

高等学校化学学报 ›› 2024, Vol. 45 ›› Issue (3): 20230474.doi: 10.7503/cjcu20230474
收稿日期:2023-11-13
出版日期:2024-03-10
发布日期:2023-12-29
通讯作者:
孟庆华
E-mail:qhmeng@sjtu.edu.cn
Received:2023-11-13
Online:2024-03-10
Published:2023-12-29
Contact:
MENG Qinghua
E-mail:qhmeng@sjtu.edu.cn
摘要:
研究了湿度和有机物小分子等储存耐候条件对声学增强材料谐振频率偏移值的影响, 探讨了由不同硅铝比分子筛组成的声学增强材料在不同湿度及有机物小分子气氛中储存后的谐振频率及偏移值的变化规律. 研究结果表明, 由既定硅铝比分子筛组成的声学增强材料在25 ℃/50%相对湿度(RH)和25 ℃/95%RH条件下储存后, 其谐振频率偏移值未发生明显变化. 氮气吸附实验结果表明, 25 ℃下氮气分子占据了较少的比表面积, 水分子的存在不影响声学增强材料对氮气分子的吸附. 在25 ℃/50%RH条件下放置后, 声学增强材料在25 ℃/95%RH条件下吸附的水分子会逐渐脱附. 声学增强材料在不同种类有机物小分子气氛中储存后, 其谐振频率偏移值呈不同的变化趋势. 对于尺寸较小的有机物小分子, 由于存在可逆吸附, 声学增强材料的谐振频率偏移值未有明显变化; 对于尺寸较大或具有小尺寸端基基团的有机物小分子, 由于存在嵌入式吸附, 声学增强材料的谐振频率偏移值明显降低.
中图分类号:
TrendMD:
孟庆华, 史超. 分子筛型声学增强材料的储存耐候性能. 高等学校化学学报, 2024, 45(3): 20230474.
MENG Qinghua, SHI Chao. Storage Weathering Resistance of Zeolitic Acoustically Enhanced Materials. Chem. J. Chinese Universities, 2024, 45(3): 20230474.
| Cell parameter | Orthorhombic | Monoclinic | Cell parameter | Orthorhombic | Monoclinic |
|---|---|---|---|---|---|
| a/nm | 2.0070 | 2.0107 | β/(°) | 90 | 90 |
| b/nm | 1.9920 | 1.9879 | γ/(°) | 90 | 90 |
| c/nm | 1.3420 | 1.3369 | Space group | Pnma | P21/n |
| α/(°) | 90 | 90.67 |
Table 1 Cell parameters of MFI under orthorhombic and monoclinic crystal systems[14]
| Cell parameter | Orthorhombic | Monoclinic | Cell parameter | Orthorhombic | Monoclinic |
|---|---|---|---|---|---|
| a/nm | 2.0070 | 2.0107 | β/(°) | 90 | 90 |
| b/nm | 1.9920 | 1.9879 | γ/(°) | 90 | 90 |
| c/nm | 1.3420 | 1.3369 | Space group | Pnma | P21/n |
| α/(°) | 90 | 90.67 |
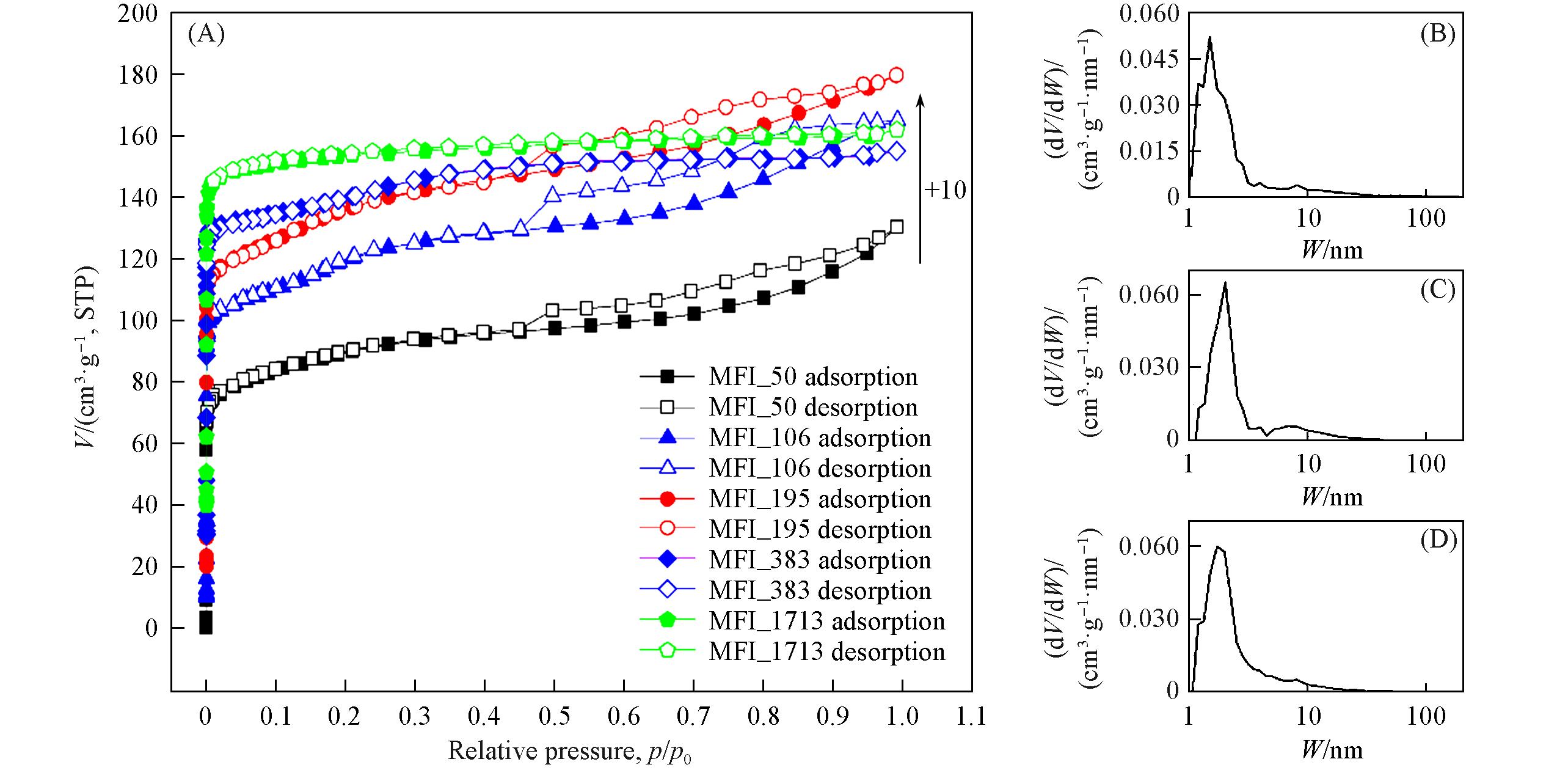
Fig.2 N2 adsorption⁃desorption isotherms(‒196 ℃) of zeolitic AEMs with various SARs(A) and pore size distribution of MFI_50(B), MFI_106(C) and MFI_195(D)“+10” implies the increasement with 10 cm3/g, STP from previous one.
| Sample | SAR | Composition | Stotal/(m2·g-1) | Vtotal/(mL·g-1) | Vmicro/(cm3·g-1) | Monolayer N2 adsorptionVm/(cm3·g-1, STP) |
|---|---|---|---|---|---|---|
| MFI_50 | 49.5 | Si94.1Al1.9O192Na1.9 | 328.47 | 0.2006 | 0.1235 | 75.467 |
| MFI_106 | 105.7 | Si95.1Al0.9O192Na0.9 | 393.66 | 0.2393 | 0.1726 | 90.445 |
| MFI_195 | 194.9 | Si95.51Al0.49O192Na0.49 | 415.42 | 0.2466 | 0.1761 | 95.444 |
| MFI_383 | 383.0 | Si95.75Al0.25O192Na0.25 | 429.05 | 0.1932 | 0.1796 | 98.576 |
| MFI_1713 | 1713.3 | Si95.944Al0.056O192Na0.056 | 451.09 | 0.1882 | 0.1791 | 103.64 |
Table 2 Chemical compositions and textural properties of MFI zeolite with various SARs*
| Sample | SAR | Composition | Stotal/(m2·g-1) | Vtotal/(mL·g-1) | Vmicro/(cm3·g-1) | Monolayer N2 adsorptionVm/(cm3·g-1, STP) |
|---|---|---|---|---|---|---|
| MFI_50 | 49.5 | Si94.1Al1.9O192Na1.9 | 328.47 | 0.2006 | 0.1235 | 75.467 |
| MFI_106 | 105.7 | Si95.1Al0.9O192Na0.9 | 393.66 | 0.2393 | 0.1726 | 90.445 |
| MFI_195 | 194.9 | Si95.51Al0.49O192Na0.49 | 415.42 | 0.2466 | 0.1761 | 95.444 |
| MFI_383 | 383.0 | Si95.75Al0.25O192Na0.25 | 429.05 | 0.1932 | 0.1796 | 98.576 |
| MFI_1713 | 1713.3 | Si95.944Al0.056O192Na0.056 | 451.09 | 0.1882 | 0.1791 | 103.64 |
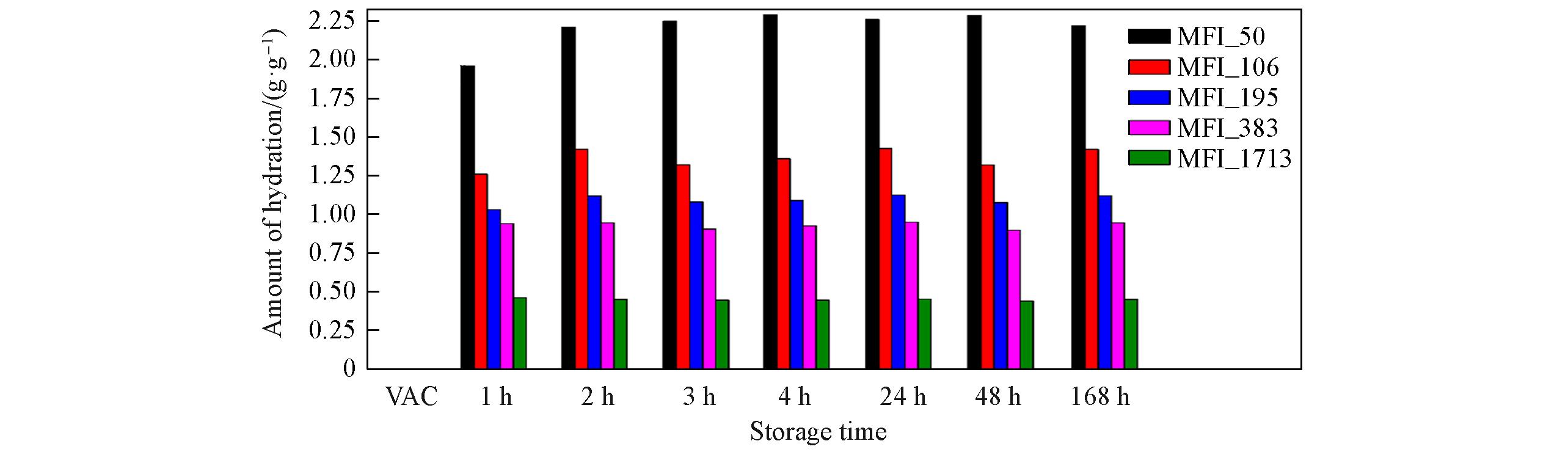
Fig.4 Amount of hydration of zeolitic AEM with various SARs after storage for different time at 25 ℃/50%RHVAC means storage in vacuum at 80 ℃ for 2 h.
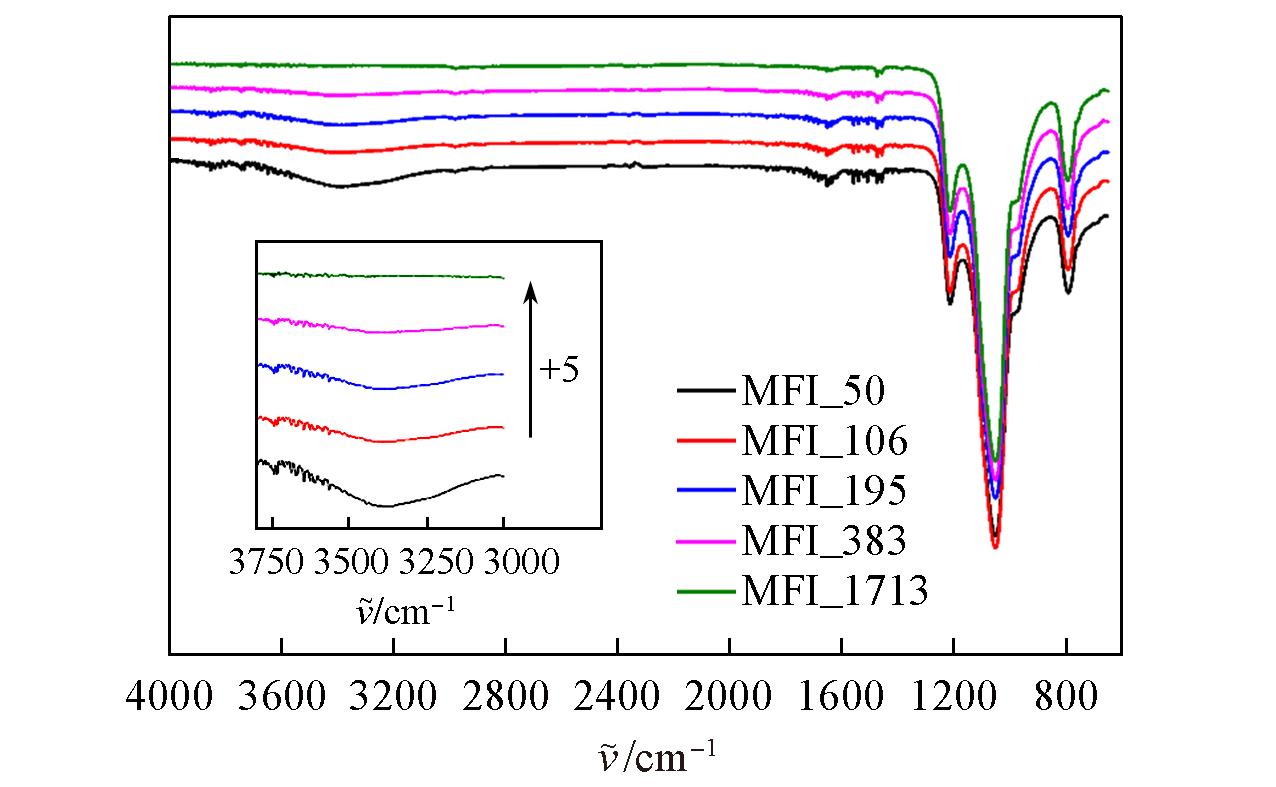
Fig.5 FTIR spectra of zeolitic AEMs with various SARs after storage at 25 ℃/50%RH for 168 hInset: zeolitic AEM’s transmittance between 3000 and 3800 cm-1; “+5” implies the increasement with 5% transmittance from previous one.
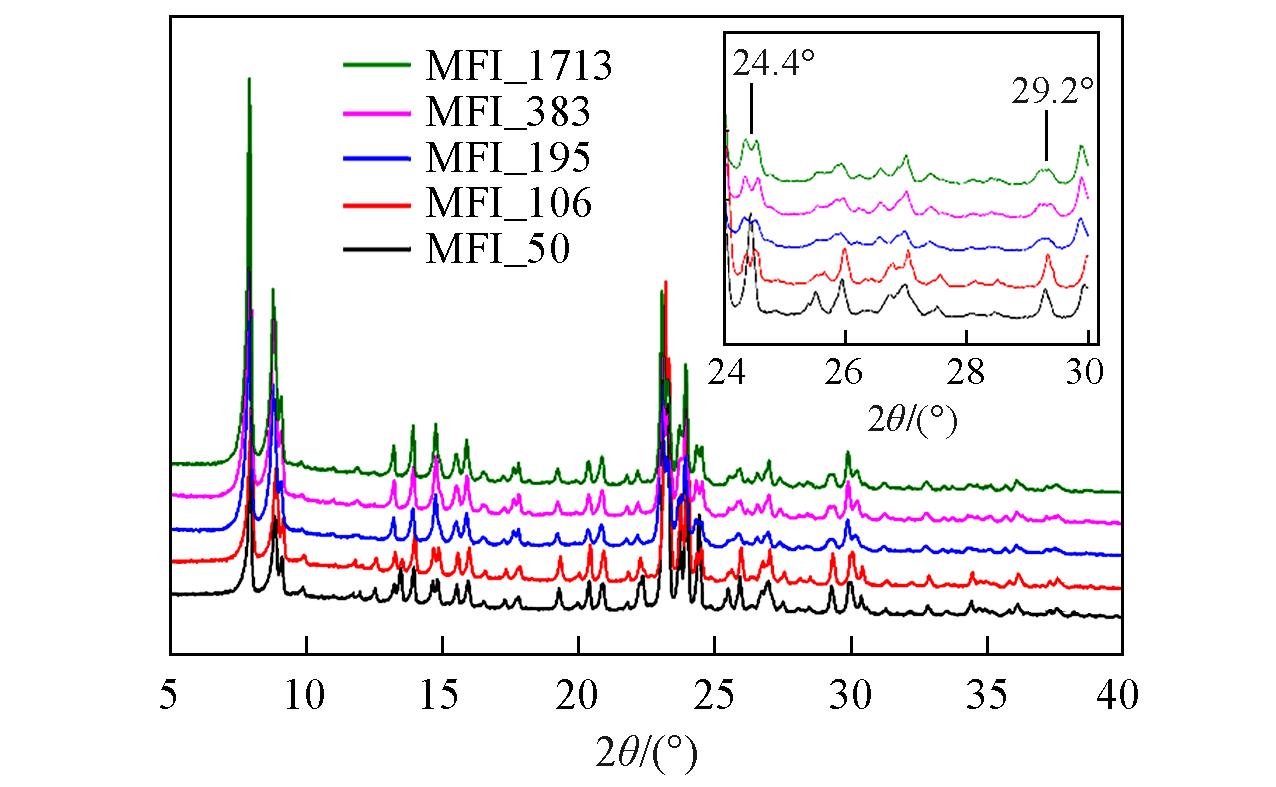
Fig.6 XRD patterns of zeolitic AEMs with various SARs after storage at 25 ℃/50%RH for 168 hInset: the evolution details of diffraction peaks between 24° and 30°.
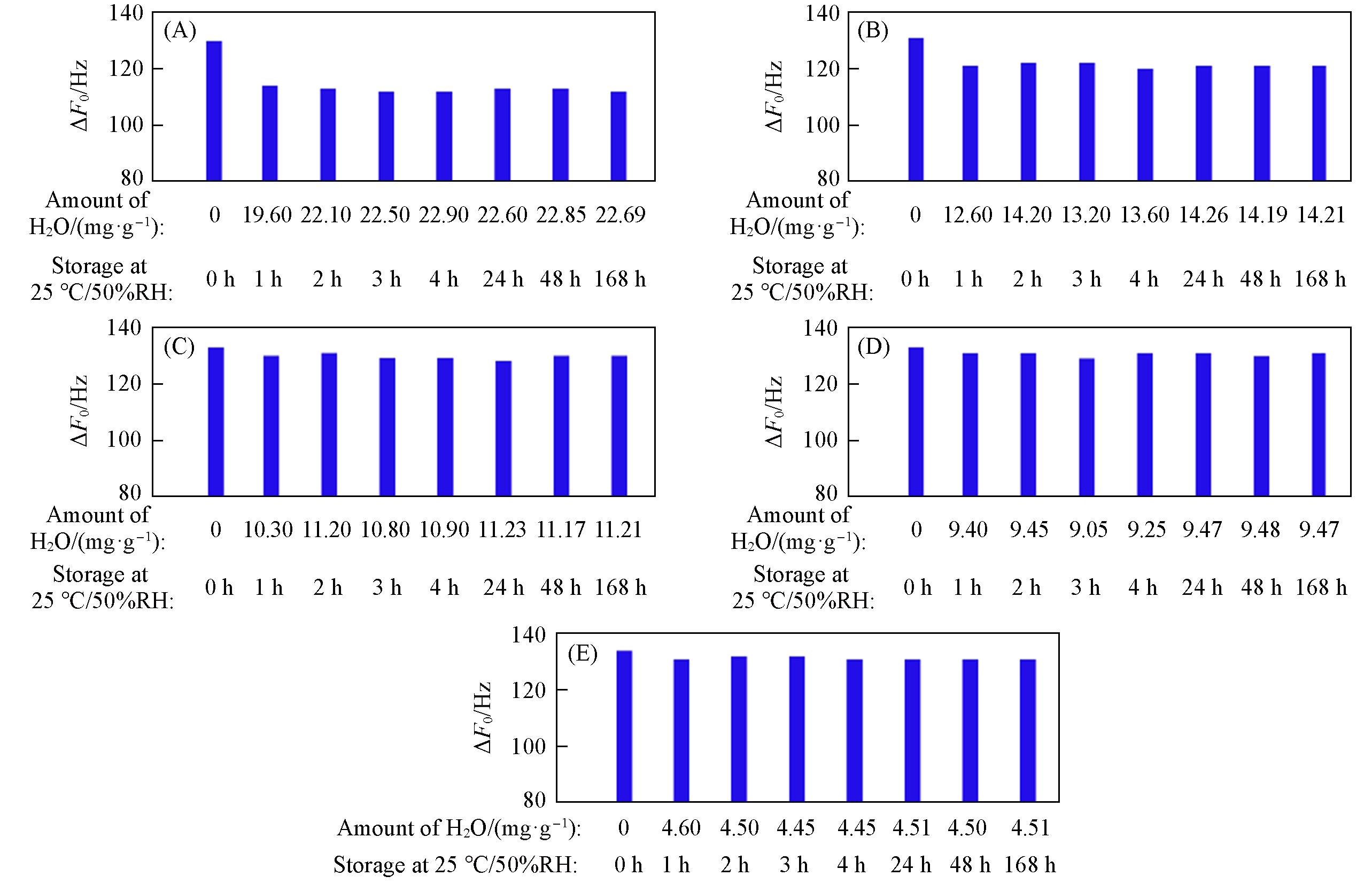
Fig.7 ΔF0 of zeolitic AEMs with various SARs stored for different time at 25 ℃/50%RH after storage in vacuum at 80 ℃ for 2 h(A) MFI_50; (B) MFI_106; (C) MFI_195; (D) MFI_383; (E) MFI_1713.
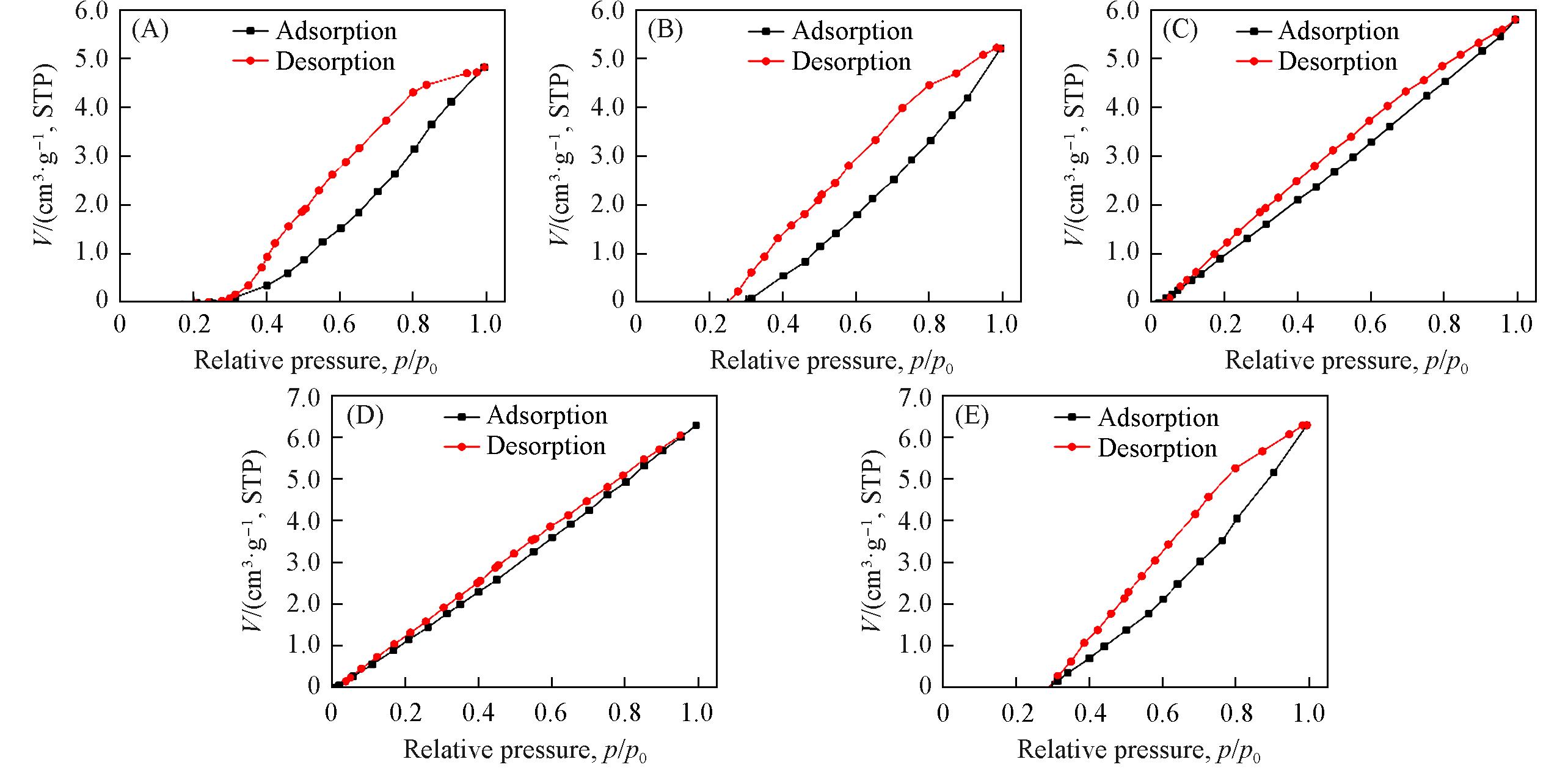
Fig.8 N2 adsorption⁃desorption isotherms(25 ℃) of zeolitic AEMs after 168 h storage at 25 ℃/50%RH(A) MFI_50; (B) MFI_106; (C) MFI_195; (D) MFI_383; (E) MFI_1713.
| Sample | SAR | Monolayer N2 adsorption Vm/(cm3·g-1, STP) |
|---|---|---|
| MFI_50 | 49.5 | 4.8152 |
| MFI_106 | 105.7 | 5.2029 |
| MFI_195 | 194.9 | 5.8008 |
| MFI_383 | 383.0 | 6.2827 |
| MFI_1713 | 1713.3 | 6.2804 |
Table 3 Monolayer N2 adsorption(25 ℃) of zeolitic AEMs after 168 h storage under 25 ℃/50%RH
| Sample | SAR | Monolayer N2 adsorption Vm/(cm3·g-1, STP) |
|---|---|---|
| MFI_50 | 49.5 | 4.8152 |
| MFI_106 | 105.7 | 5.2029 |
| MFI_195 | 194.9 | 5.8008 |
| MFI_383 | 383.0 | 6.2827 |
| MFI_1713 | 1713.3 | 6.2804 |
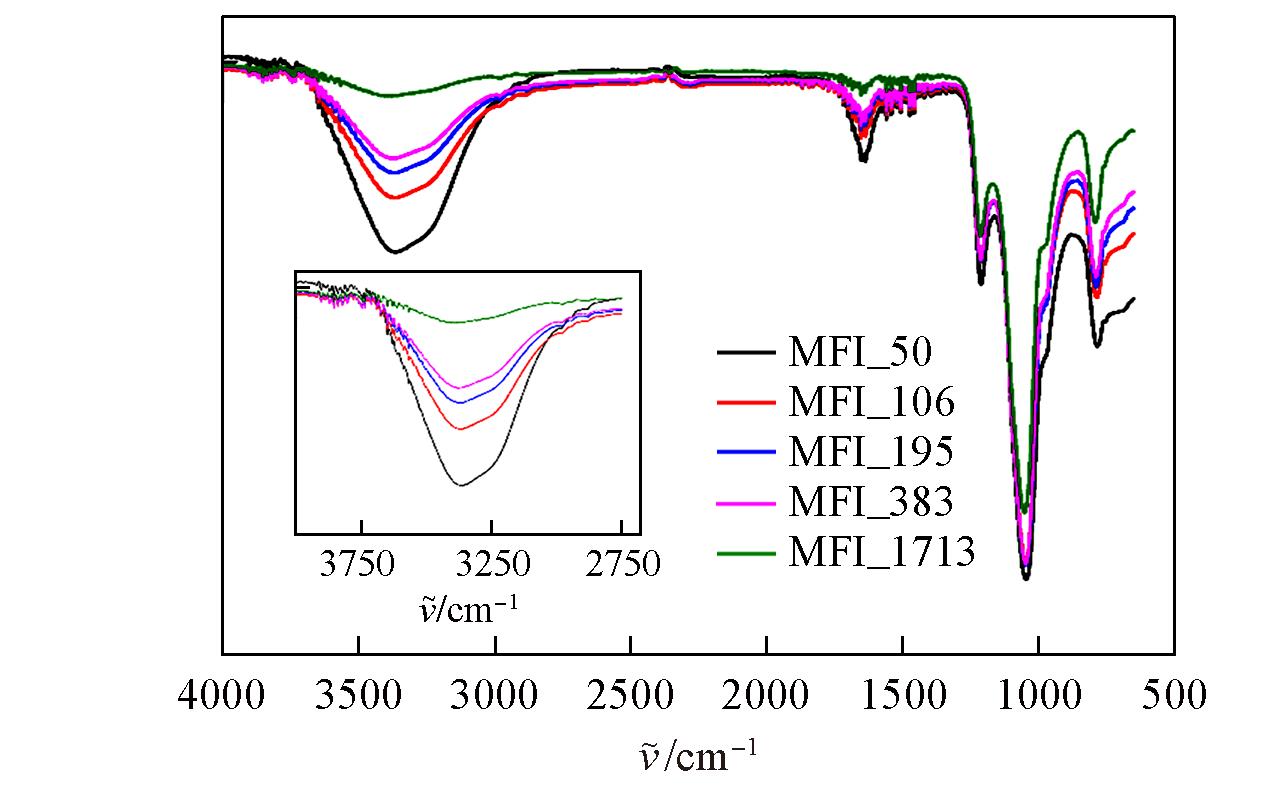
Fig.10 FTIR spectra of zeolitic AEMs with various SARs after 168 h storage at 25 ℃/95%RHInset: zeolitic AEM’s transmittance between 2750 and 4000 cm-1.
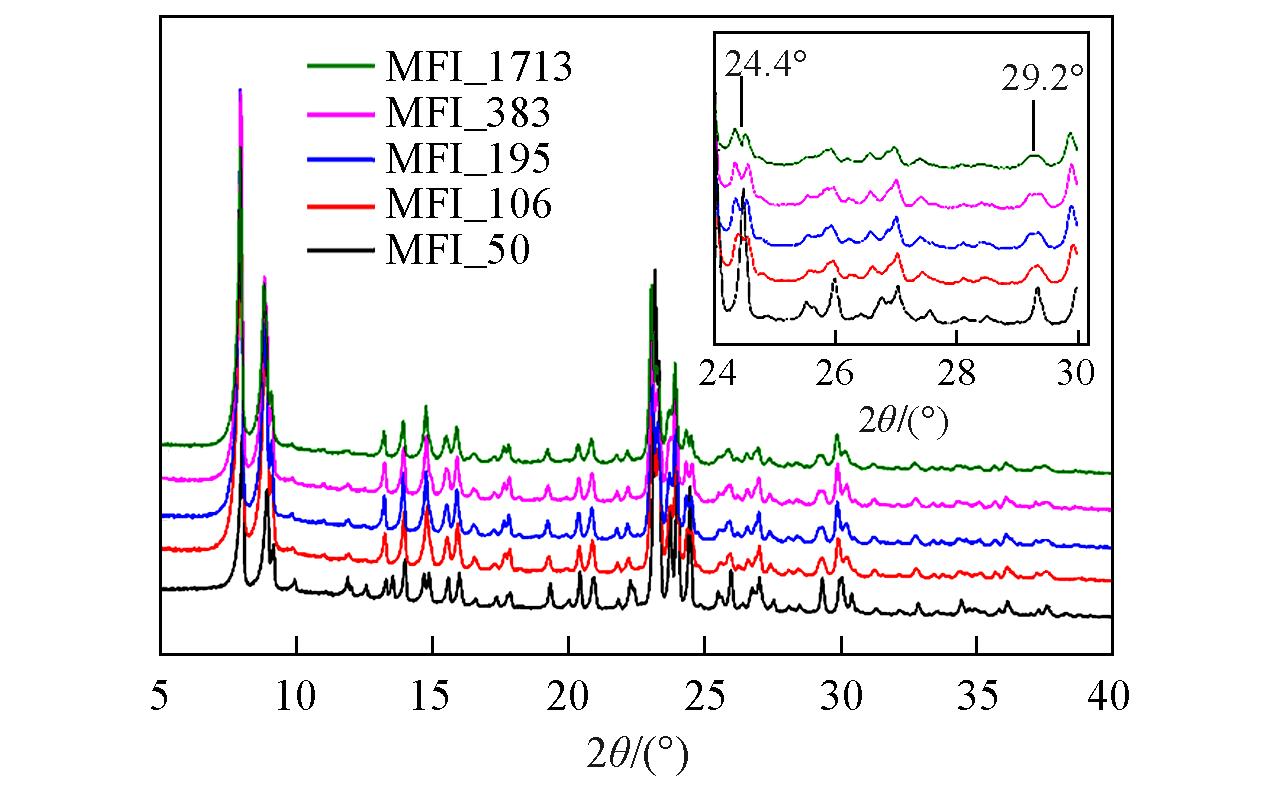
Fig.11 XRD patterns of zeolitic AEM with various SARs after 168 h storage at 25 ℃/95%RHInset: the evolution details of diffraction peaks between 24° and 30°.
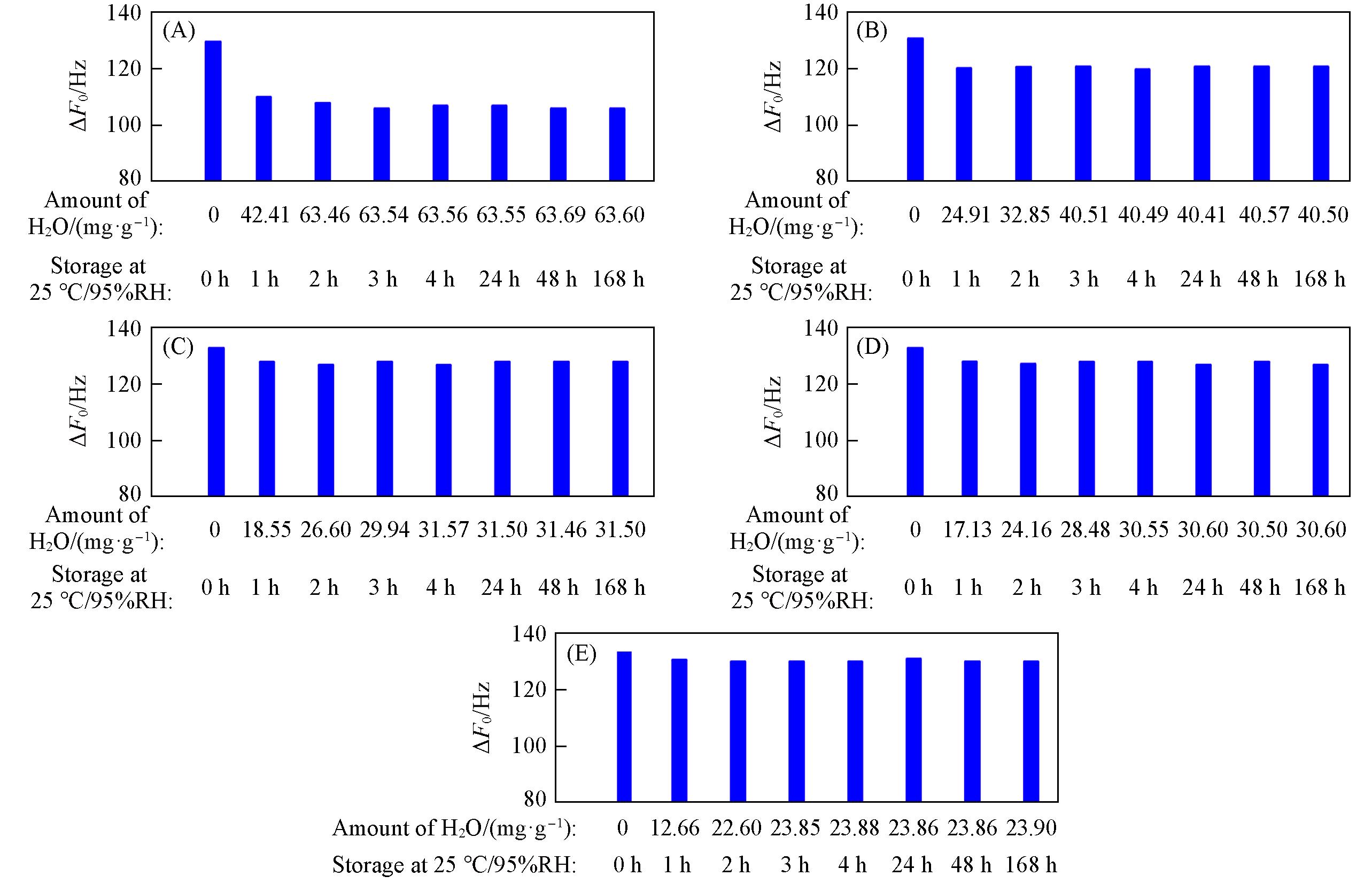
Fig.12 ΔF0 of zeolitic AEM with various SARs stored for different time at 25 ℃/95%RH after storage in vacuum at 80 ℃ for 2 h(A) MFI_50; (B) MFI_106; (C) MFI_195; (D) MFI_383; (E) MFI_1713.
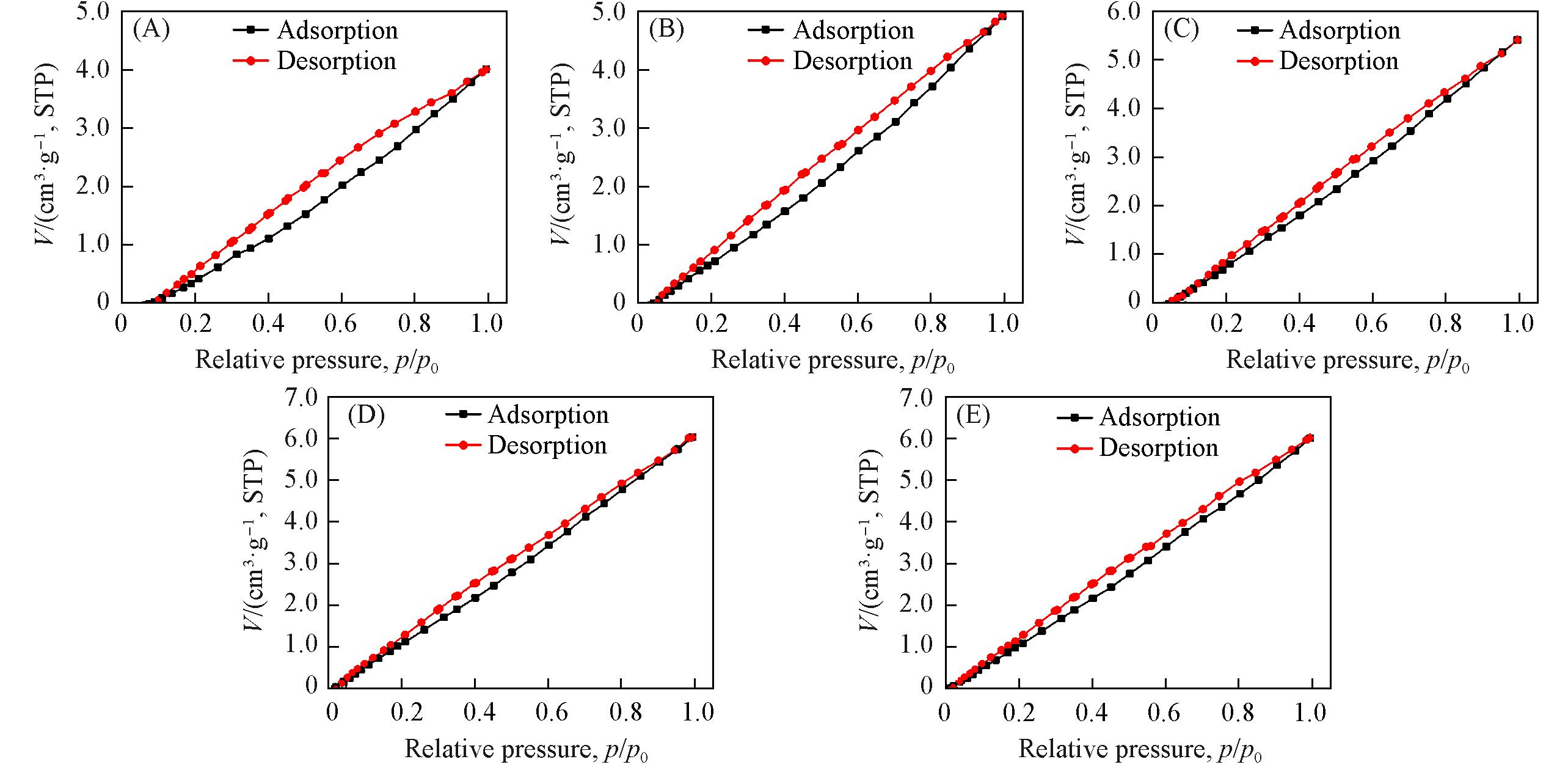
Fig.13 N2 adsorption⁃desorption isotherms(25 ℃) of zeolitic AEMs after 168 h storage at 25 ℃/95%RH(A) MFI_50; (B) MFI_106; (C) MFI_195; (D) MFI_383; (E) MFI_1713.
| Sample | SAR | Monolayer N2 adsorptionVm/(cm3·g-1, STP) | Sample | SAR | Monolayer N2 adsorptionVm/(cm3·g-1, STP) |
|---|---|---|---|---|---|
| MFI_50 | 49.5 | 4.0086 | MFI_383 | 383.0 | 6.0360 |
| MFI_106 | 105.7 | 4.9216 | MFI_1713 | 1713.3 | 6.0197 |
| MFI_195 | 194.9 | 5.4134 |
Table 4 Monolayer N2 adsorption(25 ℃) of zeolitic AEM after 168 h storage at 25 ℃/95%RH
| Sample | SAR | Monolayer N2 adsorptionVm/(cm3·g-1, STP) | Sample | SAR | Monolayer N2 adsorptionVm/(cm3·g-1, STP) |
|---|---|---|---|---|---|
| MFI_50 | 49.5 | 4.0086 | MFI_383 | 383.0 | 6.0360 |
| MFI_106 | 105.7 | 4.9216 | MFI_1713 | 1713.3 | 6.0197 |
| MFI_195 | 194.9 | 5.4134 |
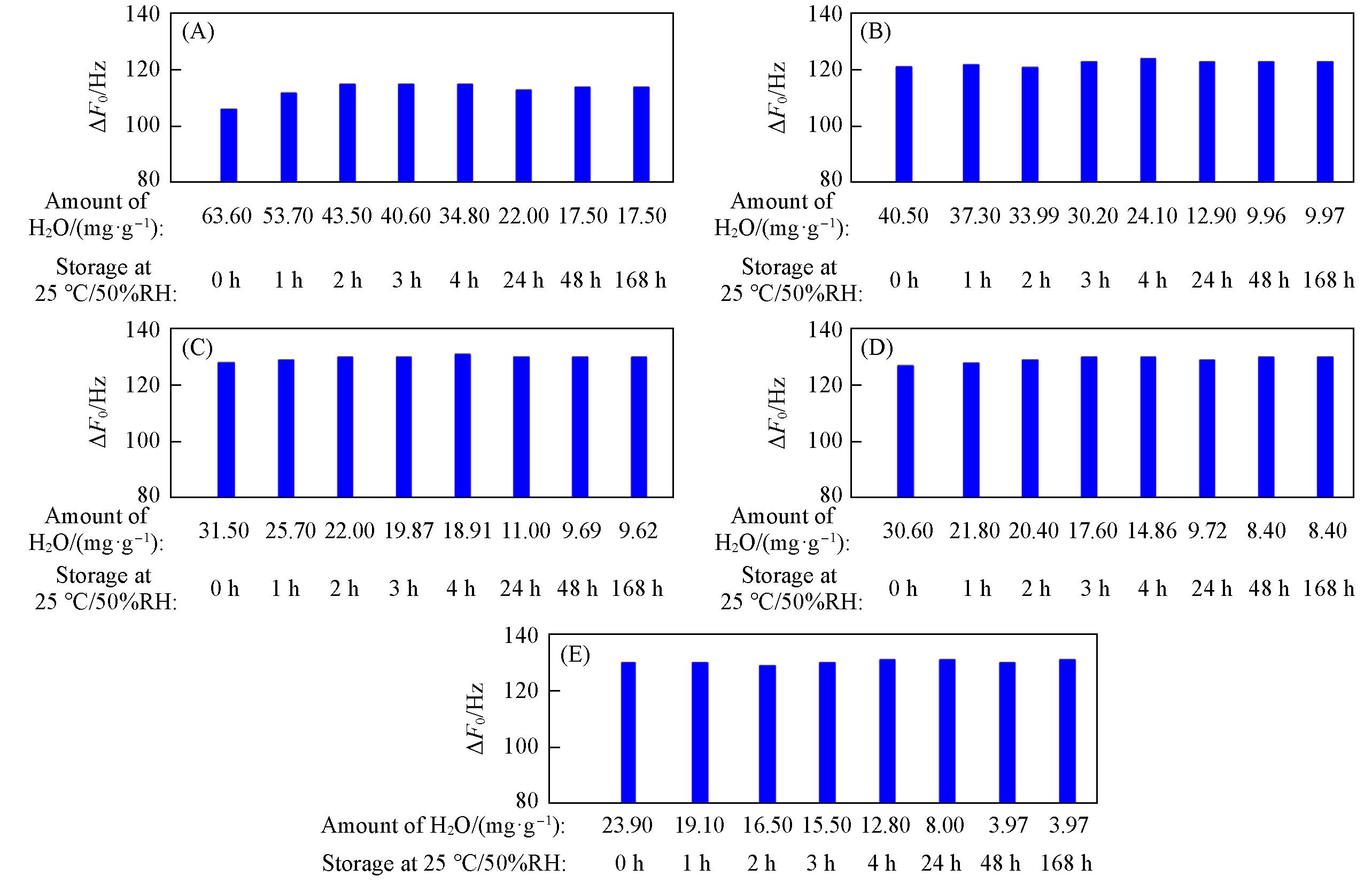
Fig.15 ΔF0 of zeolitic AEM with various SARs stored for different time at 25 ℃/50%RH after 168 h storage at 25 ℃/95%RH(A) MFI_50; (B) MFI_106; (C) MFI_195; (D) MFI_383; (E) MFI_1713.
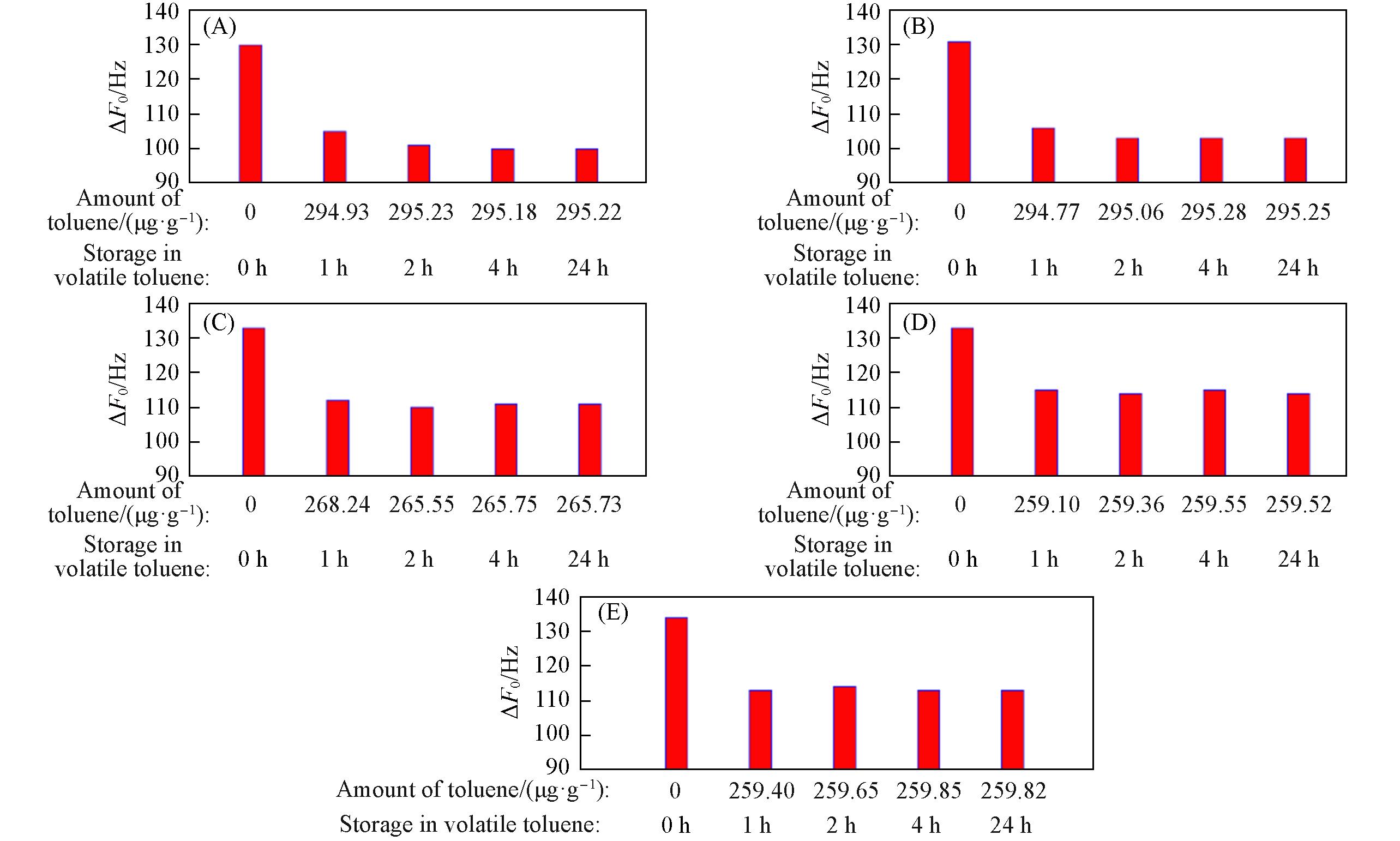
Fig.16 ΔF0 of zeolitic AEMs with various SARs stored for different time in volatile toluene after storage in vacuum at 80 ℃ for 2 h(A) MFI_50; (B) MFI_106; (C) MFI_195; (D) MFI_383; (E) MFI_1713.
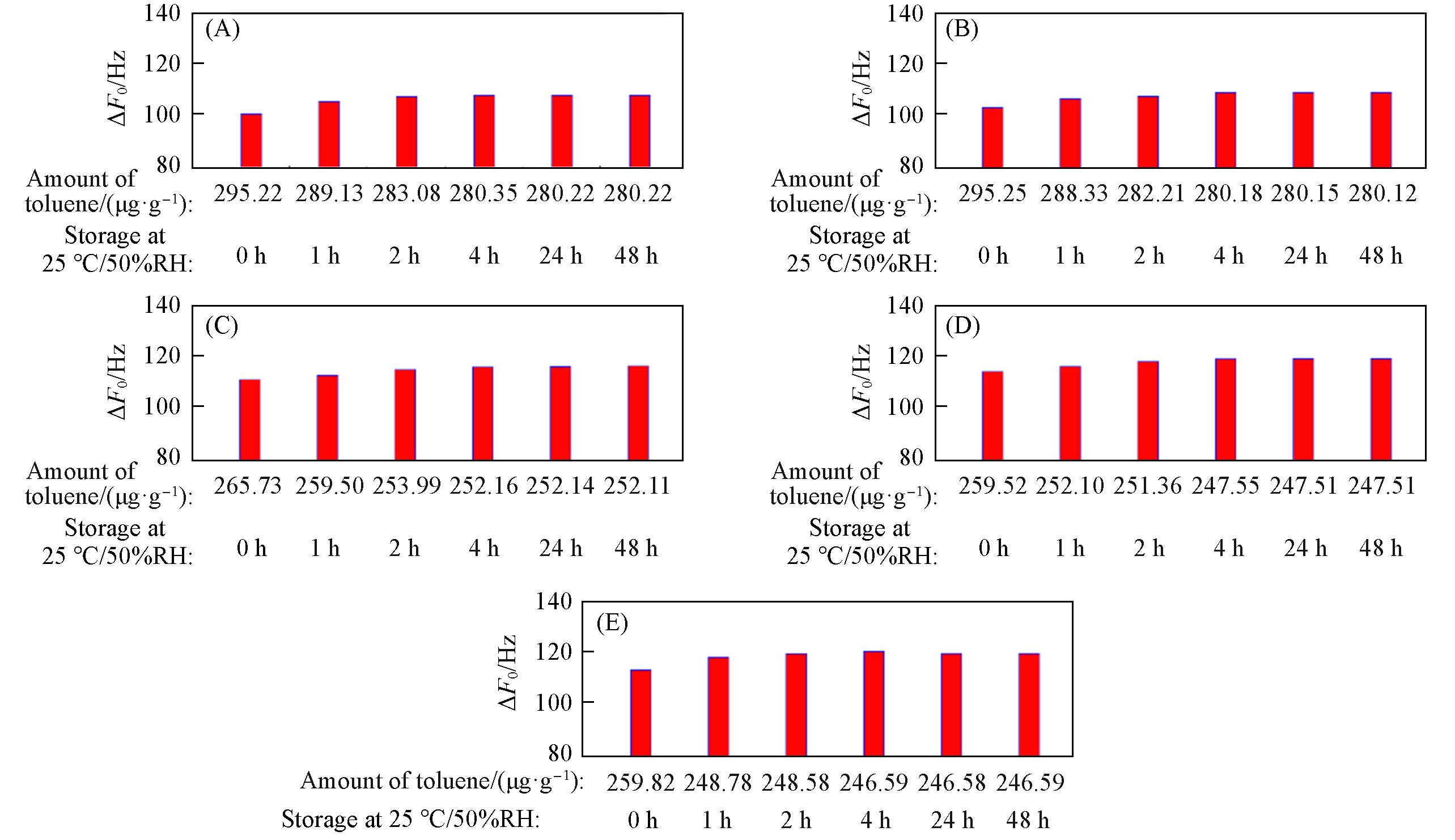
Fig.17 ΔF0 of zeolitic AEMs with various SARs stored for different time at 25 ℃/50%RH after 24 h storage in volatile toluene(A) MFI_50; (B) MFI_106; (C) MFI_195; (D) MFI_383; (E) MFI_1713.
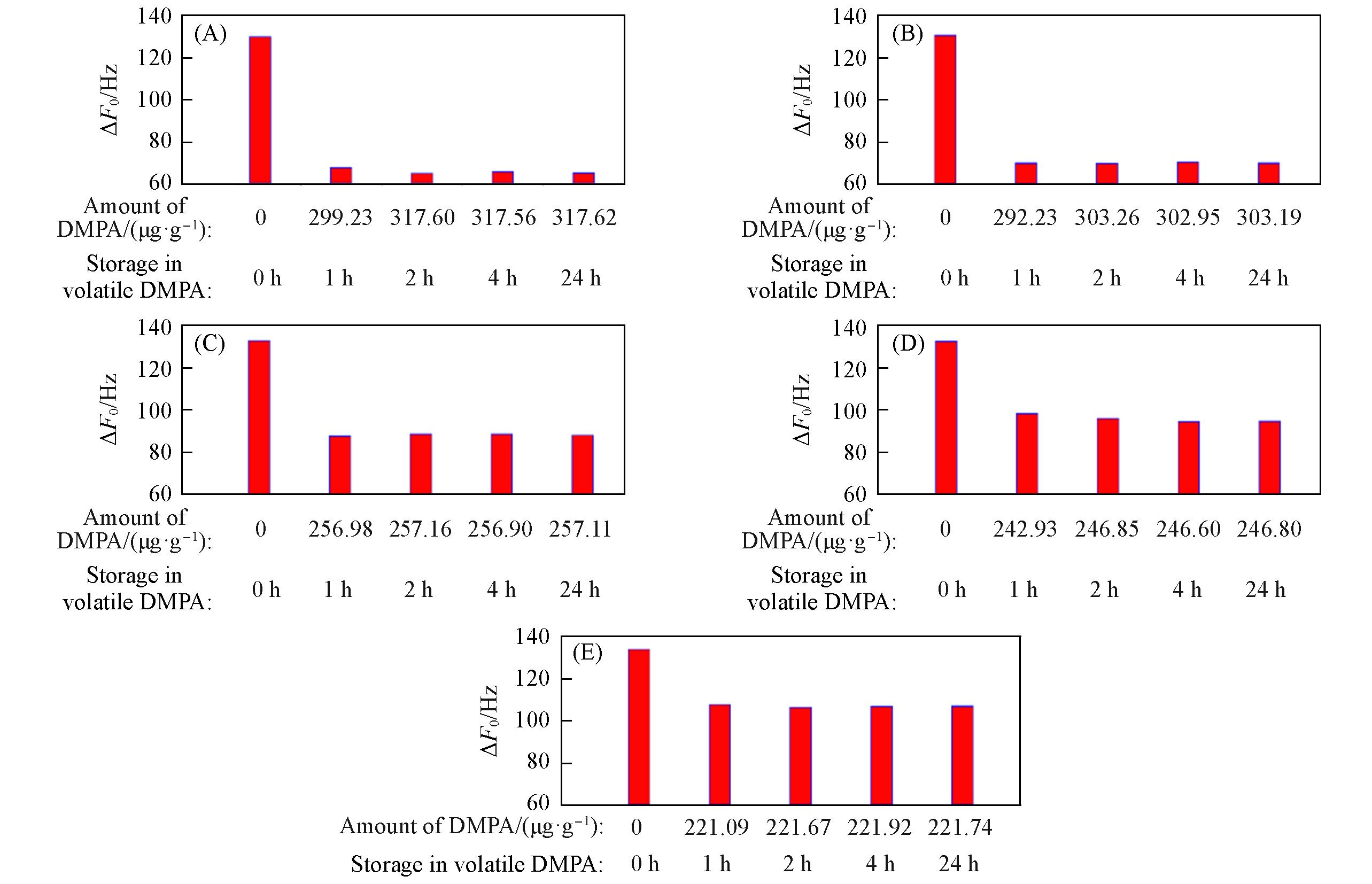
Fig.18 ΔF0 of zeolitic AEM with various SARs stored for different time in volatile DMPA after storage in vacuum at 80 ℃ for 2 h(A) MFI_50; (B) MFI_106; (C) MFI_195; (D) MFI_383; (E) MFI_1713.
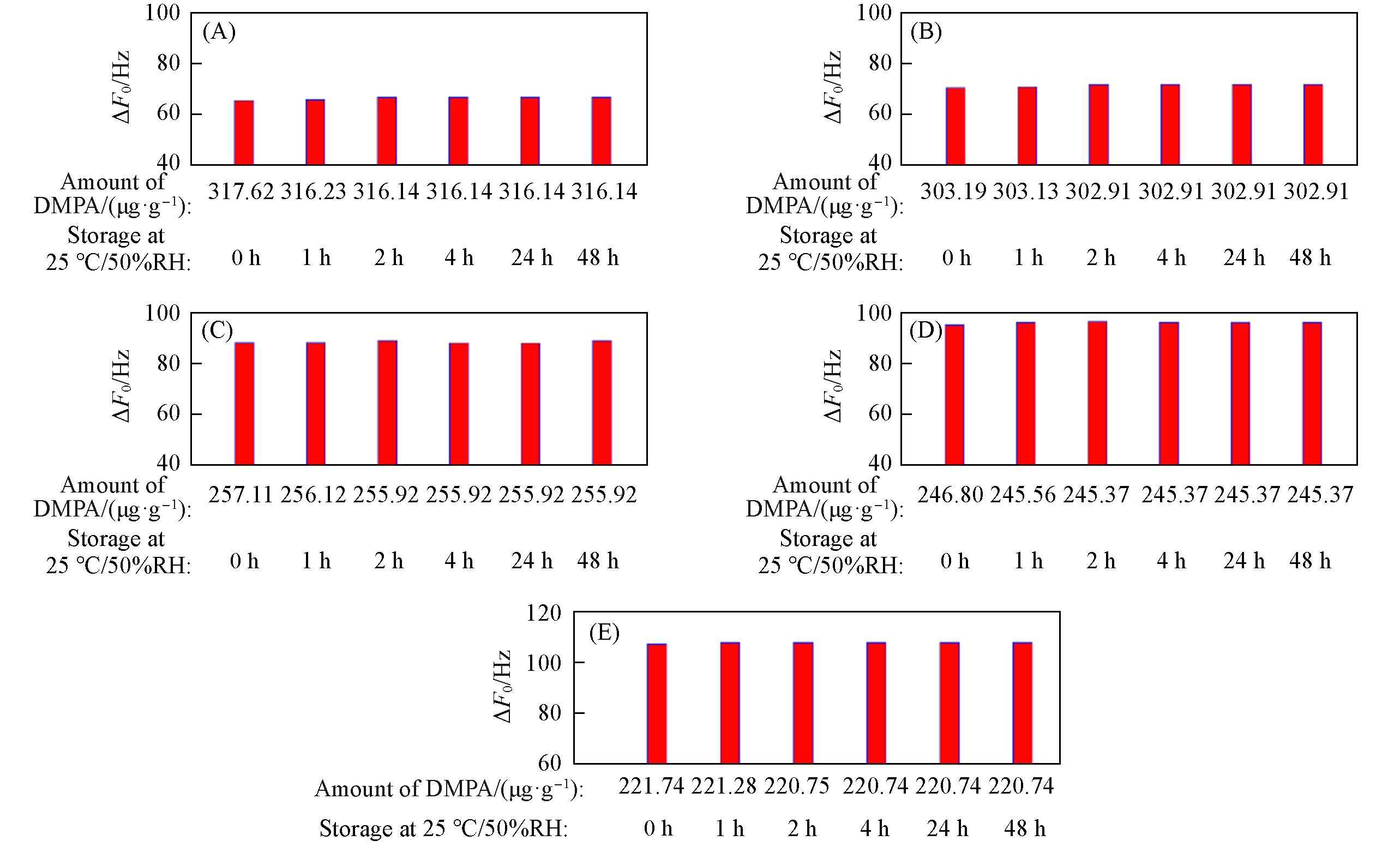
Fig.19 ΔF0 of zeolitic AEM with various SARs stored for different time at 25 ℃/50%RH after 24 h storage in volatile DMPA(A) MFI_50; (B) MFI_106; (C) MFI_195; (D) MFI_383; (E) MFI_1713.
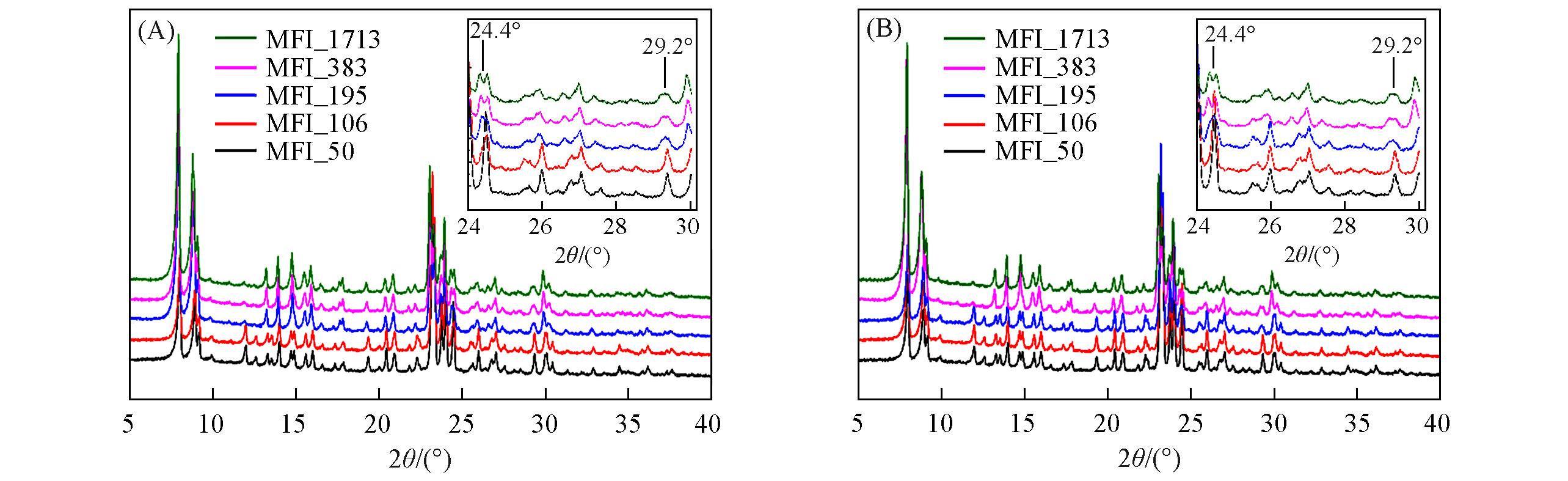
Fig.20 XRD patterns of zeolitic AEMs with various SARs after 24 h storage in volatile toluene(A) and DMPA(B)Insets: the evolution details of diffraction peaks between 24° and 30°.
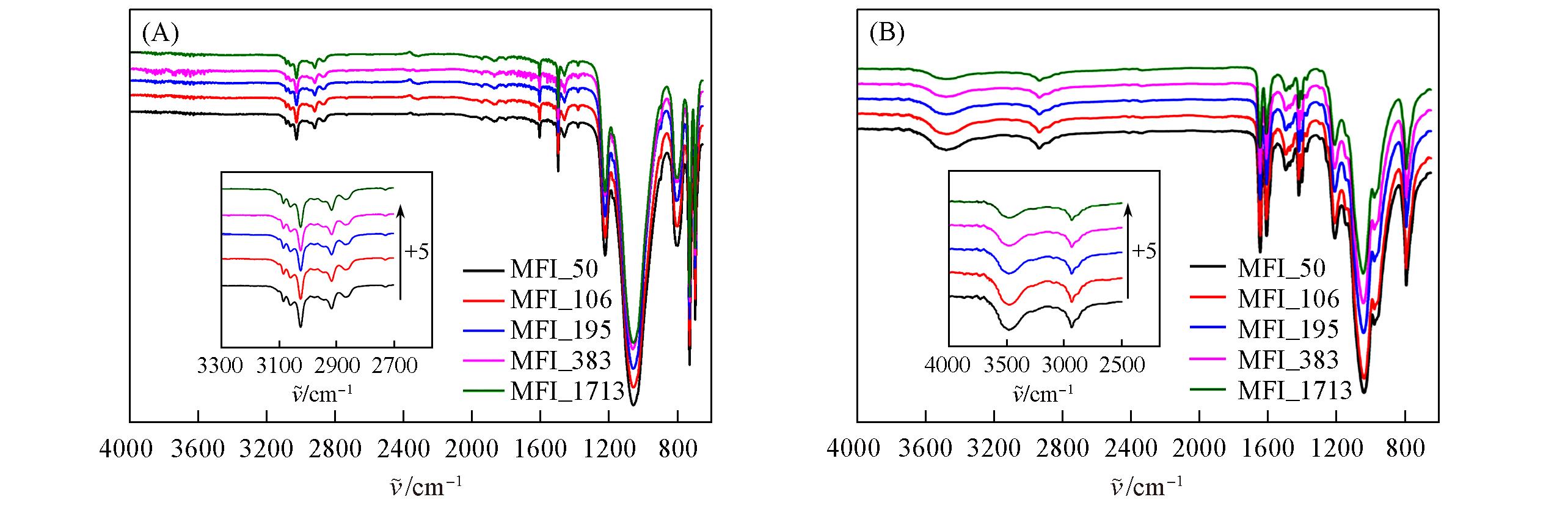
Fig.21 FTIR spectra of zeolitic AEMs with various SARs after 24 h storage in volatile toluene(A) and DMPA(B)Insets zeolitic AEMs’ transmittance between 2500 and 4000 cm-1. “+5” implies the increasement with 5% transmittance from previous one.
| 1 | Forest L., Gibiat V., Hooley A., J. Non⁃Cryst. Solids, 2001, 285(1—3), 230—235 |
| 2 | Tascan M., Vaughn E. A., Text. Res. J., 2008, 78(4), 289—296 |
| 3 | Tascan M., Vaughn E. A., J. Eng. Fibers Fabr., 2008, 3(2), 32—38 |
| 4 | del Rey R., Alba J., Arenas J. P., Sanchis V. J., Appl. Acoust., 2012, 73(6/7), 604—609 |
| 5 | Wrigh J. R., J. Audio Eng. Soc., 2003, 51(4), 244—247 |
| 6 | Venegas R., Umnova O., J. Acoust. Soc. Am., 2016, 140(2), 755—766 |
| 7 | Bechwati F., Avis M. R., Bull D. J., Cox T. J., Hargreaves J. A., Moser D., Ross D. K., Umnova O., Venegas R., J. Acoust. Soc. Am., 2012, 132(1), 239—248 |
| 8 | Zhang X. Y., Che S. A., Chem. Res. Chinese Universities, 2020, 36(5), 760—767 |
| 9 | Papakyriacou M., Kobler J., Sauer J., Loudspeaker System with Improved Sound, EP 2424270B1, 2010⁃08⁃23 |
| 10 | Jiang Y. W., Xu D. P., Kwon J. H., Jiang Z. X., Kim J. H., Hwang S. M., J. Mech. Sci. Technol., 2019, 33(8), 3679—3683 |
| 11 | Xu R. R., Pang W. Q., Yu J. J., Huo Q. S., Chen J. S., Chemistry of Zeolite and Related Porous Materials, Science Press, Beijing, 2004, 1—4 |
| 徐如人, 庞文琴, 于吉红, 霍启升, 陈接胜. 分子筛与多孔材料化学, 北京: 北京科学出版社, 2004, 1—4 | |
| 12 | Qi G. G., Meng X. J., Chem. J. Chinese Universities, 2023, 44(10), 20230227 |
| 戚刚刚, 孟祥举. 高等学校化学学报, 2023, 44(10), 20230227 | |
| 13 | Ardit M., Martucci A., Cruciani G., J. Phys. Chem. C, 2015, 119(13), 7351—7359 |
| 14 | Lempesis N., Smatsi N., Mavrantzas V. G., Pratsinis S. E., J. Phys. Chem. C, 2018, 122(11), 6217—6229 |
| 15 | Hay D. G., Jaeger H., J. Chem. Soc., Chem. Commun., 1984, 21, 1433 |
| 16 | Liu Z. Y., Zhang W. J., Lin B. X., Yu Q., Wei G. X., Zhu W., J. Fuel Chem. Technol., 1985, 13(2), 106—114 |
| 刘振义, 张婉静, 林炳雄, 于勤, 魏国祥, 朱伟. 燃料化学学报, 1985, 13(2), 106—114 | |
| 17 | Sethia G., Pillai R. S., Dangi G. P., Somani R. S., Bajaj H. C., Jasra R. V., Ind. Eng. Chem. Res., 2010, 49(5), 2353—2362 |
| 18 | Kabalan I., Michelin L., Rigolet S., Marichal C., Daou T. J., Lebeau B., Paillaud J. L., Solid State Sci., 2016, 58, 111—114 |
| 19 | Wu E. L., Lawton S. L.,Olson D. H., Rohrman A. C., Kokotailo G. T., J. Phys. Chem., 1979, 83(21), 2777—2781 |
| 20 | Baerlocher C., McCusker L. B., 2001, http://www.iza⁃structure.org/databases |
| 21 | Olson D. H., Kokotailo G. T., Lawton S. L., Meier W. M., J. Phys. Chem., 1981, 85(15), 2238—2243 |
| 22 | van Koningsveld H., Jansen J. C., van Bekkum H., Zeolites, 1990, 10(4), 235—242 |
| 23 | Chen N. Y., J. Phys. Chem., 1976, 80(1), 60—64 |
| 24 | Tsutsumi K., Mizoe K., Colloids and Surfaces, 1989, 37, 29—38 |
| 25 | Catuzo G. L., Santilli C. V., Martins L., Catal. Today, 2021, 381, 215—223 |
| 26 | Guo W. G., Xin Q., Zhang H., Liang J., Chin. J. Catal., 1981, 2(1), 36—41 |
| 郭文珪, 辛勤, 张慧, 梁娟. 催化学报, 1981, 2(1), 36—41 | |
| 27 | Ison A., Gorte R. J., J. Catal., 1984, 89(1), 150—158 |
| 28 | Miao C. L., Charaterization of Zeolite Catalyst by FT⁃IR, Dalian University of Technology, Dalian, 2010 |
| 苗翠兰. 用红外光谱法表征ZSM⁃5型沸石催化剂的研究, 大连: 大连理工大学, 2010 | |
| 29 | Isernia L. F., Mater. Res., 2013, 16(4), 792—802 |
| 30 | Ohlin L., Bazin P., Thibault⁃Starzyk F., Hedlund J., Grahn M., J. Phys. Chem. C, 2013, 117(33), 16972—16982 |
| 31 | Huang H. F., Rong W. J., Gu Y. Y., Chang R. Q., Lu H. F., Acta Scientiae Cicumstantiae, 2014, 34(12), 3144—3151 |
| 黄海凤, 戎文娟, 顾勇义, 常仁芹, 卢晗锋. 环境科学学报, 2014, 34(12), 3144—3151 | |
| 32 | Flanigen E. M., Bennett J. M., Grose R. W., Cohen J. P., Patton R. L., Kirchner R. M., Smith J. V., Nature, 1978, 271(5645), 512—516 |
| 33 | Kokotailo G. T., Lawton S. L., Olson D. H., Meier W. M., Nature, 1978, 272(5652), 437—438 |
| 34 | Zhu J. H., Xu Y., Wang Y., Zhou S. L., Zhou C. F., Acta Physico⁃Chimica Sinica, 2004, 20(S1), 946—966 |
| 朱建华, 徐杨, 王英, 周仕禄, 周春芳. 物理化学学报, 2004, 20(S1), 946—966 | |
| 35 | Wu E. L., Lawton S. L., Olson D. H., Rohrman A. C., Kokotailo G. T., J. Phys. Chem., 1979, 83(21), 2777—2781 |
| 36 | Mentzen B. F., Gelin P., Mater. Res. Bull., 1995, 30(3), 373—380 |
| 37 | van Koningsveld H., Jansen J. C., Microporous Mater., 1996, 6(3), 159—167 |
| [1] | 杨颖楠, 孙启明. EAB分子筛的合成及在甲醇制烯烃反应中的应用[J]. 高等学校化学学报, 2023, 44(8): 20230119. |
| [2] | 熊文朋, 褚月英, 王强, 徐君, 邓风. 丙酮羰基13C化学位移表征分子筛限域效应的理论计算[J]. 高等学校化学学报, 2023, 44(8): 20230063. |
| [3] | 徐德义, 丁超俊, 李芳, 刘月明, 何鸣元. 磷改性失活TS-1高效催化C5=裂解制备C2=/C3=反应的研究[J]. 高等学校化学学报, 2023, 44(8): 20230094. |
| [4] | 赵晨, 谭涓, 韩京京, 刘瑞, 冯春硕, 刘靖. ZSM-22分子筛的形貌控制合成及催化长链正构烷烃制生物航空煤油性能[J]. 高等学校化学学报, 2023, 44(11): 20230208. |
| [5] | 戚刚刚, 孟祥举. 中国沸石分子筛基础研究近年来的突破性进展[J]. 高等学校化学学报, 2023, 44(10): 20230227. |
| [6] | 谢小兰, 刘湛, 吕佳敏, 余申, 李小云, 苏宝连, 陈丽华. Cu基等级孔ZSM-5分子筛单晶的制备及硝基苯加氢催化性能[J]. 高等学校化学学报, 2023, 44(10): 20230109. |
| [7] | 姚伊婷, 吕佳敏, 余申, 刘湛, 李昱, 李小云, 苏宝连, 陈丽华. 等级孔微孔-介孔Fe2O3/ZSM-5中空分子筛催化材料的制备及催化苄基化性能[J]. 高等学校化学学报, 2022, 43(8): 20220090. |
| [8] | 陈玮琴, 吕佳敏, 余申, 刘湛, 李小云, 陈丽华, 苏宝连. 有机杂化介孔Beta分子筛的合成及在苯甲醇烷基化反应中的应用[J]. 高等学校化学学报, 2022, 43(6): 20220086. |
| [9] | 李志光, 齐国栋, 徐君, 邓风. Sn-Al-β分子筛酸性在葡萄糖转化反应中作用的固体NMR研究[J]. 高等学校化学学报, 2022, 43(6): 20220138. |
| [10] | 李加富, 张凯, 王宁, 孙启明. 分子筛限域单原子金属催化剂的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220032. |
| [11] | 孟祥龙, 杨歌, 郭海玲, 刘晨光, 柴永明, 王纯正, 郭永梅. 纳米分子筛的合成及硫化氢吸附性能[J]. 高等学校化学学报, 2022, 43(3): 20210687. |
| [12] | 魏李娜, 彭莉, 朱锋, 顾鹏飞, 顾学红. 中空纤维Au-CeZr/FAU催化膜的制备及在富氢气氛CO选择性氧化反应中的应用[J]. 高等学校化学学报, 2022, 43(10): 20220175. |
| [13] | 李海勃, 肖长发, 江龙, 黄云, 淡宜. MCM-41分子筛负载氯化铝催化丙烯酸甲酯与1-辛烯共聚[J]. 高等学校化学学报, 2021, 42(9): 2974. |
| [14] | 李奕川, 朱国富, 王宇, 柴永明, 刘晨光, 何盛宝. 基底表面性质与前驱液化学环境对原位定向构筑钛硅分子筛膜的影响[J]. 高等学校化学学报, 2021, 42(9): 2934. |
| [15] | 罗强强, 金少青, 孙洪敏, 杨为民. 液相酸溶液后补钛合成Ti-MWW分子筛[J]. 高等学校化学学报, 2021, 42(9): 2742. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
