催化剂高效光驱动CO2环加成[J]. 高等学校化学学报, 2022, 43(9): 20220349. |
| [3] | 王茹玥, 魏呵呵, 黄凯, 伍晖. 单原子材料的冷冻合成[J]. 高等学校化学学报, 2022, 43(9): 20220428. |
| [4] | 王新天, 李攀, 曹越, 洪文浩, 耿忠璇, 安志洋, 王昊宇, 王桦, 孙斌, 朱文磊, 周旸. 单原子材料在二氧化碳催化中的技术经济分析与产业化应用前景[J]. 高等学校化学学报, 2022, 43(9): 20220347. |
| [5] | 唐全骏, 刘颖馨, 孟蓉炜, 张若天, 凌国维, 张辰. 单原子催化在海洋能源领域的应用[J]. 高等学校化学学报, 2022, 43(9): 20220324. |
| [6] | 楚宇逸, 兰畅, 罗二桂, 刘长鹏, 葛君杰, 邢巍. 单原子铈对弱芬顿效应活性位点氧还原稳定性的提升[J]. 高等学校化学学报, 2022, 43(9): 20220294. |
| [7] | 杨静怡, 李庆贺, 乔波涛. 铱单原子和纳米粒子在N2O分解反应中的协同催化[J]. 高等学校化学学报, 2022, 43(9): 20220388. |
| [8] | 林高鑫, 王家成. 单原子掺杂二硫化钼析氢催化的进展和展望[J]. 高等学校化学学报, 2022, 43(9): 20220321. |
| [9] | 任诗杰, 谯思聪, 刘崇静, 张文华, 宋礼. 铂单原子催化剂同步辐射X射线吸收谱的研究进展[J]. 高等学校化学学报, 2022, 43(9): 20220466. |
| [10] | 汪思聪, 庞贝贝, 刘潇康, 丁韬, 姚涛. XAFS技术在单原子电催化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220487. |
| [11] | 秦永吉, 罗俊. 单原子催化剂在CO2转化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220300. |
| [12] | 姚青, 俞志勇, 黄小青. 单原子催化剂的合成及其能源电催化应用的研究进展[J]. 高等学校化学学报, 2022, 43(9): 20220323. |
| [13] | 范建玲, 唐灏, 秦凤娟, 许文静, 谷鸿飞, 裴加景, 陈文星. 氮掺杂超薄碳纳米片复合铂钌单原子合金催化剂的电化学析氢性能[J]. 高等学校化学学报, 2022, 43(9): 20220366. |
| [14] | 林治, 彭志明, 贺韦清, 沈少华. 单原子与团簇光催化: 竞争与协同[J]. 高等学校化学学报, 2022, 43(9): 20220312. |
| [15] | 程前, 杨博龙, 吴文依, 向中华. S掺杂Fe-N-C高活性氧还原反应催化剂[J]. 高等学校化学学报, 2022, 43(9): 20220341. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||