

高等学校化学学报 ›› 2023, Vol. 44 ›› Issue (1): 20220619.doi: 10.7503/cjcu20220619
吴钰洁1, 黄文治1, 潘俊达1, 石凯祥1,2, 刘全兵1,2( )
)
收稿日期:2022-09-15
出版日期:2023-01-10
发布日期:2022-10-25
通讯作者:
刘全兵
E-mail:liuqb@gdut.edu.cn
基金资助:
WU Yujie1, HUANG Wenzhi1, PAN Junda1, SHI Kaixiang1,2, LIU Quanbing1,2( )
)
Received:2022-09-15
Online:2023-01-10
Published:2022-10-25
Contact:
LIU Quanbing
E-mail:liuqb@gdut.edu.cn
Supported by:摘要:
锂硫电池具有理论能量密度高等优势, 被认为是最有前景的一类新型二次电池. 硫正极存在硫和硫化锂的导电性差、 可溶性多硫化物的扩散/穿梭、 循环过程中硫的体积膨胀以及氧化还原过程慢等问题, 严重制约着电池的活性和循环稳定性. 设计“蛋黄-蛋壳”结构纳米反应器应用于锂硫电池正极, 可通过调控其“蛋黄”、 “蛋壳”和“空腔”结构缓解充放电过程中电极的体积变化, 为离子/电子输运提供快速通道, 强化对多硫化物的吸附和催化转换作用等, 进而提高电极的活性和循环性能, 有利于推进锂硫电池的商业化进程. 本文总结了“蛋黄-蛋壳”结构纳米反应器的设计和调控策略, 包括单核-单壳、 单核-多壳、 多核-单壳以及多核-多壳等, 并结合锂硫电池的工作特点和目前应用存在的问题, 对未来发展前景进行了展望.
中图分类号:
TrendMD:
吴钰洁, 黄文治, 潘俊达, 石凯祥, 刘全兵. “蛋黄-蛋壳”结构纳米反应器的设计、 调控及在锂硫电池正极中的应用研究. 高等学校化学学报, 2023, 44(1): 20220619.
WU Yujie, HUANG Wenzhi, PAN Junda, SHI Kaixiang, LIU Quanbing. Design, Regulation and Applications in Lithium-sulfur Battery Cathodes of Yolk-shell Nanoreactors. Chem. J. Chinese Universities, 2023, 44(1): 20220619.
| Classification | Material | Morphology | Synthesis method | Ref. |
|---|---|---|---|---|
| Single⁃yolk⁃single⁃shell | FeP | Nanosphere⁃in⁃nanotube | Hard template | [ |
| SiO x /C@Void@C | Nanosphere | Hard template | [ | |
| Sb2Se3@void@C | Nanorod | Hard template | [ | |
| Co3O4/C@SiO2 | Rhombic dodecahedron | Hard template | [ | |
| Co3O4@TiO2@C | Sphere | Hard template | [ | |
| Si⁃infilled capsule | Capsule | Soft template | [ | |
| Si@void@C | — | Soft template | [ | |
| Classification | Material | Morphology | Synthesis method | Ref. |
| Single⁃yolk⁃single⁃shell | Cu⁃FeP@C | Sphere | Template method | [ |
| ZnS@C | Nanorod | Self⁃sacrifice template strategy | [ | |
| NiSe2@C | Sphere | Ostwald ripening | [ | |
| CoOHCl@C | — | Ostwald ripening | [ | |
| NiCo2O4/NiO | Sphere | Ostwald ripening | [ | |
| MoSe2 | Sphere | Ostwald ripening | [ | |
| NCS/CNT | Nanosphere | Kirkendall effect | [ | |
| MnO x /C | Nanoparticle | Ostwald ripening, Kirkendall effect | [ | |
| Sn⁃Sn2Co3@CoSnO3⁃Co3O4 | Microsphere | Spray drying | [ | |
| Si@void@C/CNTs | Microsphere | Spray drying | [ | |
| Sb@C | Microsphere | Spray drying | [ | |
| Co3O4@NiCo2O4 | Rhombic dodecahedron | Seed epitaxial growth | [ | |
| Fe2N@TiO2@C | Submicrocube | Hard template | [ | |
| Single⁃yolk⁃muti⁃shell | Co3O4 | Cube | Hydrothermal method | [ |
| Co3O4 | Sphere | Hydrothermal method | [ | |
| YDS⁃FCCNs | Nanospheres | Solvothermal method | [ | |
| V2O3/C | Sphere | Sequential self⁃template | [ | |
| Fe3O4⁃GC | Microsphere | Kirkendall diffusion | [ | |
| NiO | — | Sequential templating approach | [ | |
| Cobalt sulfide MSNBs | Nanobox | Ion⁃conversion⁃exchange | [ | |
| m⁃Y⁃S Fe1-x S@C⁃3 | Hexagonal nanosheet | Hard template | [ | |
| Muti⁃yolk⁃single⁃shell | SnO2/Mn2SnO4@C | Nanobox | Hard template | [ |
| Co⁃NC@N⁃HCSs | Sphere | Hard template | [ | |
| Fe2O3@N⁃PC/Mn3O4 | Nanocapsule | Template method | [ | |
| Bi2Se3@C | Sphere | Solvothermal method | [ | |
| MnO2@NCG | Honeycomb | Sol⁃gel | [ | |
| Si@C@ZIF⁃67⁃800N | — | Sol⁃gel, MOF self⁃template | [ | |
| ZnSe/2(CoSe2)@NC | Hexahedron | Two⁃step method | [ | |
| MYS⁃Co4N@C/SeS2 | Cube | Chelation competition induced polymerization | [ | |
| TiC@C@Graphene | Fiber | Electrospinning progress | [ | |
| DCS⁃Si | Sphere | Chemical vapor deposition | [ | |
| Muti⁃yolk⁃muti⁃shell | M⁃cores@HoMS | Sphere | Hydrothermal method | [ |
| Sn NPs@NxC HoMS⁃DL | Sphere | In situ evolution of shell to core | [ |
Table 1 Design methods of yolk-shell nanoreactors
| Classification | Material | Morphology | Synthesis method | Ref. |
|---|---|---|---|---|
| Single⁃yolk⁃single⁃shell | FeP | Nanosphere⁃in⁃nanotube | Hard template | [ |
| SiO x /C@Void@C | Nanosphere | Hard template | [ | |
| Sb2Se3@void@C | Nanorod | Hard template | [ | |
| Co3O4/C@SiO2 | Rhombic dodecahedron | Hard template | [ | |
| Co3O4@TiO2@C | Sphere | Hard template | [ | |
| Si⁃infilled capsule | Capsule | Soft template | [ | |
| Si@void@C | — | Soft template | [ | |
| Classification | Material | Morphology | Synthesis method | Ref. |
| Single⁃yolk⁃single⁃shell | Cu⁃FeP@C | Sphere | Template method | [ |
| ZnS@C | Nanorod | Self⁃sacrifice template strategy | [ | |
| NiSe2@C | Sphere | Ostwald ripening | [ | |
| CoOHCl@C | — | Ostwald ripening | [ | |
| NiCo2O4/NiO | Sphere | Ostwald ripening | [ | |
| MoSe2 | Sphere | Ostwald ripening | [ | |
| NCS/CNT | Nanosphere | Kirkendall effect | [ | |
| MnO x /C | Nanoparticle | Ostwald ripening, Kirkendall effect | [ | |
| Sn⁃Sn2Co3@CoSnO3⁃Co3O4 | Microsphere | Spray drying | [ | |
| Si@void@C/CNTs | Microsphere | Spray drying | [ | |
| Sb@C | Microsphere | Spray drying | [ | |
| Co3O4@NiCo2O4 | Rhombic dodecahedron | Seed epitaxial growth | [ | |
| Fe2N@TiO2@C | Submicrocube | Hard template | [ | |
| Single⁃yolk⁃muti⁃shell | Co3O4 | Cube | Hydrothermal method | [ |
| Co3O4 | Sphere | Hydrothermal method | [ | |
| YDS⁃FCCNs | Nanospheres | Solvothermal method | [ | |
| V2O3/C | Sphere | Sequential self⁃template | [ | |
| Fe3O4⁃GC | Microsphere | Kirkendall diffusion | [ | |
| NiO | — | Sequential templating approach | [ | |
| Cobalt sulfide MSNBs | Nanobox | Ion⁃conversion⁃exchange | [ | |
| m⁃Y⁃S Fe1-x S@C⁃3 | Hexagonal nanosheet | Hard template | [ | |
| Muti⁃yolk⁃single⁃shell | SnO2/Mn2SnO4@C | Nanobox | Hard template | [ |
| Co⁃NC@N⁃HCSs | Sphere | Hard template | [ | |
| Fe2O3@N⁃PC/Mn3O4 | Nanocapsule | Template method | [ | |
| Bi2Se3@C | Sphere | Solvothermal method | [ | |
| MnO2@NCG | Honeycomb | Sol⁃gel | [ | |
| Si@C@ZIF⁃67⁃800N | — | Sol⁃gel, MOF self⁃template | [ | |
| ZnSe/2(CoSe2)@NC | Hexahedron | Two⁃step method | [ | |
| MYS⁃Co4N@C/SeS2 | Cube | Chelation competition induced polymerization | [ | |
| TiC@C@Graphene | Fiber | Electrospinning progress | [ | |
| DCS⁃Si | Sphere | Chemical vapor deposition | [ | |
| Muti⁃yolk⁃muti⁃shell | M⁃cores@HoMS | Sphere | Hydrothermal method | [ |
| Sn NPs@NxC HoMS⁃DL | Sphere | In situ evolution of shell to core | [ |

Fig.1 Schematic diagram of single⁃core⁃single⁃shell synthesis method(A) Hard template; (B) soft template; (C) Ostwald ripening; (D) Kirkendall effect.
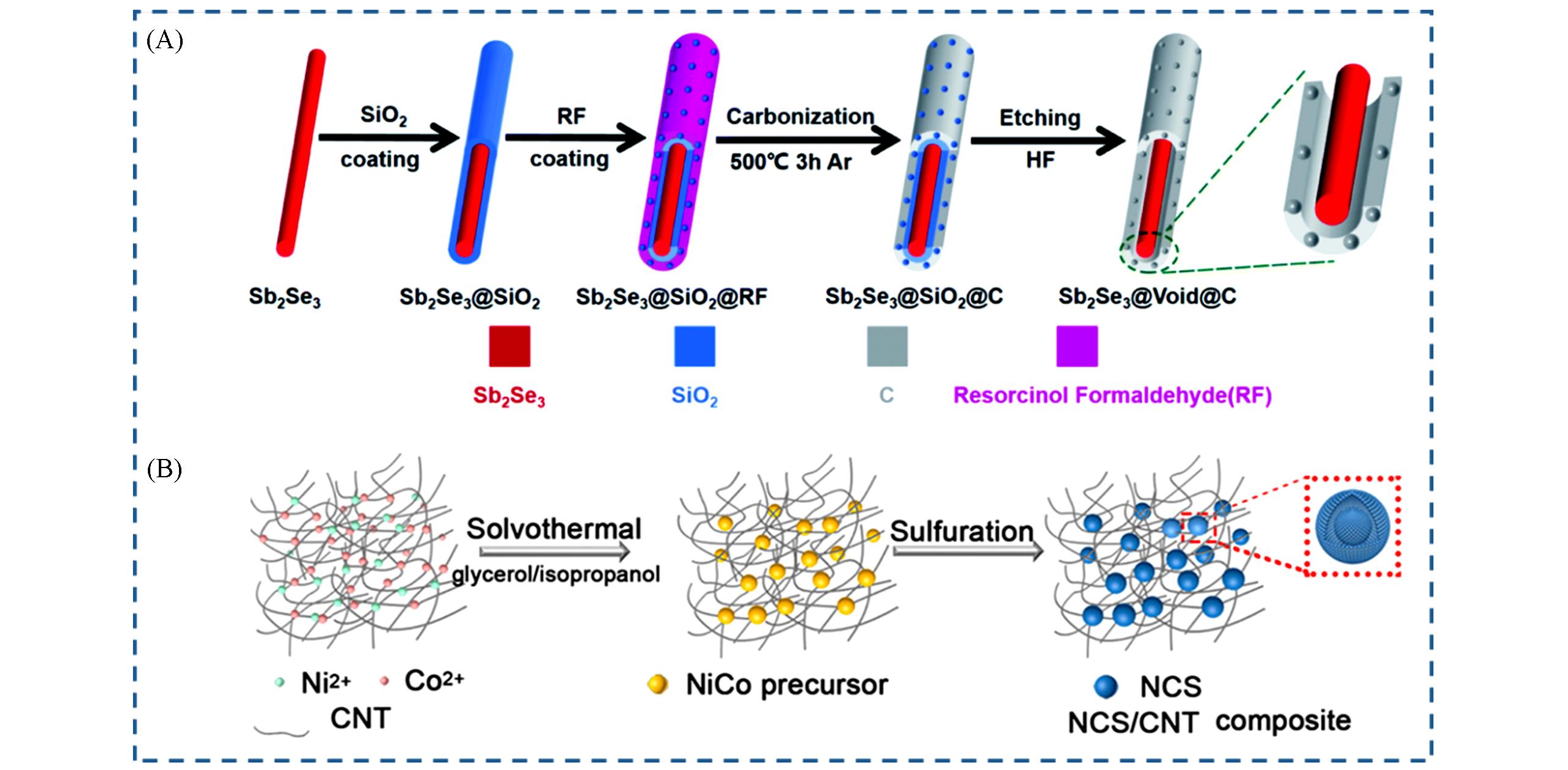
Fig.2 Hard template method(A)[13] and Kirkendall effect(B)[24] of single⁃core⁃single⁃shell nanoreactors(A) Copyright 2021, the Royal Society of Chemistry; (B) Copyright 2021, American Chemical Society.
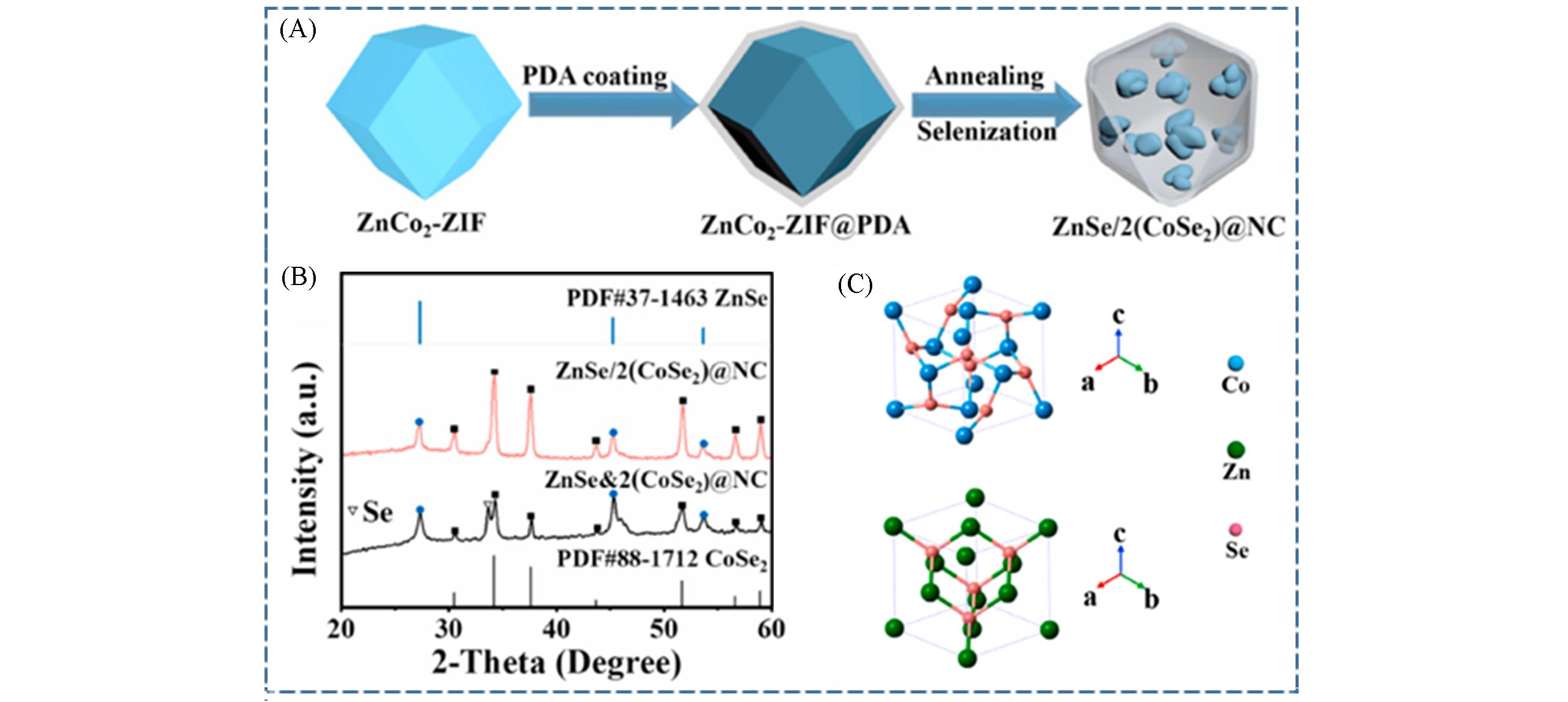
Fig.4 Design method of multi⁃cores⁃single⁃shell nanoreactors[45](A) Schematic illustration of the synthesis procedures of ZnSe/2(CoSe2)@NC; (B) XRD patterns of ZnSe/2(CoSe2)@NC and ZnSe&2(CoSe2)@NC; (C) crystal structure of ZnSe and CoSe2. Copyright 2021, Elsevier.
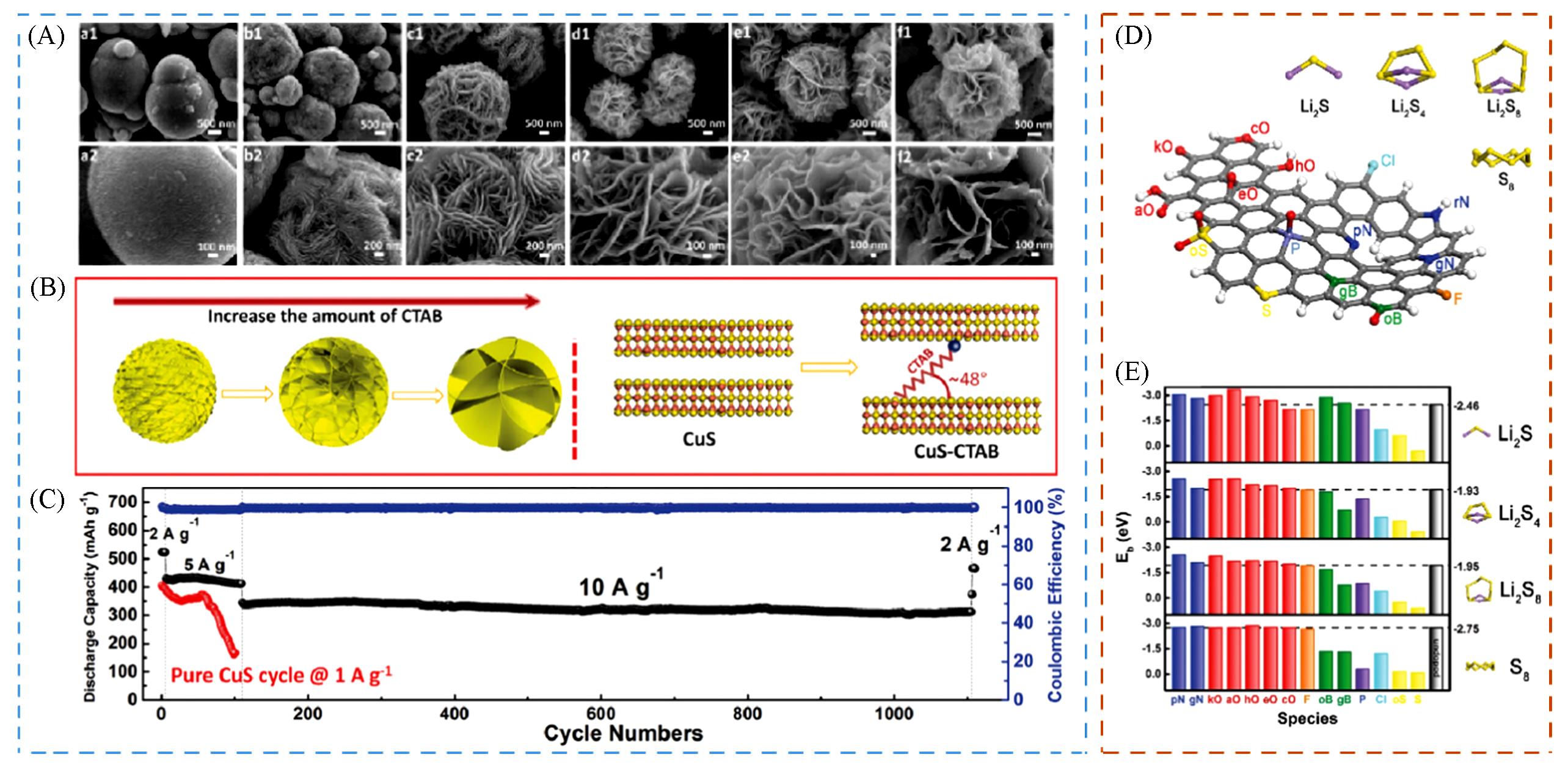
Fig.5 Regulatory strategies for tunable channel shell(A—C)[68] and functionalized shell(D, E)[71](A) SEM images of CuS microspheres with adding a 0 g(a1), 0.05 g(b1), 0.1 g(c1), 0.2 g(d1) , 0.3 g(e1) and 0.5 g(f1) of CTAB; (B) schematic of morphological changing processes of CuS-CTAB microspheres with increasing the amount of CTAB and the interlayer space change of CuS-CTAB after inserting the CTAB; (C) cycling performance at different current densities and the corresponding CE of CuS-CTAB-0.2 microspheres, and cycling performance of the synthesized pure CuS without intercalation of CTAB for comparison; (D) schematic illustration of X-doped nanocarbon materials(X = B, N, O, F, P, S, Cl); (E) the binding energy Eb(eV) of Li2S, Li2S4, Li2S8, and S8 interacting with X-doped GNRs(a modal to simulate the copious boundary and edge regions of CNTs and graphene).(A—C) Copyright 2018, Wiley-VCH. (D, E) Copyright 2016, Wiley-VCH.
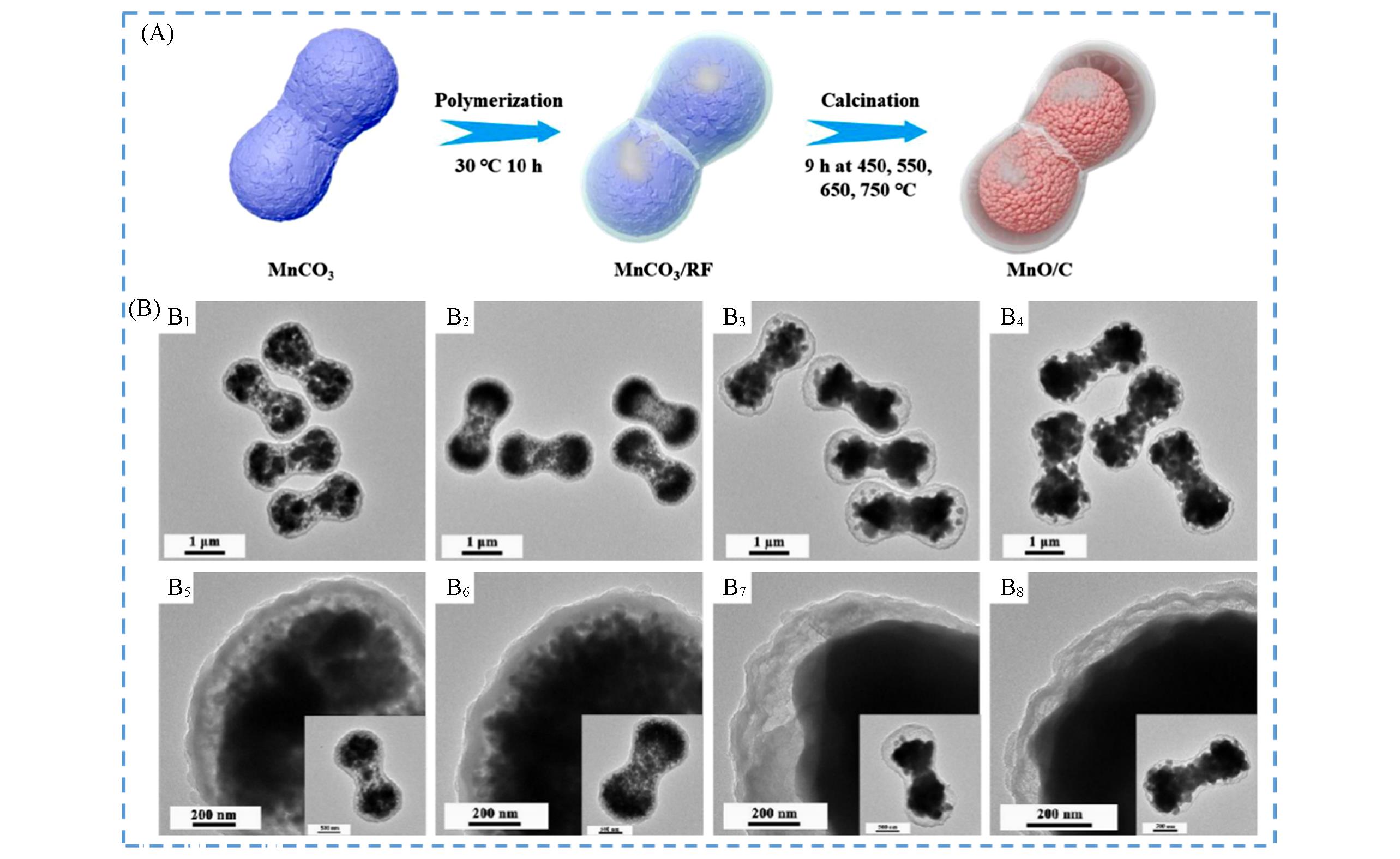
Fig.6 Schematic diagram of dumbbell⁃like MnO/C microspheres(A) and TEM images of dumbbell⁃like MnO/C microspheres under different temperatures(B)[88]Temperature/℃: (B1, B5) 450; (B2, B6) 550; (B3, B7) 650; (B4, B8) 750. Copyright 2020, Elsevier.
| Classification | Host material | S content,w(%) | Highest reversible capacity/(mA·h·g-1) | Cycling performance | Ref. | |
|---|---|---|---|---|---|---|
| Retention/(mA·h·g-1) | Cycle | |||||
| Carbon⁃based composite | BCN@HCS | 70.0 | 1075(at 0.2C) | 700(at 1C) | 500 | [ |
| N⁃C NSs | 70.0 | 879.4(at 0.1C) | 616(at 1C) | 500 | [ | |
| HPC@S⁃PANi | 65.2 | 833.3(at 0.5 A/g) | 650.2(at 1 A/g) | 200 | [ | |
| NYSC | 75.4 | 930(at 0.1C) | 909(at 0.2C) | 500 | [ | |
| GNC | 60.0 | 1236(at 0.1C) | 810(at 1C) | 100 | [ | |
| rGO/N⁃YSHCS | 70.0 | 826(at 0.1C) | 800(at 0.2C) | 100 | [ | |
| Ploymer⁃based composite | RGO/PANI | 67.0 | — | 690(at 0.1C) | 70 | [ |
| S@void@PPy | 98.4 | — | 650(at 0.2 A/g) | 200 | [ | |
| Metal⁃based composite | MnO2@NCG | 80.0 | 1436(at 0.1C) | 871.5(at 1C) | 1000 | [ |
| Co⁃VN@C | 70.0 | 1379.2(at 0.1C) | 602(at 1C) | 300 | [ | |
| In2O3@C | 84.0 | 1042.9(at 0.2C) | 440.8(at 1C) | 1000 | [ | |
| FeSe2@C | 82.0 | 1295(at 0.1C) | 684(at 1C) | 700 | [ | |
| S@void@TiO2 | 72.4 | 1082(at 0.2C) | 766(at 0.2C) | 1000 | [ | |
| YS⁃ZnO | 76.7 | 1355(at 0.5C) | 1212(at 0.5C) | 500 | [ | |
| V2O3⁃VN@NC | 69.8 | 1220(at 0.1C) | 618(at 1C) | 800 | [ | |
| Fe3O4@void@C | 66.5 | 1010(at 0.1C) | 625(at 0.2C) | 500 | [ | |
| Co⁃NC@N⁃HCSs | 80.8 | 685.5(at 0.2C) | 399.7(at 1C) | 450 | [ | |
| Fe2O3@N⁃PC/Mn3O4 | 70.1 | 1319(at 0.2C) | 1122(at 0.5C) | 200 | [ | |
| Co@BNCNTs YS | 75.1 | 950(at 0.2C) | 700.2(at 1C) | 400 | [ | |
| MoS2/Ni3S2 | 64.7 | 1242.2(at 0.2C) | 739(at 1C) | 1000 | [ | |
| THC@CoSe2 | 61.0 | 985.3(at 0.1C) | 438.5(at 1C) | 1000 | [ | |
| MnO2@HCS | 66.3 | 887(at 0.1C) | 705(at 1C) | 500 | [ | |
| rGO/VO2 | 70.0 | 892.7(at 0.2C) | 334.9(at 1C) | 400 | [ | |
| TiO2⁃CNFs@void@TiN@C | 68.0 | 1103.5(at 0.5C) | 675.8(at 1C) | 1000 | [ | |
| Fe2N@C | 60.0 | 1361(at 0.1C) | 734(at 1C) | 600 | [ | |
Table 2 Applications for lithium-sulfur battery cathodes of yolk-shell nanoractors
| Classification | Host material | S content,w(%) | Highest reversible capacity/(mA·h·g-1) | Cycling performance | Ref. | |
|---|---|---|---|---|---|---|
| Retention/(mA·h·g-1) | Cycle | |||||
| Carbon⁃based composite | BCN@HCS | 70.0 | 1075(at 0.2C) | 700(at 1C) | 500 | [ |
| N⁃C NSs | 70.0 | 879.4(at 0.1C) | 616(at 1C) | 500 | [ | |
| HPC@S⁃PANi | 65.2 | 833.3(at 0.5 A/g) | 650.2(at 1 A/g) | 200 | [ | |
| NYSC | 75.4 | 930(at 0.1C) | 909(at 0.2C) | 500 | [ | |
| GNC | 60.0 | 1236(at 0.1C) | 810(at 1C) | 100 | [ | |
| rGO/N⁃YSHCS | 70.0 | 826(at 0.1C) | 800(at 0.2C) | 100 | [ | |
| Ploymer⁃based composite | RGO/PANI | 67.0 | — | 690(at 0.1C) | 70 | [ |
| S@void@PPy | 98.4 | — | 650(at 0.2 A/g) | 200 | [ | |
| Metal⁃based composite | MnO2@NCG | 80.0 | 1436(at 0.1C) | 871.5(at 1C) | 1000 | [ |
| Co⁃VN@C | 70.0 | 1379.2(at 0.1C) | 602(at 1C) | 300 | [ | |
| In2O3@C | 84.0 | 1042.9(at 0.2C) | 440.8(at 1C) | 1000 | [ | |
| FeSe2@C | 82.0 | 1295(at 0.1C) | 684(at 1C) | 700 | [ | |
| S@void@TiO2 | 72.4 | 1082(at 0.2C) | 766(at 0.2C) | 1000 | [ | |
| YS⁃ZnO | 76.7 | 1355(at 0.5C) | 1212(at 0.5C) | 500 | [ | |
| V2O3⁃VN@NC | 69.8 | 1220(at 0.1C) | 618(at 1C) | 800 | [ | |
| Fe3O4@void@C | 66.5 | 1010(at 0.1C) | 625(at 0.2C) | 500 | [ | |
| Co⁃NC@N⁃HCSs | 80.8 | 685.5(at 0.2C) | 399.7(at 1C) | 450 | [ | |
| Fe2O3@N⁃PC/Mn3O4 | 70.1 | 1319(at 0.2C) | 1122(at 0.5C) | 200 | [ | |
| Co@BNCNTs YS | 75.1 | 950(at 0.2C) | 700.2(at 1C) | 400 | [ | |
| MoS2/Ni3S2 | 64.7 | 1242.2(at 0.2C) | 739(at 1C) | 1000 | [ | |
| THC@CoSe2 | 61.0 | 985.3(at 0.1C) | 438.5(at 1C) | 1000 | [ | |
| MnO2@HCS | 66.3 | 887(at 0.1C) | 705(at 1C) | 500 | [ | |
| rGO/VO2 | 70.0 | 892.7(at 0.2C) | 334.9(at 1C) | 400 | [ | |
| TiO2⁃CNFs@void@TiN@C | 68.0 | 1103.5(at 0.5C) | 675.8(at 1C) | 1000 | [ | |
| Fe2N@C | 60.0 | 1361(at 0.1C) | 734(at 1C) | 600 | [ | |
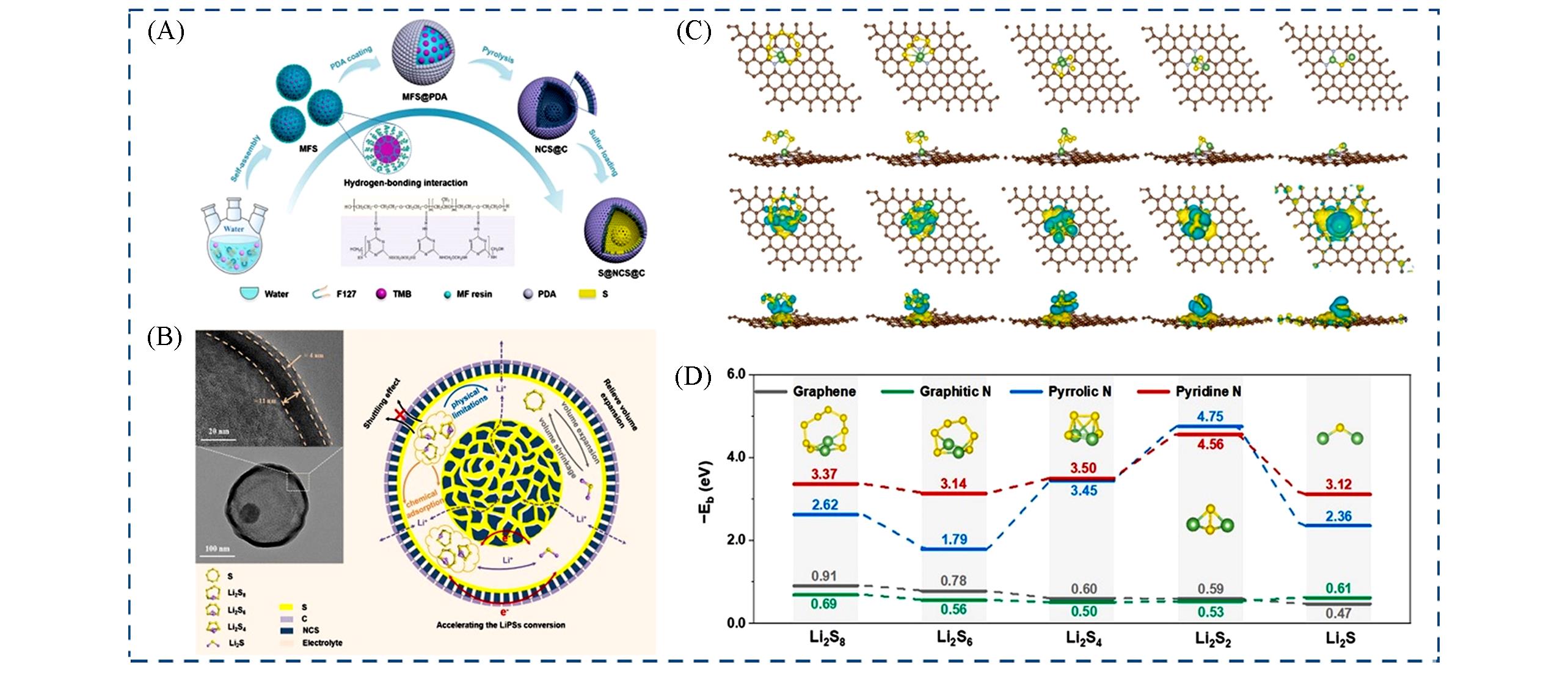
Fig.7 Application of single⁃yolk⁃multi⁃shell carbon materials in lithium⁃sulfur batteries[121](A) Schematic diagram of the preparation process of S@NCS@C-800 composites; (B) schematic illustration of the synergistic mechanism of S@NCS@C-800; (C) the optimal adsorption model of polysulfides with pyridine N and the charge density for polysulfides adsorbed on pyridine N; (D) the binding energies between different kinds of carbon and polysulfide.Copyright 2022, Elsevier.
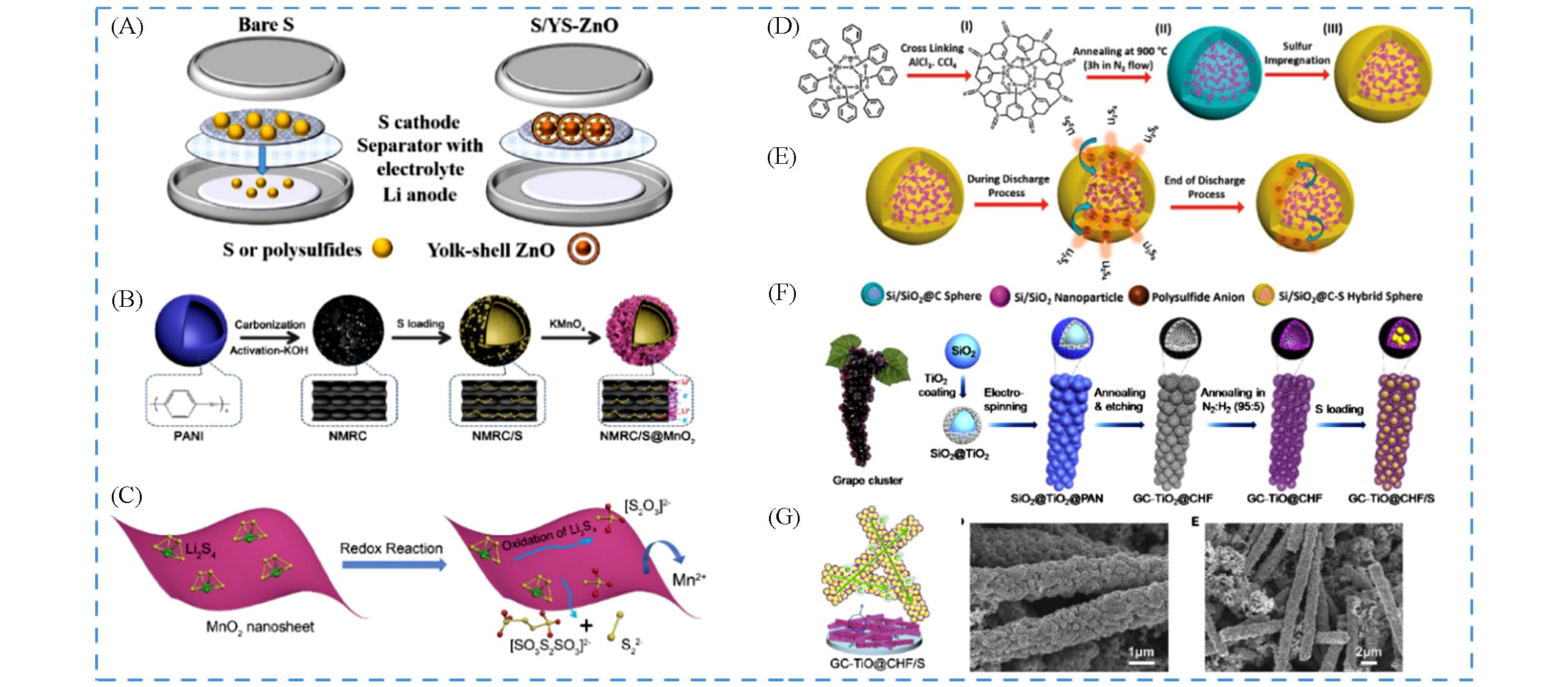
Fig.8 Application of yolk⁃shell metal oxide composite system in lithium⁃sulfur batteries(A) Schematic diagram illustrating the effect of the YS ZnO[107]; (B) the schematic synthesis process of NMRC/S@MnO2; (C) schematic illustration of the interaction between polysulfide and MnO2[128]; (D) schematic illustration of the synthesis process of the Si/SiO2@C-S hybrid spheres; (E) schematic diagram illustrating the concept of the “polysulfide reservoir” in the Si/SiO2@C-S hybrid spheres[129]; (F) schematic illustration of the synthesis process of the GC-TiO@CHF/S cathode; (G) schematic and SEM images of the GC-TiO@CHF/S electrode[131].(A) Copyright 2021, Elsevier; (B, C) Copyright 2020, Elsevier; (D, E) Copyright 2016, Wiley-VCH; (F, G) Copyright 2017, Elsevier.
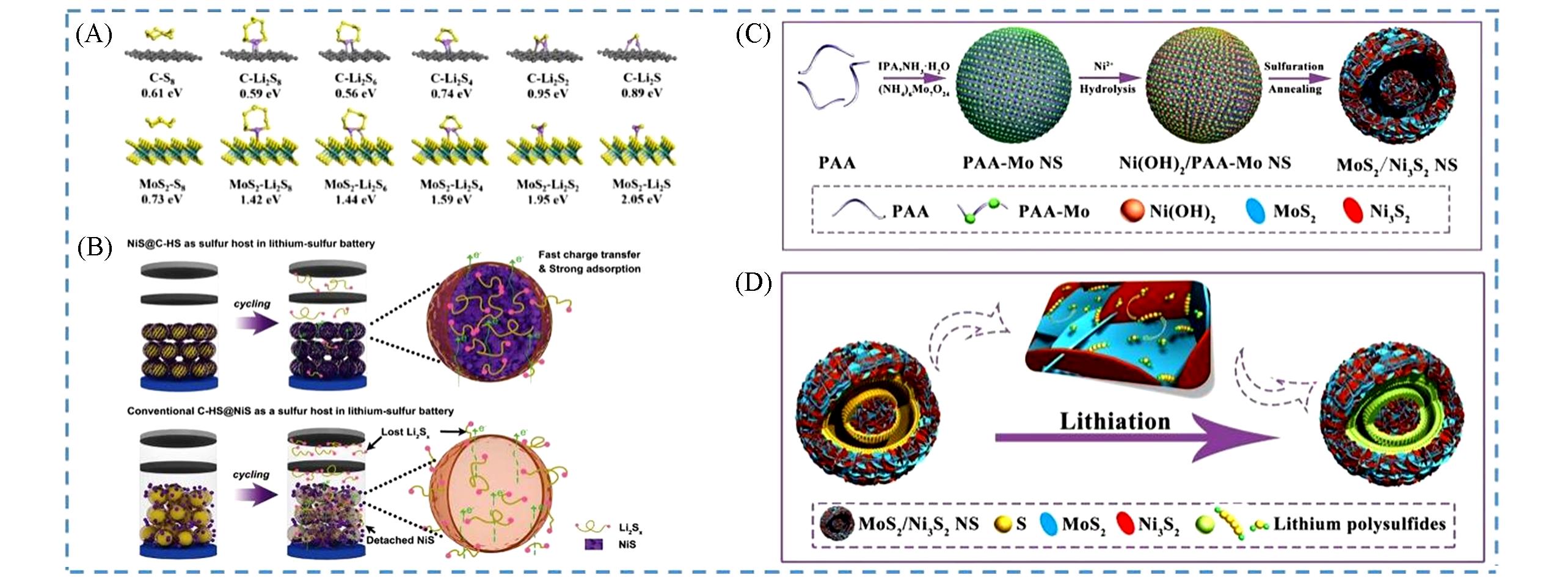
Fig.9 Application of yolk⁃shell metal sulfide composite system in lithium⁃sulfur batteries(A) Interaction schematic and corresponding binding energies of S8, Li2S8, Li2S6, Li2S4, Li2S2, and Li2S on carbon and MoS2 substrate[133]; (B) schematic illustration of NiS@C-HS and conventional C-HS@NiS as sulfur hosts for improving the performance of LSBs[134]; (C) schematic illustration of the fabrication procedure to obtain MoS2/Ni3S2 yolk-shell NS; (D) schematic illustration of the advantages of the MoS2/Ni3S2@S NS during the cycling process[110].(A) Copyright 2020, Elsevier; (B) Copyright 2017, Wiley-VCH; (C, D) Copyright 2020, Elsevier.
| 1 | Liang X., Hart C., Pang Q., Garsuch A., Weiss T., Nazar L. F., Nat. Commun., 2015, 6(1), 5682 |
| 2 | Zhao J., Yang M., Yang N., Wang J., Wang D., Chem. Res. Chinese Universities, 2020, 36(3), 313—319 |
| 3 | Pang Q., Liang X., Kwok C. Y., Nazar L. F., Nat. Energy, 2016, 1(9), 1—11 |
| 4 | Sun Q., Xi B., Li J. Y., Mao H., Ma X., Liang J., Feng J., Xiong S., Adv. Energy Mater., 2018, 8(22), 1800595 |
| 5 | Liu Y., Zhao X., Li S., Zhang Q., Wang K., Chen J., Chem. Res. Chinese Universities, 2022, 38(1), 147—154 |
| 6 | Liu T., Zhang L., Cheng B., Yu J., Adv. Energy Mater., 2019, 9(17), 1803900 |
| 7 | Yu L., Hu H., Wu H. B., Lou X. W., 2017, 29(15), 1604563 |
| 8 | Moon G. D., Nanomaterials, 2020, 10(4), 10040675 |
| 9 | Kang J., Tian X., Yan C., Wei L., Gao L., Ju J., Zhao Y., Deng N., Cheng B., Kang W., Small, 2022, 18(8), 2104469 |
| 10 | Wang J., Tang H., Wang H., Yu R., Wang D., Mater. Chem. Front., 2017, 1(3), 414—430 |
| 11 | Liu J. Y., Zhou T., Wang Y., Han T. L., Hu C. Q., Zhang H. G., Nanoscale, 2021, 13(37), 15624—15630 |
| 12 | Wang H. Q., Que X. Q., Liu Y. N., Wu X. X., Yuan Q. H., Lu J. Y., Gan W., J. Alloy. Compd., 2021, 874, 159913 |
| 13 | Zhang Y. M., Li S. J., Cheng L. L., Li Y. L., Ren X. Z., Zhang P. X., Sun L. N., Yang H. Y., J. Mater. Chem. A, 2021, 9(6), 3388—3397 |
| 14 | Zhang M., Wang C. H., Liu C., Luo R., Li J. S., Sun X. Y., Shen J. Y., Han W. Q., Wang L. J., J. Mater. Chem. A, 2018, 6(24), 11226—11235 |
| 15 | Liao W. M., Tian J. H., Shan Z. Q., Lin H. Z., Na R., J. Alloy. Compd., 2018, 739, 746—754 |
| 16 | Wu Y., Han T. L., Zhou T., Qiao X., Chen X., Zhou P., Liu J. Y., Nanotechnology, 2020, 31(33), 335403 |
| 17 | Ma J. Y., Tan H., Liu H., Chao Y. M., Part. Part. Syst. Char., 2021, 38(3), 2000288 |
| 18 | Li J., Liu Q., Zhang Y. X., Jiang J. X., Wu H. B., Yu X. Y., Chem. Eng. J., 2021, 421, 127776 |
| 19 | Xu X. J., Li F. K., Zhang D. C., Liu Z. B., Zuo S. Y., Zeng Z. Y., Liu J., Adv. Sci., 2022, 9(14), 2200247 |
| 20 | Mei H., Zhang L., Zhang K. L., Gao J. H., Zhang H. B., Huang Z. D., Xu B., Sun D. F., Electrochim. Acta, 2020, 357, 136886 |
| 21 | Kim D. H., Park G. D., Kang Y. C., Int. J. Energ. Res., 2022, 46(7), 9761—9770 |
| 22 | Yang K., Yan Y., Chen W., Zeng D., Ma C., Han Y., Zhang W., Kang H., Wen Y., Yang Y., J. Electroanal. Chem., 2019, 851, 113482 |
| 23 | Mao B. G., Guo D. L., Qin J. W., Meng T., Wang X., Cao M. H., Angew. Chem. Int. Ed., 2018, 57(2), 446—450 |
| 24 | Deng Y. Y., Wang X., Wang Z. H., Wang X. M., Li Z. W., Wang L. C., Zhou C., Chen D. Y., Luo Y. F., Energ. Fuel., 2021, 35(6), 5342—5351 |
| 25 | Yang C., Yao Y., Lian Y. B., Chen Y. J., Shah R., Zhao X. H., Chen M. Z., Peng Y., Deng Z., Small, 2019, 15(16), 1900015 |
| 26 | Park G. D., Kang Y. C., Small, 2018, 14(13), 1703957 |
| 27 | Gao Y., Qiu X., Wang X., Gu A., Chen X., Yu Z., Mater. Today Commun., 2020, 25, 101589 |
| 28 | Chen B., Qin H., Li K., Zhang B., Liu E., Zhao N., Shi C., He C., Nano Energy, 2019, 66, 104133 |
| 29 | Zhao Y. J., Ding L., Wang X. M., Yang X. M., He J. B., Yang B. J., Wang B. N., Zhang D. W., Li Z. W., J. Alloy. Compd., 2021, 861, 157945 |
| 30 | Du Y. C., Weng W. S., Zhang Z. Z., He Y. A., Xu J. Y., Yang T., Bao J. C., Zhou X. S., Chinese J. Chem., 2021, 39(7), 1878—1884 |
| 31 | Xiao J., Zhu Z., Zhang G., Li H., Li Y., Ren X., Zhang P., Mi H., Appl. Surf. Sci., 2021, 545, 149022 |
| 32 | Li J., Li F., Liao J., Li H., Dang D., Liu Q., Peng H. J., Adv. Mater. Interfaces, 2020, 7(14), 2000667 |
| 33 | Wang X., Wang J., Chen Z., Yang K., Zhang Z., Shi Z., Mei T., Qian J., Li J., Wang X., J. Alloy. Compd., 2020, 822, 153656 |
| 34 | Li Y., Zhang S., Wang S., Leng J., Jiang C., Ren X., Zhang Z., Yang Y., Tang Z., J. Mater. Chem. A, 2019, 7(33), 19234—19240 |
| 35 | Park G. D., Hong J. H., Jung D. S., Lee J. H., Kang Y. C., J. Mater. Chem. A, 2019, 7(26), 15766—15773 |
| 36 | Qin M., Lan D., Liu J. L., Liang H. S., Zhang L. M., Xing H., Xu T. T., Wu H. J., Chem. Rec., 2020, 20(2), 102—119 |
| 37 | Wang X., Chen Y., Fang Y., Zhang J., Gao S., Lou X. W., Angew. Chem. Int. Ed., 2019, 58(9), 2675—2679 |
| 38 | Shi X. L., Gan Y. M., Zhang Q. X., Wang C. Y., Zhao Y., Guan L. H., Huang W., Adv. Mater., 2021, 33(33), 2100837 |
| 39 | Tian J., Yang L. W., Zha L. X., Wang R., Huang S. J., Xu G. B., Wei T. Y., Li H. P., Cao J. X., Wei X. L., J. Power Sources, 2021, 506, 230243 |
| 40 | Sun X., Liu S., Sun W., Li Y., Wang D., Guo Q., Hong X., Xu J., Zheng C., Electrochim. Acta, 2021, 398, 139302 |
| 41 | Liu H., Chen Z., Zhou L., Pei K., Xu P., Xin L., Zeng Q., Zhang J., Wu R., Fang F., Che R., Sun D., Adv. Energy Mater., 2019, 9(37), 1901667 |
| 42 | Zhu Y., Zhao J., Li L., Xu J., Zhao X., Mi Y., Jin J., Dalton T., 2021, 50(31), 10758—10764 |
| 43 | Yan W., Chen S., Wen M., Wu Q., Yu S., J. Mater. Chem. A, 2021, 9(4), 2276—2283 |
| 44 | Liu N., Liu J., Jia D., Huang Y., Luo J., Mamat X., Yu Y., Dong Y., Hu G., Energy Storage Mater., 2019, 18, 165—173 |
| 45 | Yan W., Cao M., Fan S., Liu X., Liu T., Li H., Su J., Compos. Part B⁃Eng., 2021, 213, 108732 |
| 46 | Chen T., Kong W., Fan M., Zhang Z., Wang L., Chen R., Hu Y., Ma J., Jin Z., J. Mater. Chem. A, 2019, 7(35), 20302—20309 |
| 47 | Zhang Y., Zhang P., Li B., Zhang S., Liu K., Hou R., Zhang X., Silva S. R. P., Shao G., Energy Storage Mater., 2020, 27, 159—168 |
| 48 | Chen S., Shen L., van Aken P. A., Maier J., Yu Y., Adv. Mater., 2017, 29(21), 1605650 |
| 49 | Zhao J., Wang J., Bi R., Yang M., Wan J., Jiang H., Gu L., Wang D., Angew. Chem. Int. Ed., 2021, 60(49), 25719—25722 |
| 50 | Li B., Wang J., Bi R., Yang N., Wan J., Jiang H., Gu L., Du J., Cao A., Gao W., Wang D., Adv. Mater., 2022, 34(18), 2200206 |
| 51 | Guo S., Hu X., Hou Y., Wen Z., ACS Appl. Mater. Inter., 2017, 9(48), 42084—42092 |
| 52 | He J., Luo L., Chen Y., Manthiram A., Adv. Mater., 2017, 29(34), 1702707 |
| 53 | Du X., Zhao C., Luan Y., Zhang C., Jaroniec M., Huang H., Zhang X., Qiao S. Z., J. Mater. Chem. A, 2017, 5(40), 21560—21569 |
| 54 | Liu Z., Luo Y., Zhou M., Wang W., Gan N., Okada S., Yamaki J. I., Electrochemistry, 2015, 83(12), 1067—1070 |
| 55 | Su X., Huang J., Yan B., Hong Z., Li S., Pang B., Luo Y., Feng L., Zhou M., Xia Y., RSC Adv., 2018, 8(55), 31388—31395 |
| 56 | Park S. K., Kim J. K., Chan K. Y., J. Mater. Chem. A, 2017, 5(35), 18823—18830 |
| 57 | Cho J. S., Kang Y. C., Small, 2015, 11(36), 4673—4681 |
| 58 | Xie Q., Lin L., Ma Y., Zeng D., Yang J., Huang J., Wang L., Peng D. L., Electrochim. Acta, 2017, 226, 79—88 |
| 59 | Hong Y. J., Son M. Y., Park B. K., Kang Y. C., Small, 2013, 9(13), 2224—2227 |
| 60 | Choi S. H., Kang Y. C., ACS Appl. Mater. Inter., 2015, 7(44), 24694—24702 |
| 61 | Leng J., Wang Z., Li X., Guo H., Li H., Shih K., Yan G., Wang J., J. Mater. Chem. A, 2017, 5(29), 14996—15001 |
| 62 | Acapulco J. A. I. Jr., Hong S., Kim S. K., Park S., J. Colloid Interf. Sci., 2016, 461, 376—382 |
| 63 | Kim T. J., Yoon J. H., Yi G. R., Yoo P. J., Nanoscale, 2019, 11(28), 13650—13658 |
| 64 | Zhao Y., Li J., Wu C., Ding Y., Guan L., ChemPlusChem, 2012, 77(9), 748—751 |
| 65 | Gu L., Jiang L., Jin J., Liu J., Sun G., Carbon, 2015, 82, 572—578 |
| 66 | Zhang W., Li J., Guan P., Lv C., Yang C., Han N., Wang X., Song G., Peng Z., J. Alloy. Compd., 2020, 835, 155135 |
| 67 | Li C., Yang W., He W., Zhang X., Zhu J., Energy Storage Mater., 2021, 43, 1—19 |
| 68 | Xiao Y., Su D., Wang X., Wu S., Zhou L., Shi Y., Fang S., Cheng H. M., Li F., Adv. Energy Mater., 2018, 8(22), 1800930 |
| 69 | Yang J., Wang Y. X., Chou S. L., Zhang R., Xu Y., Fan J., Zhang W. X., Liu H. K., Zhao D., Dou S. X., Nano Energy, 2015, 18, 133—142 |
| 70 | Maihom T., Kaewruang S., Phattharasupakun N., Chiochan P., Limtrakul J., Sawangphruk M., J. Phys. Chem. C, 2018, 122(13), 7033—7040 |
| 71 | Hou T. Z., Chen X., Peng H. J., Huang J. Q., Li B. Q., Zhang Q., Li B., Small, 2016, 12(24), 3283—3291 |
| 72 | Zhou W., Xiao X., Cai M., Yang L., Nano Lett, 2014, 14(9), 5250—5256 |
| 73 | Peng Y., Zhang Y., Huang J., Wang Y., Li H., Hwang B. J., Zhao J., Carbon, 2017, 124, 23—33 |
| 74 | Zhou X., Tian J., Wu Q., Hu J., Li C., Energy Storage Mater., 2020, 24, 644—654 |
| 75 | Park S. K., Lee J. K., Kang Y. C., Adv. Funct. Mater., 2018, 28(18), 1705264 |
| 76 | Xu J., Xu L., Zhang Z., Sun B., Jin Y., Jin Q., Liu H., Wang G., Energy Storage Mater., 2022, 47, 223—234 |
| 77 | Zhang X., Zhang P., Zhang S., Zhang Y., Hou R., Liu K., Miao F., Shao G., J. Energy Chem., 2020, 51, 378—387 |
| 78 | Yu X., Sun Y., Liang F., Jiang B., Yang Z., Macromolecules, 2018, 52(1), 96—102 |
| 79 | Xiang D., Jiang B., Liang F., Yan L., Yang Z., Macromolecules,2020, 53(3), 1063—1069 |
| 80 | Jing J., Jiang B., Liang F., Yang Z., ACS Macro Lett., 2019, 8(6), 737—742 |
| 81 | Liu Z., Lu T., Song T., Yu X. Y., Lou X. W., Paik U., Energ. Environ. Sci., 2017, 10(7), 1576—1580 |
| 82 | Sun W., Liu C., Li Y., Luo S., Liu S., Hong X., Xie K., Liu Y., Tan X., Zheng C., ACS Nano, 2019, 13(10), 12137—12147 |
| 83 | Sun W., Li Y., Liu S., Liu C., Tan X., Xie K., Chem. Eng. J., 2021, 416(10), 129166 |
| 84 | Yang F., Gao H., Hao J., Zhang S., Li P., Liu Y., Chen J., Guo Z., Adv. Funct. Mater., 2019, 29(16), 1808291 |
| 85 | Guo B., Ma Q., Zhang L., Yang T., Liu D., Zhang X., Qi Y., Bao S. J., Xu M., Chem. Eng. J., 2020, 127521 |
| 86 | Wang R., Wang K., Gao S., Jiang M., Zhou M., Cheng S., Jiang K., Nanoscale, 2017, 9(39), 14881—14887 |
| 87 | Qiu W., Jiao J., Xia J., Zhong H., Chen L., Chem., 2015, 21(11), 4359—4367 |
| 88 | Chen J., Yang K., Yu H., Shah T., Zhang Q., Zhang B., Carbon, 2020, 170, 37—48 |
| 89 | Yin J., Zhang Y., Lu Q., Wu X., Jiang Z., Dang L., Ma H., Guo Y., Gao F., Yan Q., J. Mater. Chem. A, 2017, 5(25), 12757—12761 |
| 90 | Li J., Wang J., Liang X., Zhang Z., Liu H., Qian Y., Xiong S., ACS Appl. Mater. Inter., 2014, 6(1), 24—30 |
| 91 | Zhang Z., Ji Y., Li J., Tan Q., Zhong Z., Su F., ACS Appl. Mater. Inter., 2015, 7(11), 6300—6309 |
| 92 | Wang J., Tang H., Zhang L., Ren H., Yu R., Jin Q., Qi J., Mao D., Yang M., Wang Y., Liu P., Zhang Y., Wen Y., Gu L., Ma G., Su Z., Tang Z., Zhao H., Wang D., Nature Energy, 2016, 1(5), 160650 |
| 93 | Zhao X. X., Wang J. Y., Yu R. B., Wang D., J. Am. Chem. Soc., 2018, 140(49), 17114—17119 |
| 94 | Xu S., Hessel C. M., Ren H., Yu R., Jin Q., Yang M., Zhao H., Wang D., Energy Environ. Sci., 2014, 7(2), 632—637 |
| 95 | Wang J., Yang N., Tang H., Dong Z., Jin Q., Yang M., Kisailus D., Zhao H., Tang Z., Wang D., Angew. Chem., 2013, 52(25), 6417—6420 |
| 96 | Lin L., Pei F., Peng J., Fu A., Cui J., Fang X., Zheng N., Nano Energy, 2018, 54, 50—58 |
| 97 | Jin Z., Liu B., Zhang Q., Sun G., Li L., Zhang L., Wang C., Su Z., J. Alloy. Compd., 2020, 834, 155128 |
| 98 | Wu F., Zhao S., Chen L., Lu Y., Su Y., Li J., Bao L., Yao J., Zhou Y., Chen R., Electrochim. Acta, 2018, 292, 199—207 |
| 99 | Xu J., Fan H., Su D., Wang G., J. Alloy. Compd., 2018, 747, 283—292 |
| 100 | Yuan Y., Tan G., Wen J., Lu J., Ma L., Liu C., Zuo X., Shahbazian⁃Yassar R., Wu T., Amine K., Adv. Funct. Mater., 2018, 28(38), 1706443 |
| 101 | Zhang Y., Sun K., Zhan L., Wang Y., Ling L., Appl. Surf. Sci., 2018, 427, 823—829 |
| 102 | Man J. Z., Yang X., Li Z. H., Liu L., Lei G. T., Ding Y. H., Ionics, 2021, 27(6), 2455—2464 |
| 103 | Zhang M., Zhu M. F., Zhong Y., Han T. L., Sun B., Zhu S. G., Gu C. P., Kong L. T., Zhang H. G., Liu J. Y., Nanotechnology, 2020, 31(45), 455402 |
| 104 | Ren W., Xu L., Zhu L., Wang X., Ma X., Wang D., ACS Appl. Mater. Inter., 2018, 10(14), 11642—11651 |
| 105 | Liu J., Ding Y., Shen Z., Zhang H., Han T., Guan Y., Tian Y., Braun P. V., Adv. Sci., 2021, 9(3), 2103517 |
| 106 | Yu Y., Yan M., Dong W. D., Wu L., Tian Y. W., Deng Z., Chen L. H., Hasan T., Li Y., Su B. L., Chem. Eng. J., 2021, 417, 129241 |
| 107 | Zhang R., Wu M., Fan X., Jiang H., Zhao T., J. Energy Chem., 2021, 55, 136—144 |
| 108 | Zhao M., Lu Y., Yang Y., Zhang M., Yue Z., Zhang N., Peng T., Liu X., Luo Y., Nanoscale, 2021, 13(30), 13085—13094 |
| 109 | Zhou T., Shen Z., Wu,Y., Han T., Zhu M., Qiao X., Zhu Y., Zhang H., Liu J., Nanoscale, 2021, 13(16), 7744—7750 |
| 110 | Jin Z., Liang Z., Zhao M., Zhang Q., Liu B., Zhang L., Chen L., Li L., Wang C., Chem. Eng. J., 2020, 394, 124983 |
| 111 | Shao Q., Guo D., Wang C., Chen J., J. Alloy. Compd., 2020, 842, 150790 |
| 112 | Song Z., Lu X., Hu Q., Lin D., Zheng Q., Dalton T., 2020, 49(42), 14921—14930 |
| 113 | Liu S., Hong X., Li Y., Xu J., Zheng C., Xie K., RSC Adv., 2016, 6(100), 98035—98041 |
| 114 | Chen J. J., Zhang Q., Shi Y. N., Qin L. L., Cao Y., Zheng M. S., Dong Q. F., Phys. Chem. Chem. Phys., 2012, 14(16), 5376—5382 |
| 115 | Zhang W., Int. J. Electrochem. Sci., 2020, 15(8), 7925—7934 |
| 116 | Zhou L., Danilov D. L. E., Rüdiger‐A Notten P. H. L., Adv. Energy Mater., 2020, 11(15), 2001304 |
| 117 | Li X., Cheng X., Gao M., Ren D., Liu Y., Guo Z., Shang C., Sun L., Pan H., ACS Appl. Mater. Inter., 2017, 9(12), 10717—10729 |
| 118 | Zhu B., Guo G., Wu G., Zhang Y., Dong A., Hu J., Yang D., J. Alloy. Compd., 2019, 775, 776—783 |
| 119 | Yang T., Zhong Y., Liang J., Rahman M. M., Lei W., Chen Y., Part. Part. Syst. Char., 2017, 34(4), 1600281 |
| 120 | Zhang C., Hao B., Yuan C., Guo Z., Lou X. D., Angew. Chem. Int. Ed., 2012, 51, 9592 |
| 121 | Pan J., Sun Y., Wu Y., Li J., Huang W., Shi K., Lin Y., Dong H., Liu Q., Carbon, 2022, 198, 80—90 |
| 122 | Chen S., Huang X., Sun B., Zhang J., Liu H., Wang G., J. Mater. Chem. A, 2014, 2(38), 16199—16207 |
| 123 | Wang Z., Zhang S., Zhang L., Lin R., Wu X., Fang H., Ren Y., J. Power Sources, 2014, 248, 337—342 |
| 124 | Wei W., Li J., Liu D., Pan C., Liu P., Energ. Fuel., 2020, 34(6), 7676—7683 |
| 125 | Han J., Xu G., Ding B., Pan J., Dou H., MacFarlane D. R., J. Mater. Chem. A, 2014, 2(15), 5352—5357 |
| 126 | An Y., Wei P., Fan M., Chen D., Chen H., Ju Q., Tian G., Shu K., Appl. Surf. Sci., 2016, 375, 215—222 |
| 127 | Liu S., Wang Y., Zhang N., Xun Q., Zhou Z., Mater. Res. Express, 2019, 6(11), 115518 |
| 128 | Chen H., Dong W. D., Xia F. J., Zhang Y. J., Yan M., Song J. P., Zou W., Liu Y., Hu Z. Y., Liu J., Li Y., Wang H. E., Chen L. H., Su B. L., Chem. Eng. J., 2020, 381, 122746 |
| 129 | Rehman S., Guo S., Hou Y., Adv. Mater., 2016, 28(16), 3167—3172 |
| 130 | Hong Y. J., Roh K. C., Kang Y. C., Carbon, 2018, 126, 394—403 |
| 131 | Li Z., Guan B. Y., Zhang J., Lou X. W., Joule, 2017, 1(3), 576—587 |
| 132 | Wu Y., Li D., Pan J., Sun Y., Huang W., Wu M., Zhang B., Pan F., Shi K., Liu Q., J. Mater. Chem. A, 2022, 10(30), 16309—16318 |
| 133 | Shao Q., Lu P., Xu L., Guo D., Gao J., Wu Z. S., Chen J., J. Energy Chem., 2020, 51, 262—271 |
| 134 | Ye C., Zhang L., Guo C., Li D., Vasileff A., Wang H., Qiao S. Z., Adv. Funct. Mater., 2017, 27(33), 1702524 |
| 135 | Qian J., Xing Y., Yang Y., Li Y., Yu K., Li W., Zhao T., Ye Y., Li L., Wu F., Chen R., Adv. Mater., 2021, 33(25), 2100810 |
| 136 | Wang Z., Xu X., Liu Z., Ji S., Ahmed Idris S. O., Liu J., Electrochim. Acta, 2020, 332, 135482 |
| [1] | 胡平澳, 张琪, 张会茹. 锂硫电池中硒缺陷WSe2催化性能的理论研究[J]. 高等学校化学学报, 2023, 44(2): 20220595. |
| [2] | 胡栋栋, 来华, 刘宇艳, 宋颖斌, 罗欣, 张东杰, 樊志敏, 谢志民, 成中军. 形状记忆磁性润滑表面的制备及对超顺磁液滴的滑动操控[J]. 高等学校化学学报, 2023, 44(2): 20220442. |
| [3] | 赵霄朗, 杨梅, 王江艳, 王丹. 富锂正极材料结构设计和表面调控的研究进展[J]. 高等学校化学学报, 2023, 44(1): 20220263. |
| [4] | 韩付超, 李福进, 陈良, 贺磊义, 姜玉南, 徐守冬, 张鼎, 其鲁. CoSe2/C复合电催化材料修饰隔膜对高载量锂硫电池性能的影响[J]. 高等学校化学学报, 2022, 43(8): 20220163. |
| [5] | 尹肖菊, 孙逊, 赵程浩, 姜波, 赵晨阳, 张乃庆. 单原子催化剂在锂硫电池中的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220076. |
| [6] | 张诗昱, 何润合, 李永兵, 魏士俊, 张兴祥. 辐照交联制备低分子量聚丙烯腈纤维锂硫电池正极材料及其储硫机理[J]. 高等学校化学学报, 2022, 43(3): 20210632. |
| [7] | 秦文婕, 黄一倬, 罗潇, 钱旭红, 杨有军. 基于Cy7类菁染料分子探针的设计策略[J]. 高等学校化学学报, 2022, 43(12): 20220567. |
| [8] | 许文哲, 张皓. 超分子相互作用主导的纳米药物成核[J]. 高等学校化学学报, 2022, 43(10): 20220264. |
| [9] | 陈铭苏, 张会茹, 张琪, 刘家琴, 吴玉程. 锂硫电池中钴磷共掺杂MoS2催化性能的第一性原理研究[J]. 高等学校化学学报, 2021, 42(8): 2540. |
| [10] | 李辉阳, 朱思颖, 李莎, 张桥保, 赵金保, 张力. 锂离子电池硅氧化物负极首次库伦效率的影响因素与提升策略[J]. 高等学校化学学报, 2021, 42(8): 2342. |
| [11] | 耿传楠, 化五星, 凌国维, 陶莹, 张辰, 杨全红. 锂硫电池中的催化作用: 材料与表征[J]. 高等学校化学学报, 2021, 42(5): 1331. |
| [12] | 巴智晨, 梁大鑫, 谢延军. MXenes复合材料的发展: 界面调控及结构设计[J]. 高等学校化学学报, 2021, 42(4): 1225. |
| [13] | 王冶, 张晓思, 孙丽婧, 李冰, 刘琳, 杨淼, 田鹏, 刘仲毅, 刘中民. 有机硅烷辅助合成特殊形貌SAPO分子筛[J]. 高等学校化学学报, 2021, 42(3): 683. |
| [14] | 詹舒辉, 赵亚松, 杨乃亮, 王丹. 石墨炔孔结构: 设计、 合成和应用[J]. 高等学校化学学报, 2021, 42(2): 333. |
| [15] | 姜沁源, 周晨晖, 蒙海兵, 韩莹, 张如范. 二维金属有机框架材料的合成及电催化应用[J]. 高等学校化学学报, 2021, 42(2): 556. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||