

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (1): 40.doi: 10.7503/cjcu20200362
所属专题: 分子筛功能材料 2021年,42卷,第1期
王彬宇1, 李莉1, 李菁2, 靳科研1, 张少卿1, 张佳楠3, 闫文付1( )
)
收稿日期:2020-06-18
出版日期:2021-01-10
发布日期:2021-01-12
通讯作者:
闫文付
E-mail:yanw@jlu.edu.cn
基金资助:
WANG Binyu1, LI Li1, LI Jing2, JIN Keyan1, ZHANG Shaoqing1, ZHANG Jianan3, YAN Wenfu1( )
)
Received:2020-06-18
Online:2021-01-10
Published:2021-01-12
Contact:
YAN Wenfu
E-mail:yanw@jlu.edu.cn
Supported by:摘要:
系统总结了以工业固体废料为原料合成沸石分子筛材料的最新研究进展, 讨论了以粉煤灰、 珍珠岩工业废料、 煤矸石、 流体催化裂化(FCC)废催化剂、 锂矿渣、 铝土矿渣、 废瓷料和废弃玻璃等工业固体废料为原料, 合成LTA, FAU, MFI, CHA, GIS, SOD, ANA和KFI沸石分子筛材料的工艺方法, 及其在污水中重金属离子的脱除、 空气中CO2的捕获、 氮氧化物的选择性还原等实际应用中的性能, 并对未来工业固体废料合成沸石分子筛的发展趋势进行了展望.
中图分类号:
TrendMD:
王彬宇, 李莉, 李菁, 靳科研, 张少卿, 张佳楠, 闫文付. 用工业固体废料合成沸石分子筛的研究进展. 高等学校化学学报, 2021, 42(1): 40.
WANG Binyu, LI Li, LI Jing, JIN Keyan, ZHANG Shaoqing, ZHANG Jianan, YAN Wenfu. Recent Progresses on the Synthesis of Zeolites from the Industrial Solid Wastes. Chem. J. Chinese Universities, 2021, 42(1): 40.
| Raw material | Elemental composition(%, mass fraction) | Crystal phase | Ref. | |||||
|---|---|---|---|---|---|---|---|---|
| Fly ash | Al2O3 | SiO2 | CaO | Fe2O3 | K2O | MgO | Mullite, quartz | [ |
| (40.26) | (48.97) | (2.29) | (3.07) | (0.49) | (0.4) | |||
| MnO | Na2O | P2O5 | SO3 | TiO2 | LOI | |||
| (0.02) | (0.11) | (0.3) | (0.27) | (1.52) | (2.26) | |||
| Perlite industrial waste | O | Si | Al | K | Na | Fe | Mica, quartz, feldspars | [ |
| (71.79) | (19.60) | (4.24) | (2.17) | (1.26) | (0.40) | |||
| Ca | Mg | |||||||
| (0.39) | (0.17) | |||||||
| Coal gangue | SiO2 | TiO2 | Al2O3 | Fe2O3 | MgO | CaO | Kaolinite | [ |
| (33.50) | (0.74) | (27.96) | (1.68) | (0.48) | (1.11) | |||
| Na2O | K2O | H2O | P2O5 | MnO | LOI | |||
| (0.17) | (0.35) | (0.43) | (0.079) | (0.02) | (34.24) | |||
| Spent FCC catalyst | SiO2 | Al2O3 | NaO2 | Fe2O3 | TiO2 | La2O3 | FAU(Y) | [ |
| (49.5) | (45.1) | (1.4) | (1.7) | (1.0) | (0.5) | |||
| P2O5 | Other | |||||||
| (0.5) | (0.2) | |||||||
| Lithium slag | SiO2 | Al2O3 | SO3 | CaO | Fe2O3 | K2O | Quartz, spodumene | [ |
| (71.73) | (25.16) | (1.58) | (0.21) | (0.58) | (0.38) | |||
| Na2O | TiO2 | |||||||
| (0.06) | (0.03) | |||||||
| Bauxite residue | Na2O | MgO | Al2O3 | SiO2 | P2O5 | K2O | Gibbsite, calcite, hematite | [ |
| (4.03) | (0.21) | (11.46) | (7.89) | (0.09) | (0.45) | |||
| CaO | TiO2 | MnO | Fe2O | |||||
| (3.53) | (4.82) | (0.21) | (36.8) | |||||
| Waste porcelain | SiO2 | Al2O3 | Na2O | K2O | Fe2O3 | CaO | Mullite, quartz, trydimite | [ |
| (65.1) | (23.0) | (4.40) | (1.89) | (1.80) | (1.26) | |||
| TiO2 | ZrO2 | MgO | P2O5 | ZnO | BaO | |||
| (0.88) | (0.87) | (0.133) | (0.096) | (0.227) | (0.133) | |||
| Waste glass | SiO2 | CaO | Fe2O3 | Na2O | MgO | Bi2O3 | Amorphous | [ |
| (66.1) | (24.9) | (2.4) | (1.7) | (1.2) | (0.5) | |||
| Al2O3 | K2O | TiO2 | ||||||
| (1.1) | (0.9) | (0.3) | ||||||
Table 1 Composition and crystal phase of industrial solid waste
| Raw material | Elemental composition(%, mass fraction) | Crystal phase | Ref. | |||||
|---|---|---|---|---|---|---|---|---|
| Fly ash | Al2O3 | SiO2 | CaO | Fe2O3 | K2O | MgO | Mullite, quartz | [ |
| (40.26) | (48.97) | (2.29) | (3.07) | (0.49) | (0.4) | |||
| MnO | Na2O | P2O5 | SO3 | TiO2 | LOI | |||
| (0.02) | (0.11) | (0.3) | (0.27) | (1.52) | (2.26) | |||
| Perlite industrial waste | O | Si | Al | K | Na | Fe | Mica, quartz, feldspars | [ |
| (71.79) | (19.60) | (4.24) | (2.17) | (1.26) | (0.40) | |||
| Ca | Mg | |||||||
| (0.39) | (0.17) | |||||||
| Coal gangue | SiO2 | TiO2 | Al2O3 | Fe2O3 | MgO | CaO | Kaolinite | [ |
| (33.50) | (0.74) | (27.96) | (1.68) | (0.48) | (1.11) | |||
| Na2O | K2O | H2O | P2O5 | MnO | LOI | |||
| (0.17) | (0.35) | (0.43) | (0.079) | (0.02) | (34.24) | |||
| Spent FCC catalyst | SiO2 | Al2O3 | NaO2 | Fe2O3 | TiO2 | La2O3 | FAU(Y) | [ |
| (49.5) | (45.1) | (1.4) | (1.7) | (1.0) | (0.5) | |||
| P2O5 | Other | |||||||
| (0.5) | (0.2) | |||||||
| Lithium slag | SiO2 | Al2O3 | SO3 | CaO | Fe2O3 | K2O | Quartz, spodumene | [ |
| (71.73) | (25.16) | (1.58) | (0.21) | (0.58) | (0.38) | |||
| Na2O | TiO2 | |||||||
| (0.06) | (0.03) | |||||||
| Bauxite residue | Na2O | MgO | Al2O3 | SiO2 | P2O5 | K2O | Gibbsite, calcite, hematite | [ |
| (4.03) | (0.21) | (11.46) | (7.89) | (0.09) | (0.45) | |||
| CaO | TiO2 | MnO | Fe2O | |||||
| (3.53) | (4.82) | (0.21) | (36.8) | |||||
| Waste porcelain | SiO2 | Al2O3 | Na2O | K2O | Fe2O3 | CaO | Mullite, quartz, trydimite | [ |
| (65.1) | (23.0) | (4.40) | (1.89) | (1.80) | (1.26) | |||
| TiO2 | ZrO2 | MgO | P2O5 | ZnO | BaO | |||
| (0.88) | (0.87) | (0.133) | (0.096) | (0.227) | (0.133) | |||
| Waste glass | SiO2 | CaO | Fe2O3 | Na2O | MgO | Bi2O3 | Amorphous | [ |
| (66.1) | (24.9) | (2.4) | (1.7) | (1.2) | (0.5) | |||
| Al2O3 | K2O | TiO2 | ||||||
| (1.1) | (0.9) | (0.3) | ||||||
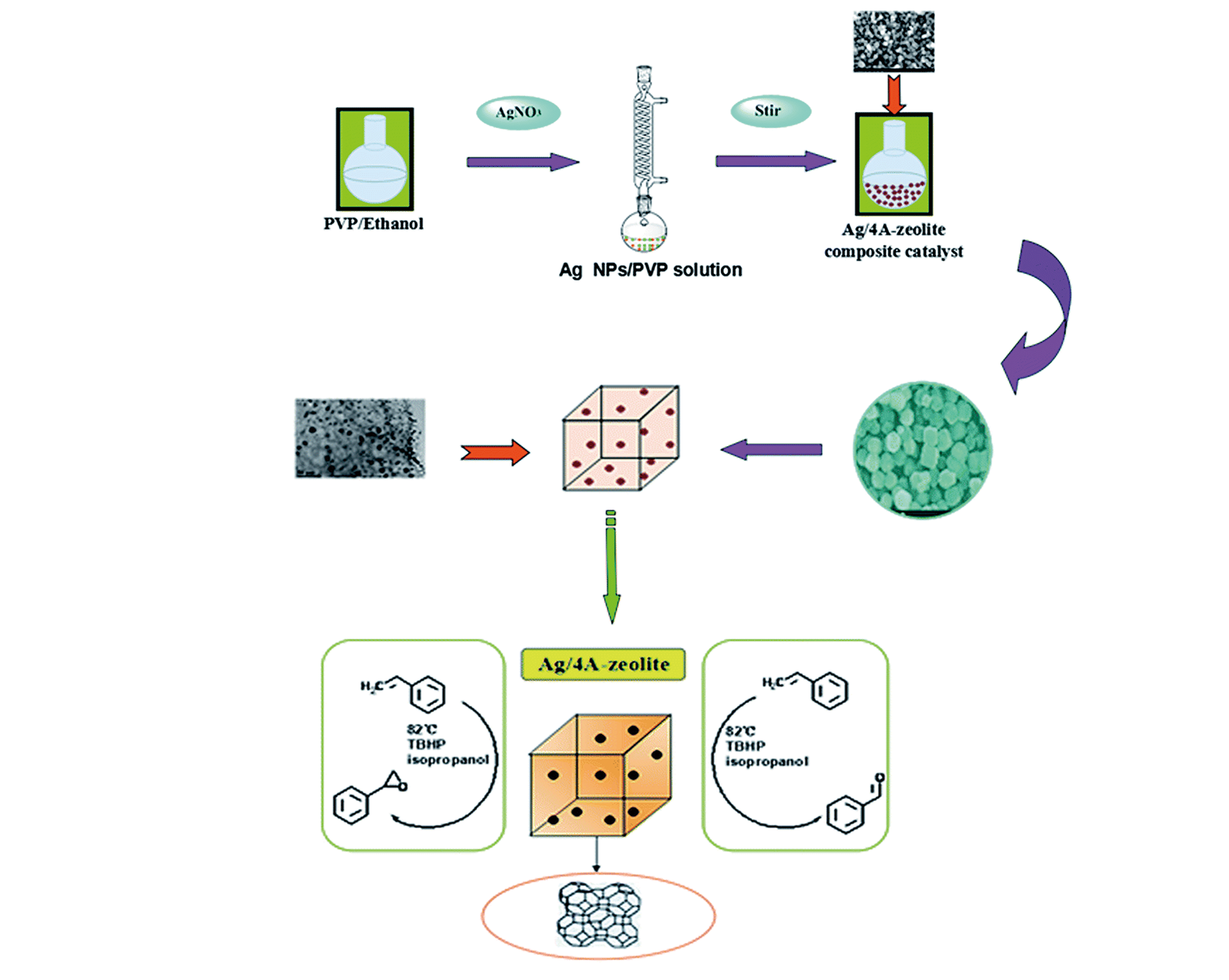
Fig.3 Scheme showing preparation of the Ag NPs/4A?zeolite composite catalyst and the chemical equation of styrene epoxidation[59]Copyright 2015, Royal Society of Chemistry.
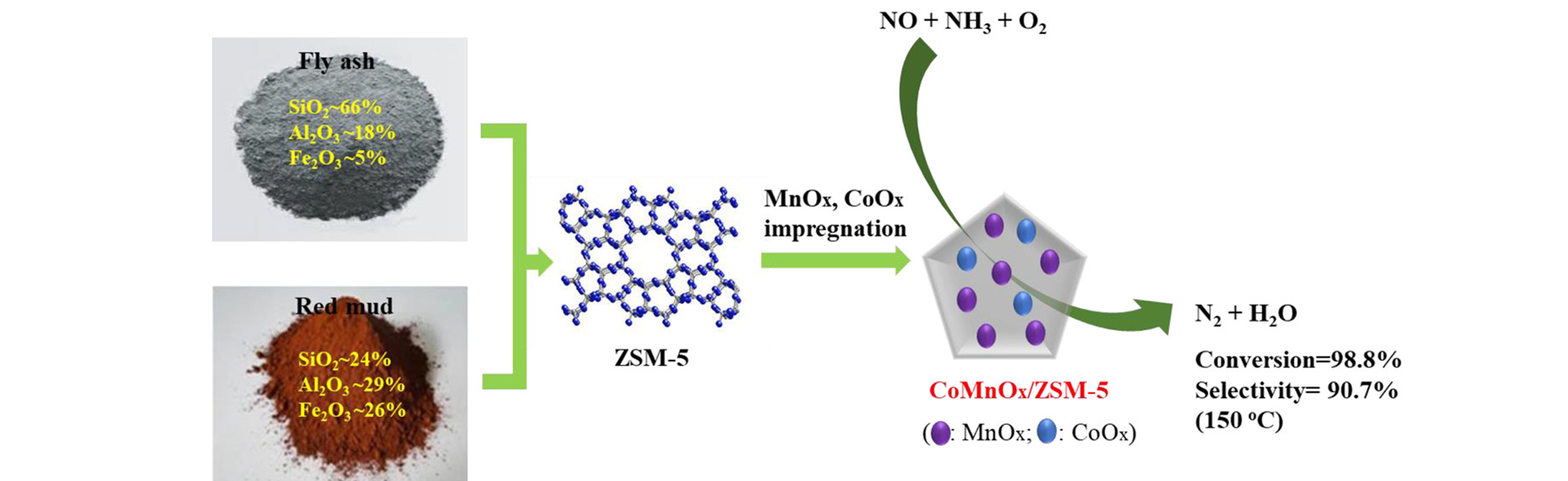
Fig.5 Co? and Mn?coimpregnated ZSM?5 prepared from recycled industrial solid wastes for low?temperature NH3?SCR[90]Copyright 2019, American Chemical Society.
| Raw material | Pre?treatment method | Crystallization method | Main product | Crystal size/μm | Ref. |
|---|---|---|---|---|---|
| Fly ash | NaOH solution pretreatment | Microwave hydrothermal | LTA | 2—5 | [ |
| crystallization | |||||
| Alkali fusion | Hydrothermal crystallization | LTA | 8—10 | [ | |
| Alkali fusion | Ultrasonic hydrothermal | FAU(X) | 0.5 | [ | |
| crystallization | |||||
| Alkali fusion(Na2CO3), acid leaching | Hydrothermal crystallization | GIS | 2.84—5.82 | [ | |
| Roasting, alkali fusion, acid leaching | Hydrothermal crystallization | ZSM?5 | 2—3 | [ | |
| Acid leaching | Hydrothermal crystallization | ZSM?5 | 5—8 | [ | |
| Acid leaching | Hydrothermal crystallization | ZSM?5 | 0.4—0.5 | [ | |
| Alkali fusion(KOH) | Hydrothermal crystallization | CHA | 4—8 | [ | |
| Alkali fusion(KOH) | Hydrothermal crystallization | CHA | 5—8 | [ | |
| Alkali fusion | Hydrothermal crystallization | FAU(Y) | 7—8 | [ | |
| Perlite industrial waste | No treatment | Hydrothermal crystallization | Zeolite?Pc | 1 | [ |
| No treatment | Hydrothermal crystallization | FAU(X) | 10—25 | [ | |
| No treatment | Hydrothermal crystallization | FAU(X) | 1 | [ | |
| Coal gangue | Roasting, alkali fusion | Hydrothermal crystallization | LTA | 1.7 | [ |
| Roasting | Hydrothermal crystallization | LTA | 1—1.5 | [ | |
| Roasting | Hydrothermal crystallization | LTA | 1.5 | [ | |
| Roasting | Hydrothermal crystallization | LTA | 1.5—2 | [ | |
| Roasting, alkali fusion | Hydrothermal crystallization | FAU(X) | 2.5—3 | [ | |
| Alkali fusion | Hydrothermal crystallization | FAU(X) | 12—17 | [ | |
| No treatment | Hydrothermal crystallization | CHA | 2—7 | [ | |
| No treatment | Ultrasonic hydrothermal | CHA | 1.5—7 | [ | |
| crystallization | |||||
| Roasting, acid leaching | Hydrothermal crystallization | ZSM?5 | 2—4 | [ | |
| Spent FCC catalyst | Alkali fusion(Na2CO3) | Hydrothermal crystallization | LTA | 2 | [ |
| Acid leaching | Hydrothermal crystallization | LTA | 1—2.5 | [ | |
| Ball milling | Hydrothermal crystallization | FAU(X) | 1 | [ | |
| Alkali fusion | Hydrothermal crystallization | FAU(Y) | 0.2 | [ | |
| Ammonium sulfate activation method, | Hydrothermal crystallization | FAU(Y) | 0.4—1.2 | [ | |
| acid leaching | |||||
| Microwave acid treatment | Microwave hydrothermal | FAU(Y) | 0.1—0.3 | [ | |
| crystallization | |||||
| Lithium slag | No treatment | Dynamic hydrothermal | SOD | 20 | [ |
| crystallization | |||||
| Alkali fusion | Hydrothermal crystallization | FAU(X) | 1—3 | [ | |
| Bauxite residue | Alkali fusion | Hydrothermal crystallization | LTA | 0.2—1 | [ |
| Acid leaching, alkali fusion | Hydrothermal crystallization | LTA | 5—10 | [ | |
| No treatment | Hydrothermal crystallization | ZSM?5 | 2—5 | [ | |
| Alkali fusion | Hydrothermal crystallization | ZSM?5 | 1—2 | [ | |
| Alkali fusion, acid leaching | Hydrothermal crystallization | LTA | 1—10 | [ | |
| Waste porcelain | Alkali fusion | Hydrothermal crystallization | EMT | 0.3—1 | [ |
| Alkali fusion | Hydrothermal crystallization | FAU | 0.5 | [ | |
| Alkaline Liquor | Hydrothermal crystallization | LTA | 2—4 | [ | |
| Waste glass | Acid leaching | Hydrothermal crystallization | LTA | 2—3 | [ |
| NaOH solution pretreatment, | Hydrothermal crystallization | LTA | 2—5 | [ | |
| Sodium sulfide treatment | |||||
| Roasting | Hydrothermal crystallization | GIS | 2—5 | [ | |
| No treatment | Hydrothermal crystallization | ANA | 5—10 | [ |
Table 2 Activation methods, crystallization methods and size distribution of synthetic zeolites from industrial solid wastes
| Raw material | Pre?treatment method | Crystallization method | Main product | Crystal size/μm | Ref. |
|---|---|---|---|---|---|
| Fly ash | NaOH solution pretreatment | Microwave hydrothermal | LTA | 2—5 | [ |
| crystallization | |||||
| Alkali fusion | Hydrothermal crystallization | LTA | 8—10 | [ | |
| Alkali fusion | Ultrasonic hydrothermal | FAU(X) | 0.5 | [ | |
| crystallization | |||||
| Alkali fusion(Na2CO3), acid leaching | Hydrothermal crystallization | GIS | 2.84—5.82 | [ | |
| Roasting, alkali fusion, acid leaching | Hydrothermal crystallization | ZSM?5 | 2—3 | [ | |
| Acid leaching | Hydrothermal crystallization | ZSM?5 | 5—8 | [ | |
| Acid leaching | Hydrothermal crystallization | ZSM?5 | 0.4—0.5 | [ | |
| Alkali fusion(KOH) | Hydrothermal crystallization | CHA | 4—8 | [ | |
| Alkali fusion(KOH) | Hydrothermal crystallization | CHA | 5—8 | [ | |
| Alkali fusion | Hydrothermal crystallization | FAU(Y) | 7—8 | [ | |
| Perlite industrial waste | No treatment | Hydrothermal crystallization | Zeolite?Pc | 1 | [ |
| No treatment | Hydrothermal crystallization | FAU(X) | 10—25 | [ | |
| No treatment | Hydrothermal crystallization | FAU(X) | 1 | [ | |
| Coal gangue | Roasting, alkali fusion | Hydrothermal crystallization | LTA | 1.7 | [ |
| Roasting | Hydrothermal crystallization | LTA | 1—1.5 | [ | |
| Roasting | Hydrothermal crystallization | LTA | 1.5 | [ | |
| Roasting | Hydrothermal crystallization | LTA | 1.5—2 | [ | |
| Roasting, alkali fusion | Hydrothermal crystallization | FAU(X) | 2.5—3 | [ | |
| Alkali fusion | Hydrothermal crystallization | FAU(X) | 12—17 | [ | |
| No treatment | Hydrothermal crystallization | CHA | 2—7 | [ | |
| No treatment | Ultrasonic hydrothermal | CHA | 1.5—7 | [ | |
| crystallization | |||||
| Roasting, acid leaching | Hydrothermal crystallization | ZSM?5 | 2—4 | [ | |
| Spent FCC catalyst | Alkali fusion(Na2CO3) | Hydrothermal crystallization | LTA | 2 | [ |
| Acid leaching | Hydrothermal crystallization | LTA | 1—2.5 | [ | |
| Ball milling | Hydrothermal crystallization | FAU(X) | 1 | [ | |
| Alkali fusion | Hydrothermal crystallization | FAU(Y) | 0.2 | [ | |
| Ammonium sulfate activation method, | Hydrothermal crystallization | FAU(Y) | 0.4—1.2 | [ | |
| acid leaching | |||||
| Microwave acid treatment | Microwave hydrothermal | FAU(Y) | 0.1—0.3 | [ | |
| crystallization | |||||
| Lithium slag | No treatment | Dynamic hydrothermal | SOD | 20 | [ |
| crystallization | |||||
| Alkali fusion | Hydrothermal crystallization | FAU(X) | 1—3 | [ | |
| Bauxite residue | Alkali fusion | Hydrothermal crystallization | LTA | 0.2—1 | [ |
| Acid leaching, alkali fusion | Hydrothermal crystallization | LTA | 5—10 | [ | |
| No treatment | Hydrothermal crystallization | ZSM?5 | 2—5 | [ | |
| Alkali fusion | Hydrothermal crystallization | ZSM?5 | 1—2 | [ | |
| Alkali fusion, acid leaching | Hydrothermal crystallization | LTA | 1—10 | [ | |
| Waste porcelain | Alkali fusion | Hydrothermal crystallization | EMT | 0.3—1 | [ |
| Alkali fusion | Hydrothermal crystallization | FAU | 0.5 | [ | |
| Alkaline Liquor | Hydrothermal crystallization | LTA | 2—4 | [ | |
| Waste glass | Acid leaching | Hydrothermal crystallization | LTA | 2—3 | [ |
| NaOH solution pretreatment, | Hydrothermal crystallization | LTA | 2—5 | [ | |
| Sodium sulfide treatment | |||||
| Roasting | Hydrothermal crystallization | GIS | 2—5 | [ | |
| No treatment | Hydrothermal crystallization | ANA | 5—10 | [ |
| 1 | Xu R. R., Pang W. Q., Huo Q. S., Chemistry of Zeolites and Porous Materials, Science Press, Beijing, 2015(徐如人, 庞文琴, 霍启升. 分子筛与多孔材料化学, 北京: 科学出版社, 2015) |
| 2 | Yilmaz B., Müller U., Top. Catal., 2009, 52(6/7), 888—895 |
| 3 | Zhang W., Ji Y. Y., Peng H., Dai S. Y., Liu Y., Zhang J. M., Wang D. M., Chem. J. Chinese Universities, 2018, 39(9), 1985—1992(张武, 纪妍妍, 彭涵, 戴少英, 刘莹, 张纪梅, 王冬梅. 高等学校化学学报, 2018, 39(9), 1985—1992) |
| 4 | Li G., Li H., Zhao H., Cai T., Li Y., Guan S., Chem. Res. Chinese Universities, 2018, 34(6), 877―881 |
| 5 | Jiri C., Avelino C., Stacey Z., Zeolites and Catalysis: Synthesis, Reactions and Applications, John Wiley & Sons, Weinheim, 2010 |
| 6 | Kulprathipanja S., Zeolites in Industrial Separation and Catalysis, John Wiley & Sons, Weinheim, 2010 |
| 7 | Xiao F. S., Meng X., Zeolites in Sustainable Chemistry: Synthesis, Characterization and Catalytic Applications, Springer, Berlin, 2015 |
| 8 | Li Y., Xu L., Bai P., Rong G., Zhang D., Diwu J., Yan W., Chai Z., Wang S., Environ. Sci. : Nano, 2019, 6(3), 736—746 |
| 9 | Wang S., Peng Y., Chem. Eng. J., 2010, 156(1), 11—24 |
| 10 | Li Y., Bai P., Yan Y., Yan W., Shi W., Xu R., Micropor. Mesopor. Mater., 2019, 273, 203—211 |
| 11 | Rodney P. T., Risto H., Ion Exchange in Molecular Sieves by Conventional Techniques, Springer, Berlin, 2002 |
| 12 | Li Y., Simon A. O., Jiao C., Zhang M., Yan W., Rao H., Liu J., Zhang J., Micropor. Mesopor. Mater., 2020, 302, 110244 |
| 13 | Ren X., Qu R., Liu S., Zhao H., Wu W., Song H., Zheng C., Wu X., Gao X., Aerosol Air Qual. Res., 2020, 20(5), 1127—1144 |
| 14 | Shao G. Q., Zhu H., Ma W., Yan P., Ma J. L., Chem. J. Chinese Universities, 2019, 40(11), 2265—2273(邵国泉, 朱惠, 马伟, 闫攀, 马继龙. 高等学校化学学报, 2019, 40(11), 2265—2273) |
| 15 | Yang R. T., Gas Separation by Adsorption Processes Butterworth, Boston, 1987 |
| 16 | Yang R. T., Adsorbents_ Fundamentals and Applications, Wiley⁃Interscience, New York, 2003 |
| 17 | Baerlocher C., McCusker L. B., Database of Zeolite Structures, http://www. iza⁃structure. org/databases/ |
| 18 | Guo J. H., Yan W. F., Shi W., Xu R. R., Chem. J. Chinese Universities, 2018, 39(5), 841—848(郭俊辉, 闫文付, 师唯, 徐如人. 高等学校化学学报, 2018, 39(5), 841—848) |
| 19 | Fang X. J., Liu R. Y., Lin S., Shi L., Wang R. W., Li Y., Li J. Y., Chem. J. Chinese Universities, 2019, 40(5), 867—872(房夕杰, 刘瑞云, 林森, 石磊, 王润伟, 李乙, 李俊英. 高等学校化学学报, 2019, 40(5), 867—872) |
| 20 | Collins F., Rozhkovskaya A., Outram J. G., Millar G. J., Micropor. Mesopor. Mater., 2020, 291, 109667 |
| 21 | Qiu Y., Wu D., Yan L., Zhou Y., RSC Adv., 2016, 6(43), 36942—36953 |
| 22 | Luo Y., Wu Y., Ma S., Zheng S., Zhang Y., Chu P. K., Environ. Sci. Pollut. Res. Int., 2020, 1—14 |
| 23 | Feng W., Wan Z., Daniels J., Li Z., Xiao G., Yu J., Xu D., Guo H., Zhang D., May E. F., Li G., J. Cleaner Prod., 2018, 202, 390—400 |
| 24 | Osacký M., Pálková H., Hudec P., Czímerová A., Galusková D., Vítková M., Micropor. Mesopor. Mater., 2020, 294, 109852 |
| 25 | Qian T., Li J., Adv. Powder Technol., 2015, 26(1), 98—104 |
| 26 | Basaldella E., Torressanchez R., Conconi M., Appl. Clay. Sci., 2009, 42(3/4), 611—614 |
| 27 | Chen D., Hu X., Shi L., Cui Q., Wang H., Yao H., Appl. Clay. Sci., 2012, 59/60, 148—151 |
| 28 | Belviso C., Agostinelli E., Belviso S., Cavalcante F., Pascucci S., Peddis D., Varvaro G., Fiore S., Micropor. Mesopor. Mater., 2015, 202, 208—216 |
| 29 | Maatoug N., Delahay G., Tounsi H., Waste Manag., 2018, 74, 267—278 |
| 30 | Kim J. C., Choi M., Song H. J., Park J. E., Yoon J. H., Park K. S., Lee C. G., Kim D. W., Mater. Chem. Phys., 2015, 166, 20—25 |
| 31 | Hosseini Asl S. M., Javadian H., Khavarpour M., Belviso C., Taghavi M., Maghsudi M., J. Cleaner Prod., 2019, 208, 1131—1147 |
| 32 | Toniolo N., Boccaccini A. R., Ceram. Int., 2017, 43(17), 14545—14551 |
| 33 | Vassilev S. G., Vassileva C. G., Energy Fuels, 2005, 19, 1084—1098 |
| 34 | Xu H., Wu L., Shi T., Liu W., Qi S., Sci. China: Technol. Sci., 2014, 57(6), 1127—1134 |
| 35 | Behin J., Bukhari S. S., Dehnavi V., Kazemian H., Rohani S., Chem. Eng. Technol., 2014, 37(9), 1532—1540 |
| 36 | Boycheva S., Marinov I., Miteva S., Zgureva D., Sustainable Chem. Pharm., 2020, 15, 100217 |
| 37 | Czuma N., Zarębska K., Motak M., Gálvez M. E., Da Costa P., Fuel, 2020, 267, 117139 |
| 38 | Lim Y., Yu J., Park S., Kim M., Chen S., Bakri N. A. B., Sabri N., Bae S., Kim H. S., Bioresour. Technol., 2020, 307, 123201 |
| 39 | Zhang Y., Kang W., Han H., Wang H., Chen Y., Gong X., Zhai C., Song H., J. Am. Ceram. Soc., 2019, 102(12), 7665—7677 |
| 40 | Liu X., Zeng S., Wang R., Zhang Z., Qiu S., Chem. Res. Chinese Universities, 2018, 34(3), 350―357 |
| 41 | Cui K., Fang Y., Xu D., Zhang Y., Han L., Che S., Chem. Res. Chinese Universities, 2019, 35(3), 359―362 |
| 42 | Wang Q., Yang X., Jiang Y., Huo H., Li D., Lin K., Xu X., Chem. Res. Chinese Universities, 2018, 34(1), 13—18 |
| 43 | Wang C., Wang D., Zheng S., Fang X., Zhang W., Tian Y., Lin H., Lu H., Jiang L., Chem. Res. Chinese Universities, 2018, 34(6), 983―988 |
| 44 | Li J., Shi Y., Fu X., Huang J., Y. Z., S. D., Zhang F., React. Kinet., Mech. Catal., 2019, 128, 289—314 |
| 45 | Hossini Asl S. M., Masomi M., Tajbakhsh M., J. Cleaner Prod., 2020, 258, 120688 |
| 46 | Rajakrishnamoorthy P., Karthikeyan D., Saravanan C. G., Mater. Today: Proc., 2020, 22, 499—506 |
| 47 | Feng R., Chen K., Yan X., Hu X., Zhang Y., Wu J., Catalysts, 2019, 9(10), 788—800 |
| 48 | Du T., Fang X., Wei Y., Shang J., Zhang B., Liu L., Energ. Fuel., 2017, 31(4), 4301—4307 |
| 49 | Che S., Fang X., Li S., Chen X., Du T., Z. Anorg. Allg. Chem., 2019, 645(24), 1365—1371 |
| 50 | Rayalu S., Meshram S. U., Hasan M. Z., J. Hazard. Mater., 2000, 77, 123—131 |
| 51 | Ren X., Liu S., Qu R., Xiao L., Hu P., Song H., Wu W., Zheng C., Wu X., Gao X., Micropor. Mesopor. Mater., 2020, 295, 109940 |
| 52 | Król M., Morawska J., Mozgawa W., Pichór W., Mater. Sci. Pol., 2014, 32(3), 503—513 |
| 53 | Christidis G. E., Paspaliaris I., Kontopoulos A., Appl. Clay Sci., 1999, 15, 305—324 |
| 54 | Król M., Mozgawa W., Morawska J., Pichór W., Micropor. Mesopor. Mater., 2014, 196, 216—222 |
| 55 | Król M., Matras E., Mozgawa W., Int. J. Environ. Sci. Technol., 2016, 13(11), 2697—2704 |
| 56 | Zhang Y., Ling T. C., Constr. Build. Mater., 2020, 234, 117424 |
| 57 | Frías M., Sanchez de Rojas M. I., García R., Juan Valdés A., Medina C., Cem. Concr. Compos., 2012, 34(5), 678—683 |
| 58 | Querol X., Izquierdo M., Monfort E., Alvarez E., Font O., Moreno T., Alastuey A., Zhuang X., Lu W., Wang Y., Int. J. Coal. Geol., 2008, 75(2), 93—104 |
| 59 | Hu X., Bai J., Wang J., Li C., Xu W., RSC Adv., 2015, 5(4), 2968—2973 |
| 60 | Zhou J., Zheng F., Li H., Wang J., Bu N., Hu P., Gao J. M., Zhen Q., Bashir S., Liu J., Chem. Eng. J., 2020, 381, 122698 |
| 61 | Li H., Zheng F., Wang J., Zhou J., Huang X., Chen L., Hu P., Gao J. M., Zhen Q., Bashir S., Liu J. L., Chem. Eng. J., 2020, 390, 124513 |
| 62 | Lu X., Shi D., Chen J., Environ. Earth Sci., 2017, 76(17), 591—601 |
| 63 | Lu X., Chen J., J. Water Reuse Desalin., 2018, 8(1), 94—101 |
| 64 | Chen J., Lu X., J. Mater. Cycles Waste Manage., 2017, 20(1), 489—495 |
| 65 | Ge Q., Moeen M., Tian Q., Xu J., Feng K., Environ. Sci. Pollut. Res. Int., 2020, 27(7), 7398—7408 |
| 66 | Han J., Jin X., Song C., Bi Y., Liu Q., Liu C., Ji N., Lu X., Ma D., Li Z., Green Chem., 2020, 22(1), 219—229 |
| 67 | Han J., Ha Y., Guo M., Zhao P., Liu Q., Liu C., Song C., Ji N., Lu X., Ma D., Li Z., Ultrason. Sonochem., 2019, 59, 104703 |
| 68 | Li H., Cheng R., Liu Z., Du C., Sci. Total. Environ., 2019, 683, 638—647 |
| 69 | Akcil A., Veglio F., Ferella F., Okudan M. D., Tuncuk A., Waste Manag., 2015, 45, 420—433 |
| 70 | Ferella F., Innocenzi V., Maggiore F., Resour., Conserv. Recycl., 2016, 108, 10—20 |
| 71 | Xue Y., Wei X., Zhao H., Wang T., Xiao Y., J. Cleaner Prod., 2020, 259, 120830 |
| 72 | Gonzalez M. R., Pereyra A. M., Bosch P., Fetter G., Lara V. H., Basaldella E. I., J. Mater. Sci., 2016, 51(11), 5061—5072 |
| 73 | Monzón J. D., Gonzalez M. R., Mardones L. E., Conconi M. S., Pereyra A. M., Basaldella E. I., Mater. Today Commun., 2019, 21, 100624 |
| 74 | Ferella F., Leone S., Innocenzi V., De Michelis I., Taglieri G., Gallucci K., J. Cleaner Prod., 2019, 230, 910—926 |
| 75 | Basaldella E. I., Paladino J. C., Solari M., Valle G. M., Appl. Catal. B, 2006, 66(3/4), 186—191 |
| 76 | Kothari Z., Niwate Y., Patel D. P., Chudasama C., Kalpana G., Jasra R. V., Clean Technol. Environ. Policy, 2018, 21(2), 471—477 |
| 77 | Liu X., Li L., Yang T., Yan Z., J. Porous. Mater, 2011, 19(1), 133—139 |
| 78 | Zheng S., He L., Yao H., Ren S., Yu H., Zhang J., China Pet. Process. Petrochem. Technol., 2015, 17(4), 46—54 |
| 79 | Le T., Wang Q., Ravindra A. V., Li X., Ju S., J. Alloy. Compd., 2019, 776, 437—446 |
| 80 | Jandová J., Dvořák P., Vu H. N., Hydrometallurgy, 2010, 103(1—4), 12—18 |
| 81 | Li J., Lian P., Huang S., Huang L., J. Environ. Manage., 2020, 265, 110551 |
| 82 | Yiren W., Dongmin W., Yong C., Dapeng Z., Ze L., Constr. Build. Mater., 2019, 203, 304—313 |
| 83 | Xing P., Wang C., Zeng L., Ma B., Wang L., Chen Y., Yang C., ACS Sustainable Chem. Eng., 2019, 7(10), 9498—9505 |
| 84 | Lin G., Zhuang Q., Cui Q., Wang H., Yao H., Chinese. J. Chem. Eng., 2015, 23(11), 1768—1773 |
| 85 | Hind A. R., Bhargava S. K., Grocott S. C., Colloids Surf. A, 1999, 146, 359—374 |
| 86 | Xue S., Zhu F., Kong X., Wu C., Huang L., Huang N., Hartley W., Environ. Sci. Pollut. Res. Int., 2016, 23(2), 1120—1132 |
| 87 | Gomes H. I., Mayes W. M., Rogerson M., Stewart D. I., Burke I. T., J. Cleaner Prod., 2016, 112, 3571—3582 |
| 88 | Ma D., Wang Z., Guo M., Zhang M., Liu J., Waste Manag., 2014, 34(11), 2365—2372 |
| 89 | Riski O., Hartanto D., Ni’mah Y. L., Hartati, Prasetyoko D., AIP Conf. Proc., 2018, 2049, 020038 |
| 90 | Zhang S., Zhang C., Wang Q., Ahn W. S., Ind. Eng. Chem. Res., 2019, 58(51), 22857—22865 |
| 91 | Xie W. M., Zhou F. P., Bi X. L., Chen D. D., Li J., Sun S. Y., Liu J. Y., Chen X. Q., J. Hazard. Mater., 2018, 358, 441—449 |
| 92 | Quyen D. T. N., Loc L. C., Ha H. K. P., Nga D. T. H., Tri N., Van N. T. T., AIP Conf. Proc., 2017, 1878, 020034 |
| 93 | Belviso C., Kharchenko A., Agostinelli E., Cavalcante F., Peddis D., Varvaro G., Yaacoub N., Mintova S., Micropor. Mesopor. Mater., 2018, 270, 24—29 |
| 94 | Ma L., Han L., Chen S., Hu J., Chang L., Bao W., Wang J., Fuel Process. Technol., 2019, 189, 39—48 |
| 95 | Dong W. X., Cho W., Bao Q. F., Shen H. J., J. Ceram., 2018, 39(2), 170—173(董伟霞, Cho Woo Seok, 包启富, 沈慧娟. 陶瓷学报, 2018, 39(2), 170—173) |
| 96 | Tan L., Yu F., Chen X. W., China Ceram. Ind., 2011, 18(3), 21—23(谭丽, 余峰, 陈学文. 中国陶瓷工业, 2011, 18(3), 21—23) |
| 97 | Li L., Liu W., You Q., Chen M., Zeng Q., J. Cleaner Prod., 2020, 259, 120853 |
| 98 | Wajima T., Ikegami Y., Ceram. Int., 2007, 33(7), 1269—1274 |
| 99 | Wajima T., Ikegami Y., Ceram. Int., 2009, 35(7), 2983—2986 |
| 100 | Ibrahim S., Meawad A., Constr. Build. Mater., 2018, 182, 451—458 |
| 101 | Terzano R., D'Alessandro C., Spagnuolo M., Romagnoli M., Medici L., CLEAN — Soil, Air, Water, 2015, 43(1), 133—140 |
| 102 | Di L. G., Glass, 2019,(6), 52—55(狄磊刚, 玻璃, 2019,(6), 52—55) |
| 103 | Tsujiguchi M., Kobashi T., Utsumi Y., Kakimori N., Nakahira A., Tsujiguchi M., J. Am. Ceram. Soc., 2014, 97(1), 114—119 |
| 104 | Yao Z., Wu D., Liu J., Wu W., Zhao H., Tang J., J. Hazard. Mater., 2017, 324(Pt B), 673—680 |
| 105 | Kang Y., Swain B., Im B., Yoon J. H., Park K. H., Lee C. G., Kim D. G., Metals, 2019, 9(12), 1240—1248 |
| 106 | Majdinasab A. R., Manna P. K., Wroczynskyj Y., van Lierop J., Cicek N., Tranmer G. K., Yuan Q., Mater. Chem. Phys., 2019, 221, 272—287 |
| 107 | Majdinasab A. R., Yuan Q., Ceram. Int., 2019, 45(2), 2400—2410 |
| 108 | Vigil de la Villa Mencía R., Goiti E., Ocejo M., Giménez R. G., Micropor. Mesopor. Mater., 2020, 293, 109817 |
| 109 | Charnell J. F., J. Cryst. Gr., 1971, 8(3), 291—294 |
| [1] | 姚伊婷, 吕佳敏, 余申, 刘湛, 李昱, 李小云, 苏宝连, 陈丽华. 等级孔微孔-介孔Fe2O3/ZSM-5中空分子筛催化材料的制备及催化苄基化性能[J]. 高等学校化学学报, 2022, 43(8): 20220090. |
| [2] | 李志光, 齐国栋, 徐君, 邓风. Sn-Al-β分子筛酸性在葡萄糖转化反应中作用的固体NMR研究[J]. 高等学校化学学报, 2022, 43(6): 20220138. |
| [3] | 陈玮琴, 吕佳敏, 余申, 刘湛, 李小云, 陈丽华, 苏宝连. 有机杂化介孔Beta分子筛的合成及在苯甲醇烷基化反应中的应用[J]. 高等学校化学学报, 2022, 43(6): 20220086. |
| [4] | 李加富, 张凯, 王宁, 孙启明. 分子筛限域单原子金属催化剂的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220032. |
| [5] | 孟祥龙, 杨歌, 郭海玲, 刘晨光, 柴永明, 王纯正, 郭永梅. 纳米分子筛的合成及硫化氢吸附性能[J]. 高等学校化学学报, 2022, 43(3): 20210687. |
| [6] | 魏李娜, 彭莉, 朱锋, 顾鹏飞, 顾学红. 中空纤维Au-CeZr/FAU催化膜的制备及在富氢气氛CO选择性氧化反应中的应用[J]. 高等学校化学学报, 2022, 43(10): 20220175. |
| [7] | 李海勃, 肖长发, 江龙, 黄云, 淡宜. MCM-41分子筛负载氯化铝催化丙烯酸甲酯与1-辛烯共聚[J]. 高等学校化学学报, 2021, 42(9): 2974. |
| [8] | 李奕川, 朱国富, 王宇, 柴永明, 刘晨光, 何盛宝. 基底表面性质与前驱液化学环境对原位定向构筑钛硅分子筛膜的影响[J]. 高等学校化学学报, 2021, 42(9): 2934. |
| [9] | 罗强强, 金少青, 孙洪敏, 杨为民. 液相酸溶液后补钛合成Ti-MWW分子筛[J]. 高等学校化学学报, 2021, 42(9): 2742. |
| [10] | 田润赛, 卢芊, 张洪滨, 张渤, 冯源源, 魏金香, 冯季军. 氮杂碳原位包覆Cu2O/Co3O4@C异质结构复合材料的设计构筑及高效储锂性能[J]. 高等学校化学学报, 2021, 42(8): 2592. |
| [11] | 张旭, 阙家乾, 侯月新, 吕佳敏, 刘湛, 雷坤皓, 余申, 李小云, 陈丽华, 苏宝连. 等级孔介孔-微孔TS-1分子筛单晶的合成及催化氯丙烯环氧化性能[J]. 高等学校化学学报, 2021, 42(8): 2529. |
| [12] | 王磊, 孙毯毯, 闫娜娜, 马超, 刘晓娜, 田鹏, 郭鹏, 刘中民. 利用适用于SAPO-34的有机结构导向剂合成SSZ-13分子筛[J]. 高等学校化学学报, 2021, 42(6): 1716. |
| [13] | 王冶, 张晓思, 孙丽婧, 李冰, 刘琳, 杨淼, 田鹏, 刘仲毅, 刘中民. 有机硅烷辅助合成特殊形貌SAPO分子筛[J]. 高等学校化学学报, 2021, 42(3): 683. |
| [14] | 李健, 于明明, 孙源, 冯文华, 冯兆池, 吴剑峰. 水溶液pH对甲烷低温氧化制备甲醇的影响[J]. 高等学校化学学报, 2021, 42(3): 776. |
| [15] | 王勇, 董彪, 孙娇, 董德录, 孙连坤. 基于分子筛模板的银/硅铝无定形结构复合材料的合成及光谱性质[J]. 高等学校化学学报, 2021, 42(10): 3233. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||