

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (3): 448.doi: 10.7503/cjcu20180521
收稿日期:2019-07-24
出版日期:2019-03-10
发布日期:2019-08-21
作者简介:联系人简介: 李 颖, 女, 博士, 副教授, 主要从事电化学传感检测、 光电催化等方面的研究. E-mail: 基金资助:
LI Ying*( ), KANG Junjun, ZHAO Xueru, XU Wenkai, QI Qi
), KANG Junjun, ZHAO Xueru, XU Wenkai, QI Qi
Received:2019-07-24
Online:2019-03-10
Published:2019-08-21
Contact:
LI Ying
E-mail:liying_791190@163.com
Supported by:摘要:
以金修饰磁性石墨烯(Au@Fe3O4@RGO)为载体, 通过表面分子印迹技术, 选择水环境中邻苯二甲酸二正丁酯(DBP)为模板分子, 制备了金修饰磁性石墨烯基分子印迹复合膜(Au@Fe3O4@RGO-MIP); 通过扫描电子显微镜(TEM)、 X射线衍射(XRD)、 傅里叶变换红外光谱(FTIR)等测试手段对其进行了分析表征. 以Au@Fe3O4@RGO-MIP作为传感器识别元件的敏感材料, 利用循环伏安(CV)、 电化学阻抗谱(EIS)和差分脉冲(DPV)等电化学分析方法, 对构建的分子印迹电化学传感器进行了性能分析, 结果表明, 该传感器对水环境中DBP的响应平衡时间为6 min, 在0.01~0.1 μmol/L范围内, DBP浓度与响应电流之间呈现良好的线性关系, 检出限为0.3049 nmol/L(S/N=3).
中图分类号:
TrendMD:
李颖, 康君君, 赵雪茹, 徐文凯, 齐琦. 金修饰磁性石墨烯基分子印迹复合材料的制备及对水中邻苯二甲酸二正丁酯的电化学传感检测. 高等学校化学学报, 2019, 40(3): 448.
LI Ying, KANG Junjun, ZHAO Xueru, XU Wenkai, QI Qi. Preparation of Gold-modified Magnetic Graphene-based Molecularly Imprinted Composites and Electrochemical Sensing Detection of Dinbutyl Phthalate in Water†. Chem. J. Chinese Universities, 2019, 40(3): 448.

Fig.3 FTIR spectra(A) of GO(a), Au@RGO(b), Fe3O4@RGO(c), Au@Fe3O4@RGO(d) and Au@Fe3O4@RGO-MIP(e) and ultraviolet(UV) spectra(B) of Au@RGO(a), Au@Fe3O4@RGO(b) and Au@Fe3O4@RGO-MIP(c)
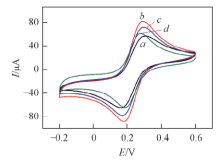
Fig.4 CV curves of Au@RGO(a), Au@Fe3O4@RGO(b), Au@Fe3O4@RGO-MIP(c), Au@Fe3O4@RGO-MIP(d) after binding templateCV curves were recorded between -0.5 V to 0.5 V in 5 mmol/L K3[Fe(CN)6] solution containing 0.1 mol/L KCl at a scan rate of 50 mV/s.
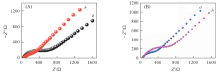
Fig.5 Electrochemical impedance spectra of Au@RGO(a), Au@Fe3O4@RGO(b)(A), Au@Fe3O4@RGO-MIP(a) and Au@Fe3O4@RGO-MIP(b)(B) after binding templateEIS of various electrodes were recorded between -0.5 V to 0.5 V in 5 mmol/L K3[Fe(CN)6] solution containing 0.1 mol/L KCl at a scan rate of 50 mV/s.
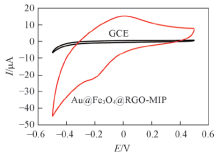
Fig.6 CV curves of DBP detectionCV curves of various electrodes were recorded between -0.5 V to 0.5 V in 0.1 mol/L PBS solution at a scan rate of 50 mV/s.
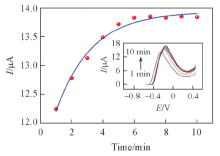
Fig.7 Adsorption kinetic curve on the response to DBP for Au@Fe3O4@RGO-MIPs electrode in PBS solution containing 10 μmol/L DBPThe insert is the DBP for Au@Fe3O4@RGO-MIP at different times. DPV of various electrodes were recorded between -0.5 V to 0.5 V in 0.1 mol/L PBS solution at a scan rate of 50 mV/s, the amplitude of 50 mV, the pulse width of 50 ms, pulse cycle of 200 ms.
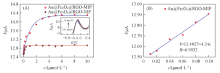
Fig.8 Different concentration curves of DBP on Au@Fe3O4@RGO-MIP and Au@Fe3O4@RGO-NIP(A) and linear relationship curve of Au@Fe3O4@RGO-MIP detects DBP(B)Inset is the DPV curves of Au@Fe3O4@RGO-MIP for different concentrations of DBP. DPV of various electrodes were recorded between -0.5 V to 0.5 V in 0.1 mol/L PBS solution at a scan rate of 50 mV/s, the amplitude of 50 mV, the pulse width of 50 ms, pulse cycle of 200 ms.
| Sensor | LOD/(nmol·L-1) | Linear range/(μmol·L-1) | Ref. |
|---|---|---|---|
| MWCNTs@GONRs/GCE | 25.15 | 1.44—229.93 | [ |
| DBP-MMIP-CL | 2.09 | 20.8—38400 | [ |
| MGO@Au NPs-MIPs | 0.80 | 2.5—5 | [ |
| Nano-Ni(OH)2 QCM | 17.96 | 0.018—0.072 | [ |
| Au@Fe3O4@RGO-MIP | 0.305 | 0.01—0.1 | This work |
Table 1 Comparison with the other electrochemical sensors for determination of DBP
| Sensor | LOD/(nmol·L-1) | Linear range/(μmol·L-1) | Ref. |
|---|---|---|---|
| MWCNTs@GONRs/GCE | 25.15 | 1.44—229.93 | [ |
| DBP-MMIP-CL | 2.09 | 20.8—38400 | [ |
| MGO@Au NPs-MIPs | 0.80 | 2.5—5 | [ |
| Nano-Ni(OH)2 QCM | 17.96 | 0.018—0.072 | [ |
| Au@Fe3O4@RGO-MIP | 0.305 | 0.01—0.1 | This work |
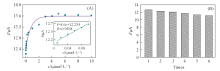
Fig.10 Response curve of Au@Fe3O4@RGO-MIP modified electrode to DBP concentration in lake water(A) and reproducibility of Au@Fe3O4@RGO-MIP for the detection of DBP(B)Inset of (A) is the calibration curve.
| [1] | Li J., Su Q., Li K. Y., Sun C. F., Zhang W. B., Food Chem., 2013, 141(4), 3714—3720 |
| [2] | Wang T., Wang J., Zhang C. L., Yang Z., Dal X. P., Cheng M. S., Hou X. H., Analyst, 2015, 140(15), 5308—5316 |
| [3] | Barp L., Purcaro G., Franchina F. A., Zoccali M., Sciarrone D., Tranchida P. Q., Mondello L., Anal. Chim. Acta, 2015, 887, 237—244 |
| [4] | Du L. P., Ma L. J., Qiao Y., Lu Y., Xiao D. G., Food Chem., 2016, 197, 1200—1206 |
| [5] | Liu M., Peng Q. Q., Cheng Y. F., Tang Q., Feng Q., Food Chem., 2015, 176, 12—16 |
| [6] | Liu Q., Yang H. J., Shi Y. X., Shu L., Chinese Journal of Eco-Agriculture, 2012, 20(8), 968—975 |
| (刘庆, 杨红军, 史衍玺, 舒龙. 中国生态农业学报, 2012, 20(8), 968—975) | |
| [7] | US Environmental Protection Agency, Phthalates Action Plan, 2012-03-12 |
| [8] | Zhang S. H., Yang Q., Li Z., Wang W. J., Zang X. H., Zang X. H., Wang C., Wang Z., Food Chem., 2019, 263, 258—264 |
| [9] | Xu W. Z., Zhang X. M., Huang W. H., Nie Y. J., Yang W. M., Chinese J. Anal. Chem., 2017, 45(4), 521—528 |
| (徐婉珍, 张枭明, 黄卫红, 聂仪晶, 杨文明. 分析化学, 2017, 45(4), 521—528) | |
| [10] | Javier G. S., Miguel Ángel G. C., Bárbara S. R., Javier H. B., Miguel Ángel R. D., Chemosphere, 2019, 201, 254—261 |
| [11] | Dai X. Y., Wu P., Environmental Science Guide, 2017, 36(1), 80—82 |
| (戴轩宇, 吴鹏. 环境科学导刊, 2017, 36(1), 80—82) | |
| [12] | Liang Y. R., Zhang Z. M., Liu Z. J., Wang K., Wu X. Y., Zeng K., Meng H., Zhang Z., Biosens. Bioelectron., 2017, 91, 199—202 |
| [13] | Yang X., Zhang Z. H., Cheng X., Zhang M. L., Luo L. J., Peng M. J., Nie L. H., Chinese J. Anal. Chem., 2012, 40(6), 870—876 |
| (杨潇, 张朝晖, 陈星, 张明磊, 罗丽娟, 彭密军, 聂丽华. 分析化学, 2012, 40(6), 870—876) | |
| [14] | Zeng H.H.., Li X. Q., Hao W. L., Zhang L. Z., Wei T., Zhao X. F., Liu Y. Y., Li J. H.,J. Hazard. Mater., 2017, 324, 250—257 |
| [15] | Zhao L. J., Gao L., Yi L. X., Jiang C., Zhao K., Spectroscopy and Spectral Analysis, 2017, 37(1), 109—113 |
| (赵丽君, 高磊, 衣玲学, 姜晨, 赵昆. 光谱学与光谱分析, 2017, 37(1), 109—113) | |
| [16] | He F., Tian Y. X., Xu Z. L., Luo L., Yang J. Y., Wang H., Sun Y. M., Du Q. F., Shen Y. D., J. Toxicol. Environ. Health Part A, 2019, 81(4), 80—88 |
| [17] | Feng N. X., Yu J., Mo C. H., Zhao H. M., Li Y. W., Wu B. X., Cai Q. Y., Li H., Zhou D. M., Wong M. H., Sci. Total Environ., 2019, 616, 117—127 |
| [18] | Guo J. B., Chen W., Jiang L. J., Ma F., Zheng G. C., Journal of Applied and Environmental Biology, 2014, 20(6), 1104—1110 |
| (郭静波, 陈微, 姜丽杰, 马放, 郑国臣. 应用与环境生物学报, 2014, 20(6), 1104—1110) | |
| [19] | Yáñez-Sedeño P., Campuzano S., Pingarrón J. M., Analytica Chimica Acta, 2017, 960, 1—17 |
| [20] | Zamora-Gálvez A., Ait-Lahcen A., Mercante L. A., Analytical Chemistry, 2016, 88(7), 3573—3584 |
| [21] | Han Q., Shen X., Zhu W. Y., Zhu C. H., Zhou X. M., Jiang H. J., Biosensors and Bioelectronics, 2016, 79, 180—186 |
| [22] | Yola M. L., Eren T., Atar N., Sens. Actuators, B, 2015, 210, 149—157 |
| [23] | Hummers Jr.W. S.., Offeman R. E.,J. Am. Chem. Soc., 1958, 80(6), 1339 |
| [24] | Kovtyukhova N. I., Ollivier P. J., Martin B. R., Mallouk T. E., Chizhik C. A., Buzaneva E. V., Gorchinskiy A. D., Chem. Mater., 1999, 11(3), 771—778 |
| [25] | Li S. H., Liu C. H., Yin G. H., Zhang Q., Luo J. H., Wu N. C., Biosens. Bioelectron., 2017, 91, 687—691 |
| [26] | Tanhaei M., Mahjoub A. R., Safarifard V., Mater. Lett., 2019, 41, 189—195 |
| [27] | Li D., Liu Y. R., Lin B. P., Sun Y., Yang H., Chemical Progress, 2015, 27(4), 404—415 |
| (李丹, 刘玉荣, 林保平, 孙莹, 杨洪. 化学进展, 2015, 27(4), 404—415) | |
| [28] | Lu X. J., Preparation, Characterization and Supercapacitance Characteristics of Graphene—Based Conductive Polymer Composites, Nanjing Aerospace University, Naijing, 2011 |
| (卢向军. 石墨烯基导电聚合物复合材料的制备、 表征及其超电容特性研究, 南京: 南京航空航天大学, 2011) | |
| [29] | Wang Y., Zhai F., Hasebe Y., Jia H. M., Zhang Z. Q., Bioelectrochem. Bioenerg., 2019, 122, 174—182 |
| [30] | Hao J. X., Wu K. B., Wan C. D., Tang Y., Talanta, 2019, 185, 550—556 |
| [31] | Mathew G., Dey P., Das R., Chowdhury S. D., Das M. P., Veluswamy P., Neppolian B., Das J., Biosens. Bioelectron., 2019, 115, 53—60 |
| [32] | Tung V. C., Allen M. J., Yang Y., Kaner R. B., Nat. Nanotechnol., 2009, 4(1), 25—29 |
| [33] | Zhang J., Ma J., Fan X., Peng W., Zhang G., Catal. Commun., 2017, 89, 148—151 |
| [34] | Dabiri M., Lehi N. F., Movahed S. K., Catal. Lett., 2016, 146(9), 1674—1686 |
| [35] | Wang D. M., Duan H. C., Lü J. H., Lü C. L., Journal of Materials Chemistry A, 2017, 5(10), 5088—5097 |
| [36] | Yan G. W., Zhang Y., Di W. H., Qin W. P., Dyes Pigm., 2019, 159, 28—34 |
| [37] | Qiu H.M.., Fan L. L., Li X. J., Li L. L., Sun M., Luo C. N.,J. Pharmaceut. Biomed. Anal., 2013, 75, 123—129 |
| [38] | Li X. J., Wang X. X., Li L. L., Duan H. M., Luo C. N., Talanta, 2015, 131, 354—360 |
| [39] | Hu R. H., Zhang K. H., Fan G. K., Luo Z. Y., Li G., Meas. Sci. Technol., 2015, 26, 055102 |
| [1] | 王瑞娜, 孙瑞粉, 钟添华, 池毓务. 大尺寸石墨烯量子点组装体的制备及电化学发光性能[J]. 高等学校化学学报, 2022, 43(8): 20220161. |
| [2] | 李玉龙, 谢发婷, 管燕, 刘嘉丽, 张贵群, 姚超, 杨通, 杨云慧, 胡蓉. 基于银离子与DNA相互作用的比率型电化学传感器用于银离子的检测[J]. 高等学校化学学报, 2022, 43(8): 20220202. |
| [3] | 王君旸, 刘争, 张茜, 孙春燕, 李红霞. DNA银纳米簇在功能核酸荧光生物传感器中的应用[J]. 高等学校化学学报, 2022, 43(6): 20220010. |
| [4] | 闫嘉森, 韩现英, 党兆涵, 李建刚, 何向明. 石蜡/膨胀石墨/石墨烯复合相变储热材料的制备及性能[J]. 高等学校化学学报, 2022, 43(6): 20220054. |
| [5] | 曹磊, 陈美君, 袁刚, 常钢, 张修华, 王升富, 何汉平. 冠醚功能化的栅控石墨烯场效应晶体管的制备及对汞离子的检测[J]. 高等学校化学学报, 2022, 43(4): 20210688. |
| [6] | 郑雪莲, 杨翠翠, 田维全. 全椅式边含薁缺陷石墨烯纳米片的二阶非线性光学性质[J]. 高等学校化学学报, 2022, 43(3): 20210806. |
| [7] | 杨隽阁, 高成乾, 李博鑫, 尹德忠. 改性氧化石墨烯稳定Pickering乳液法制备高导热相变整体材料[J]. 高等学校化学学报, 2022, 43(2): 20210593. |
| [8] | 张志博, 尚涵, 徐文轩, 韩广东, 崔金声, 杨皓然, 李瑞鑫, 张生辉, 徐欢. 氧化石墨烯插层聚β-羟基丁酸酯复合材料的结晶形态与宏观性能[J]. 高等学校化学学报, 2022, 43(2): 20210566. |
| [9] | 胡波, 朱昊辰. 双层氧化石墨烯纳米体系中受限水的介电常数[J]. 高等学校化学学报, 2022, 43(2): 20210614. |
| [10] | 俞彬, 谌小燕, 赵越, 陈卫昌, 肖新颜, 刘海洋. 氧化石墨烯基钴卟啉复合材料的电催化析氢反应[J]. 高等学校化学学报, 2022, 43(2): 20210549. |
| [11] | 王雪丽, 宋相伟, 解颜宁, 杜妮阳, 王振新. 部分还原氧化石墨烯的制备、 表征及对人宫颈癌HeLa细胞的体外杀伤作用[J]. 高等学校化学学报, 2022, 43(2): 20210595. |
| [12] | 魏闯宇, 陈艳丽, 姜建壮. 基于乙硫基取代的三层酞菁铕二聚体修饰ITO电极构筑电化学多巴胺和尿酸传感器[J]. 高等学校化学学报, 2022, 43(1): 20210582. |
| [13] | 马丽娟, 高升启, 荣祎斐, 贾建峰, 武海顺. Sc, Ti, V修饰B/N掺杂单缺陷石墨烯的储氢研究[J]. 高等学校化学学报, 2021, 42(9): 2842. |
| [14] | 赵凌云, 黄汉雄, 罗杜宇, 苏逢春. 复合材料柔软性对倒金字塔微结构阵列传感器性能的影响[J]. 高等学校化学学报, 2021, 42(9): 2953. |
| [15] | 潘晓君, 鲍容容, 潘曹峰. 可穿戴柔性触觉传感器的研究进展[J]. 高等学校化学学报, 2021, 42(8): 2359. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||