

高等学校化学学报 ›› 2015, Vol. 36 ›› Issue (10): 1945.doi: 10.7503/cjcu20150327
张艳青1,2, 郑华艳1, 章日光1, 李忠1( ), 王宝俊1, 赵秋勇2
), 王宝俊1, 赵秋勇2
收稿日期:2015-04-22
出版日期:2015-10-10
发布日期:2015-10-09
作者简介:联系人简介: 李 忠, 男, 博士, 教授, 主要从事催化和一碳化学研究. E-mail:基金资助:
ZHANG Yanqing1,2, ZHENG Huayan1, ZHANG Riguang1, LI Zhong1,*( ), WANG Baojun1, ZHAO Qiuyong2
), WANG Baojun1, ZHAO Qiuyong2
Received:2015-04-22
Online:2015-10-10
Published:2015-10-09
Contact:
LI Zhong
E-mail:lizhong@tyut.edu.cn
Supported by:摘要:
基于密度泛函理论方法构建并优化了CuMY(M为碱金属阳离子)分子筛的稳定构型, 采用速控步骤CO插入CH3O形成CH3OCO反应, 研究了碱金属阳离子对Cu+Y分子筛中活性中心周围电子环境及催化甲醇氧化羰基化合成碳酸二甲酯性能的影响. 计算结果表明, Li+, Na+和K+稳定落位于Y分子筛小笼中, 且随着金属离子半径的增大, CH3OH, CO, CH3O在CuMY上的吸附能和CO/CH3O的共吸附能均逐渐增加, CO插入CH3O反应的过渡态结构稳定性逐渐降低, 活化能逐渐上升, 相应的反应活性逐渐下降. 而落位在超笼中Ⅱ*位的Rb+与Cs+则随着离子半径的增大, 反应过渡态的结构稳定性提高, 克服的活化能降低, 反应活性升高. 不同CuMY分子筛上催化活性顺序为CuLiY-Ⅰ'>CuCsY-Ⅱ*>CuNaY-Ⅰ'>CuRbY-Ⅱ*>CuKY-Ⅰ'>CuCuY-Ⅰ', 其中CuLiY-Ⅰ'分子筛克服速控反应的活化能垒(52.74 kJ/mol)最低.
中图分类号:
TrendMD:
张艳青, 郑华艳, 章日光, 李忠, 王宝俊, 赵秋勇. 碱金属阳离子对Cu+Y催化甲醇氧化羰基化性能影响的密度泛函理论研究. 高等学校化学学报, 2015, 36(10): 1945.
ZHANG Yanqing, ZHENG Huayan, ZHANG Riguang, LI Zhong, WANG Baojun, ZHAO Qiuyong. Density Functional Theory Investigation on the Effect of Alkali Metal Cations on the Catalytic Performance for Cu+Y Zeolites in Oxidative Carbonylation of Methanol†. Chem. J. Chinese Universities, 2015, 36(10): 1945.
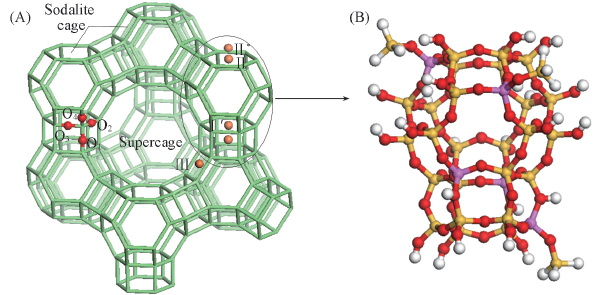
Fig.1 Structure of the faujasite(A) Stereodiagram of the faujasite-framework with cation sites and different crystallographic oxygen positions; (B) the cluster model Y zeolite. Red, yellow, purple, white and orange balls stand for O, Si, Al, H atoms, and cation sites, respectively.
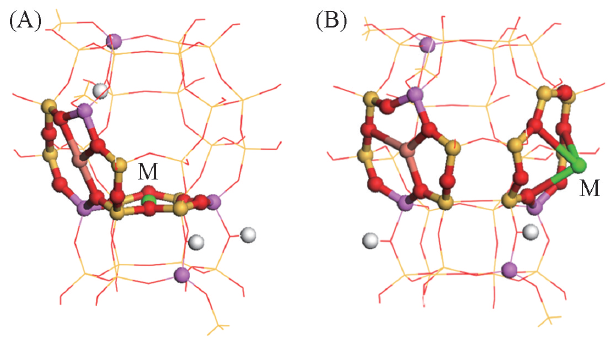
Fig.2 Stable structures of CuMY zeolitesOrange and green balls stand for Cu+ and alkali metal cations, respectively, others see Fig.1. (A) CuMY-Ⅰ'(M=Li, Na, K); (B) CuMY-Ⅱ*(M=Rb, Cs).
| Metal cation | Location | Eb/(kJ·mol-1) | q(M)/e |
|---|---|---|---|
| Li+ | Ⅰ' | 605.88 | 0.623 |
| Na+ | Ⅰ' | 525.34 | 0.747 |
| K+ | Ⅰ' | 444.98 | 0.804 |
| Rb+ | Ⅱ* | 387.96 | 0.784 |
| Cs+ | Ⅱ* | 355.92 | 0.860 |
Table 1 Location of alkali metal cations after opti-mization, the net charge[q(M)] and the binding energies(Eb) of metal cations at different sites of Y zeolites
| Metal cation | Location | Eb/(kJ·mol-1) | q(M)/e |
|---|---|---|---|
| Li+ | Ⅰ' | 605.88 | 0.623 |
| Na+ | Ⅰ' | 525.34 | 0.747 |
| K+ | Ⅰ' | 444.98 | 0.804 |
| Rb+ | Ⅱ* | 387.96 | 0.784 |
| Cs+ | Ⅱ* | 355.92 | 0.860 |
| Catalyst | Eb/(kJ·mol-1) | q(Cu)/e | q(M)/e | Electronic configuration |
|---|---|---|---|---|
| CuCuY-Ⅰ'[ | 647.39 | 0.393 | 0.340 | 3d9.9534s0.4254p0.229 |
| CuLiY-Ⅰ' | 658.63 | 0.385 | 0.617 | 3d9.9524s0.4334p0.230 |
| CuNaY-Ⅰ' | 672.20 | 0.367 | 0.758 | 3d9.9424s0.4554p0.235 |
| CuKY-Ⅰ' | 690.10 | 0.340 | 0.821 | 3d9.9314s0.4974p0.233 |
| CuRbY-Ⅱ* | 670.70 | 0.343 | 0.783 | 3d9.9414s0.4764p0.239 |
| CuCsY-Ⅱ*[ | 673.21 | 0.345 | 0.801 | 3d9.9434s0.4774p0.235 |
Table 2 Binding energies(Eb), net charge [q(Cu/M)] and electronic configurations of Cu+ of CuMY zeolite
| Catalyst | Eb/(kJ·mol-1) | q(Cu)/e | q(M)/e | Electronic configuration |
|---|---|---|---|---|
| CuCuY-Ⅰ'[ | 647.39 | 0.393 | 0.340 | 3d9.9534s0.4254p0.229 |
| CuLiY-Ⅰ' | 658.63 | 0.385 | 0.617 | 3d9.9524s0.4334p0.230 |
| CuNaY-Ⅰ' | 672.20 | 0.367 | 0.758 | 3d9.9424s0.4554p0.235 |
| CuKY-Ⅰ' | 690.10 | 0.340 | 0.821 | 3d9.9314s0.4974p0.233 |
| CuRbY-Ⅱ* | 670.70 | 0.343 | 0.783 | 3d9.9414s0.4764p0.239 |
| CuCsY-Ⅱ*[ | 673.21 | 0.345 | 0.801 | 3d9.9434s0.4774p0.235 |
| Catalyst | Eads/(kJ·mol-1) | ||||||
|---|---|---|---|---|---|---|---|
| Adsorbed CH3OH | Adsorbed CO | Co-adsorbed CO | Adsorbed CH3O | Co-adsorbed CH3O | Co-adsorbed CO/CH3O | TS | |
| CuCuY-Ⅰ' | 103.17 | 154.40 | 42.35 | 177.32 | 110.10 | 241.11 | 101.80 |
| CuLiY-Ⅰ' | 111.61 | 156.47 | 87.56 | 172.35 | 150.11 | 218.49 | 165.75 |
| CuNaY-Ⅰ' | 117.28 | 158.25 | 91.46 | 176.66 | 143.42 | 224.25 | 148.69 |
| CuKY-Ⅰ' | 124.29 | 162.37 | 95.15 | 179.79 | 113.17 | 234.61 | 141.98 |
| CuRbY-Ⅱ* | 117.88 | 165.34 | 96.98 | 193.58 | 157.53 | 230.12 | 141.77 |
| CuCsY-Ⅱ* | 103.17 | 153.66 | 95.89 | 175.11 | 145.62 | 222.59 | 167.19 |
Table 3 Adsorption energies of single CH3OH(CO, CH3O), co-adsorbed CO/CH3O and transition state(TS) involving in CO insertion into CH3O to CH3OCO on CuMY zeolites
| Catalyst | Eads/(kJ·mol-1) | ||||||
|---|---|---|---|---|---|---|---|
| Adsorbed CH3OH | Adsorbed CO | Co-adsorbed CO | Adsorbed CH3O | Co-adsorbed CH3O | Co-adsorbed CO/CH3O | TS | |
| CuCuY-Ⅰ' | 103.17 | 154.40 | 42.35 | 177.32 | 110.10 | 241.11 | 101.80 |
| CuLiY-Ⅰ' | 111.61 | 156.47 | 87.56 | 172.35 | 150.11 | 218.49 | 165.75 |
| CuNaY-Ⅰ' | 117.28 | 158.25 | 91.46 | 176.66 | 143.42 | 224.25 | 148.69 |
| CuKY-Ⅰ' | 124.29 | 162.37 | 95.15 | 179.79 | 113.17 | 234.61 | 141.98 |
| CuRbY-Ⅱ* | 117.88 | 165.34 | 96.98 | 193.58 | 157.53 | 230.12 | 141.77 |
| CuCsY-Ⅱ* | 103.17 | 153.66 | 95.89 | 175.11 | 145.62 | 222.59 | 167.19 |
| Catalyst | q/e | ||||
|---|---|---|---|---|---|
| CH3OH | CO | co-adsorbed CO | CH3O | co-adsorbed CH3O | |
| CuCuY-Ⅰ'[ | 0.247 | 0.426 | 0.395 | -0.116 | -0.124 |
| CuLiY-Ⅰ' | 0.173 | 0.404 | 0.354 | -0.065 | -0.158 |
| CuNaY-Ⅰ' | 0.241 | 0.417 | 0.364 | -0.066 | -0.121 |
| CuKY-Ⅰ' | 0.248 | 0.420 | 0.365 | -0.078 | -0.072 |
| CuRbY-Ⅱ* | 0.258 | 0.414 | 0.373 | -0.102 | -0.174 |
| CuCsY-Ⅱ*[ | 0.243 | 0.409 | 0.378 | -0.098 | -0.161 |
Table 4 Net charge of single CH3OH(CO, CH3O) and co-adsorbed CO/CH3O on CuMY zeolites
| Catalyst | q/e | ||||
|---|---|---|---|---|---|
| CH3OH | CO | co-adsorbed CO | CH3O | co-adsorbed CH3O | |
| CuCuY-Ⅰ'[ | 0.247 | 0.426 | 0.395 | -0.116 | -0.124 |
| CuLiY-Ⅰ' | 0.173 | 0.404 | 0.354 | -0.065 | -0.158 |
| CuNaY-Ⅰ' | 0.241 | 0.417 | 0.364 | -0.066 | -0.121 |
| CuKY-Ⅰ' | 0.248 | 0.420 | 0.365 | -0.078 | -0.072 |
| CuRbY-Ⅱ* | 0.258 | 0.414 | 0.373 | -0.102 | -0.174 |
| CuCsY-Ⅱ*[ | 0.243 | 0.409 | 0.378 | -0.098 | -0.161 |
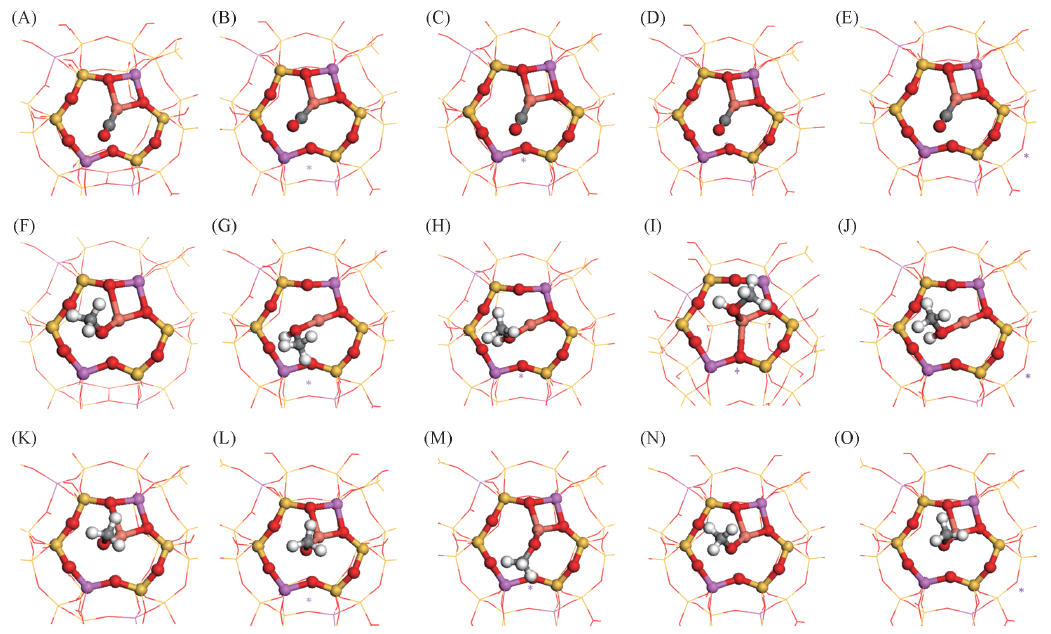
Fig.3 Stable configurations of CO, CH3OH and CH3O adsorbed on CuY and CuMY zeolitesBond lengths are in nm.(A) CO on CuCuY-Ⅰ', dC—O=0.1148, dCu—C=0.1788; (B) CO on CuLiY-Ⅰ' , dC—O=0.1148, dCu—C=0.1787; (C) CO on CuNaY-Ⅰ' , dC—O=0.1154, dCu—C=0.1784; (D) CO on CuKY-Ⅰ' , dC—O=0.1148, dCu—C=0.1788; (E) CO on CuRbY-Ⅱ* , dC—O=0.1149, dCu—C=0.1786; (F) CH3OH on CuCuY-Ⅰ' , dCu—OCH4=0.1910; (G) CH3OH on CuLiY-Ⅰ' , dCu—OCH4=0.1929; (H) CH3OH on CuNaY-Ⅰ' , dCu—OCH4=0.1919; (I) CH3OH on CuKY-Ⅰ' , dCu—OCH4=0.2098; (J) CH3OH on CuRbY-Ⅱ*, dCu—OCH4=0.1932; (K) CH3O on CuCuY-Ⅰ' , dCu—OCH3=0.1810; (L) CH3O on CuLiY-Ⅰ' , dCu—OCH3=0.1804; (M) CH3O on CuNaY-Ⅰ' , dCu—OCH3=0.1801; (N) CH3O on CuKY-Ⅰ', dCu—OCH3=0.1799; (O) CH3O on CuRbY-Ⅱ* , dCu—OCH3=0.1808.
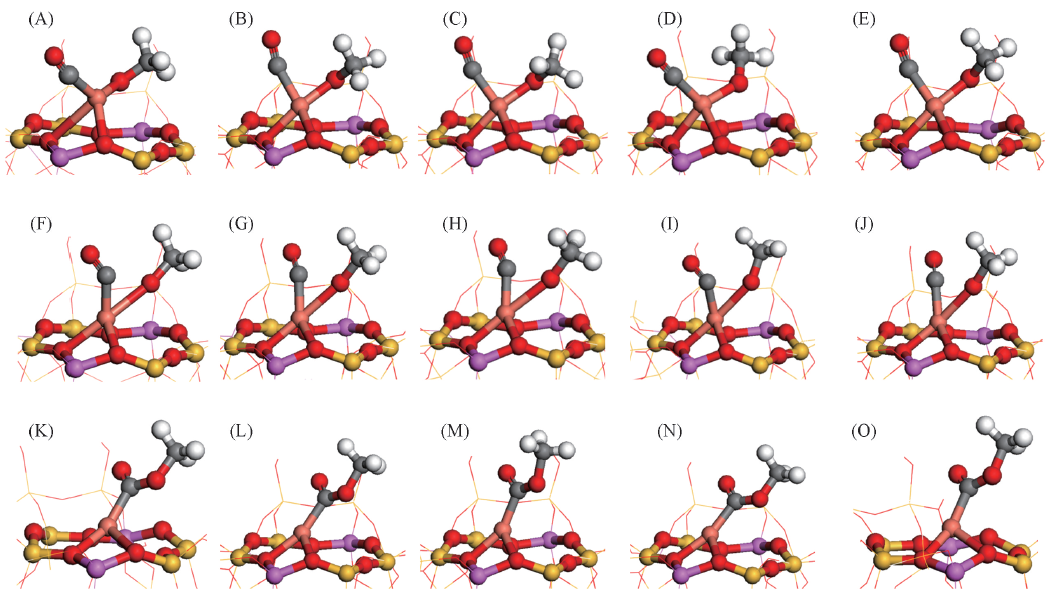
Fig.4 Stable configurations of co-adsorbed CO and CH3O on CuMY zeolitesBond lengths are in nm.(A) CO/CH3O on CuCuY-Ⅰ' , dC—O=0.1145, dCu—O=0.1844, dC—OCH3=0.3312; (B) CO/CH3O on CuLiY-Ⅰ', dC—O=0.1142, dCu—O=0.1882, dC—OCH3=0.2488; (C) CO/CH3O on CuNaY-Ⅰ', dC—O=0.1140, dCu—O=0.1879, dC—OCH3=0.2670; (D) CO/CH3O on CuKY-Ⅰ' , dC—O=0.1142, dCu—O=0.1845, dC—OCH3=0.3123; (E) CO/CH3O on CuRbY-Ⅱ* , dC—O=0.1140, dCu—O=0.1889, dC—OCH3=0.2539; (F) TS on CuCuY-Ⅰ', dC—O=0.1150, dCu—O=0.2356, dC—OCH3=0.2041; (G) TS on CuLiY-Ⅰ', dC—O=0.1179, dCu—O=0.2298, dC—OCH3=0.1845; (H) TS on CuNaY-Ⅰ', dC—O=0.1150, dCu—O=0.2353, dC—OCH3=0.1889; (I) TS on CuKY-Ⅰ', dC—O=0.1193, dCu—O=0.2414, dC—OCH3=0.2012; (J) TS on CuRbY-Ⅱ* , dC—O=0.1180, dCu—O=0.2275, dC—OCH3=0.1858; (K) CH3OCO on CuCuY-Ⅰ' , dC—O=0.1225, dCu—O=0.2817, dC—OCH3=0.1336; (L) CH3OCO on CuLiY-Ⅰ', dC—O=0.1224, dCu—O=0.2824, dC—OCH3=0.1332; (M) CH3OCO on CuNaY-Ⅰ' , dC—O=0.1224, dCu—O=0.2805, dC—OCH3=0.1332; (N) CH3OCO on CuKY-Ⅰ', dC—O=0.1223, dCu—O=0.2816, dC—OCH3=0.1331; (O) CH3OCO on CuRbY-Ⅱ* , dC—O=0.1220, dCu—O=0.2726, dC—OCH3=0.1339.
| [1] | Keller N., Rebmann G., Keller V., J. Mol. Catal. A:Chem., 2010, 317(1/2), 1—18 |
| [2] | Pacheco M. A., Marshall C.L., Energ. Fuel, 1997, 11(1), 2—29 |
| [3] | Romano U., Tesel R., Maurl M. M., Rebora P., Ind. Eng. Chem. Prod. Res. Dev., 1980, 19(3), 396—403 |
| [4] | Delledonne D., Rivetti F., Romano U., Appl. Catal. A: Gen., 2001, 221(1/2), 241—251 |
| [5] | Richter M., Fait M. J. G., Eckelt R., Schneider M., Radnik J., Heidemann D., Fricke R., J. Catal., 2007, 245(1), 11—24 |
| [6] | Zhang Y. H., Briggs D. N., Smit E. D., Bell A. T., J. Catal., 2007, 251(2), 443—452 |
| [7] | King S.T., J. Catal., 1996, 161(2), 530—538 |
| [8] | Gao Z., He M.Y., Dai Y. Y., Zeolite Catalysis and Separation Technology, China Petrochemical Press, Beijing, 1999 |
| (高滋, 何鸣元, 戴逸云. 沸石催化与分离技术, 北京: 中国石化出版社, 1999) | |
| [9] | Zheng H., Qi J., Zhang R., Li Z., Wang B., Ma X., Fuel Process. Technol., 2014, 128(0), 310—318 |
| [10] | Fu T., Zheng H., Niu Y., Wang R., Li Z., Acta Chimica Sinica, 2011, 69(15), 1765—1772 |
| (付廷俊, 郑华艳, 牛燕燕, 王瑞玉, 李忠. 化学学报,2011, 69(15), 1765—1772) | |
| [11] | Kieger S., Delahay G., Coq B., Appl. Catal. B: Environ., 2000, 25(1), 1—9 |
| [12] | Zhang Z., Wong T.T., Sachtler W.M.H., J. Catal., 1991, 128(1), 13—22 |
| [13] | Keane M. A., Microporous Mater., 1995, 3(4/5), 385—394 |
| [14] | Seiki T., Nakato A., Nishiyama S., Tsuruya S., Phys. Chem. Chem. Phys., 2003, 5(17), 3818—3826 |
| [15] | Ren J., Wang D.L., Pei Y.L., Qin Z. F., Lin J.Y., Li Z., 2013, 34(11), 2594—2600 |
| (任军, 王冬蕾, 裴永丽, 秦志峰, 林建英, 李忠. 高等学校化学学报,2013, 34(11), 2594—2600) | |
| [16] | Nam J. K., Choi M. J., Cho D. H., Suh J. K., Kim S. B., J. Mol. Catal. A:Chem., 2013, 370(0), 7—13 |
| [17] | Zhang Y.H., Bell A.T., J. Catal., 2008, 255(2), 153—161 |
| [18] | Delley B., J.Chem.Phys., 2000, 113(18), 7756—7764 |
| [19] | Delley B., J.Chem.Phys., 1990, 92(1), 508—517 |
| [20] | Perdew J. P., Burke K., Ernzerhof M., Phys. Rev. Lett., 1996, 77(18), 3865—3868 |
| [21] | Halgren T.A., Lipscomb W.N., Chem. Phys. Lett., 1977, 49(2), 225—232 |
| [22] | Zheng X., Bell A.T., J. Phys. Chem. C, 2008, 112(13), 5043—5047 |
| [23] | Drake I.J., Zhang Y., Briggs D., Lim B., Chau T., Bell A. T., J. Phys. Chem. B, 2006, 110(24), 11654—11664 |
| [24] | Rejmak P., Sierka M., Sauer J., Phys. Chem. Chem. Phys., 2007, 9(40), 5446—5456 |
| [25] | Hill J. R., Freeman C.M., Delley B., J. Phys. Chem. A, 1999, 103(19), 3772—3777 |
| [26] | Sierka M., Eichler U., Datka J., Sauer J., J. Phys. Chem. B, 1998, 102(33), 6397—6404 |
| [27] | Kim H.S., Bae D., Lim W. T. , Seff K., J. Phys. Chem. C, 2012, 116(16), 9009—9018 |
| [28] | Su H., Kim H. S., Seo S. M., Ko S. O., Suh J. M., Kim G. H., Lim W. T., Bull. Korean Chem. Soc., 2012, 33(8), 2785—2788 |
| [29] | Frising T., Leflaive P., Microporous Mesoporous Mater., 2008, 114(1—3), 27—63 |
| [30] | Berthomieu D., Ducéré J. M., Goursot A., J. Phys. Chem. B, 2002, 106(30), 7483—7488 |
| [31] | Palomino G. T., Bordiga S., Zecchina A., J. Phys. Chem. B, 2000, 104(36), 8641—8651 |
| [32] | Berthomieu D., Krishnamurty S., Coq B., Delahay G., Goursot A., J. Phys. Chem. B, 2001, 105(6), 1149—1156 |
| [33] | Anderson S.A., Root T.W., J. Mol. Catal. A: Chem., 2004, 220(2), 247—255 |
| [34] | Bak J.,Clausen S., Appl. Spectrosc., 1999, 53(6), 697—700 |
| [35] | Zhang R., Song L., Wang B., Li Z., J. Comput. Chem., 2012, 33(11), 1101—1110 |
| [36] | Zhang R., Wang G., Wang B., J. Catal., 2013, 305, 238—255 |
| [37] | Hammond G. S.,J. Am. Chem. Soc., 1955, 77(2), 334—338 |
| [1] | 何鸿锐, 夏文生, 张庆红, 万惠霖. 羟基氧化铟团簇与二氧化碳和甲烷作用的密度泛函理论研究[J]. 高等学校化学学报, 2022, 43(8): 20220196. |
| [2] | 黄汉浩, 卢湫阳, 孙明子, 黄勃龙. 石墨炔原子催化剂的崭新道路:基于自验证机器学习方法的筛选策略[J]. 高等学校化学学报, 2022, 43(5): 20220042. |
| [3] | 刘洋, 李旺昌, 张竹霞, 王芳, 杨文静, 郭臻, 崔鹏. Sc3C2@C80与[12]CPP纳米环之间非共价相互作用的理论研究[J]. 高等学校化学学报, 2022, 43(11): 20220457. |
| [4] | 程媛媛, 郗碧莹. ·OH自由基引发CH3SSC |
| [5] | 王园月, 安梭梭, 郑旭明, 赵彦英. 5-巯基-1, 3, 4-噻二唑-2-硫酮微溶剂团簇的光谱和理论计算研究[J]. 高等学校化学学报, 2022, 43(10): 20220354. |
| [6] | 周成思, 赵远进, 韩美晨, 杨霞, 刘晨光, 贺爱华. 硅烷类外给电子体对丙烯-丁烯序贯聚合的调控作用[J]. 高等学校化学学报, 2022, 43(10): 20220290. |
| [7] | 钟声广, 夏文生, 张庆红, 万惠霖. 电中性团簇MCu2Ox(M=Cu2+, Ce4+, Zr4+)上甲烷和二氧化碳直接合成乙酸的理论研究[J]. 高等学校化学学报, 2021, 42(9): 2878. |
| [8] | 马丽娟, 高升启, 荣祎斐, 贾建峰, 武海顺. Sc, Ti, V修饰B/N掺杂单缺陷石墨烯的储氢研究[J]. 高等学校化学学报, 2021, 42(9): 2842. |
| [9] | 黄罗仪, 翁约约, 黄旭慧, 王朝杰. 车前草中黄酮类成分结构和性质的理论研究[J]. 高等学校化学学报, 2021, 42(9): 2752. |
| [10] | 王建, 张红星. 四配位铂磷光发射体结构与光物理性质关系的理论研究[J]. 高等学校化学学报, 2021, 42(7): 2245. |
| [11] | 胡伟, 刘小峰, 李震宇, 杨金龙. 金刚石纳米线氮空位色心的表面与尺寸效应[J]. 高等学校化学学报, 2021, 42(7): 2178. |
| [12] | 杨一莹, 朱荣秀, 张冬菊, 刘成卜. 金催化炔基苯并二𫫇英环化合成8-羟基异香豆素的理论研究[J]. 高等学校化学学报, 2021, 42(7): 2299. |
| [13] | 柳扬, 李清波, 孙杰, 赵显. Ga对在AlN衬底上直接生长石墨烯的远程催化[J]. 高等学校化学学报, 2021, 42(7): 2271. |
| [14] | 郑若昕, 张颖, 徐昕. 低标度XYG3双杂化密度泛函的开发与测评[J]. 高等学校化学学报, 2021, 42(7): 2210. |
| [15] | 应富鸣, 计辰儒, 苏培峰, 吴玮. 基于完全活性空间自洽场的杂化多组态密度泛函方法λ-DFCAS[J]. 高等学校化学学报, 2021, 42(7): 2218. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||