

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (12): 3579.doi: 10.7503/cjcu20210527
收稿日期:2021-07-21
出版日期:2021-12-10
发布日期:2021-09-22
通讯作者:
王力
E-mail:wanglimerry@jmu.edu.cn
基金资助:
SHUAI Die1, ZHAO Meijuan1, CHEN Bingnian2, WANG Li1( )
)
Received:2021-07-21
Online:2021-12-10
Published:2021-09-22
Contact:
WANG Li
E-mail:wanglimerry@jmu.edu.cn
Supported by:摘要:
合成并表征了4种过渡金属钒取代的Keggin型磷钼酸盐Na3+nPMo12-nVnO40(PMo12-nVn)(n=2, 3, 4, 5). 酶动力学实验结果表明, 4种多酸对酪氨酸酶的抑制类型为可逆的混合型抑制, 过渡金属钒取代的个数会影响酪氨酸酶的抑制效果. 当所加酶量为500 U/mL时, PMo10V2, PMo9V3, PMo8V4和PMo7V5对酪氨酸酶的半抑制率(IC50)分别为(7.046±0.506), (12.128±0.574), (12.362±0.802)和(9.860±1.490) mmol/L. 分子对接研究表明, 化合物与酪氨酸酶之间主要存在氢键和范德华力. 细胞实验结果表明, 在0~200 μmol/L范围内, 4种多酸未对B16黑素瘤细胞产生细胞毒性, 且能显著抑制黑色素的生成. 此外, 1,1-二苯基-2-三硝基苯肼(DPPH)与2,2'-联氮-双-3-乙基苯并噻唑啉-6-磺酸(ABTS)自由基清除实验显示, 4种多酸具有良好的抗氧化能力, 清除DPPH自由基能力优于ABTS.
中图分类号:
TrendMD:
帅蝶, 赵美娟, 陈丙年, 王力. 4种Keggin型磷钼酸盐对蘑菇酪氨酸酶活性和黑色素生成的抑制及抗氧化作用. 高等学校化学学报, 2021, 42(12): 3579.
SHUAI Die, ZHAO Meijuan, CHEN Bingnian, WANG Li. Inhibitory Effect of Four Kinds of Keegin-type Phosphomolybdate on Tyrosinase and Melanin Formation and Its Antioxidant Activities. Chem. J. Chinese Universities, 2021, 42(12): 3579.
| Compd. | IC50/(mmol·L-1) | Inhibitory mechanism | Inhibitory type | Inhibitory constant | |
|---|---|---|---|---|---|
| KI/(mmol·L-1) | KIS/(mmol·L-1) | ||||
| PMo11V[ | 0.522±0.003 | Reversible | Competitive | 0.172 | — |
| PMo10V2 | 7.046±0.506 | Reversible | Mixed?type | 12.482 | 66.315 |
| PMo9V3 | 12.128±0.574 | Reversible | Mixed?type | 15.431 | 14.013 |
| PMo8V4 | 12.362±0.802 | Reversible | Mixed?type | 10.870 | 46.191 |
| PMo7V5 | 9.860±1.490 | Reversible | Mixed?type | 7.222 | 4.563 |
Table 1 Enzyme kinetic results of Keggin-type vanadium-substituted phosphomolybdate on tyrosinase
| Compd. | IC50/(mmol·L-1) | Inhibitory mechanism | Inhibitory type | Inhibitory constant | |
|---|---|---|---|---|---|
| KI/(mmol·L-1) | KIS/(mmol·L-1) | ||||
| PMo11V[ | 0.522±0.003 | Reversible | Competitive | 0.172 | — |
| PMo10V2 | 7.046±0.506 | Reversible | Mixed?type | 12.482 | 66.315 |
| PMo9V3 | 12.128±0.574 | Reversible | Mixed?type | 15.431 | 14.013 |
| PMo8V4 | 12.362±0.802 | Reversible | Mixed?type | 10.870 | 46.191 |
| PMo7V5 | 9.860±1.490 | Reversible | Mixed?type | 7.222 | 4.563 |
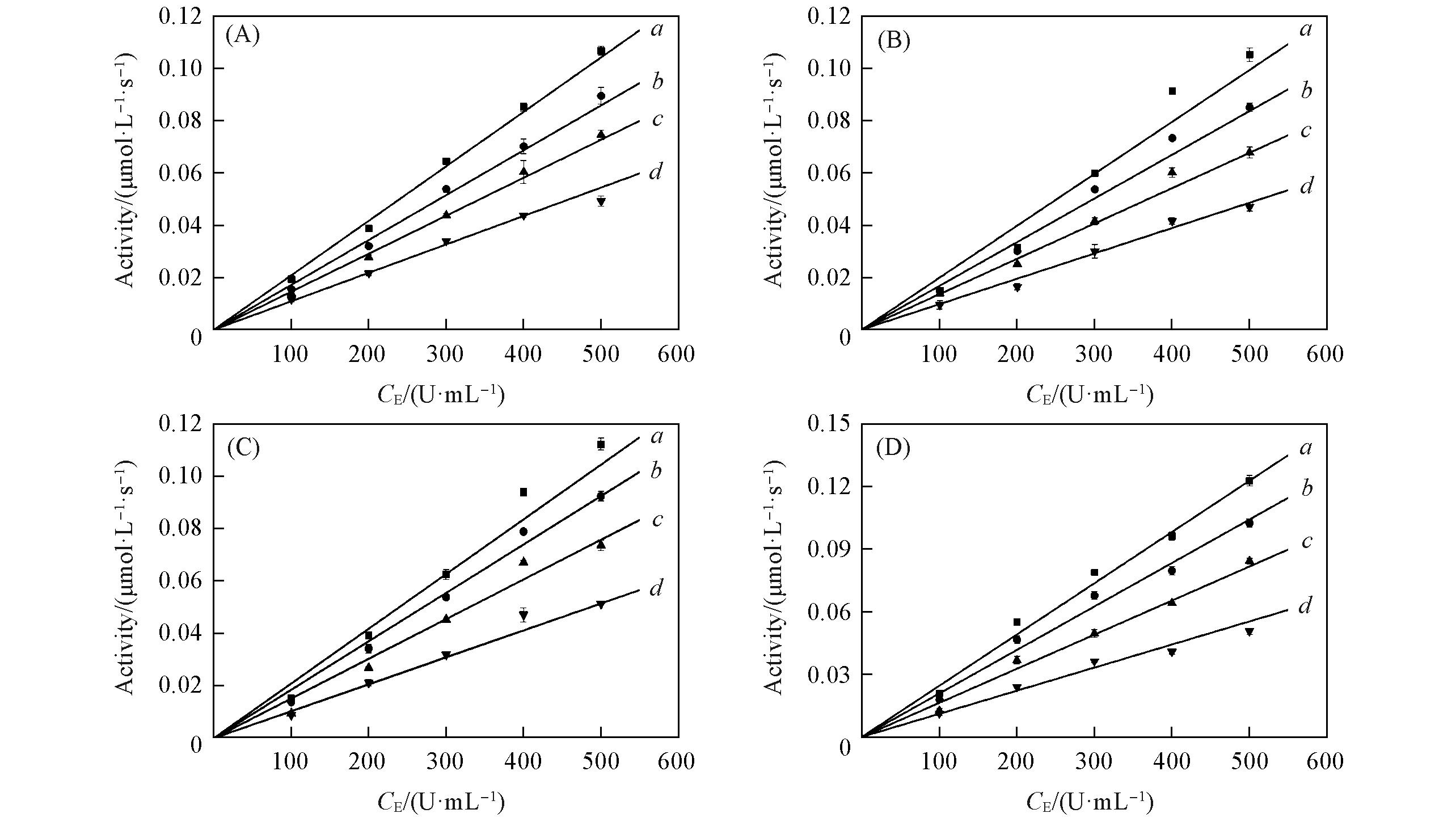
Fig.5 Inhibitory mechanism of PMo10V2(A), PMo9V3(B), PMo8V4(C) and PMo7V5(D) on the tyrosinase(A) The concentrations of PMo10V2 were 0(a), 1.6(b), 2.4(c), 3.6(d) mmol/L, respectively; (B) the concentrations of PMo9V3 were 0(a), 1(b), 3(c), 5(d) mmol/L, respectively; (C) the concentrations of PMo8V4 were 0(a), 1(b), 2(c), 3(d) mmol/L, respectively; (D) the concentrations of PMo7V5 were 0(a), 1(b), 2(c), 3(d) mmol/L, respectively.
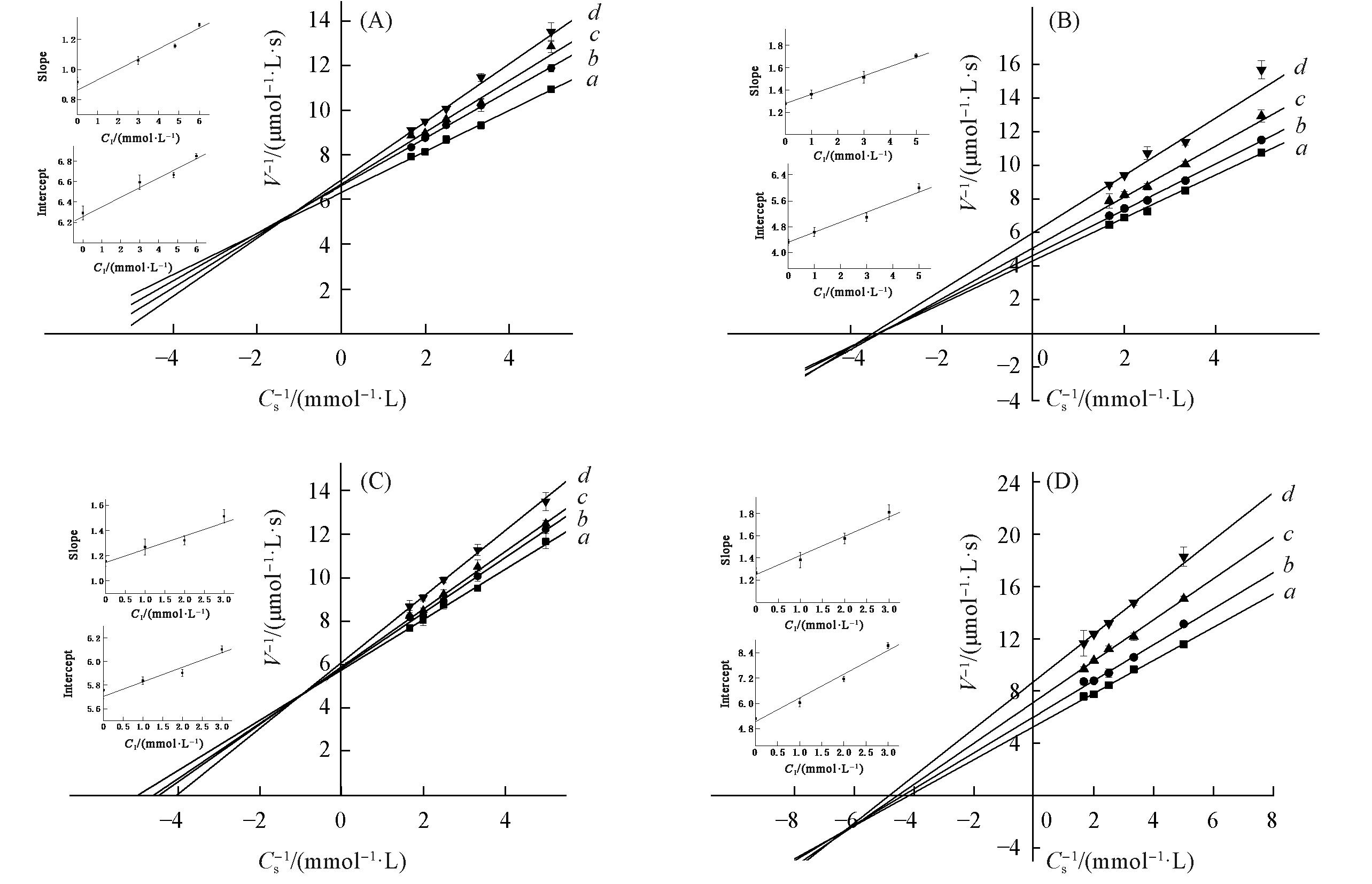
Fig.6 Inhibitory type and constants of PMo10V2(A), PMo9V3(B), PMo8V4(C) and PMo7V5(D) on the tyrosinase(A) The concentrations of PMo10V2 were 0(a), 3(b), 4.8(c), 6 mmol/L(d), respectively;(B) the concentrations of PMo9V3 were 0(a), 1(b), 3(c), 5 mmol/L(d), respectively;(C) the concentrations of PMo8V4 were 0(a), 1(b), 2(c), 3 mmol/L(d), respectively;(D) the concentrations of PMo7V5 were 0(a), 1(b), 2(c), 3 mmol/L(d), respectively. Cs- 1 represents the reciprocal of substrate concentration; V-1 represents the reciprocal of enzyme reaction rate. Insets represent the secondary plot of the slope and intercept, respectively, versus the concentrations of PMo10V2, PMo9V3, PMo8V4 and PMo7V5 to determine the inhibition constants(K1 and KIS).
| Compd. | Force type | Amino acid |
|---|---|---|
| PMo9V3 | Hydrogen bond | His85, His94, His259, Asn260 |
| van der Waals force | His61, Asn81, Cys83, Thr84, Phe90, His244, Glu256, His263, Phe264, Val283, Ala286, Phe292 | |
| PMo8V4 | Hydrogen bond | His94, His259, Val283, Ala286 |
| van der Waals force | His61, Cys83, Thr84, His85, Phe90, Asn260, His263, Ala287, Phe292, His295, His296 | |
| PMo7V5 | Hydrogen bond | His259, Asn260 |
| van der Waals force | His61, Cys83, Thr84, His85, Gly86, Phe90, His244, Val248, His263, Phe264, Gly281, Val283, Phe292 |
Table 2 Molecular docking of compounds
| Compd. | Force type | Amino acid |
|---|---|---|
| PMo9V3 | Hydrogen bond | His85, His94, His259, Asn260 |
| van der Waals force | His61, Asn81, Cys83, Thr84, Phe90, His244, Glu256, His263, Phe264, Val283, Ala286, Phe292 | |
| PMo8V4 | Hydrogen bond | His94, His259, Val283, Ala286 |
| van der Waals force | His61, Cys83, Thr84, His85, Phe90, Asn260, His263, Ala287, Phe292, His295, His296 | |
| PMo7V5 | Hydrogen bond | His259, Asn260 |
| van der Waals force | His61, Cys83, Thr84, His85, Gly86, Phe90, His244, Val248, His263, Phe264, Gly281, Val283, Phe292 |
| 1 | Solomon E. I., Sundaram U. M., Machonkin T. E., Chem. Rev., 1996, 96(7), 2563—2605 |
| 2 | Shao L. L., Wang X. L., Chen K., Dong X. W., Kong L. M., Zhao D. Y., Hider R. C., Zhou T., Food Chem., 2018, 242, 174—181 |
| 3 | Lehraiki A., Abbe P., Rocchi S., J. Invest. Dermatol., 2014, 134(10), 2589—2597 |
| 4 | Niu C., Yin L., Aisa H. A., Int. J. Mol. Sci., 2018, 19(3), 746 |
| 5 | Jin K. S., Oh Y. N., Hyun S. K., Kwon H. J., Kim B. W., Food Chem. Toxicol., 2014, 68, 38—43 |
| 6 | Huang Y. C., Liu K. C., Chiou Y. L., Yang C. H., Chen T. H., Li T. T., Liu L. L., Chem. Biol. Interact., 2013, 205(3), 157—164 |
| 7 | Lin L. C., Dong Y., Zhao H. F., Wen L. R., Yang B., Zhao M. M., Food Chem., 2011, 129(3), 884—889 |
| 8 | Dianat S., Tangestaninejad S., Yadollahi B., Bordbar A. K., Moghadam M., Mirkhani V., Mohammadpoor-Baltork I., J. Mol. Liq., 2012, 174, 76—79 |
| 9 | Wang S. T., Hou Y., Wang E. B., Li Y. G., Hu C. W., Chem. J. Chinese Universities, 2004, 25(1), 27—29(王树涛, 侯煜, 王恩波, 李阳光, 胡长文. 高等学校化学学报, 2004, 25(1), 27—29) |
| 10 | Fu H., Wang X. L., Chen W. L., Meng J. X., Li Y. G., Wang E. B., Chem. J. Chinese Universities, 2011, 32(3), 650—654(傅海, 王晓兰, 陈维林, 孟靖昕, 李阳光, 王恩波. 高等学校化学学报, 2011, 32(3), 650—654) |
| 11 | Wang X. L., Wang E. B., Xu X. X., Li Y. G., Chem. J. Chinese Universities, 2008, 29(10), 1937—1940(王晓兰, 王恩波, 徐欣欣, 李阳光. 高等学校化学学报, 2008, 29(10), 1937—1940) |
| 12 | Zhang L. D., Wei Y. G., Feng X. L., Guo H. J., Chem. J. Chinese Universities, 2005, 26(8), 1395—1398(张丽丹, 魏永革, 冯秀玲, 郭洪猷. 高等学校化学学报, 2005, 26(8), 1395—1398) |
| 13 | Cen Q., Xiao W., Liu Y. Q., Wang Q., Nafady A., Ma S. Q., Chem. Res. Chinese Universities, 2020, 36(6), 1128—1135 |
| 14 | Zhang W. Q., Lin Z. G., Chi Y. N., Hong J., Yan L., Hu C. W., Chem. Res. Chinese Universities, 2020, 36(6), 1161—1167 |
| 15 | Qi Y., Xiang Y., Wang J., Qi Y. F., Li J., Niu J. Q., Zhong J., Antiviral Res., 2013, 100(2), 392—398 |
| 16 | Pu F., Wang E. B., Jiang H. Y., Ren J. S., Molecular Biosystems, 2013, 9(1), 113—120 |
| 17 | Bijelic A., Aureliano M., Rompel A., Chem. Commun., 2018, 54(10), 1153—1169 |
| 18 | Chen B. N., Feng Z. G., Wang L., Chem. J. Chinese Universities, 2011, 32(5), 1033—1036(陈丙年, 冯珍鸽, 王力. 高等学校化学学报, 2011, 32(5), 1033—1036) |
| 19 | Zhao M. J., Chen X. S., Chi G. X., Shuai D., Wang L., Chen B. N., Li J., Inorganic Chemistry Frontiers, 2020, 7(22), 4320—4332 |
| 20 | Hu X. Y., Chen B. N., Wang L., Chen F. H., Chem. Res. Chinese Universities, 2012, 28(5), 862—865 |
| 21 | Bijelic A., Aureliano M., Rompel A., Angew. Chem. Int. Ed., 2019, 58(10), 2980—2999 |
| 22 | Blazevic A., Rompel A., Coord. Chem. Rev., 2016, 307(Part 1), 42—64 |
| 23 | Liu S. X., Liu Y. Y., Wang E. B., Zeng Y., Li Z. L., Chem. J. Chinese Universities, 1996, 17(8), 1188—1190(刘术侠, 刘彦勇, 王恩波, 曾毅, 李泽琳. 高等学校化学学报, 1996, 17(8), 1188—1190) |
| 24 | Liu S. X., Li Y. X., Hang Z. B., Wang E. B., Zeng Y., Li Z. L., Chem. J. Chinese Universities, 2002, 23(5), 777—782(刘术侠, 李阳先, 韩正波, 王恩波, 曾毅, 李泽淋. 高等学校化学学报, 2002, 23(5), 777—782) |
| 25 | Xing R., Zheng A. P., Wang F., Wang L., J. Mol. Sci., 2015, 31, 436—440(邢蕊, 郑阿萍, 王芳, 王力. 分子科学学报, 2015, 31(5), 436—440) |
| 26 | Zhang F. M., Guo M. P., Ge H. Q., Wang J., Front. Chem. Eng. China, 2007, 1(3), 296—299 |
| 27 | Tsigdinos G. A., Hallada C. J., Inorg. Chem., 1968, 7, 149—176 |
| 28 | Chen Q. X., Song K. K., Qiu L., Liu X. D., Huang H., Guo H. Y., Food Chem., 2005, 91(2), 269—274 |
| 29 | Jones K., Hughes J., Hong M., Jia Q., Orndorff S., Pigment Cell Res., 2002, 15(5), 335—340 |
| 30 | Jiang M. M., Tian C. R., Wang X. J., Jiangsu J. Agric. Sci., 2008, 24(2), 194—198(蒋萌蒙, 田呈瑞, 王向军. 江苏农业学报, 2008, 24(2), 194—198) |
| 31 | Zheng A. P., Wang F., Xing R., Jiang A. H., Wang L., Chem. J. Chinese Universities, 2014, 35(3), 476—481(郑阿萍, 王芳, 邢蕊, 蒋爱华, 王力. 高等学校化学学报, 2014, 35(3), 476—481) |
| 32 | Ismaya W. T., Rozeboom H. J., Weijn A., Mes J. J., Fusetti F., Wichers H. J., Dijkstra B. W., Biochemistry, 2011, 50(24), 5477—5486 |
| 33 | Yang Y. F., Sun X., Ni H., Du X. P., Chen F., Jiang Z. D., Li Q. B., J. Agric. Food. Chem., 2019, 67(46), 12741—12751 |
| 34 | Ali M., Khan K. M., Salar U., Ashraf M., Taha M., Wadood A., Hamid S., Riaz M., Ali B., Shamim S., Ali F., Perveen S., Mol. Divers., 2018, 22(4), 841—861 |
| 35 | Xing R., Wang F., Dong L., Zheng A. P., Wang L., Su W. J., Lin T., Food Chem., 2016, 197(Part A), 205—211 |
| 36 | Chung S. W., Ha Y. M., Kim Y. J., Song S., Lee H., Suh H., Chung H. Y., Arch. Pharm. Res., 2009, 32(2), 289—294 |
| 37 | Teng H., Fan X. Y., Lv Q. Y., Zhang Q., Xiao J. B., Qian Y. W., Zheng B. D., Gao H., Gao S. H., Chen L., Food Chem., 2020, 330, 127030 |
| 38 | Zhu Y. J., Qiu L., Zhou J. J., Guo H. Y., Hu Y. H., Li Z. C., Wang Q., Chen Q. X., Liu B., J. Enzyme Inhib. Med. Chem., 2010, 25(6), 798—803 |
| 39 | Wang L. Y., Ma M. T., Yu Z. P., Du S. K., Food Chem., 2021, 352, 129399 |
| 40 | Ashooriha M., Khoshneviszadeh M., Khoshneviszadeh M., Moradi S. E., Rafiei A., Kardan M., Emami S., Bioorg. Chem., 2019, 82, 414—422 |
| 41 | Thaipong K., Boonprakob U., Crosby K., Cisneros-Zevallos L., Byrne D. H., Journal Of Food Composition And Analysis, 2006, 19(6/7), 669—675 |
| 42 | Guo J.,Cai L. L., Xie T., Zhou W. Z., Yang S. K., Xie Q., Liu Q. B., Qin J., J. Functional Materials, 2019, 50(9), 9120—9128 (郭军, 蔡丽丽, 解田, 周维珍, 杨三可, 谢泉, 刘其斌, 秦军. 功能材料, 2019, 50(9), 9120—9128) |
| 43 | Zheng A. P., Acta Sci. Natura Univ. Innermongolia, Natural Science Ed., 2018, 33(3), 204—207, 218(郑阿萍. 内蒙古民族大学学报(自然科学版), 2018, 33(3), 204—207, 218) |
| 44 | Xing R., Functionality Study of Polyoxometalates as Tyrosinase Inhibitor, Jimei University, Xiamen, 2016(邢蕊. 多酸型酪氨酸酶抑制剂的功能性研究, 厦门: 集美大学, 2016) |
| 45 | Gong R. M., J. Anhui Normal Univ.(Nat. Sci.) , 1998, 21(1), 46—49(龚仁敏. 安徽师大学报(自然科学版), 1998, 21(1), 46—49) |
| 46 | Xie H., Hang Y. T., Shao P. L., Zhang L. F., Zheng A. R., Sci. Tech. Food Ind., 2017, 38(22), 37—41(谢惠, 韩娅婷, 邵佩兰, 张丽芬, 郑安然. 食品工业科技, 2017, 38(22), 37—41) |
| 47 | Wang M., Tian Z. Y., Wang W. X., Food Res. Int., 2018, 39(22), 66—72(王敏, 田珍燕, 王蔚新. 食品研究与开发, 2018, 39(22), 66—72) |
| [1] | 金睿明, 穆晓清, 徐岩. 生物-化学法合成黑色素前体5, 6-二羟基吲哚[J]. 高等学校化学学报, 2022, 43(8): 20220134. |
| [2] | 张太文, 郭军, 张丹, 袁常梅, 邱双艳. trz-Cl-Cu-PMo12的合成、 表征及催化氧化碘离子性能[J]. 高等学校化学学报, 2022, 43(10): 20220215. |
| [3] | 蒋君, 宫田田, 张成鹏, 刘晓倩, 赵俊伟. 吡啶二羧酸修饰的稀土嵌入碲钨酸盐的合成及电化学生物识别性质[J]. 高等学校化学学报, 2022, 43(1): 20210561. |
| [4] | 初明月, 李峰博, 高宁, 杨昕, 于婷婷, 马慧媛, 杨桂欣, 庞海军. 轮型多金属氧酸盐复合物膜的制备及在检测亚硝酸盐中的应用[J]. 高等学校化学学报, 2022, 43(1): 20210579. |
| [5] | 陈慧娜, 李新雄, 郑寿添. 铌多酸三维框架材料的研究进展[J]. 高等学校化学学报, 2022, 43(1): 20210625. |
| [6] | 梁宇, 刘欢, 宫丽阁, 王春晓, 王春梅, 于凯, 周百斌. 联咪唑修饰{SiW12O40}杂化物的合成及超级电容性能[J]. 高等学校化学学报, 2022, 43(1): 20210556. |
| [7] | 黄罗仪, 翁约约, 黄旭慧, 王朝杰. 车前草中黄酮类成分结构和性质的理论研究[J]. 高等学校化学学报, 2021, 42(9): 2752. |
| [8] | 雷晓彤, 金怡卿, 孟烜宇. 基于分子模拟方法预测PIP2在双孔钾通道TREK-1上结合位点的研究[J]. 高等学校化学学报, 2021, 42(8): 2550. |
| [9] | 胡皓程, 李文利, 张嘉宁, 刘宇博. 黑木耳寡糖的提取、 结构表征及生物活性[J]. 高等学校化学学报, 2021, 42(8): 2465. |
| [10] | 李聪聪, 刘明皓, 韩佳睿, 朱镜璇, 韩葳葳, 李婉南. 基于分子动力学模拟的VmoLac非特异性底物催化活性的理论研究[J]. 高等学校化学学报, 2021, 42(8): 2518. |
| [11] | 杨依然, 姚华, 闫江红, 孙志恒, 张余, 房雪晴, 李绪文, 金永日. 薤中新的甾体皂苷类化学成分[J]. 高等学校化学学报, 2021, 42(6): 1742. |
| [12] | 赵雪, 张安歌, 田洪瑞, 王赫男, 霍海燕, 刘术侠. 基于Keggin型杂多酸的无机-有机杂化物的合成和光催化活性[J]. 高等学校化学学报, 2021, 42(6): 1723. |
| [13] | 彭海月, 汪婷, 李国瑞, 黄静. 黑色素的合成及小分子对其功能的调控[J]. 高等学校化学学报, 2021, 42(11): 3357. |
| [14] | 杨举, 苏丽娇, 李灿花, 鲁佳佳, 杨俊丽, 古捷, 杨丽, 杨丽娟. 甘松新酮/磷酸盐柱[6]芳烃主客体络合行为[J]. 高等学校化学学报, 2021, 42(10): 3099. |
| [15] | 张爱芹, 王嫚, 申刚义, 金军. 多溴联苯醚与人血清白蛋白相互作用的表面等离子体共振及分子对接[J]. 高等学校化学学报, 2020, 41(9): 2054. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||