

高等学校化学学报 ›› 2022, Vol. 43 ›› Issue (10): 20220264.doi: 10.7503/cjcu20220264
许文哲,张皓
收稿日期:2022-04-19
出版日期:2022-10-10
发布日期:2022-05-18
基金资助:Received:2022-04-19
Online:2022-10-10
Published:2022-05-18
Contact:
ZHANG Hao
E-mail:hao_zhang@jlu.edu.cn
Supported by:摘要:
纳米沉淀法是目前制备纳米药物的主要途径, 是指通过向药物的良溶剂中引入不良溶剂产生过饱和体系, 进而形成纳米尺度药物颗粒的方法. 该方法操控灵活, 能够大范围地选择药物分子、 溶剂、 载体、 表面活性剂及其它赋形剂, 实现对纳米药物成核及生长过程的调控. π-π堆积和疏水相互作用等分子间弱相互作用能够主导纳米药物成核, 从而用于制备高生物安全性的无载体纳米药物(CFNs). 目前超分子自组装在成核过程中的具体作用、 协同效应及调控方法尚缺少归纳总结. 根据纳米沉淀法的成核理论, 本文对超分子相互作用在成核过程中的重要贡献进行了诠释; 基于目前单药自组装CFNs的进展, 对多药共组装CFNs的优势进行了强调; 并将超分子相互作用主导成核的概念拓展到通过金属离子螯合形成的CFNs. 从理论上阐明了超分子相互作用在纳米药物成核过程中的主导作用, 将极大促进以高生物安全、 多功能及以联合治疗为标志的下一代CFNs的发展.
中图分类号:
TrendMD:
许文哲, 张皓. 超分子相互作用主导的纳米药物成核. 高等学校化学学报, 2022, 43(10): 20220264.
XU Wenzhe, ZHANG Hao. Supramolecular Interactions-mediated Nanodrug Nucleation. Chem. J. Chinese Universities, 2022, 43(10): 20220264.
| Abbreviation | Full name | Abbreviation | Full name | Abbreviation | Full name |
|---|---|---|---|---|---|
| AAs | Amino acids | CPT | Camptothecin | ICG | Indocyanine green |
| ATO | Atovaquone | Cur | Curcumin | Ir | Irinotecan |
| BBR | Berberine | DOX | Doxorubicin | MTX | Methotrexate |
| BC | Betulonic acid | FA | Folic acid | PAAs | Polyamino acids |
| CA | Cinnamic acid | FZ | Phenylsulfonylfuroxan | PTX | Paclitaxel |
| Cb | Chlorambucil | GA | Gambogic acid | UA | Ursolic acid |
| Ce6 | Chlorin e6 | HCPT | 10?Hydroxycamptothecin |
Table 1 Abbreviations and full names of the drugs in this review
| Abbreviation | Full name | Abbreviation | Full name | Abbreviation | Full name |
|---|---|---|---|---|---|
| AAs | Amino acids | CPT | Camptothecin | ICG | Indocyanine green |
| ATO | Atovaquone | Cur | Curcumin | Ir | Irinotecan |
| BBR | Berberine | DOX | Doxorubicin | MTX | Methotrexate |
| BC | Betulonic acid | FA | Folic acid | PAAs | Polyamino acids |
| CA | Cinnamic acid | FZ | Phenylsulfonylfuroxan | PTX | Paclitaxel |
| Cb | Chlorambucil | GA | Gambogic acid | UA | Ursolic acid |
| Ce6 | Chlorin e6 | HCPT | 10?Hydroxycamptothecin |
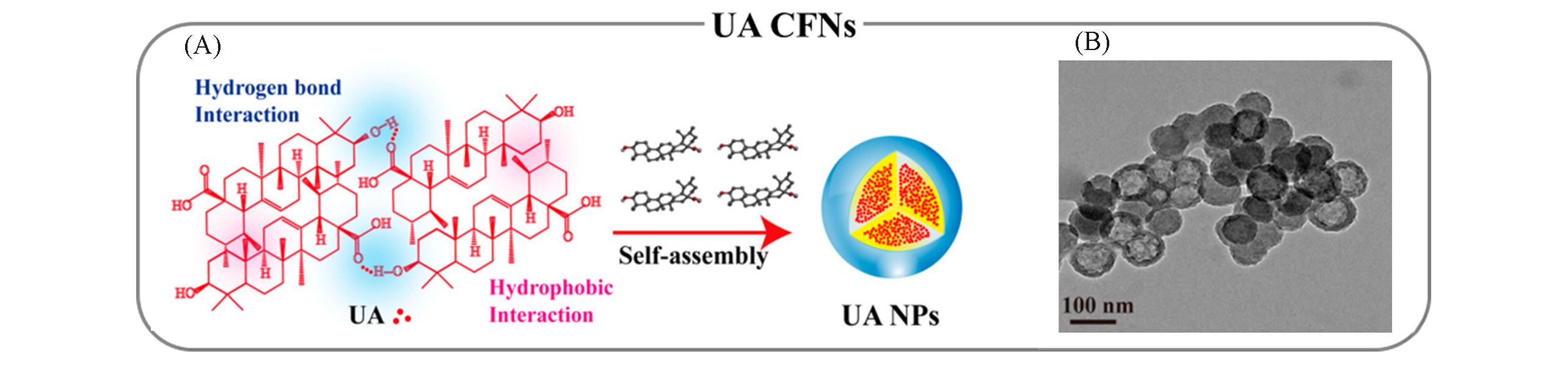
Fig.2 Representative natural drug?based self?assembled CFNs through predominant supramolecular interactions[34](A) Schematic illustration of the nanoprecipitation process of UA CFNs and the involved supramolecular interactions; (B) transmission electron microscope(TEM) image of UA CFNs. Copyright 2018, American Chemical Society.
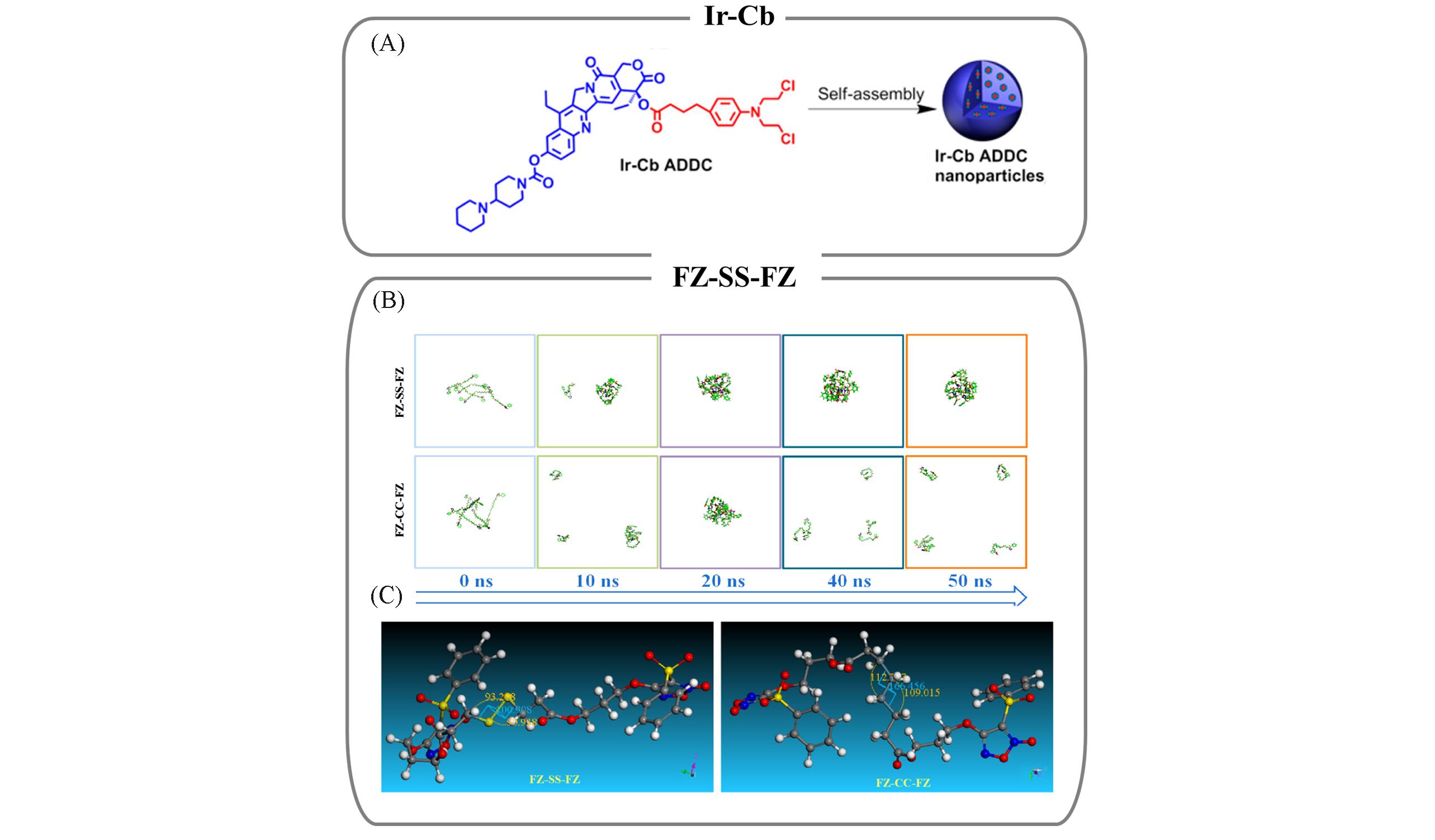
Fig.3 CFNs from drugs conjugated with complementary hydrophobic drugs and representative mechanism(A) Constitutional formula of Ir-Cb prodrugs and the preparation of Ir-Cb CFNs[40]; (B) self-assembly evolution of FZ-SS-FZ and FZ-CC-FZ CFNs studied by MD simulations; (C) Materials Studio simulation results of bond angles and dihedral angles of FZ-SS-FZ and FZ-CC-FZ prodrugs. When the bond angle and dihedral angle are closer to 90°, the CFNs is more stable. The bond angles of —SS— and —CC— are 93.2°/90.9° and 112.7°/109.0°, respectively. The dihedral angles of C—SS—C and C—CC—C are 100.9° and 166.4°, respectively[43].(A) Copyright 2014, American Chemical Society; (B, C) Copyright 2021, American Chemical Society.
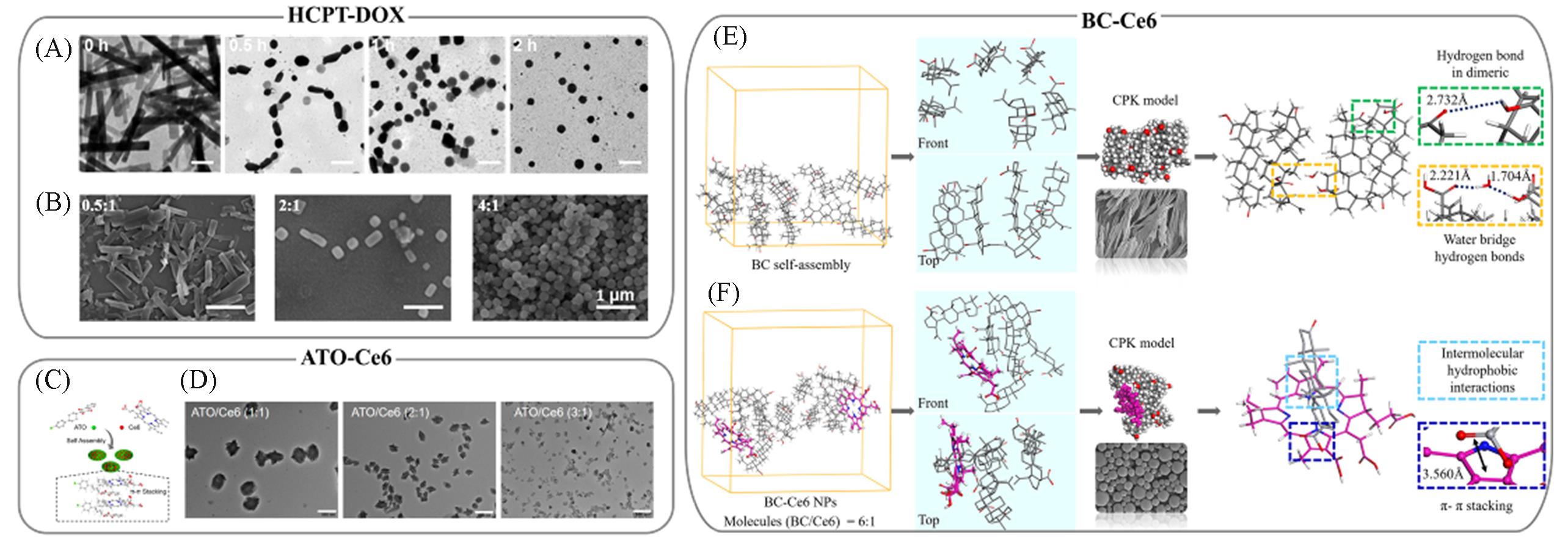
Fig.5 Representative π?system multidrug co?assembled CFNs with controllable size and morphology(A) Morphology evolution of HCPT-DOX CFNs characterized by TEM at different time points, the scale bar is 500 nm; (B) TEM images of HCPT-DOX CFNs at different DOX-to-HCPT molar ratios[54]; (C) schematic illustration of co-assembly between ATO and Ce6 through π?π stacking; (D) TEM images of ATO-Ce6 CFNs at different ATO-to-Ce6 molar ratios, the scale bar is 500 nm[56]; MD simulation results of BC self-assembled(E) and BC-Ce6 co-assembled CFNs(F)[29].(A, B) Copyright 2015, American Chemical Society; (C, D) Copyright 2020, American Chemical Society; (E, F) Copyright 2020, American Chemical Society.
| CFNs | Hydrodynamic diameter/nm | Zeta potential/mV | Stability duration/d | Ref. |
|---|---|---|---|---|
| Coniferyl ferulate?Ce6 | 159.1 | Negative | 5 | [ |
| NLG919?Ce6 | — | Negative | 5 | [ |
| α?Tocopherol succinate?DOX | 199.9 | Negative | 7 | [ |
| ICG?PTX | 112±1.06 | -34.5 | 7(buffer solution) | [ |
| ICG@UA/PTX | ca. 130.8 | -30.8 | 8 | [ |
| Ce6?gambogic acid | 142.2±14.9 | -27.5±4.6 | 9 | [ |
| ATO?Ce6 | 193.5 | -23.31 | 9(water, PBS, or cell culture medium) | [ |
| BC?Ce6 | 231.4±8.4 | -5.2±2.23 | 10 | [ |
| UA?lactobionic acid?ICG | 116.4±2.4 | -30.6±1.8 | 30 | [ |
| PTX?ICG | 140±1.4 | -36±2.2 | Over 30 | [ |
Table 2 Summary of hydrodynamic diameter, zeta potential, and stability duration of CFNs
| CFNs | Hydrodynamic diameter/nm | Zeta potential/mV | Stability duration/d | Ref. |
|---|---|---|---|---|
| Coniferyl ferulate?Ce6 | 159.1 | Negative | 5 | [ |
| NLG919?Ce6 | — | Negative | 5 | [ |
| α?Tocopherol succinate?DOX | 199.9 | Negative | 7 | [ |
| ICG?PTX | 112±1.06 | -34.5 | 7(buffer solution) | [ |
| ICG@UA/PTX | ca. 130.8 | -30.8 | 8 | [ |
| Ce6?gambogic acid | 142.2±14.9 | -27.5±4.6 | 9 | [ |
| ATO?Ce6 | 193.5 | -23.31 | 9(water, PBS, or cell culture medium) | [ |
| BC?Ce6 | 231.4±8.4 | -5.2±2.23 | 10 | [ |
| UA?lactobionic acid?ICG | 116.4±2.4 | -30.6±1.8 | 30 | [ |
| PTX?ICG | 140±1.4 | -36±2.2 | Over 30 | [ |
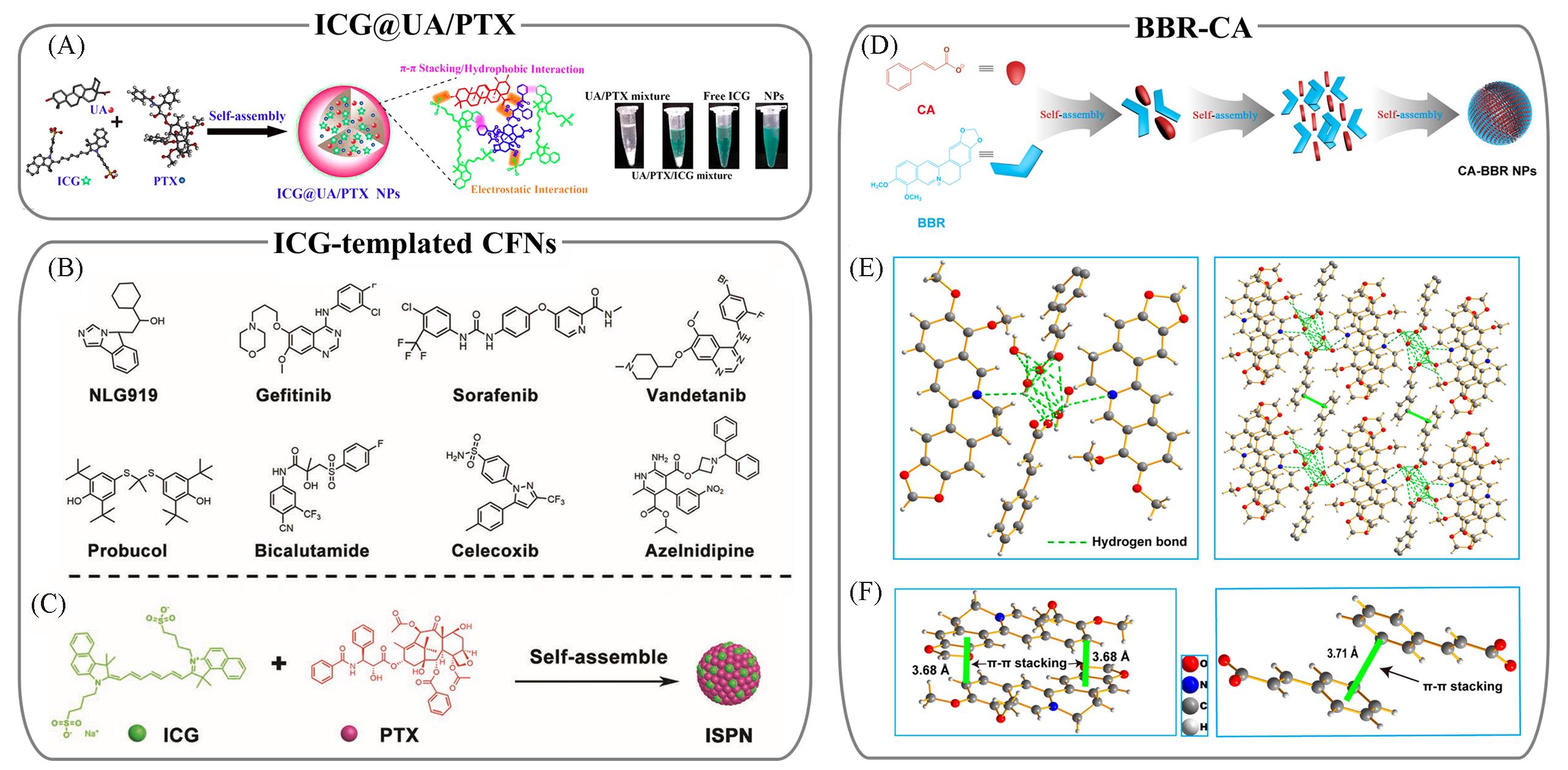
Fig.6 Representative π?system multidrug co?assembled CFNs characterized with function integration and type enrichment(A) Schematic illustration of co-assembled ICG@UA/PTX CFNs through π?π stacking, hydrophobic interaction and electrostatic interaction, and optical images of the corresponding constitutions[60]; (B) a library of small molecule drugs for ICG-templated CFNs; (C) schematic illustration of co-assembled ICG/PTX CFNs by adding PTX-contained organic solution into ICG-predissolved aqueous media[62]; (D) self-assembly process of BBR-CA CFNs; (E) formation of BBR-CA nucleus by hydrogen bonding(left) and 3D stacking of BBR-CA nucleus through π?π stacking (right); (F) inter-BBR(left) and inter-CA(right) π?π stacking[72].(A) Copyright 2017, American Chemical Society; (B, C) Copyright 2019, Wiley Online Library; (D—F) Copyright 2019, American Chemical Society.
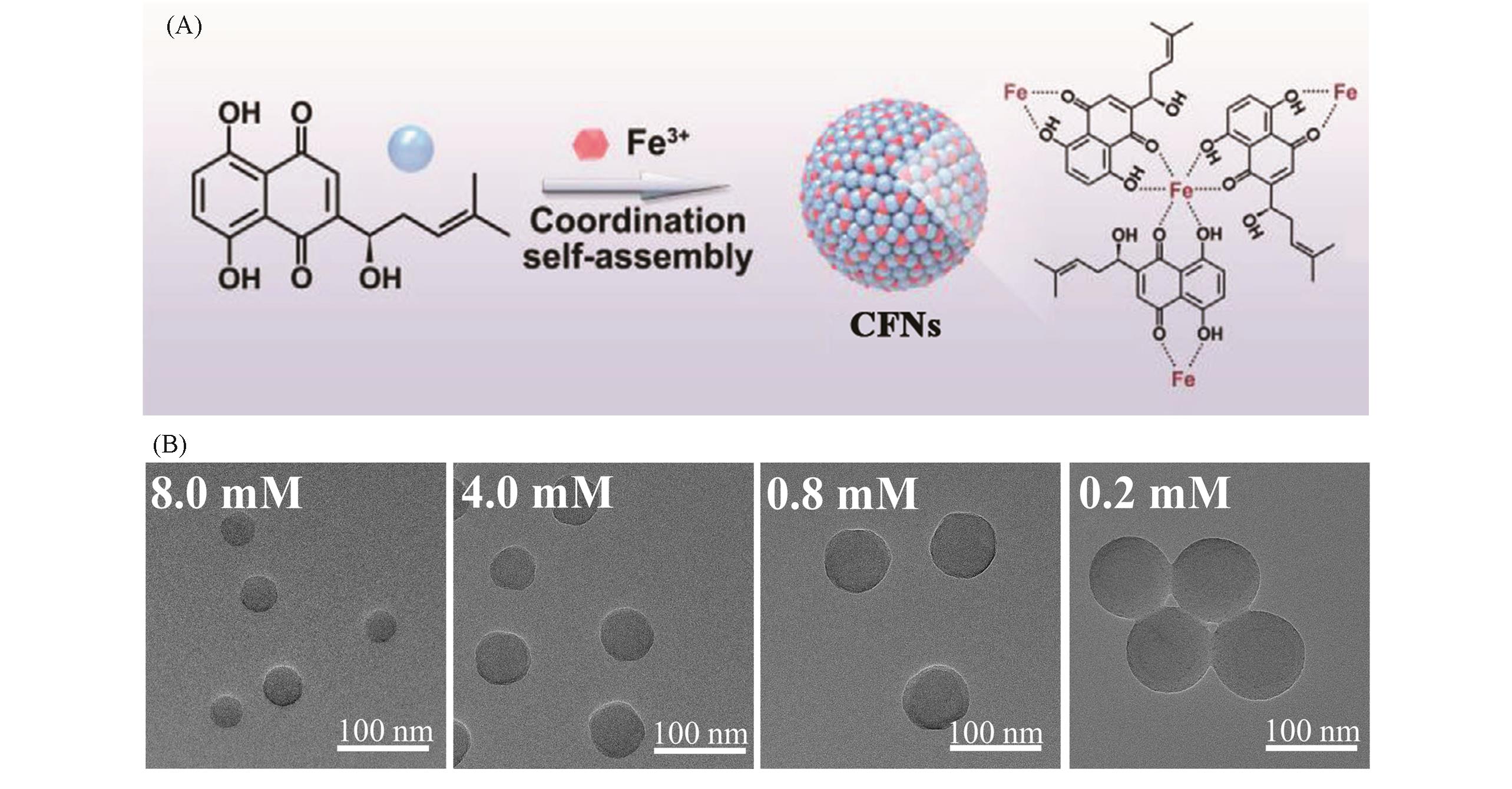
Fig.7 Schematic illustration of the formation of Fe(Ⅲ)?shikonin CFNs driven by coordination interaction(A) and TEM images of Fe(Ⅲ)?shikonin CFNs prepared with different Fe(Ⅲ) concentrations(B)[76]Copyright 2021, Wiley Online Library.
| 1 | Salvioni L., Rizzuto M. A., Bertolini J. A., Pandolfi L., Colombo M., Prosperi D., Cancers, 2019, 11, 1855 |
| 2 | US Food and Drug Administration, https://www.fda.gov/regulatory⁃information/search⁃fda⁃guidance⁃documents/considering⁃whether⁃fda⁃regulated⁃product⁃involves⁃application⁃nanotechnology |
| 3 | Zhao Y., Cao W. Q., Liu Y., Chem. J. Chinese Universities, 2020, 41(5), 909—923 |
| 4 | Sun Q. R., Zhao N., Liu S. W., Xin H., Zhang H., Zhang L. N., Chem. J. Chinese Universities, 2021, 42(10),3225—3232 |
| 5 | Edis Z., Wang J., Waqas M. K., Ijaz M., Ijaz M., Int. J. Nanomedicine, 2021, 16, 1313—1330 |
| 6 | Barenholz Y., J. Control. Release, 2012, 160, 117—134 |
| 7 | Shi J., Kantoff P. W., Wooster R., Farokhzad O. C., Nat. Rev. Cancer, 2017, 17, 20—37 |
| 8 | Qin S. Y., Zhang A. Q., Cheng S. X., Rong L., Zhang X. Z., Biomaterials, 2017, 112, 234—247 |
| 9 | Mei H., Cai S., Huang D., Gao H., Cao J., He B., Bioact. Mater., 2022, 8, 220—240 |
| 10 | Karaosmanoglu S., Zhou M., Shi B., Zhang X., Williams G. R., Chen X., J. Control. Release, 2021, 329, 805—832 |
| 11 | Jiang S., Fu Y., Zhang X., Yu T., Lu B., Du J., Front. Bioeng. Biotechnol., 2021, 9, 799806 |
| 12 | Ge W., Wang L., Zhang J., Ou C., Si W., Wang W., Zhang Q., Dong X., Adv. Mater. Interfaces, 2020, 8, 2001602 |
| 13 | Qiao Y., Wei Z., Qin T., Song R., Yu Z., Yuan Q., Du J., Zeng Q., Zong L., Duan S., Pu X., Chin. Chem. Lett., 2021, 32, 2877—2881 |
| 14 | Zhang J., Li Y., An F. F., Zhang X., Chen X., Lee C. S., Nano Lett., 2015, 15, 313—318 |
| 15 | Malamatari M., Taylor K. M. G., Malamataris S., Douroumis D., Kachrimanis K., Drug Discov. Today, 2018, 23, 534—547 |
| 16 | D'Addio S. M., Prud'homme R. K., Adv. Drug. Deliv. Rev., 2011, 63, 417—426 |
| 17 | Lepeltier E., Bourgaux C., Couvreur P., Adv. Drug. Deliv. Rev., 2014, 71, 86—97 |
| 18 | Martinez R. C. J., Tarhini M., Badri W., Miladi K., Greige⁃Gerges H., Nazari Q. A., Galindo R. S. A., Roman R. A., Fessi H., Elaissari A., Int. J. Pharm., 2017, 532, 66—81 |
| 19 | Prabagar B., Yang Y., Shi Z., Chem. Soc. Rev., 2021, 50, 11249—11269 |
| 20 | Li G., Sun B., Li Y., Luo C., He Z., Sun J., Small, 2021, 17, e2101460 |
| 21 | Zhang Y., Fang F., Li L., Zhang J., ACS Biomater. Sci. Eng., 2020, 6, 4816—4833 |
| 22 | Zhang X., Li N., Zhang S., Sun B., Chen Q., He Z., Luo C., Sun J., Med. Res. Rev., 2020, 40, 1754—1775 |
| 23 | Azadbakht R., Hakimi M., Khanabadi J., Spectrochim. Acta. A. Mol. Biomol. Spectrosc., 2021, 250, 119236 |
| 24 | Lee J. H., Kim T. M., Choi I. G., Choi J. W., Nanomaterials, 2021, 11, 1790 |
| 25 | Mora⁃Huertas C. E., Fessi H., Elaissari A., Adv. Colloid Interface Sci., 2011, 163, 90—122 |
| 26 | Beck⁃Broichsitter M., Colloids and Surfaces A, 2021, 625, 126928 |
| 27 | Li X., Shen J., Wang B., Feng X., Mao Z., Sui X., ACS Sustain. Chem. Eng., 2021, 9, 5470—5480 |
| 28 | Nikoubashman A., Lee V. E., Sosa C., Prud'homme R. K., Priestley R. D., Panagiotopoulos A. Z., ACS Nano, 2016, 10, 1425—1433 |
| 29 | Cheng J., Zhao H., Wang J., Han Y., Yang X., ACS Appl. Mater. Interfaces, 2020, 12, 43488—43500 |
| 30 | Yan X., Bernard J., Ganachaud F., Adv. Colloid Interface Sci., 2021, 294, 102474 |
| 31 | Hicham F., Packiarajan M., Kenrick L. V., Debra M. S., Klaas H., Karl V. W., Joseph G. S., J. Am. Chem. Soc., 2001, 123, 3854—3855 |
| 32 | Chen Z., Farag M. A., Zhong Z., Zhang C., Yang Y., Wang S., Wang Y., Adv. Drug Deliv. Rev., 2021, 176, 113870 |
| 33 | Shao J., Fang Y., Zhao R., Chen F., Yang M., Jiang J., Chen Z., Yuan X., Jia L., Asian J. Pharm. Sci., 2020, 15, 685—700 |
| 34 | Fan L., Zhang B., Xu A., Shen Z., Guo Y., Zhao R., Yao H., Shao J. W., Mol. Pharm., 2018, 15, 2466—2478 |
| 35 | Li W., Yang Y., Wang C., Liu Z., Zhang X., An F., Diao X., Hao X., Zhang X., Chem. Commun.(Camb.), 2012, 48, 8120—8122 |
| 36 | Zhang J., Li S., An F. F., Liu J., Jin S., Zhang J. C., Wang P. C., Zhang X., Lee C. S., Liang X. J., Nanoscale, 2015, 7, 13503—13510 |
| 37 | He D., Zhang W., Deng H., Huo S., Wang Y. F., Gong N., Deng L., Liang X. J., Dong A., Chem. Commun.(Camb.), 2016, 52, 14145—14148 |
| 38 | Hou M., Xue P., Gao Y. E., Ma X., Bai S., Kang Y., Xu Z., Biomater. Sci., 2017, 5, 1889—1897 |
| 39 | Huang L., Wan J., Wu H., Chen X., Bian Q., Shi L., Jiang X., Yuan A., Gao J., Wang H., Nano Today, 2021, 36, 101030 |
| 40 | Huang P., Wang D., Su Y., Huang W., Zhou Y., Cui D., Zhu X., Yan D., J. Am. Chem. Soc., 2014, 136, 11748—11756 |
| 41 | Zhang T., Huang P., Shi L., Su Y., Zhou L., Zhu X., Yan D., Mol. Pharm., 2015, 12, 2328—2336 |
| 42 | Cheng C., Sui B., Wang M., Hu X., Shi S., Xu P., Adv. Healthc. Mater., 2020, 9, e2001128 |
| 43 | Gu G., Chen C., Zhang S., Yin B., Wang J., ACS Appl. Mater. Interfaces, 2021, 13, 50682—50694 |
| 44 | Onoue S., Yamada S., Chan H. K., Int. J. Nanomedicine, 2014, 9, 1025—1037 |
| 45 | Faul C. F. J., Antonietti M., Adv. Mater., 2003, 15, 673—683 |
| 46 | Wang Z., Wang L., Liu S., Zhang M., Li Y., Rong L., Liu Y., Zhang H., Nanoscale, 2022, 14, 2186—2198 |
| 47 | Xu Z., Hou M., Shi X., Gao Y. E., Xue P., Liu S., Kang Y., Biomater. Sci., 2017, 5, 444—454 |
| 48 | Xu Z., Shi X., Hou M., Xue P., Gao Y. E., Liu S., Kang Y., Colloids Surf. B Biointerfaces, 2017, 150, 50—58 |
| 49 | Shen Y., Jin E., Zhang B., Murphy C. J., Sui M., Zhao J., Wang J., Tang J., Fan M., Kirk E. V., Murdoch W. J., J. Am. Chem. Soc., 2010, 132, 4259—4265 |
| 50 | Sun T., Lin W., Zhang W., Xie Z., Chem Asian J., 2016, 11, 3174—3177 |
| 51 | Kinnear C., Moore T. L., Rodriguez⁃Lorenzo L., Rothen⁃Rutishauser B., Petri⁃Fink A., Chem. Rev., 2017, 117, 11476—11521 |
| 52 | Chen F., Zhao Y., Pan Y., Xue X., Zhang X., Kumar A., Liang X. J., Mol. Pharm., 2015, 12, 2237—2244 |
| 53 | Li Y., Lin J., Cai Z., Wang P., Luo Q., Yao C., Zhang Y., Hou Z., Liu J., Liu X., J. Control. Release, 2020, 321, 222—235 |
| 54 | Zhao Y., Chen F., Pan Y., Li Z., Xue X., Okeke C. I., Wang Y., Li C., Peng L., Wang P. C., Ma X., Liang X. J., ACS Appl. Mater. Interfaces, 2015, 7, 19295—19305 |
| 55 | Wen Y., Zhang W., Gong N., Wang Y. F., Guo H. B., Guo W., Wang P. C., Liang X. J., Nanoscale, 2017, 9, 14347—14356 |
| 56 | Zhao L. P., Zheng R. R., Chen H. Q., Liu L. S., Zhao X. Y., Liu H. H., Qiu X. Z., Yu X. Y., Cheng H., Li S. Y., Nano Lett., 2020, 20, 2062—2071 |
| 57 | Li X., Kong R., Li Y., Huang J., Zhou X., Li S., Cheng H., Chem. Commun.(Camb.), 2022, 58, 3917—3920 |
| 58 | Zhao L. P., Zheng R. R., Huang J. Q., Chen X. Y., Deng F. A., Liu Y. B., Huang C. Y., Yu X. Y., Cheng H., Li S. Y., ACS Nano, 2020, 14, 17100—17113 |
| 59 | Zheng R. R., Zhao L. P., Liu L. S., Deng F. A., Chen X. Y., Jiang X. Y., Wang C., Yu X. Y., Cheng H., Li S. Y., Biomater. Sci., 2021, 9, 3445—3452 |
| 60 | Guo Y., Jiang K., Shen Z., Zheng G., Fan L., Zhao R., Shao J., ACS Appl. Mater. Interfaces, 2017, 9, 43508—43519 |
| 61 | Lan J. S., Liu L., Zeng R. F., Qin Y. H., Hou J. W., Xie S. S., Yue S., Yang J., Ho R. J. Y., Ding Y., Zhang T., Chem. Eng. J., 2021, 407, 127212 |
| 62 | Feng B., Niu Z., Hou B., Zhou L., Li Y., Yu H., Adv. Funct. Mater., 2019, 30, 1906605 |
| 63 | Zhao R., Zheng G., Fan L., Shen Z., Jiang K., Guo Y., Shao J. W., Acta Biomater., 2018, 70, 197—210 |
| 64 | Lin J., Li C., Guo Y., Zou J., Wu P., Liao Y., Zhang B., Le J., Zhao R., Shao J. W., J. Mater. Chem. B, 2019, 7, 6914—6923 |
| 65 | Zhang Z., Wang Y., Ma Q., Zhang S., Liu H., Zhao B., Liu R., Wang W., Du B., Zhong Y., Kong D., Chem. Eng. J., 2021, 406, 126801 |
| 66 | Lin H., Chen Y., Shi J., Chem. Soc. Rev., 2018, 47, 1938—1958 |
| 67 | Yu C., Zhou M., Zhang X., Wei W., Chen X., Zhang X., Nanoscale, 2015, 7, 5683—5690 |
| 68 | Jiang K., Han L., Guo Y., Zheng G., Fan L., Shen Z., Zhao R., Shao J., J. Mater. Chem. B, 2017, 5, 9121—9129 |
| 69 | Liu C., Liu Q., Chen L., Li M., Yin J., Zhu X., Chen D., Adv. Healthc. Mater., 2020, 9, e2000899 |
| 70 | Wang X. L., Wang Z. X., Chem. J. Chinese Universities, 2018, 39(10), 2185—2191 |
| 71 | Yang B., Wang K., Zhang D., Sun B., Ji B., Wei L., Li Z., Wang M., Zhang X., Zhang H., Kan Q., Luo C., Wang Y., He Z., Sun J., Biomater. Sci., 2018, 6, 2965—2975 |
| 72 | Huang X., Wang P., Li T., Tian X., Guo W., Xu B., Huang G., Cai D., Zhou F., Zhang H., Lei H., ACS Appl. Mater. Interfaces, 2020, 12, 227—237 |
| 73 | Shan L., Gao G., Wang W., Tang W., Wang Z., Yang Z., Fan W., Zhu G., Zhai K., Jacobson O., Dai Y., Chen X., Biomaterials, 2019, 210, 62—69 |
| 74 | Liu T., Feng J., Zhang X. Z., Journal of Functional Polymers, 2019, 32, 421—433 |
| 75 | Xu C., Wang Y., Yu H., Tian H., Chen X., ACS Nano, 2018, 12, 8255—8265 |
| 76 | Feng W., Shi W., Liu S., Liu H., Liu Y., Ge P., Zhang H., Adv. Healthc. Mater., 2022, 11, e2101926 |
| 77 | Yang G. G., Zhou D. J., Pan Z. Y., Yang J., Zhang D. Y., Cao Q., Ji L. N., Mao Z. W., Biomaterials, 2019, 216, 119280 |
| 78 | Pucci C., Martinelli C., De Pasquale D., Battaglini M., di Leo N., Degl'Innocenti A., Belenli Gumus M., Drago F., Ciofani G., ACS Appl. Mater. Interfaces, 2022, 14, 15927—15941 |
| 79 | Wang Y., Xu C., Meng M., Lin L., Hu Y., Hao K., Sheng S., Zhang S., Wu J., Liu F., Jiang X., Tian H., Chen X., Nano Today, 2021, 40, 101266 |
| 80 | Tu L., Fan Z., Zhu F., Zhang Q., Zeng S., Chen Z., Ren L., Hou Z., Ye S., Li Y., J. Mater. Chem. B, 2020, 8, 5667—5681 |
| 81 | Zhao L., Zheng R., Liu L., Chen X., Guan R., Yang N., Chen A., Yu X., Cheng H., Li S., Biomaterials, 2021, 275, 120970 |
| 82 | Liu J., Zuo W., Jin Q., Liu C., Liu N., Tian H., Zhu X., Mater. Sci. Eng. C Mater. Biol. Appl., 2021, 129, 112351 |
| 83 | Thi T. T. H., Suys E. J. A., Lee J. S., Nguyen D. H., Park K. D., Truong N. P., Vaccines(Basel), 2021, 9, 359 |
| 84 | Scalise M., Pochini L., Galluccio M., Console L., Indiveri C., Front. Oncol., 2017, 7, 306 |
| 85 | Li Y., Jiang Y., Zheng Z., Du N., Guan S., Guo W., Tang X., Cui J., Zhang L., Liu K., Yu Q., Gan Z., Adv. Mater., 2022, 34, e2110490 |
| [1] | 丁鑫, 师红东, 刘扬中. 以人血清白蛋白为载体的Ru(Ⅲ)和全反式维甲酸共运输纳米药物的构建及抗肿瘤转移作用[J]. 高等学校化学学报, 2021, 42(10): 3040. |
| [2] | 赵宇, 曹琬晴, 刘阳. 聚合物纳米药物载体的研究进展[J]. 高等学校化学学报, 2020, 41(5): 909. |
| [3] | 李林, 许鑫汝, 李英奇, 张彩凤. 靶向纳米钻石-甲氨蝶呤药物体系制备及与MCF-7细胞作用[J]. 高等学校化学学报, 2019, 40(9): 1998. |
| [4] | 朱钦富, 胡可珍, 李小杰, 陈明清. pH响应性树枝状聚合物-金纳米粒子复合药物载体的制备[J]. 高等学校化学学报, 2019, 40(5): 1065. |
| [5] | 杨兵, 马於光, 沈家骢. π-共轭分子堆积、光电性能与超分子调控[J]. 高等学校化学学报, 2008, 29(12): 2643. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
