

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (6): 1140.doi: 10.7503/cjcu20150857
• Physical Chemistry • Previous Articles Next Articles
KONG Danni, JIANG Tao, ZHANG Yiying, CAO Fahai*( )
)
Received:2015-11-08
Online:2016-06-10
Published:2016-05-26
Contact:
CAO Fahai
E-mail:fhcao@ecust.edu.cn
Supported by:CLC Number:
TrendMD:
KONG Danni, JIANG Tao, ZHANG Yiying, CAO Fahai. Catalytic Performance of Activated Carbon Supported Pt-Ni Bimetallic Catalyst for Glycerol in situ Hydrogenolysis†[J]. Chem. J. Chinese Universities, 2016, 37(6): 1140.
| Catalyst | Specific surface areaa/(m2·g-1) | Pore volumea/(cm3·g-1) | Average pore diametera/nm | Mean particle sizeb/nm |
|---|---|---|---|---|
| AC | 806 | 0.53 | 5.3 | |
| Pt-Fe/AC | 783 | 0.45 | 5.1 | 2.3 |
| Pt-Ni/AC | 768 | 0.49 | 5.1 | 2.1 |
| Pt-Co/AC | 763 | 0.51 | 5.2 | 2.3 |
| Pt-Zn/AC | 691 | 0.52 | 5.2 | 2.3 |
| Pt-Cu/AC | 523 | 0.49 | 4.7 | 4.9 |
Table 1 Physicochemical property of Pt-M/AC catalysts
| Catalyst | Specific surface areaa/(m2·g-1) | Pore volumea/(cm3·g-1) | Average pore diametera/nm | Mean particle sizeb/nm |
|---|---|---|---|---|
| AC | 806 | 0.53 | 5.3 | |
| Pt-Fe/AC | 783 | 0.45 | 5.1 | 2.3 |
| Pt-Ni/AC | 768 | 0.49 | 5.1 | 2.1 |
| Pt-Co/AC | 763 | 0.51 | 5.2 | 2.3 |
| Pt-Zn/AC | 691 | 0.52 | 5.2 | 2.3 |
| Pt-Cu/AC | 523 | 0.49 | 4.7 | 4.9 |
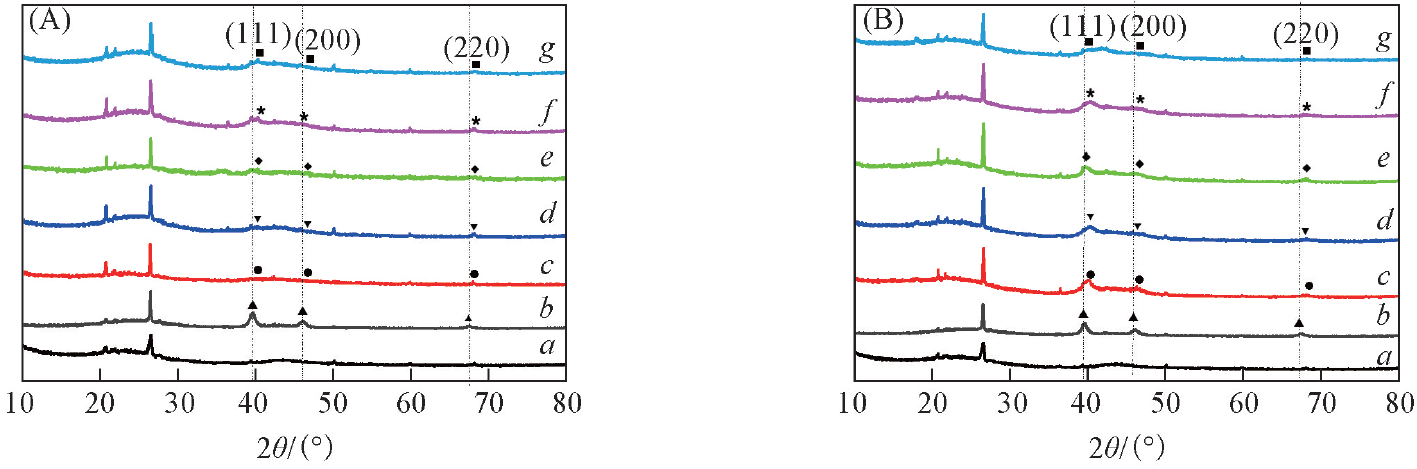
Fig.1 XRD patterns of the Pt-M/AC catalysts before(A) and after(B) reactiona. AC; b. Pt/AC; c. Pt-Ni/AC; d. Pt-Co/AC; e. Pt-Fe/AC; f. Pt-Zn/AC; g. Pt-Cu/AC.
| Catalyst | Conversion(%) | Reaction rate/ (μmol·g-1·min-1) | Selectivity(%) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 1,2-PDO | EG | 2-PO | 1-PO | CH4 | CO2 | Othersb | |||
| 2%Pt-2%Fe/AC | 99.8 | 169.6 | 48.7 | 7.5 | 9.8 | 9.0 | 1.5 | 13.0 | 10.5 |
| 2%Pt-2%Ni/AC | 98.3 | 167.0 | 60.5 | 9.1 | 9.9 | 7.8 | 1.1 | 10.5 | 1.1 |
| 2%Pt-2%Co/AC | 94.2 | 159.9 | 60.1 | 6.9 | 10.5 | 9.5 | 0.5 | 10.4 | 2.1 |
| 2%Pt-2%Zn/AC | 85.1 | 144.6 | 50.3 | 10.7 | 12.0 | 9.8 | 0.8 | 10.1 | 6.3 |
| 2%Pt-2%Cu/AC | 54.7 | 92.9 | 58.5 | 12.2 | 9.1 | 3.1 | 0.1 | 2.6 | 14.4 |
Table 2 Glycerol in situ hydrogenolysis on different Pt-M/AC catalystsa
| Catalyst | Conversion(%) | Reaction rate/ (μmol·g-1·min-1) | Selectivity(%) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 1,2-PDO | EG | 2-PO | 1-PO | CH4 | CO2 | Othersb | |||
| 2%Pt-2%Fe/AC | 99.8 | 169.6 | 48.7 | 7.5 | 9.8 | 9.0 | 1.5 | 13.0 | 10.5 |
| 2%Pt-2%Ni/AC | 98.3 | 167.0 | 60.5 | 9.1 | 9.9 | 7.8 | 1.1 | 10.5 | 1.1 |
| 2%Pt-2%Co/AC | 94.2 | 159.9 | 60.1 | 6.9 | 10.5 | 9.5 | 0.5 | 10.4 | 2.1 |
| 2%Pt-2%Zn/AC | 85.1 | 144.6 | 50.3 | 10.7 | 12.0 | 9.8 | 0.8 | 10.1 | 6.3 |
| 2%Pt-2%Cu/AC | 54.7 | 92.9 | 58.5 | 12.2 | 9.1 | 3.1 | 0.1 | 2.6 | 14.4 |
| Catalyst | Conversion(%) | Reaction rate/ (μmol·g-1·min-1) | Selectivity(%) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 1,2-PDO | EG | 2-PO | 1-PO | CH4 | CO2 | Othersb | |||
| 2%Ni/AC | 45.7 | 77.6 | 50.6 | 35.8 | 13.6 | 0 | 0 | 0 | 0 |
| 2%Pt/AC | 80.5 | 136.7 | 45.1 | 4.6 | 15.4 | 14.2 | 2.9 | 14.9 | 2.9 |
| 1%Pt-1%Ni/AC | 85.6 | 145.4 | 56.3 | 5.8 | 11.6 | 11.9 | 1.6 | 11.5 | 1.3 |
| 2%Pt-1%Ni/AC | 93.0 | 158.0 | 60.9 | 8.2 | 10.6 | 5.6 | 1.0 | 12.6 | 1.1 |
| 5%Pt-1%Ni/AC | 98.5 | 167.3 | 56.8 | 8.2 | 11.2 | 7.6 | 1.4 | 13.2 | 1.6 |
| 2%Pt-2%Ni/AC | 98.3 | 167.0 | 60.5 | 9.1 | 9.9 | 7.8 | 1.3 | 10.3 | 1.1 |
| 2%Pt-3%Ni/AC | 98.0 | 166.4 | 58.3 | 8.8 | 9.7 | 7.2 | 2.3 | 12.3 | 1.4 |
| 2%Pt-5%Ni/AC | 97.8 | 166.1 | 57.8 | 9.4 | 9.3 | 7.1 | 2.7 | 12.1 | 1.6 |
Table 3 Glycerol in situ hydrogenolysis on Pt-Ni/AC catalysts with different mass ratios of Pt/Nia
| Catalyst | Conversion(%) | Reaction rate/ (μmol·g-1·min-1) | Selectivity(%) | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 1,2-PDO | EG | 2-PO | 1-PO | CH4 | CO2 | Othersb | |||
| 2%Ni/AC | 45.7 | 77.6 | 50.6 | 35.8 | 13.6 | 0 | 0 | 0 | 0 |
| 2%Pt/AC | 80.5 | 136.7 | 45.1 | 4.6 | 15.4 | 14.2 | 2.9 | 14.9 | 2.9 |
| 1%Pt-1%Ni/AC | 85.6 | 145.4 | 56.3 | 5.8 | 11.6 | 11.9 | 1.6 | 11.5 | 1.3 |
| 2%Pt-1%Ni/AC | 93.0 | 158.0 | 60.9 | 8.2 | 10.6 | 5.6 | 1.0 | 12.6 | 1.1 |
| 5%Pt-1%Ni/AC | 98.5 | 167.3 | 56.8 | 8.2 | 11.2 | 7.6 | 1.4 | 13.2 | 1.6 |
| 2%Pt-2%Ni/AC | 98.3 | 167.0 | 60.5 | 9.1 | 9.9 | 7.8 | 1.3 | 10.3 | 1.1 |
| 2%Pt-3%Ni/AC | 98.0 | 166.4 | 58.3 | 8.8 | 9.7 | 7.2 | 2.3 | 12.3 | 1.4 |
| 2%Pt-5%Ni/AC | 97.8 | 166.1 | 57.8 | 9.4 | 9.3 | 7.1 | 2.7 | 12.1 | 1.6 |
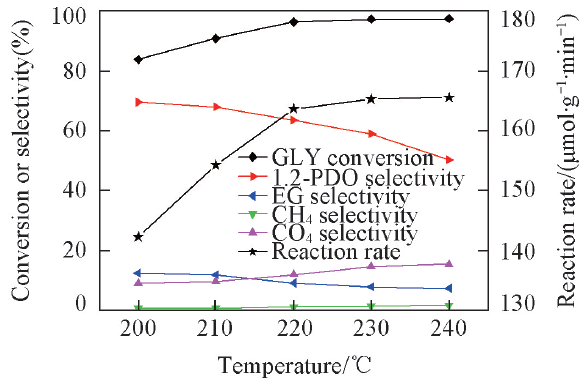
Fig.5 Effect of reaction temperature on glycerol in situ hydrogenolysisReaction conditions: 0.2 g catalyst(2%Pt-2%Ni/AC), 1.0 MPa N2 pressure, 15 g 10%(mass fraction) glycerol aqueous solution, 8 h.
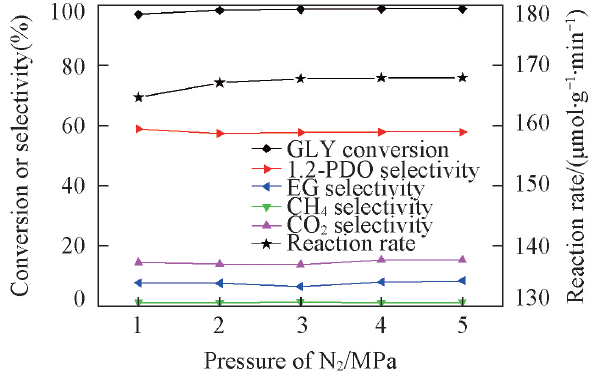
Fig.6 Effect of N2 pressure on glycerol in situ hydrogenolysisReaction conditions: 0.2 g catalyst(2%Pt-2%Ni/AC), 220 ℃, 15 g 10%(mass fraction) glycerol aqueous solution, 8 h.
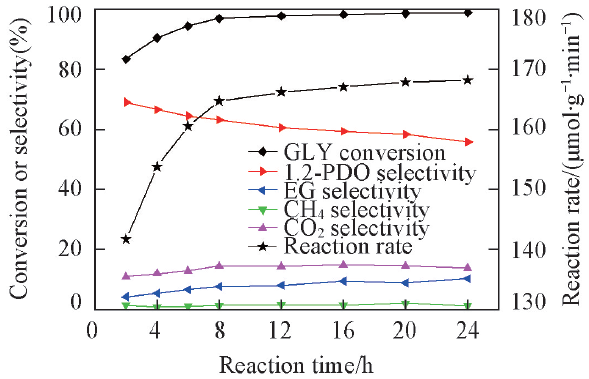
Fig.7 Effect of reaction time on glycerol in situ hydrogenolysisReaction condition: 0.2 g catalyst(2%Pt-2%Ni/AC), 220 ℃, 1.0 MPa N2 pressure, 15 g 10%(mass fraction) glycerol aqueous solution
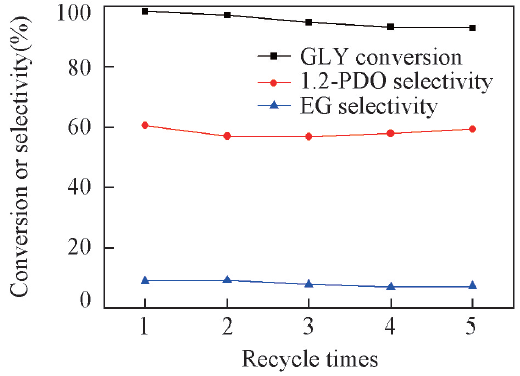
Fig.8 Reuse of Pt-Ni/AC catalyst for glycerol in situ hydrogenolysiReaction condition: 0.2 g catalyst(2%Pt-2%Ni/AC), 1.0 MPa N2 pressure, 15 g 10%(mass fraction) glycerol aqueous solution, 220 ℃, 8 h.
| [1] | Zhou C. H., Beltramini J. N., Fan Y. X., Lu G. Q., Chem. Soc. Rev., 2008, 37, 527—549 |
| [2] | Dam J. T., Hanefeld D., Chem. Sus. Chem., 2011, 4, 1017—1034 |
| [3] | Wang S., Yin K.H., Zhang Y. C., Liu H. C., ACS Catalysis, 2013, 3, 2112—2121 |
| [4] | Yu W. Q., Zhao J., Ma H., Miao H., Song Q., Xua J., Appl. Catal. A, 2010, 383, 73—78 |
| [5] | Kurosaka T., Maruyama H., Naribayashi I., Sasaki Y., Catal. Commun., 2008, 9, 1360—1363 |
| [6] | Martin A., Armbruster U., Gandarias I., Arias P. L., Eur. J. Lipid Sci. Technol., 2013, 115, 9—27 |
| [7] | D’Hondt E., de Vyver S. V., Sels B. F., Jacobs P. A.,Chem. Commun., 2008, 6011—6012 |
| [8] | Roy D., Subramaniam B., Chaudhari R. V., Catal. Today, 2010, 156, 31—37 |
| [9] | Yin A. Y., Guo X. Y., Dai W. L., Fan K. N., Green Chem., 2009, 11, 1514—1516 |
| [10] | Barbelli M. L., Santori G. F., Nichio N. N., Bioresour. Technol., 2012, 111, 500—503 |
| [11] | Meryemoglu B., Hesenov A., Irmak S., Atanur O. M., Erbatur O., Int. J. Hydrogen Energy, 2010, 35(22), 12580—12587 |
| [12] | Venkateswara R. C., Viswanathan B., J. Phys. Chem. C, 2009, 113, 18907—18913 |
| [13] | Antolini E., Salgado J. R. C., da Silva R. C., Gonzalez E. R., Mater. Chem. Phys., 2007, 101, 395—403 |
| [14] | Hu Y. J., Wu P., Zhang H., Cai C. X., Electrochim. Acta, 2012, 85, 314—321 |
| [15] | Zhao Y. C., Yang X. L., Tian J. N., Wang F. Y., Zhan L., Int. J. Hydrogen Energy, 2010, 35, 3249—3257 |
| [16] | Kyung W. P., Jong H. C., Boo K. K., Seol A. L., Yung E. S., J. Phys. Chem. B, 2002, 106, 1869—1877 |
| [17] | Dasari M. A., Kiatsimkul P. P., Sutterlin W. R., Suppes G. J., Appl. Catal. A, 2004, 281, 225—231 |
| [18] | Wang S., Zhang D., Ma Y. Y., Zhang H., Gao J., Nie Y. T., Sun X. H., Appl. Mater. Interfaces, 2014, 6, 12429—12435 |
| [19] | Wang Z. B., Zuo P. J., Wang G. J., Du C. Y., Yin G. P., J. Phys. Chem. C, 2008, 112, 6582—6587 |
| [20] | Gao J., Hou Z. Y., Liu X. S., Zeng Y. W., Luo M. F., Zheng X. M., Int. J. Hydrogen Energy, 2009, 34(9), 3734—3742 |
| [21] | Zhang H., Yin Y. J., Hu Y. J., Li C.Y., Wu P., Wei S.H., Cai C. X., J. Phys. Chem. C, 2010, 114, 11861—11867 |
| [22] | Liang Y. M., Zhang H. M., Zhong H. X., Zhu X. B., Tian Z. Q., Xu Q. Y., Yi B. L., J. Catal., 2006, 238, 468—476 |
| [23] | Hu Y. J., He F. Y., Ben A. L., Chen C. Y., J. Electroanal. Chem., 2014, 726, 55—61 |
| [24] | Mu R., Fu Q., Xu H., Zhang H., Huang Y.Y., Jiang Z., Zhang S., Tan D. L., Bao X. H., J. Am. Chem. Soc., 2011, 133,1978—1986 |
| [25] | Jiang T., Huai Q., Geng T., Ying W. Y., Xiao T. C., Cao F. H., Biomass Bioenergy, 2015, 78, 71—79 |
| [26] | Gordon D., Weather B., Calvin H., J. Catal., 1982, 77, 460—472 |
| [27] | Feng J., Fu H. Y., Wang J. B., Li R. X., Chen H., Li X. J., Catal. Commun., 2008, 9, 1458—1464 |
| [1] | GE Yicong, NIE Wanli, SUN Guofeng, CHEN Jiaxuan, TIAN Chong. Silver-catalyzed [5+1] Cyclization of 2-Vinylanilines with Benzisoxazoles [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220142. |
| [2] | WANG Mingzhi, ZHENG Yanping, WENG Weizheng. Catalytic Methane Combustion over CeO2 Supported PdO and Ce1‒x Pd x O2‒δ Species [J]. Chem. J. Chinese Universities, 2022, 43(4): 20210816. |
| [3] | GAO Jing, HE Wentao, WANG Xinxin, XIANG Yushu, LONG Lijuan, QIN Shuhao. Preparation of DOPO Derivative Modified Carbon Nanotubes and Their Effect on Flame Retardancy of Polylactic Acid [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210670. |
| [4] | LI Xiaohui, WEI Aijia, MU Jinping, HE Rui, ZHANG Lihui, WANG Jun, LIU Zhenfa. Effects of SmPO4 Coatingon Electrochemical Performance of High-voltage LiNi0.5Mn1.5O4 Cathode Materials [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210546. |
| [5] | ZHANG Liling, LIU Liu, ZHENG Mingqiu, FANG Wenkai, LIU Da, TANG Hongwu. Dual Signal Detection of HPV16 DNA by CRISPR/Cas12a Biosensing System Based on Upconversion Luminescent Resonance Energy Transfer [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220412. |
| [6] | WANG Yuanyue, AN Suosuo, ZHENG Xuming, ZHAO Yanying. Spectroscopic and Theoretical Studies on 5-Mercapto-1,3,4-thiadiazole-2-thione Microsolvation Clusters [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220354. |
| [7] | LI Dan, XIAO Liping, FAN Jie. Inorganic-based Surface Materials with Anti-SARS-CoV-2 Properties and Their Mechanisms of Action [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220301. |
| [8] | LIANG Yu, LIU Huan, GONG Lige, WANG Chunxiao, WANG Chunmei, YU Kai, ZHOU Baibin. Synthesis and Supercapacitor Properties of Biimidazole-modified {SiW12O40} Hybrid [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210556. |
| [9] | ZHOU Yonghui, HUANG Rujun, YAN Jianyang, LI Yajun, QIU Huanhuan, YANG Jinxuan, ZHENG Youxuan. Synthesis and Electroluminescence Properties of Two Iridium(Ⅲ) Complexes with Nitrogen Heterocycle Structures [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210415. |
| [10] | LIU Huazheng, PAN Xiaoguang, LI Hua, WAN Renzhong, LIU Xigong. Na2CO3-catalyzed 1,6-Conjugate Addition of Trimethylsilyl Azide to δ-CF3-δ-Aryl-disubstituted Para-Quinone Methides: Efficient Construction of Diarylmethanes Bearing CF3- and N3-Substituted Quaternary Stereocenters [J]. Chem. J. Chinese Universities, 2021, 42(9): 2772. |
| [11] | LI Haibo, XIAO Changfa, JIANG Long, HUANG Yun, DAN Yi. Copolymerization of Methyl Acrylate and 1-Octene Catalyzed by the Loaded Aluminum Chloride on MCM-41 Molecular Sieve [J]. Chem. J. Chinese Universities, 2021, 42(9): 2974. |
| [12] | LUO Qiangqiang, JIN Shaoqing, SUN Hongmin, YANG Weimin. Post-synthesis of Ti-MWW Zeolite via Titanium Incorporation in Liquid Acid Solution [J]. Chem. J. Chinese Universities, 2021, 42(9): 2742. |
| [13] | WANG Meiyin, HUANG Daofeng, CHEN Xin, ZHOU Junfu, REN Yuanhang, YE Lin, YUE Bin, HE Heyong. Liquid Phase Assembly of Mesoporous CsxH3-xPW12O40 and Characterization of Their Acidity [J]. Chem. J. Chinese Universities, 2021, 42(9): 2734. |
| [14] | HU Chuanchuan, PANG Jingxiang, HE Chuangchuang, LI Wei, SUN Shutao. Sc(OTf)3 Catalyzed 1,6-Conjugate Allylation of δ-CN p-QMs: Synthesis of Allyl Substituted Diarylacetonitrile Compounds [J]. Chem. J. Chinese Universities, 2021, 42(9): 2805. |
| [15] | MENG Fanwei, GAO Qi, YE Qing, LI Chenxi. Potassium Poisoning Mechanism of Cu-SAPO-18 Catalyst for Selective Catalytic Reduction of NOx by Ammonia [J]. Chem. J. Chinese Universities, 2021, 42(9): 2832. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||