

高等学校化学学报 ›› 2022, Vol. 43 ›› Issue (12): 20220556.doi: 10.7503/cjcu20220556
收稿日期:2022-08-20
出版日期:2022-12-10
发布日期:2022-10-10
通讯作者:
刘定斌
E-mail:liudb@nankai.edu.cn
基金资助:
YE Zhuo1,2, JI Moxuan2, LIU Dingbin2( )
)
Received:2022-08-20
Online:2022-12-10
Published:2022-10-10
Contact:
LIU Dingbin
E-mail:liudb@nankai.edu.cn
Supported by:摘要:
心血管疾病(Cardiovascular disease, CVD)是全球疾病致死的主要原因之一. 动脉粥样硬化(Atherosclerosis, AS)是引发各种CVD的首要危险因素, 其发生发展通常经历持续的慢性炎症过程. 因此, 及时高效地检测AS, 对于早期评估、 诊断和治疗CVD具有重要临床意义. 光学探针成像拥有极高的灵敏度和空间分辨率以及超快的信号采集处理速度, 被广泛应用于生物医学检测与成像. 本文综合评述了6种常见用于AS成像的光学探针, 涉及小分子荧光探针、 聚集诱导发光(Aggregate-induced emission, AIE)纳米探针、 量子点探针、 上转换纳米探针、 光声探针和多模态探针等; 并对各种探针的优缺点进行了分析比较. 在此基础上, 展望了光学探针在AS成像领域的发展前景, 并提出了相应建议.
中图分类号:
TrendMD:
叶卓, 吉墨轩, 刘定斌. 动脉粥样硬化光学成像探针研究进展. 高等学校化学学报, 2022, 43(12): 20220556.
YE Zhuo, JI Moxuan, LIU Dingbin. Advances of Optical Probes in Atherosclerosis Imaging. Chem. J. Chinese Universities, 2022, 43(12): 20220556.
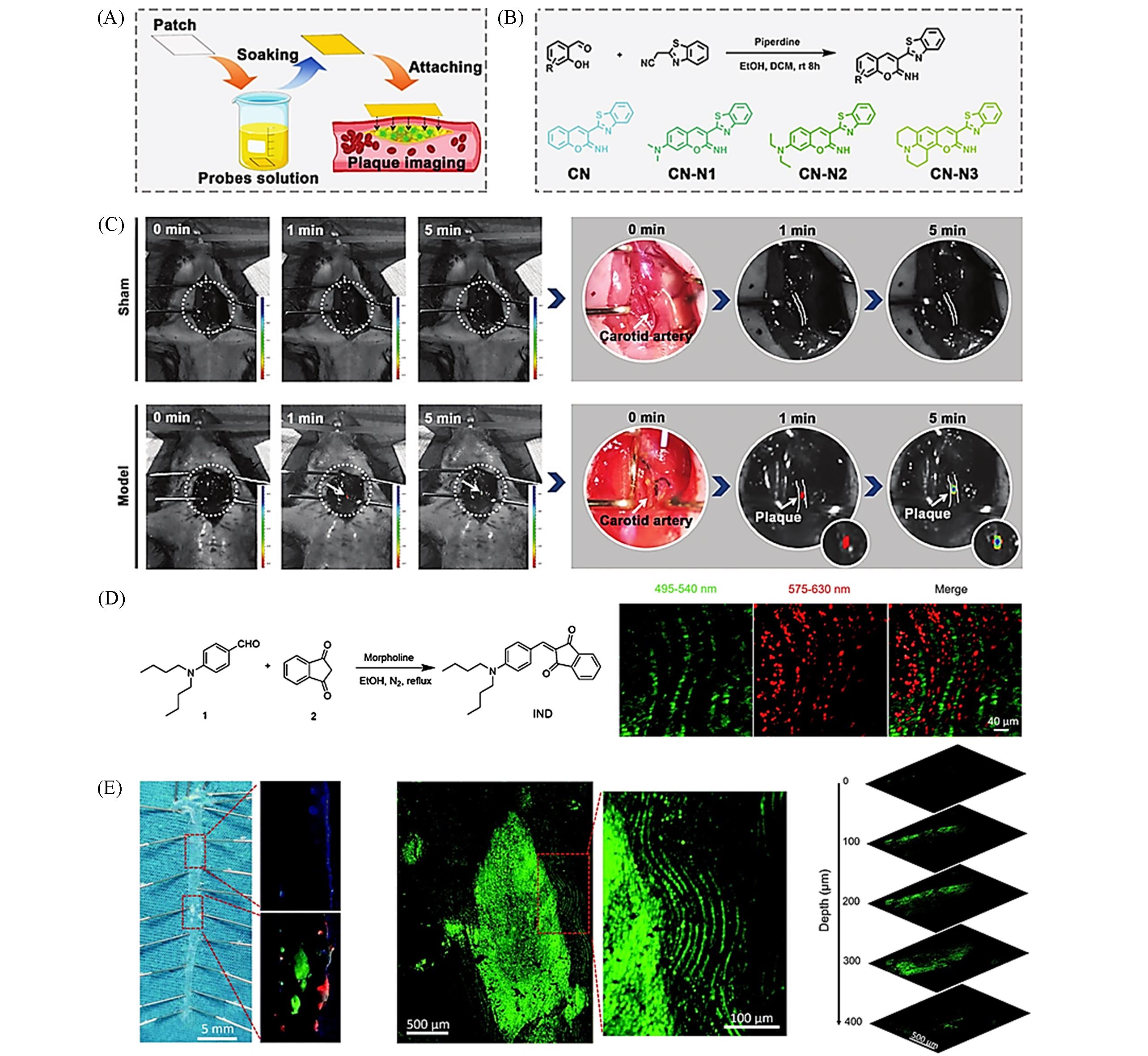
Fig.1 Lipid⁃responsive small molecule fluorescent probe(A) Intraoperative fluorescent imaging process using in situ foam-cell-specific probe-soaked patch for accurate location of atherosclerotic plaque; (B) synthetic route for probes used in the present study; (C) AS model imaging procedure: the left carotid artery is exposed to CN-N2-soaked filter paper; in vivo imaging was performed at 1 and 5 min[52]; (D) synthetic route to IND; (E) fluorescent images of lipid accumulation in the aorta of an ApoE-/- mice labeled with IND[53].(A—C) Copyright 2021, Wiley-VCH; (D, E) Copyright 2019, Royal Society of Chemistry.
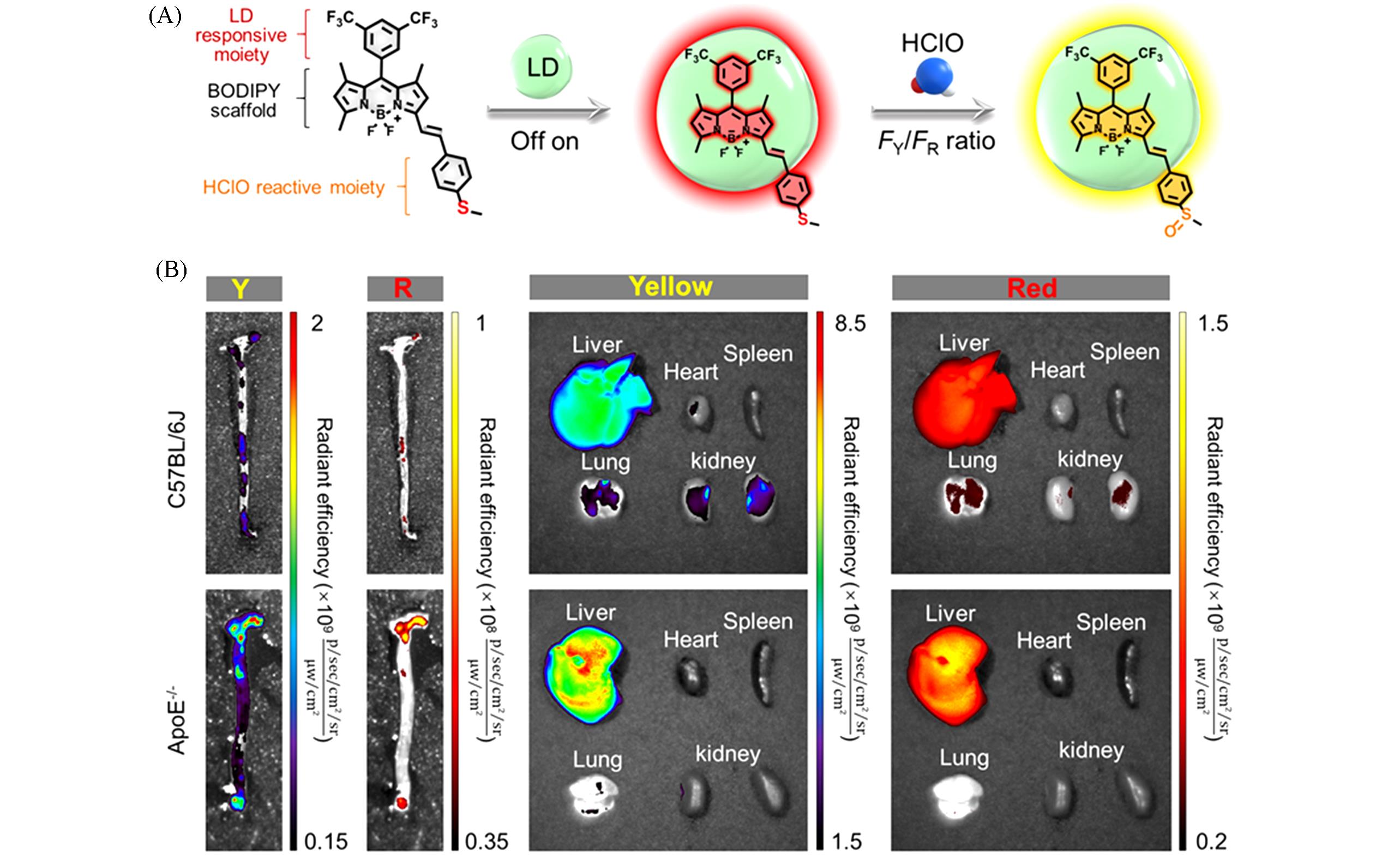
Fig.2 Dual⁃target responsive small molecule fluorescent probe[54](A) Schematic illustration of iSHERLOCK for “off-on” and ratiometric detection of LDs and HClO; FY and FR represent the fluorescent intensities(FI) in the yellow and red channels, respectively; (B) ex vivo imaging of aortas and main organs in C57BL/6J(control) and ApoE-/- mice(12 week high-fat diet feeding) after intravenous injection of MTB-B-CF3-incorporated Ac-LDL complexes.Copyright 2022, Wiley-VCH.
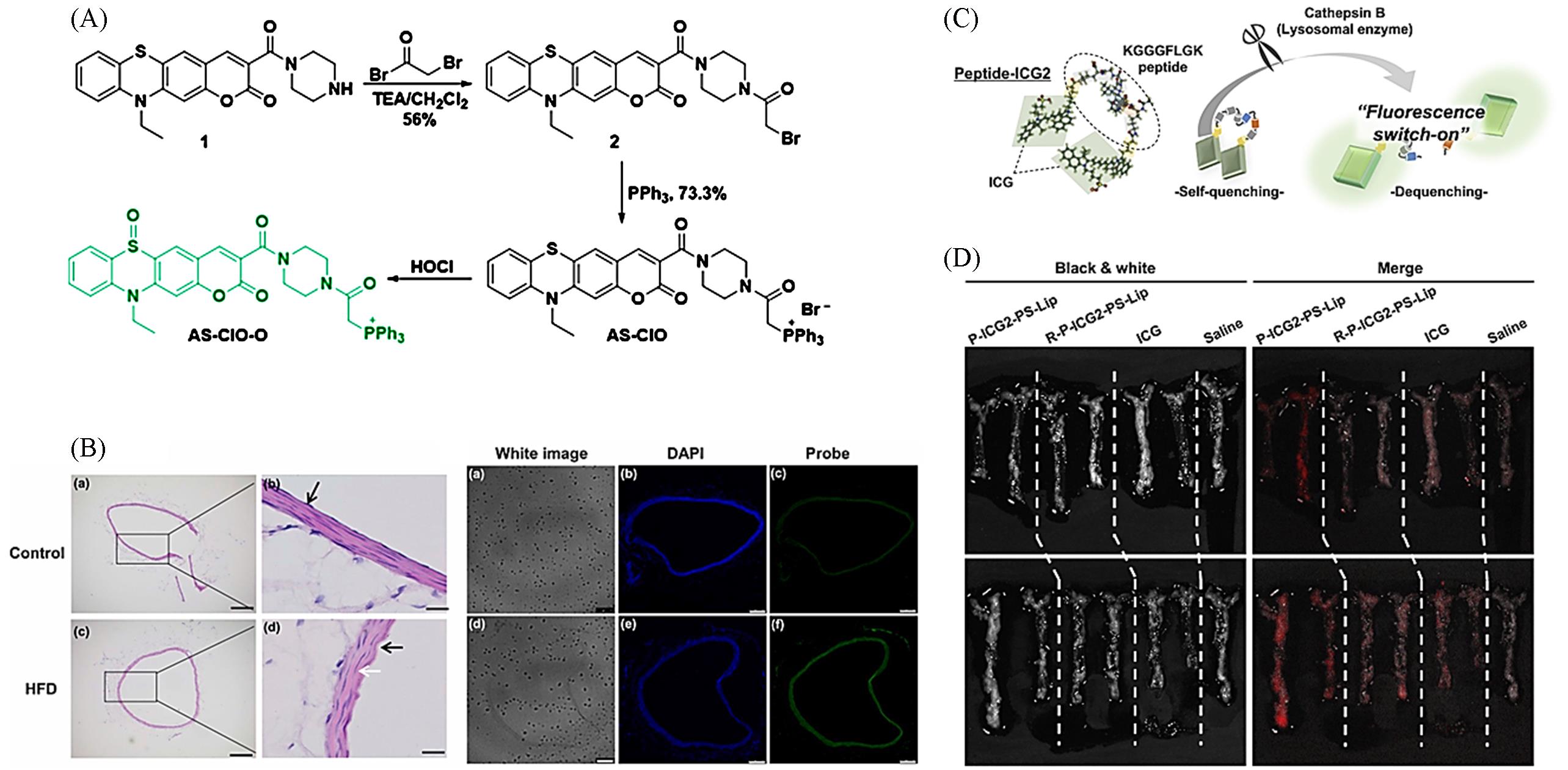
Fig.3 Other types of responsive small molecule fluorescent probes(A) The synthesis of AS-ClO and its reaction with HOCl; (B) H&E staining and fluorescence imaging of abdominal aorta sections from normal mice or HFD-mice[55]; (C) structural design and fluorescence-switch function of Peptide-ICG2; (D) fluorescence activation of P-ICG2-PS-Lip in atherosclerotic plaques[56].(A, B) Copyright 2021, Elsevier; (C, D) Copyright 2019, Elsevier.
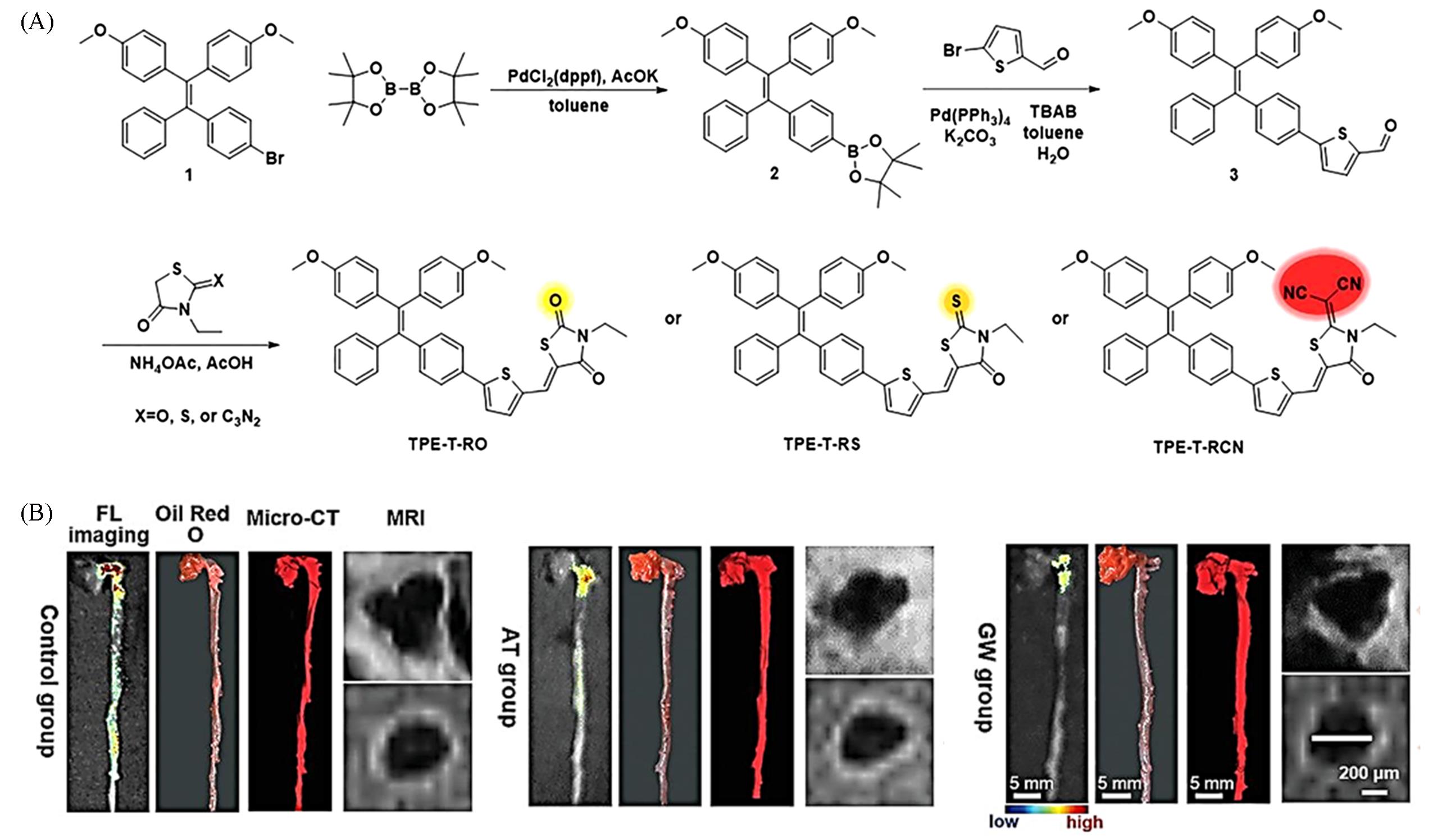
Fig.4 AIE probe for early imaging of AS plaques[60](A) The synthesis and optical property of a series of rhodanine derivatives-based AIEgens; synthetic route to TPE-T-RO, TPE-T-RS, and TPE-T-RCN; (B) representative FL imaging, Oil Red O staining, Micro-CT and MRI images of the same whole aorta isolated from mice with different treatments.Copyright 2021, Wiley-VCH.
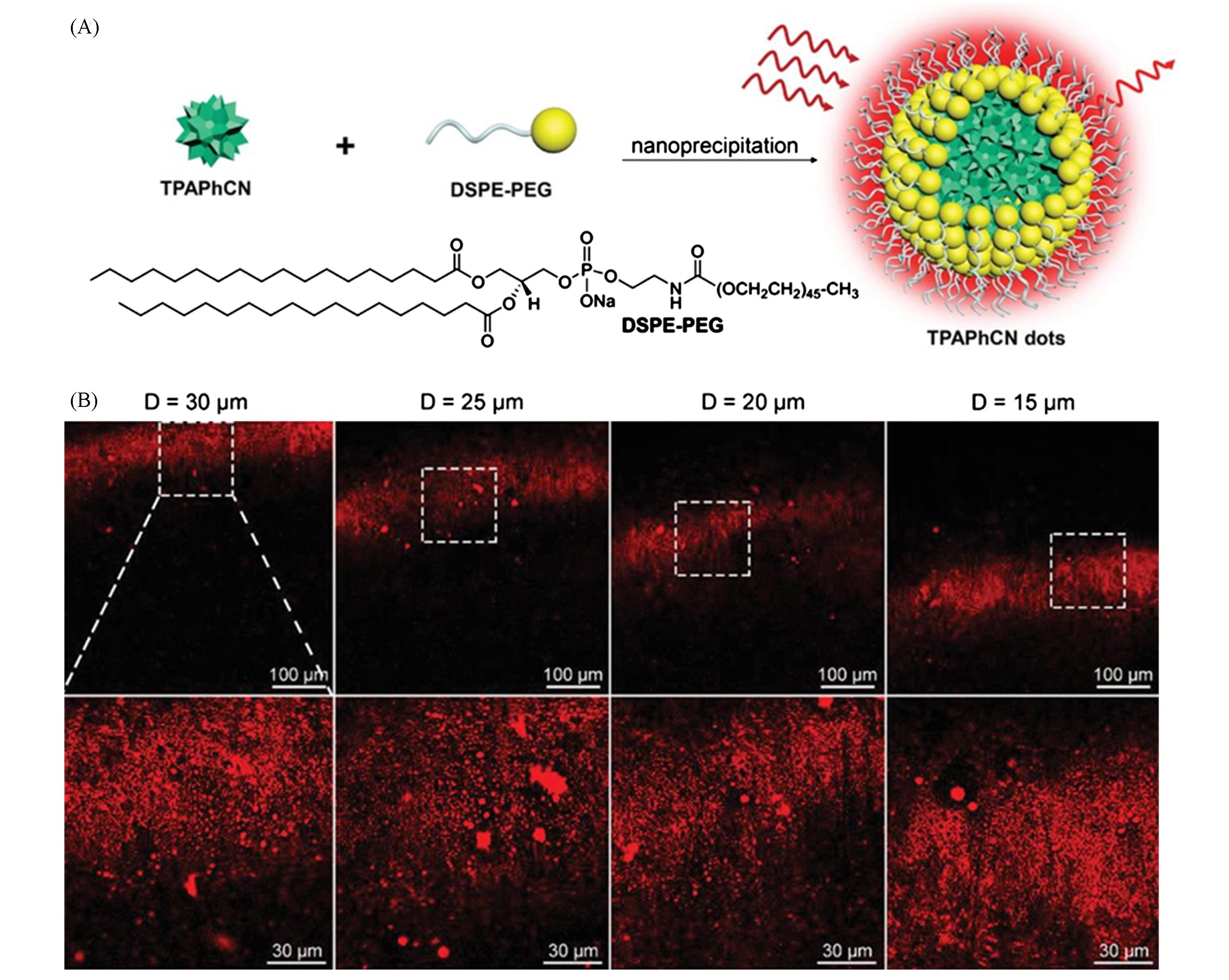
Fig.5 Three⁃photon AIE probe for deep AS plaque imaging[58](A) Schematic illustration of preparation of TPAPhCN dots; (B) intravital 3PF images of TPAPhCN-dots-labeled atherosclerotic plaques on the carotid artery vessel wall at different depths.Copyright 2021, Wiley-VCH.
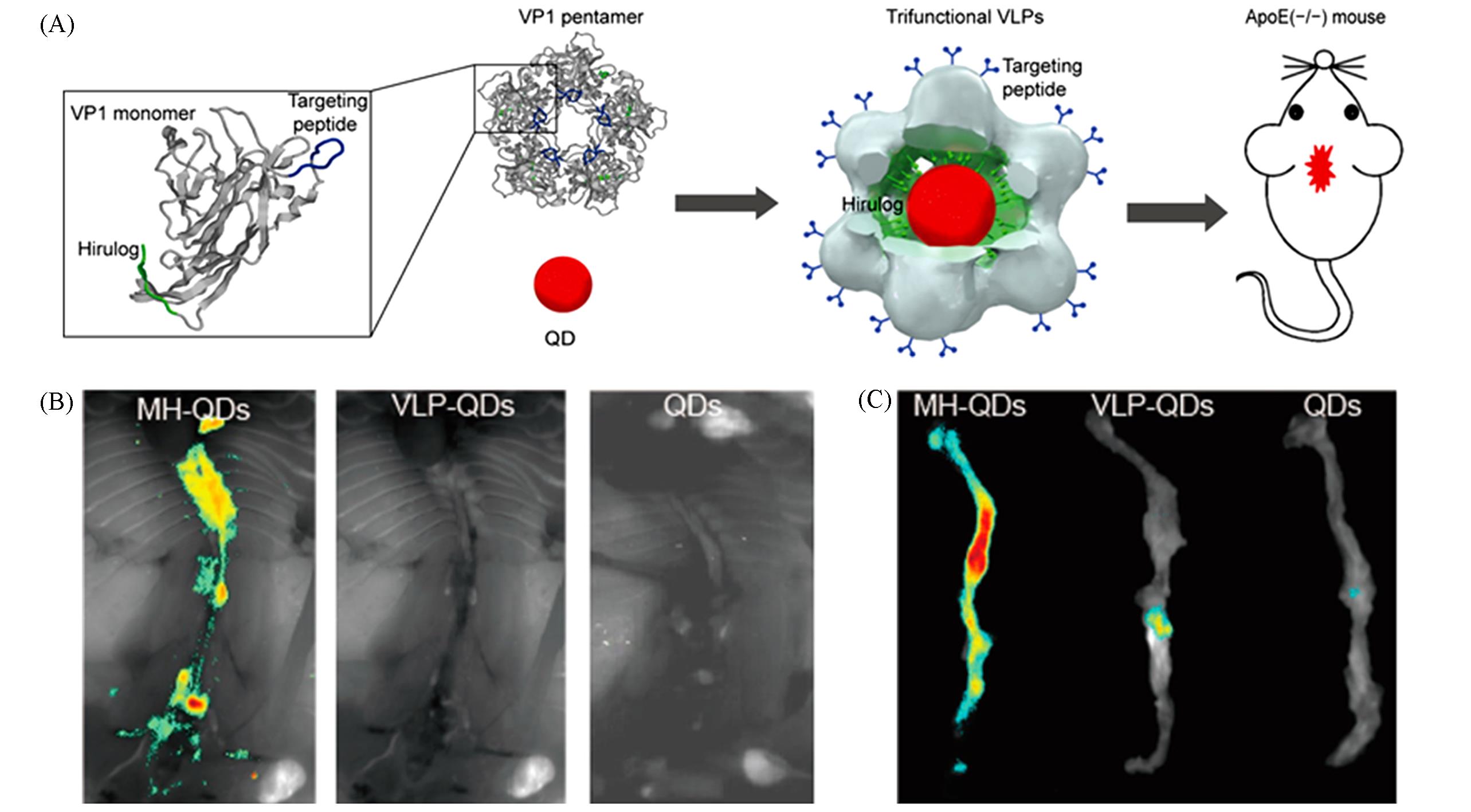
Fig.6 Quantum dot probes for in vivo imaging of AS[69](A) Schematic of study design. Twelve VP1 pentamers with targeting peptides and Hirulog peptides package one QD800 to form trifunctional virus-like particles(VLPs); trifunctional VLPs were injected into ApoE-/- mice via the tail vein for in vivo imaging; (B) in situ fluorescence images of aortas taken from mice injected with MH-QDs, VLP-QDs, and QDs; (C) ex vivo fluorescence of aortas from ApoE-/- mice injected with MH-QDs, VLP-QDs, and QDs.Copyright 2016, American Chemical Society.
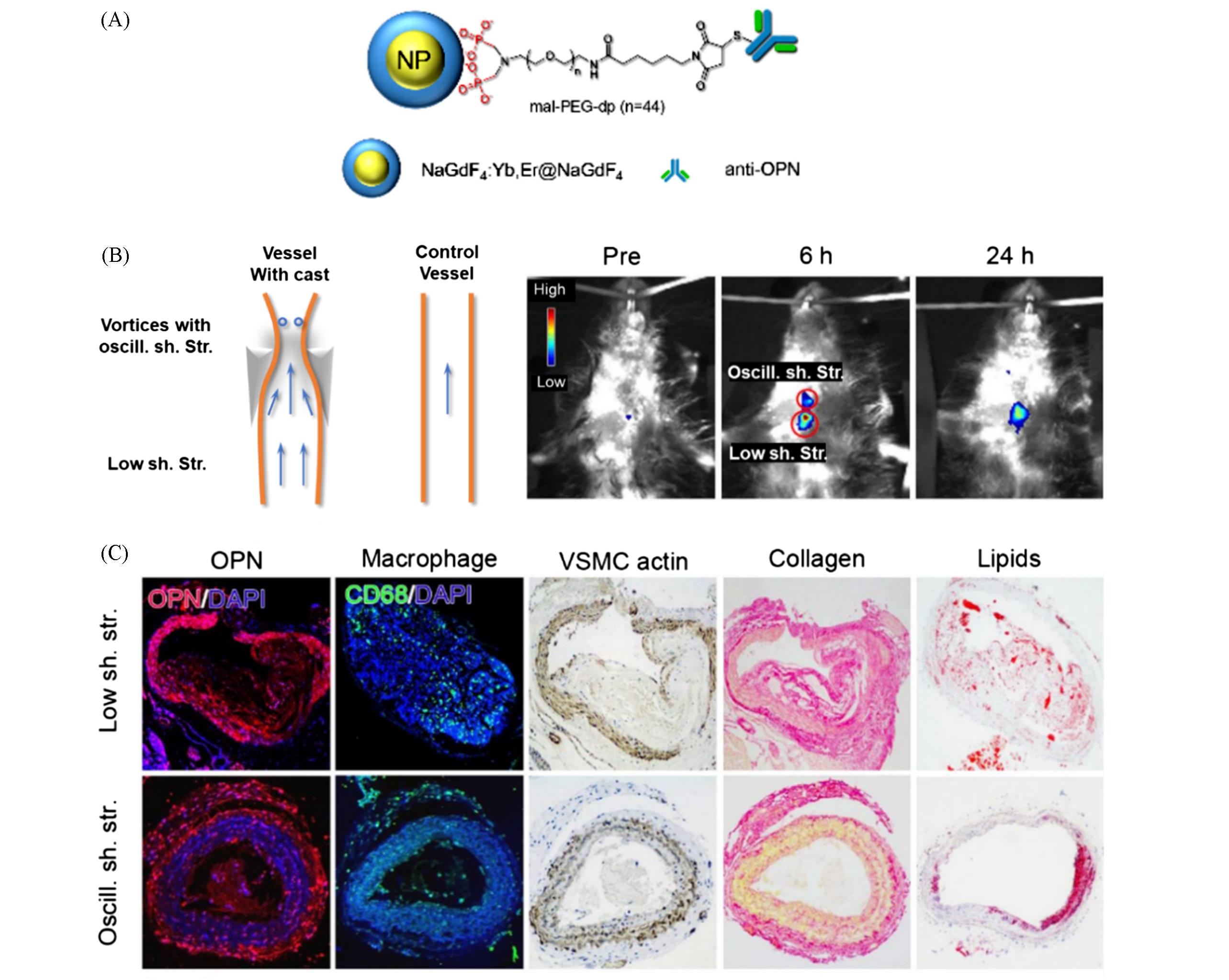
Fig.7 Upconversion luminescent nanoparticle probe for in vivo imaging of AS[77](A) A schematic drawing of the UCNP-anti-OPN probe; (B) in vivo upconversion luminescent images captured before and at different time points after intravenous injection of the UCNP-anti-OPN probe(the region of interest is encircled with red circles for showing the oscillatory shear stress(top) and lowered shear stress region (bottom) of the constrained vessel); (C) histological analyses.Copyright 2017, American Chemical Society.
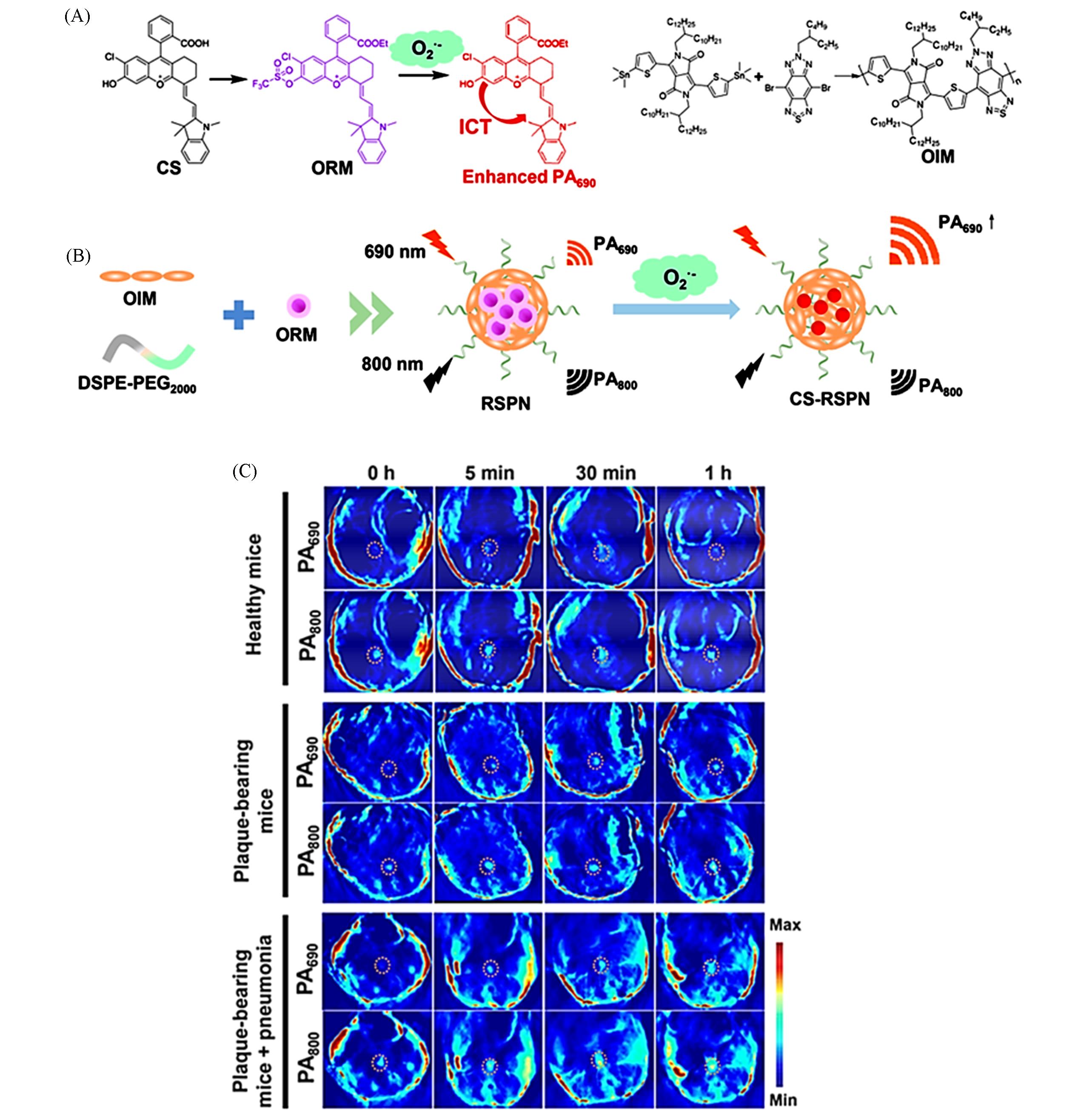
Fig.8 ROS responsive PA probe for in vivo imaging of AS[96](A) Chemical structures of CS, ORM, and OIM and the responsive mechanism for ORM to ·O2-; (B) one-step self-assembly of RSPN; (C) PA images after injection of RSPN of healthy mice, plaque-bearing mice and plaque-bearing mice complicated with pneumonia after injection of RSPN. Aorta regions were depicted by dotted circles.Copyright 2021, American Chemical Society.
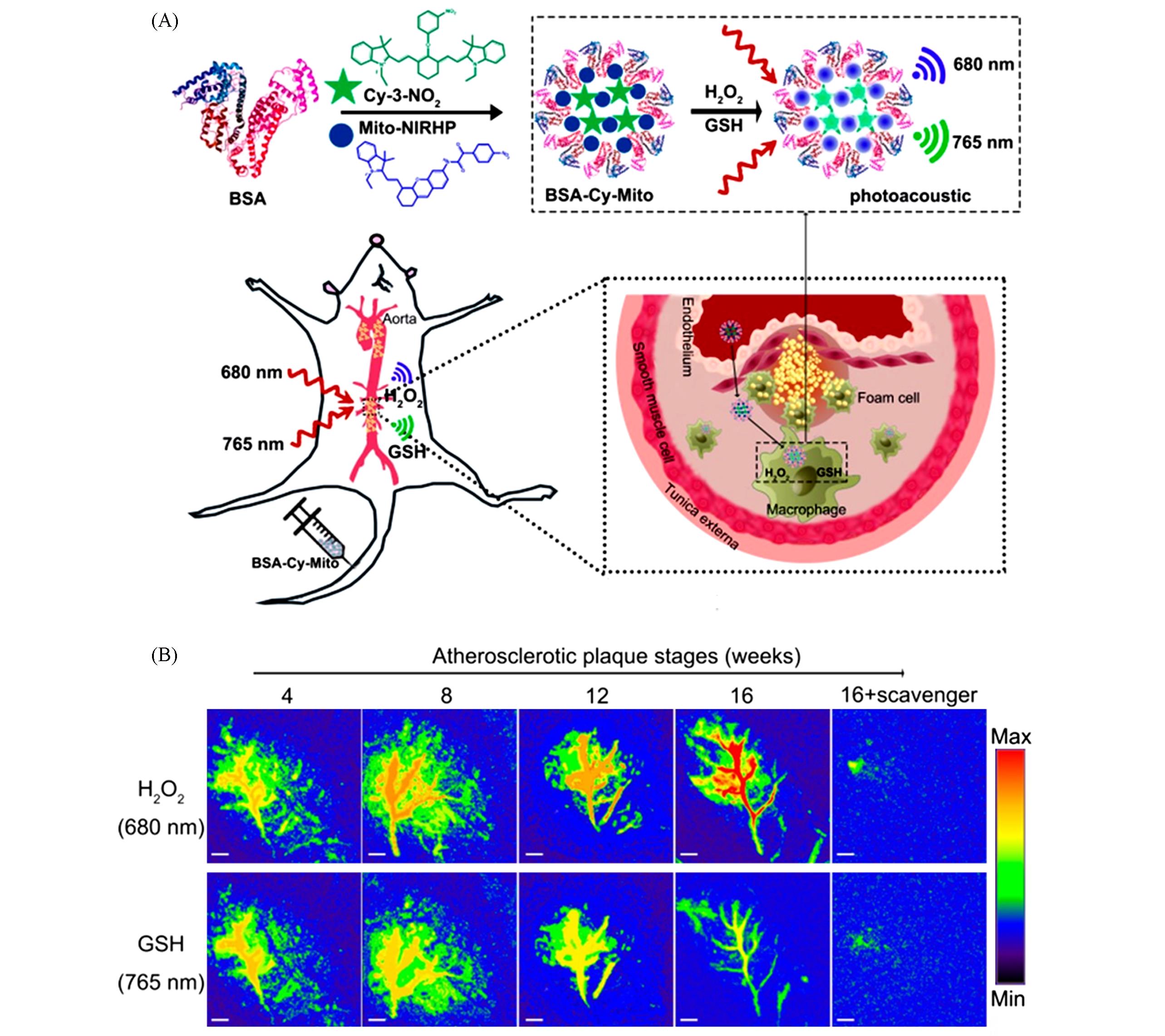
Fig.9 Dual⁃target responsive PA probe for in vivo imaging of AS[97](A) Molecular structures of GSH/H2O2-responsive BSA-Cy-Mito nanoprobe and illustration of the BSA-Cy-Mito nanoprobe for in vivo PA imaging of redox state to assess atherosclerotic plaque vulnerability; (B) representative PA imaging of abdominal aortas of plaque-bearing ApoE-/- mice at different stages injecting with BSA-Cy-Mito nanoprobe or pretreated with a scavenger.Copyright 2018, American Chemical Society.
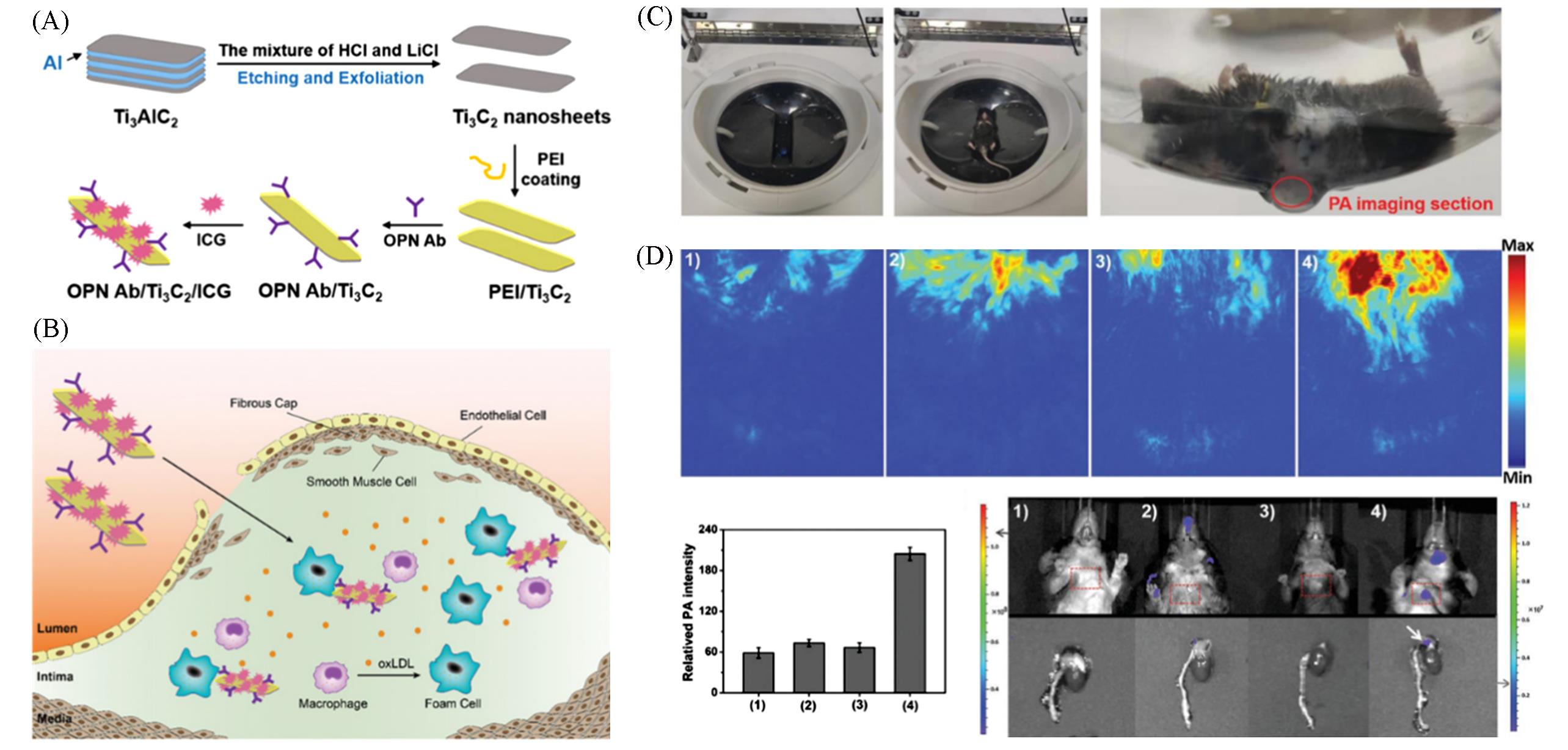
Fig.10 Targeted PA probe for in vivo imaging of AS[95](A, B) The preparation of OPN Ab/Ti3C2/ICG nanoprobes(A) and targeted recognition of OPN Ab/Ti3C2/ICG PA nanoprobes toward foam cells and vulnerable AS plaque(B); (C) the picture of PA measurement of AS model mice; (D) the in vivo PA imaging of experimental mice.Copyright 2020, Wiley-VCH.
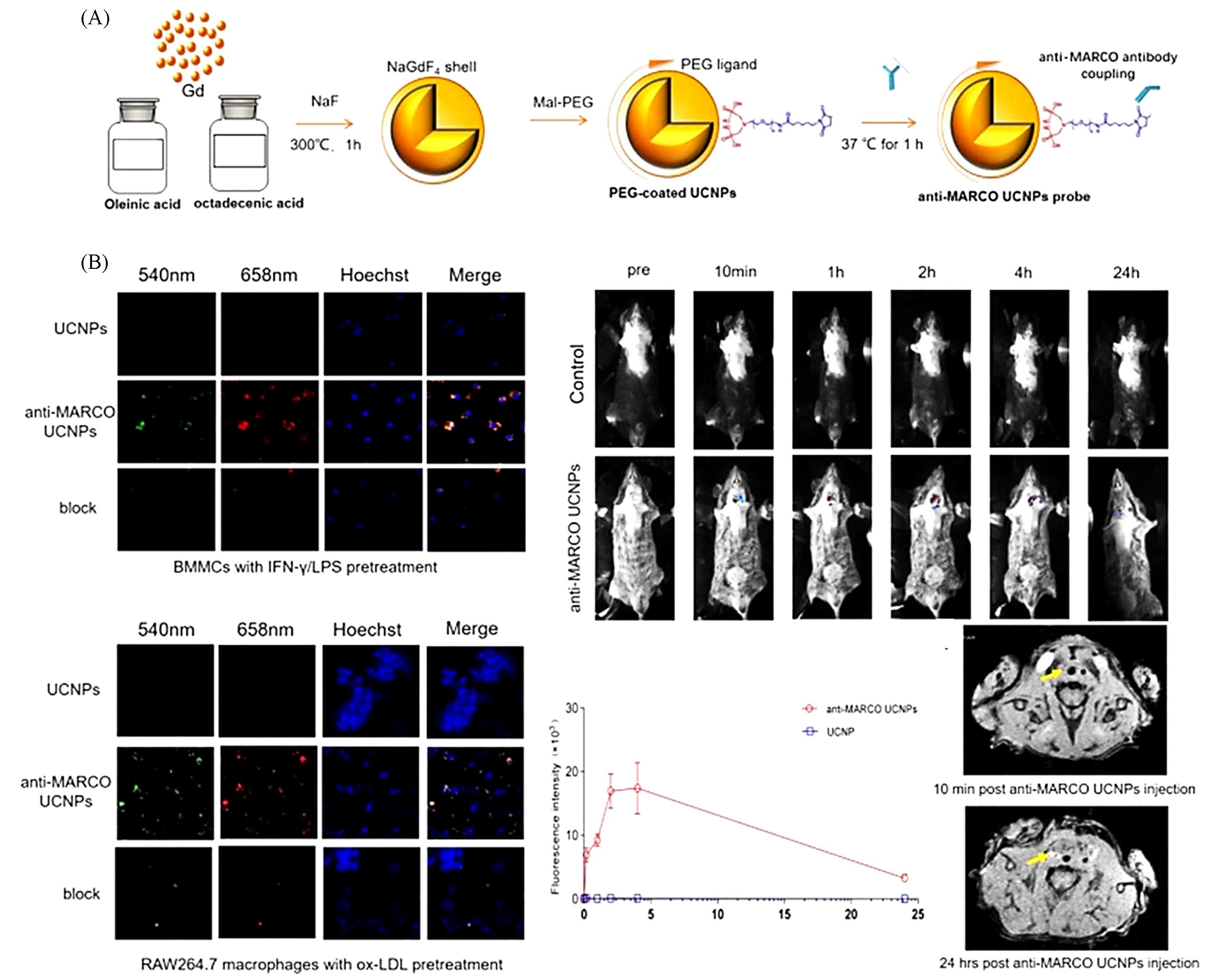
Fig.11 Multimodal probe for in vivo imaging of AS[100](A) Scheme of anti-MARCO UCNP probe construct; (B) in vitro and in vivo upconversion luminescent imaging of binding ability of anti-MARCO-UCNPs to M1 macrophage.Copyright 2019, Elsevier.
| 1 | Stewart J., Addy K., Campbell S., Wilkinson P., JRSM Cardiovasc. Dis., 2020, 9, 2048004020949326 |
| 2 | Amosse J., Martinez M. C., Le Lay S., Stem Cell Investig., 2017, 4(12), doi:10.21037/sci.2017.11.07 |
| 3 | Thayer J. F., Yamamoto S. S., Brosschot J. F., Int. J. Cardiol., 2010, 141(2), 122—131 |
| 4 | Benjamin E. J., Virani S. S., Callaway C. W., Chamberlain A. M., Chang A. R., Cheng S., Chiuve S. E., Cushman M., Delling F. N., Deo R., de Ferranti S. D., Ferguson J. F., Fornage M., Gillespie C., Isasi C. R., Jimenez M. C., Jordan L. C., Judd S. E., Lackland D., Lichtman J. H., Lisabeth L., Liu S. M., Longenecker C. T., Lutsey P. L., Mackey J. S., Matchar D. B., Matsushita K., Mussolino M. E., Nasir K., O'Flaherty M., Palaniappan L. P., Pandey A., Pandey D. K., Reeves M. J., Ritchey M. D., Rodriguez C. J., Roth G. A., Rosamond W. D., Sampson U. K. A., Satou G. M., Shah S. H., Spartano N. L., Tirschwell D. L., Tsao C. W., Voeks J. H., Willey J. Z., Wilkins J. T., Wu J. H. Y., Alger H. M., Wong S. S., Muntner P., Circulation, 2018, 137(12), e67—e492 |
| 5 | Dimmeler S., EMBO Mol. Med., 2011, 3(12), 697 |
| 6 | Libby P., Ridker P. M., Maseri A., Circulation, 2002, 105(9), 1135—1143 |
| 7 | Tabas I., Nat. Rev. Immunol., 2010, 10(1), 36—46 |
| 8 | Alfonso F., Rivero F., Circ. Cardiovasc. Inte., 2017, 10(12), e006143 |
| 9 | Alfonso F., Virmani R., Eur. Heart. J., 2015, 36(22), 1357—1360 |
| 10 | Libby P., J. Lipid. Res., 2009, 50, S352—S357 |
| 11 | Alfonso F., Joner M., Jacc⁃Cardiovasc. Imag., 2017, 10(8), 892—896 |
| 12 | Finn A. V., Nakano M., Narula J., Kolodgie F. D., Virmani R., Arterioscl. Throm. Vas., 2010, 30(7), 1282—1292 |
| 13 | Jaffer F. A., Libby P., Weissleder R., J. Am. Coll. Cardiol., 2006, 47(7), 1328—1338 |
| 14 | Ntziachristos V., Ripoll J., Wang L. H. V., Weissleder R., Nat. Biotechnol., 2005, 23(3), 313—320 |
| 15 | Lovell J. F., Jin C. S., Huynh E., Jin H. L., Kim C., Rubinstein J. L., Chan W. C. W., Cao W. G., Wang L. V., Zheng G., Nat. Mater., 2011, 10(4), 324—332 |
| 16 | Miao Q. Q., Xie C., Zhen X., Lyu Y., Duan H. W., Liu X. G., Jokerst J. V., Pu K. Y., Nat. Biotechnol., 2017, 35(11), 1102—1110 |
| 17 | Willmann J. K., van Bruggen N., Dinkelborg L. M., Gambhir S. S., Nat. Rev. Drug Discov., 2008, 7(7), 591—607 |
| 18 | Grenier N., Merville P., Combe C., Nat. Rev. Nephrol., 2016, 12(6), 348—359 |
| 19 | Ning X. H., Lee S., Wang Z. R., Kim D., Stubblefield B., Gilbert E., Murthy N., Nat. Mater., 2011, 10(8), 602—607 |
| 20 | Smith A. M., Mancini M. C., Nie S. M., Nat. Nanotechnol., 2009, 4(11), 710—711 |
| 21 | So M. K., Xu C. J., Loening A. M., Gambhir S. S., Rao J. H., Nat. Biotechnol., 2006, 24(3), 339—343 |
| 22 | Hong G. S., Antaris A. L., Dai H. J., Nat. Biomed. Eng., 2017, 1(1), 1—22 |
| 23 | Chan J., Dodani S. C., Chang C. J., Nat. Chem., 2012, 4(12), 973—984 |
| 24 | Park S. M., Aalipour A., Vermesh O., Yu J. H., Gambhir S. S., Nat. Rev. Mater., 2017, 2(5), 1—20 |
| 25 | Peterson B. S., Ann. Ny. Acad. Sci., 2003, 1008(1), 219—327 |
| 26 | Weissleder R., Science, 2006, 312(5777), 1168—1171 |
| 27 | Jaffer F. A., Libby P., Weissleder R., Circulation, 2007, 116(9), 1052—1061 |
| 28 | Rudin M., Weissleder R., Nat. Rev. Drug Discov., 2003, 2(2), 123—131 |
| 29 | Jokerst J. V., Gambhir S. S., Acc. Chem. Res., 2011, 44(10), 1050—1060 |
| 30 | Thorek D. L., Robertson R., Bacchus W. A., Hahn J., Rothberg J., Beattie B. J., Grimm J., Am. J. Nucl. Med. Molec., 2012, 2(2), 163—173 |
| 31 | Que E. L., Domaille D. W., Chang C. J., Chem. Rev., 2008, 108(5), 1517—1549 |
| 32 | Weissleder R., Mahmood U., Radiology, 2001, 219(2), 316—333 |
| 33 | Massoud T. F., Gambhir S. S., Trends. Mol. Med., 2007, 13(5), 183—191 |
| 34 | Lin V. S., Chen W., Xian M., Chang C. J., Chem. Soc. Rev., 2015, 44(14), 4596—4618 |
| 35 | Bartling S. H., Stiller W., Semmler W., Kiessling F., Curr. Med. Imaging., 2007, 3(1), 45—59 |
| 36 | Attia M. F., Wallyn J., Anton N., Vandamme T. F., Crit. Rev. Ther. Drug., 2018, 35(5), 391—431 |
| 37 | Aime S., Castelli D. D., Crich S. G., Gianolio E., Terreno E., Acc. Chem. Res., 2009, 42(7), 822—831 |
| 38 | Ni D. L., Bu W. B., Ehlerding E. B., Cai W. B., Shi J. L., Chem. Soc. Rev., 2017, 46(23), 7438—7468 |
| 39 | Terreno E., Delli Castelli D., Viale A., Aime S., Chem. Rev., 2010, 110(5), 3019—3042 |
| 40 | Deshpande N., Needles A., Willmann J. K., Clin. Radiol., 2010, 65(7), 567—581 |
| 41 | Culjat M. O., Goldenberg D., Tewari P., Singh R. S., Ultrasound Med. Biol., 2010, 36(6), 861—873 |
| 42 | Chen H. M., Zhang W. Z., Zhu G. Z., Xie J., Chen X. Y., Nat. Rev. Mater., 2017, 2(7), 11—18 |
| 43 | Herschman H. R., Science, 2003, 302(5645), 605—608 |
| 44 | Gambhir S. S., Nat. Rev. Cancer, 2002, 2(9), 683—693 |
| 45 | Shokeen M., Anderson C. J., Acc. Chem. Res., 2009, 42(7), 832—841 |
| 46 | Dobrucki L. W., Sinusas A. J., Nat. Rev. Cardiol., 2010, 7(1), 38—47 |
| 47 | Ametamey S. M., Honer M., Schubiger P. A., Chem. Rev., 2008, 108(5), 1501—1516 |
| 48 | Ntziachristos V., Razansky D., Chem. Rev., 2010, 110(5), 2783—2794 |
| 49 | Maguire E. M., Pearce S. W. A., Xiao Q. Z., Vasc. Pharmacol., 2019, 112, 54—71 |
| 50 | Ayyappan J. P., Paul A., Goo Y. H., Mol. Med. Rep., 2016, 13(6), 4527—4534 |
| 51 | Li S. F., Zhuang W. H., Chen J. R., Li G. C., Li C. M., Chen L., Liao Y. B. A., Chen M., Wang Y. B., J. Mater. Chem. B, 2021, 9(19), 4050—4055 |
| 52 | Zheng J. R., Zhao S. S., Mao Y. J., Du Z. C., Li G., Sang M. M., Small, 2022, 18(5), 2104471 |
| 53 | Situ B., Gao M., He X. J., Li S. W., He B. R., Guo F. X., Kang C. M., Liu S., Yang L., Jiang M. J., Hu Y. W., Tang B. Z., Zheng L., Mater. Horizons, 2019, 6(3), 546—553 |
| 54 | Ye Z., Ji M. X., Wu K. F., Yang J., Liu A. A., Sun W., Ding D., Liu D. B., Angew. Chem. Int. Ed., 2022, 61(29), e 202204518 |
| 55 | Wang B. Y., Yuan F. Y., Wang S., Duan R., Ren W. X., Hou J. T., Sensor. Actuat. B⁃Chem., 2021, 348, 130695 |
| 56 | Narita Y., Shimizu K., Ikemoto K., Uchino R., Kosugi M., Maess M. B., Magata Y., Oku N., Ogawa M., J. Control. Release, 2019, 302, 105—115 |
| 57 | Li Q. Q., Li Z., Acc. Chem. Res., 2020, 53(4), 962—973 |
| 58 | Wang S. W., Li X. Q., Chong S. Y., Wang X. Y., Chen H., Chen C. J., Ng L. G., Wang J. W., Liu B., Adv. Mater., 2021, 33(11), 2007490 |
| 59 | Li H. D., Yao Q. C., Xu F., Li Y. Q., Kim D., Chung J., Baek G., Wu X. F., Hillman P. F., Lee E. Y., Ge H. Y., Fan J. L., Wang J. Y., Nam S. J., Peng X. J., Yoon J. Y., Angew. Chem., 2020, 132(25), 10272—10281 |
| 60 | Wang K., Gao H. Q., Zhang Y. W., Yan H. Y., Si J. H., Mi X. Y., Xia S. A., Feng X. Q., Liu D. B., Kong D. L., Wang T., Ding D., Adv. Mater., 2022,34(9), 2106994 |
| 61 | Hoover E. E., Squier J. A., Nat. Photonics, 2013, 7(2), 93—101 |
| 62 | Wang S. W., Chen H., Liu J., Chen C. J., Liu B., Adv. Funct. Mater., 2020, 30(30), 2002546 |
| 63 | de Arquer F. P. G., Talapin D. V., Klimov V. I., Arakawa Y., Bayer M., Sargent E. H., Science, 2021, 373(6555), eaaz8541 |
| 64 | Yoffe A. D., Adv. Phys., 2001, 50(1), 1—208 |
| 65 | Lim S. Y., Shen W., Gao Z. Q., Chem. Soc. Rev., 2015, 44(1), 362—381 |
| 66 | Hennessy K., Badolato A., Winger M., Gerace D., Atatüre M., Gulde S., Fält S., Hu E. L., Imamoğlu A., Nature, 2007, 445(7130), 896—899 |
| 67 | Xue Q., Zhang H. J., Zhu M. S., Pei Z. X., Li H. F., Wang Z. F., Huang Y., Huang Y., Deng Q. H., Zhou J., Du S. Y.,Huang Q., Zhi C. Y., Adv. Mater., 2017, 29(15), 1604847 |
| 68 | Zhuang Y. R., Lin B. X., Yu Y., Wang Y. M., Zhang L., Cao Y. J., Guo M. L., Food Chem., 2021, 356, 129720 |
| 69 | Sun X. X., Li W., Zhang X. W., Qi M., Zhang Z. P., Zhang X. E., Cui Z. Q., Nano Lett., 2016, 16(10), 6164—6171 |
| 70 | Chen G. Y., Qju H. L., Prasad P. N., Chen X. Y., Chem. Rev., 2014, 114(10), 5161—5214 |
| 71 | Zhang F., Shi Q. H., Zhang Y. C., Shi Y. F., Ding K. L., Zhao D. Y., Stucky G. D., Adv. Mater., 2011, 23(33), 3775—3779 |
| 72 | Xu C. T., Svenmarker P., Liu H. C., Wu X., Messing M. E., Wallenberg L. R., Andersson⁃Engels S., ACS Nano, 2012, 6(6), 4788—4795 |
| 73 | Haase M., Schäfer H., Angew. Chem. Int. Ed., 2011, 50(26), 5808—5829 |
| 74 | Wu Y. M., Cen Y., Huang L. J., Yu R. Q., Chu X., Chem. Commun., 2014, 50(36), 4759—4762 |
| 75 | Su Q. Q., Han S. Y., Xie X. J., Zhu H. M., Chen H. Y., Chen C. K., Liu R. S., Chen X. Y., Wang F., Liu X. G., J. Am. Chem. Soc., 2012, 134(51), 20849—20857 |
| 76 | Hudson D. E., Hudson D. O., Wininger J. M., Richardson B. D., Photomed. Laser Surg., 2013, 31(4), 163—168 |
| 77 | Qiao R. R., Qiao H. Y., Zhang Y., Wang Y. B., Chi C. W., Tian J., Zhang L. F., Cao F., Gao M. Y., ACS Nano, 2017, 11(2), 1816—1825 |
| 78 | Quillard T., Croce K., Jaffer F. A., Weissleder R., Libby P., Thromb. Haemostasis, 2011, 105(5), 828—836 |
| 79 | Qiao R. R., Liu C. H., Liu M. H., Hu H., Liu C. Y., Hou Y., Wu K. C., Lin Y. N., Liang J. M., Gao M. Y., ACS Nano, 2015, 9(2), 2120—2129 |
| 80 | Liu C. Y., Hou Y., Gao M. Y., Adv. Mater., 2014, 26(40), 6922—6932 |
| 81 | Scatena M., Liaw L., Giachelli C. M., Arterioscl. Throm. Vas., 2007, 27(11), 2302—2309 |
| 82 | Matsui Y., Rittling S. R., Okamoto H., Inobe M., Jia N., Shimizu T., Akino M., Sugawara T., Morimoto J., Kimura C., Kon S., Denhardt D., Kitabatake A., Uede T., Arterioscl. Throm. Vas., 2003, 23(6), 1029—1034 |
| 83 | Wang L. H. V., Hu S., Science, 2012, 335(6075), 1458—1462 |
| 84 | de la Zerda A., Zavaleta C., Keren S., Vaithilingam S., Bodapati S., Liu Z., Levi J., Smith B. R.,Ma T. J., Oralkan O., Cheng Z., Chen X. Y., Dai H. J., Khuri⁃Yakub B. T., Gambhir S. S., Nat. Nanotechnol., 2008, 3(9), 557—562 |
| 85 | Yeager D., Chen Y. S., Litovsky S., Emelianov S., Theranostics, 2014, 4(1), 36—46 |
| 86 | Nam K., Stanczak M., Lyshckik A., Machado P., Kono Y., Forsberg F., Shaw C. M., Eisenbrey J. K., Biomed. Phys. Eng. Express, 2018, 4(3), 032002 |
| 87 | Cao Y. C., Kole A., Hui J., Zhang Y., Mai J. Y., Alloosh M., Sturek M., Cheng J. X., Sci. Rep., 2018, 8, 2400 |
| 88 | Iskander⁃Rizk S., Wu M., Springeling G., van Beusekom H. M. M., Mastik F., Hekkert M. T., Beurskens R. H. S. H., Hoogendoorn A., Hartman E. M. J., van der Steen A. F. W., Wentzel J. J., van Soest G., EuroIntervention, 2019, 15(5), 452—456 |
| 89 | Gao F., Feng X. H., Zhang R. C., Liu S. Y., Ding R., Kishor R., Zheng Y. J., Sci. Rep., 2017, 7,626 |
| 90 | Gao F., Bai L. Y., Liu S. Y., Zhang R. C., Zhang J. T., Feng X. H., Zheng Y. J., Zhao Y. L., Nanoscale, 2017, 9(1), 79—86 |
| 91 | Gao F., Bai L., Feng X., Tham H. P., Zhang R., Zhang Y., Liu S., Zhao L., Zheng Y., Zhao Y., Small, 2016, 12(38), 5239—5244 |
| 92 | Yao J. J., Kaberniuk A. A., Li L., Shcherbakova D. M., Zhang R. Y., Wang L. D., Li G., Verkhusha V. V., Wang L. H. V., Nat. Methods, 2016, 13(1), 67—73 |
| 93 | Wu C. X., Zhang Y. J., Li Z., Li C. Y., Wang Q. B., Nanoscale, 2016, 8(25), 12531—12539 |
| 94 | Xie Z. H., Yang Y. Q., He Y. Q., Shu C. Y., Chen D., Zhang J. K., Chen J. Q., Liu C. B., Sheng Z. H., Liu H. D., Liu J., Gong X. J., Song L., Dong S. H., Theranostics, 2020, 10(10), 4694—4704 |
| 95 | Ge X. X., Cui H. T., Kong J., Lu S. Y., Zhan R., Gao J. N., Xu Y. K., Lin S. Y., Meng K. X., Zu L. Y., Guo S. J., Zheng L. M., Adv. Mater., 2020, 32(38), 2000037 |
| 96 | Ma Y., Xu L., Yin B. L., Shang J. H., Chen F. F., Xu J. T., Song Z. L., Nan B., Song G. S., Zhang X. B., Nano Lett., 2021, 21(10), 4484—4493 |
| 97 | Gao W., Li X., Liu Z. H., Fu W., Sun Y. H., Cao W. H., Tong L. L., Tang B., Anal. Chem., 2019, 91(1), 1150—1156 |
| 98 | Willmann J. K., van Bruggen N., Dinkelborg L. M., Gambhir S. S., Nat. Rev. Drug Discov., 2008, 7(7), 591—607 |
| 99 | Qiao H. Y., Wang Y. B., Zhang R. H., Gao Q. S., Liang X., Gao L., Jiang Z. H., Qiao R. R., Han D., Zhang Y., Qiu Y., Tian J., Gao M. Y., Cao F., Biomaterials, 2017, 112, 336—345 |
| 100 | Qin M., Wang L., Li F., Yang M., Song L., Tian F., Yukht A., Shah P. K., Rothenberg M. E., Sharifi B. G., Atherosclerosis, 2017, 263, 82—91 |
| 101 | Jaffer F. A., Calfon M. A., Rosenthal A., Mallas G., Razansky R. N., Mauskapf A., Weissleder R., Libby P., Ntziachristos V., J. Am. Coll. Cardiol., 2011, 57(25), 2516—2526 |
| 102 | Wang Y. B., Zhang Y., Wang Z., Zhang J. B., Qiao R. R., Xu M. Q., Yang N., Gao L., Qiao H. Y., Gao M. Y., Cao F., Biomaterials, 2019, 219, 119378 |
| [1] | 徐心昱, 张乐天, 曹晖, 马原, 刘柳卉, 宋国胜, 张晓兵. 脂质响应型探针用于动脉粥样硬化成像及治疗的研究进展[J]. 高等学校化学学报, 2022, 43(12): 20220383. |
| [2] | 赵恒智, 余方志, 李翔菲, 李乐乐. 基于DNA与上转换纳米颗粒相结合的生物传感与成像研究进展[J]. 高等学校化学学报, 2022, 43(12): 20220626. |
| [3] | 张钤, 刘雅薇, 王帆, 刘凯, 张洪杰. 稀土纳米材料在高分辨活体成像及诊疗中的应用[J]. 高等学校化学学报, 2022, 43(12): 20220552. |
| [4] | 唐玉静, 胡敏, 王霞, 王启刚. 载酶纳米催化体系用于疾病诊疗的研究进展[J]. 高等学校化学学报, 2022, 43(12): 20220640. |
| [5] | 卢美如, 张宏宇, 石百媚, 孙茂忠, 徐丽广, 胥传来, 匡华. 手性纳米材料: 生物成像、 生物传感与治疗[J]. 高等学校化学学报, 2022, 43(12): 20220683. |
| [6] | 朱兆田, 李圣凯, 宋明慧, 蔡芯琪, 宋志灵, 陈龙, 陈卓. 多功能金属石墨纳米囊的生物医学应用进展[J]. 高等学校化学学报, 2021, 42(9): 2701. |
| [7] | 黄池宝, 康帅, 潘淇, 吕国岭. 衍生于咔唑的双氰基二苯代乙烯型双光子荧光脂筏探针[J]. 高等学校化学学报, 2021, 42(8): 2443. |
| [8] | 柯梦婷, 袁江培, 张恒, 方煜. 多孔配位聚合物靶向亚细胞器用于生物成像和诊疗[J]. 高等学校化学学报, 2021, 42(11): 3295. |
| [9] | 张雨, 荆江博, 邵玥明, 殷鑫, 徐斌, 温晓玉. 基于聚集诱导发光的纳米粒子用于肝癌细胞靶向成像[J]. 高等学校化学学报, 2019, 40(11): 2382. |
| [10] | 方超, 朱焓毓, 刘晔, 赵外欧, 李亚鹏, 王静媛. 过氧化氢敏感的靶向荧光载药纳米粒子的制备及在动脉粥样硬化中的应用[J]. 高等学校化学学报, 2018, 39(9): 2071. |
| [11] | 刘晔, 姚顺雨, 方超, 赵外欧, 王静媛, 李亚鹏. 靶向髓过氧化酶的双模态探针分子的合成与表征[J]. 高等学校化学学报, 2018, 39(7): 1573. |
| [12] | 亢艳艳, 戚卉, 周淼, 张颖, 庄家琪, 杨军星, 王博蔚. Ag∶InP/ZnSe纳米晶的合成与生物成像[J]. 高等学校化学学报, 2017, 38(9): 1512. |
| [13] | 张海鹏, 韩冰, 贾致真, 丁荣波, 徐斌, 徐蔚青, 范志民. 载药荧光纳米粒子的制备及在乳腺癌MCF-7细胞系的应用及效果评价[J]. 高等学校化学学报, 2017, 38(5): 860. |
| [14] | 胡齐, 方超, 赵外鸥, 李亚鹏, 陈霞, 王静媛. 动脉粥样硬化造影剂PGMA-EDA-g-PEG-g-DS@IO的合成及性能[J]. 高等学校化学学报, 2015, 36(10): 2061. |
| [15] | 石渊渊, 王玉兰, 李志强. SMS高表达对细胞内和培养基中SM水平的影响[J]. 高等学校化学学报, 2008, 29(10): 1982. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||