

高等学校化学学报 ›› 2026, Vol. 47 ›› Issue (5): 20260003.doi: 10.7503/cjcu20260003
收稿日期:2026-01-01
出版日期:2026-05-10
发布日期:2026-02-05
通讯作者:
杨海波
E-mail:hbyang@chem.ecnu.edu.cn
基金资助:
LI Weijian, XU Xiaoqin, WANG Wei, YANG Haibo( )
)
Received:2026-01-01
Online:2026-05-10
Published:2026-02-05
Contact:
YANG Haibo
E-mail:hbyang@chem.ecnu.edu.cn
Supported by:摘要:
传统有机发光材料在聚集态下常因π-π堆积导致荧光猝灭, 严重制约了其固态应用性能. 聚集诱导发光现象(AIE)的发现为解决这一难题提供了革命性思路, 但聚集诱导发光小分子仍存在功能单一、 结构调控性有限等问题. 因此, 将聚集诱导发光单元与具有精确三维拓扑结构的树枝状分子相结合, 发展聚集诱导发光树枝状分子, 成为实现高效固态发光、 多功能集成及智能响应的重要策略. 本文综合评述了该领域的最新进展, 重点聚焦以四苯基乙烯和9,10-二苯乙烯基蒽为核心代表的两类体系. 通过分析其可控合成策略、 多尺度结构表征方法及构效关系, 揭示了树枝状分子骨架的精确结构如何通过限制分子内运动显著提升荧光量子产率、 调控发光颜色, 并赋予材料优异的聚集诱导发光性能. 评述了引入轮烷单元实现的动态响应特性, 及其在人工光捕获系统、 圆偏振发光和智能信息加密等方面的创新应用. 聚集诱导发光树枝状分子通过结构精确设计, 不仅能有效克服聚集导致的荧光猝灭效应, 更能实现发光性能的调控与多重功能的协同集成. 最后, 探讨了该领域在精准合成、 动态行为机理解析及生物医学应用转化等方面面临的挑战, 展望了未来发展趋势, 为新一代高性能、 智能化发光材料的设计与开发提供了重要参考.
中图分类号:
TrendMD:
李伟健, 徐小琴, 王威, 杨海波. 聚集诱导发光树枝状分子的研究进展. 高等学校化学学报, 2026, 47(5): 20260003.
LI Weijian, XU Xiaoqin, WANG Wei, YANG Haibo. Recent Advances in AIE-active Dendrimers. Chem. J. Chinese Universities, 2026, 47(5): 20260003.
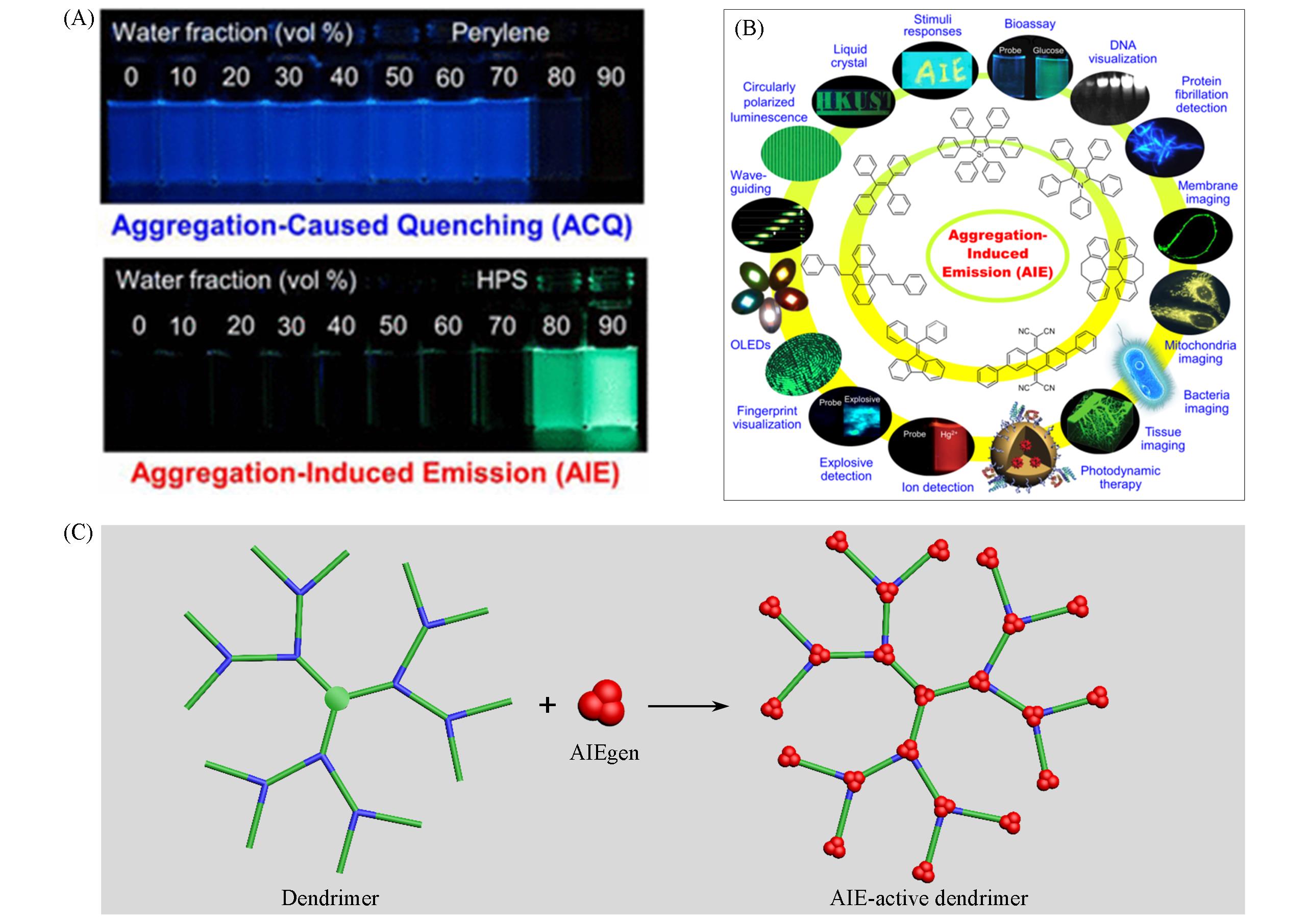
Fig.1 Fluorescence photographs of ACQ molecules and AIEgens(A), typical AIEgens and their diverse applications(B) [5], and cartoon representation of AIE⁃active dendrimers(C)(A, B) Copyright 2015, American Chemical Society.
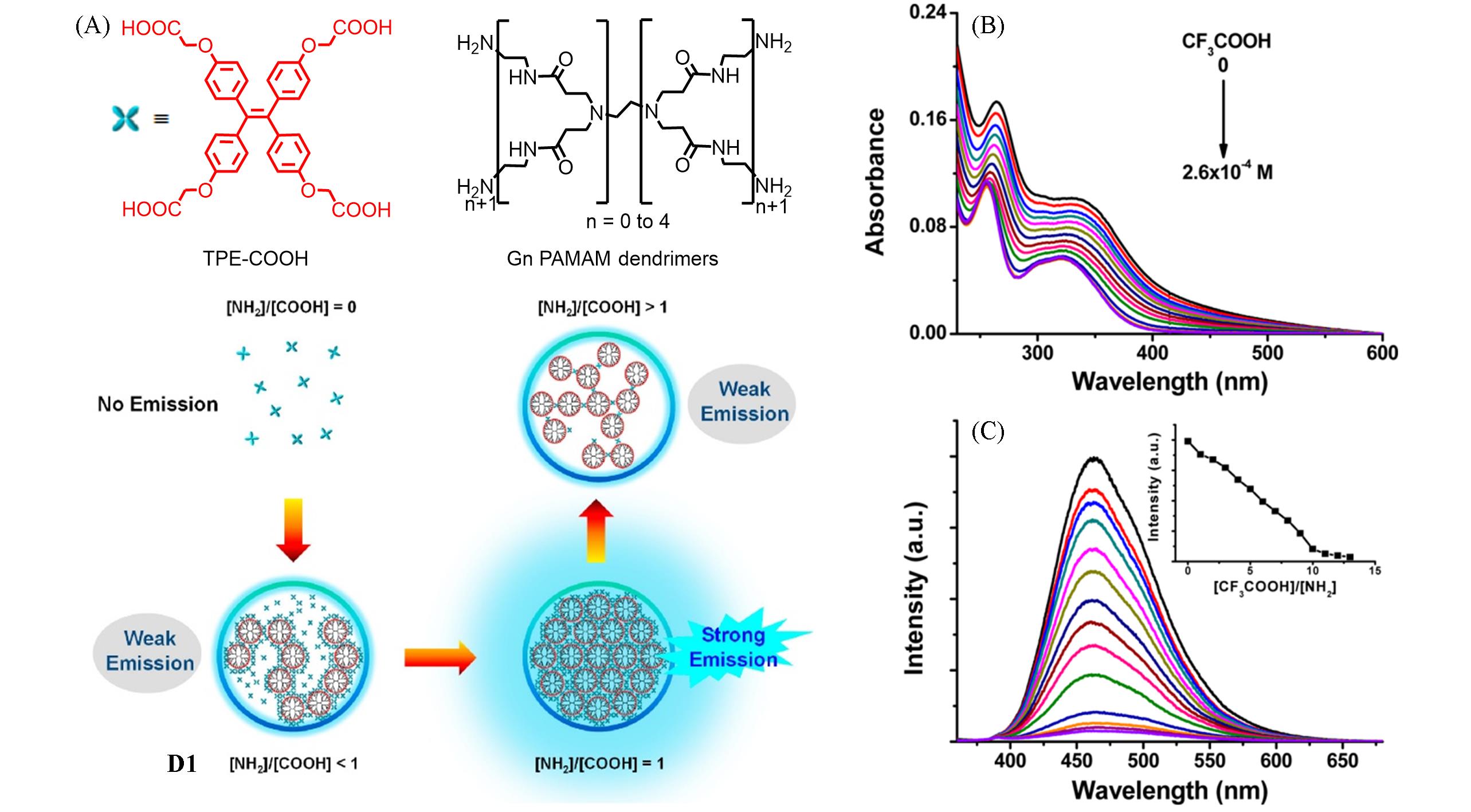
Fig.2 Schematic illustration for the formation of the AIE⁃active dendrimer D1 via self⁃assembly of the G4⁃PAMAM dendrimer and TPE⁃COOH AIEgen(A), absorption(B) and fluorescence(C) spectra of D1 in methanol dispersion during addition of CF3COOH[38]Inset in (C): plot of the fluorescence intensity at 464 nm versus [CF3COOH]/[NH2]. Copyright 2015, American Chemical Society.
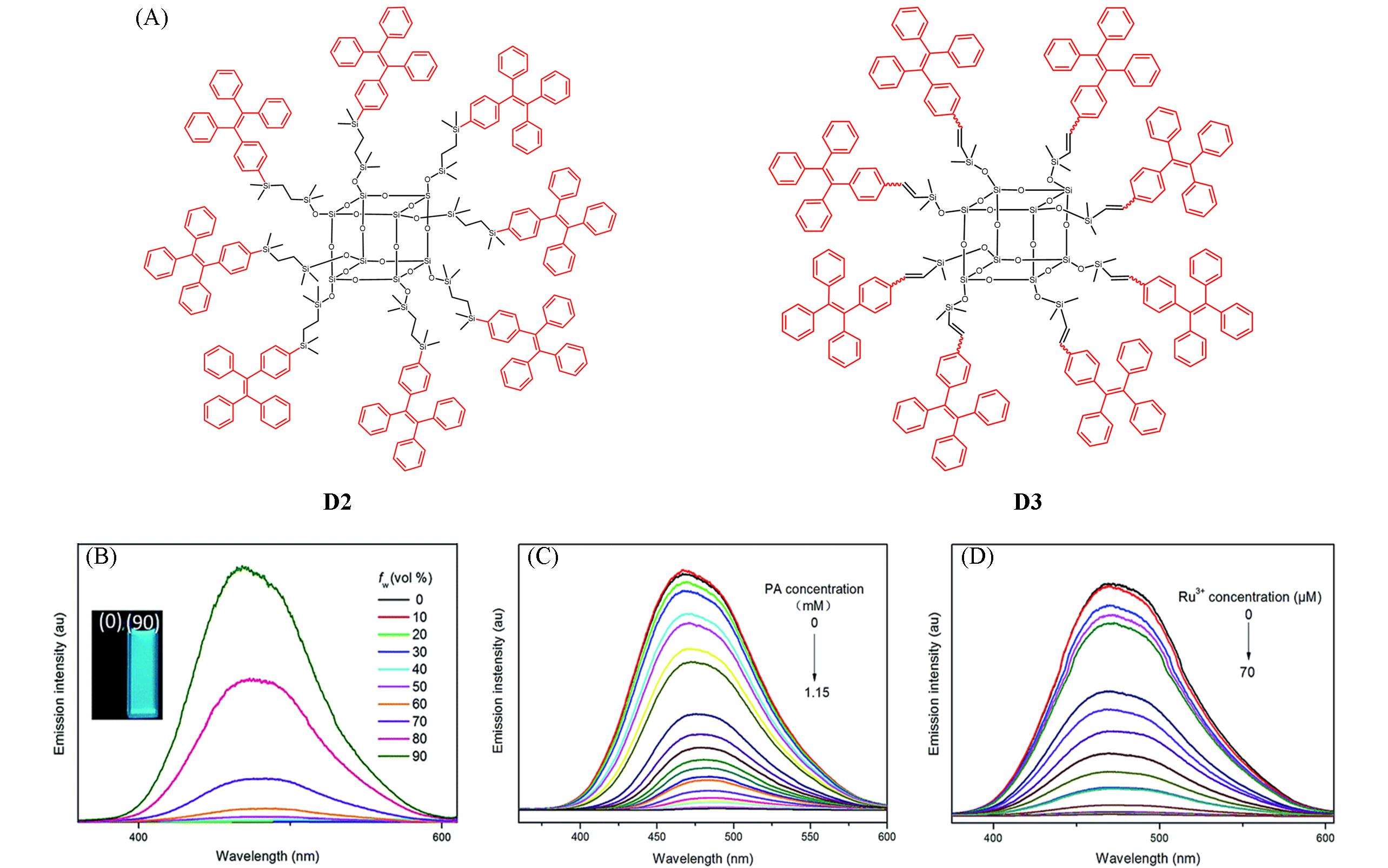
Fig.3 Chemical structures of AIE⁃active dendrimers D2 and D3(A), fluorescence spectra of D2 in THF⁃water mixtures(B) and fluorescence spectra of D2 in THF⁃water mixtures with 90% water content containing different amounts of PA(C) and Ru3+ ions(D)[39]Inset of (B): photographs of D2 in THF and THF-water(volume ratio, 1∶9) mixture taken under illumination of a handheld UV lamp.Copyright 2015, the Royal Society of Chemistry.
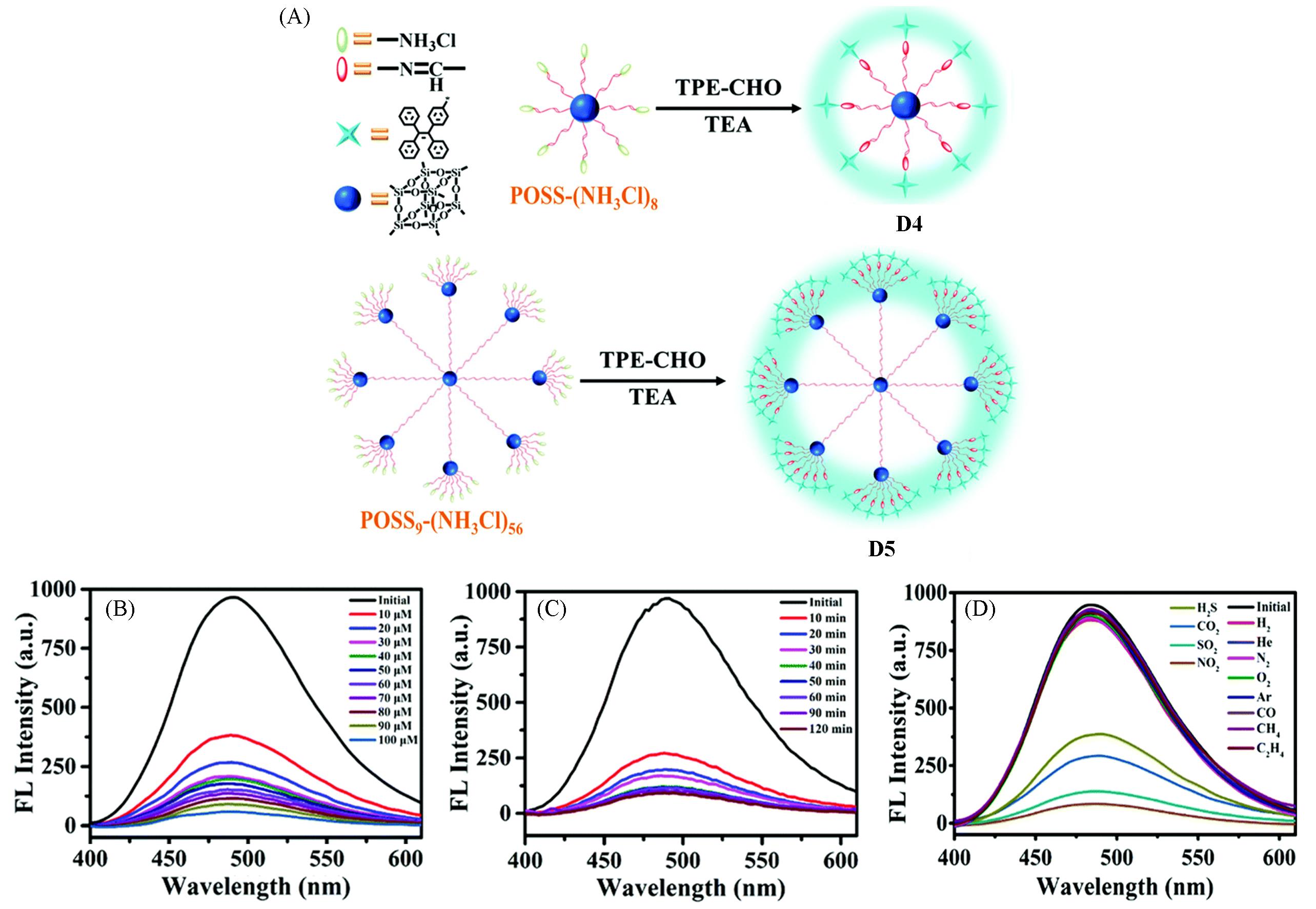
Fig.4 Synthetic routes of AIE⁃active dendrimers D4 and D5(A), fluorescence spectra of D5 in THF/water mixtures with different concentrations of Ru3+(B), D5 in response to different times in the presence of Ru3+(C) and D5 in THF/water mixtures in response to different gases(D)[40]Copyright 2016, the Royal Society of Chemistry.
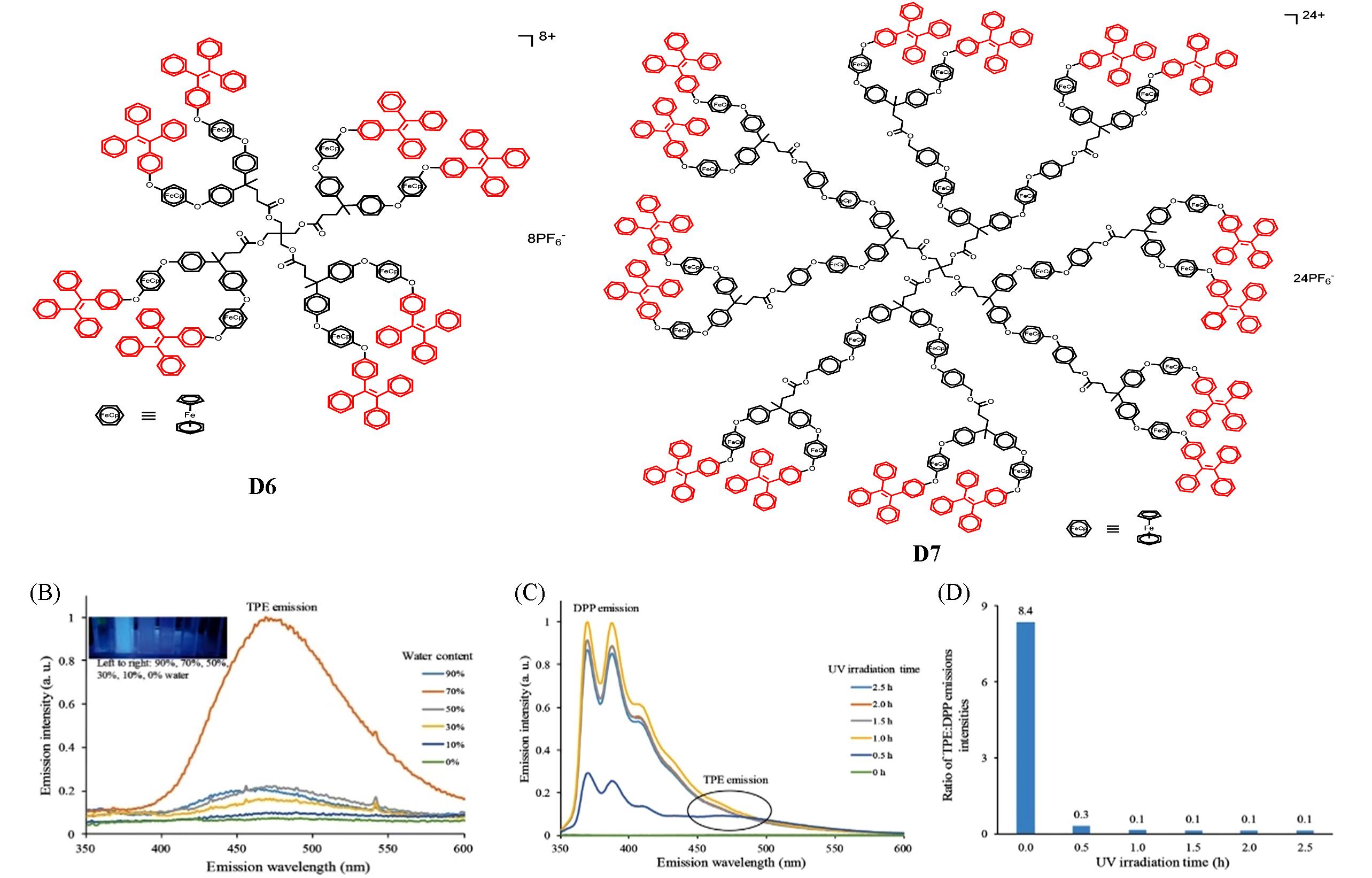
Fig.5 Chemical structures of AIE⁃active dendrimers D6 and D7(A), fluorescence spectra of D6 in THF/H2O mixtures(B), fluorescence spectra of D6(C), and TPE∶DPP emissions intensities tuned by photochemical reaction time for 0, 0.5, 1.0, 1.5, 2.0, 2.5 h in THF/H2O(volume ratio 3∶7)(D)[41]Copyright 2016, Wiley⁃VCH.
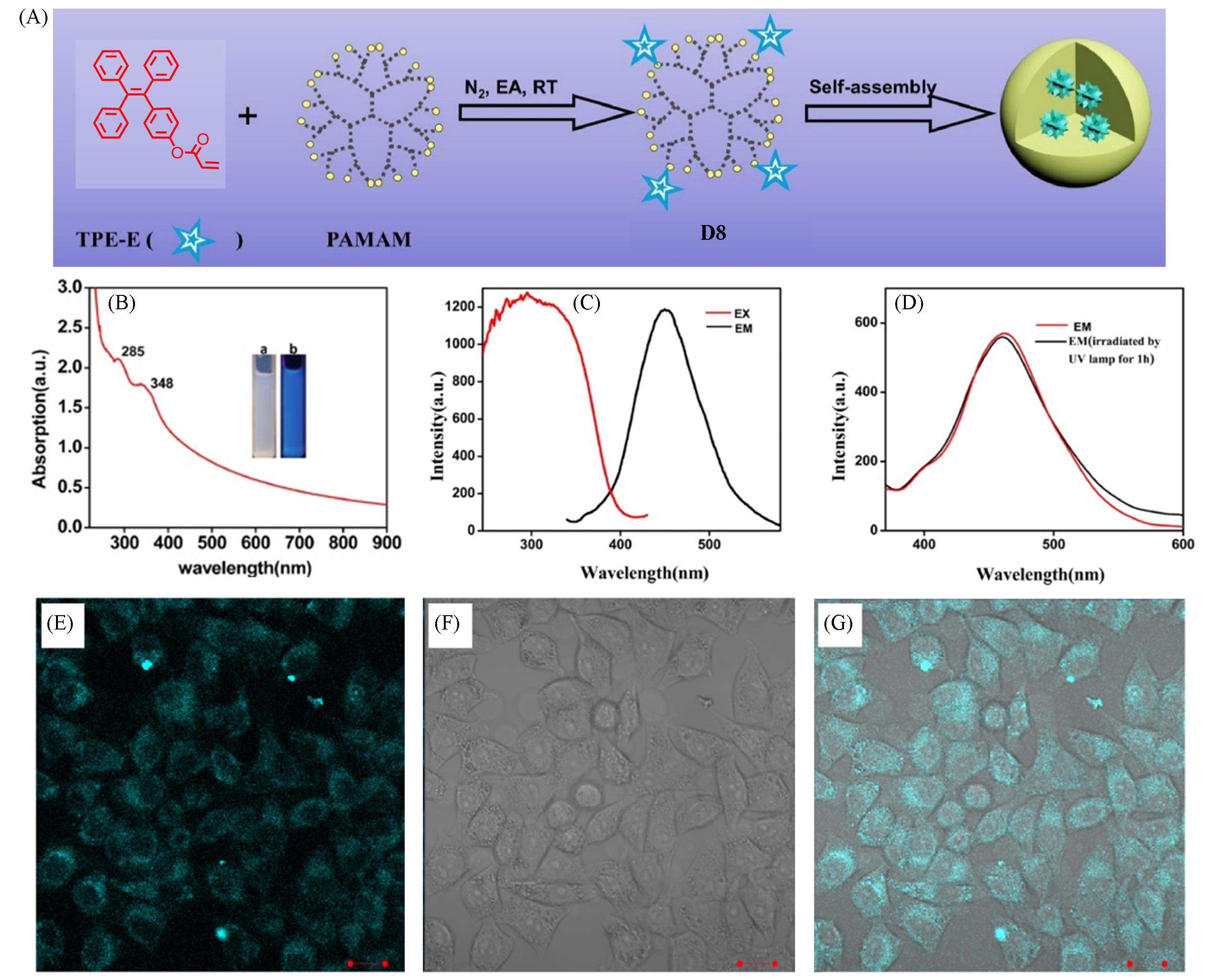
Fig.6 Fabrication and biological applications of AIE⁃active dendrimers[42](A) Synthesis and self-assembly of AIE-active dendrimers D8; (B) UV-Vis spectrum of D8; (C) fluorescence excitation(EX) and emission(EM) spectra of D8; (D) EM spectra of D8 after irradiated by UV lamp for 1 h; (E—G) CLSM images of D8 nanoparticles using a 405 nm laser: (E) fluorescent image, (F) bright fields, (G) merged image. Inset of (B): photographs of D8 in water under natural light(left) and UV light at 365 nm(right).Copyright 2017, Elsevier.
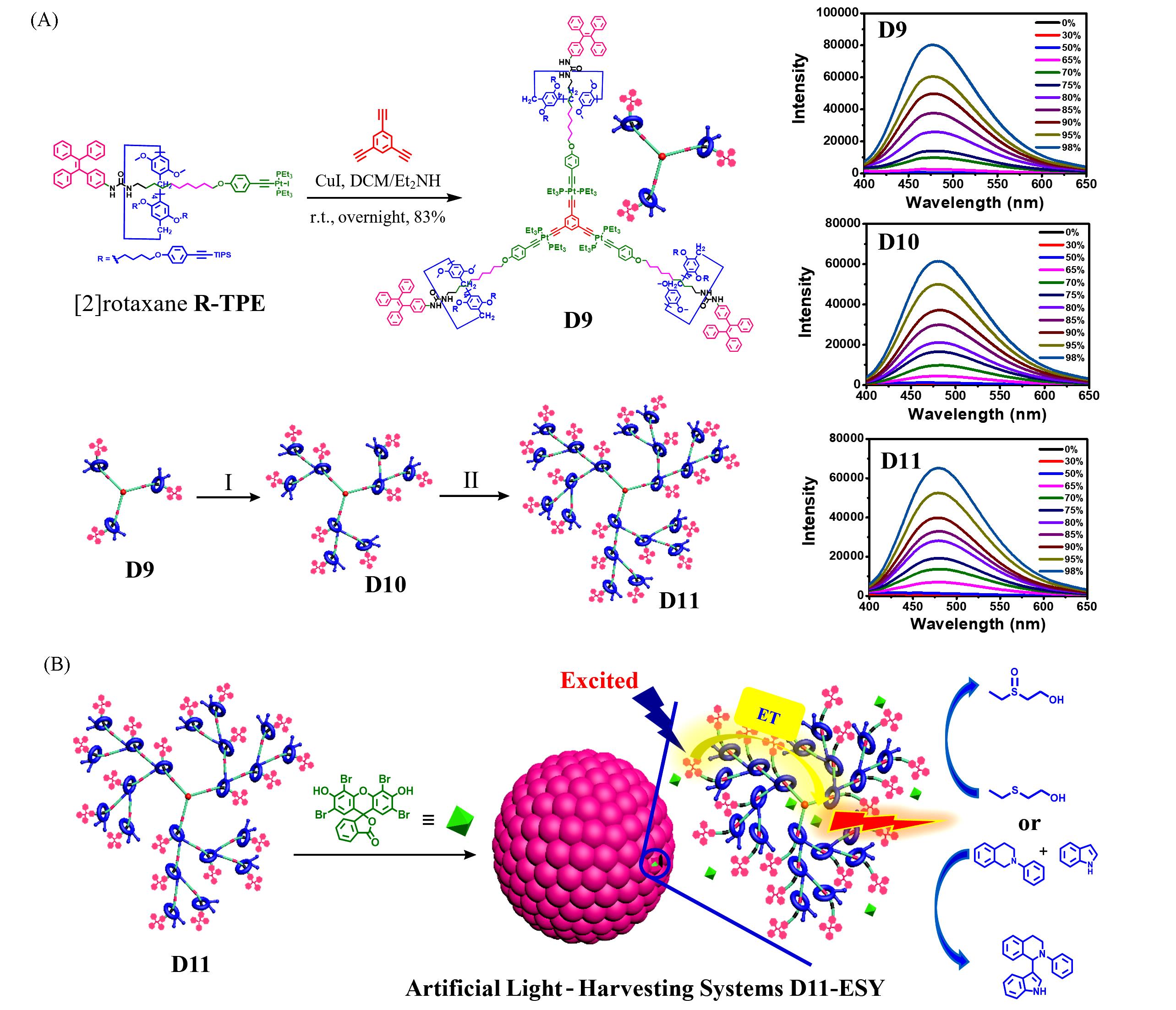
Fig.7 Synthesis of the AIE⁃active rotaxane dendrimers D9—D11 through the controllable divergent approach from [2]rotaxane R⁃TPE(left) and fluorescence spectra of in DCM/MeOH with various MeOH fractions(right)(A) and the construction of artificial light harvesting systems based on AIE⁃active rotaxane dendrimers D11 and ESY as efficient photocatalysts(B)[49]
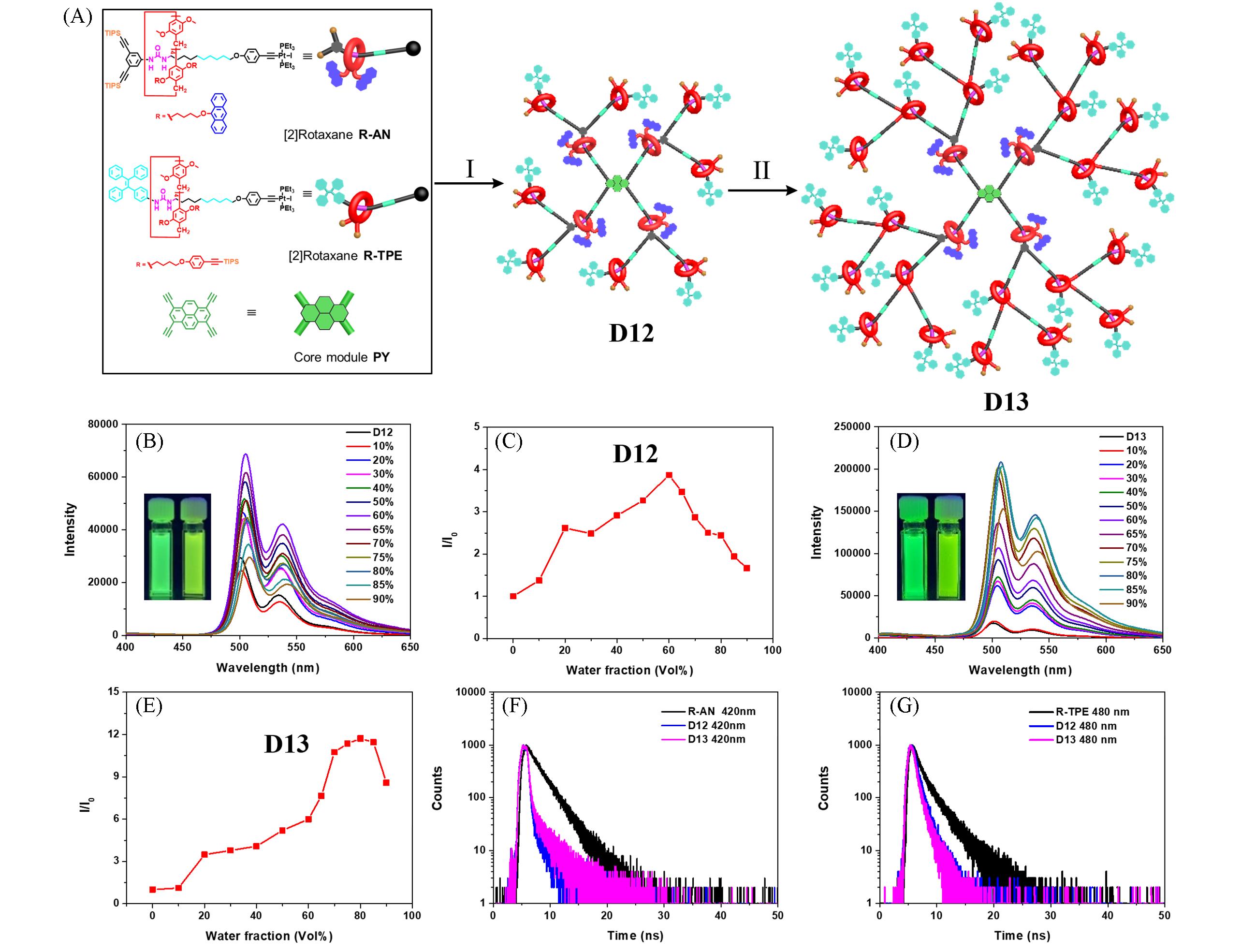
Fig.8 Cartoon representation of AIE⁃active rotaxane dendrimers D12 and D13 for efficient light harvesting(A), fluorescence spectra of D12(B) and D13(D) in THF/H2O with various H2O fractions, plot of the relative emission intensity(I/I0) of D12(C) and D13(E) versus the composition of the THF/water mixture, and time⁃resolved fluorescence decay curves of D12(F) and D13(G) and [2]rotaxane monomers(375 nm excitation and 420, 480 nm detection, respectively)[50]Reaction conditions (I): (a) R⁃AN, PY, CuI, DCM/Et2NH, r.t., overnight, 81%; (b) Bu4NF⋅3H2O, THF, r.t., 2 h, 87%; (c) R⁃TPE, CuI, DCM/Et2NH, r.t., overnight, 70%. Reaction conditions (II): (a) Bu4NF⋅3H2O, THF, r.t., 2 h, 70%; (b) R⁃TPE, CuI, DCM/Et2NH, r.t., overnight, 71%.Copyright 2022, Elsevier.
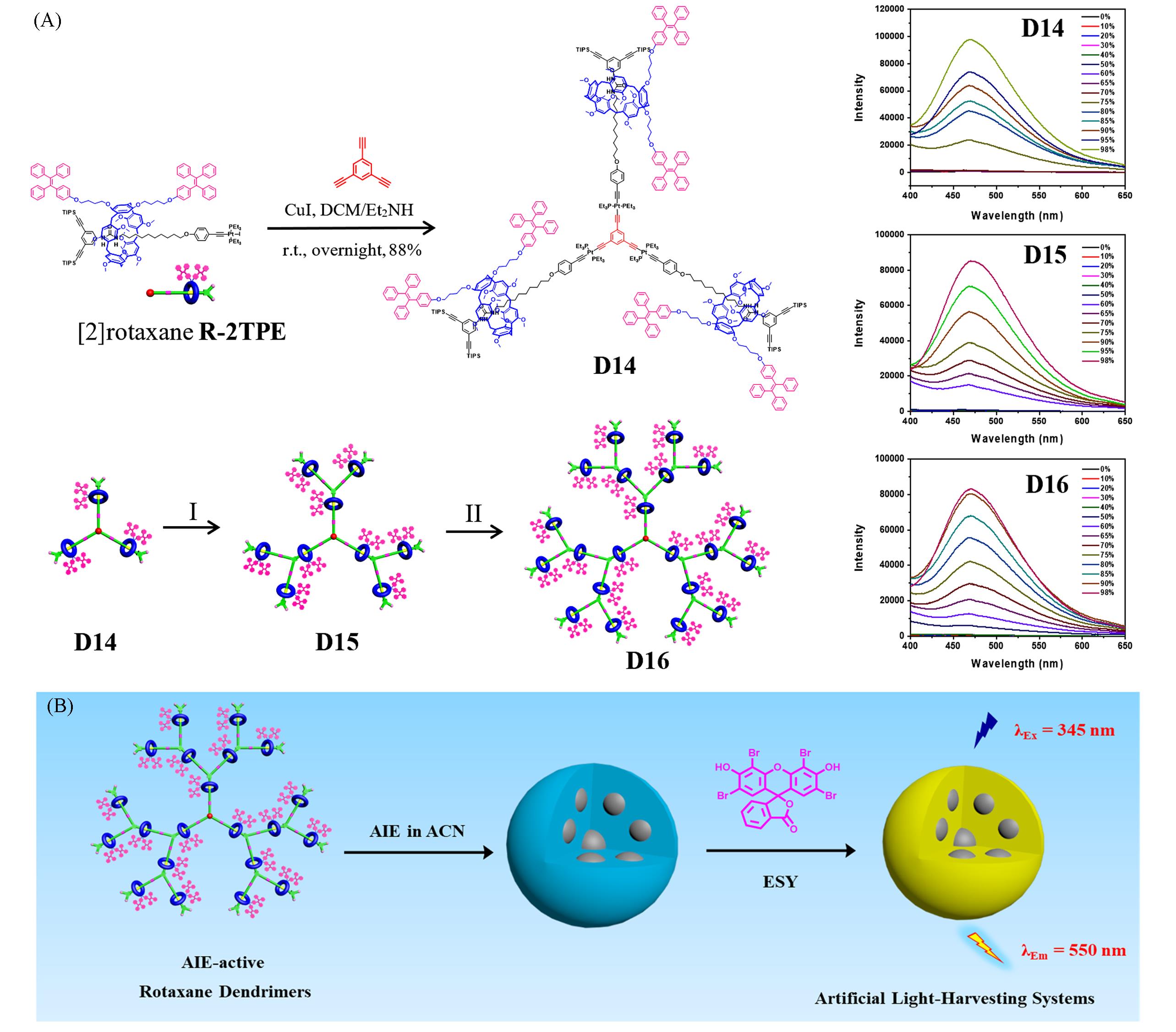
Fig.9 Synthesis of the AIE⁃active rotaxane dendrimers D14—D16 through the controllable divergent approach from [2]rotaxane R⁃2TPE(left) and fluorescence spectra in DCM/ACN with various ACN fractions(right)(A) and the construction of artificial light harvesting systems based on AIE⁃active rotaxane dendrimers and ESY(B)[51]Reaction conditions (I): (a) Bu4NF⋅3H2O, THF, r.t., 2 h; (b) R⁃2TPE, CuI, DCM/Et2NH, r.t., overnight, 72%. Reaction conditions (II): (a) Bu4NF⋅3H2O, THF, r.t., 2 h; (b) R⁃2TPE, CuI, DCM/Et2NH, r.t., overnight, 78%.Copyright 2025, the Royal Society of Chemistry.
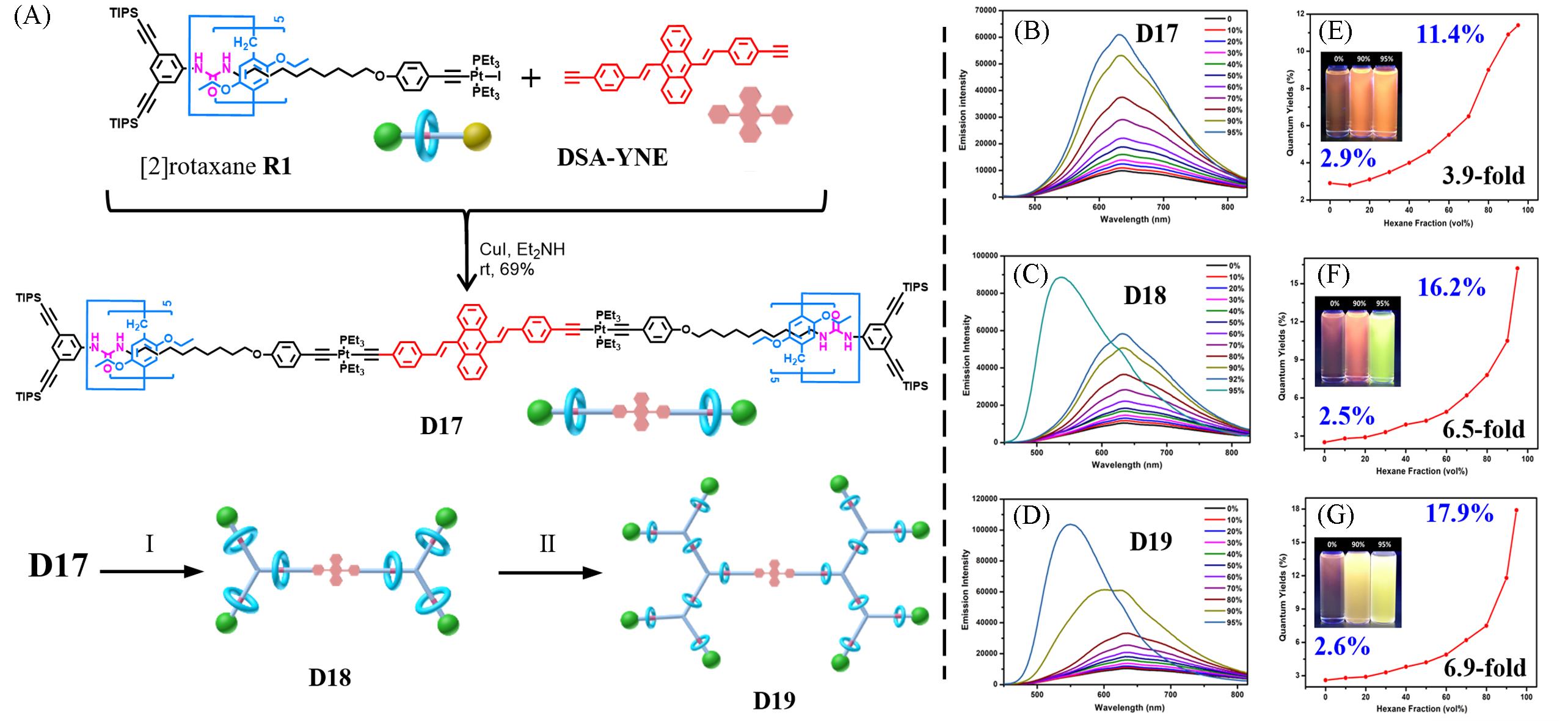
Fig.10 Synthesis of the AIE⁃active rotaxane dendrimers D17—D19 through the controllable divergent approach(A), fluorescence spectra(B, D, F) and fluorescence quantum yields(C, E, G) of D17(B, C), D18(D, E), and D19(G, F) in DCM/n‑hexane with different n‑hexane fractions[58]The insets present photographs of the corresponding solutions(from left to right: 0, 90%, and 95% n‑hexane) irradiated by a UV lamp. Reaction conditions (I): (a) Bu4NF⋅3H2O, THF, r.t., 2 h; (b) R1, CuI, Et2NH, r.t., 8 h, 54%. Reaction conditions (II): (a) Bu4NF⋅3H2O, THF, r.t., 2 h; (b) R1, CuI, Et2NH, r.t., 8 h, 37%.Copyright 2019, the Royal Society of Chemistry.
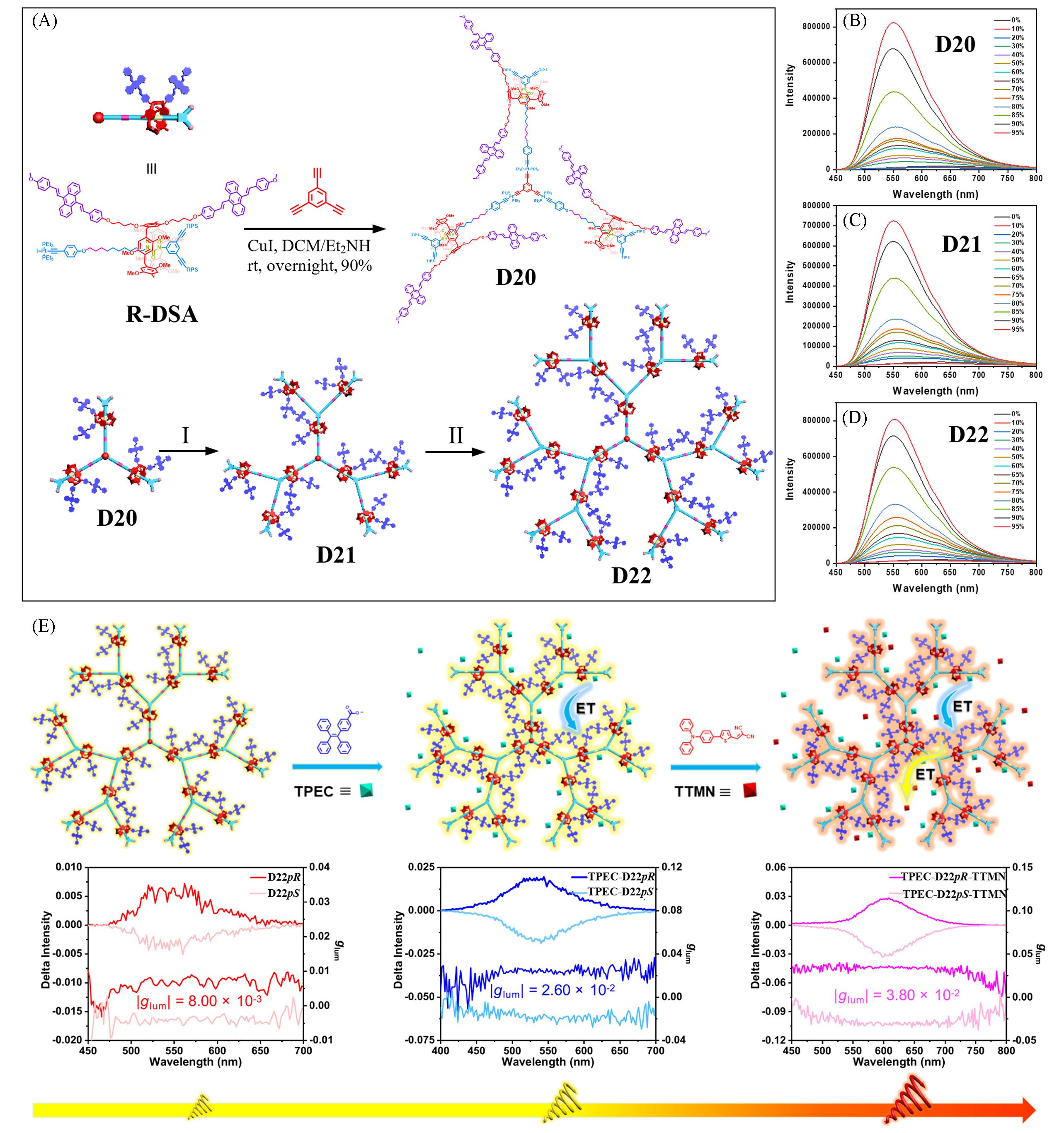
Fig.11 Synthesis of the AIE⁃active rotaxane dendrimers D20—D22 through the controllable divergent approach(A), fluorescence spectra of D20(B), D21(C), and D22(D) in THF/H2O mixture with various H2O fractions, and boosting the CPL performances of chiral artificial light harvesting systems based on chiral AIE⁃active rotaxane dendrimers along with the two⁃step FRET process(E)[59]Reaction conditions (I): (a) Bu4NF⋅3H2O, THF, r.t., 2 h; (b) R⁃DSA, CuI, DCM/Et2NH, r.t., overnight, 80%. Reaction conditions (II): (a) Bu4NF⋅3H2O, THF, r.t., 2 h; (b) R⁃DSA, CuI, DCM/ Et2NH, r.t., overnight, 82%.Copyright 2025, Wiley-VCH.
| [1] | Luo J., Xie Z., Lam J. W. Y., Cheng L., Chen H., Qiu C., Kwok H. S., Zhan X., Liu Y., Zhu D., Tang B. Z., Chem. Commun., 2001, 1740—1741 |
| [2] | Lam J. W. Y., Tang B. Z., Chem. Soc. Rev., 2011, 40, 5361—5388 |
| [3] | Yang S. Y., Chen Y., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Chem. Soc. Rev., 2024, 53, 5366—5393 |
| [4] | Zhu F. Y., Mei L. J., Tian R., Li C., Wang Y. L., Xiang S. L., Zhu M. Q., Tang B. Z., Chem. Soc. Rev., 2024, 53, 3350—3383 |
| [5] | Leung N. L. C., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Chem. Rev., 2015, 115, 11718—11940 |
| [6] | Duo Y., Han L., Yang Y., Wang Z., Wang L., Chen J., Xiang Z., Yoon J., Luo G., Tang B. Z., Chem. Rev., 2024, 124, 11242—11347 |
| [7] | Ding D., Li K., Liu B., Tang B. Z., Acc. Chem. Res., 2013, 46, 2441—2453 |
| [8] | Wang B., Liu Y., Chen X., Liu X. T., Liu Z., Lu C., Chem. Soc. Rev., 2024, 53, 10189—10215 |
| [9] | Feng H. T., Yuan Y. X., Xiong J. B., Zheng Y. S., Tang B. Z., Chem. Soc. Rev., 2018, 47, 7452—7476 |
| [10] | Wu W., Tang R., Li Q., Li Z., Chem. Soc. Rev., 2015, 44, 3997—4022 |
| [11] | Wang Y., Nie J., Fang W., Yang L., Hu Q., Wang Z., Sun J. Z., Tang B. Z., Chem. Rev., 2020, 120, 4534—4577 |
| [12] | Feng G., Liu B., Acc. Chem. Res., 2018, 51, 1404—1414 |
| [13] | Shi Y., Yin G., Yan Z., Sang P., Wang M., Brzozowski R., Eswara P., Wojtas L., Zheng Y., Li X., Cai J., J. Am. Chem. Soc., 2019, 141, 12697—12706 |
| [14] | Liu D., Chen M., Li K., Li Z., Huang J., Wang J., Jiang Z., Zhang Z., Xie T., Newkome G. R., Wang P., J. Am. Chem. Soc., 2020, 142, 7987—7994 |
| [15] | Li Y., Dong Y., Cheng L., Qin C., Nian H., Zhang H., Yu Y., Cao L., J. Am. Chem. Soc., 2019, 141, 8412—8415 |
| [16] | Liu N., Lin T., Wu M., Luo H. K., Huang S. L., Hor T. S. A., J. Am. Chem. Soc., 2019, 141, 24, 9448—9452 |
| [17] | Shi Y. E, Ma J., Feng A., Wang Z., Rogach A. L., Aggregate, 2021, 2, e112 |
| [18] | Xu H., Cheng P., Aggregate, 2024, 5, e518 |
| [19] | He Y. M., Feng Y., Fan Q. H., Acc. Chem. Res., 2014, 47, 2894—2906 |
| [20] | Wu P., Feldman A. K., Nugent A. K., Hawker C. J., Scheel A., Voit B., Pyun J., Fréchet J. M. J., Sharpless K.B., Fokin V. V., Angew. Chem. Int. Ed., 2004, 43, 3928—3932 |
| [21] | Fischer M., Vögtle F., Angew. Chem. Int. Ed., 1999, 38, 884—905 |
| [22] | Liu X., Zeng Y., Zhang X., Yu T., Chen J., Li Y., Sci. China Chem., 2015, 58, 390—399 |
| [23] | Liu S., Sci. China Chem., 2019, 62, 925—926 |
| [24] | Wang X., Gao P., Wang J., Yang Y., You Y., Wu D., CCS Chem., 2021, 3, 1093—1104 |
| [25] | Arrigo A., Puntoriero F., Ganga G. L., Campagna S., Burian M., Bernstorff S., Amenitsch H., Chem, 2017, 3, 494—508 |
| [26] | Jiang D. L., Aida T., Nature, 1997, 388, 454—456 |
| [27] | Percec V., Glodde M., Bera T. K., Miura Y., Shiyanovskaya I., Singer K. D., Balagurusamy V. S. K., Heiney P. A., Schnell I., Rapp A., Spiess H. W., Hudson S. D., Duan H., Nature, 2002, 419, 384—387 |
| [28] | Helms B., Meijer E. W., Science, 2006, 313, 929—930 |
| [29] | Tomalia D. A., Science, 1991, 252, 1231—1231 |
| [30] | Astruc D., Nature Chem., 2012, 4, 255—267 |
| [31] | Shiraishi K., Kashiwabara T., Sanji T., Tanaka M., New J. Chem., 2009, 33, 1680—1684 |
| [32] | Chen Q., Zhang D., Zhang G., Yang X., Feng Y., Fan Q., Zhu D., Adv. Funct. Mater., 2010, 20, 3244—3251 |
| [33] | Xu B., Zhang J., Fang H., Ma S., Chen Q., Sun H., Im C., Tian W., Polym. Chem., 2014, 5, 479 |
| [34] | Abd-El-Aziz A. S., Abdelghani A. A., Wagner B. D., Bissessur R., Macromol. Rapid Commun., 2019, 40, 1800711 |
| [35] | Leung M. K., Lin Y. S., Lee C. C., Chang C. C., Wang Y. X., Kuo C. P., Singh N., Lin K. R., Hu C. W., Tsengc C. Y., Ho K. C., RSC Adv., 2013, 3, 22219 |
| [36] | Chen Y., Lv Y., Han Y., Zhu B., Zhang F., Bo Z., Liu C. Y., Langmuir, 2009, 25, 8548—8555 |
| [37] | Huang G., Ma B., Chen J., Peng Q., Zhang G., Fan Q., Zhang D., Chem. Eur. J., 2012, 18, 3886—3892 |
| [38] | Liu X., Zeng Y., Liu J., Li P., Zhang D., Zhang X., Yu T., Chen J., Yang G., Li Y., Langmuir, 2015, 31, 4386—4393 |
| [39] | Xiang K., He L., Li Y., Xu C., Li S., RSC Adv., 2015, 5, 97224 |
| [40] | Zuo Y., Wang X., Yang Y., Huang D., Yang F., Shen H., Wu D., Polym. Chem., 2016, 7, 6432—6436 |
| [41] | Abd⁃El⁃Aziz A. S., Agatemor C., Etkin N., Wagner B., Macromol. Rapid Commun., 2016, 37, 1235—1241 |
| [42] | Lv Q., Liu M., Wang K., Mao L., Xu D., Zeng G., Liang S., Deng F., Zhang X., Wei Y., J. Taiwan Inst. Chem. E., 2017, 75, 292—298 |
| [43] | Xu X. Q., Wang X. Q., Wang W., Chinese Chem. Lett., 2023, 34, 107665 |
| [44] | Wang X. Q., Li W. J., Wang W., Yang H. B., Acc. Chem. Res., 2021, 54, 4091—4106 |
| [45] | Qu S., Deng J., Wang W., Liu Y., Yang M., Yang L., Gao W., Liu, G., Wang Y., Ding Y., Cheng L., Yu W., Zhang Z., Yan X., CCS Chem., 2025, 7, 3849—3859 |
| [46] | Wang P. L., Chen P., Yang R., Tetlow D. J., Zhang Z. H., Han J., Fielden S. D. P., Howlader P., Zhang L., Leigh D. A., Angew. Chem. Int. Ed., 2025, 64, e202513115 |
| [47] | Chen L., Sheng X., Li G., Huang F., Chem. Soc. Rev., 2022, 51, 7046—7065 |
| [48] | Zhu K., Baggi, G., Loeb S. J., Nature Chem., 2018, 10, 625—630 |
| [49] | Li W. J., Wang X. Q., Zhang D. Y., Hu Y. X., Xu W. T., Xu L., Wang W., Yang H. B., Angew. Chem. Int. Ed., 2021, 60, 18761—18768 |
| [50] | Li W. J., Jiang H., Wang X. Q., Zhang D. Y., Zhu Y., Ke Y., Wang W., Yang H. B., Mater. Today Chem., 2022, 24, 100874 |
| [51] | Xu X. Q., Song Y. R., Cao J. H., Li W. J., Zhu Y., Zhang D. Y., Wang W., Wang X. Q., Yang H. B., Chem. Sci., 2025, 16, 5786 |
| [52] | Wang Z., Xu B., Zhang L., Zhang J., Ma T., Zhang J., Fu X., Tian W., Nanoscale, 2013, 5, 2065—2072 |
| [53] | Xu B., He J., Dong Y., Chen F., Yu W., Tian W., Chem. Commun., 2011, 47, 6602—6604 |
| [54] | He J., Xu B., Chen F., Xia H., Li K., Ye L., Tian W., J. Phys. Chem. C, 2009, 113, 22, 9892—9899 |
| [55] | Chen P. Y., Zhang G. Y., Li J. G., Ma L. J., Zhou J. Y., Zhu M. G., Li S., Wang Z., Chem. Res. Chinese Universities, 2024, 40(2), 293—304 |
| [56] | Xiong J. Y., Wu M. J., Yao L. Y., Chem. Res. Chinese Universities, 2024, 40(5), 887—893 |
| [57] | Sun Z. H., Yin P. P., He S. Y., Zhang K. G., Pan X. R., Wang J. Y., Hao P. N., Zhou Z., Yang X. G., Ma L. F., Tan C. L., Chem. Res. Chinese Universities, 2025, 41(3), 519—524 |
| [58] | Wang X. Q., Wang W., Li W. J., Qin Y., Yin G. Q., Jiang W. L., Li X., Wu S., Yang H. B., Org. Chem. Front., 2019, 6, 1686—1691 |
| [59] | Xu X. Q., Li W. J., Zhang D. Y., Zhu Y., Xu W. T., Wang Y., Wang X. Q., Wang W., Yang H. B., Angew. Chem. Int. Ed., 2025, 64, e202419434 |
| [1] | 张哲浩, 宛新华, 张洁. 螺旋聚苯乙炔衍生物圆偏振发光材料的构象调控、 自组装与功能化[J]. 高等学校化学学报, 2026, 47(5): 20260064. |
| [2] | 焦海丽, 郑小燕. 限域环境诱导有机发光增强的理论研究进展[J]. 高等学校化学学报, 2026, 47(5): 20260060. |
| [3] | 曾宪平, 秦毅, 王东. 聚集诱导发光探针用于手术导航[J]. 高等学校化学学报, 2026, 47(5): 20260066. |
| [4] | 翟哲, 刘雷静, 田文晶. 聚集诱导发光分子探针的设计及在细胞器成像中的应用[J]. 高等学校化学学报, 2026, 47(5): 20260078. |
| [5] | 尹诗琪, 郑志刚, 何心桐, 王世敏, 顾星桂, 王二静. 从原生到功能: AIE天然产物的发光、 改性及应用[J]. 高等学校化学学报, 2026, 47(5): 20260042. |
| [6] | 朱高桦, 舒菊, 耿江涛, 马夫龙, 熊玲红, 何学文. 原位激活型近红外二区聚集诱导发光探针应用于肿瘤中过氧化氢的高灵敏成像[J]. 高等学校化学学报, 2026, 47(5): 20260058. |
| [7] | 潘晶珂, 齐馨, 张露露, 王贝贝, 吕超. 主客体作用力构建的聚集诱导发光型超分子聚合物的合成与应用研究[J]. 高等学校化学学报, 2026, 47(5): 20260130. |
| [8] | 吴锐, 李政, 李琪, 石佳俊, 赵艳, 冯维旭, 颜红侠. 含Si, P, B的非传统超支化发光聚合物研究进展[J]. 高等学校化学学报, 2026, 47(5): 20250411. |
| [9] | 张凯为, 秦安军. 聚集诱导发光镧系配合物[J]. 高等学校化学学报, 2026, 47(5): 20260013. |
| [10] | 武宇乾, 夏阳, 李琬卉, 王小野. 基于非手性聚合物的共组装圆偏振发光材料[J]. 高等学校化学学报, 2026, 47(5): 20260006. |
| [11] | 李雨婷, 罗亮. 拉曼成像技术的前沿进展及与聚集体科学的交叉研究[J]. 高等学校化学学报, 2026, 47(4): 20260008. |
| [12] | 方瑾钰, 黄瀚玮, 宋航, 吴谦, 赵征, 唐本忠. 智能响应, 精准点亮: 酶响应的聚集诱导发光材料在生物医学中的诊疗新策略[J]. 高等学校化学学报, 2026, 47(4): 20260009. |
| [13] | 孙妍, 朱东霞. 近红外激发有机发光材料的构筑及在疾病治疗中的应用[J]. 高等学校化学学报, 2026, 47(4): 20260002. |
| [14] | 杨湛, 邓皇俊, 池振国. 具有聚集诱导发光性质的氢键有机框架的研究进展[J]. 高等学校化学学报, 2026, 47(4): 20260012. |
| [15] | 李银, 汤睿霖, 瞿超, 程亮慧, 胡玉玺, 吴钰祥, 王志明. 离子化策略实现马来酸酐水溶性光敏探针的制备及其在高效抗菌中的应用[J]. 高等学校化学学报, 2026, 47(4): 20250413. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||