

高等学校化学学报 ›› 2022, Vol. 43 ›› Issue (5): 20220076.doi: 10.7503/cjcu20220076
尹肖菊1, 孙逊2, 赵程浩2, 姜波2, 赵晨阳2, 张乃庆1( )
)
收稿日期:2022-01-30
出版日期:2022-05-10
发布日期:2022-04-07
通讯作者:
张乃庆
E-mail:znqmww@163.com
基金资助:
YIN Xiaoju1, SUN Xun2, ZHAO Chenghao2, JIANG Bo2, ZHAO Chenyang2, ZHANG Naiqing1( )
)
Received:2022-01-30
Online:2022-05-10
Published:2022-04-07
Contact:
ZHANG Naiqing
E-mail:znqmww@163.com
Supported by:摘要:
锂硫电池由于具有能量密度高、 成本低等突出特点, 已经成为下一代高能量密度电化学储能体系的重要发展方向之一. 但锂硫电池的发展仍然存在硫利用率低、 循环寿命短及倍率性能差等亟待解决的关键问题. 单原子催化剂具有高的原子利用率和原子级尺度的结构可调变性等突出特点, 在锂硫电池研究领域受到了广泛的关注. 本文从正极、 负极、 隔膜/中间层3个方面总结了单原子催化剂在锂硫电池中的最新研究进展. 最后, 还对单原子催化剂在锂硫电池中未来的研究发展方向以及需解决的关键科学和技术问题进行了展望, 以期推动单原子催化材料在锂硫电池中的进一步广泛应用.
中图分类号:
TrendMD:
尹肖菊, 孙逊, 赵程浩, 姜波, 赵晨阳, 张乃庆. 单原子催化剂在锂硫电池中的研究进展. 高等学校化学学报, 2022, 43(5): 20220076.
YIN Xiaoju, SUN Xun, ZHAO Chenghao, JIANG Bo, ZHAO Chenyang, ZHANG Naiqing. Research Progress of Single Atomic Catalysts in Lithium-sulfur Batteries. Chem. J. Chinese Universities, 2022, 43(5): 20220076.
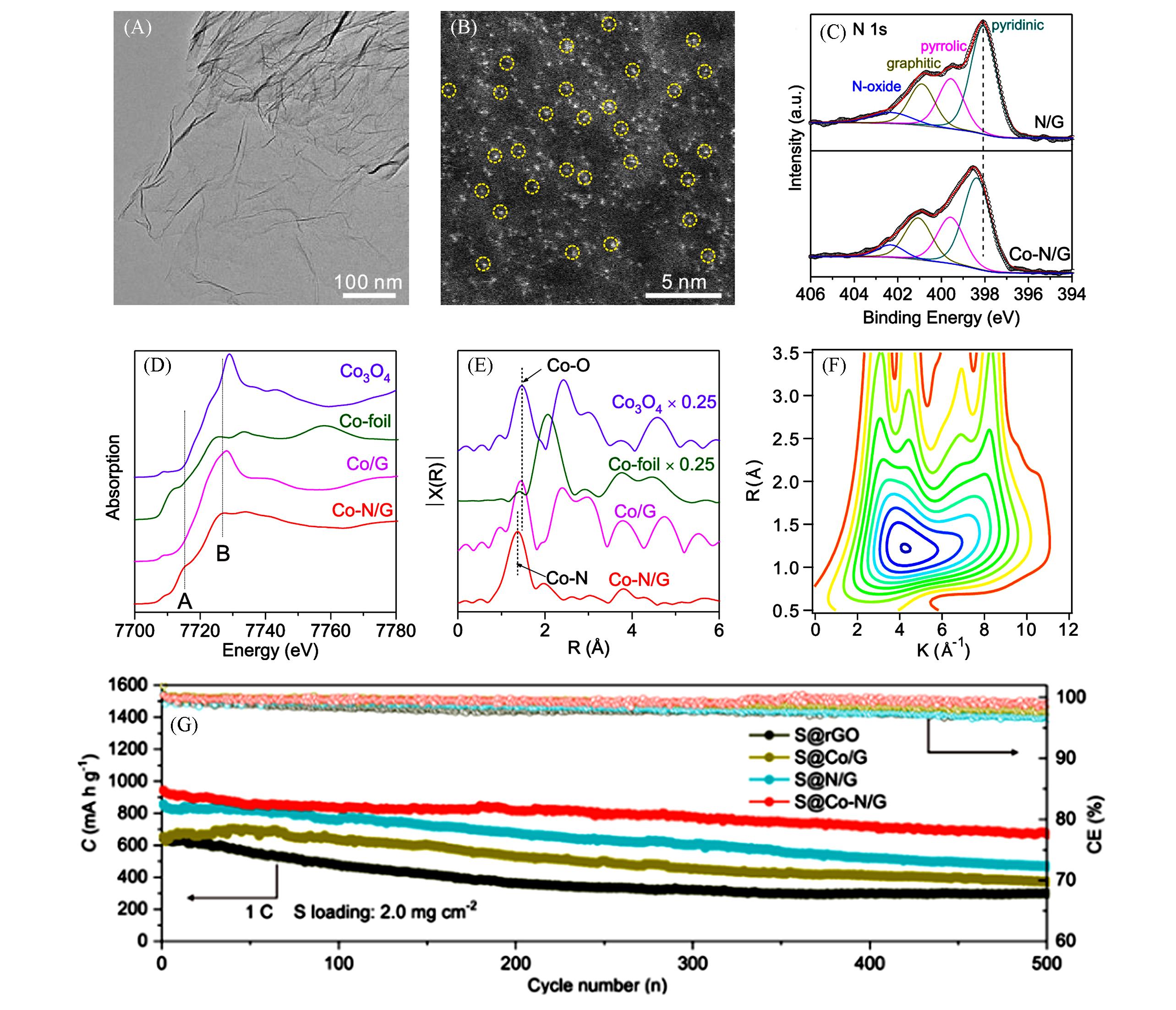
Fig.2 Structure and cycle perfomance of the Co?N/G composite[33]TEM(A) and HAADF-STEM(B) images of Co-N/G; (C) high-resolution XPS N1s spectra of Co-N/G and N/G; XANES(D) and FT-EXAFS(E) in R space for Co-N/G and reference samples including Co/G, Co-foil and Co3O4; (F) wavelet transforms for Co-N/G; (G) cycle performance of different cathode. Copyright 2019, American Chemical Society.
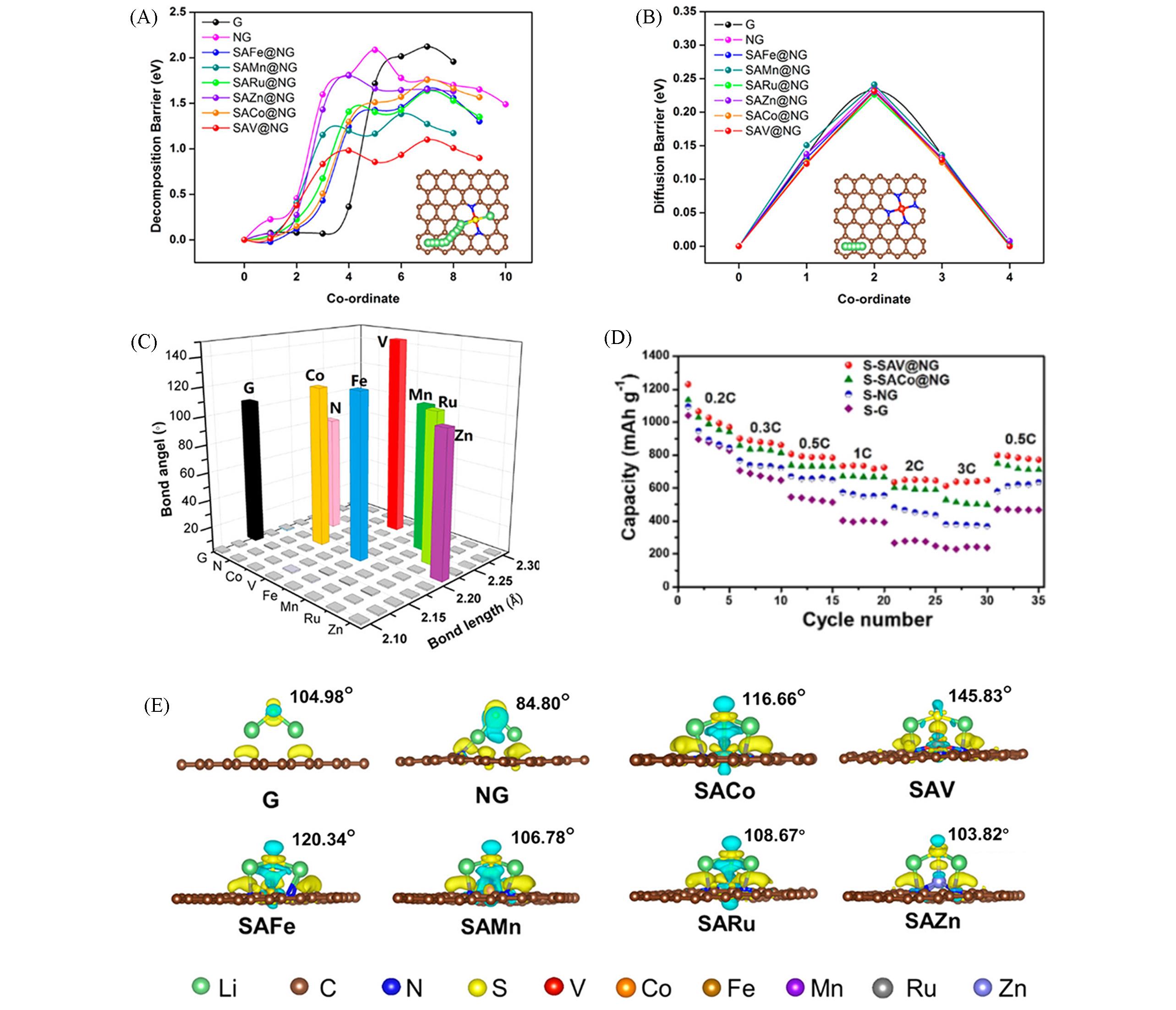
Fig.3 Theoretical understanding for Li2S decomposition, Li ion diffusion, and anchoring effect[38]Decomposition barriers of Li2S(A) and lithium ion diffusion barriers(B) on different substrates including graphene, NG, SAFe@NG, SAMn@NG, SARu@NG, SAZn@NG, SACo@NG and SAV@NG; the inset images in (A) and (B) are the detailed decomposition path of Li2S and diffusion pathway of lithium atom on SAV@NG, respectively; (C) bond angle(Li—S—Li) of Li2S and bond length(Li—S) of Li2S adsorption on the graphene, NG, SACo@NG, SAV@NG, SAFe@NG, SAMn@NG, SARu@NG and SAZn@NG, respectively; (D) rate performance of the S-G, S-NG, S-SACo@NG and S-SAV@NG electrodes; (E) side view for charge density difference of Li2S adsorption on the graphene, NG, SACo@NG, SAV@NG, SAFe@NG, SAMn@NG, SARu@NG and SAZn@NG, respectively; the yellow and blue sections represent the electron accumulate and lose region; the iso-surface is set to 0.003 eV/nm3. Copyright 2020, American Chemical Society.
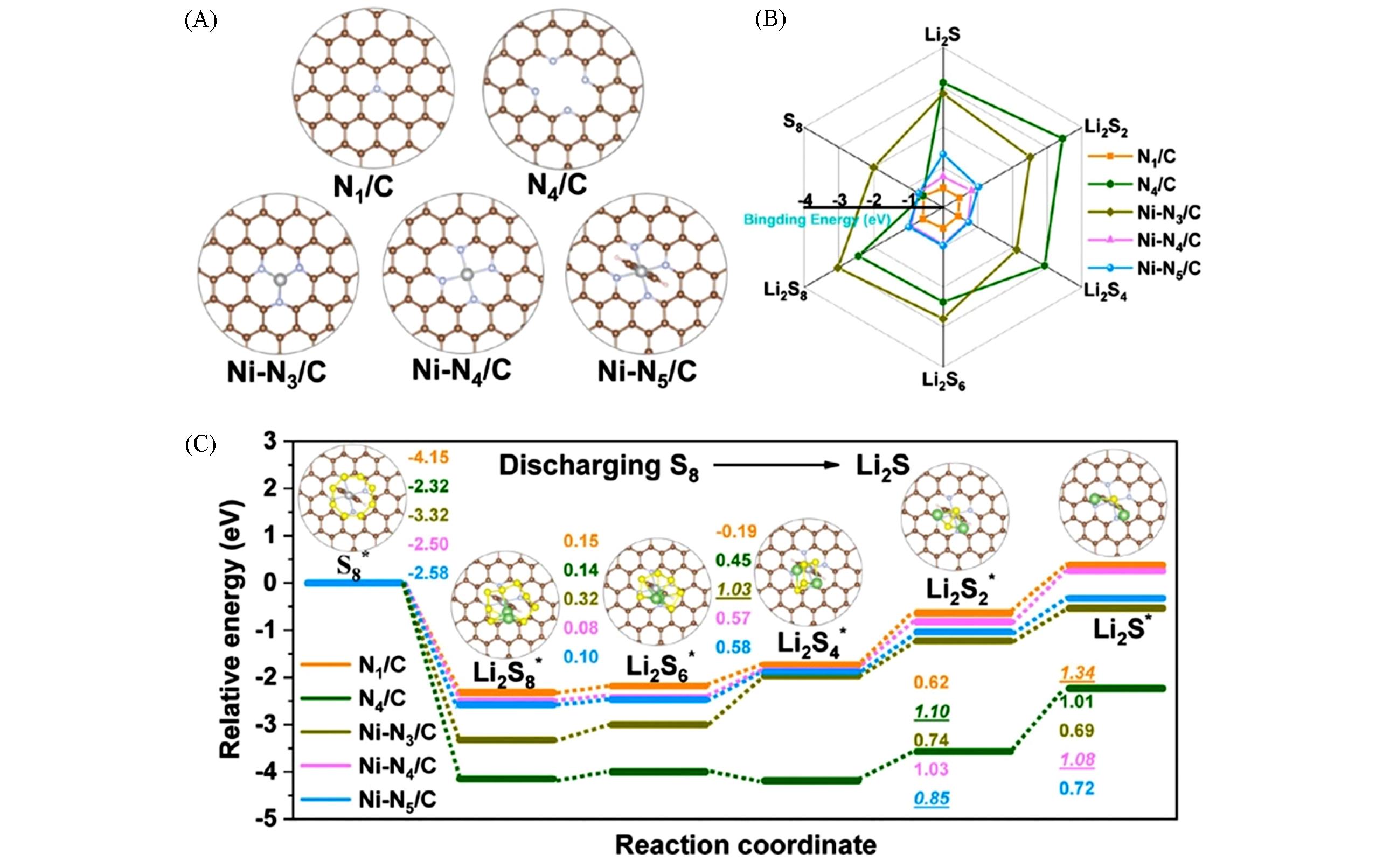
Fig.4 Computational design of Ni?N5/C[43](A) Structural models of N1/C, N4/C, Ni-N3/C, Ni-N4/C and Ni-N5/C; (B) binding energies of LiPSs on the five catalyst models; (C) energy profiles for the discharging process from S8 to Li2S on the five catalyst models; inset images are the optimized structures of LiPSs adsorption configurations on Ni-N5/C. Copyright 2021, American Chemical Society.
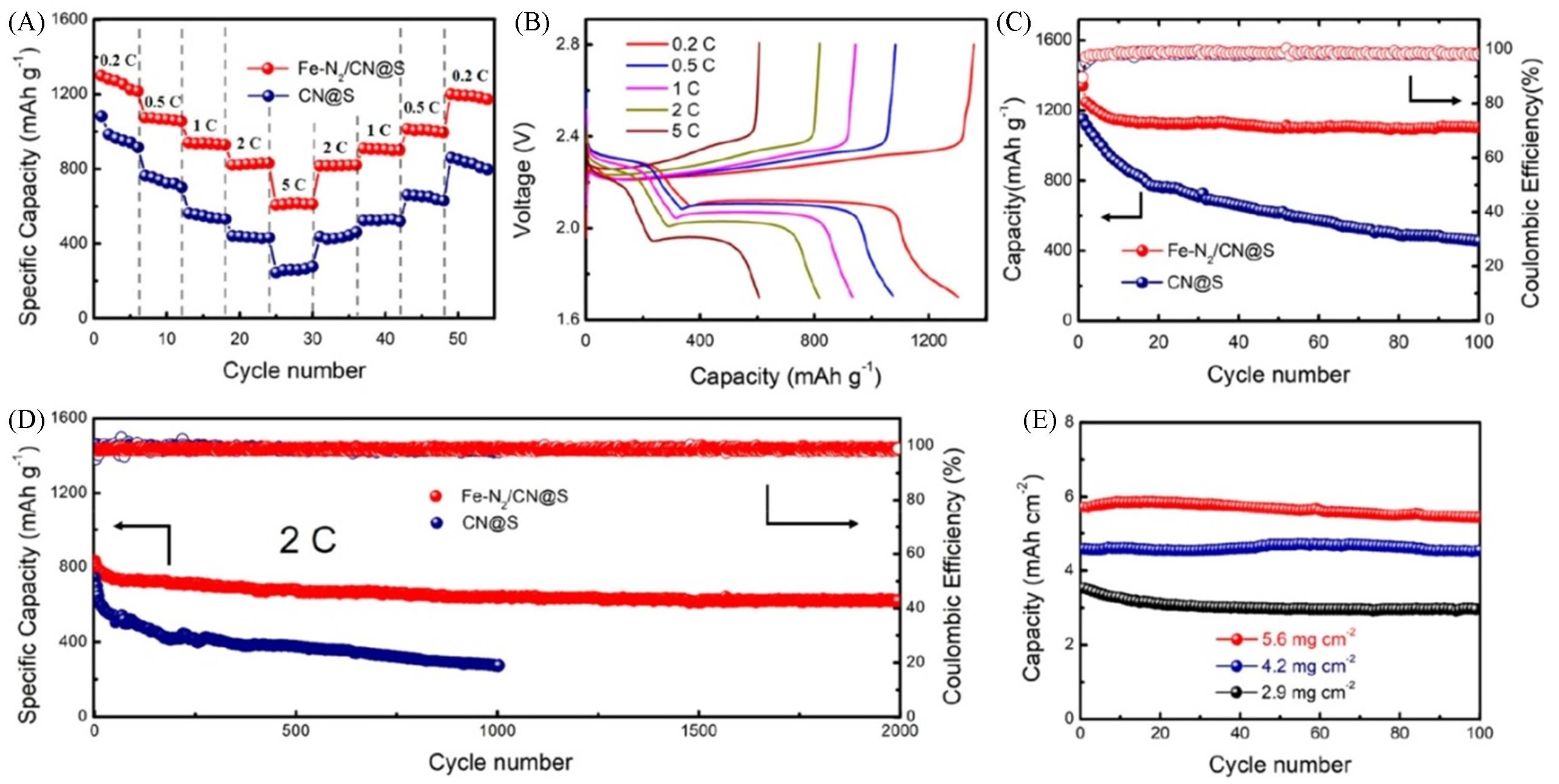
Fig.5 Electrochemical performance of Li?S batteries based on Fe?N2/CN@S and CN@S cathodes[47]Rate performance(A) and galvanostatic charge-discharge curves(B) of batteries based on Fe-N2/CN@S cathodes at 0.2C—5C; cycle performance at 0.2C(C), long-term cycle stability at 2C(D) and cycle performance with different sulfur loading cathodes at 0.2C(E). Copyright 2020, American Chemical Society.
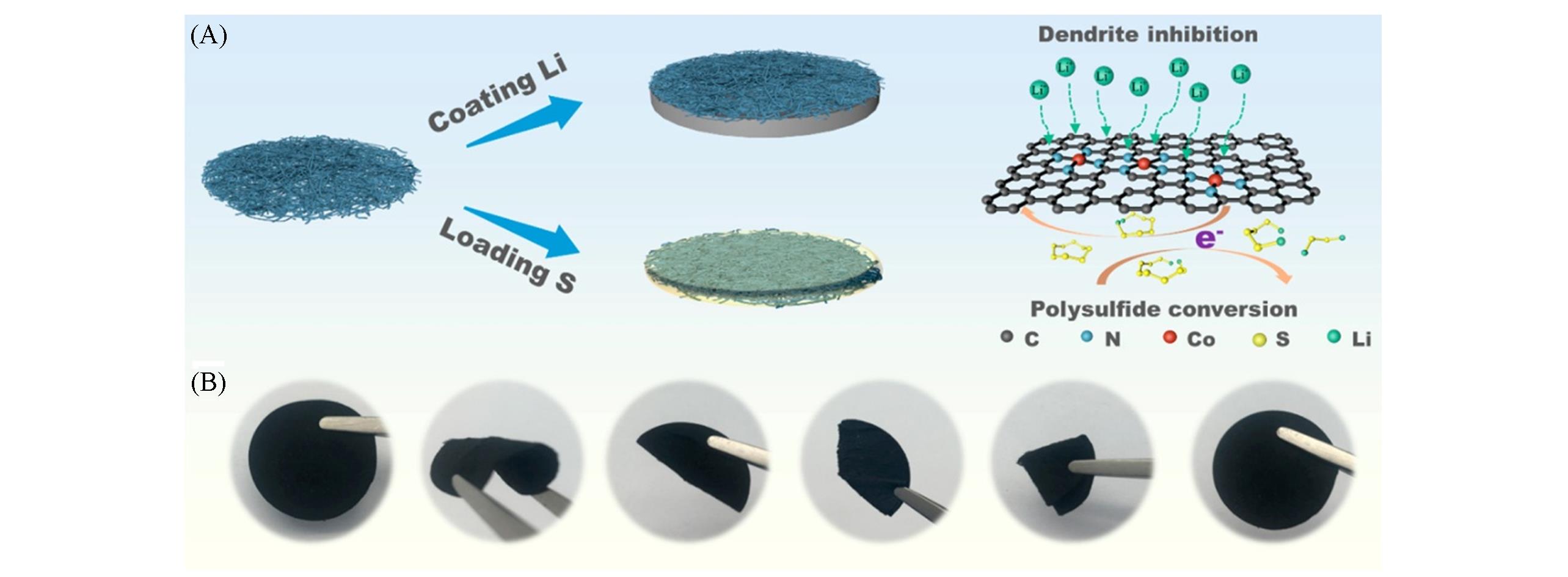
Fig.6 Functional illustration of the fibrous skeleton Co?PCNF[53](A) Schematic diagram illustrating the structural configuration and design principle of the dual-functional fibrous skeleton harnes-sing single-atomic Co-N x species; (B) digital photos showing the flexibility and robustness of the fibrous skeleton. Copyright 2021, American Chemical Society.
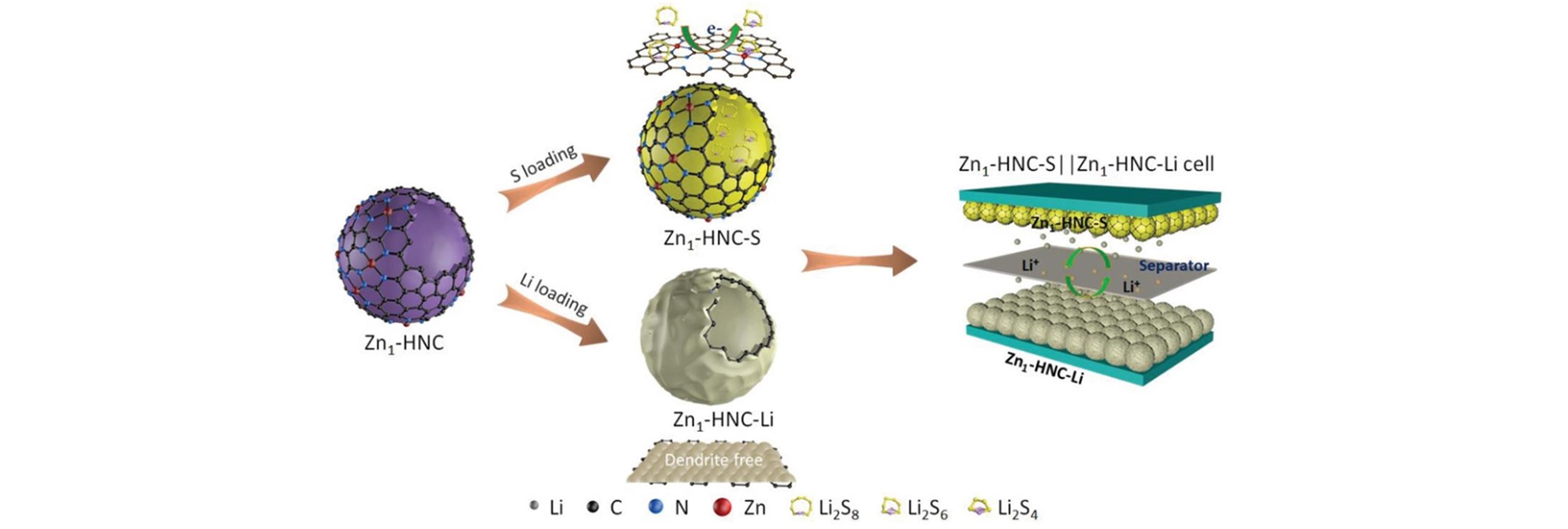
Fig.7 Synthesis schematic of the dual?functional Zn1?HNC nanoreactors for Zn1?HNC?S cathode and the Zn1?HNC?Li anode of the Zn1?HNC?S||Zn1?HNC?Li full battery[54]Copyright 2020, Wiley-VCH GmbH.
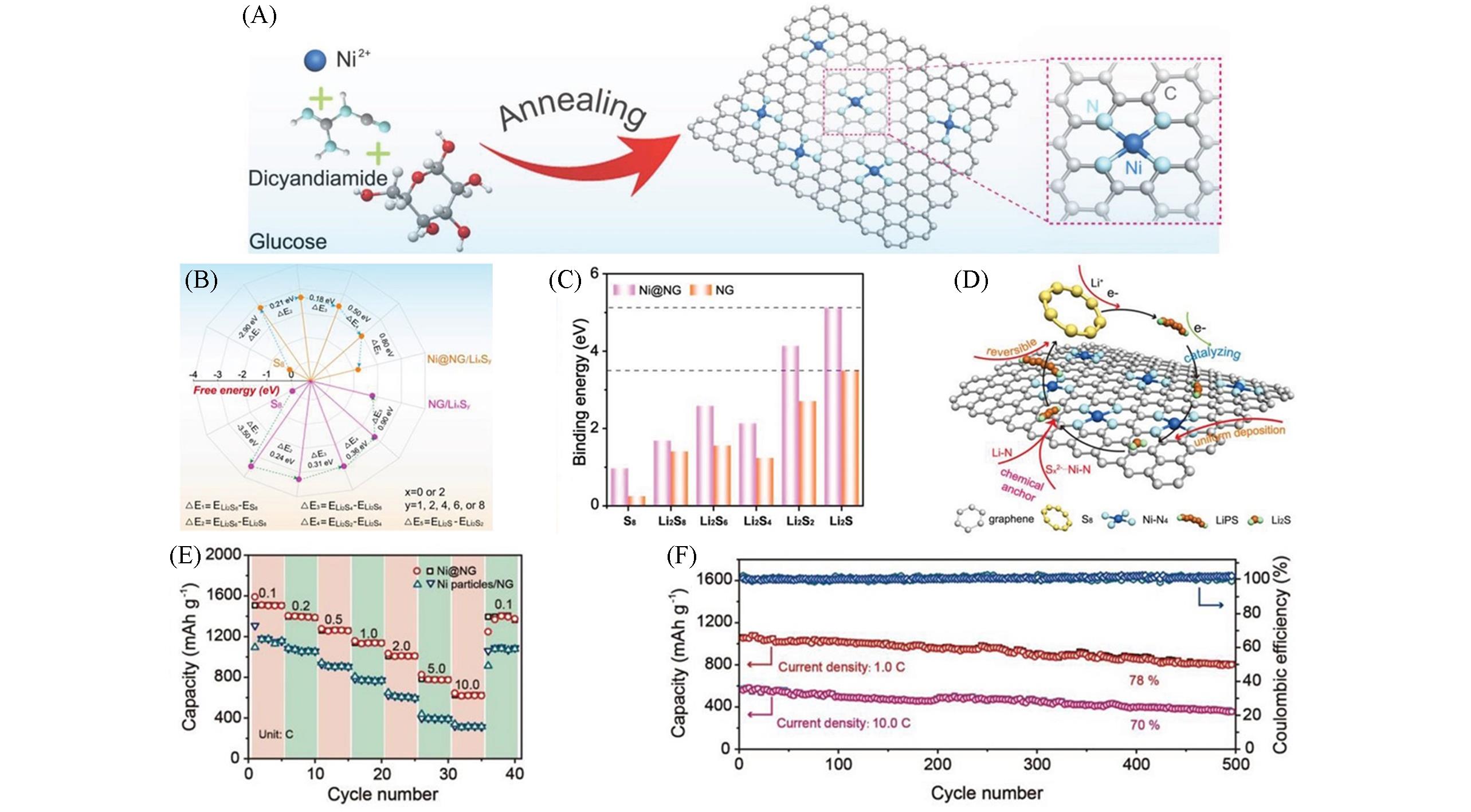
Fig.8 Schematic illustration and electrochemical performance of Ni@NG[34](A) Schematic illustration of the preparation of Ni@NG with the Ni-N4 sites; (B) the free-energy diagrams of LiPS on Ni@NG or NG; (C) binding energies between the LiPS and the Ni@NG or NG; (D) the catalytic mechanism of the LiPS on the surface of Ni@NG in electrochemical process; (E) the rate performance of the Li-S batteries based on the Ni@NG or Ni particles/NG modified separators; (F) cycling stability of the Li-S batteries based on the Ni@NG modified separator. Copyright 2019, Wiley‐VCH Verlag GmbH & Co. KGaA, Weinheim.
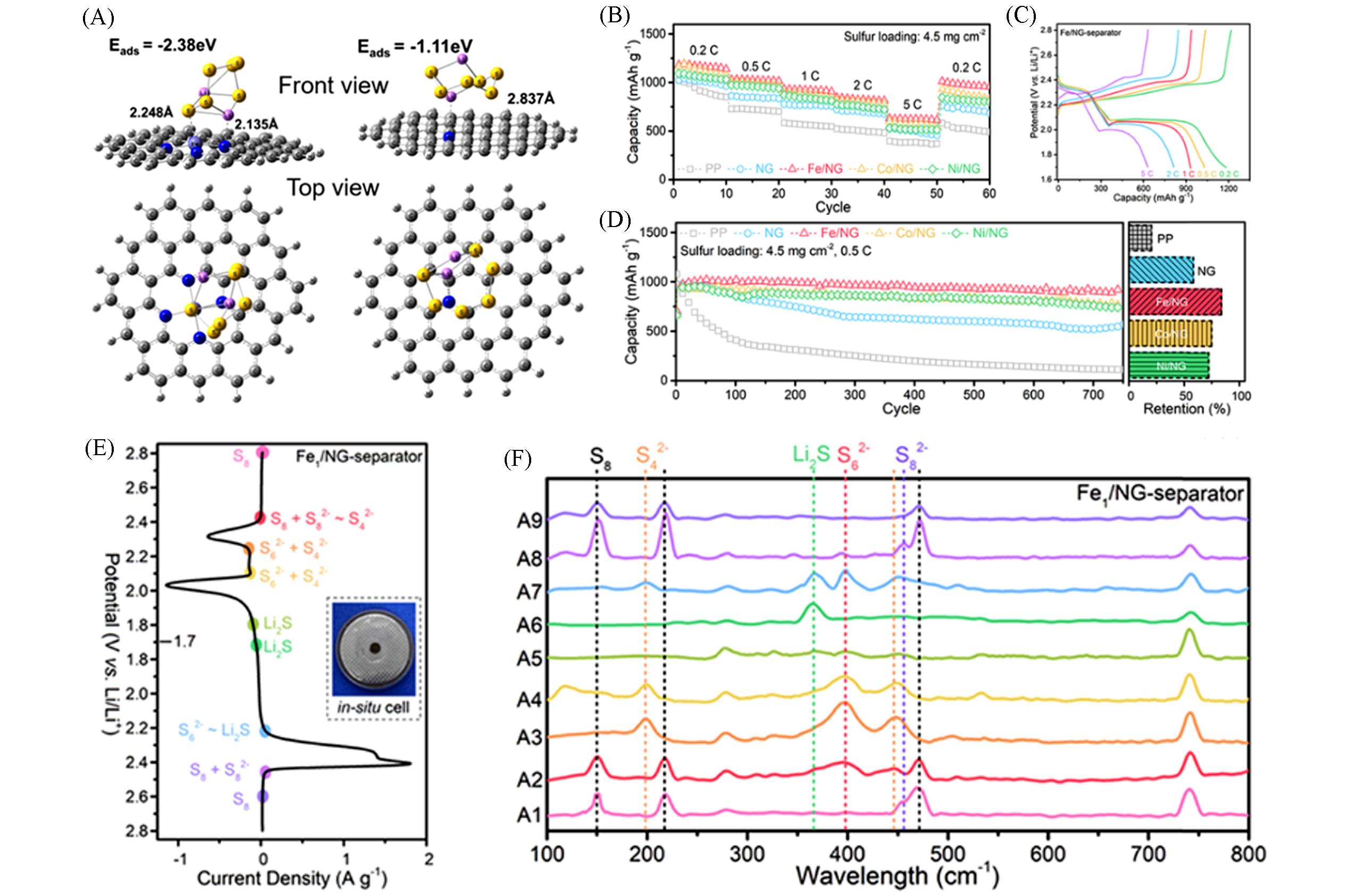
Fig.9 Theoretical calculation and experimental characterization of NG and Fe1/NG in Li?S batteries[57](A) Density functional theory(DFT) calculations of the Li2S6 adsorption on NG and Fe1/NG; rate capabilities(B), charge-discharge curves(C) and cycling performance(D) of the Li-S batteries; (E) CV profiles of the Li-S cell with the PP separator or the Fe1/NG-modified separator; inset: digital photo of the in situ Raman cell; (F) in situ Raman spectra of the Li-S cell with the Fe1/NG-modified separator at different voltages as indicated in (E). Copyright 2019, American Chemical Society.
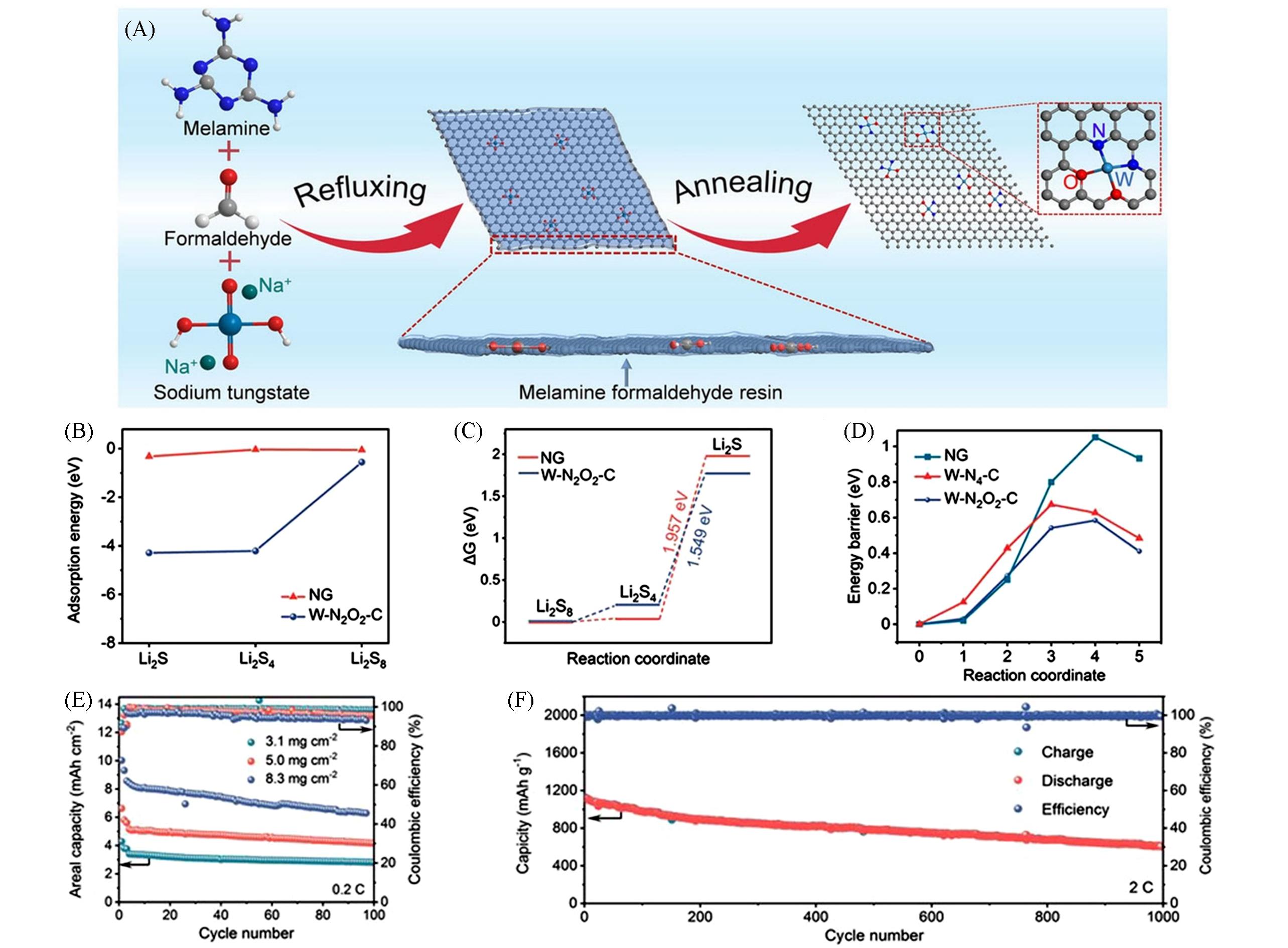
Fig.10 Schematic illustration and electrochemical performance of W/NG[58](A) Illustration of the preparation process of W/NG;(B) adsorption energies of NG and W/NG for Li2S8, Li2S4, and Li2S; (C) Gibbs free energy changes of Li2S8, Li2S4, and Li2S on NG and W/NG; (D) energy profiles for the decomposition of Li2S on NG, W-N4-C and W-O2N2-C; cycling performance of the LSBs based on W/NG@PP high sulfur loading(E) and 2C(F), respectively. Copyright 2021, Wiley‐VCH GmbH.
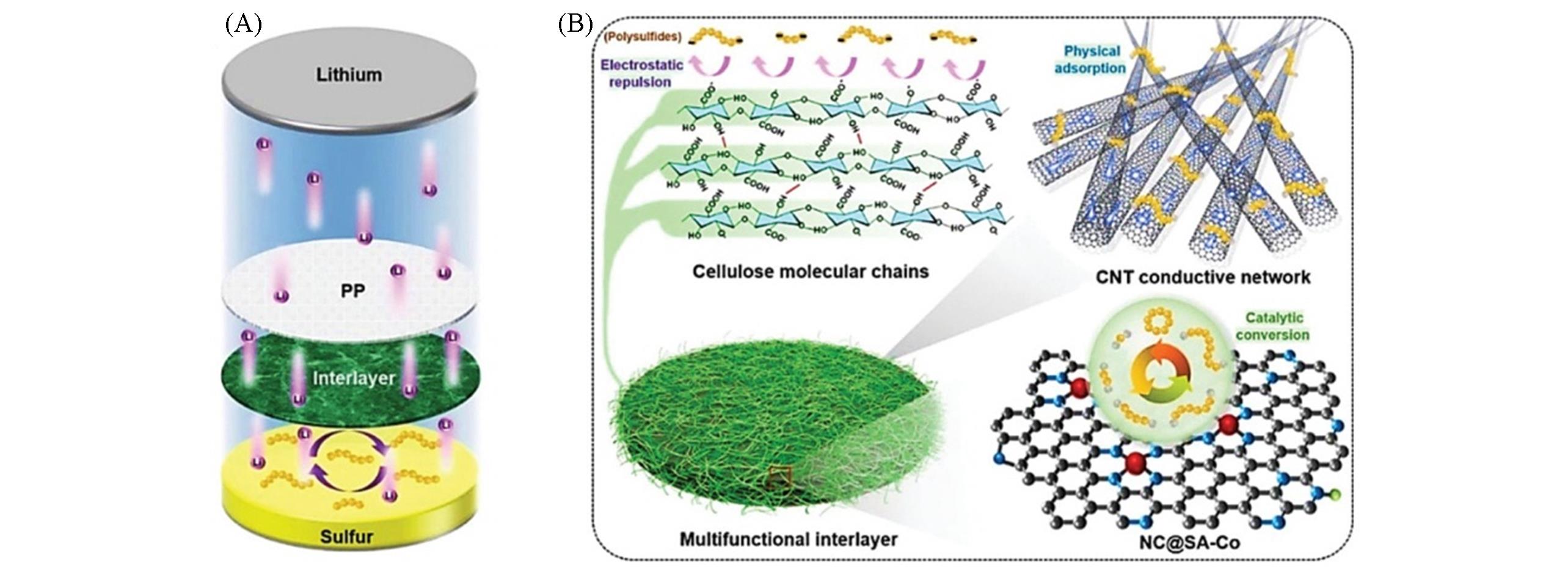
Fig.11 Schematic illustration of the multifunctional interlayer[59](A) Schematic illustration of the operation of the Li-S batteries with the configuration of the multifunctional C-C-N-Co interlayer; (B) schematic illustration of the C-C-N-Co interlayer with different functions. Copyright 2020, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim.
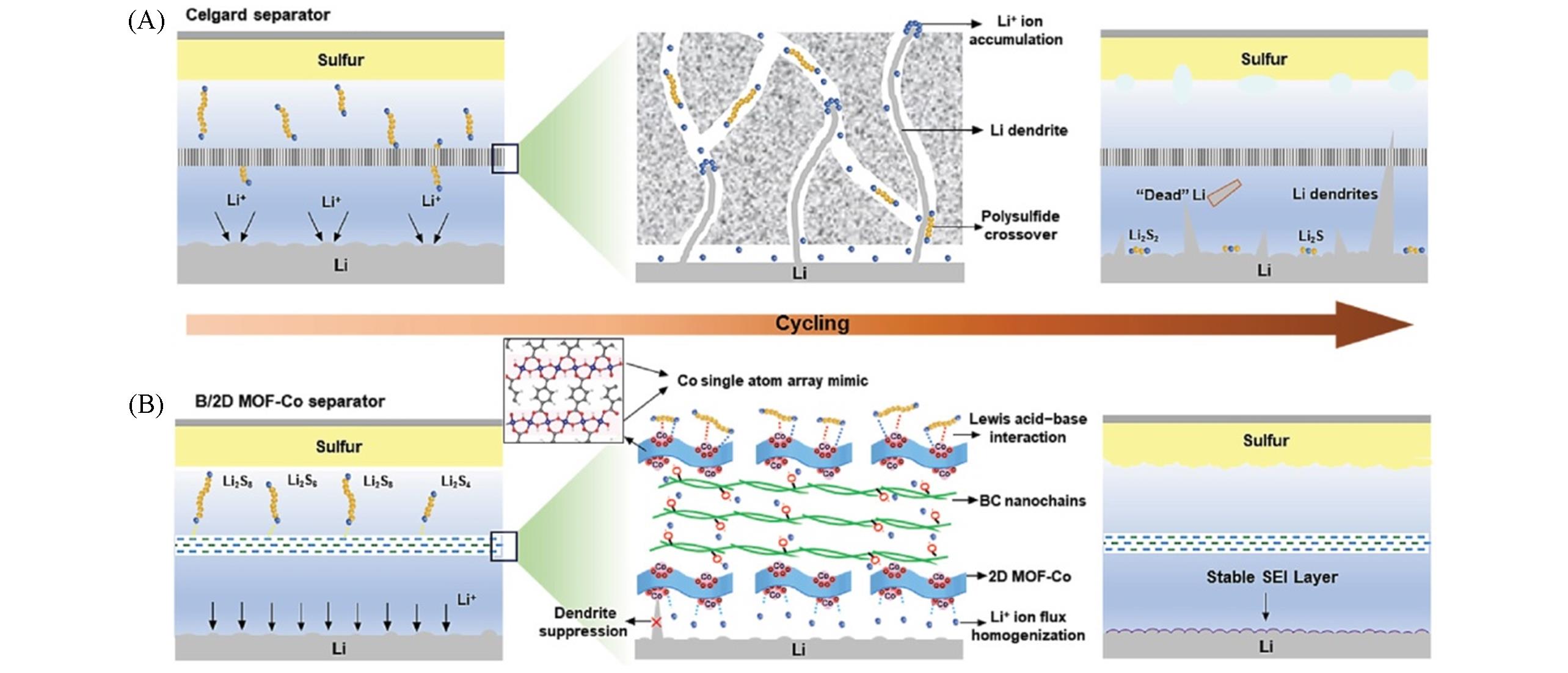
Fig.12 Schematic illustration for the Li?S batteries with celgard and B/2D MOF?Co separators[60](A) With a celgard separator: continuous Li dendrite growth and polysulfides shuttle; (B) with a B/2D MOF-Co separator: numerous Co-O4 moieties array on 2D MOF-Co nanosheets contribute to anchoring polysulfides via Lewis acid-base interaction and homogenizing Li+ ion flux, resulting in effective suppression of dendrites and polysulfide diffusion. Copyright 2020, Wiley-VCH Verlag GmbH & Co. KGaA, Weinheim.
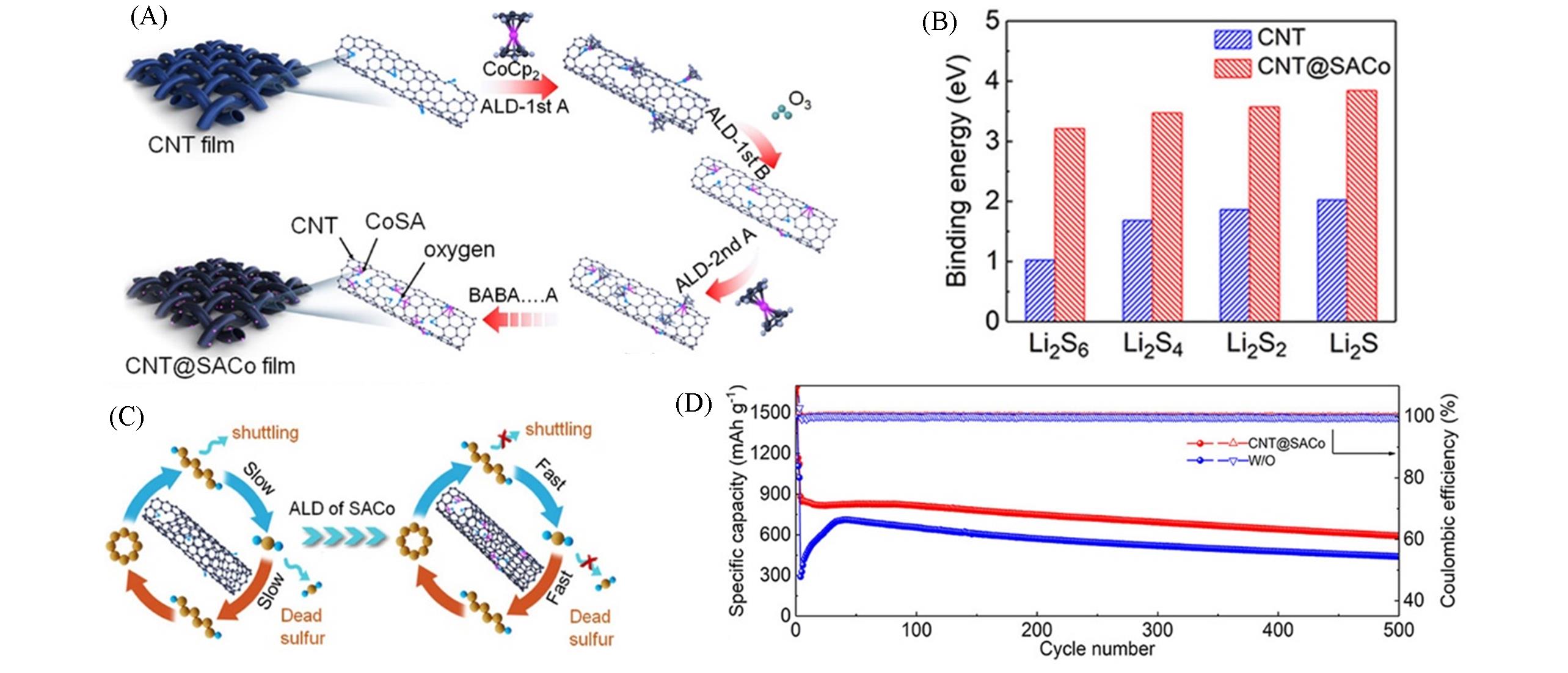
Fig.13 Schematic illustration and electrochemical performance of CNT@SACo interlayer[61](A) Schematic illustration of the preparation process of the CNT@SACo interlayer by the ALD method; (B) binding energies of polysulfides on the surfaces of carbon and SACo-decorated carbon; (C) schematic illustration of the electrochemical reaction of sulfur on the CNT and CNT@SACo; (D) cycling performances at 1C. Copyright 2020, American Chemical Society.
| 38 | Zhou G., Zhao S., Wang T., Yang S. Z., Johannessen B., Chen H., Liu C., Ye Y., Wu Y., Peng Y., Liu C., Jiang S. P., Zhang Q., Cui Y., Nano Lett., 2020, 20(2), 1252—1261 |
| 39 | Han Z., Zhao S., Xiao J., Zhong X., Sheng J., Lv W., Zhang Q., Zhou G., Cheng H. M., Adv. Mater., 2021, 33(44), 2105947 |
| 40 | Han X., Zhang Z., Xu X., J. Mater. Chem. A, 2021, 9(20), 12225—12235 |
| 41 | Liu Y., Wei Z., Zhong B., Wang H., Xia L., Zhang T., Duan X., Jia D., Zhou Y., Huang X., Energy Stor. Mater., 2021, 35, 12—18 |
| 42 | Wang Y., Adekoya D., Sun J., Tang T., Qiu H., Xu L., Zhang S., Hou Y., Adv. Funct. Mater., 2018, 29(5), 1807485 |
| 43 | Zhang S., Ao X., Huang J., Wei B., Zhai Y., Zhai D., Deng W., Su C., Wang D., Li Y., Nano Lett., 2021, 21(22), 9691—9698 |
| 44 | Zhang Y., Liu J., Wang J., Zhao Y., Luo D., Yu A., Wang X., Chen Z., Angew. Chem. Int. Ed. Engl., 2021, 60(51), 26622—26629 |
| 45 | Wang C., Song H., Yu C., Ullah Z., Guan Z., Chu R., Zhang Y., Zhao L., Li Q., Liu L., J. Mater. Chem. A, 2020, 8(6), 3421— 3430 |
| 46 | Shao Q., Xu L., Guo D., Su Y., Chen J., J. Mater. Chem. A, 2020, 8(45), 23772—23783 |
| 47 | Qiu Y., Fan L., Wang M., Yin X., Wu X., Sun X., Tian D., Guan B., Tang D., Zhang N., ACS Nano, 2020, 14(11), 16105—16113 |
| 48 | Zhang D., Wang S., Hu R., Gu J., Cui Y., Li B., Chen W., Liu C., Shang J., Yang S., Adv. Funct. Mater., 2020, 30(30), 2002471 |
| 49 | Han B., Guo Y., Huang Y., Xi W., Xu J., Luo J., Qi H., Ren Y., Liu X., Qiao B., Angew. Chem. Int. Ed. Engl., 2020, 59(29), 11824—11829 |
| 50 | Yang J., Li W., Wang D., Li Y., Adv. Mater., 2020, 32(49), 2003300 |
| 51 | Zhao C., Xu G. L., Yu Z., Zhang L., Hwang I., Mo Y. X., Ren Y., Cheng L., Sun C. J., Ren Y., Zuo X., Li J. T., Sun S. G., Amine K., Zhao T., Nat. Nanotechnol., 2021, 16(2), 166—173 |
| 52 | Wang D., Ma K., Hao J., Zhang W., Wang C., Xu C., Shi H., Ji Z., Yan X., Gu Y., Nano Energy, 2021, 89, 106426 |
| 53 | Huang T., Sun Y., Wu J., Jin J., Wei C., Shi Z., Wang M., Cai J., An X. T., Wang P., Su C., Li Y. Y., Sun J., ACS Nano, 2021, 15(9), 14105—14115 |
| 54 | Shi H., Ren X., Lu J., Dong C., Liu J., Yang Q., Chen J., Wu Z. S., Adv. Energy Mater., 2020, 10(39), 2002271 |
| 55 | Liu D., Wu C., Chen S., Ding S., Xie Y., Wang C., Wang T., Haleem Y. A., Ur Rehman Z., Sang Y., Liu Q., Zheng X., Wang Y., Ge B., Xu H., Song L., Nano Res., 2018, 11(4), 2217—2228 |
| 56 | Cao G., Wang Z., Bi D., Zheng J., Lai Q., Liang Y., Chem. Eur. J., 2020, 26(45), 10314—10320 |
| 57 | Zhang K., Chen Z., Ning R., Xi S., Tang W., Du Y., Liu C., Ren Z., Chi X., Bai M., Shen C., Li X., Wang X., Zhao X., Leng K., Pennycook S. J., Li H., Xu H., Loh K. P., Xie K., ACS Appl. Mater. Interfaces, 2019, 11(28), 25147—25154 |
| 58 | Wang P., Xi B., Zhang Z., Huang M., Feng J., Xiong S., Angew. Chem. Int. Ed. Engl., 2021, 60(28), 15563—15571 |
| 59 | Li Y., Zhou P., Li H., Gao T., Zhou L., Zhang Y., Xiao N., Xia Z., Wang L., Zhang Q., Gu L., Guo S., Small Methods, 2020, 4(3), 1900701 |
| 60 | Li Y., Lin S., Wang D., Gao T., Song J., Zhou P., Xu Z., Yang Z., Xiao N., Guo S., Adv. Mater., 2020, 32(8), 1906722 |
| 61 | Lin Q., Ding B., Chen S., Li P., Li Z., Shi Y., Dou H., Zhang X., ACS Applied Energy Materials, 2020, 3(11), 11206—11212 |
| 1 | Bruce P. G., Freunberger S. A., Hardwick L. J., Tarascon J. M., Nat. Mater., 2011, 11(1), 19—29 |
| 2 | Ji X., Nazar L. F., J. Mater. Chem., 2010, 20(44), 9821—9826 |
| 3 | Park J., Yu S. H., Sung Y. E., Nano Today, 2018, 18, 35—64 |
| 4 | Kumar R., Liu J., Hwang J. Y., Sun Y. K., J. Mater. Chem. A, 2018, 6(25), 11582—11605 |
| 5 | Seh Z. W., Sun Y., Zhang Q., Cui Y., Chem. Soc. Rev., 2016, 45(20), 5605—5634 |
| 6 | Yang Y., Zheng G., Cui Y., Chem. Soc. Rev., 2013, 42(7), 3018—3032 |
| 7 | Zhang S. S., J. Power Sources, 2013, 231, 153—162 |
| 8 | Pang Q., Liang X., Kwok C. Y., Nazar L. F., Nat. Energy, 2016, 1(9), 1—11 |
| 9 | Peng H. J., Huang J. Q., Cheng X. B., Zhang Q., Adv. Energy Mater., 2017, 7(24), 1700260 |
| 10 | Ji X., Lee K. T., Nazar L. F., Nat. Mater., 2009, 8(6), 500—506 |
| 11 | Xin S., Gu L., Zhao N. H., Yin Y. X., Zhou L. J., Guo Y. G., Wan L. J., J. Am. Chem. Soc., 2012, 134(45), 18510—18513 |
| 12 | Sahore R., Levin B. D. A., Pan M., Muller D. A., Disalvo F. J., Giannelis E. P., Adv. Energy Mater., 2016, 6(14), 1600134 |
| 13 | Zhou W., Wang C., Zhang Q., Abruña H. D., He Y., Wang J., Mao S. X., Xiao X., Adv. Energy Mater., 2015, 5(16), 1500117 |
| 14 | Zhou W., Xiao X., Cai M., Yang L., Nano Lett., 2014, 14(9), 5250—5256 |
| 15 | Pei F., An T., Zang J., Zhao X., Fang X., Zheng M., Dong Q., Zheng N., Adv. Energy Mater., 2016, 6(8), 1502539 |
| 16 | Zhou G., Zhao Y., Manthiram A., Adv. Energy Mater., 2015, 5(9), 1402263 |
| 17 | Yao H., Zheng G., Hsu P. C., Kong D., Cha J. J., Li W., Seh Z. W., Mcdowell M. T., Yan K., Liang Z., Narasimhan V. K., Cui Y., Nat. Commun., 2014, 5(1), 1—9 |
| 18 | Gu X., Tong C. J., Wen B., Liu L. M., Lai C., Zhang S., Electrochim. Acta, 2016, 196, 369—376 |
| 19 | Seh Z. W., Yu J. H., Li W., Hsu P. C., Wang H., Sun Y., Yao H., Zhang Q., Cui Y., Nat. Commun., 2014, 5(1), 1—8 |
| 20 | Deng D. R., Xue F., Jia Y. J., Ye J. C., Bai C. D., Zheng M. S., Dong Q. F., ACS Nano, 2017, 11(6), 6031—6039 |
| 21 | Geng C. N., Hua W. X., Ling G. W., Tao Y., Zhang C., Yang Q. H., Chem. J. Chinese Universities, 2021, 42(5), 1331—1339 |
| 耿传楠, 化五星, 凌国维, 陶莹, 张辰, 杨全红. 高等学校化学学报, 2021, 42(5), 1331—1339 | |
| 22 | Maurer F., Jelic J., Wang J., Gänzler A., Dolcet P., Wöll C., Wang Y., Studt F., Casapu M., Grunwaldt J. D., Nat. Catal., 2020, 3(10), 824—833 |
| 23 | Kim Y., Collinge G., Lee M. S., Khivantsev K., Cho S. J., Glezakou V. A., Rousseau R., Szanyi J., Kwak J. H., Angew. Chem. Int. Ed. Engl., 2021, 60(42), 22769—22775 |
| 24 | Min Y., Zhou X., Chen J. J., Chen W., Zhou F., Wang Z., Yang J., Xiong C., Wang Y., Li F., Yu H. Q., Wu Y., Nat. Commun., 2021, 12(1), 1—10 |
| 25 | Zhuang Z., Kang Q., Wang D., Li Y., Nano Res., 2020, 13(7), 1856—1866 |
| 26 | Fang L., Feng Z., Cheng L., Winans R. E., Li T., Small Methods, 2020, 4(10), 2000315 |
| 27 | Xu K., Zhu M., Zhu L., Li D., Zhang W., Huang T., Zhu Y., Qian Y., J. Mater. Chem. A, 2020, 8(15), 7100—7108 |
| 28 | Li B. Q., Kong L., Zhao C. X., Jin Q., Chen X., Peng H. J., Qin J. L., Chen J. X., Yuan H., Zhang Q., Huang J. Q., InfoMat, 2019, 1(4), 533—541 |
| 29 | Wang J., Jia L., Lin H., Zhang Y., ChemSusChem, 2020, 13(13), 3404—3411 |
| 30 | Wang F., Li J., Zhao J., Yang Y., Su C., Zhong Y. L., Yang Q. H., Lu J., ACS Mater. Lett., 2020, 2(11), 1450—1463 |
| 31 | Wang J., Ding B., Lu X., Nara H., Sugahara Y., Yamauchi Y., Adv. Mater. Interfaces, 2021, 8(8), 2002159 |
| 32 | Wang J., Jia L., Zhong J., Xiao Q., Wang C., Zang K., Liu H., Zheng H., Luo J., Yang J., Fan H., Duan W., Wu Y., Lin H., Zhang Y., Energy Stor. Mater., 2019, 18, 246—252 |
| 33 | Du Z., Chen X., Hu W., Chuang C., Xie S., Hu A., Yan W., Kong X., Wu X., Ji H., Wan L. J., J. Am. Chem. Soc., 2019, 141(9), 3977—3985 |
| 34 | Zhang L., Liu D., Muhammad Z., Wan F., Xie W., Wang Y., Song L., Niu Z., Chen J., Adv. Mater., 2019, 31(40), 1903955 |
| 35 | Xia H., Qu G., Yin H., Zhang J., J. Mater. Chem. A, 2020, 8(31), 15358—15372 |
| 36 | Liu Z., Zhou L., Ge Q., Chen R., Ni M., Utetiwabo W., Zhang X., Yang W., ACS Appl. Mater. Interfaces, 2018, 10(23), 19311—19317 |
| 37 | Xie J., Li B. Q., Peng H. J., Song Y. W., Zhao M., Chen X., Zhang Q., Huang J. Q., Adv. Mater., 2019, 31(43), 1903813 |
| [1] | 楚宇逸, 兰畅, 罗二桂, 刘长鹏, 葛君杰, 邢巍. 单原子铈对弱芬顿效应活性位点氧还原稳定性的提升[J]. 高等学校化学学报, 2022, 43(9): 20220294. |
| [2] | 杨静怡, 李庆贺, 乔波涛. 铱单原子和纳米粒子在N2O分解反应中的协同催化[J]. 高等学校化学学报, 2022, 43(9): 20220388. |
| [3] | 林高鑫, 王家成. 单原子掺杂二硫化钼析氢催化的进展和展望[J]. 高等学校化学学报, 2022, 43(9): 20220321. |
| [4] | 任诗杰, 谯思聪, 刘崇静, 张文华, 宋礼. 铂单原子催化剂同步辐射X射线吸收谱的研究进展[J]. 高等学校化学学报, 2022, 43(9): 20220466. |
| [5] | 秦永吉, 罗俊. 单原子催化剂在CO2转化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220300. |
| [6] | 林治, 彭志明, 贺韦清, 沈少华. 单原子与团簇光催化: 竞争与协同[J]. 高等学校化学学报, 2022, 43(9): 20220312. |
| [7] | 滕镇远, 张启涛, 苏陈良. 聚合物单原子光催化剂的载流子分离和表面反应机制[J]. 高等学校化学学报, 2022, 43(9): 20220325. |
| [8] | 王茹玥, 魏呵呵, 黄凯, 伍晖. 单原子材料的冷冻合成[J]. 高等学校化学学报, 2022, 43(9): 20220428. |
| [9] | 韩付超, 李福进, 陈良, 贺磊义, 姜玉南, 徐守冬, 张鼎, 其鲁. CoSe2/C复合电催化材料修饰隔膜对高载量锂硫电池性能的影响[J]. 高等学校化学学报, 2022, 43(8): 20220163. |
| [10] | 赵润瑶, 纪桂鹏, 刘志敏. 吡咯氮配位单原子铜催化剂的电催化二氧化碳还原性能[J]. 高等学校化学学报, 2022, 43(7): 20220272. |
| [11] | 谷雨, 奚宝娟, 李江潇, 熊胜林. 单原子催化剂在氧还原反应中的分子级调控[J]. 高等学校化学学报, 2022, 43(5): 20220036. |
| [12] | 吴俊, 何观朝, 费慧龙. 自支撑单原子膜电极在能源电催化中的应用[J]. 高等学校化学学报, 2022, 43(5): 20220051. |
| [13] | 庄嘉豪, 王定胜. 单原子催化的关键进展与未来挑战[J]. 高等学校化学学报, 2022, 43(5): 20220043. |
| [14] | 张宏伟, 陈雯, 赵美淇, 马超, 韩云虎. 单原子催化剂在电化学中的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220129. |
| [15] | 夏天, 万家炜, 于然波. 异原子配位结构碳基单原子电催化剂结构与性能相关性的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220162. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||