

高等学校化学学报 ›› 2026, Vol. 47 ›› Issue (5): 20260013.doi: 10.7503/cjcu20260013
收稿日期:2026-01-04
出版日期:2026-05-10
发布日期:2026-02-10
通讯作者:
秦安军
E-mail:msqinaj@scut.edu.cn
基金资助:Received:2026-01-04
Online:2026-05-10
Published:2026-02-10
Contact:
QIN Anjun
E-mail:msqinaj@scut.edu.cn
Supported by:摘要:
聚集诱导发光(AIE)镧系配合物结合了镧系离子独特的光物理性质与AIE特性, 在多个领域具有应用价值. 然而, 迄今尚未有文献对该领域的研究进展进行系统梳理. 本文基于镧系中心数目总结了AIE镧系配合物的研究进展, 并阐述了其作用机制及在刺激响应和生物成像等领域的应用. 最后, 简要探讨了AIE镧系配合物当前面临的挑战及未来研究方向.
中图分类号:
TrendMD:
张凯为, 秦安军. 聚集诱导发光镧系配合物. 高等学校化学学报, 2026, 47(5): 20260013.
ZHANG Kaiwei, QIN Anjun. Lanthanide Complexes with Aggregation-induced Emission. Chem. J. Chinese Universities, 2026, 47(5): 20260013.
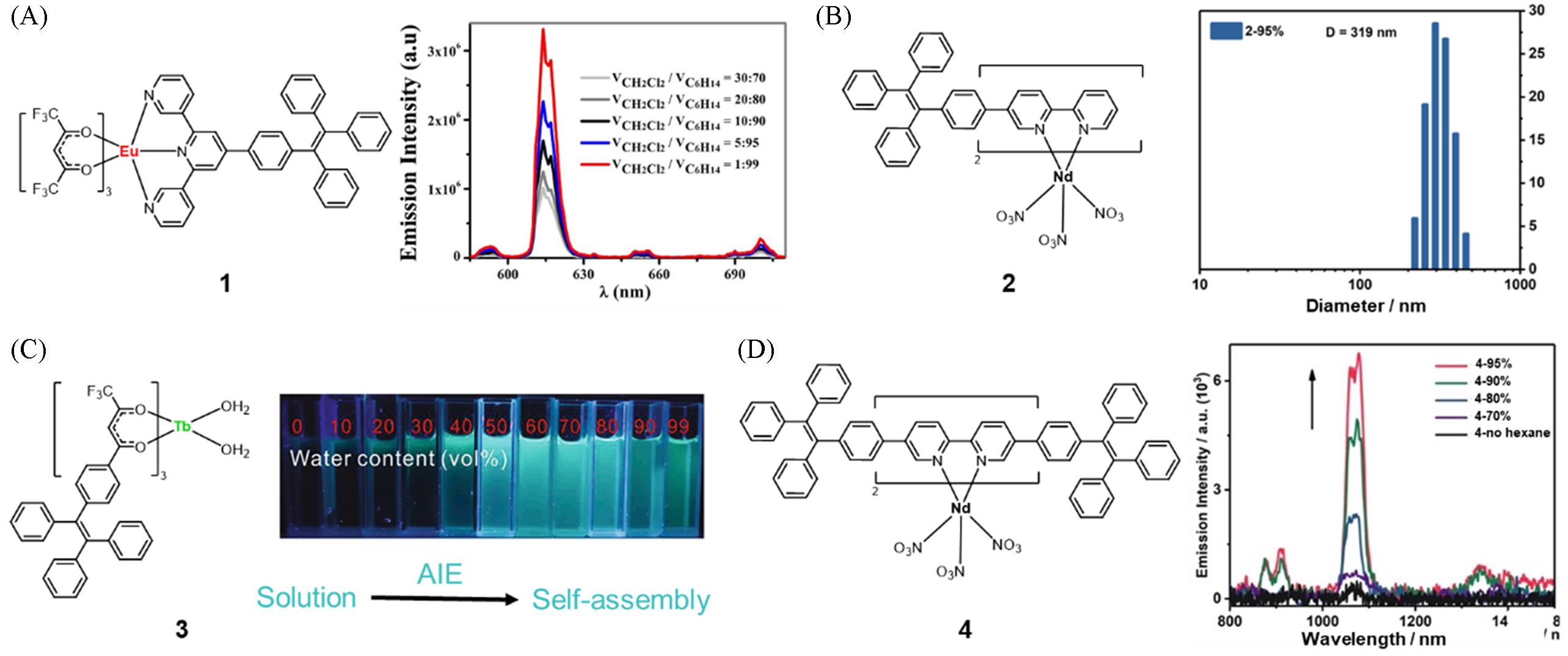
Fig.2 Molecular structure of complex 1 and photoluminescence(PL) spectra of complex 1 in dichloromethane with different volume fractions of n⁃hexane(A)[20], molecular structure of complex 2 and DLS of complex 2 in n⁃hexane/chloroform mixtures with the n⁃hexane content of 95%(B)[22], molecular structure of complex 3 and fluorescent photographs of complex 3(C)[23] and molecular structure of complex 4 and PL spectra of complex 4 in chloroform with different volume fractions of n⁃hexane(D)[22](A) Copyright 2015, Springer Nature; (B, D) Copyright 2021, John Wiley and Sons; (C) Copyright 2021, Elsevier Ltd.
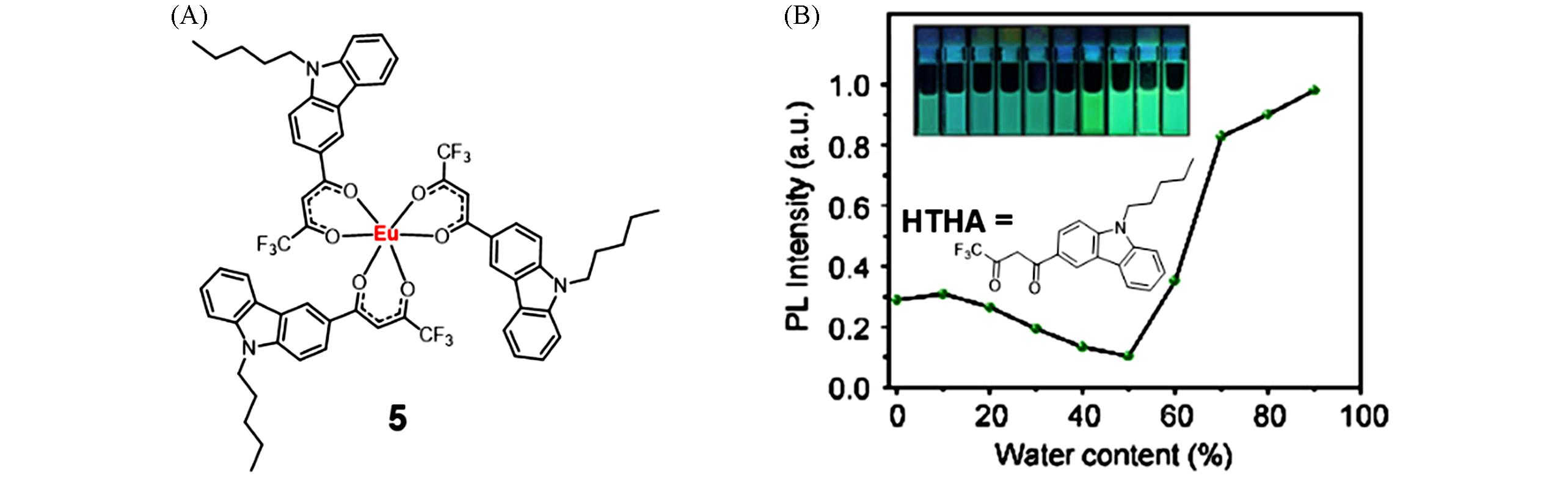
Fig.3 Molecular structure of complex 5(A) and PL intensity profile of ligand HTHA at 470 nm in ethanol with different volume fractions of water and fluorescent photographs of HTHA(B)[25]
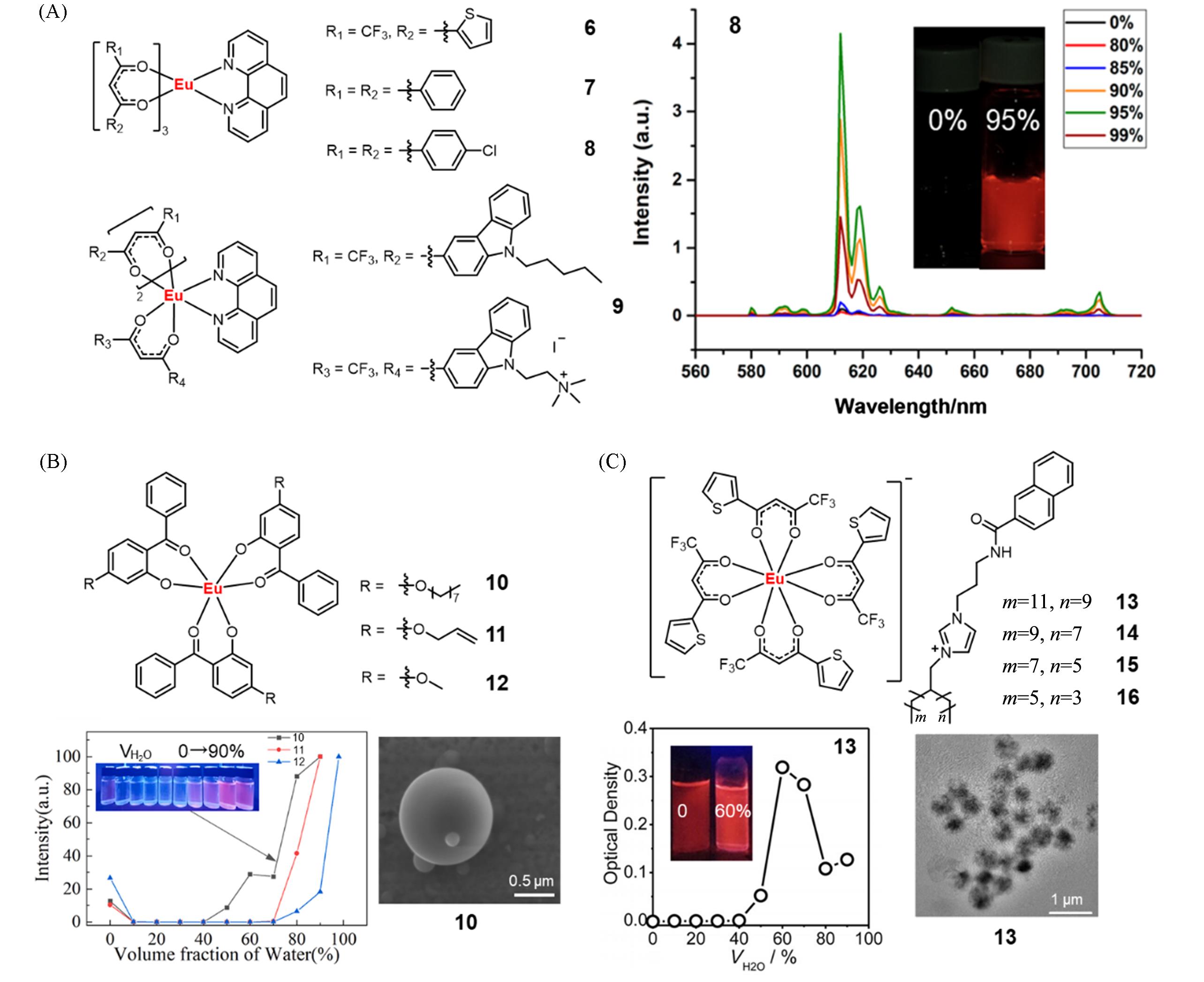
Fig.4 Molecular structure of β⁃diketone complexes 6—9 and PL spectra of complex 8 in THF with different volume fraction of water(A)[29], molecular structure of complexes 10—12 and PL intensity profile of complexes 10—12 at 615 nm in acetone with different volume fractions of water, SEM image of complex 10 with the water volume fraction of 90%(B)[31], molecular structure of complexes 13—16 and optical density of complex 13 at 500 nm in ethanol with different volume fractions of water, TEM image of aggregates formed by complex 13 in ethanol/water mixtures with water volume fraction of 60%(C)[32](A) Copyright 2019, MDPI; (B) Copyright 2025, Elsevier Ltd.; (C) Copyright 2018, John Wiley and Sons.
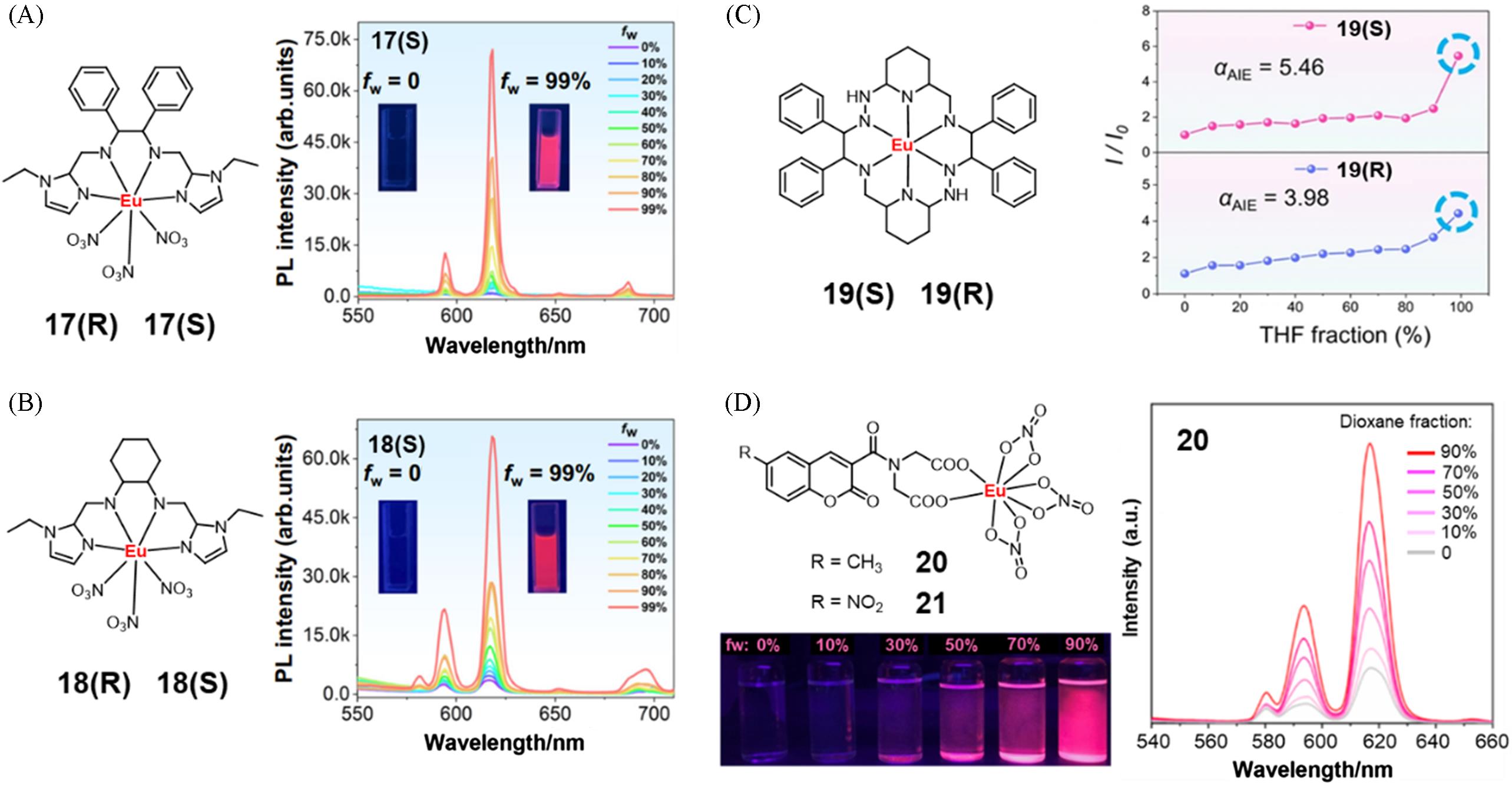
Fig.5 Molecular structure of chiral complexes 17(R) and 17(S) and PL spectra of complex 17(S) in DMSO with different fraction of glycerin(A)[33], molecular structure of chiral complexes 18(R) and 18(S) and PL spectra of complex 18(S) in DMSO with different fraction of glycerin(B)[33], molecular structure of chiral complexes 19(R) and 19(S) and PL intensity profile of complexes 19(R) and 19(S) at 611 nm in DMSO with different fraction of THF(C)[35], and molecular structure of complexes 20 and 21, fluorescent photographs and PL spectra of complex 20 in DMF with different fraction of 1,4⁃dioxane(D)[36](A) Copyright 2024, Springer Nature; (B) Copyright 2024, Springer Nature; (C) Copyright 2025, Springer Nature; (D) Copyright 2020, John Wiley and Sons.
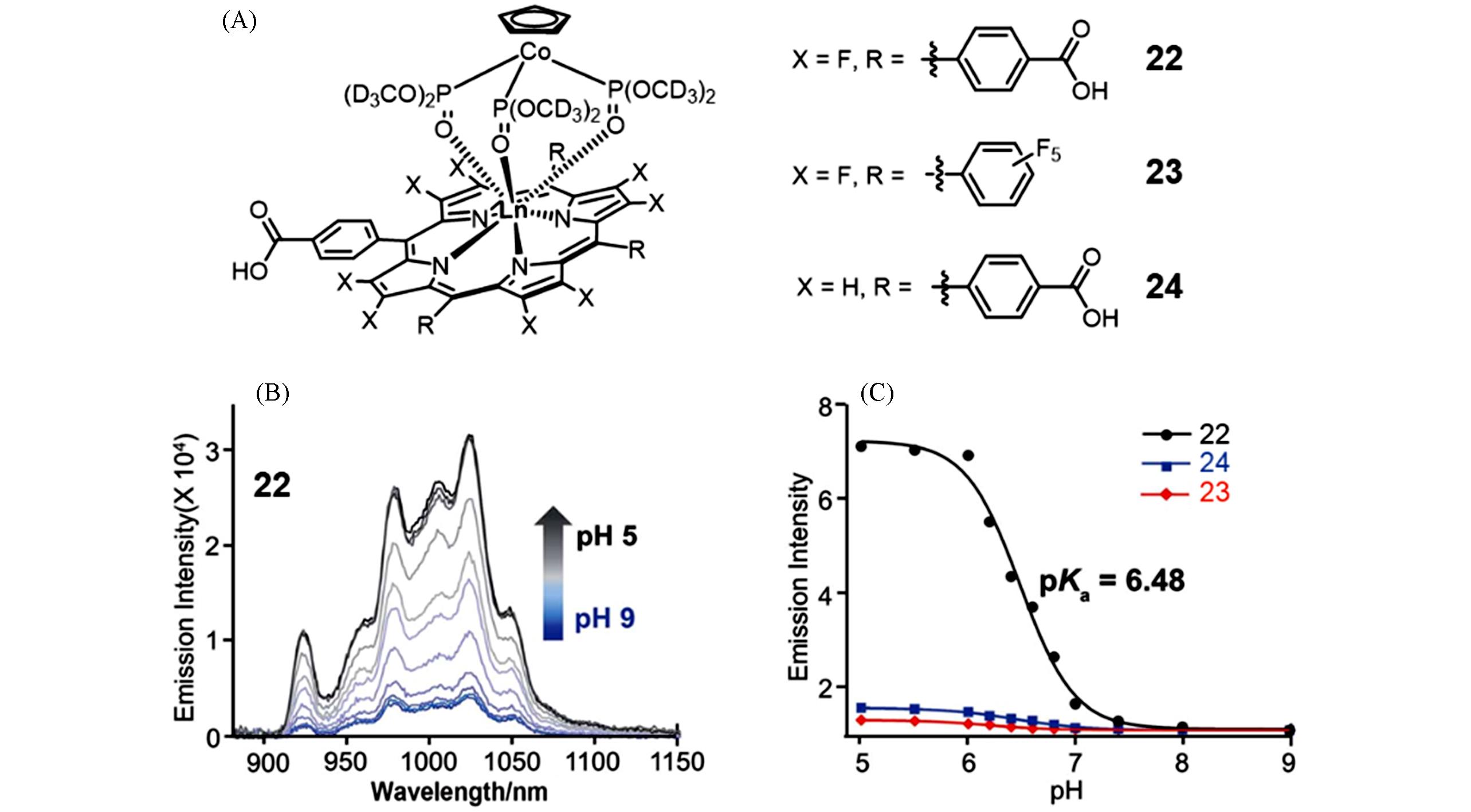
Fig.6 Molecular structure of complexes 22—24(A), fluorescence emission spectra of complex 22 in PBS buffer(pH=5.0—9.0)(B), and the normalized emission intensity ratio of complexes 22—24 according to the value at pH 9.0 versus the pH values in the pH range 5.0—9.0(C)[37]
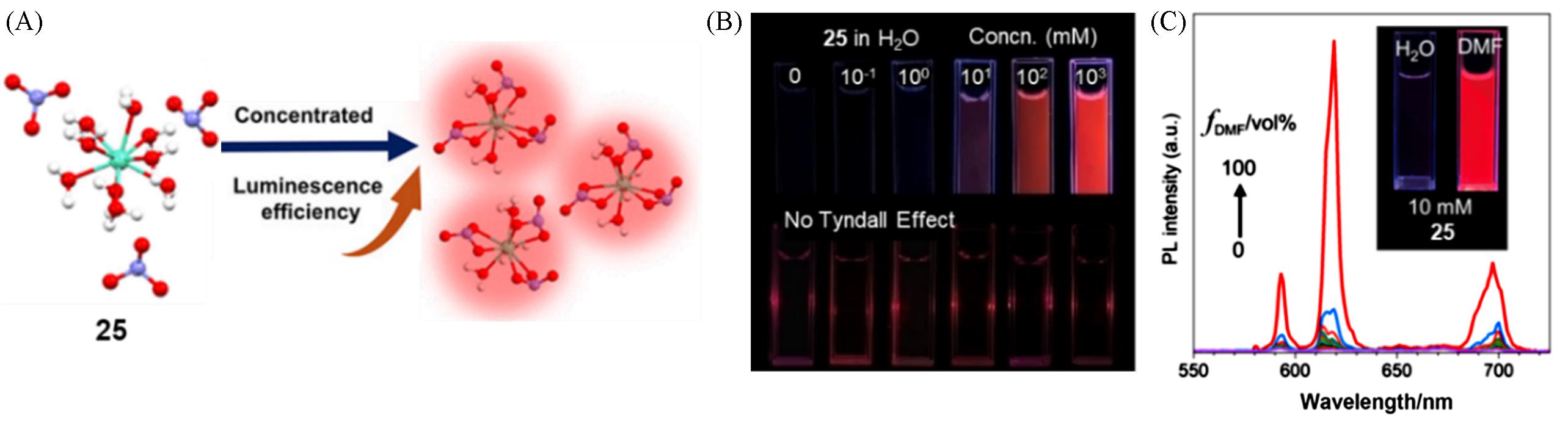
Fig.7 Schematic diagram of the structural changes of complex 25 from low concentration to aggregated state(A), fluorescence photographs of complex 25 solutions and laser for Tyndall effect test(B) and fluorescence emission spectra of complex 25 in water with different fraction of DMF and fluorescence photograph complex 25 in water and DMF(C)[39]
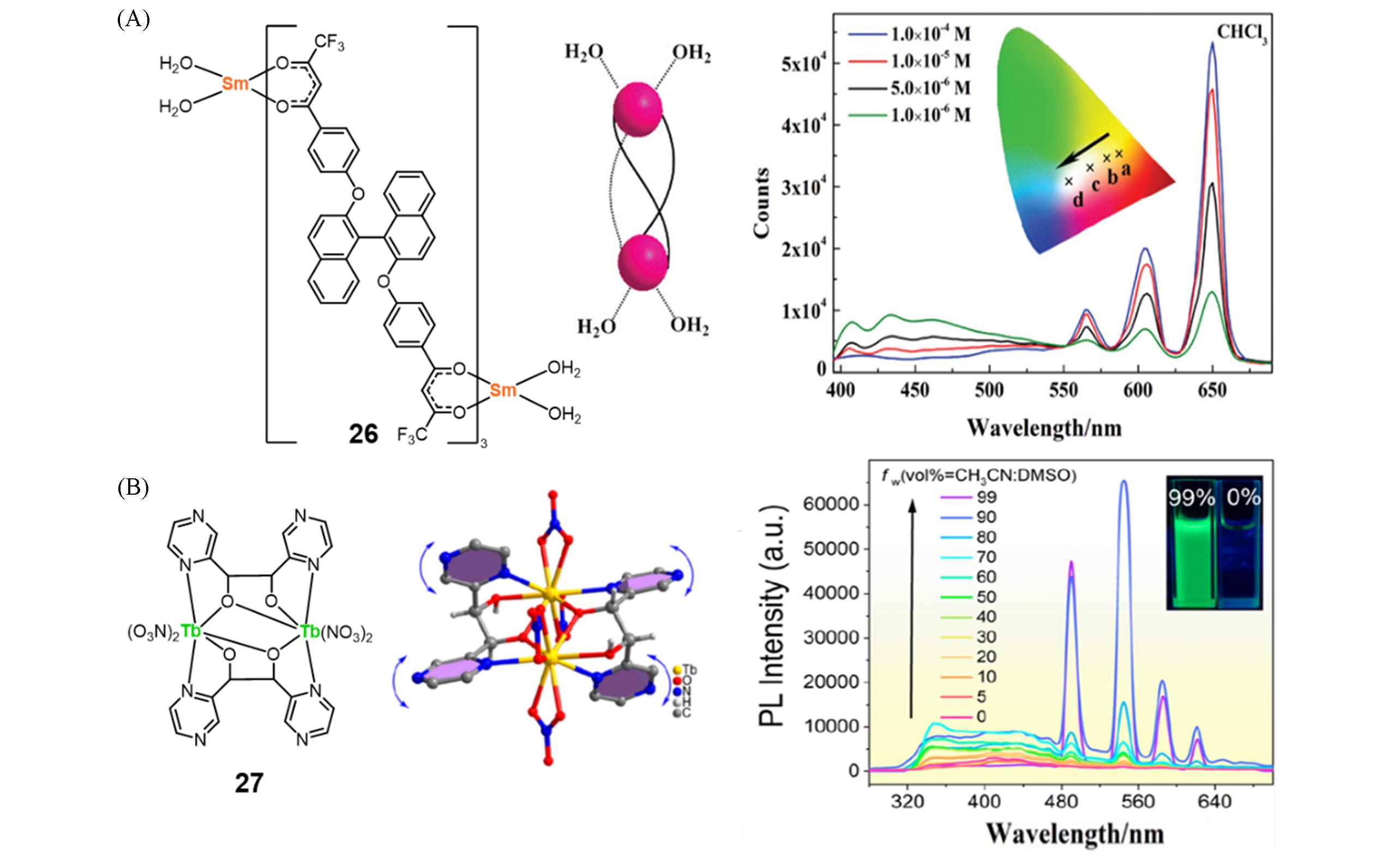
Fig.8 Molecular structure and schematic diagram of complex 26, PL spectra of complex 26 in THF at different concentrations(A)[40] and molecular structure and structural diagram of complex 27, PL spectra of complex 27 in DMSO with different fraction of CH3CN(B)[41](A) Copyright 2014, the Royal Society of Chemistry; (B) copyright 2023, American Chemical Society.
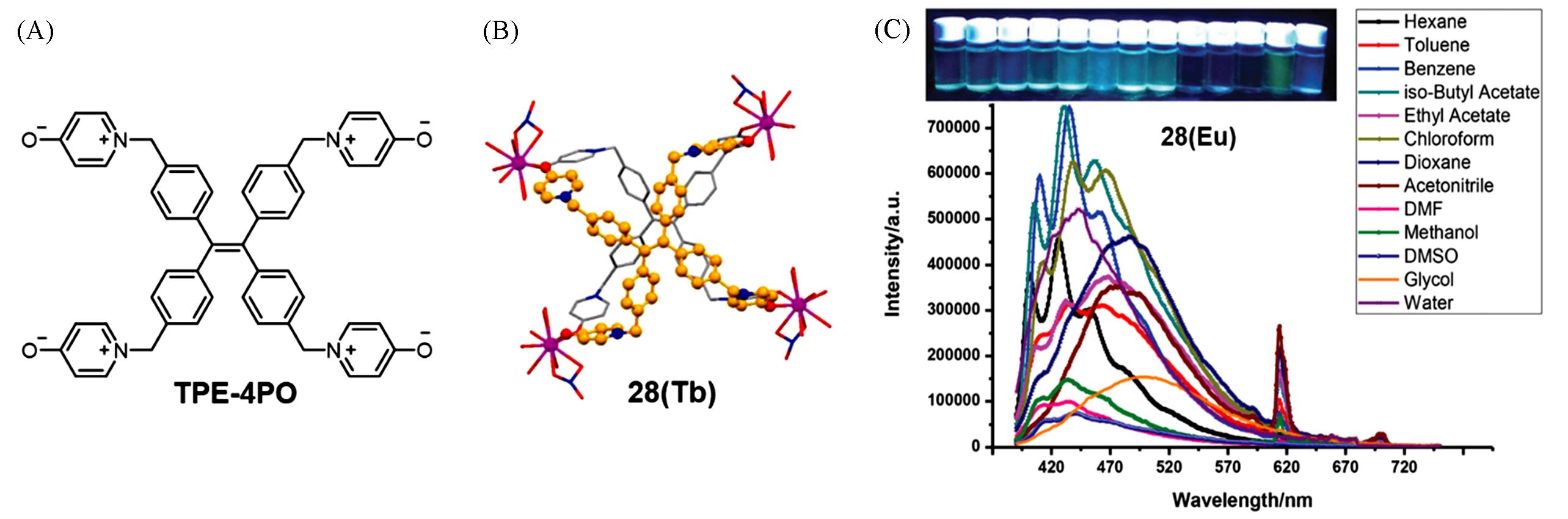
Fig.9 Molecular structure of ligand TPE⁃4PO(A), the coordination environment of Tb3+ ions and TPE⁃4PO ligands in the crystal of complex 28(Tb)(B) and fluorescent photographs and PL spectra of complex 28(Eu) in various solvents(C)[45]
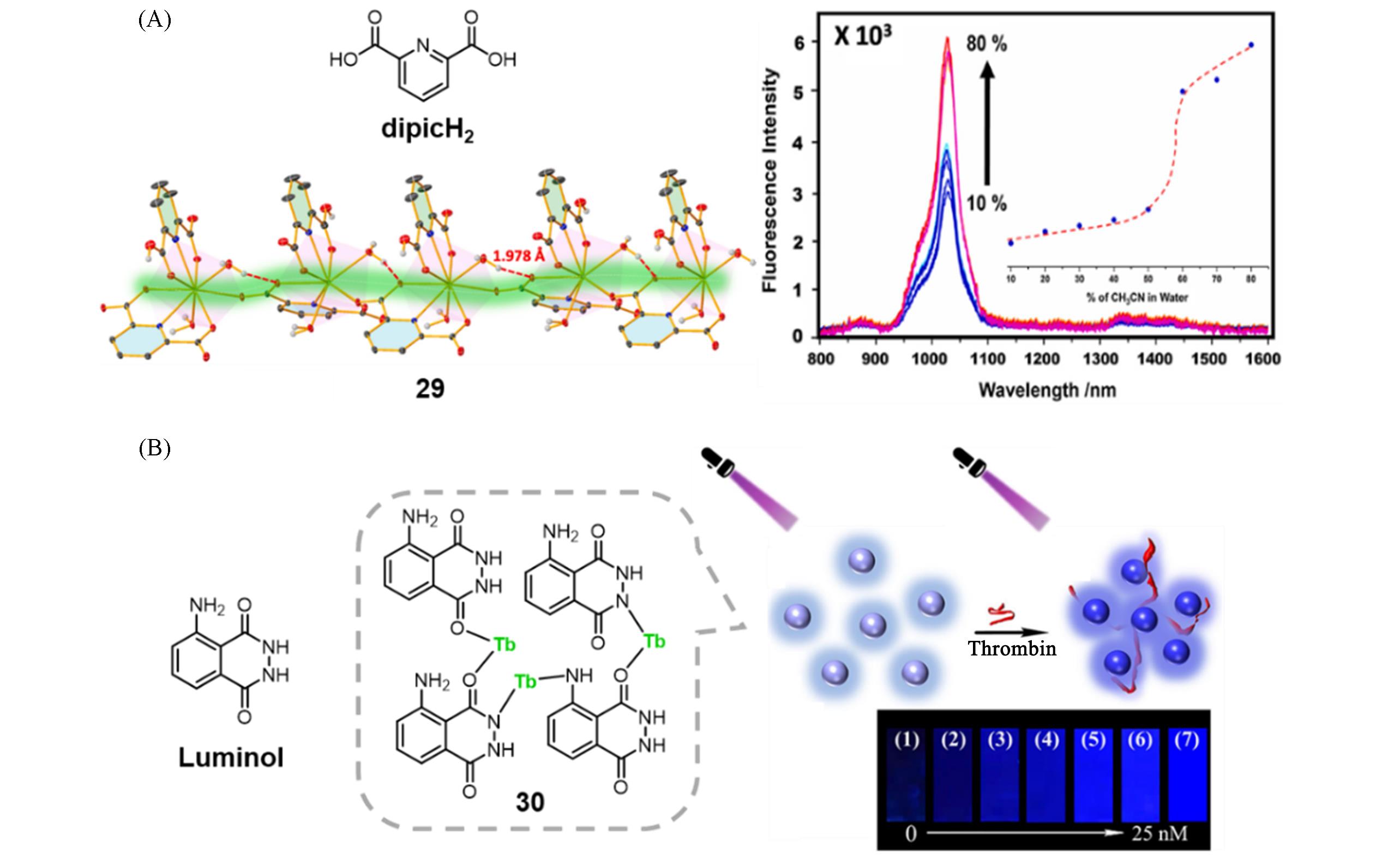
Fig.10 Molecular structure of ligand dipicH2 and crystal structure of polymer 29, PL spectra of polymer 29 in CH3CN with different fraction of water(A)[46] and molecular structure of ligand luminol and polymer 30, fluorescent photographs of the polymer 30 dispersed in Tris buffer in the presence of various concentrations of thrombin(B)[49](A) Copyright 2025, Elsevier Ltd.; (B) Copyright 2019, Springer Nature.
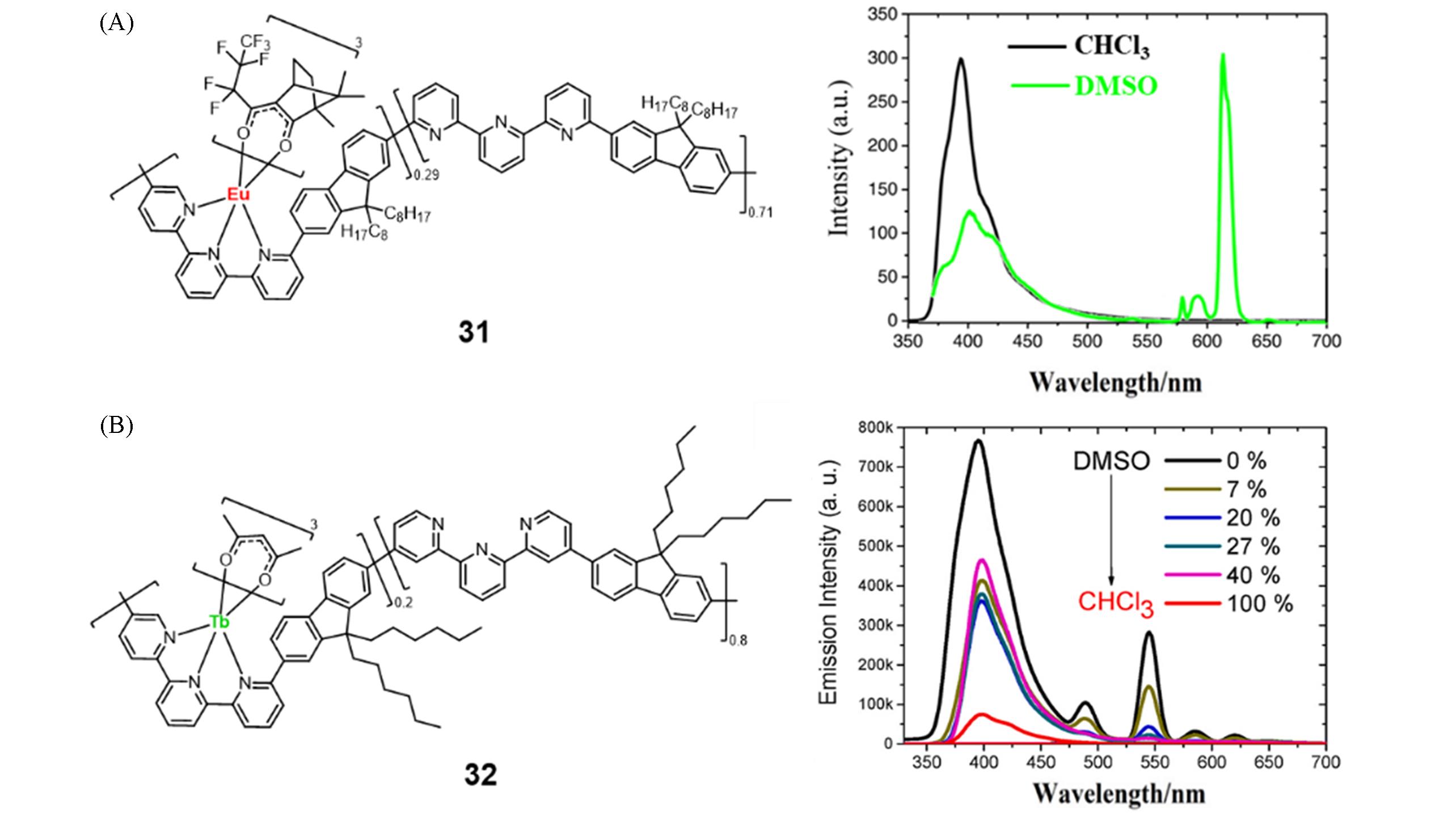
Fig.11 Molecular structure, PL spectra in chloroform and DMSO of metallopolymer 31(A)[50] and molecular structure of metallopolymer 32 and PL spectra of metallopolymer 32 in DMSO/chloroform mixture with different chloroform fractions(B)[51](A) Copyright 2020, Elsevier Ltd.; (B) copyright 2021, Elsevier Ltd.
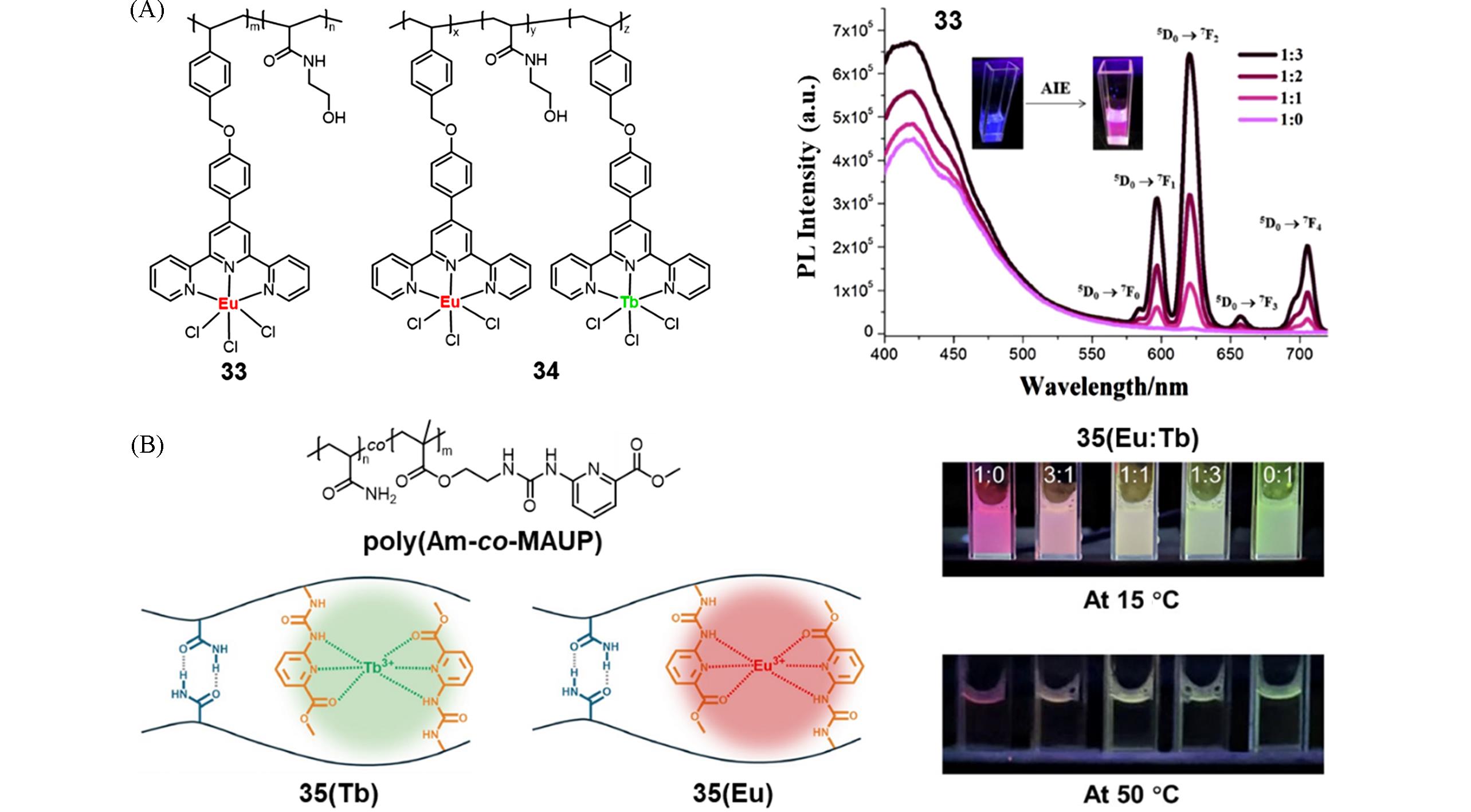
Fig.12 Molecular structure of polymers 33 and 34 and PL spectra in DMF with different fraction of 1,4⁃dioxane(A)[56] and molecular structure of poly(Am⁃co⁃MAUP) and schematic diagram of 35(Tb) and 35(Eu), fluorescent photographs of 35(Ln) consisting of different ratios of Eu3+ to Tb3+ at 15 ℃ and 50 ℃(B)[58](A) Copyright 2018, Elsevier Ltd.; (B) copyright 2025, Science China Press.
| System type | Structural flexibility | Representative applications |
|---|---|---|
| Mononuclear | High | Chemical sensing, bioimaging, and anti⁃counterfeiting |
| Dinuclear | Moderate | Solid⁃state lighting and stimulus⁃responses |
| Polynuclear | Low to moderate | Solid⁃state lighting, chemical sensing, and biosensing |
Table 1 Comparative summary of AIE lanthanide complexes with different number of Ln centers
| System type | Structural flexibility | Representative applications |
|---|---|---|
| Mononuclear | High | Chemical sensing, bioimaging, and anti⁃counterfeiting |
| Dinuclear | Moderate | Solid⁃state lighting and stimulus⁃responses |
| Polynuclear | Low to moderate | Solid⁃state lighting, chemical sensing, and biosensing |
| Ln3+ ion | Emission range(Luminescent color) | Characteristic emission peak/nm | Luminescence lifetime |
|---|---|---|---|
| Tb3+ | Visible(Green) | 545 | ms scale(0.5—2.0 ms) |
| Eu3+ | Visible(Red) | 613 | ms scale(0.2—1.0 ms) |
| Sm3+ | Visible(Orange) | 645 | μs scale(1—50 μs) |
| Yb3+ | NIR⁃II | 980 | μs scale(1—200 μs) |
| Nd3+ | NIR⁃II | 1064 | μs scale(<10 μs) |
Table 2 Comparative properties of Ln3+ ions in AIE lanthanide complexes
| Ln3+ ion | Emission range(Luminescent color) | Characteristic emission peak/nm | Luminescence lifetime |
|---|---|---|---|
| Tb3+ | Visible(Green) | 545 | ms scale(0.5—2.0 ms) |
| Eu3+ | Visible(Red) | 613 | ms scale(0.2—1.0 ms) |
| Sm3+ | Visible(Orange) | 645 | μs scale(1—50 μs) |
| Yb3+ | NIR⁃II | 980 | μs scale(1—200 μs) |
| Nd3+ | NIR⁃II | 1064 | μs scale(<10 μs) |
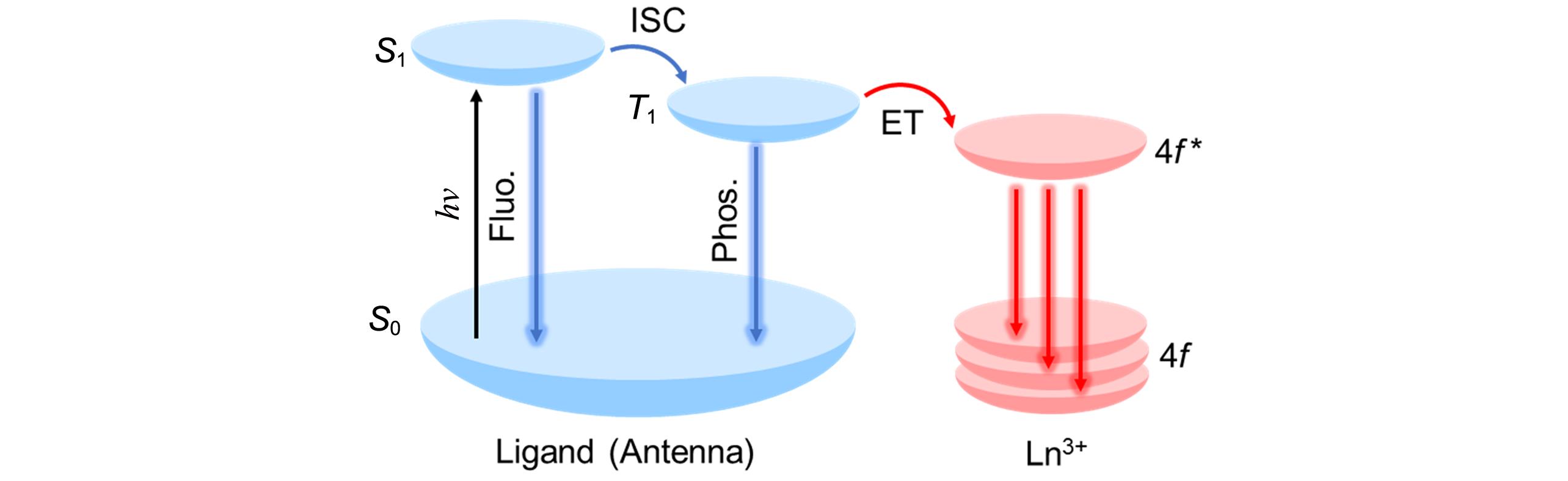
Fig.14 Energy level diagram illustrating the antenna effect and sequential energy transfer processes for lanthanide complexes with characteristic f⁃f electronic transitions
| Mechanism | Dominant motion/vibration targeted | Key trigger for AIE |
|---|---|---|
Restriction of intramolecular rotation(RIR) | Intramolecular rotation(flexible moieties) | Aggregation⁃induced steric hindrance/intermolecular interactions |
Restriction of intramolecular vibration(RIV) | Intramolecular vibration(ligand backbones) | Aggregation⁃induced conformational rigidity |
Electronic⁃vibrational decoupling(EVD) | Electronic⁃vibrational coupling | Concentration increase/solvent⁃induced ligand exchange |
Ln3+⁃coordination⁃induced aggregation | Cluster dispersion⁃aggregation transition | Ln3+⁃ligand coordination |
Table 3 Summary of core AIE mechanisms in lanthanide complexes
| Mechanism | Dominant motion/vibration targeted | Key trigger for AIE |
|---|---|---|
Restriction of intramolecular rotation(RIR) | Intramolecular rotation(flexible moieties) | Aggregation⁃induced steric hindrance/intermolecular interactions |
Restriction of intramolecular vibration(RIV) | Intramolecular vibration(ligand backbones) | Aggregation⁃induced conformational rigidity |
Electronic⁃vibrational decoupling(EVD) | Electronic⁃vibrational coupling | Concentration increase/solvent⁃induced ligand exchange |
Ln3+⁃coordination⁃induced aggregation | Cluster dispersion⁃aggregation transition | Ln3+⁃ligand coordination |
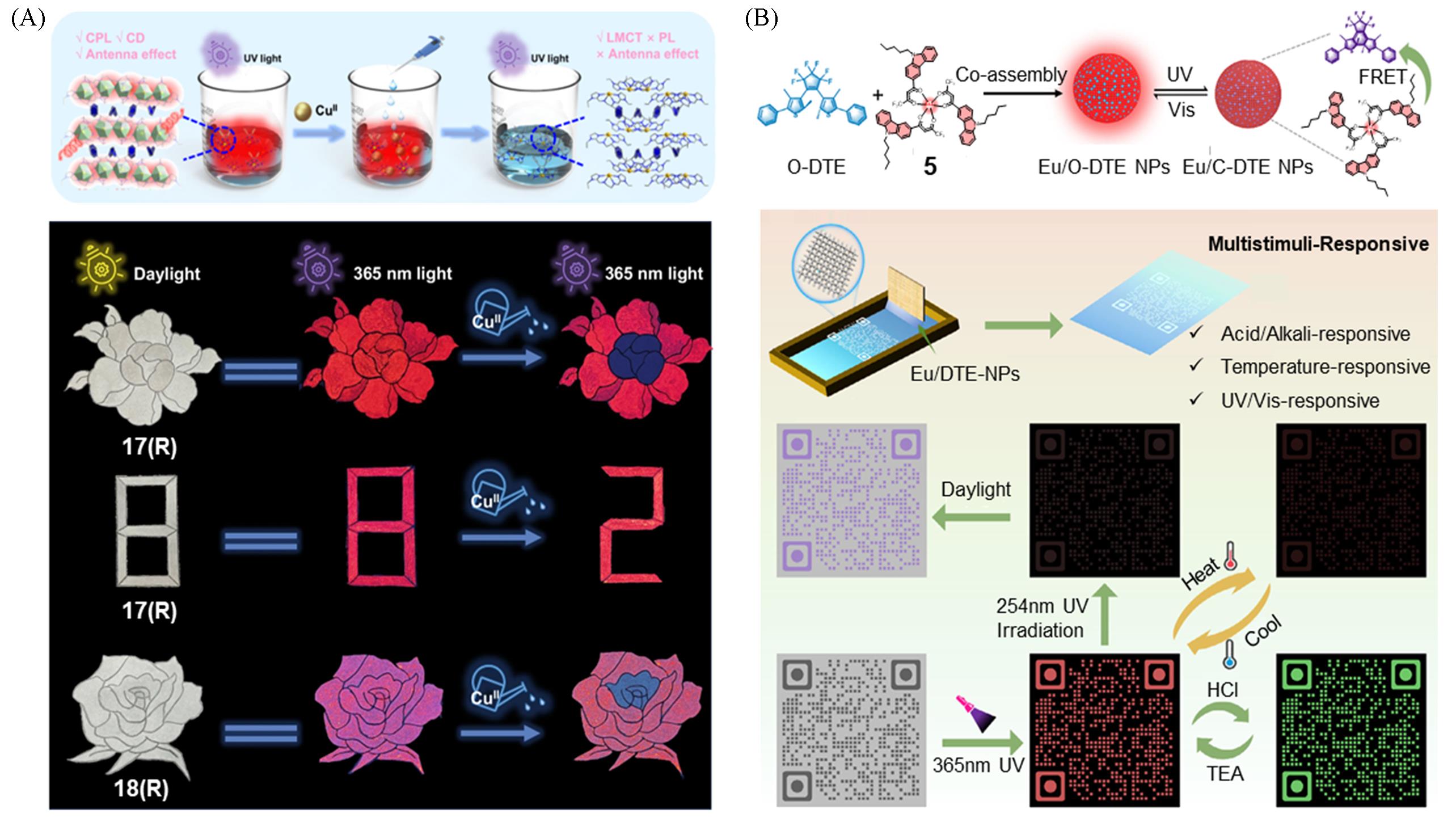
Fig.15 Schematic diagram of the response mechanism of the complexes 17(R, S) and 18(R, S) to copper ions and its real applications(A)[33] and schematic illustration of 5 NPs assemblies and Eu/DTE⁃NPs co⁃assemblies and application of Eu/DTE⁃NPs in multidimensional dynamic information encryption(B)[25](A) Copyright 2024, Springer Nature; (B) copyright 2025, John Wiley and Sons.
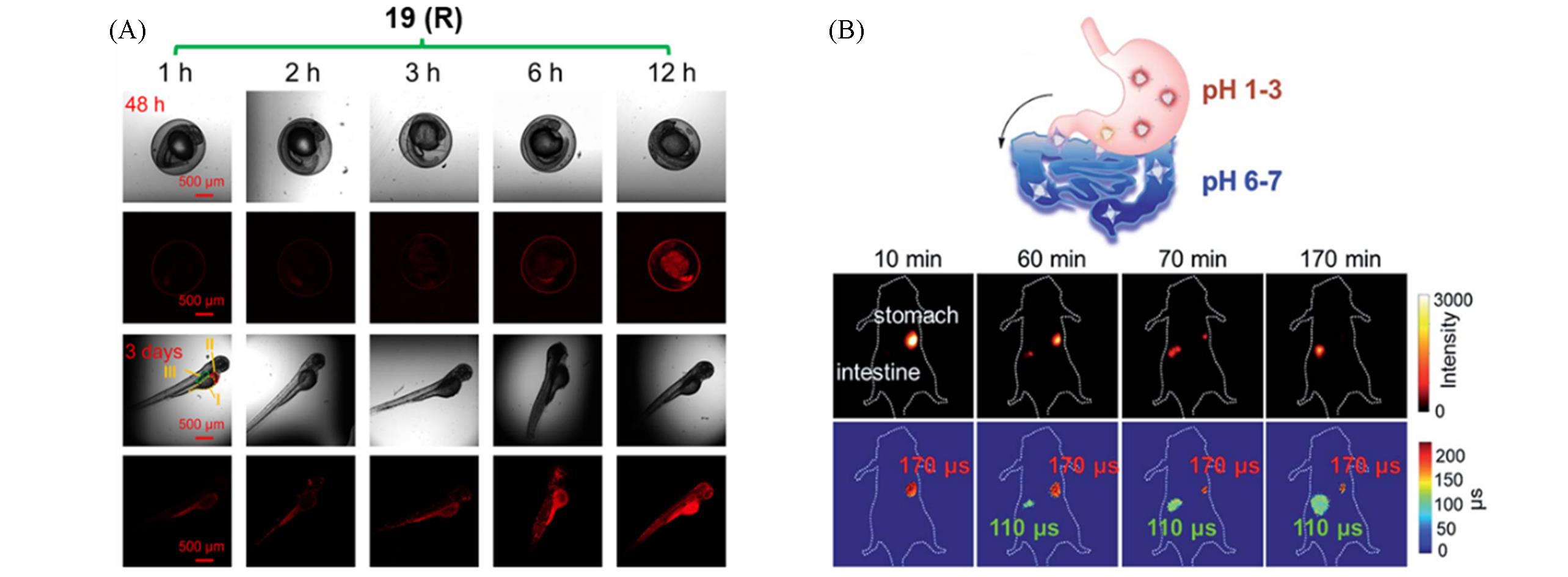
Fig.16 CLSM images and fluorescence intensity diagrams of zebrafish embryos(48 h after fertilization) and larvae(day 3) after incubation with complex 19(R)(A)[35] and schematic illustration of the metabolic process of complex 22 from the stomach to the intestine, and NIR fluorescence intensity imaging of complex 22(B)[37](A) Copyright 2025, Springer Nature; (B) copyright 2019, the Royal Society of Chemistry.
| [13] | Furet E., Costuas K., Rabiller P., Maury O., J. Am. Chem. Soc., 2008, 130(7), 2180—2183 |
| [14] | Thibon A., Pierre V. C., Anal. Bioanal. Chem., 2009, 394(1), 107—120 |
| [15] | Wang T., Wang S. F., Liu Z. Y., He Z. Y., Yu P., Zhao M. Y., Zhang H. X., Lu L. F., Wang Z. X., Wang Z. Y., Zhang W. A., Fan Y., Sun C. X., Zhao D. Y., Liu W. M., Bünzli J. C. G., Zhang F., Nat. Mater., 2021, 20(11), 1571—1578 |
| [16] | Xu J., Luo R., Luo Z. C., Xu J., Mu Z., Bian H. Y., Chan S., Y., Tan. B. Y. H., Chi D. Z., An Z. F., Xing G. C., Qin X., Gong C. Y., Wu Y. M., Liu X. G., Nat. Photon., 2025, 19(1), 71—78 |
| [17] | Andres J., Hersch R. D., Moser J. E., Chauvin A. S., Adv. Funct. Mater., 2014, 24(32), 5029—5036 |
| [18] | Yang Y. L., Hu X. M., Yang Z., Huang W., Adv. Funct. Mater., 2025, 35(2), 2412970 |
| [19] | Fernández⁃Fariña S., Kotova O., Donohoea S. R., Gunnlaugsson T., Chem. Soc. Rev., 2025, 54(23), 11226—11265 |
| [20] | Zhang Y., Jiao P. C., Xu H. B., Tang M. J., Yang X. P., Huang S. M., Deng J. G., Sci. Rep., 2015, 5(1), 9335 |
| [21] | Wu J. J., Zhao C., Zhu Z. H., Li X. L., Ashebr T. G., Tang J. K., Chem. Asian J., 2022, 17(23), e202200913 |
| [22] | Zhuo H., Guan D. B., He J. C., Xu H. B., Zeng M. H., Chem. Eur. J., 2021, 27(65), 16204—16211 |
| [23] | Su P. R., Liang L. J., Wang T., Zhou P. P., Cao J., Liu W. S., Tang Y., Chem. Eng. J., 2021, 413, 127408 |
| [24] | Wang S. Y., Wang L., Fang F., Ma X., Guo Y. Y., Wang R. D., Zhang S. S., Zhang Z., Du L., Zhao Q. H., Inorg. Chem. Front., 2023, 10(18), 5258—5269 |
| [25] | Liang L. J., Yang X., Yan X. Y., Kou Y., Zhang Y. H., Su P. R., Tang Y., Adv. Mater., 2026, 38(2), e14252 |
| [26] | de Sá G. F., Malta O. L., de Mello⁃Donegá C., Simas A. M., Longo R. L., Santa⁃Cruz P. A., da Silva E. F. Jr., Coord. Chem. Rev., 2000, 196(1), 165—195 |
| [27] | Kai T., Kishimoto M., Akita M., Yoshizawa M., Chem. Commun., 2018, 54(8), 956—959 |
| [28] | Xu M. J., Wang J. H., Liao Z. X., Liu J. Q., Yi H., Hao H. X., Li F., Xu S. Y., Liu F. F., Peng Y. L., Wang S. L., Cao X. D., Zhang P. F., Luminescence, 2025, 40(8), e70289 |
| [29] | Wong H. Y., Chan W. T. K., Law G. L., Molecules, 2019, 24(4), 662 |
| [30] | Su P. R., Wang T., Zhou P. P., Yang X. X., Feng X. X., Zhang M. N., Liang L. J., Tang Y., Yan C. H., Natl. Sci. Rev., 2022, 9(1), nwab016 |
| [31] | Chen F. F., Wang J. M., Xu W., Ren Z. Z., Peng G., Huang T., Zhao F., J. Alloy. Compd., 2025, 1010, 177421 |
| [32] | Zhang G. P., Zhu H. X., Chen M. J., Pietraszkiewicz M., Pietraszkiewicz O., Li H. G., Hao J. C., Chem. Eur. J., 2018, 24(59), 15912—15920 |
| [33] | Li Y. L. Wang H. L., Zhu Z. H., Wang Y. F., Liang F. P., Zou H. H., Nat. Commun., 2024, 15(1), 2896 |
| [34] | Wang H. L., Yu B., Liang F. P., Zou H. H., Chin. Chem. Lett., 2025, 111346 |
| [35] | Qin W. W., Yu B., Zhang G. H., Tang M. J., Zou H. H., Liang F. P., Zhu Z. H., Rare Met., 2025, 44(12), 10390—10403 |
| [36] | Yang S. S., Guo Z. P., Hu Z. Q., Guo D. C., Luminescence, 2021, 36(2), 306—315 |
| [37] | Ning Y. Y., Cheng S. M., Wang J. X., Liu Y. W., Feng W., Li F. Y., Zhang J. L., Chem. Sci., 2019, 10(15), 4227—4235 |
| [38] | Bodedla G. B., Zhu X., Wong W. Y., Aggregate, 2023, 4(3), e330 |
| [39] | Wang Y. F., Wang J. J., Zhang S. W., Tang N., Ou X. W., Jiang J. H., Ma F. L., Alam P., Qiu Z. J., Wang W. J., Zhao Z., Lam J. W. Y., Tang B. Z., ACS Nano, 2025, 19(27), 25042—25051 |
| [40] | Leng J. Q., Li H. F., Chen P., Sun W. B., Gao T., Yan P. F., Dalton Trans., 2014, 43(32), 12228—12235 |
| [41] | Ai J. F., Li Y. L., Wang H. L., Liang F. P., Zhu Z. H., Zou H. H., Inorg. Chem., 2023, 62(48), 19552—19564 |
| [42] | Nakai T., Shima K., Shoji S., Fushimi K., Hasegawa Y., Kitagawa Y., Front. Chem., 2023, 11, 1154012 |
| [43] | Pan J. K., Chen Z., Wang H., Li X. P., Yu X. J., Sci. China Mater., 2025, 68(10), 3485—3510 |
| [44] | Yang D. Q., Li H. M., Li H. R., Coord. Chem. Rev., 2024, 514, 215875 |
| [45] | Zhu Y. X., Wei Z. W., Pan M., Wang H. P., Zhang J. Y., Su C. Y., Dalton Trans., 2016, 45(3), 943—950 |
| [46] | Raghuvanshi K., Verma A., Sunkari S. S., J. Lumin., 2025, 283, 121284 |
| [47] | Tong Y. J., Yu L. D., Wu L. L., Cao S. P., Zhang L., Xia X. H., Qiu J. D., Chem. Commun., 2018, 54(54), 7487—7490 |
| [48] | Tong Y. J., Song A. M., Yu L. D., Liang R. P., Qiu J. D., Microchim. Acta, 2019, 187(1), 53 |
| [49] | Qi W. J., Zhao M. Y., Fu Y. L., He H. K., Tian X., Wu D., Zhang Y., Hu P. P., Dyes Pigm., 2020, 172, 107797 |
| [50] | Campos E. C. G., Turchetti D. A., Santana A. J., Domingues R. A., Duarte L., Atvars T. D. Z., Akcelrud L., Synth. Met., 2021, 273, 116686 |
| [51] | Campos E. C. G., Turchetti D. A., Zanlorenzi C., Domingues R. A, Duarte L. G. T. A., Atvars T. D. Z., Akcelrud L., Polymer, 2021, 229, 123990 |
| [52] | Campos E. C. G., Turchetti D. A., Domingues R. A., Akcelrud L. C., Synth. Met., 2025, 311, 117802 |
| [53] | Zhang S. X., Yin W. D., Yang Z. M., Shah I., Yang Y., Li Z., Zhang S. J., Zhang B., Lei Z. Q., Ma H. C., Anal. Chem., 2020, 92(1), 7808—7815 |
| [54] | Huang Y. J., Feng W. X., Zhou Z. P., Zheng H. Z., Zhao Y., Yan H. X., Lv X. Q., J. Mater. Chem. C, 2022, 10(19), 7586—7593 |
| [55] | Feng W. X., Huang Y. J., Zhao Y., Tian W., Yan H. X., ACS Appl. Mater. Interfaces, 2023, 15(13), 17211—17221 |
| [56] | Zhang Z., Chang H., Kang Y. F., Li X. P., Jiang H. E., Xue B. L., Wang Y. Y., Lv X. Q., Zhu X. J., Sensor. Actuat. B⁃Chem., 2019, 282, 999—1007 |
| [57] | Zhang Z., Chen Y. X., Chang H., Wang Y. Y., Li X. P., Zhu X. J., J. Mater. Chem. C, 2020, 8(6), 2205—2210 |
| [58] | Fan X. Z., Zhou Z. H., Luo M. Q., Banquy X., Zhang J. W., Zhu X. X., Zhang C. Z., Sci. China Chem., 2025, 68(11), 5949—5959 |
| [59] | You J. G., Lu C. Y., Kumar A. S. K., Tseng W. L., Nanoscale, 2018, 10(37), 17691—17698 |
| [60] | Ma F. H., Deng L., Wang T. T., Zhang A. M., Yang M. H., Li X. Q., Chen X., Microchim. Acta, 2023, 190(8), 291 |
| [61] | Ma J., Cong X., Ou K. D., Liao Y. G., Yang Y. J., Wang H., Sensor. Actuat. B⁃Chem., 2023, 390, 133904 |
| [62] | Huang X. M., Chen H. Y., Huang R., Shi Y. D., Ye R. H., Qiu B., Microchim. Acta, 2024, 191(1), 54 |
| [63] | Mu J., Zhang H. F., Huang Z. Z., Jia Q., Spectrochim. Acta A, 2023, 291, 122388 |
| [64] | Pan T. T., Zhou T., Tu Y. F., Yan J. L., Talanta, 2021, 227, 122197 |
| [65] | Miao W. J., Wang L., Liu Q., Guo S., Zhao L. Z., Peng J. J., Chem. Asian J., 2021, 16(3), 247—251 |
| [66] | Madni A., Zhang Y. C., Zhang L. Y., Qiang Y. N., Qi S. D., Zhai H. L., Food Compos. Anal., 2025, 148, 108429— |
| [67] | Saraci F., Quezada⁃Novoa V., Donnarumma P. R., Howarth A. J., Chem. Soc. Rev., 2020, 49(22), 7949—7977 |
| [68] | Xu H., Cheng P., Aggregate, 2024, 5(3), e518 |
| [69] | Yin H. Q., Wang X. Y., Yin X. B., J. Am. Chem. Soc., 2019, 141(38), 15166—15173 |
| [70] | Yang L., Dou Y., Qin L., Chen L. L., Xu M. Z., Kong C., Zhang D. P., Zhou Z., Wang S. N., Inorg. Chem., 2020, 59(22), 16644—16653 |
| [71] | Li G. Y., Tong C. L., Anal. Chim. Acta, 2020, 1133, 11—19 |
| [72] | Li Z. J., Jiang F. L., Yu M. X., Li S. C., Chen L., Hong M. C., Nat. Commun., 2022, 13(1), 2142 |
| [73] | Xu S. J., Xiong G., Zhang X. Y., Huang K., Qin D. B., Zhao B., Cryst. Growth Des., 2023, 23(6), 4214—4221 |
| [74] | Zhai X. Y., Kou Y., Liang L. J., Liang P. Y., Su P. R., Tang Y., Inorg. Chem., 2023, 62(45), 18533—18542 |
| [75] | Yan Z. W., Ling Y., Zeng Q., Sun Z., Li N. B., Luo H. Q., Sensor. Actuat. B⁃Chem., 2024, 417, 136169 |
| [76] | Li Y. L., Wang H. L., Xiao Z. X., Ai J. F., Liang F. P., Zhu Z. H., Zou H. H., ACS Appl. Mater. Interfaces, 2024, 16(45), 62301—62313 |
| [77] | Wang H. L., Li Y. H., Zou H. H., Liang F. P., Zhu Z. H., Adv. Mater., 2025, 37(29), 2502742 |
| [78] | Li Z. J., Zhang J. P., Wu Z. Y., Lei H., Zhao Y. F., Qi W. Q., Gao X., Jiang F. L., Liu Y. S., Chen L., Hong M. C., Adv. Sci., 2025, 12(4), e10147 |
| [79] | Li X. Q., Zhao J. W., Wang Y. L., Liu W., Zhao S. S., Chen X., Tian T. Y., Zhang H., Sun Q., Zhao Z., Inorg. Chem., 2025, 64(34), 17313—17321 |
| [80] | Zhang J., He B. Z., Hu Y. P., Alam P., Zhang H. K., Lam J. W. Y., Tang B. Z., Adv. Mater., 2021, 33(32), 2008071 |
| [1] | Luo J., Xie Z., Lam J. W. Y., Cheng L., Chen H., Qiu C., Kwok H. S., Zhan X., Liu Y., Zhu D., Tang B. Z., Chem. Commun., 2001, (18), 1740—1741 |
| [2] | Han P. B., Xia E. H., Qin A. J., Tang B. Z., Coord. Chem. Rev., 2022, 473, 214843 |
| [3] | Yang S. Y., Zhang L., Kong F. C., Chen Y. Y., Li W. J., Wang F., Liu C., He X., Xiao X. D., Wang J., Sun J. W., Chow P. C. Y., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Chem, 2025, 11(5), 102534 |
| [4] | Yan D., Wang D., Tang B. Z., Nat. Rev. Bioeng., 2025, 3(11), 976—991 |
| [5] | Chen W. Z., Guan H. T., Lu Y. F., Zeng G. H., Gu D., Guo K. Y., Jiang C., Liu H. X., Aggregate, 2025, 6(4), e70008 |
| [6] | Mei J., Hong Y. N., Lam J. W. Y., Qin A. J., Tang Y. H., Tang B. Z., Adv. Mater., 2014, 26(31), 5429—5479 |
| [7] | Mei J., Leung N. L. C., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Chem. Rev., 2015, 115(21), 11718—11940 |
| [8] | Yang S. Y., Chen Y. Y., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Chem. Soc. Rev., 2024, 53(11), 5366—5393 |
| [9] | Pei Y., Sun Y., Huang M. J., Zhang Z. J., Yan D. Y., Cui J., Zhu D. X., Zeng Z. B., Wang D., Tang B. Z., Biosensors, 2022, 12(12), 1104 |
| [10] | Mauro M., Cebrián C., Isr. J. Chem., 2018, 58(8), 901—914 |
| [11] | Sathish V., Ramdass A., Thanasekaran P., Lu K. L., Rajagopal S., J. Photoch. Photobio. C, 2015, 23, 25—44 |
| [12] | Wang F., Liu X. G., Chem. Soc. Rev., 2009, 38(4), 976—989 |
| [1] | 朱高桦, 舒菊, 耿江涛, 马夫龙, 熊玲红, 何学文. 原位激活型近红外二区聚集诱导发光探针应用于肿瘤中过氧化氢的高灵敏成像[J]. 高等学校化学学报, 2026, 47(5): 20260058. |
| [2] | 潘晶珂, 齐馨, 张露露, 王贝贝, 吕超. 主客体作用力构建的聚集诱导发光型超分子聚合物的合成与应用研究[J]. 高等学校化学学报, 2026, 47(5): 20260130. |
| [3] | 尹诗琪, 郑志刚, 何心桐, 王世敏, 顾星桂, 王二静. 从原生到功能: AIE天然产物的发光、 改性及应用[J]. 高等学校化学学报, 2026, 47(5): 20260042. |
| [4] | 焦海丽, 郑小燕. 限域环境诱导有机发光增强的理论研究进展[J]. 高等学校化学学报, 2026, 47(5): 20260060. |
| [5] | 曾宪平, 秦毅, 王东. 聚集诱导发光探针用于手术导航[J]. 高等学校化学学报, 2026, 47(5): 20260066. |
| [6] | 翟哲, 刘雷静, 田文晶. 聚集诱导发光分子探针的设计及在细胞器成像中的应用[J]. 高等学校化学学报, 2026, 47(5): 20260078. |
| [7] | 吴锐, 李政, 李琪, 石佳俊, 赵艳, 冯维旭, 颜红侠. 含Si, P, B的非传统超支化发光聚合物研究进展[J]. 高等学校化学学报, 2026, 47(5): 20250411. |
| [8] | 李伟健, 徐小琴, 王威, 杨海波. 聚集诱导发光树枝状分子的研究进展[J]. 高等学校化学学报, 2026, 47(5): 20260003. |
| [9] | 李雨婷, 罗亮. 拉曼成像技术的前沿进展及与聚集体科学的交叉研究[J]. 高等学校化学学报, 2026, 47(4): 20260008. |
| [10] | 方瑾钰, 黄瀚玮, 宋航, 吴谦, 赵征, 唐本忠. 智能响应, 精准点亮: 酶响应的聚集诱导发光材料在生物医学中的诊疗新策略[J]. 高等学校化学学报, 2026, 47(4): 20260009. |
| [11] | 孙妍, 朱东霞. 近红外激发有机发光材料的构筑及在疾病治疗中的应用[J]. 高等学校化学学报, 2026, 47(4): 20260002. |
| [12] | 杨湛, 邓皇俊, 池振国. 具有聚集诱导发光性质的氢键有机框架的研究进展[J]. 高等学校化学学报, 2026, 47(4): 20260012. |
| [13] | 李银, 汤睿霖, 瞿超, 程亮慧, 胡玉玺, 吴钰祥, 王志明. 离子化策略实现马来酸酐水溶性光敏探针的制备及其在高效抗菌中的应用[J]. 高等学校化学学报, 2026, 47(4): 20250413. |
| [14] | 任奥成, 李青云, 吉晓帆. 荧光超分子聚合物网络[J]. 高等学校化学学报, 2026, 47(4): 20250390. |
| [15] | 张阳戴翼, 邵研, 姜世梅. 基于动态氢键网络协同调控AIE与力学性能的多响应水凝胶[J]. 高等学校化学学报, 2026, 47(4): 20250381. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
