

高等学校化学学报 ›› 2026, Vol. 47 ›› Issue (5): 20260060.doi: 10.7503/cjcu20260060
• 综合评述 • 上一篇
收稿日期:2026-01-30
出版日期:2026-05-10
发布日期:2026-03-25
通讯作者:
郑小燕
E-mail:xiaoyanzheng@bit.edu.cn
基金资助:
JIAO Haili1,2, ZHENG Xiaoyan2( )
)
Received:2026-01-30
Online:2026-05-10
Published:2026-03-25
Contact:
ZHENG Xiaoyan
E-mail:xiaoyanzheng@bit.edu.cn
Supported by:摘要:
聚集诱导发光(AIE)分子为固态有机发光材料的应用与开发提供了新路径. 然而, 溶液态AIE体系的发光效率受限于其分子内运动导致的非辐射跃迁衰变, 通过构建“限域”环境能够有效抑制其非辐射衰减通道, 从而实现发光增强. 目前, 实验上调控发光的限域环境多样, 且发光调控机制不同, 但其微观机制尚不清楚. 本文综合评述了近年来多尺度理论模拟揭示不同限域环境中AIE发光增强机制的研究进展. 系统阐述了不同限域环境[如无定形聚集、 (共)结晶、 高压、 主客体包合、 细胞膜及光化学反应]对分子构象、 分子堆积、 电子结构及激发态动力学的调控规律, 阐明了分子结构、 限域环境与发光性能之间的构效关系, 从理论上揭示了限域诱导发光增强的微观机制, 为高性能AIE材料的设计与性能调控提供了理论依据, 有助于推动其在光电、 生物成像及传感等领域的应用.
中图分类号:
TrendMD:
焦海丽, 郑小燕. 限域环境诱导有机发光增强的理论研究进展. 高等学校化学学报, 2026, 47(5): 20260060.
JIAO Haili, ZHENG Xiaoyan. Progress on Theoretical Study of Organic Luminescence Enhancement Induced by Confined Environments. Chem. J. Chinese Universities, 2026, 47(5): 20260060.
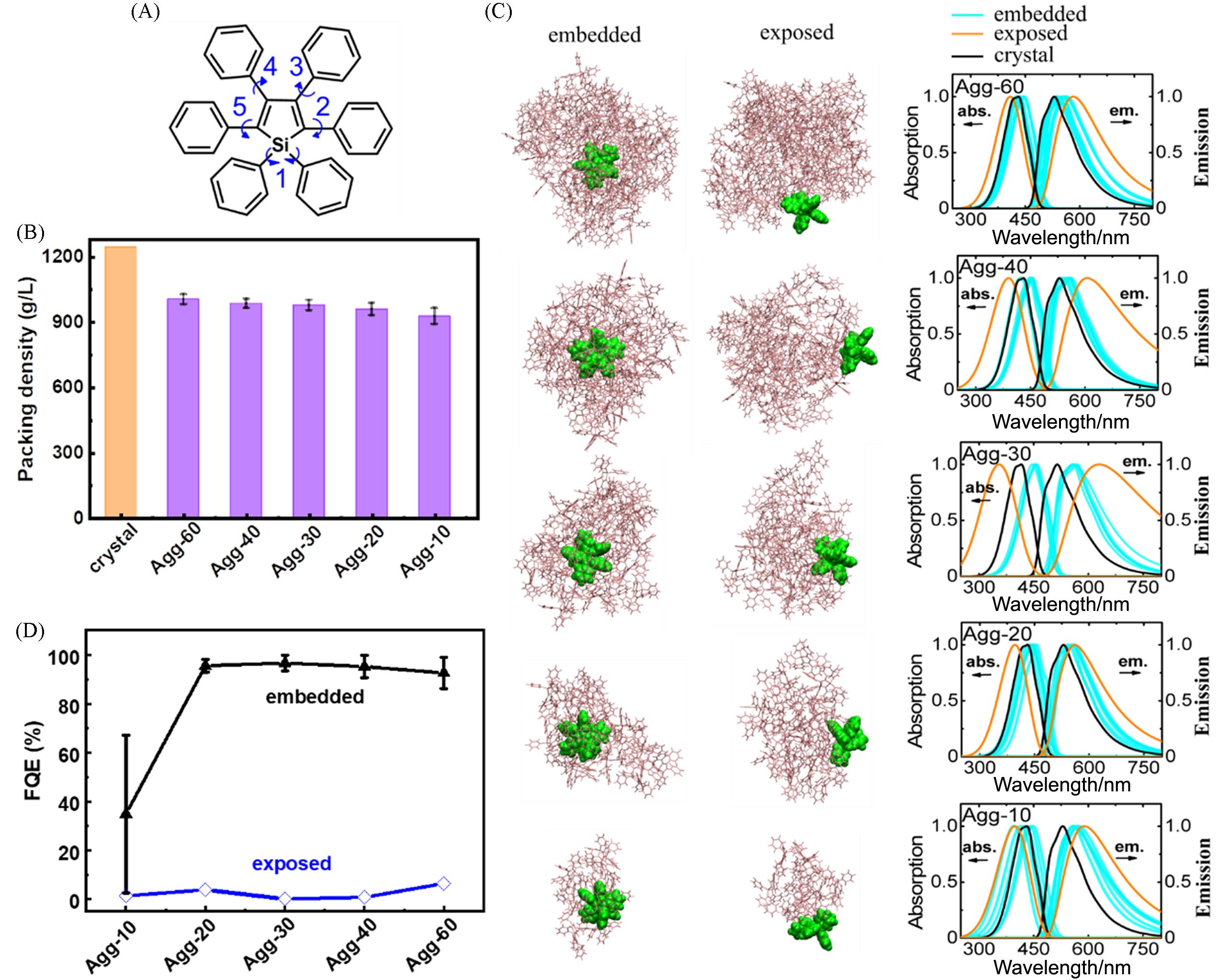
Fig.2 Molecular structure of HPS with key dihedral angles between each phenyl group and the central silacycle highlighted by blue arrows(A), the average packing density of HPS aggregates with different sizes and crystals(B), the representative QM/MM models of the embedded and exposed HPS molecules in aggregates with different sizes, as well as the calculated vibrationally resolved normalized absorption(left) and emission(right) spectra for five embedded(cyan line) and one exposed HPS molecule(orange line) in aggregates, the corresponding spectra of the HPS crystal are shown(black line) for comparison(C) and the FQE of embedded and exposed HPS molecule in amorphous aggregates with different sizes(D)[20]Copyright 2016, the Royal Society of Chemistry.
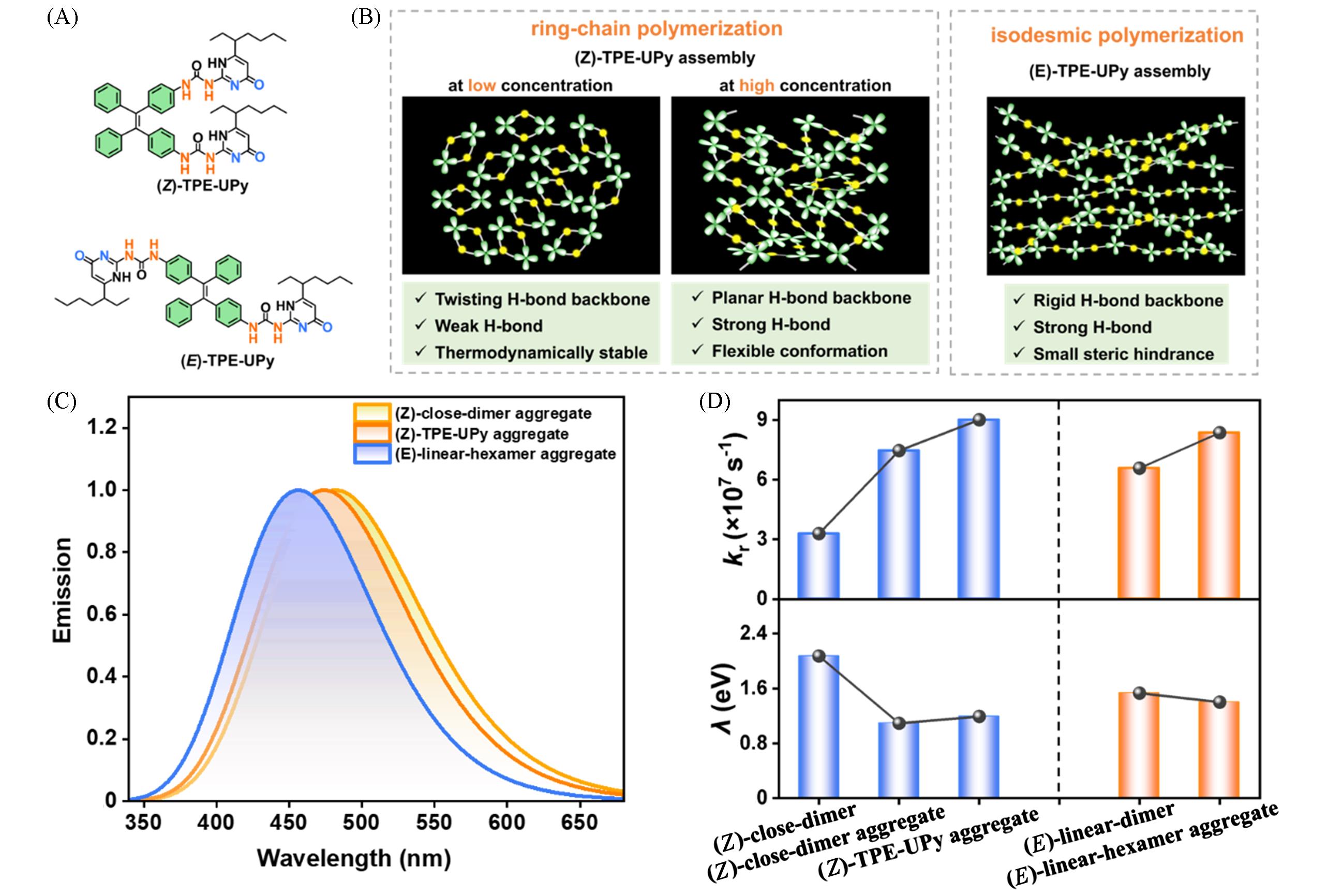
Fig.3 Chemical structures of (Z)⁃/(E)⁃TPE⁃UPy(A), schematic diagrams of (Z)⁃TPE⁃UPy and (E)⁃TPE⁃UPy assemblies(B), the emission spectra of three representative aggregates(C) and the krand λ of five representative systems(D)[56]Copyright 2023, the Royal Society of Chemistry.
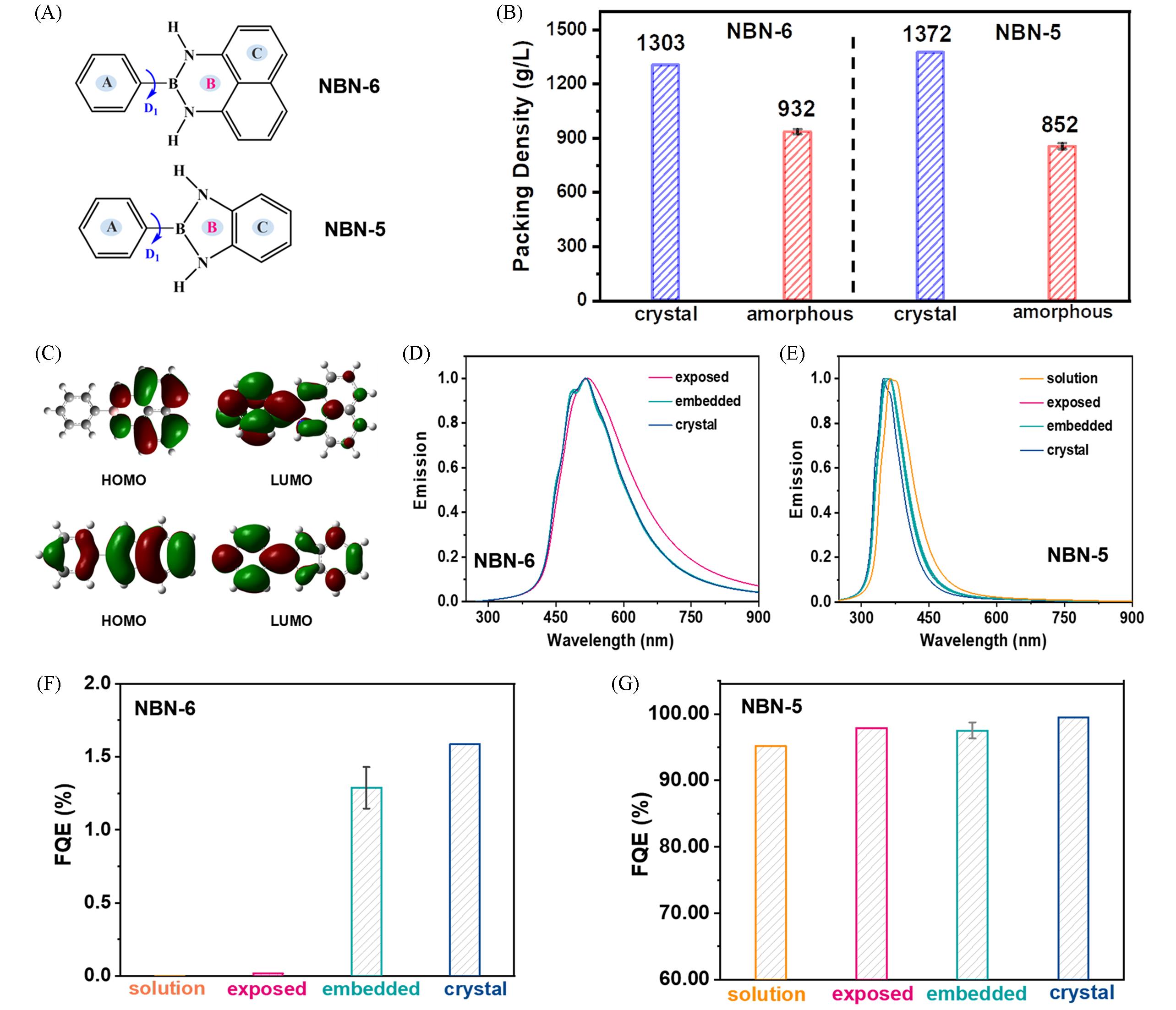
Fig.4 Chemical structures of two representative NBN⁃doped PAH systems: six⁃membered NBN⁃6 and five⁃membered NBN⁃5(A), the packing density of NBN⁃6 and NBN⁃5 in amorphous aggregates and crystals(B), the electron density contours of HOMO and LUMO in S0 geometry in methanol(C), the calculated emission spectra of NBN⁃6(D) and NBN⁃5(E) in different environments and the FQE of NBN⁃6(F) and NBN⁃5(G) in different environments[62]Copyright 2021, the Royal Society of Chemistry.
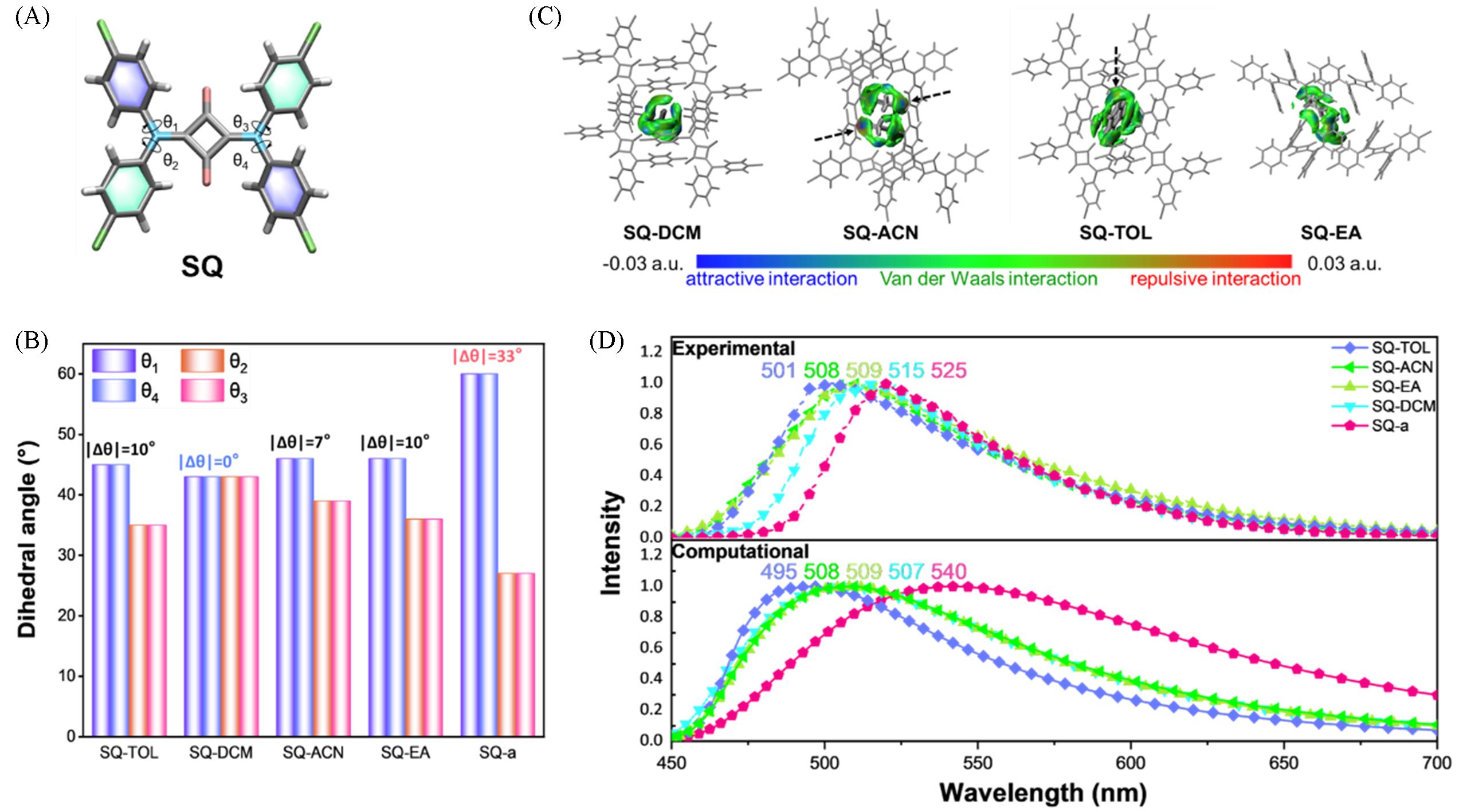
Fig.5 Molecular structure of SQ with its four chlorobenzene rings and one central squaraine core(A, the key dihedral angle between four chlorobenzene ring and the central squaraine core are marked as θ1, θ2, θ3, and θ4), key dihedral angles and their relative difference(Δθ, Δθ=|θ1-θ2|) of all five SQ⁃based systems(B), the independent gradient model analysis of four solvated co⁃crystals(C) and experimental and calculated emission spectra of five different SQ⁃based crystals(D)[70]Copyright 2024, Elsevier B.V.
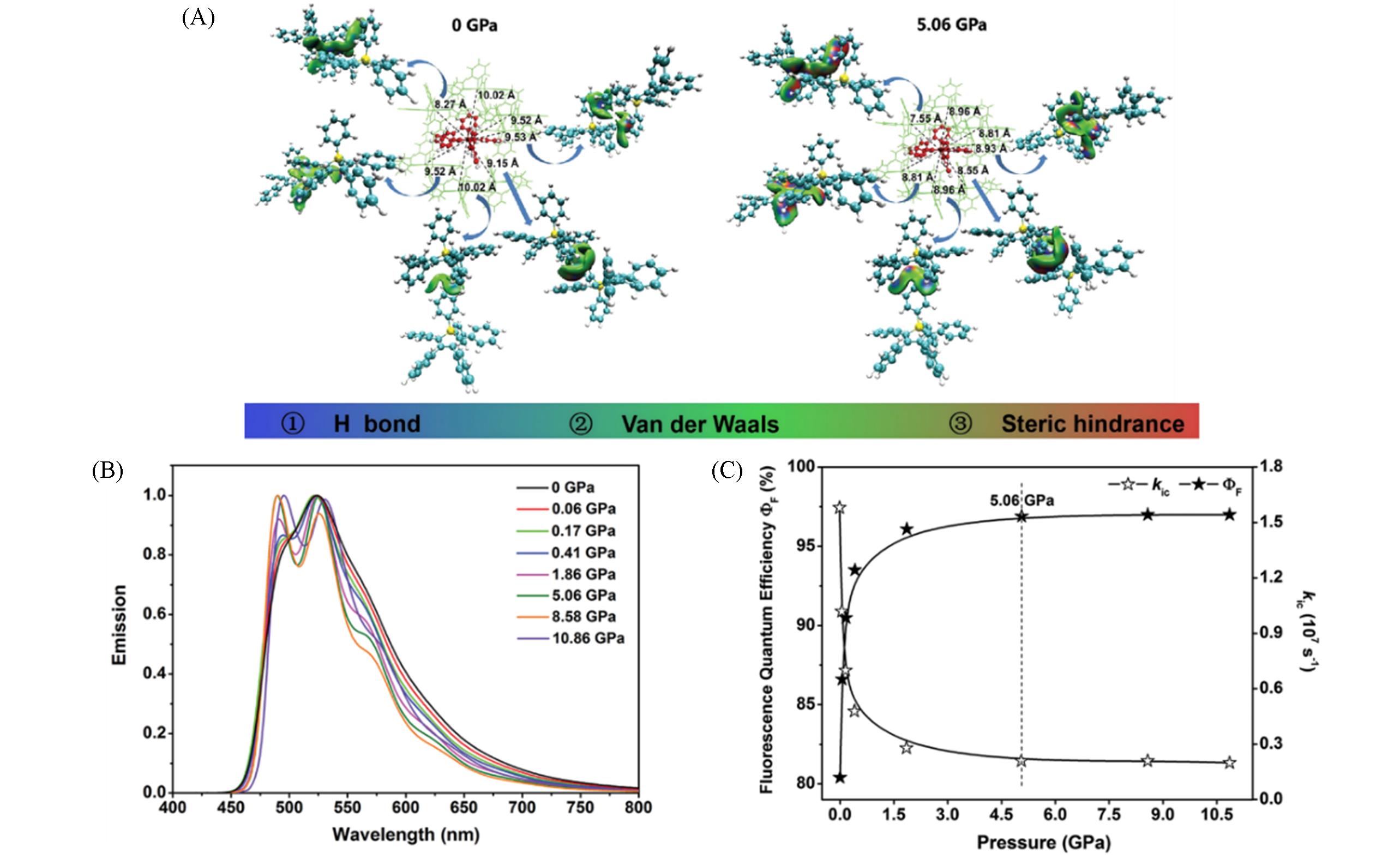
Fig.6 Molecular packing structures within ~1 nm of the QM centroid and relevant intermolecular interactions in the HPS aggregates at 0 GPa and 5.06 GPa(A), calculated vibrationally resolved normalized emission spectra for HPS in aggregates at different pressures(B) and calculated kic and FQE of HPS in aggregates at different pressures(C)[73]Copyright 2019, the Royal Society of Chemistry.
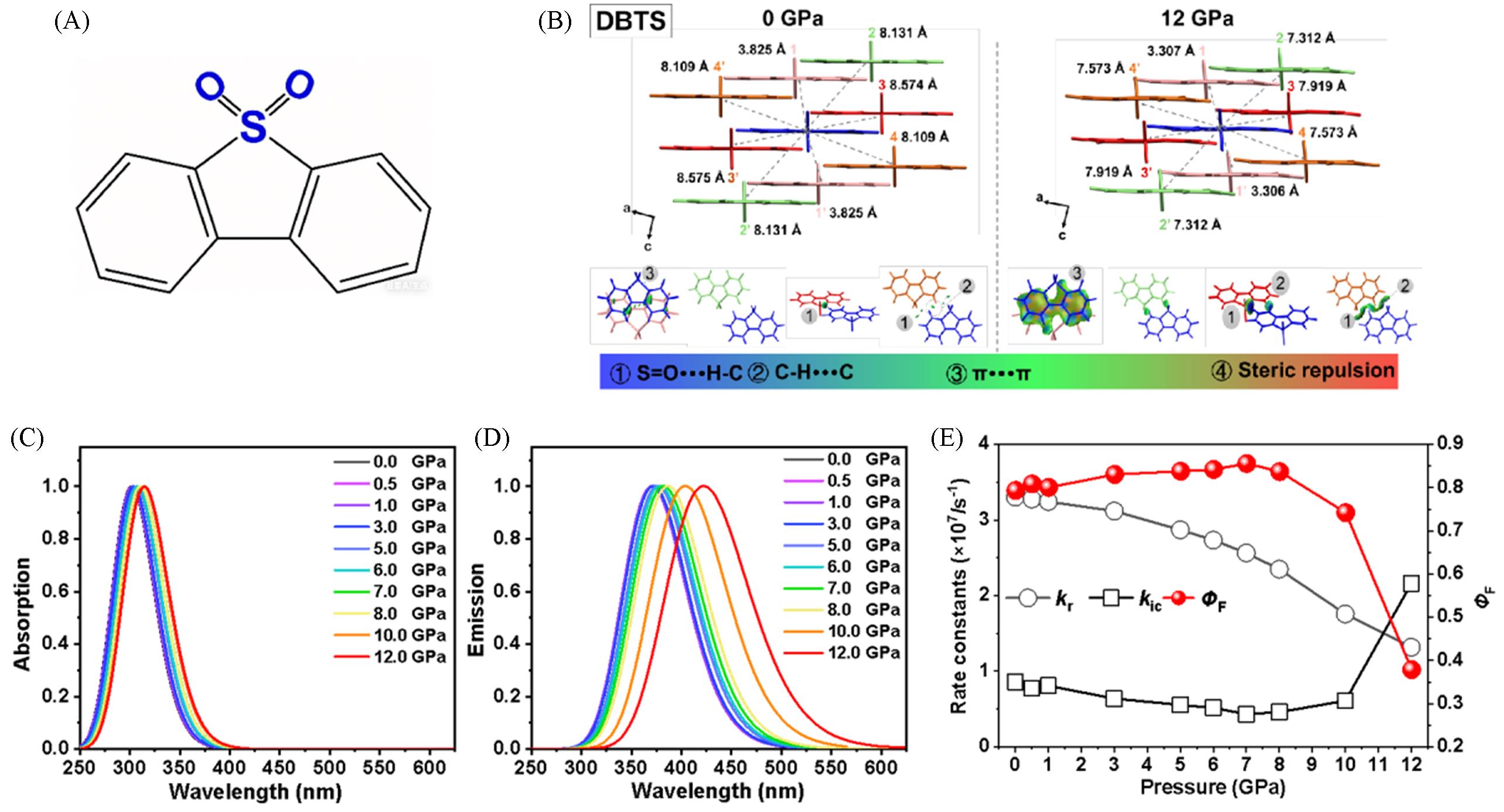
Fig.7 Chemical structure of DBTS(A), molecular packing of DBTS and the IGM isosurfaces of the dimers between QM molecules and surrounding molecules(0.01 au) at pressures of 0 and 12 GPa(B), calculated normalized absorption(C) and emission(D) spectra of DBTS crystals at different pressures and calculated kr, kic, and ΦF of DBTS crystals at different pressures(E)[78]Copyright 2022, the American Chemical Society.
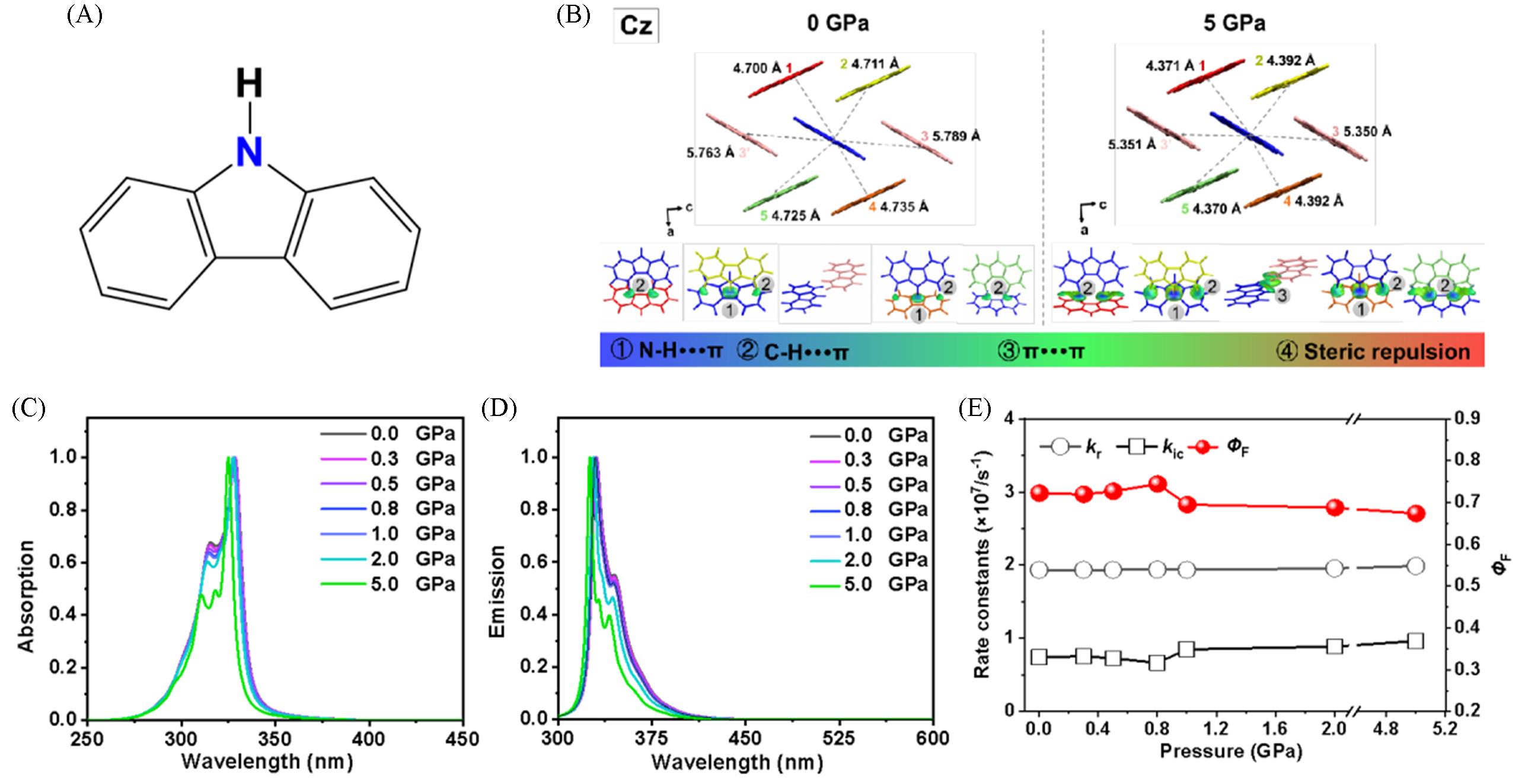
Fig.8 Chemical structures of Cz(A), molecular packing of Cz and the IGM isosurfaces of the dimers between QM molecules and surrounding molecules(0.01 au) at pressures of 0 and 5 GPa(B), calculated normalized absorption(C) and emission(D) spectra of Cz crystals at different pressures and calculated kr, kic and ΦF of Cz crystals at different pressures(E)[78]Copyright 2022, American Chemical Society.
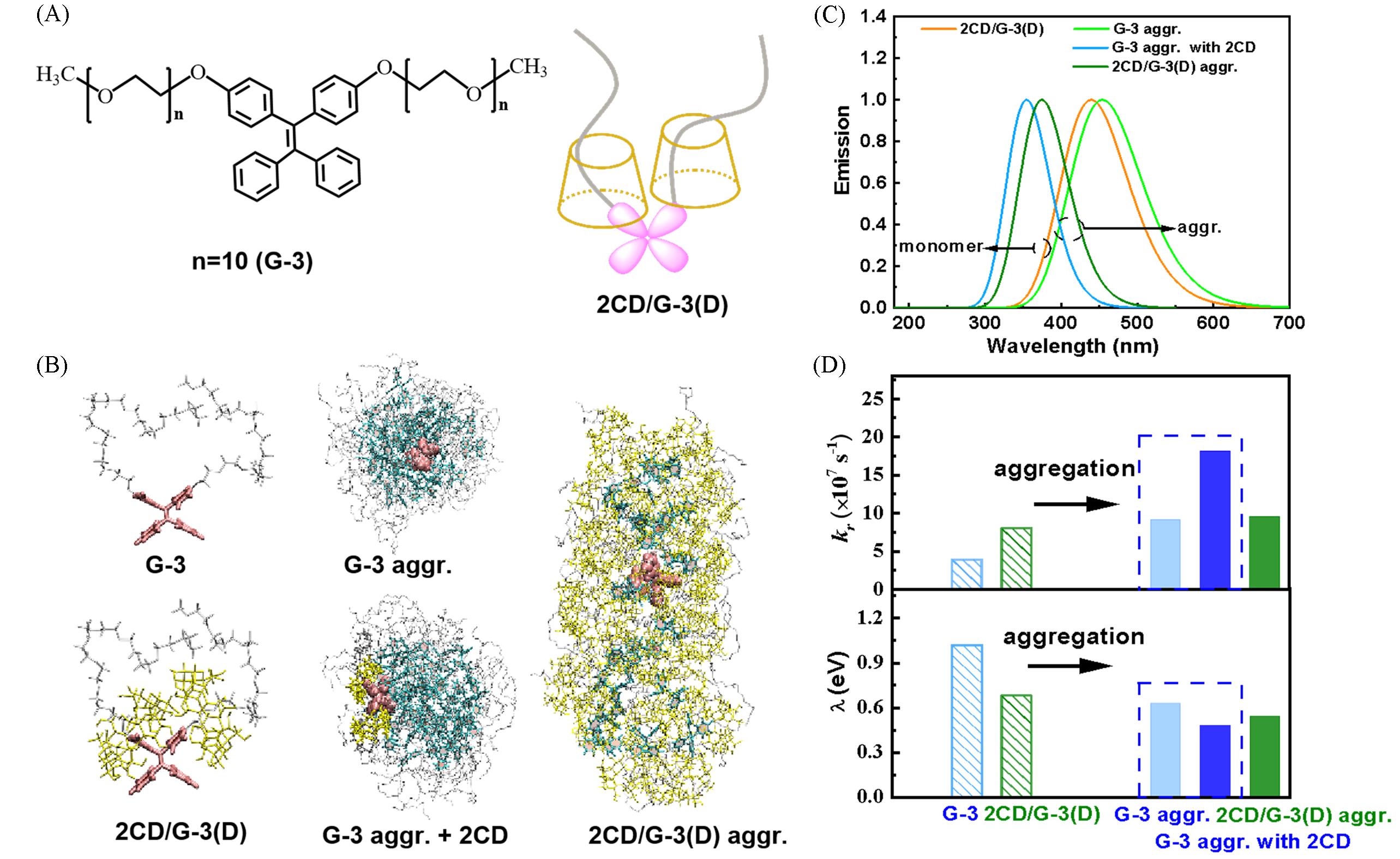
Fig.9 Schematic diagrams of guest molecule G⁃3 and host⁃guest inclusion 2CD/G⁃3(D)(A), the QM/MM models of the representative systems: monomers[G⁃3, host⁃guest inclusion 2CD/G⁃3(D)] and aggregates[G⁃3, G⁃3 aggregate with 2CD and 2CD/G⁃3(D)](B), the emission spectra of monomer and aggregates(C) and the krand λ of the monomers and aggregates(D)[34]Copyright 2021, the Royal Society of Chemistry.
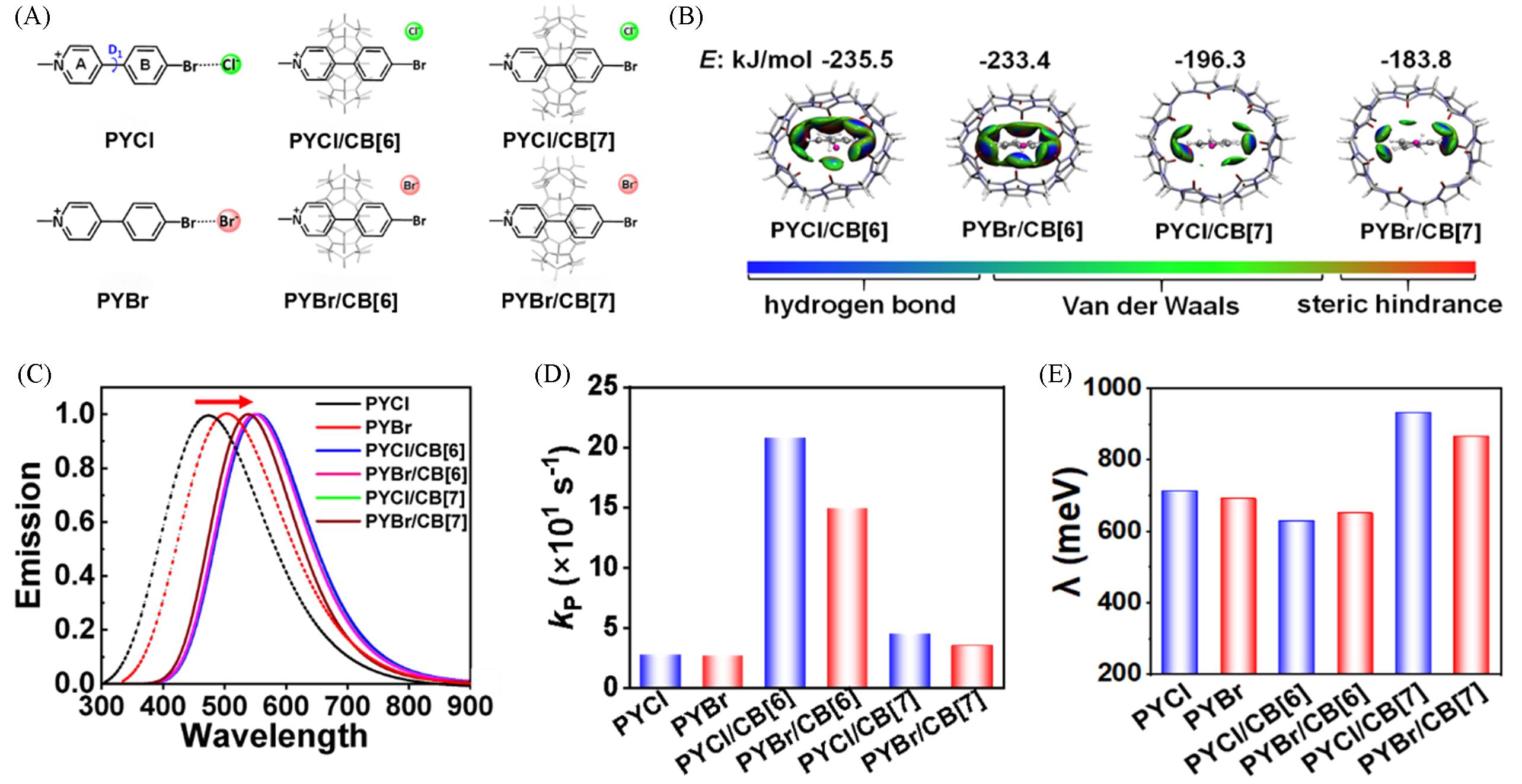
Fig.10 Chemical structures of two guests(PYCl and PYBr) and four host⁃guest complexes(PYCl/CB[6], PYBr/CB[6], PYCl/CB[7] and PYBr/CB[7])(A), the calculated binding energies between guests and hosts, and the intermolecular interactions of the host⁃guest complexes obtained via IGM analysis(B), the calculated phosphorescence spectra(C), phosphorescence radiative rate constant(kP)(D) and λ(E)of the studied systems in crystalline state[85]Copyright 2024, the Royal Society of Chemistry.
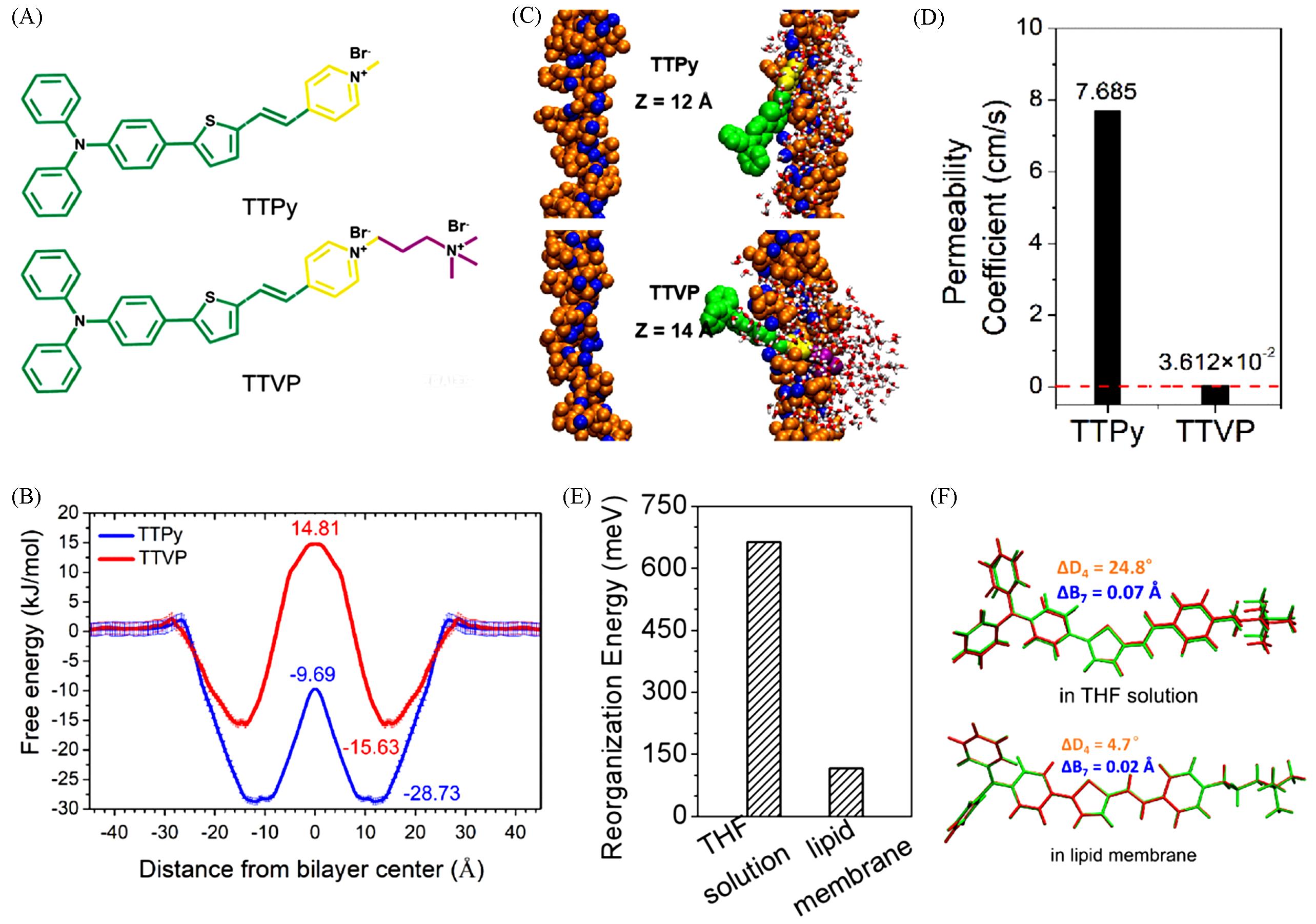
Fig.11 Chemical structures of TTPy and TTVP(A), free energy profiles of TTPy and TTVP across the membrane(B), snapshots of TTPy and TTVP at the most stable positions in lipid membranes(C), the calculated permeability coefficients of two AIEgens(D), calculated total reorganization energies of TTVP(E) and superposition of optimized structures at the S0 and S1 states for TTVP in the lipid membrane and dilute THF solution(F)[74]Copyright 2019, the Royal Society of Chemistry.
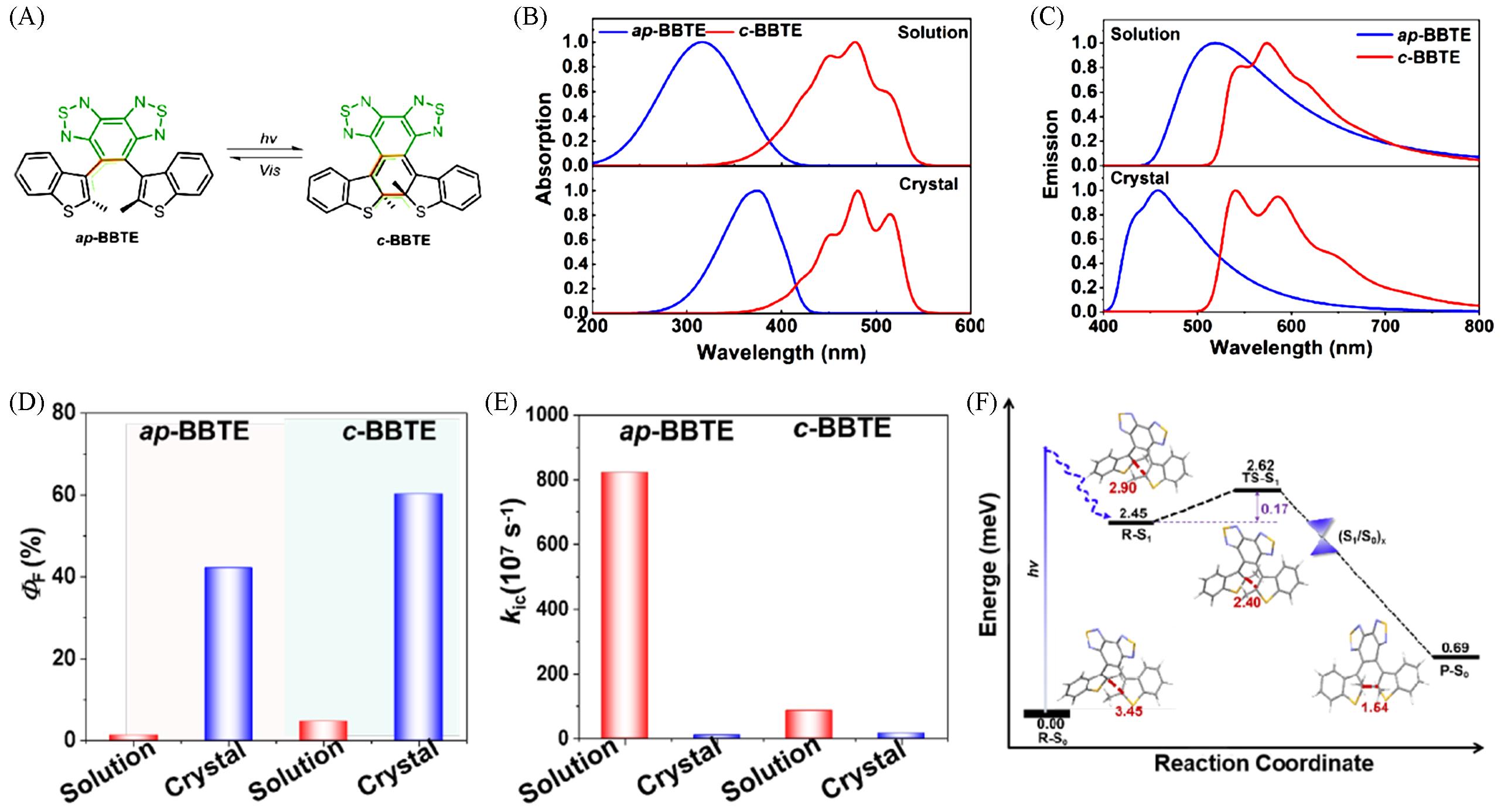
Fig.12 Reversible photocyclization reaction from ap⁃BBTE to c⁃BBTE(A), absorption(B) and emission(C) spectra of ap⁃BBTE and c⁃BBTE in solution and crystalline state, the ΦF(D)and kic(E) of ap⁃BBTE and c⁃BBTE in dilute THF solution and the crystalline state, and potential energy profiles(in eV) for photocyclization reaction in singlet states (S1) of ap⁃BBTE(F, bond distances are given in Å, 1 Å=0.1 nm)[95]Copyright 2024, American Chemical Society.
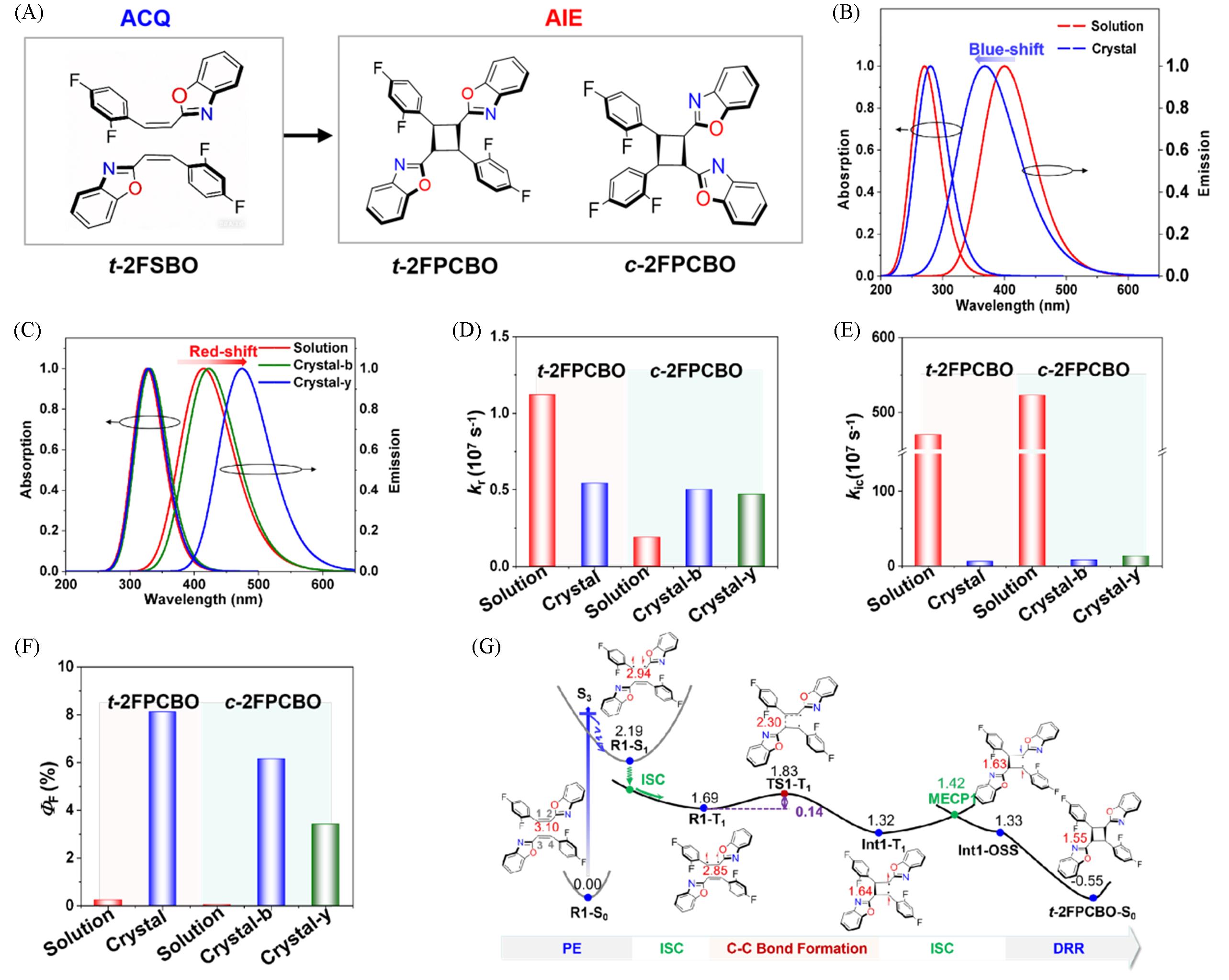
Fig.13 Schematic illustration of the [2+2] photocycloaddition reaction of t⁃2FSBO and the formation of dimers t⁃2FPCBO and c⁃2FPCBO(A), the calculated absorption and emission spectra of t⁃2FPCBO(B), c⁃2FPCBO⁃b and c⁃2FPCBO⁃y(C), calculated kr(D), kic(E) and ΦF(F)of t⁃2FPCBO and c⁃2FPCBO in dilute solution and crystalline states and potential energy profiles(energy in eV) of the UV light⁃induced [2+2] cycloaddition reaction of t⁃2FSBO dimer(G)[96]Copyright 2024, the Royal Society of Chemistry.
| [1] | Hong Y. I., Lam J., W. Y., Tang B. Z., Chem. Commun., 2009, 29, 4332—4353 |
| [2] | Fan X., Sun J., Wang F., Chu Z., Wang P., Dong Y., Hu R., Tang B. Z., Zou D., Chem. Commun., 2008, 26, 2989—2991 |
| [3] | Wang R., Hu M., Chem. Commun., 2025, 61, 4991—4994 |
| [4] | Zheng S., Zhu T., Wang Y., Yang T., Yuan W. Z., Angew. Chem. Int. Ed., 2020, 59, 10018—10022 |
| [5] | Yang S., Jia Q., Ou X., Sun F., Song C., Zhao T., Kwok R. T. K., Sun J., Zhao Z., Lam J. W. Y., Wang Z., Tang B. Z., J. Am. Chem. Soc., 2025, 147, 3570—3583 |
| [6] | Qu R., Zhen X., Jiang X., CCS Chem., 2022, 4(2), 401—419 |
| [7] | Xu R., Zhang P., Shen Q., Zhou Y., Wang Z., Xu Y., Meng L., Dang D., Tang B. Z., Coord. Chem. Rev., 2023, 477, 214944 |
| [8] | Peng Q., Shuai Z., Aggregate, 2021, 2, e91 |
| [9] | Huang S., Feng B., Cheng X., Huang X., Ding J., Yu K., Dong J., Zeng W., Chem. Eng. J., 2023, 476, 1—11 |
| [10] | Chen M., Nie H., Song B., Li L. Z., Sun J. Z., Qin A. J., Tang B. Z., J. Mater. Chem. C, 2016, 4(14), 2901—2908 |
| [11] | Zhao Z., Chen B., Geng J. L., Chang Z. F., Aparicio‐Ixta L., Nie H., Goh C. C., Ng L. G., Qin A., Ramos‐Ortiz G., Liu B., Tang B. Z., Part. Part. Syst. Charact., 2013, 31(4), 481—491 |
| [12] | Zhao N., Chen S., Hong Y.Tang B. Z., Chem. Commun., 2015, 51(71), 13599—13602 |
| [13] | Zhang L., Liu W., Huang X., Zhang G., Wang X., Wang Z., Zhang D., Jiang X., Analyst, 2015, 140(17), 5849—5854 |
| [14] | Yuan Y., Zhang R., Cheng X., Xu S., Liu B., Chem. Sci., 2016, 7(7), 4245—4250 |
| [15] | Wang H., Liu G., Gao H., Wang Y., Polym. Chem., 2015, 6(26), 4715—4718 |
| [16] | Shi H., Xin D., Gu X., Zhang P., Peng H., Chen S., Lin G., Zhao Z., Tang B. Z., J. Mater. Chem. C, 2016, 4(6), 1228—1237 |
| [17] | Ravindran E., Varathan E., Subramanian V., Somanathan N., J. Mater. Chem. C, 2016, 4(34), 8027—8040 |
| [18] | Bao Y., de Keersmaecker H., Corneillie S., Yu F., Mizuno H., Zhang G., Hofkens J., Mendrek B., Kowalczuk A., Smet M., Chem. Mater., 2015, 27(9), 3450—3455 |
| [19] | Tong S., Dai J., Sun J., Liu Y., Ma X., Liu Z., Ma T., Tan J., Yao Z., Wang S., Zheng H., Wang K., Hong F., Yu X., Gao C., Gu X., Nat. Commun., 2022, 13(1), 5234 |
| [20] | Zheng X., Peng Q., Zhu L., Xie Y., Huang X., Shuai Z., Nanoscale, 2016, 8, 15173—15180 |
| [21] | Hu R., Qin A., Tang B. Z., Prog. Polym. Sci., 2020, 100, 101176 |
| [22] | Dong Y., Lam J. W. Y., Qin A., Liu J., Li Z., Tang B. Z., Sun J., Kwok H. S., Appl. Phys. Lett., 2007, 91(1), 011111 |
| [23] | Yuan H., Wang K., Yang K., Liu B., Zou B., J. Phys. Chem. Lett., 2014, 5(17), 2968—2973 |
| [24] | Wu M., Liu H., Liu H., Lu T., Wang S., Niu G., Sui L., Bai F., Yang B., Wang K., Yang X., Zou B., J. Phys. Chem. Lett., 2022, 13(11), 2493—2499 |
| [25] | Fu Z., Liu H., Zhao J., Zhang X., Zheng X., Yang B., Yang X., Wang K., Zou B., J. Mater. Chem. C, 2021, 9(41), 14578—14582 |
| [26] | Li D., Lu F., Wang J., Hu W., Cao X. M., Ma X., Tian H., J. Am. Chem. Soc., 2018, 140(5), 1916—1923 |
| [27] | Zhang Z., Chen Y., Liu Y., Angew. Chem. Int. Ed., 2019, 58(18), 6028—6032 |
| [28] | Zhang Z. Y., Xu W. W., Xu W. S., Niu J., Sun X., Liu Y., Angew. Chem. Int. Ed., 2020, 59(42), 18748—18754 |
| [29] | Wang H., Xing H., Gong J., Zhang H., Zhang J., Wei P., Yang G., Lam J. W. Y., Lu R., Mater. Horiz., 2020, 7(6), 1566—1572 |
| [30] | Ding L., Wang Z. Y., Yao Z. F., Liu N. F., Wang X. Y., Zhou Y. Y., Luo L. F., Shen Z. H., Wang J. Y., Pei J., Macromolecules, 2021, 54(12), 5815—5824 |
| [31] | Shi W., Wei R., Zhang D., Meng L., Xie J., Cai K., Zhao D., Angew. Chem. Int. Ed., 2022, 61(37), e202208635 |
| [32] | Yang J., Peng Q., Chin. J. Chem. Phys., 2022, 35(1), 38—51 |
| [33] | Zhang H., Zhao Z., Turley A. T., Wang L., Mcgonigal P. R., Tu Y., Li Y., Wang Z., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Adv. Mater., 2020, 32(36), 2001457 |
| [34] | Yang J. F., Peng Q., Xue R. D., Li Z. S., Zheng X. Y., Mater. Chem. Front., 2021, 5(4), 1806—1816 |
| [35] | Yang J., Peng Q., Phys. Chem. Chem. Phys., 2024, 26(26), 18418—18425 |
| [36] | Yang J., Fang M., Li Z., Aggregate, 2020, 1(1), 6—18 |
| [37] | Li Q., Li Z., Acc. Chem. Res., 2020, 53(4), 962—973 |
| [38] | Chen J., Law C. C. W., Lam J. W. Y., Dong Y., Lo S. M. F., Williams I. D., Zhu D., Tang B. Z., Chem. Mater., 2003, 15(7), 1535—1546 |
| [39] | Dong Y., Lam J. W. Y., Qin A., Sun J., Liu J., Li Z., Sun J., Sung H. H. Y., Williams I. D., Kwok H. S., Tang B. Z., Chem. Commun., 2007, (31), 3255—3257 |
| [40] | Guo W. J., Ma S., Wang H., Qiao L., Chen L., Hong C., Liu B., Zheng X., Peng H. Q., Aggregate, 2024, 5(1), e415 |
| [41] | Jiang L., Cao S., Cheung P. P. H., Zheng X., Leung C. W. T., Peng Q., Shuai Z., Tang B. Z., Yao S., Huang X., Nat. Commun., 2017, 8, 15639 |
| [42] | Yang J., Lv J., Jiao Y., Zheng X., Peng Q., Aggregate, 2025, 6, e70029 |
| [43] | Wang H., Zheng X., Phys. Chem. Chem. Phys., 2022, 24(32), 19011—19028 |
| [44] | Amabilino D. B., Smith D. K., Steed J. W., Chem. Soc. Rev., 2017, 46(9), 2404—2420 |
| [45] | Lou X. Y., Yang Y. W., Aggregate, 2020, 1(1), 19—30 |
| [46] | Liu M., Zhang L., Wang T., Chem. Rev., 2015, 115(15), 7304—7397 |
| [47] | You L., Zha D., Anslyn E. V., Chem. Rev., 2015, 115(15), 7840—7892 |
| [48] | Guo W. J., Peng T., Zhu W., Ma S., Wang G., Li Y., Liu B., Peng H. Q., Aggregate, 2023, 4, e297 |
| [49] | Hu R., Lam J. W. Y., Liu J., Sung H. H. Y., Williams I. D., Yue Z., Wong K. S., Yuen M. M. F., Tang B. Z., Polym. Chem., 2012, 3(6), 1481—1489 |
| [50] | Peng H., Liu B., Wei P., Zhang P., Zhang H., Zhang J., Li K., Li Y., Cheng Y., Lam J. W. Y., Zhang W., Lee C., Tang B. Z., ACS Nano, 2019, 13(1), 839—846 |
| [51] | Wang Z., Cheng X., Qin A., Zhang H., Sun J. Z., Tang B. Z., J. Phys. Chem. B, 2018, 122(7), 2165—2176 |
| [52] | Liu Y., Cao Y., Zhang X., Lin Y., Li W., Demir B., Searles D. J., Whittaker A. K., Zhang A., ACS Nano, 2021, 15(12), 20067—20078 |
| [53] | Mahadevi A. S., Sastry G. N., Chem. Rev., 2016, 116(5), 2775—2825 |
| [54] | Datta S., Saha M. L.Stang P. J., Acc. Chem. Res., 2018, 51(9), 2047—2063 |
| [55] | Peng H. Q., Zheng X., Han T., Kwok R. T. K., Lam J. W. Y., Huang X., Tang B. Z., J. Am. Chem. Soc., 2017, 139(29), 10150—10156 |
| [56] | Wang H., Yang J., Zheng X., Phys. Chem. Chem. Phys., 2023, 25(20), 14387—14399 |
| [57] | Yoshii R., Hirose A., Tanaka K., Chujo Y., Chem. Eur. J., 2014, 20(27), 8320—8324 |
| [58] | Neena K. K., Thilagar P., J. Mater. Chem. C, 2016, 4(48), 11465—11473 |
| [59] | Wan W. M., Tian D., Jing Y. N., Zhang X. Y., Wu W., Ren H., Bao H. L., Angew. Chem. Int. Ed., 2018, 57(47), 15510—15516 |
| [60] | Ju C. W., Li B., Li L., Yan W., Cui C., Ma X., Zhao D., J. Am. Chem. Soc., 2021, 143(15), 5903—5916 |
| [61] | Kalluvettukuzhy N. K., Thilagar P., Organometallics, 2017, 36(14), 2692—2701 |
| [62] | Zeng Y., Yang J., Zheng X., Phys. Chem. Chem. Phys., 2021, 23, 23986—23997 |
| [63] | Huang Q., Li W., Mao Z., Qu L., Li Y., Zhang H., Yu T., Yang Z., Zhao J., Zhang Y., Aldred M. P., Chi Z., Nat. Commun., 2019, 10(1), 3017 |
| [64] | Huang Q., Li W., Mao Z., Zhang H., Li Y., Ma D., Wu H., Zhao J., Yang Z., Zhang Y., Gong L., Aldred M. P., Chi Z., Chem, 2021, 7(5), 1321—1332 |
| [65] | Li L., Ma H., Zhang J., Zhao E., Hao J., Huang H., Li H., Li P., Gu X., Tang B. Z., J. Am. Chem. Soc., 2021, 143(10), 3856—3864 |
| [66] | Shi Y., Wang S., Tao W., Guo J., Xie S., Ding Y., Xu G., Chen C., Sun X., Zhang Z., He Z., Wei P., Tang B. Z., Nat. Commun., 2022, 13(1), 1882 |
| [67] | Shi Y., Ding Y., Tao W., Wei P., ACS Appl. Mater. Interface, 2022, 14(31), 36071—36078 |
| [68] | Huang Q., Li W., Yang Z., Zhao J., Li Y., Mao Z., Yang Z., Liu S., Zhang Y., Chi Z., CCS Chem., 2022, 4(5), 1643—1653 |
| [69] | Chen L., Yuan Z., Zhang H., Ye Y., Yang Y., Xiang F., Cai K., Xiang S., Chen B., Zhang Z., Angew. Chem. Int. Ed., 2022, 61(51), e202213959 |
| [70] | Yang X., Zeng Y., Wei H., Gu X., Zheng X., Surf. Interfaces, 2024, 52, 104911 |
| [71] | Lu X., Sun Y., Zhang Z., Shuai Z., Hu W., Chin. Chem. Lett., 2021, 32(3), 1233—1236 |
| [72] | Gu Y., Wang K., Dai Y., Xiao G., Ma Y., Qiao Y., Zou B., J. Phys. Chem. Lett., 2017, 8(17), 4191—4196 |
| [73] | Zhang T., Shi W., Wang D., Zhuo S., Peng Q., Shuai Z., J. Mater. Chem. C, 2019, 7, 1388—1398 |
| [74] | Zheng X., Wang D., Xu W., Cao S., Peng Q., Tang B. Z., Mater. Horiz., 2019, 6(10), 2016—2023 |
| [75] | Zhao J., Zheng X., Front. Chem., 2022, 9, 808957 |
| [76] | Zhang T., Jiang Y., Niu Y., Wang D., Peng Q., Shuai Z., J. Phys. Chem. A, 2014, 118(39), 9094—9104 |
| [77] | Tian S., Ma H., Wang X., Lv A., Shi H., Geng Y., Li J., Liang F., Su Z. M., An Z., Huang W., Angew. Chem. Int. Ed., 2019, 58(20), 6645—6649 |
| [78] | Zhao J., Zeng Y., Zheng X., Chem. Mater., 2022, 34(23), 10711—10720 |
| [79] | Hong M., Ravva M. K., Winget P., Brédas J. L., Chem. Mater., 2016, 28(16), 5791—5798 |
| [80] | Liang G., Lam J. W. Y., Qin W., Li J., Xie N., Tang B. Z., Chem. Commun., 2014, 50(14), 1725—1727 |
| [81] | Liow S. S., Zhou H., Sugiarto S., Guo S., Chalasani M. L. S., Verma N. K., Xu J., Loh X. J., Biomacromolecules, 2017, 18(3), 886—897 |
| [82] | Wang J., Huang Z., Ma X., Tian H., Angew. Chem., 2020, 59(25), 9928—9933 |
| [83] | Song S., Zheng H. F., Li D. M., Wang J. H., Feng H. T., Zhu Z. H., Chen Y. C., Zheng Y. S., Org. Lett., 2014, 16(8), 2170—2173 |
| [84] | Cai Y., Du L., Samedov K., Gu X., Qi F., Sung H. H. Y., Patrick B. O., Yan Z., Jiang X., Zhang H., Lam J. W. Y., Williams I. D., Lee Phillips D., Qin A., Tang B. Z., Chem. Sci., 2018, 9(20), 4662—4670 |
| [85] | Luo X., Zeng Y., Wei H., Zheng X., Phys. Chem. Chem. Phys., 2024, 26(18), 14071—14078 |
| [86] | Luo J., Xie Z., Lam J. W., Cheng L., Chen H., Qiu C., Kwok H. S., Zhan X., Liu Y., Zhu D., Tang B. Z., Chem. Commun, 2001, 18, 1740—1741 |
| [87] | Mei J., Leung N. L. C., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Chem. Rev., 2015, 115(21), 11718—11940 |
| [88] | Hong Y., Lam J. W. Y., Tang B. Z., Chem. Soc. Rev., 2011, 40(11), 5361—5388 |
| [89] | Shi H., Liu J., Geng J., Tang B. Z., Liu B., J. Am. Chem. Soc., 2012, 134(23), 9569—9572 |
| [90] | Wang D., Lee M. M. S., Shan G., Kwok R. T. K., Lam J. W. Y., Su H., Cai Y., Tang B. Z., Adv. Mater., 2018, 30(39), 1802105 |
| [91] | Wang D., Su H. F., Kwok R. T. K., Hu X. L., Zou H., Luo Q. X., Lee M. S., Xu W. H., Lam J. W. Y., Tang B. Z., Chem. Sci., 2018, 9, 3685—3693 |
| [92] | Zhang J. J., Tian H., Adv. Opt. Mater., 2018, 6(6), 1701278 |
| [93] | Hong Y., Lam J. W. Y., Tang B. Z., Chem. Commun., 2009, 29, 4332—4353 |
| [94] | Yang H., Li M. Q., Li C., Luo Q. F., Zhu M. Q., Tian H., Zhu W. H., Angew. Chem. Int. Ed., 2020, 59(22), 8560—8570 |
| [95] | Yang J. F., Wei H. R., Ou Q., Li Q. S., Peng Q., Zheng X. Y., J. Phys. Chem. A, 2024, 128, 217—224 |
| [96] | Wei H. R., Zhu H. Y., Li Q. S., Zheng X. Y., J. Mater. Chem. C, 2024, 12(7), 2613—2622 |
| [97] | Xiong Z., Zhang X., Liu L., Zhu Q., Wang Z., Feng H., Qian Z., Chem. Sci., 2021, 12(32), 10710—10723 |
| [1] | 朱高桦, 舒菊, 耿江涛, 马夫龙, 熊玲红, 何学文. 原位激活型近红外二区聚集诱导发光探针应用于肿瘤中过氧化氢的高灵敏成像[J]. 高等学校化学学报, 2026, 47(5): 232. |
| [2] | 潘晶珂, 齐馨, 张露露, 王贝贝, 吕超. 主客体作用力构建的聚集诱导发光型超分子聚合物的合成与应用研究[J]. 高等学校化学学报, 2026, 47(5): 209. |
| [3] | 尹诗琪, 郑志刚, 何心桐, 王世敏, 顾星桂, 王二静. 从原生到功能: AIE天然产物的发光、 改性及应用[J]. 高等学校化学学报, 2026, 47(5): 75. |
| [4] | 曾宪平, 秦毅, 王东. 聚集诱导发光探针用于手术导航[J]. 高等学校化学学报, 2026, 47(5): 54. |
| [5] | 翟哲, 刘雷静, 田文晶. 聚集诱导发光分子探针的设计及在细胞器成像中的应用[J]. 高等学校化学学报, 2026, 47(5): 36. |
| [6] | 吴锐, 李政, 李琪, 石佳俊, 赵艳, 冯维旭, 颜红侠. 含Si, P, B的非传统超支化发光聚合物研究进展[J]. 高等学校化学学报, 2026, 47(5): 179. |
| [7] | 李伟健, 徐小琴, 王威, 杨海波. 聚集诱导发光树枝状分子的研究进展[J]. 高等学校化学学报, 2026, 47(5): 136. |
| [8] | 张凯为, 秦安军. 聚集诱导发光镧系配合物[J]. 高等学校化学学报, 2026, 47(5): 95. |
| [9] | 李雨婷, 罗亮. 拉曼成像技术的前沿进展及与聚集体科学的交叉研究[J]. 高等学校化学学报, 2026, 47(4): 20260008. |
| [10] | 方瑾钰, 黄瀚玮, 宋航, 吴谦, 赵征, 唐本忠. 智能响应, 精准点亮: 酶响应的聚集诱导发光材料在生物医学中的诊疗新策略[J]. 高等学校化学学报, 2026, 47(4): 20260009. |
| [11] | 孙妍, 朱东霞. 近红外激发有机发光材料的构筑及在疾病治疗中的应用[J]. 高等学校化学学报, 2026, 47(4): 20260002. |
| [12] | 杨湛, 邓皇俊, 池振国. 具有聚集诱导发光性质的氢键有机框架的研究进展[J]. 高等学校化学学报, 2026, 47(4): 20260012. |
| [13] | 李银, 汤睿霖, 瞿超, 程亮慧, 胡玉玺, 吴钰祥, 王志明. 离子化策略实现马来酸酐水溶性光敏探针的制备及其在高效抗菌中的应用[J]. 高等学校化学学报, 2026, 47(4): 20250413. |
| [14] | 任奥成, 李青云, 吉晓帆. 荧光超分子聚合物网络[J]. 高等学校化学学报, 2026, 47(4): 20250390. |
| [15] | 程建硕, 叶文彦, 周璐璐, 刘谋为, 李忠宇, 唐子然, 俞婉婷, 朱亮亮. 光激发诱导的生物大分子自组装[J]. 高等学校化学学报, 2026, 47(4): 20250398. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||