

高等学校化学学报 ›› 2020, Vol. 41 ›› Issue (7): 1407.doi: 10.7503/cjcu20200205
• 庆祝《高等学校化学学报》复刊40周年专栏 • 上一篇 下一篇
收稿日期:2020-04-14
出版日期:2020-07-10
发布日期:2020-06-10
通讯作者:
游书力
E-mail:slyou@sioc.ac.cn
基金资助:
ZHU Min1,2,ZHANG Xiao1,YOU Shuli1,2,*( )
)
Received:2020-04-14
Online:2020-07-10
Published:2020-06-10
Contact:
Shuli YOU
E-mail:slyou@sioc.ac.cn
Supported by:摘要:
总结了近年来可见光促进苯及其衍生物去芳构化反应的研究进展, 主要分为基态与激发态化学两方面, 并对该领域的发展前景进行了展望.
中图分类号:
TrendMD:
朱敏, 张霄, 游书力. 可见光促进的苯及衍生物去芳构化反应. 高等学校化学学报, 2020, 41(7): 1407.
ZHU Min, ZHANG Xiao, YOU Shuli. Visible-light-promoted Dearomatization of Benzene and Derivatives†. Chem. J. Chinese Universities, 2020, 41(7): 1407.
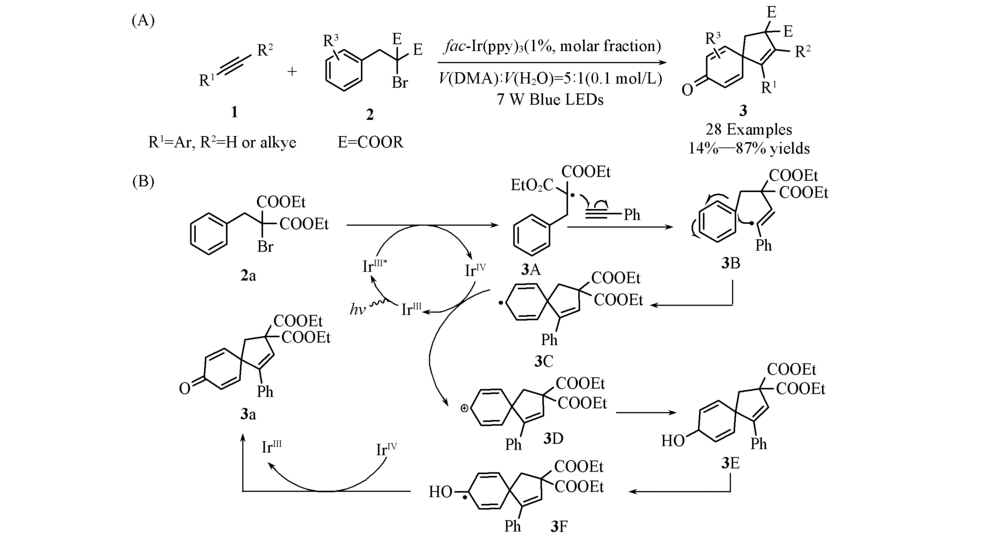
Scheme 2 General reaction(A) and proposed mechanism(B) of visible-light-induced radical dearomative cyclization of benzene derivatives[22] Copyright 2020, American Chemical Society.
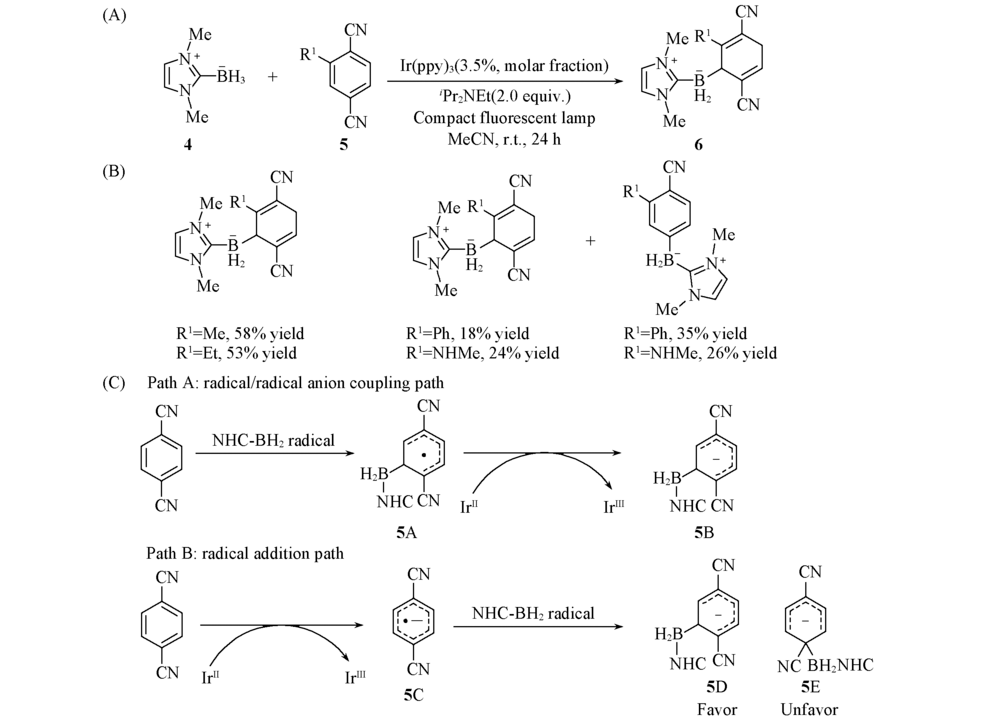
Scheme 3 General reaction(A), substrate scope(B) and proposed mechanism(C) of visible-light-induced dearomative 1,4-hydroboration of electron-poor benzenes[23] Copyright 2020, American Chemical Society.
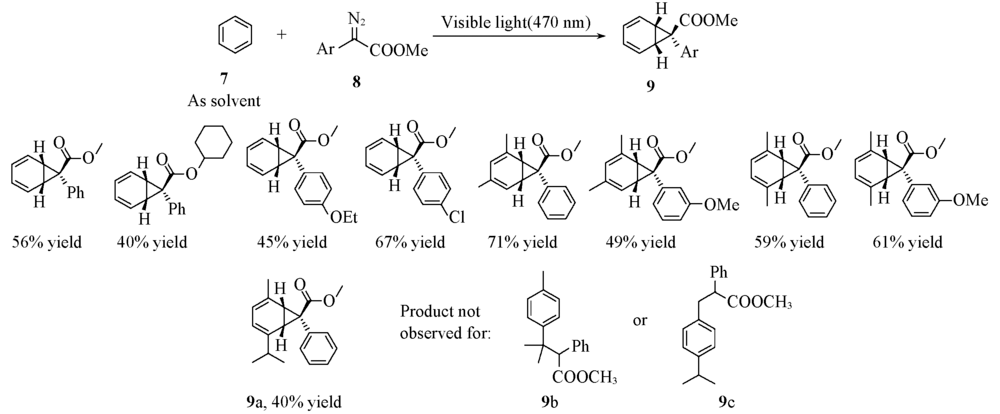
Scheme 4 General reaction(A) and substrate scope(B) of visible-light-mediated [2+1] dearomative cycloaddition reactions of arenes[24] Copyright 2019, American Chemical Society.
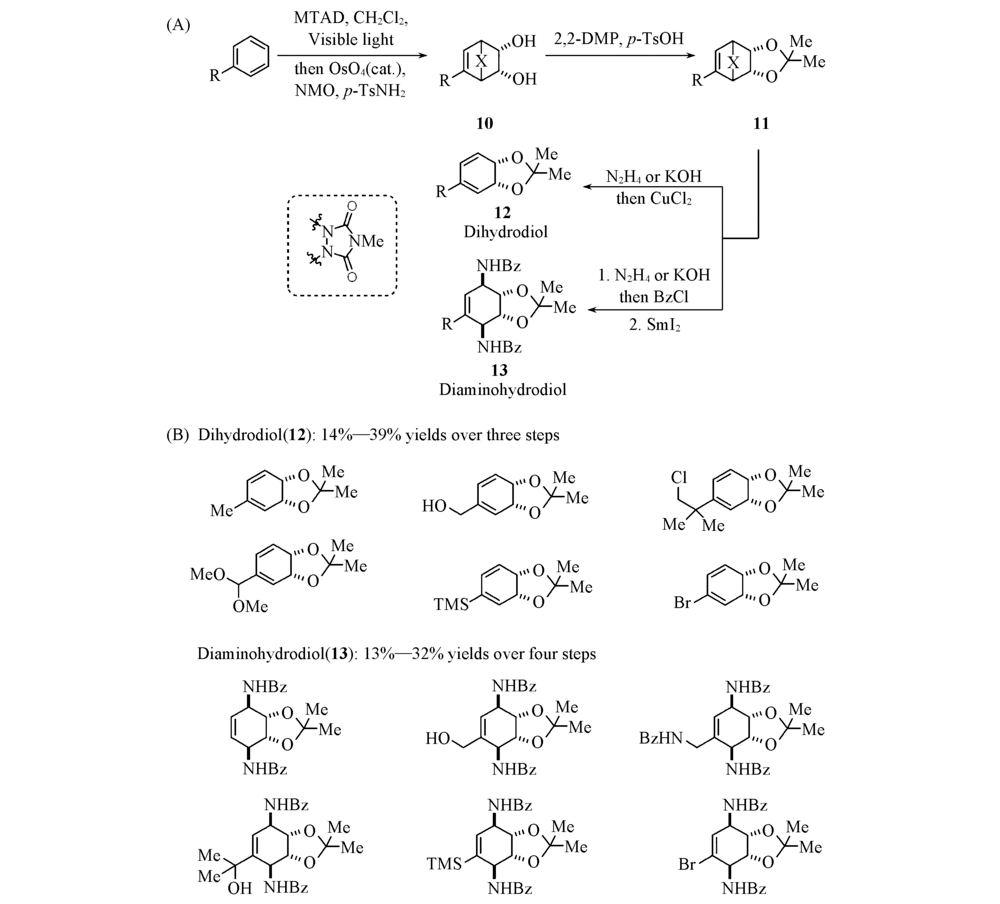
Scheme 5 General reaction(A) and substrate scope(B) of arenophile-mediated dearomative dihydroxylation and product derivatizations[26] Copyright 2016, Springer Nature Limited.
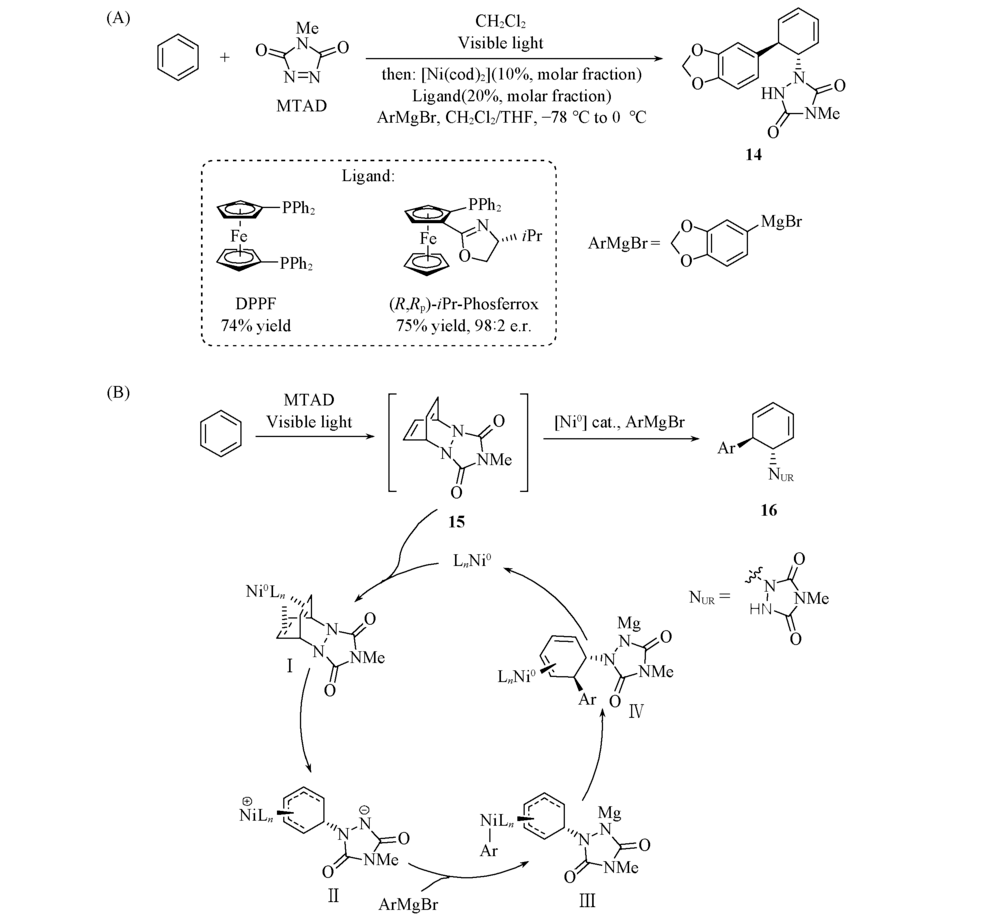
Scheme 6 Optimized reaction conditions(A) and proposed mechanism(B) of arenophile-mediated, Ni-catalyzed dearomative trans-1,2-carboamination of benzene derivatives[27] Copyright 2018, American Chemical Society.
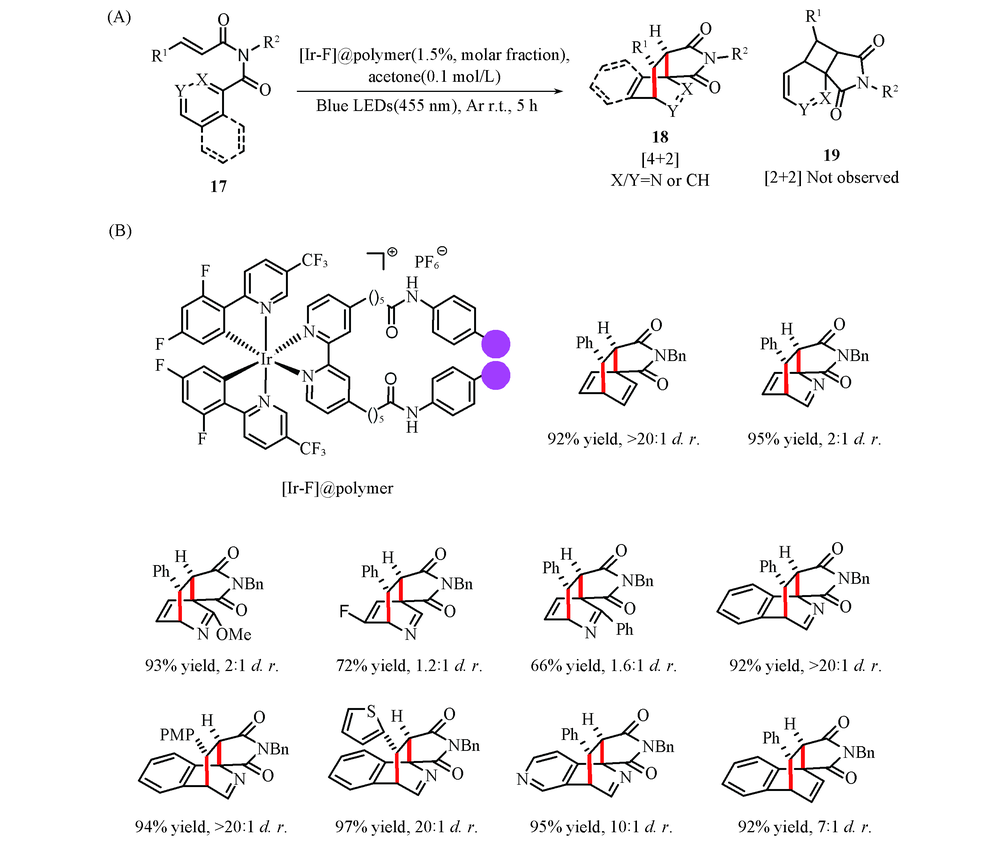
Scheme 7 General reaction(A) and substrate scope(B) of visible-light-mediated [4+2] dearomative cycloaddition of pyridine and benzene derivatives via energy transfer[32] Copyright 2019, Elesevier Company.
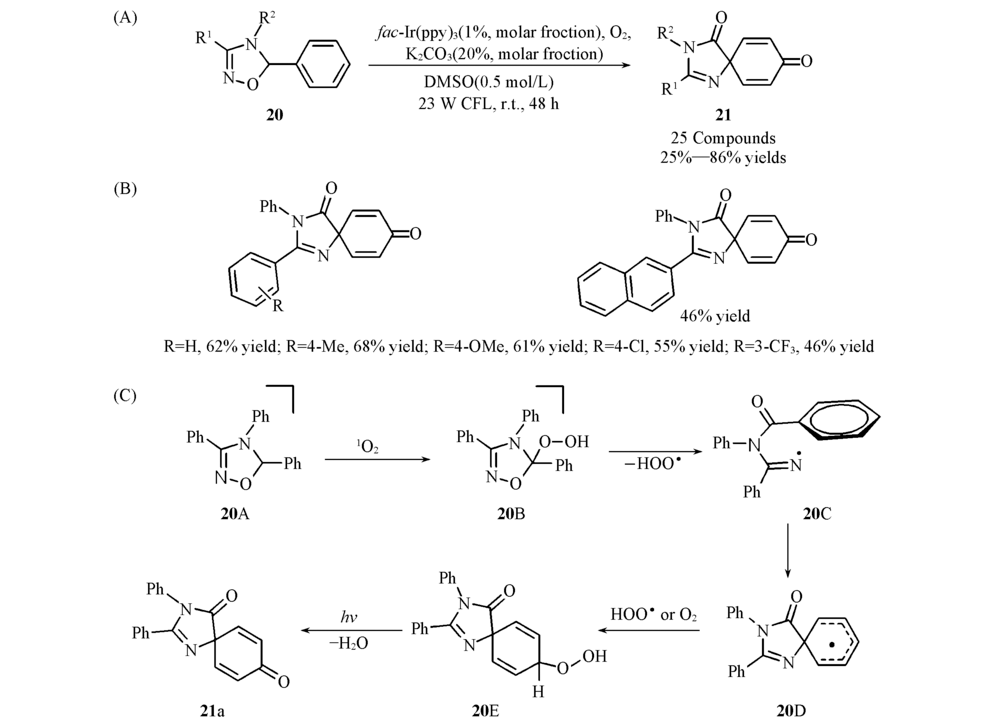
Scheme 8 General reaction(A), substrate scope(B) and proposed mechanism(C) of visible-light-promoted double functionalization of benzenes via energy transfer[33] Copyright 2019, American Chemical Society.
| [1] |
Pape A. R., Kaliappan K. P., Kündig E. P., Chem. Rev., 2000, 100, 2917—2940
doi: 10.1021/cr9902852 URL pmid: 11749310 |
| [2] |
Zhuo C. X., Zhang W., You S. L., Angew. Chem. Int. Ed., 2012, 51, 12662—12686
doi: 10.1002/anie.201204822 URL |
| [3] |
Zhuo C. X., Zheng C., You S. L., Acc. Chem. Res., 2014, 47, 2558—2573
doi: 10.1021/ar500167f URL pmid: 24940612 |
| [4] |
Duan D. H., Yin Q., Wang S. G., Gu Q., You S. L., Acta Chim. Sinica, 2014, 72, 1001—1004
doi: 10.6023/A14060497 URL |
|
( 段德河, 殷勤, 王守国, 顾庆, 游书力. 化学学报, 2014, 72, 1001—1004)
doi: 10.6023/A14060497 URL |
|
| [5] |
Wu W. T., Zhang L., You S. L., Chem. Soc. Rev., 2016, 45, 1570—1580
doi: 10.1039/c5cs00356c URL pmid: 26796922 |
| [6] |
Zheng C., You S. L., Chem., 2016, 1, 830—857
doi: 10.1016/j.chempr.2016.11.005 URL |
| [7] |
Wu W. T., Zhang L. M You S. L., Acta Chim. Sinica, 2017, 75, 419
doi: 10.6023/A17020049 URL |
|
( 吴文挺, 张立明, 游书力. 化学学报, 2017, 75, 419—438)
doi: 10.6023/A17020049 URL |
|
| [8] |
Southgate E. H., Holycross D. R., Sarlah D., Angew. Chem. Int. Ed., 2017, 56, 15049—15052
doi: 10.1002/anie.201709712 URL |
| [9] |
Hernandez L. W., Pospech J., Klöeckner U., Bingham T. W., Sarlah D., J. Am. Chem. Soc., 2017, 139, 15656—15659
doi: 10.1021/jacs.7b10351 URL pmid: 29059521 |
| [10] |
Dennis D. G., Okumura M., Sarlah D., J. Am. Chem. Soc., 2019, 141, 10193—10198
URL pmid: 31244190 |
| [11] |
Bingham T. W., Hernandez L. W., Olson D. G., Svec R. L., Hergenrother P. J., Sarlah D., J. Am. Chem. Soc., 2019, 141, 657—670
URL pmid: 30520639 |
| [12] |
Zheng C., You S. L., Nat. Prod. Rep., 2019, 36, 1589—1605
URL pmid: 30839047 |
| [13] |
Boyd D. R., Bugg T. D. H., Org. Biomol. Chem., 2006, 4, 181—192
doi: 10.1039/b513226f URL pmid: 16391757 |
| [14] |
Rosillo M., Domınguez G., Perez-Castells J., Chem. Soc. Rev., 2007, 36, 1589—1604
URL pmid: 17721584 |
| [15] |
Liebov B. K., Harman W. D., Chem. Rev., 2017, 117, 13721—13755
doi: 10.1021/acs.chemrev.7b00480 URL pmid: 29064228 |
| [16] |
Wertjes W. C., Southgate E. H., Sarlah D., Chem. Soc. Rev., 2018, 47, 7996—8017
URL pmid: 30073226 |
| [17] |
Bryce-Smith D., Gilbert A., Tetrahedron, 1976, 32, 1309—1326
doi: 10.1016/0040-4020(76)85002-8 URL |
| [18] | Bryce-Smith D., Gilbert A., Tetrahedron, 1977, 33, 2459—2489 |
| [19] | McCullough J. J., Chem. Rev., 1987, 87, 811—860 |
| [20] |
Remy R., Bochet C. G., Chem. Rev., 2016, 116, 9816—9849
URL pmid: 27340900 |
| [21] | Okumura M., Sarlah D., Eur. J. Org. Chem., 2020, 2020, 1259—1273 |
| [22] |
Dong W., Yuan Y., Xie X., Zhang Z. G., Org. Lett., 2020, 22, 528—532
URL pmid: 31908167 |
| [23] |
Dai W., Geib S. J., Curran D. P., J. Am. Chem. Soc., 2020, 142, 6261—6267
URL pmid: 32101418 |
| [24] |
Guo Y., Nguyen T. V., Koenigs R. M., Org. Lett., 2019, 21, 8814—8818
URL pmid: 31617724 |
| [25] | Hamrock S. J., Sheridan R. S., J. Am. Chem. Soc., 1989, 111, 9247—9249 |
| [26] |
Southgate E. H., Pospech J., Fu J., Holycross D. R., Sarlah D., Nat. Chem., 2016, 8, 922—928
URL pmid: 27657867 |
| [27] |
Hernandez L. W., Klöckner U., Pospech J., Hauss L., Sarlah D., J. Am. Chem. Soc., 2018, 140, 4503—4507
URL pmid: 29544244 |
| [28] |
James M. J., Schwarz J. L., Strieth-Kalthoff F., Wibbeling B., Glorius F., J. Am. Chem. Soc., 2018, 140, 8624—8628
doi: 10.1021/jacs.8b03302 URL |
| [29] |
Zhu M., Zheng C., Zhang X., You S. L., J. Am. Chem. Soc., 2019, 141, 2636—2644
URL pmid: 30653315 |
| [30] |
Cheng Y. Z., Zhao Q. R., Zhang X., You S. L., Angew. Chem. Int. Ed., 2019, 58, 18069—18074
doi: 10.1002/anie.v58.50 URL |
| [31] | Zhu M., Huang X. L., Xu H., Zheng C., Zhang X., You S. L., CCS Chem., 2020, 2, 652—664 |
| [32] | Ma J., Strieth-Kalthoff F., Dalton T., Freitag M., Schwarz J. L., Bergander K., Daniliuc C., Glorius F., Chem., 2019, 5, 2854—2864 |
| [33] |
Soni V. K., Hwang H. S., Moon Y. K., Park S. W., You Y., Cho E. J., J. Am. Chem. Soc., 2019, 141, 10538—10545
URL pmid: 31244191 |
| [1] | 李威, 罗飘, 黄廉湛, 崔志明. 基于聚苯乙烯磺酸的锂金属负极界面保护层的设计[J]. 高等学校化学学报, 2022, 43(8): 20220166. |
| [2] | 葛怡聪, 聂万丽, 孙国峰, 陈稼轩, 田冲. 银催化2-烯基苯胺与苯并异噁唑的[5+1]环化反应[J]. 高等学校化学学报, 2022, 43(8): 20220142. |
| [3] | 刘苏毓, 丁飞, 李茜, 樊春海, 冯景. 偶氮苯类DNA纳米机器[J]. 高等学校化学学报, 2022, 43(8): 20220122. |
| [4] | 高健, 冯奕钰, 方文宇, 王慧, 葛婧, 封伟. 基于低温热释放的烷基接枝相变偶氮苯材料[J]. 高等学校化学学报, 2022, 43(8): 20220146. |
| [5] | 翁美琪, 商桂铭, 王家泰, 李盛华, 樊志, 林松, 郭敏杰. 有机磷神经毒剂分子印迹聚合物的模拟模板分子[J]. 高等学校化学学报, 2022, 43(8): 20220136. |
| [6] | 赵永梅, 穆叶舒, 洪琛, 罗稳, 田智勇. 双萘酰亚胺衍生物用于检测水溶液中的苦味酸[J]. 高等学校化学学报, 2022, 43(3): 20210765. |
| [7] | 陈潇禄, 袁珍闫, 仲迎春, 任浩. 机械球磨制备三苯胺基PAF-106s及C2烃吸附性质[J]. 高等学校化学学报, 2022, 43(3): 20210771. |
| [8] | 伍泽鑫, 朱渊杰, 王泓中, 王均安, 贺英. 甲基修饰的咔唑/二苯砜基AIE-TADF蓝光材料及其OLED器件[J]. 高等学校化学学报, 2022, 43(11): 20220371. |
| [9] | 张伶育, 张继龙, 曲泽星. RDX分子内振动能量重分配的动力学研究[J]. 高等学校化学学报, 2022, 43(10): 20220393. |
| [10] | 周永慧, 黄如军, 严健洋, 李亚军, 邱欢欢, 杨进轩, 郑佑轩. 两种基于氮杂环结构铱(Ⅲ)配合物的合成及有机电致发光性能[J]. 高等学校化学学报, 2022, 43(1): 20210415. |
| [11] | 左怀龙, 雷思敏, 张锐, 李玉新, 陈伟. 新型异喹啉衍生物的设计合成及抑菌活性[J]. 高等学校化学学报, 2021, 42(9): 2766. |
| [12] | 胡串串, 庞靖祥, 贺闯闯, 李伟, 孙书涛. Sc(OTf)3催化δ⁃腈基对亚甲基苯醌的1,6⁃共轭烯丙基化: 烯丙基二芳基乙腈类化合物的合成[J]. 高等学校化学学报, 2021, 42(9): 2805. |
| [13] | 彭小明, 吴健群, 戴红玲, 杨展宏, 许莉, 许高平, 胡锋平. Ni-N-C单原子催化剂活化过硫酸盐降解苯酚[J]. 高等学校化学学报, 2021, 42(8): 2581. |
| [14] | 付志男, 谈云龙, 肖谷雨, 颜德岳. 含全氟联苯结构的磺化聚二氮杂萘酮醚氧膦质子交换膜的制备与性能[J]. 高等学校化学学报, 2021, 42(8): 2635. |
| [15] | 谢璠, 陈珊珊, 卓龙海, 陆赵情, 高坤, 代啓阳. 聚对二甲苯纳米纤维阵列的CVD液晶模板法制备及降解性能[J]. 高等学校化学学报, 2021, 42(8): 2643. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||