

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (1): 74.doi: 10.7503/cjcu20200461
所属专题: 分子筛功能材料 2021年,42卷,第1期
邹润1,董霄1,矫义来2,Carmine D'AGOSTINO1,闫文付3,范晓雷1
收稿日期:2020-07-16
出版日期:2021-01-10
发布日期:2021-01-12
基金资助:
ZOU Run1, DONG Xiao1, JIAO Yilai2, Carmine D'AGOSTINO1, YAN Wenfu3, FAN Xiaolei1( )
)
Received:2020-07-16
Online:2021-01-10
Published:2021-01-12
Contact:
FAN Xiaolei
E-mail:xiaolei.fan@manchester.ac.uk
Supported by:摘要:
等级孔分子筛是一类具有两种或多种以特定形式排布的孔结构的分子筛材料. 多层等级的孔结构使得分子筛孔道内的分子扩散得到显著改善, 进而提升了其在吸附和非均相催化等领域的应用性能. 等级孔分子筛的制备策略通常有两种, 即“自上而下”后处理法(如对母样分子筛进行脱铝、 脱硅产生介孔)和“自下而上”合成法(如软模板、 硬模板法). 本文主要对近20年来等级孔分子筛的合成方法进行了梳理, 并着重介绍了具有较高应用潜力的“自上而下”制备法. 鉴于合成等级孔分子筛的主要目的是提高分子的晶内扩散, 对近年来客体分子在等级孔分子筛内扩散的实验研究也进行了简要综述. 此外, 本文还综合评述了等级孔分子筛与传统分子筛在催化应用中的对比, 以展示前者在提升催化性能方面(如活性、 选择性等)的独特优势.
中图分类号:
TrendMD:
邹润, 董霄, 矫义来, Carmine, 闫文付, 范晓雷. 等级孔分子筛的可控合成、 扩散研究及催化应用. 高等学校化学学报, 2021, 42(1): 74.
ZOU Run, DONG Xiao, JIAO Yilai, Carmine D'AGOSTINO, YAN Wenfu, FAN Xiaolei. Controllable Synthesis, Diffusion Study and Catalysis of Hierarchical Zeolites. Chem. J. Chinese Universities, 2021, 42(1): 74.
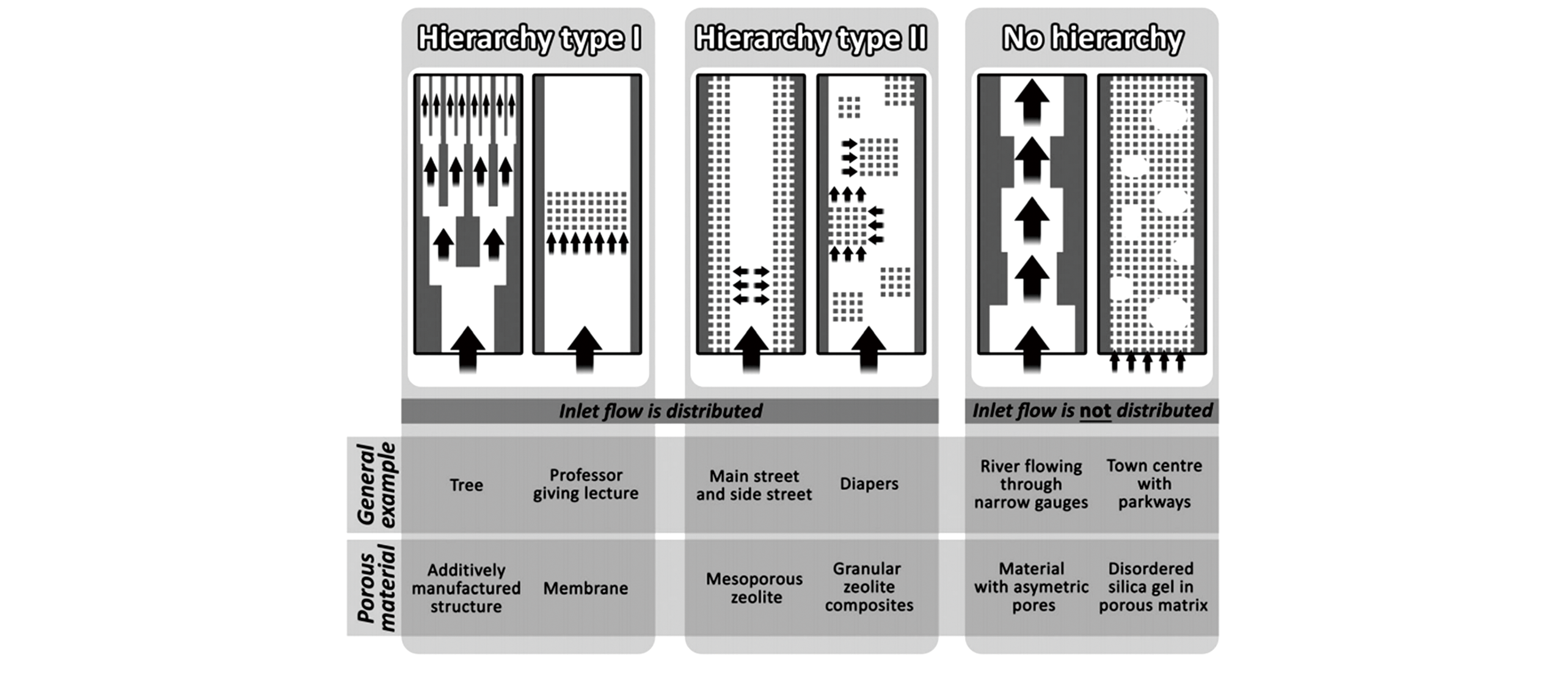
Fig.1 Schematics of the classification for different meso?/micro?pore systems in zeolitic materials[14]Copyright 2016, the Royal Society of Chemistry.
| Type | Template | Framework | Information of porestructure | Ref. |
|---|---|---|---|---|
Hard template | Carbon nanoparticles | MFI(ZSM?5) | Intracrystalline; ca. 12. 5 or 34. 5 nm | [ |
| Carbon aerogel | MFI(ZSM?5) | Intracrystalline; 9―25 nm | [ | |
| Carbon nanotube | MFI(ZSM?5), FAU(Y) | Intracrystalline; 20―30 nm | [ | |
| 3DOM carbon | MFI(silicalite?1) | Intracrystalline; ca. 6 nm | [ | |
| BEA(Beta) | Intracrystalline; 32 nm and 95 nm | [ | ||
| CaCO3 | MFI(silicalite?1) | Intracrystalline; 50―100 nm | [ | |
| Polystyrene bead | MFI(silicalite?1) | Core?shell; >200 nm | [ | |
| Polyurethane | MFI(TS?1) | Intracrystalline; ca. 4 nm | [ | |
| Wood cell | MFI(silicalite?1) | Intracrystalline; 2―20 μm | [ | |
Soft templates | Organosilane(APTMS, IBTES, PHAPTMS and ODTMS) | MFI(ZSM?5) | Intercrystalline; 2―8 nm | [73―75] |
| Amino acid(e. g. lysine) | MFI(ZSM?5) | Intracrystalline; ca. 40 nm | [ | |
| MFI(TS?1) | Intracrystalline and intercrystalline; 30―70 nm | [ | ||
| LTA | Intracrystalline; ca. 14 or 25 nm | [ | ||
| FAU(Y) | Intracrystalline; 19―24 nm | [ | ||
| Amphiphilic silane(TPHAC) | MFI(ZSM?5) | Intracrystalline; 2. 1―7. 4 nm | [ | |
| Carbohydrate(glucose and starch) | MFI(ZSM?5) | Intracrystalline; 2―25 nm | [ | |
| MFI(ZSM?5) | Intracrystalline; 2―50 nm | [ | ||
| Dual?functional template(C22?6?6) | MFI(ZSM?5) | Intracrystalline; ca. 15 nm | [ | |
| Silanised polymer(PEI) | MFI(ZSM?5) | Intracrystalline; 2―5 nm | [ | |
| Cationic polymer(PDADMAC) | BEA(Beta) | Intracrystalline; 2―50 nm | [ |
Table 1 Preparation of hierarchical zeolites via the bottom-up templating methods
| Type | Template | Framework | Information of porestructure | Ref. |
|---|---|---|---|---|
Hard template | Carbon nanoparticles | MFI(ZSM?5) | Intracrystalline; ca. 12. 5 or 34. 5 nm | [ |
| Carbon aerogel | MFI(ZSM?5) | Intracrystalline; 9―25 nm | [ | |
| Carbon nanotube | MFI(ZSM?5), FAU(Y) | Intracrystalline; 20―30 nm | [ | |
| 3DOM carbon | MFI(silicalite?1) | Intracrystalline; ca. 6 nm | [ | |
| BEA(Beta) | Intracrystalline; 32 nm and 95 nm | [ | ||
| CaCO3 | MFI(silicalite?1) | Intracrystalline; 50―100 nm | [ | |
| Polystyrene bead | MFI(silicalite?1) | Core?shell; >200 nm | [ | |
| Polyurethane | MFI(TS?1) | Intracrystalline; ca. 4 nm | [ | |
| Wood cell | MFI(silicalite?1) | Intracrystalline; 2―20 μm | [ | |
Soft templates | Organosilane(APTMS, IBTES, PHAPTMS and ODTMS) | MFI(ZSM?5) | Intercrystalline; 2―8 nm | [73―75] |
| Amino acid(e. g. lysine) | MFI(ZSM?5) | Intracrystalline; ca. 40 nm | [ | |
| MFI(TS?1) | Intracrystalline and intercrystalline; 30―70 nm | [ | ||
| LTA | Intracrystalline; ca. 14 or 25 nm | [ | ||
| FAU(Y) | Intracrystalline; 19―24 nm | [ | ||
| Amphiphilic silane(TPHAC) | MFI(ZSM?5) | Intracrystalline; 2. 1―7. 4 nm | [ | |
| Carbohydrate(glucose and starch) | MFI(ZSM?5) | Intracrystalline; 2―25 nm | [ | |
| MFI(ZSM?5) | Intracrystalline; 2―50 nm | [ | ||
| Dual?functional template(C22?6?6) | MFI(ZSM?5) | Intracrystalline; ca. 15 nm | [ | |
| Silanised polymer(PEI) | MFI(ZSM?5) | Intracrystalline; 2―5 nm | [ | |
| Cationic polymer(PDADMAC) | BEA(Beta) | Intracrystalline; 2―50 nm | [ |
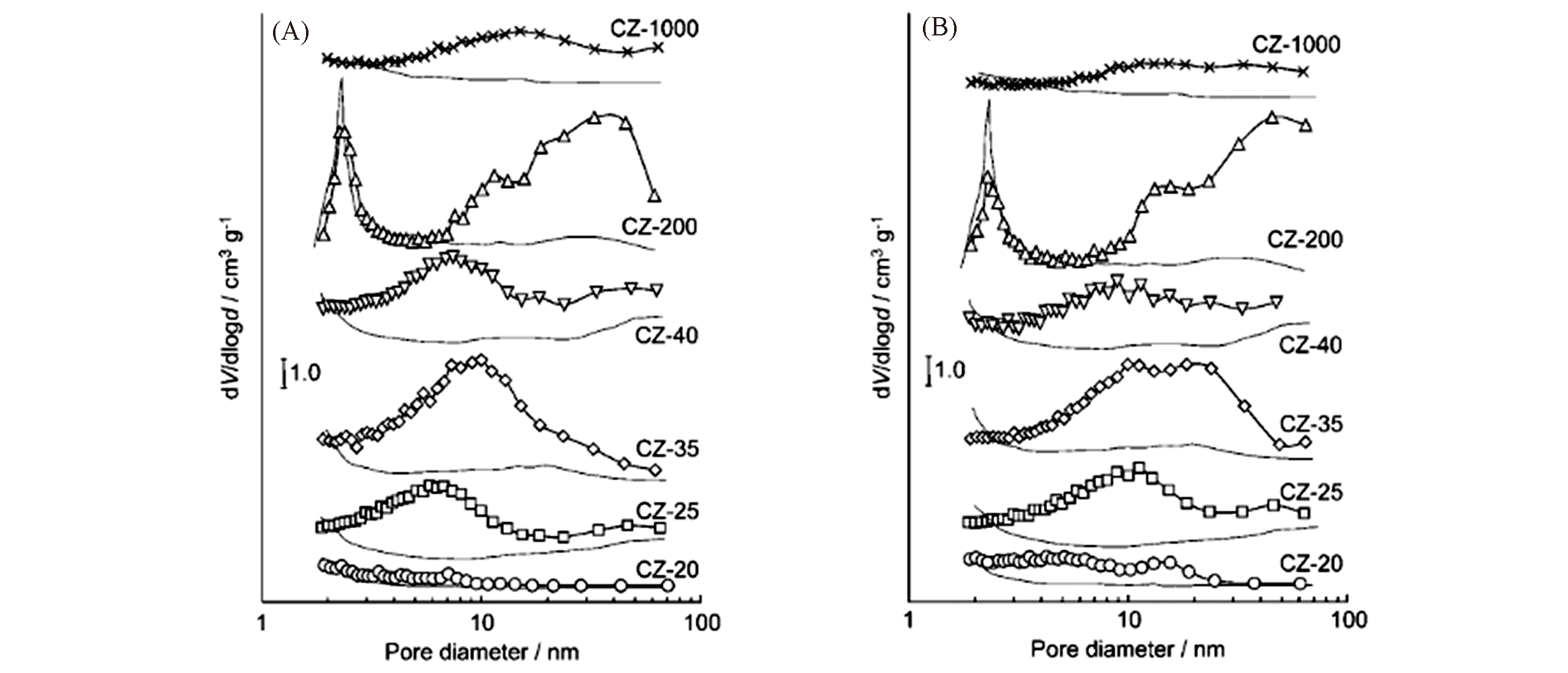
Fig.3 Pore size distribution(based on the Barrett?Joyner?Halenda, BJH, method) of the alkaline?treated commercial ZSM?5 zeolites[96]Alkaline treatment in 0.2 mol/L NaOH for 30 min at 65 ℃(A) and 85 ℃(B).Copyright 2005, Wiley?VCH Verlay GmbH&Co. kGaA, Weinheim.

Fig.4 SEM(A, B) and TEM(C, D) images of pristine ZSM?5(A, C) and mesoporous ZSM?5(B, D) treated by sequential steaming and desilication(steaming at 400 °C for 3 h and desilication at 80 °C with 0.2 mol/L NaOH for 30 min)[51]Copyright 2017, Wiley?VCH Verlay GmbH&Co. kGaA, Weinheim.

Fig.5 TEM images of parent ZSM?5(SAR=37) and mesoporous ZSM?5 obtained by sequential desilication and dealumination treatments with different acid and base concentration[58]Copyright 2011, American Chemical Society.
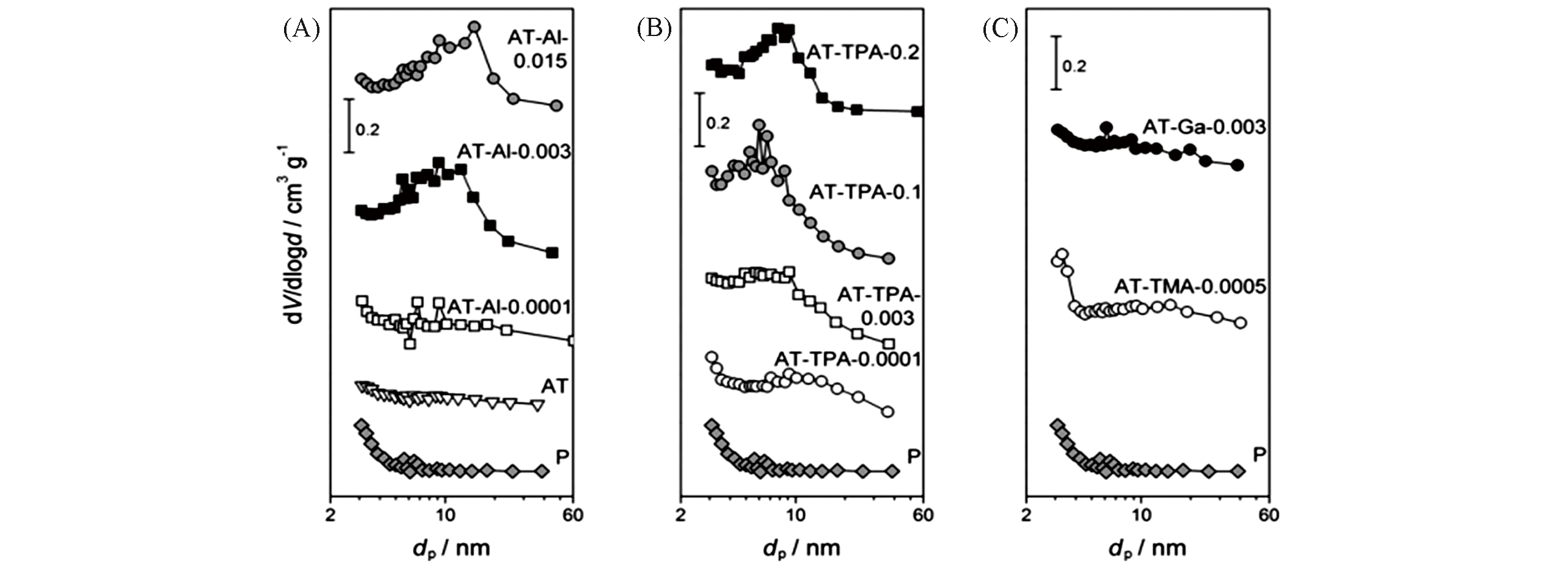
Fig.6 BJH pore size distributions of the parent and alkaline?treated silicalite?1 zeolites derived from the N2 isotherms[57]Pore?directing agent: (A) Al(OH)4+ ; (B) TPA+; (C) Ga(OH)4+ or TMA+.Copyright 2011, Wiley?VCH Verlay GmbH&Co. kGaA, Weinheim.

Fig.7 Sequential dealumination and desilication of NH4?Y zeolite[37](A) Parent Y; (B) DA2?AT2: dealuminated(0.11 mol/L EDTA) and desilicated Y(0.2mol/L NaOH); (C) DA2?AT2?AW1: dealuminated, desilicated and acid?washed Y.Copyright 2011, Wiley?VCH Verlay GmbH&Co. kGaA, Weinheim.
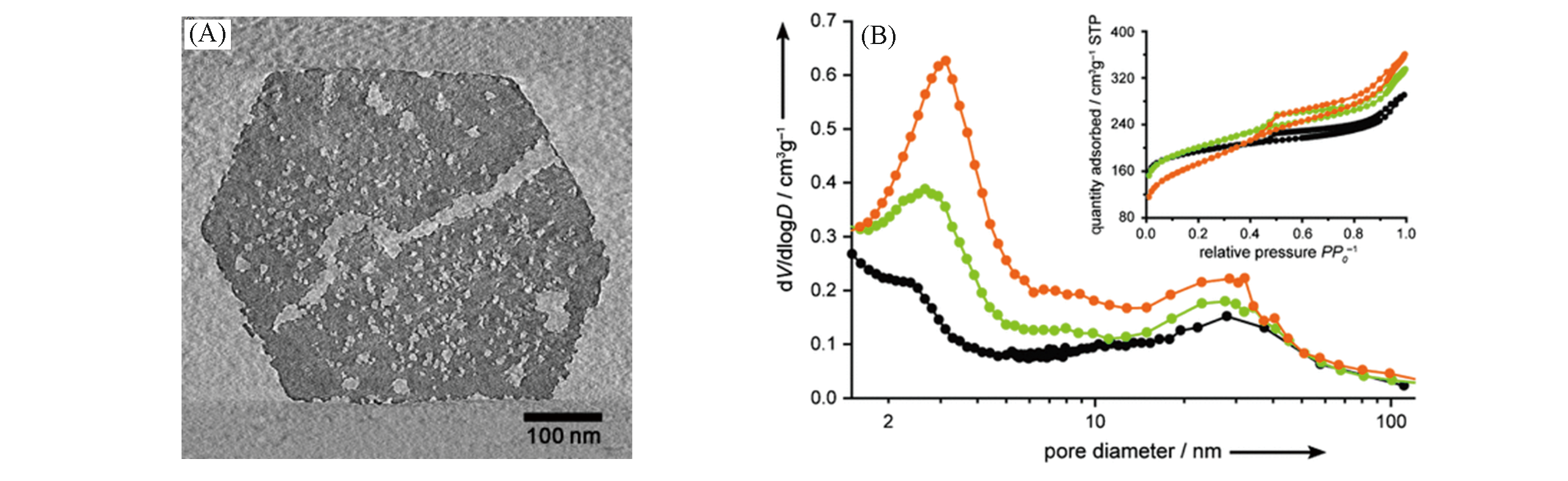
Fig.8 TEM image of desilicated USY(SAR=30) by diluted NaOH solution(0.05 mol/L, 15 min)(A) and the resulting trimodal porosity of the treated zeolite(B)[47]Copyright 2010, Wiley?VCH Verlay GmbH&Co. kGaA, Weinheim.
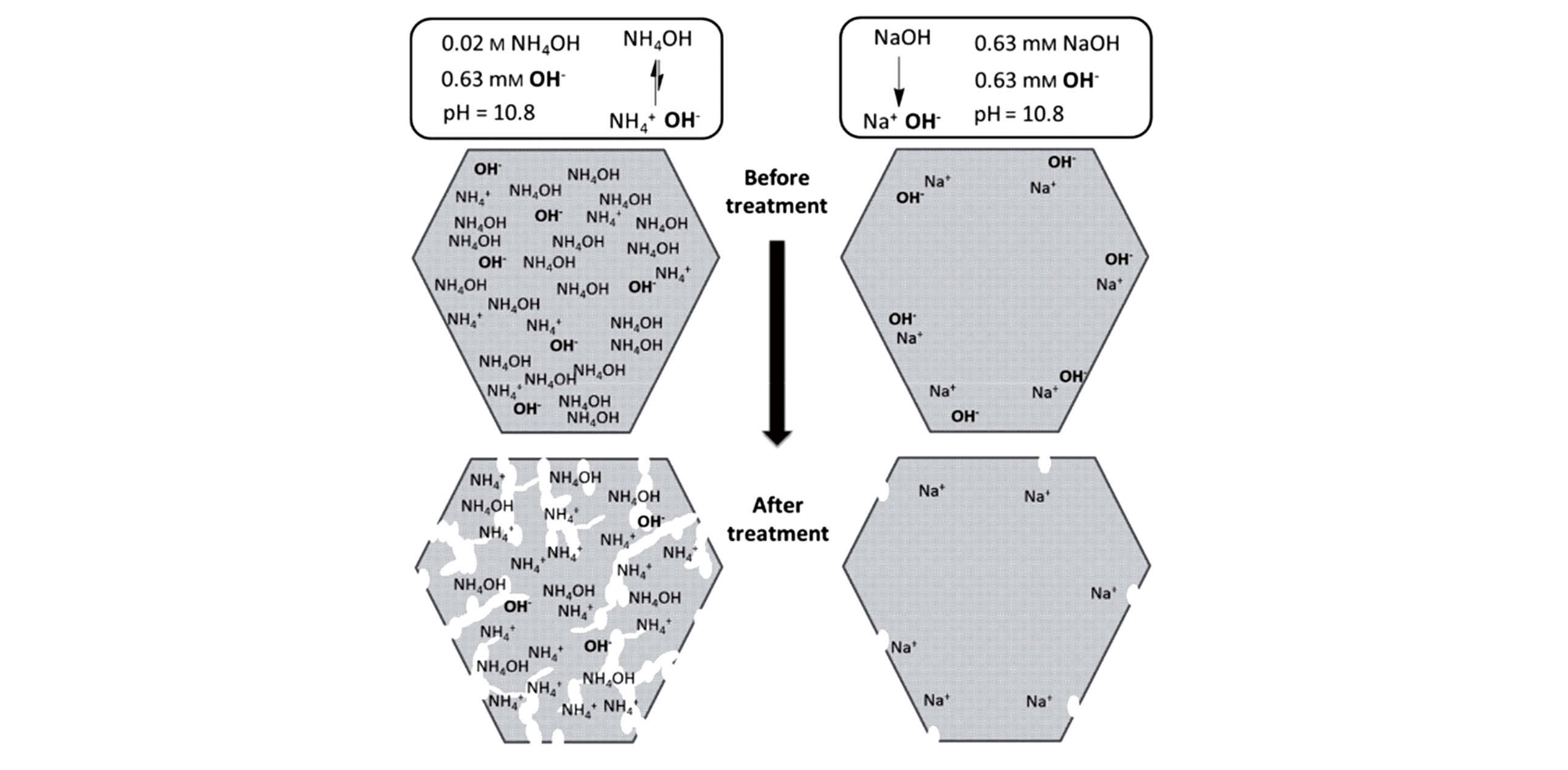
Fig.9 Comparative desilication of USY(SAR=40) by NH4OH and NaOH solutions with the same molar amount of OH-[42]Copyright 2015, Wiley?VCH Verlay GmbH&Co. kGaA, Weinheim.
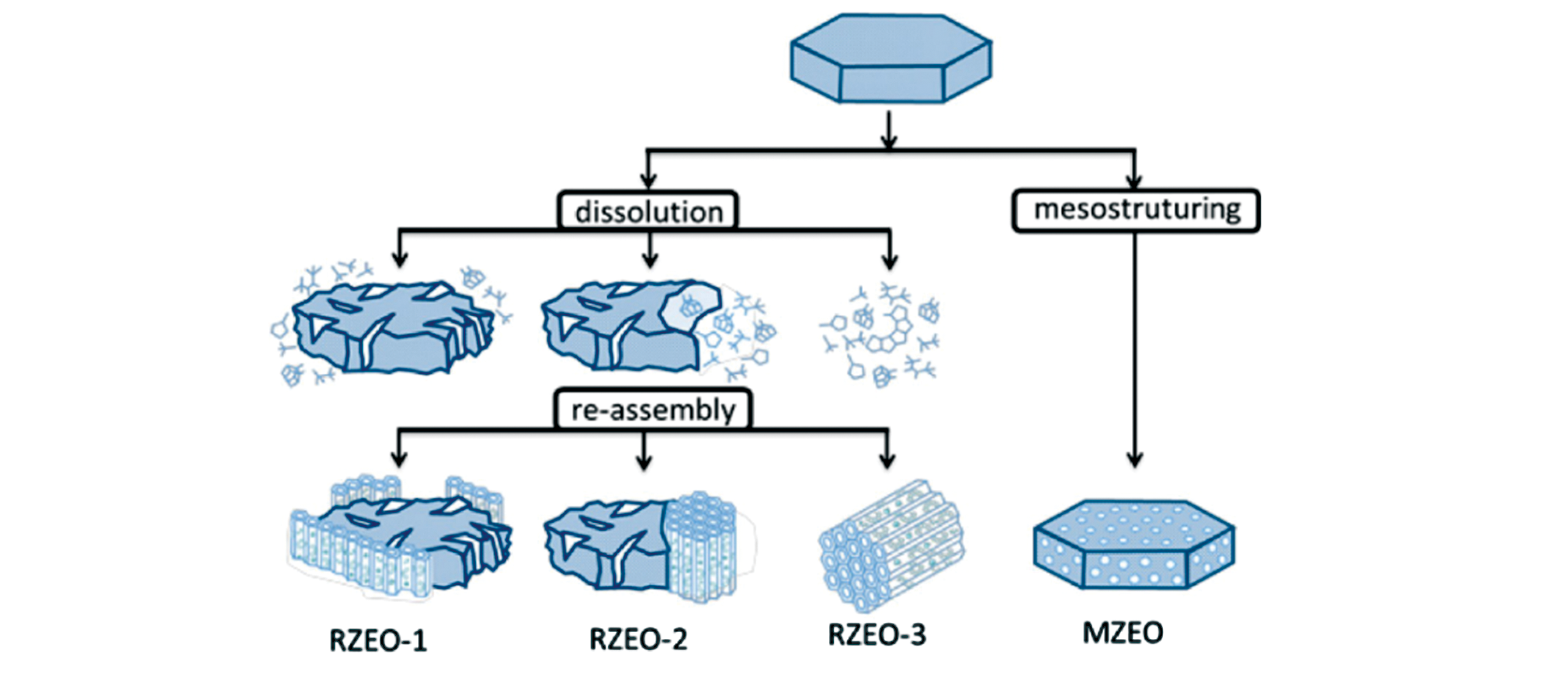
Fig.10 Recrystallisation?induced mesoporous materials RZEO(micro?meso?porous composites) and MZEO(one?phase hybrid single zeolite crystals)[20]Copyright 2017, American Chemical Society.
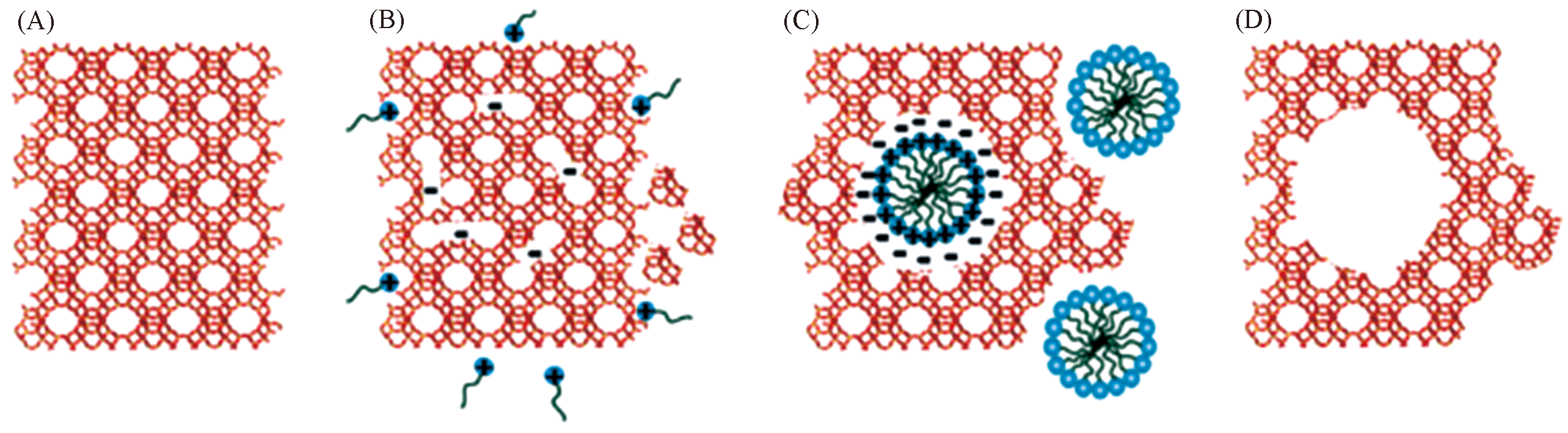
Fig.11 Mesostructuring of USY zeolite by surfactant?templated recrystallisation[46](A) Parent USY zeolites; (B) insertion of surfactants into zeolite framework; (C) rearrangement of crystal structure in USY; (D) template removal by calcination to render mesopores.Copyright 2012, the Royal Society of Chemistry.
| Topology | SAR | Treatment | Vmicro/ (cm3·g-1) | Vmeso/ (cm3·g-1) | Sext./ (m2·g-1) | Dmeso/nm | Ref. |
|---|---|---|---|---|---|---|---|
| ZSM?5 | 14 | ―* | 0.12 | 0.14 | 124 | ― | [ |
| 10―11 | Steaming+desilication | 0.1 | 0.32 | 209 | 5 | ||
| ZSM?5 | 15 | ― | 0.14 | 0.15 | 76 | ― | [ |
| ― | Desilication+acid wash(HCl) | 0.15 | 0.4 | 158 | 3 and 35 | ||
| ZSM?5 | 25 | ― | 0.16 | 0.10 | 35 | 7―10 | [ |
| 18 | Desilication | 0.13 | 0.31 | 195 | |||
| ZSM?5 | 40 | ― | 0.16 | 0.14 | 45 | ||
| 29 | Desilication | 0.13 | 0.61 | 225 | |||
| ZSM?5 | 40 | ― | 0.17 | 0.11 | 78 | ― | [ |
| 32 | Desilication(TPA) | 0.12 | 0.36 | 272 | 5 | ||
| Topology | SAR | Treatment | Vmicro/ (cm3·g-1) | Vmeso/ (cm3·g-1) | Sext./ (m2·g-1) | Dmeso/nm | Ref. |
| ZSM?5 | +∞ | 0.16 | 0.25 | 9 | [ | ||
| ― | Desilication(TPA) | 0.1 | 0.42 | 287 | ― | ||
| Y | 2. 6 | ― | 0.3 | 0.04 | 22 | ― | [ |
| < 6 | Dealumination(EDTA)+desilication | 0.2 | 0.46 | 330 | 8 | ||
| Y | 2. 55 | ― | 0.38 | 0.03 | 22 | ― | [ |
| ― | Acid wash+recrystallization(CTAB) | 0.37 | 0.16 | 243 | ― | ||
| USY | 15 | ― | 0.3 | 0.16 | 168 | ― | [ |
| ― | Recrystallization(CTAB) | 0.21 | 0.5 | 704 | 4 | ||
| USY | 15 | ― | 0.28 | 0.23 | 125 | ― | [ |
| ca. 11 | Desilication(TPA) | 0.29 | 0.42 | 253 | 5 | ||
| USY | 30 | ― | 0.33 | 0.28 | 117 | ― | [ |
| ca. 19 | Desilication(TPA) | 0.26 | 0.84 | 500 | 8 | ||
| USY | 30 | ― | 0.21 | 0.16 | 213 | 28 | [ |
| ca. 25 | Mild desilication(NaOH) | 0.16 | 0.25 | 339 | 2.7 and 27 | ||
| USY | 40 | ― | 0.27 | 0.27 | 181 | 6―30 | [ |
| ca. 40 | Mild desilication(NH4OH) | 0.16 | 0.4 | 311 | 2―6 and 6―30 |
Table 2 Porous properties of hierarchical zeolites made by different post-synthetic treatments
| Topology | SAR | Treatment | Vmicro/ (cm3·g-1) | Vmeso/ (cm3·g-1) | Sext./ (m2·g-1) | Dmeso/nm | Ref. |
|---|---|---|---|---|---|---|---|
| ZSM?5 | 14 | ―* | 0.12 | 0.14 | 124 | ― | [ |
| 10―11 | Steaming+desilication | 0.1 | 0.32 | 209 | 5 | ||
| ZSM?5 | 15 | ― | 0.14 | 0.15 | 76 | ― | [ |
| ― | Desilication+acid wash(HCl) | 0.15 | 0.4 | 158 | 3 and 35 | ||
| ZSM?5 | 25 | ― | 0.16 | 0.10 | 35 | 7―10 | [ |
| 18 | Desilication | 0.13 | 0.31 | 195 | |||
| ZSM?5 | 40 | ― | 0.16 | 0.14 | 45 | ||
| 29 | Desilication | 0.13 | 0.61 | 225 | |||
| ZSM?5 | 40 | ― | 0.17 | 0.11 | 78 | ― | [ |
| 32 | Desilication(TPA) | 0.12 | 0.36 | 272 | 5 | ||
| Topology | SAR | Treatment | Vmicro/ (cm3·g-1) | Vmeso/ (cm3·g-1) | Sext./ (m2·g-1) | Dmeso/nm | Ref. |
| ZSM?5 | +∞ | 0.16 | 0.25 | 9 | [ | ||
| ― | Desilication(TPA) | 0.1 | 0.42 | 287 | ― | ||
| Y | 2. 6 | ― | 0.3 | 0.04 | 22 | ― | [ |
| < 6 | Dealumination(EDTA)+desilication | 0.2 | 0.46 | 330 | 8 | ||
| Y | 2. 55 | ― | 0.38 | 0.03 | 22 | ― | [ |
| ― | Acid wash+recrystallization(CTAB) | 0.37 | 0.16 | 243 | ― | ||
| USY | 15 | ― | 0.3 | 0.16 | 168 | ― | [ |
| ― | Recrystallization(CTAB) | 0.21 | 0.5 | 704 | 4 | ||
| USY | 15 | ― | 0.28 | 0.23 | 125 | ― | [ |
| ca. 11 | Desilication(TPA) | 0.29 | 0.42 | 253 | 5 | ||
| USY | 30 | ― | 0.33 | 0.28 | 117 | ― | [ |
| ca. 19 | Desilication(TPA) | 0.26 | 0.84 | 500 | 8 | ||
| USY | 30 | ― | 0.21 | 0.16 | 213 | 28 | [ |
| ca. 25 | Mild desilication(NaOH) | 0.16 | 0.25 | 339 | 2.7 and 27 | ||
| USY | 40 | ― | 0.27 | 0.27 | 181 | 6―30 | [ |
| ca. 40 | Mild desilication(NH4OH) | 0.16 | 0.4 | 311 | 2―6 and 6―30 |
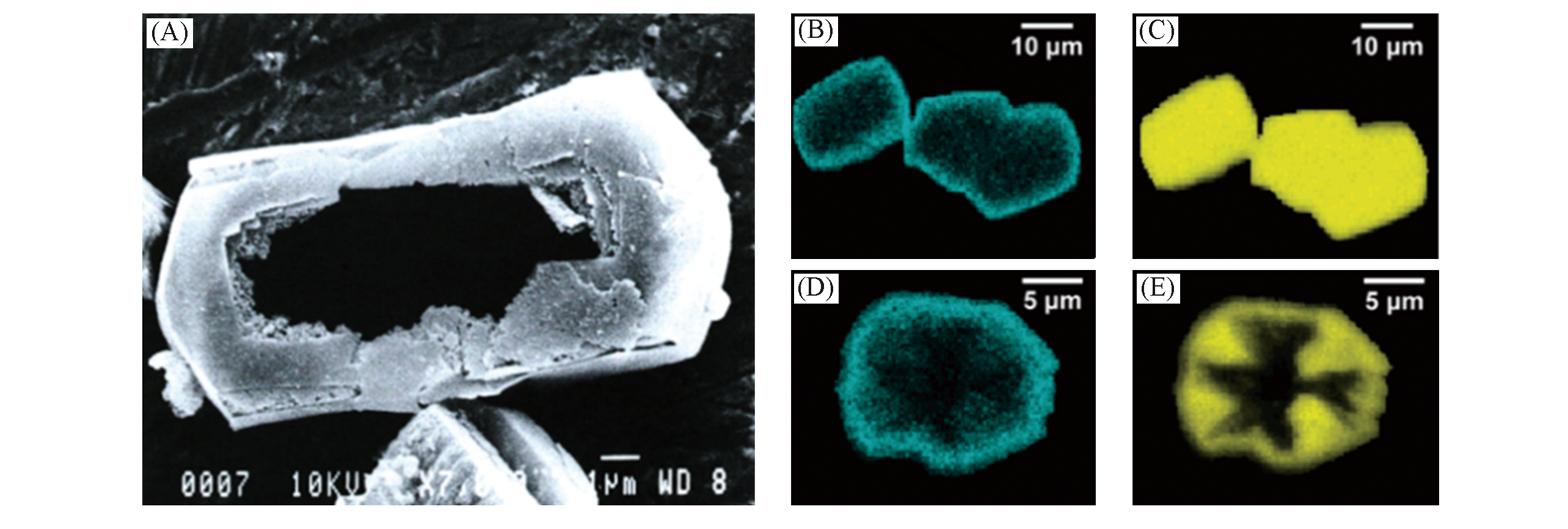
Fig.12 Al?zoned ZSM?5 to form hollow structures after desilication[59](A) SEM graphs of hollow ZSM?5 after alkaline treatment, Si/Al=75, under reflux in 0.5 mol/L Na2CO3 for 16 h; (B, D) SEM?EDX mappings of aluminium in large ZSM?5 crystal before(B) and after(D) alkaline treatment; (C, E) SEM?EDX mappings of silicon in large ZSM?5 crystal before(C) and after(E) alkaline treatment.Copyright 2006, the Royal Society of Chemistry.
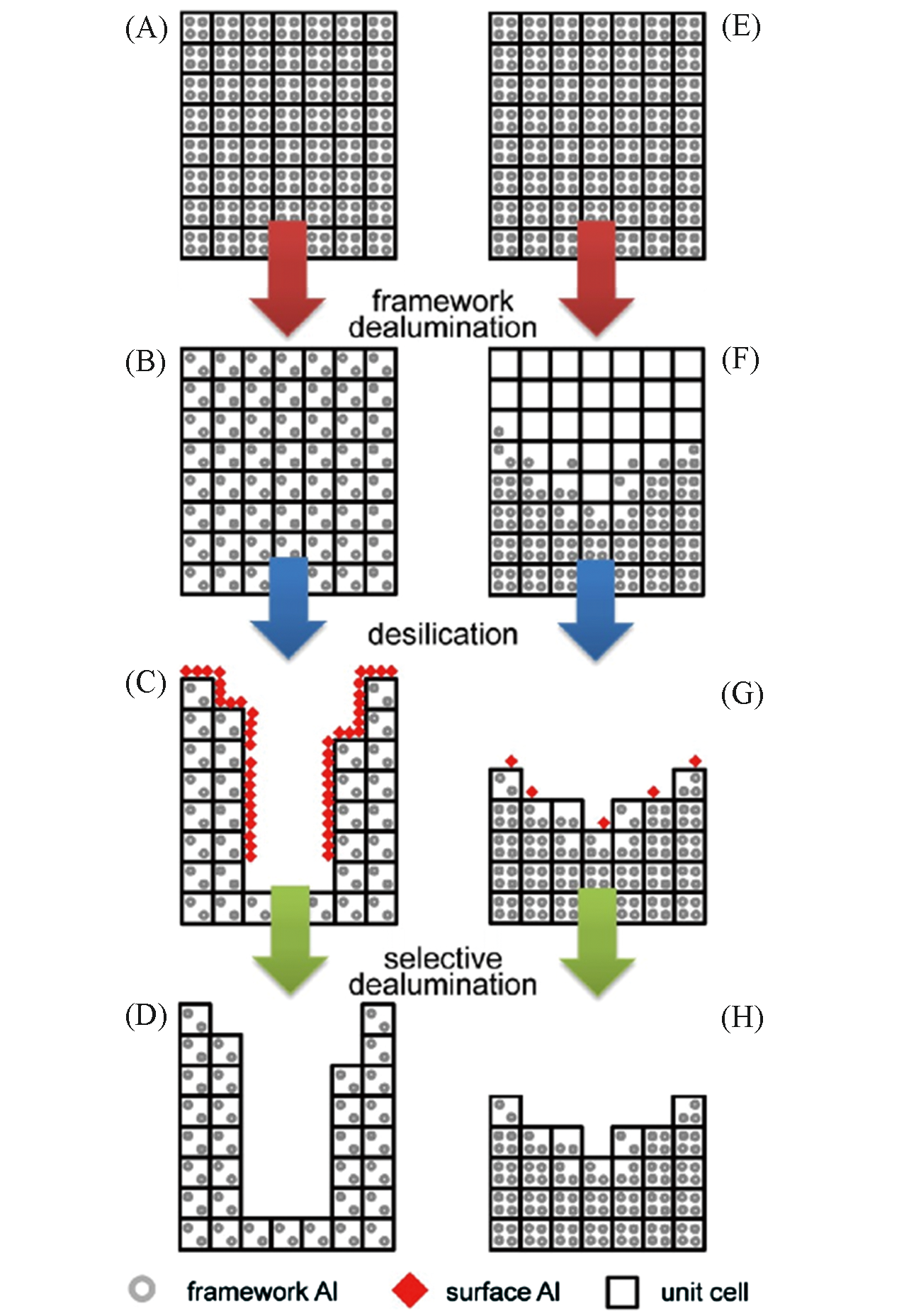
Fig.13 Schematic representation of two different sequential acid?base?acid treatments[105](A, E) parent zeolite; (B) uniform dealumination; (C) uniform dealumination promotes the effective desilication; (D, H) removal of the surface alumina debris by acid washing; (F) uneven dealumination creates Al?zoning; (G) uneven dealumination hinders the subsequent desilication.Copyright 2013, American Chemical Society.

Fig.14 TEM images of MOR zeolite before and after acid and/or chemical treatment[107](A) Parent MOR; (B) MOR treated with oxalic acid; (C) MOR treated with NH4F; (D) MOR treated with oxalic acid and NH4F; (E) larger magnification of (D).Copyright 2020, the Royal Society of Chemistry.
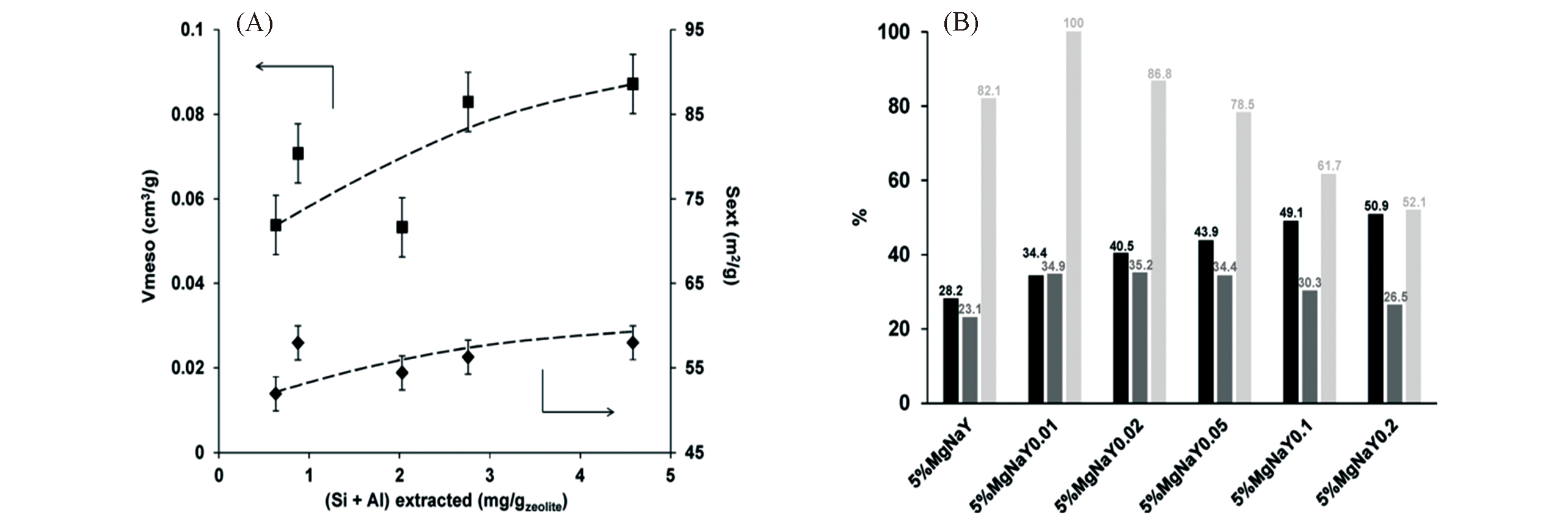
Fig.16 Comparison of mesoporous features(regarding Vmeso and Sext.) of the Na?Y parent zeolite and desilicated Y zeolites(A) and glucose conversion(black), fructose yield(grey) and selectivity to fructose(light grey) from the comparative catalysis over the parent and hierarchical Mg/Na?Y zeolites at 100 °C after 2 h(B)[111]Copyright 2018, Elsevier B. V.
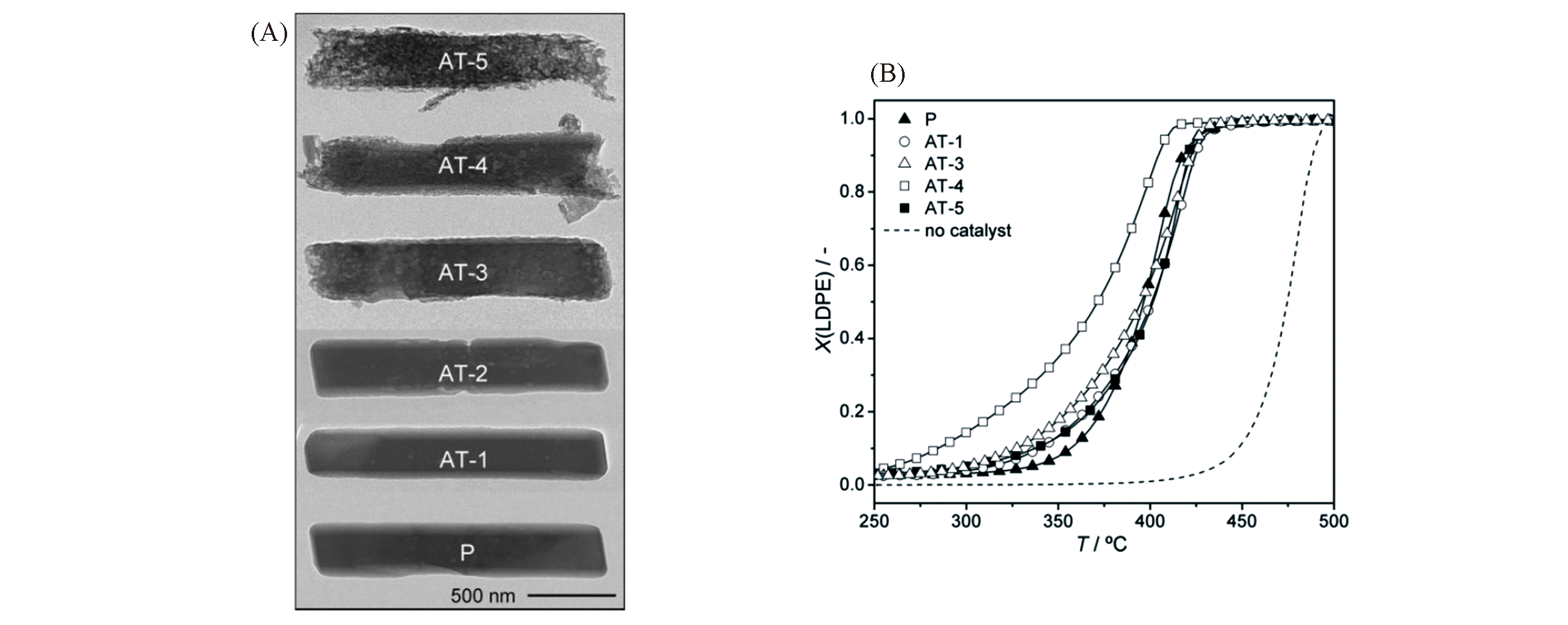
Fig.17 TEM images of the parent(P) and alkaline?treated(AT) ITQ?4 zeolites(A) and conversion(X) of LDPE as a function of reaction temperature in catalytic pyrolysis over the ITQ?4 zeolites(B)[112]Copyright 2010, Wiley?VCH Verlay GmbH&Co. kGaA, Weinheim.
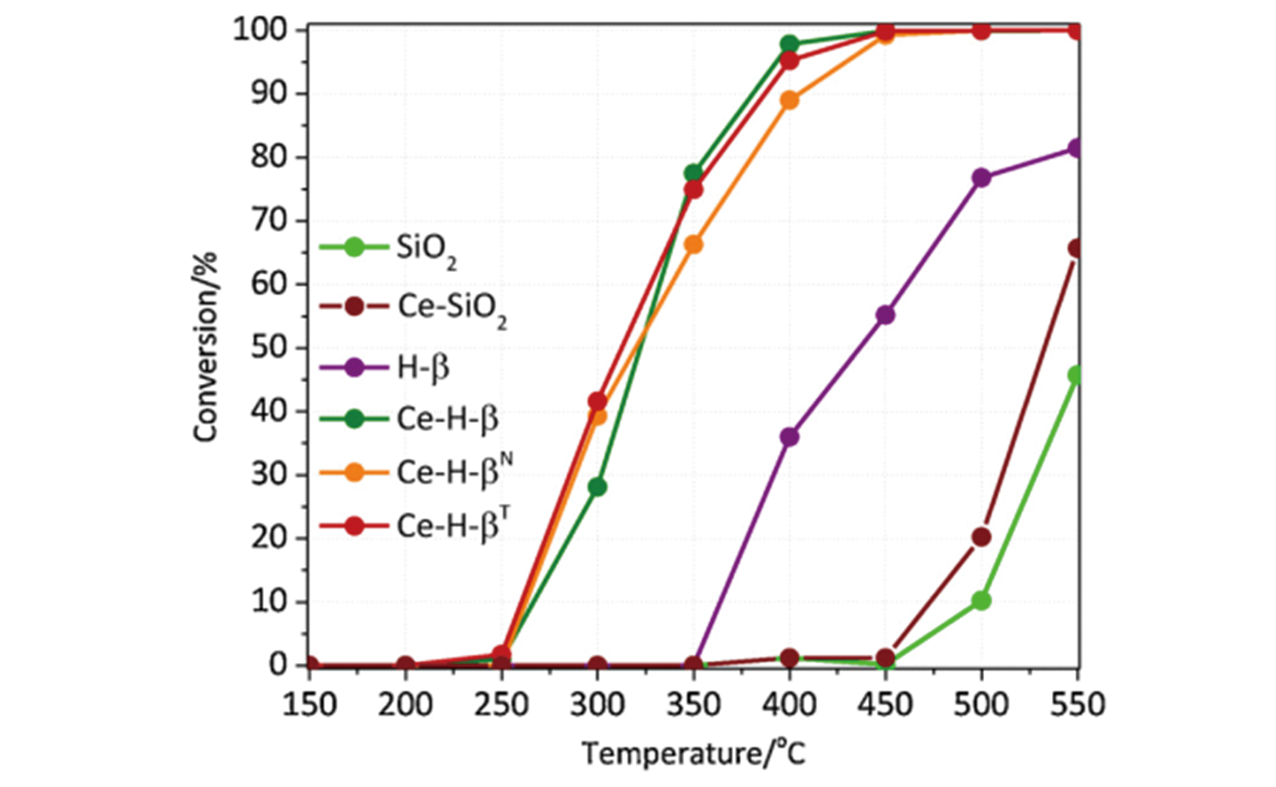
Fig.18 Trichloroethylene conversion as a function of temperature over the Ce supported on silica and beta zeolite catalysts[113]Copyright 2019, Elsevier B. V.
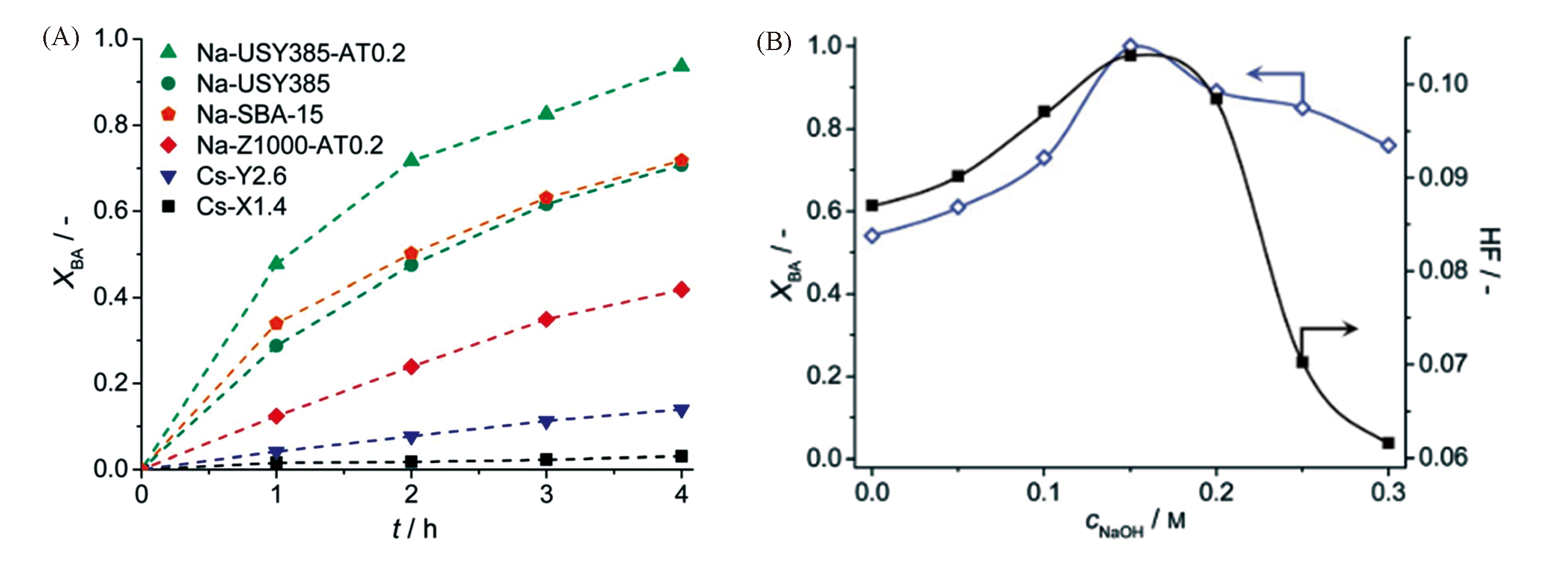
Fig.19 Benzaldehyde conversion(XBA) in the Knoevenagel condensation over different catalysts as a function of reaction time(A) and effect of NaOH concentration in desilication on XBA and hierarchy factor(HF) for desilicated USY385 zeolites(B)[114]Copyright 2014, the Royal Society of Chemistry.
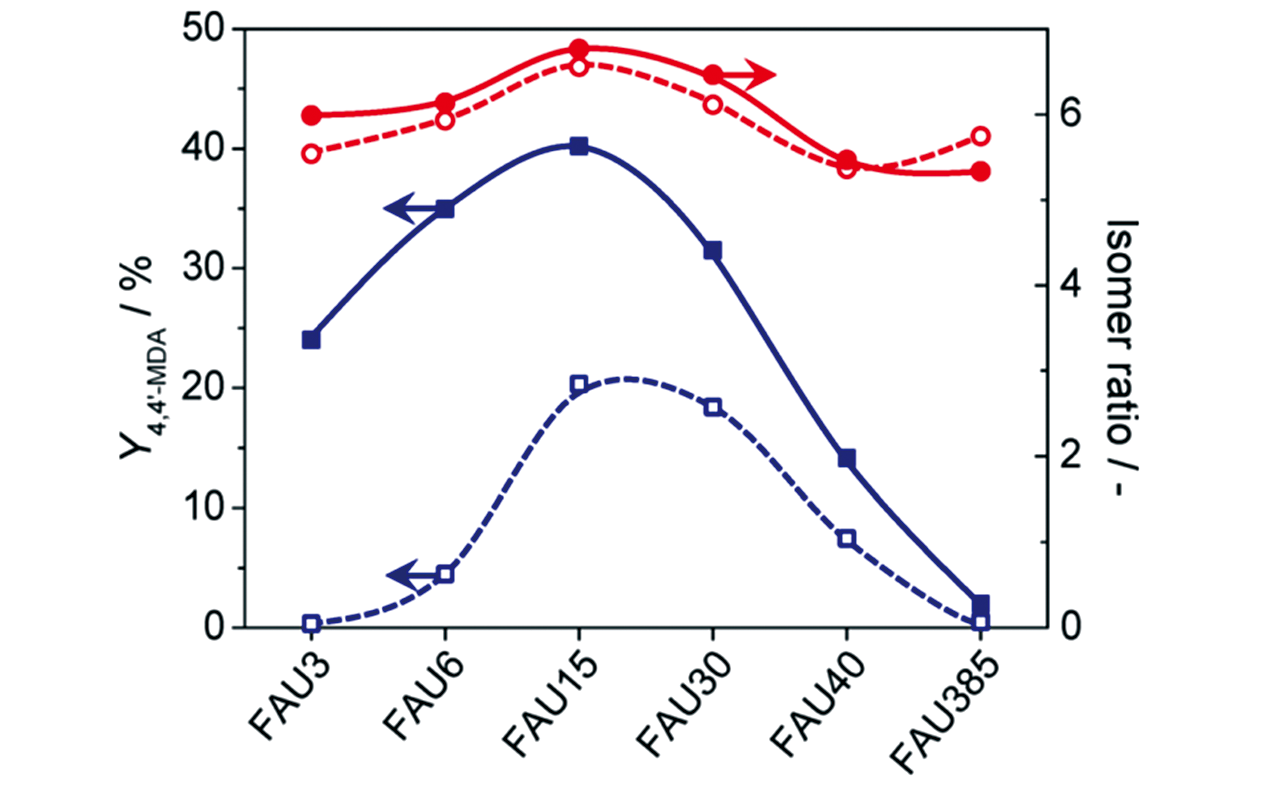
Fig.20 Yield of 4,4′?MDA(blue squares) and isomer ratio(red circles) over the parent(open symbols) and relevant hierarchical(full symbols) FAU zeolites[100]Copyright 2014, American Chemical Society.
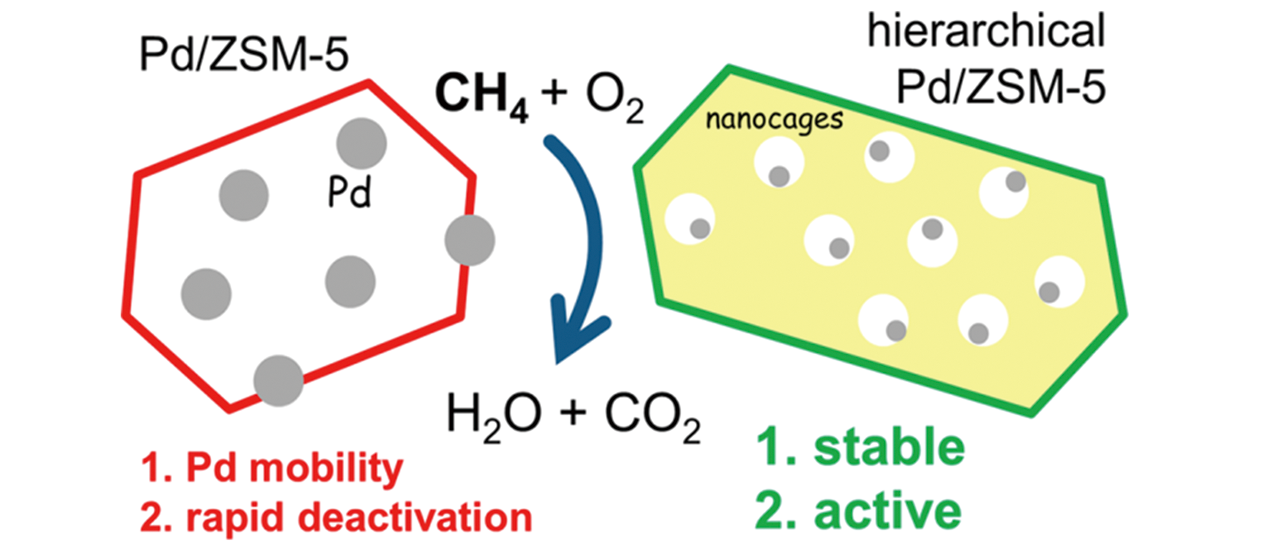
Fig.21 Schematic illustration of improving the stability of Pd nanoparticles supported on hierarchical ZSM?5 zeolites[49]Copyright 2019, American Chemical Society.

Fig.22 TEM(A) and STEM(B) images of Au/Recryst?S1 and size distribution of Au nanoparticles based on measurements of approximately 250 nanoparticles by TEM(C)[116]Copyright 2014, WILEY?VCH Verlay GmbH&Co. kGaA, Weinheim.
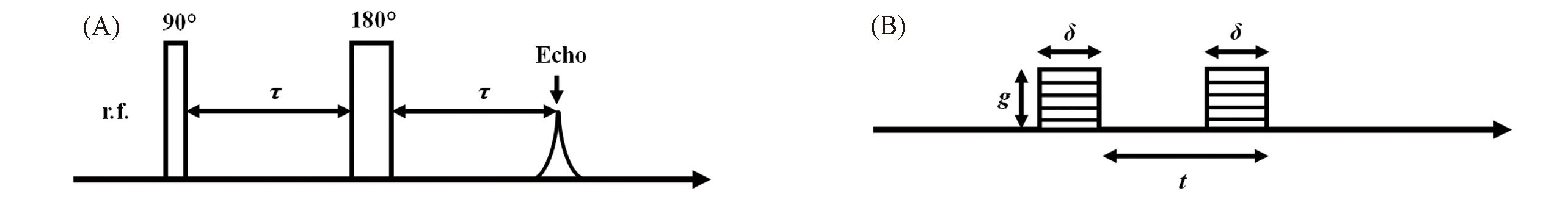
Fig.23 RF pulses for generating Hahn echo (A) and the field gradient pulses applied for self?diffusion measurement(B)(A) Thin and thick bars represent 90° and 180° RF pulses, respectively; (B) δ, pulse duration; g, magnitude of the gradient pulse; t, the time interval between the two gradient pulses.Ψ=S/S0=exp[-Dγ2g2δ2t] (1)
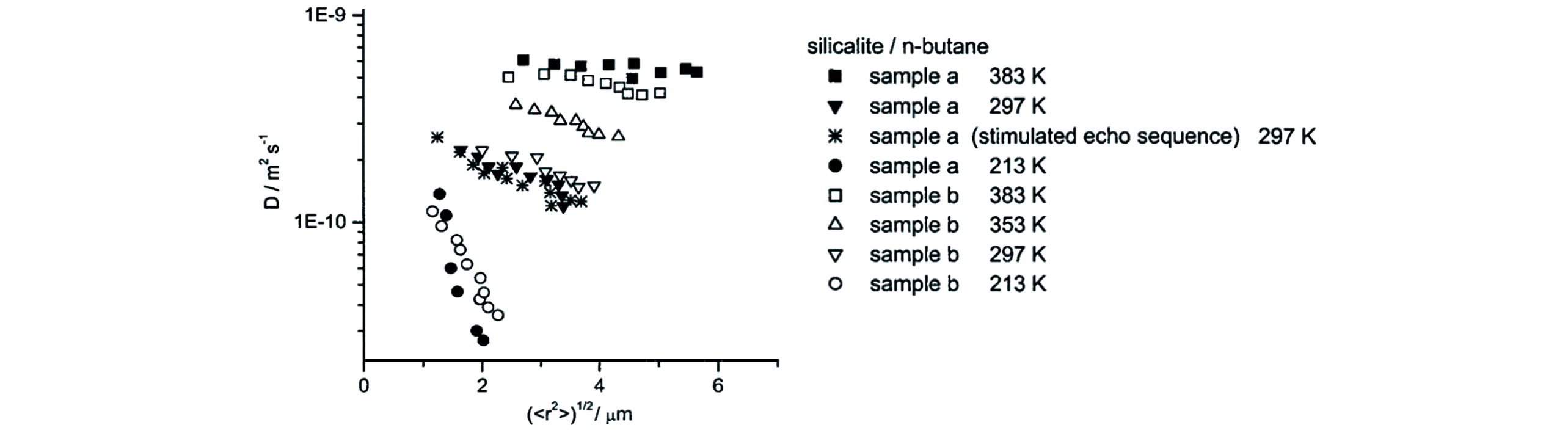
Fig.24 Diffusivity of n?butane in silicalite?1 as a function of RMSD measured at different temperatures by the PFG NMR[134]Copyright 2001, American Chemical Society.
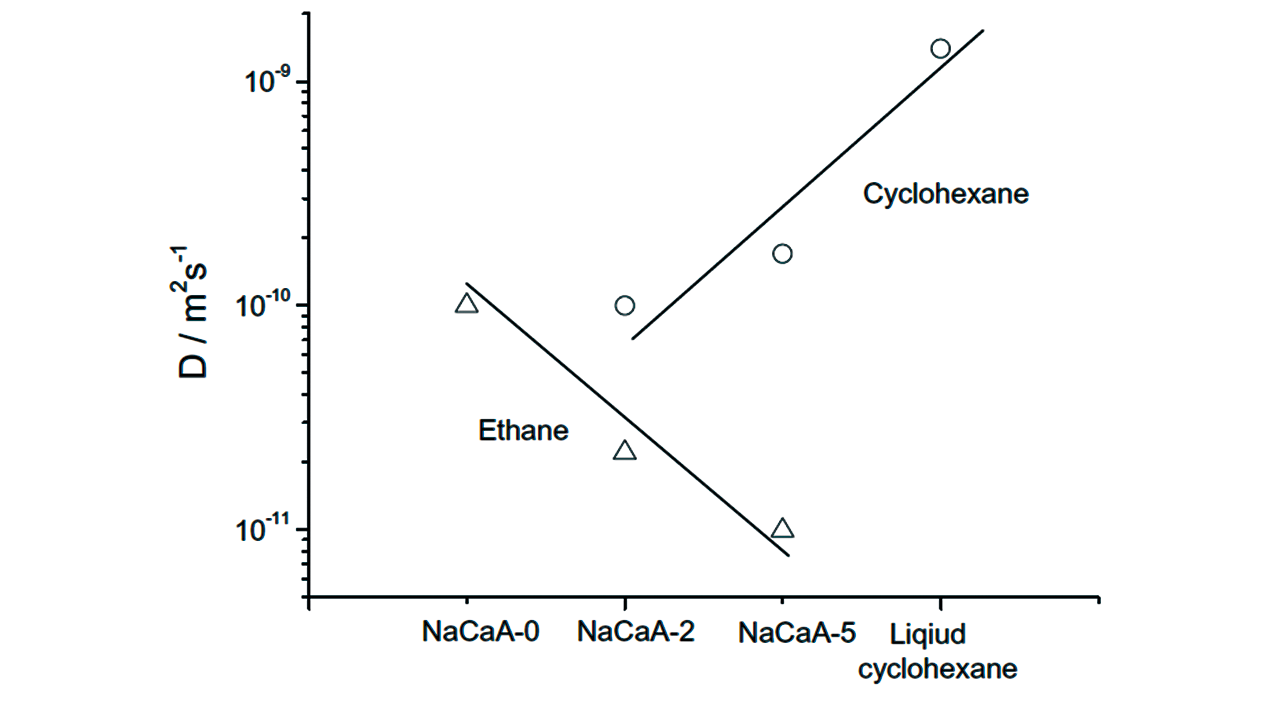
Fig.26 Comparison of the diffusivity in the space of micropores(probed with ethane) and mesopores(cyclohexane) of the LTA zeolites at room temperature[138]Copyright 2012, Elsevier Inc.
| 1 | Weckhuysen B. M., Yu J. H, Chem. Soc. Rev., 2015, 44(20), 7022—7024 |
| 2 | Coronas J., Chem. Eng. J., 2010, 156(2), 236—242 |
| 3 | Liang J., Liang Z., Zou R. Q., Zhao Y. L., Adv. Mater., 2017, 29(30), 1701139 |
| 4 | Gueudré L., Quoineaud A. A., Pirngruber G., Leflaive P., J. Phys. Chem. C, 2008, 112, 10899—10908 |
| 5 | Frising T., Leflaive P., Micropor. Mesopor. Mater., 2008, 114(1—3), 27—63 |
| 6 | Feliczak⁃Guzik A., Micropor. Mesopor. Mater., 2018, 259, 33—45 |
| 7 | Holm M. S., Taarning E., Egeblad K., Christensen C. H., Catal. Today, 2011, 168(1), 3—16 |
| 8 | Moliner M., Martínez C., Corma A., Angew. Chem. Int. Ed., 2015, 54(12), 3560—3579 |
| 9 | Pérez⁃Ramírez J., Christensen C. H., Egeblad K., Christensen C. H., Groen J. C., Chem. Soc. Rev., 2008, 37(11), 2530—2542 |
| 10 | Corma A., Díaz⁃Cabañas M. J., Jordá J. L., Martínez C., Moliner M., Nature, 2006, 443(7113), 842—845 |
| 11 | Mintova S., Gilson J. P., Valtchev V., Nanoscale, 2013, 5(15), 6693—6703 |
| 12 | Roth W. J., Nachtigall P., Morris R. E., Čejka J., Chem. Rev., 2014, 114(9), 4807—4837 |
| 13 | Tang K., Wang Y. G., Song L. J., Duan L. H., Zhang X. T., Sun Z. L., Mater. Lett., 2006, 60(17/18), 2158—2160 |
| 14 | Schwieger W., Machoke A. G., Weissenberger T., Inayat A., Selvam T., Klumpp M., Inayat A., Chem. Soc. Rev., 2016, 45(12), 3353—3376 |
| 15 | Zhao S. F., Wang W. D., Wang L. Z., Schwieger W., Wang W., Huang J., ACS Catal., 2019, 10(2), 1185—1194 |
| 16 | Zhang D. Z., Jin C. Z., Zou M. M., Huang S. J., Chemistry, 2019, 25(11), 2675—2683 |
| 17 | Issa H., Chaouati N., Toufaily J., Hamieh T., Sachse A., Pinard L., ChemCatChem, 2019, 11(18), 4581—4592 |
| 18 | Srivastava R., Catal. Today, 2018, 309, 172—188 |
| 19 | Masoumifard N., Guillet⁃Nicolas R., Kleitz F., Adv. Mater., 2018, 30(16), 1704439 |
| 20 | Sachse A., García⁃Martínez J., Chem. Mater., 2017, 29(9), 3827—3853 |
| 21 | Zhang K., Ostraat M. L., Catal. Today, 2016, 264, 3—15 |
| 22 | Verboekend D., Nuttens N., Locus R., Aelst J. V., Verolme P., Groen J. C., Pérez⁃Ramírez J., Sels B. F., Chem. Soc. Rev., 2016, 45(12), 3331—3352 |
| 23 | Wei Y., Parmentier T. E., de Jong K. P., Zečević J., Chem. Soc. Rev., 2015, 44(20), 7234—7261 |
| 24 | Javdani A., Ahmadpour J., Yaripour F., Micropor. Mesopor. Mater., 2019, 284, 443—458 |
| 25 | Čejka J., Millini R., Opanasenko M., Serrano D. P., Roth W. J., Catal. Today, 2019, 345, 2—13 |
| 26 | Yang X. Y., Chen L. H., Li Y., Rooke J. C., Sanchez C., Su B. L., Chem. Soc. Rev., 2017, 46(2), 481—558 |
| 27 | Gallego E. M., Paris C., Díaz⁃Rey M. R., Martínez⁃Armero M. E., Martínez⁃Triguero J., Martínez C., Moliner M., Corma A., Chem. Sci., 2017, 8(12), 8138—8149 |
| 28 | Sun M. H., Huang S. Z., Chen L. H., Li Y., Yang X. Y., Yuan Z. Y., Su B. L., Chem. Soc. Rev., 2016, 45(12), 3479—3563 |
| 29 | Hartmann M., Machoke A. G., Schwieger W., Chem. Soc. Rev., 2016, 45(12), 3313—3330 |
| 30 | Verboekend D., Pérez⁃Ramírez J., ChemSusChem, 2014, 7(3), 753—764 |
| 31 | Verboekend D., Mitchell S., Pérez⁃Ramírez J., Chimia, 2013, 67(5), 327—332 |
| 32 | Chal R., Gérardin C., Bulut M., Donk S. V., ChemCatChem, 2011, 3(1), 67—81 |
| 33 | Choi M., Cho H. S., Srivastava R., Venkatesan C., Choi D. H., Ryoo R., Nat. Mater., 2006, 5(9), 718—723 |
| 34 | Zhang R. X., Zhong P. N., Arandiyan H., Guan Y. N., Liu J. N., Wang N., Jiao Y. L., Fan X. L., Front. Chem. Sci. Eng., 2020, 14(2), 275—287 |
| 35 | Zhang R. X., Xu S. J, Raja D., Khusni N. B., Liu J. M., Zhang J. Y., Abdulridha S., Xiang H., Jiang S. S., Guan Y. N., Jiao Y. L., Fan X. L., Micropor. Mesopor. Mater., 2019, 278, 297—306 |
| 36 | Verboekend D., Keller T. C., Mitchell S., Pérez⁃Ramírez J., Adv. Funct. Mater., 2013, 23(15), 1923—1934 |
| 37 | Verboekend D., Vilé G., Pérez⁃Ramírez J., Adv. Funct. Mater., 2012, 22(5), 916—928 |
| 38 | Datka J., Kolidziejski W., Klinowski J., Sulikowski B., Catal. Lett., 1993, 19, 159—165 |
| 39 | Lee S., Kim H., Choi M., J. Mater. Chem. A, 2013, 1(39), 12096—12102 |
| 40 | Peng C., Liu Z. D., Yonezawa Y., Linares N., Yanaba Y., Trujillo C. A., Okubo T., Matsumoto T., García⁃Martínez J., Wakihara T., J. Mater. Chem. A, 2020, 8(2), 735—742 |
| 41 | Mehlhorn D., Rodriguez J., Cacciaguerra T., Andrei R. D., Cammarano C., Guenneau F., Gedeon A., Coasne B., Thommes M., Minoux D., Aquino C., Dath J. P., Fajula F., Galarneau A., Langmuir, 2018, 34(38), 11414—11423 |
| 42 | Aelst J. V., Verboekend D., Philippaerts A., Nuttens N., Kurttepeli M., Gobechiya E., Haouas M., Sree S. P., Denayer J. F. M., Martens J. A., Kirschhock C. E. A., Taulelle F., Bals S., Baron G. V., Jacobs P. A., Sels B. F., Adv. Funct. Mater., 2015, 25(46), 7130—7144 |
| 43 | Nuttens N., Verboekend D., Deneyer A., Aelst J. V., Sels B. F., ChemSusChem, 2015, 8(7), 1197—1205 |
| 44 | Aelst J. V., Haouas M., Gobechiya E., Houthoofd K., Philippaerts A., Sree S. P., Kirschhock C. E. A., Jacobs P., Martens J. A., Sels B. F., Taulelle F., J. Phys. Chem. C, 2014, 118(39), 22573—22582 |
| 45 | García⁃Martínez J., Li K. H., Krishnaiah G., Chem. Commun., 2012, 48(97), 11841—11843 |
| 46 | García⁃Martínez J., Johnson M., Valla J., Li K. H., Ying J. Y., Catal. Sci. Technol., 2012, 2(5), 987—994 |
| 47 | de Jong K. P., Zečević J., Friedrich H., de Jongh P. E., Bulut M., Donk S. V., Kenmogne R., Finiels A., Hulea V., Fajula F., Angew. Chem. Int. Ed., 2010, 49(52), 10074—10078 |
| 48 | Silva B. J. B., Sousa L. V., Sarmento L. R. A., Alencar S. L., Quintela P. H. L., Silva A. O. S., Appl. Catal. B, 2020, 267, 118699 |
| 49 | Petrov A. W., Ferri D., Kröcher O., van Bokhoven J. A., ACS Catal., 2019, 9(3), 2303—2312 |
| 50 | Li T., Ihli J., Wennmacher J. T. C., Krumeich F., van Bokhoven J. A., Chemistry, 2019, 25(32), 7689—7694 |
| 51 | Yang S. T., Yu C. X., Yu L. L., Miao S., Zou M. M., Jin C. Z., Zhang D. Z., Xu L. Y., Huang S. J., Angew. Chem. Int. Ed., 2017, 56(41), 12553—12556 |
| 52 | Yu L. L., Huang S. J., Miao S., Chen F. C., Zhang S., Liu Z. N., Xie S. J., Xu L. Y., Chemistry, 2015, 21(3), 1048—1054 |
| 53 | You S. J., Park E. D., Micropor. Mesopor. Mater., 2014, 186, 121—129 |
| 54 | Ahn J. H., Kolvenbach R., Neudeck C., Al⁃Khattaf S. S., Jentys A., Lercher J. A., J. Catal., 2014, 311, 271—280 |
| 55 | Sazama P., Sobalik Z., Dedecek J., Jakubec I., Parvulescu V., Bastl Z., Rathousky J., Jirglova H., Angew. Chem. Int. Ed., 2013, 52(7), 2038—2041 |
| 56 | Dapsens P. Y., Mondelli C., Pérez⁃Ramírez J., ChemSusChem, 2013, 6(5), 831—839 |
| 57 | Verboekend D., Pérez⁃Ramírez J., Chemistry, 2011, 17(4), 1137—1147 |
| 58 | Verboekend D., Mitchell S., Milina M., Groen J. C., Pérez⁃Ramírez J., J. Phys. Chem. C, 2011, 115(29), 14193—14203 |
| 59 | Groen J. C., Moulijn J. A., Pérez⁃Ramírez J., J. Mater. Chem., 2006, 16(22), 2121—2131 |
| 60 | Groen J. C., Bruckner A., Berrier E., Maldonado L., Moulijn J., Pérez⁃Ramírez J., J. Catal., 2006, 243(1), 212—216 |
| 61 | Groen J. C., Zhu W. D., Brouwer S., Huynink S. J., Kapteijn F., Moulijn J. A., Pérez⁃Ramírez J., J. Am. Chem. Soc., 2007, 129, 355—360 |
| 62 | Tao Y. S., Hattori Y., Matumoto A, Kanoh H., Kaneko K., J. Phys. Chem. B, 2005, 109, 194—199 |
| 63 | Schmidt I., Boisen A., Gustavsson E., Ståhl K., Pehrson S., Dahl S., Carlsson A., Jacobsen C. J. H., Chem. Mater., 2001, 13, 4416—4418 |
| 64 | Fan W., Snyder M. A., Kumar S., Lee P. S., Yoo W. C., McCormick A. V., Penn R. L., Stein A., Tsapatsis M., Nat. Mater., 2008, 7(12), 984—991 |
| 65 | Sun M. H., Chen L. H., Yu S., Li Y., Zhou X. G., Hu Z. Y., Sun Y. H., Xu Y., Su B. L., Angew. Chem. Int. Ed., 2020, 59, 2—12 |
| 66 | Zhu H. B., Liu Z. C., Wang Y. D., Kong D. J., Yuan X. H., Xie Z. K., Chem. Mater., 2008, 20, 1134—1139 |
| 67 | Rhodes K. H., Davis H. A., Caruso F., Zhang B. J., Mann S., Chem. Mater., 2000, 12, 2832—2834 |
| 68 | Yue M. B., Sun M. N., Xie F., Ren D. D., Micropor. Mesopor. Mater., 2014, 183, 177—184 |
| 69 | Abdulridha S., Jiang J. X., Xu S. J., Zhou Z. X., Liang H., Mao B. Y., Zhou Y. T., Garforth A. A., Jiao Y. L., Fan X. L., Green Chem., 2020, 22, 5115—5122 |
| 70 | Zhang B., Li X. R., Wu Q. F., Zhang C., Yu Y. C., Lan M. L., Wei X., Ying Z., Liu T., Liang G. F., Zhao F. Y., Green Chem., 2016, 18(11), 3315—3323 |
| 71 | Dong A. G., Wang Y., Tang Y., Ren N., Zhang Y. H., Yue Y. H., Gao Z., Adv. Mater., 2002, 14, 926—929 |
| 72 | Xiao F. S., Wang L. F., Yin C. Y., Lin K. F., Di Y., Li J. X., Xu R. R., Su D. S., Schlögl R., Yokoi T., Tatsumi T., Angew. Chem. Int. Ed., 2006, 45(19), 3090—3093 |
| 73 | Serrano D. P., Aguado J., Escola J. M., Rodriguez J. M., Peral A., J. Mater. Chem., 2008, 18(35), 4210—4218 |
| 74 | Aguado J., Serrano D. P., Rodríguez J. M., Micropor. Mesopor. Mater., 2008, 115(3), 504—513 |
| 75 | Serrano D. P., Aguado J., Escola J. M., Rodríguez J. M., Peral A., Chem. Mater., 2006, 18, 2462—2464 |
| 76 | Yang G. Y., Han J., Qiu Z. Y., Chen X. X., Feng Z. C., Yu J. H., Inorg. Chem. Front., 2020, 7(10), 1975—1980 |
| 77 | Zhang Q., Mayoral A., Terasaki O., Zhang Q., Ma B., Zhao C., Yang G. J., Yu J. H., J. Am. Chem. Soc., 2019, 141(9), 3772—3776 |
| 78 | Zhang J., Chen Z. W., Wang Y. D., Zheng G. C., Zheng H. R., Cai F. Y., Hong M., Micropor. Mesopor. Mater., 2017, 252, 79—89 |
| 79 | Zhang J., Bai S., Chen Z. W., Wang Y. D., Dong L., Zheng H. R., Cai F. Y., Hong M., J. Mater. Chem. A, 2017, 5(39), 20757—20764 |
| 80 | Feng R., Wang X. X., Lin J. W., Li Z., Hou K., Yan X. L., Hu X. Y., Yan Z. F., Rood M. J., Micropor. Mesopor. Mater., 2018, 270, 57—66 |
| 81 | Zhang M. R., Liu X. M., Yan Z. F., Mater. Lett., 2016, 164, 543—546 |
| 82 | Choi M., Na K., Kim J., Sakamoto Y., Terasaki O., Ryoo R., Nature, 2009, 461(7261), 246—249 |
| 83 | Park D. H., Kim S. S., Wang H., Pinnavaia T. J., Papapetrou M. C., Lappas A. A., Triantafyllidis K. S., Angew. Chem. Int. Ed., 2009, 48(41), 7645—7648 |
| 84 | Wang H., Pinnavaia T. J., Angew. Chem. Int. Ed., 2006, 45(45), 7603—7606 |
| 85 | Lucas A. D., Canizares P., Durán A., Carrero A., Appl. Catal. A: Gen., 1997, 154, 221—240 |
| 86 | Williams B. A., Babitz S. M., Miller J. T., Snurr R. Q., Kung H. H., Appl. Catal. A: Gen., 1999, 177, 161—175 |
| 87 | Janssen A. H., Koster A. J., de Jong K. P., J. Phys. Chem. B, 2001, 106, 11905—11909 |
| 88 | Janssen A. H., Koster A. J., de Jong K. P., Angew. Chem. Int. Ed, 2001, 40, 1102—1104 |
| 89 | Kortunov P., Vasenkov S., Kärger J., Valiullin R., Gottschalk P., Fé Elía M., Perez M., Stöcker M., Drescher B., McElhiney G., Berger C., Gläser R., Weitkamp J., J. Am. Chem. Soc., 2005, 127, 13055—13059 |
| 90 | Triantafillidis C. S., Vlessidis A. G., Nalbandian L., Evmiridis N. P., Micropor. Mesopor. Mater., 2001, 47, 369—388 |
| 91 | Inagaki S., Shinoda S., Kaneko Y., Takechi K., Komatsu R., Tsuboi Y., Yamazaki H., Kondo J. N., Kubota Y., ACS Catal., 2012, 3(1), 74—78 |
| 92 | Kerr G. T., J. Phys. Chem. C, 1968, 72, 2594—2596 |
| 93 | Akira Y., Hiromi N., Kazuhito O., Tatsuro T., Hiroshi T., Bull. Chem. Soc. Jpn, 1982, 55, 581—586 |
| 94 | Ogura M., Shin⁃ya S., Tateno J., Nara Y., Eiichi K., Masahiko M., Chem. Lett., 2000, 29, 882—883 |
| 95 | Groen J. C., Jansen J. C., Moulijn J. A., Pérez⁃Ramírez J., J. Phys. Chem. B, 2004, 108, 13062—13065 |
| 96 | Groen J. C., Peffer L. A., Moulijn J. A., Pérez⁃Ramírez J., Chemistry, 2005, 11(17), 4983—4994 |
| 97 | Verboekend D., Pérez⁃Ramírez J., Catal. Sci. Technol., 2011, 1(6), 879—890 |
| 98 | Milina M., Mitchell S., Crivelli P., Cooke D., Pérez⁃Ramírez J., Nat. Commun., 2014, 5, 3922 |
| 99 | Pérez⁃Ramírez J., Verboekend D., Bonilla A., Abelló s., Adv. Funct. Mater., 2009, 19(24), 3972—3979 |
| 100 | Keller T. C., Arras J., Wershofen S., Pérez⁃Ramírez J., ACS Catal., 2014, 5(2), 734—743 |
| 101 | Ivanova I. I., Kasyanov I. A., Maerle A. A., Zaikovskii V. I., Micropor. Mesopor. Mater., 2014, 189, 163—172 |
| 102 | Ivanova I. I, Knyazeva E. E., Chem. Soc. Rev., 2013, 42 (9), 3671—3688 |
| 103 | Dessau R. M., Valyocsik E. W., Goeke N. H., Zeolites, 1992, 12, 776—779 |
| 104 | Groen J. C., Bach T., Ziese U., Donk A. M. P., de Jong K. P., Moulijn J. A., Pérez⁃Ramírez J., J. Am. Chem. Soc., 2005, 127, 10792—10793 |
| 105 | Verboekend D., Keller T. C., Milina M., Hauert R., Pérez⁃Ramírez J., Chem. Mater., 2013, 25(9), 1947—1959 |
| 106 | Li S. H., Zheng A. M., Su Y. C., Zhang H. L., Chen L., Yang J., Ye C. H., Deng F., J. Am. Chem. Soc., 2007, 129, 11161—11171 |
| 107 | Qin Z. X., Hafiz L., Shen Y. F., Daele S. V., Boullay P., Ruaux V., Mintova S., Gilson J. P., Valtchev V., J. Mater. Chem. A, 2020, 8(7), 3621—3631 |
| 108 | Qin Z. X, Shen B. J., Yu Z. W., Deng F., Zhao L., Zhou S. G., Yuan D. L., Gao X. H., Wang B. J., Zhao H. J., Liu H. H., J. Catal., 2013, 298, 102—111 |
| 109 | Svelle S., Sommer L., Barbera K., Vennestrøm P. N. R., Olsbye U., Lillerud K. P., Bordiga S., Pan Y. H., Beato P., Catal. Today, 2011, 168(1), 38—47 |
| 110 | Wu S. M., Yang X. Y., Janiak C., Angew. Chem. Int. Ed., 2019, 58(36), 12340—12354 |
| 111 | Graça I., Bacariza M. C., Fernandes A., Chadwick D., Appl. Catal. B, 2018, 224, 660—670 |
| 112 | Verboekend D., Groen J. C., Pérez⁃Ramírez J., Adv. Funct. Mater., 2010, 20(9), 1441—1450 |
| 113 | Gołąbek K., Palomares A. E., Martínez⁃Triguero J., Tarach K. A., Kruczała K., Girman V., Góra⁃Marek K., Appl. Catal. B, 2019, 259, 118022 |
| 114 | Keller T. C., Isabettini S., Verboekend D., Rodrigues E. G., Pérez⁃Ramírez J., Chem. Sci., 2014, 5(2), 677—684 |
| 115 | Losch P., Huang W. X., Vozniuk O., Goodman E. D., Schmidt W., Cargnello M., ACS Catal., 2019, 9(6), 4742—4753 |
| 116 | Mielby J., Abildstrom J. O., Wang F., Kasama T., Weidenthaler C., Kegnaes S., Angew. Chem. Int. Ed., 2014, 53(46), 12513—12516 |
| 117 | Schneider D., Mehlhorn D., Zeigermann P., Kärger J., Valiullin R., Chem. Soc. Rev.,2016, 45, 3439—3467 |
| 118 | Randall Q. Snurr K., J., J. Phys. Chem. B, 1997, 101, 6469—6473 |
| 119 | Hoang V. T., Huang Q., Malekian A., Eić M., Do T. O., Kaliaguine S., Adsorption, 2005, 11, 421—426 |
| 120 | Ruthven D. M., Brandani S., Recent Advances in Gas Separation by Microporous Ceramic Membranes,Elsevier, Amsterdam, 2000, 187—212 |
| 121 | Ruthven D., Derrah, R., Can. J. Chem. Eng., 1972, 50(6), 743—747 |
| 122 | Jobic H., Theodorou D. N., Micropor. Mesopor. Mater., 2007, 102(1—3), 21—50 |
| 123 | Jobic H., Schmidt W., Krause C. B., Kärger J., Micropor. Mesopor. Mater., 2006, 90(1—3), 299—306 |
| 124 | Ito T., Fraissard J., J. Chem. Phys., 1982, 76(11), 5225—5229 |
| 125 | Hwang S., Parditka B., Cserháti C., Erdélyi Z., Gläser R., Haase J., Kärger J., Schmidt W., Chmelik C., Chem. Ing. Tech., 2017, 89(12), 1686—1693 |
| 126 | Kirchner T., Shakhov A., Zeigermann P., Valiullin R., Kärger J., Carbon, 2012, 50(13), 4804—4808 |
| 127 | Tanner J. E., Rev. Sci. Instrum., 1965, 36(8), 1086—1087 |
| 128 | Bingre R., Losch P., Megias⁃Sayago C., Vincent B., Pale P., Nguyen P., Louis B., ChemPhysChem, 2019, 20(21), 2874—2880 |
| 129 | Gunther J. P., Majer G., Fischer P., J. Chem. Phys., 2019, 150(12), 124201 |
| 130 | Mehlhorn D., Valiullin R., Kärger J., Cho K., Ryoo R., Materials, 2012, 5(4), 699—720 |
| 131 | Freeman R., Spin choreography, Oxford University Press Oxford, 1998 |
| 132 | Kärger J., Valiullin R., Chem. Soc. Rev., 2013, 42 (9), 4172—4197 |
| 133 | Mehlhorn D., Valiullin R., Kärger J., Cho K., Ryoo R., ChemPhysChem, 2012, 13(6), 1495—1499 |
| 134 | Vasenkov S., Böhlmann W., Galvosas P., Geier O., Liu H., Kärger J., J. Phys. Chem. B, 2001, 105(25), 5922—5927 |
| 135 | Jobic H., Ernst H., Heink W., Kärger J., Tuel A., Bée M., Micropor. Mesopor. Mater., 1998, 26, 67—75 |
| 136 | Geier O., Vasenkov S., Kärger J., J. Chem. Phys., 2002, 117(5), 1935—1938 |
| 137 | Kärger J., Vasenkov S., Micropor. Mesopor. Mater., 2005, 85(3), 195—206 |
| 138 | Mehlhorn D., Valiullin R., Kärger J., Cho K., Ryoo R., Micropor. Mesopor. Mater., 2012, 164, 273—279 |
| 139 | Jiao Y. L., Forster L., Xu S. J., Chen H. H., Han J. F., Liu X. Q., Zhou Y. T., Liu J. M., Zhang J. S., Yu J. H., D'Agostino C., Fan X. L., Angew. Chem.Int. Ed., 2020, 132, 2—11 |
| [1] | 杨静怡, 李庆贺, 乔波涛. 铱单原子和纳米粒子在N2O分解反应中的协同催化[J]. 高等学校化学学报, 2022, 43(9): 20220388. |
| [2] | 林高鑫, 王家成. 单原子掺杂二硫化钼析氢催化的进展和展望[J]. 高等学校化学学报, 2022, 43(9): 20220321. |
| [3] | 任诗杰, 谯思聪, 刘崇静, 张文华, 宋礼. 铂单原子催化剂同步辐射X射线吸收谱的研究进展[J]. 高等学校化学学报, 2022, 43(9): 20220466. |
| [4] | 汪思聪, 庞贝贝, 刘潇康, 丁韬, 姚涛. XAFS技术在单原子电催化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220487. |
| [5] | 唐全骏, 刘颖馨, 孟蓉炜, 张若天, 凌国维, 张辰. 单原子催化在海洋能源领域的应用[J]. 高等学校化学学报, 2022, 43(9): 20220324. |
| [6] | 楚宇逸, 兰畅, 罗二桂, 刘长鹏, 葛君杰, 邢巍. 单原子铈对弱芬顿效应活性位点氧还原稳定性的提升[J]. 高等学校化学学报, 2022, 43(9): 20220294. |
| [7] | 秦永吉, 罗俊. 单原子催化剂在CO2转化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220300. |
| [8] | 姚青, 俞志勇, 黄小青. 单原子催化剂的合成及其能源电催化应用的研究进展[J]. 高等学校化学学报, 2022, 43(9): 20220323. |
| [9] | 范建玲, 唐灏, 秦凤娟, 许文静, 谷鸿飞, 裴加景, 陈文星. 氮掺杂超薄碳纳米片复合铂钌单原子合金催化剂的电化学析氢性能[J]. 高等学校化学学报, 2022, 43(9): 20220366. |
| [10] | 林治, 彭志明, 贺韦清, 沈少华. 单原子与团簇光催化: 竞争与协同[J]. 高等学校化学学报, 2022, 43(9): 20220312. |
| [11] | 程前, 杨博龙, 吴文依, 向中华. S掺杂Fe-N-C高活性氧还原反应催化剂[J]. 高等学校化学学报, 2022, 43(9): 20220341. |
| [12] | 滕镇远, 张启涛, 苏陈良. 聚合物单原子光催化剂的载流子分离和表面反应机制[J]. 高等学校化学学报, 2022, 43(9): 20220325. |
| [13] | 杨静怡, 施思齐, 彭怀涛, 杨其浩, 陈亮. Ga-C3N4单原子催化剂高效光驱动CO2环加成[J]. 高等学校化学学报, 2022, 43(9): 20220349. |
| [14] | 王茹玥, 魏呵呵, 黄凯, 伍晖. 单原子材料的冷冻合成[J]. 高等学校化学学报, 2022, 43(9): 20220428. |
| [15] | 王新天, 李攀, 曹越, 洪文浩, 耿忠璇, 安志洋, 王昊宇, 王桦, 孙斌, 朱文磊, 周旸. 单原子材料在二氧化碳催化中的技术经济分析与产业化应用前景[J]. 高等学校化学学报, 2022, 43(9): 20220347. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||