

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (7): 1388.doi: 10.7503/cjcu20140288
• Analytical Chemistry • Previous Articles Next Articles
CAO Tingting, CAO Zhong*( ), HE Jinglin, LIANG Haiqin, XIAO Zhongliang*
), HE Jinglin, LIANG Haiqin, XIAO Zhongliang*
Received:2014-04-02
Online:2014-07-10
Published:2014-06-03
Contact:
CAO Zhong,XIAO Zhongliang
E-mail:zhongcao2004@163.com
Supported by:CLC Number:
TrendMD:
CAO Tingting, CAO Zhong, HE Jinglin, LIANG Haiqin, XIAO Zhongliang. Simple and Rapid Determination of Mercury Ions Based on 1,4-Dithiothreitol Assembled Gold Plate Electrode†[J]. Chem. J. Chinese Universities, 2014, 35(7): 1388.

Fig.2 Design of gold plate electrode(A) and SEM image of its section structure(B) (A) 1. Base plate; 2. gold film; 3. gold-film pin; 4. plastics film; 5. copper wire; 6. soldering tin; 7. silicone sealant.
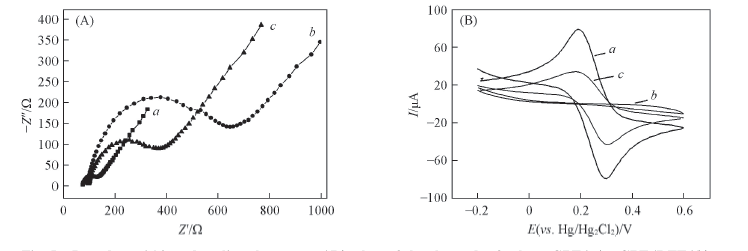
Fig.5 Impedance(A) and cyclic voltammetry(B) plots of the electrodes for bare GPE(a), GPE/DTT(b) and GPE/DTT/Hg2+(c) in the media solution containing 2.0 mmol/L K3Fe(CN)6 and 0.2 mol/L Na2SO4
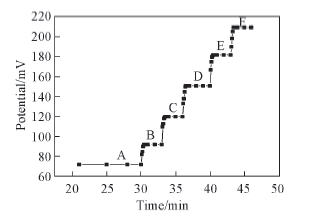
Fig.9 Dynamic response of the electrode to various concentrations of Hg2+ in Tris-HCl buffer(pH=6.0) c(Hg2+)/(mol·L-1): A. 0; B. 1.0×10-8; C. 1.0×10-7; D. 1.0×10-6; E. 1.0×10-5; F. 1.0×10-4.
| No. | Potential/mV | |
|---|---|---|
| 1.0×10-6 mol/L Hg2+ | 1.0×10-4 mol/L Hg2+ | |
| 1 | 131 | 189 |
| 2 | 130 | 188 |
| 3 | 131 | 189 |
| 4 | 131 | 189 |
| 5 | 129 | 188 |
| 6 | 130 | 187 |
| 7 | 130 | 188 |
| 8 | 131 | 188 |
| 9 | 130 | 189 |
| 10 | 131 | 190 |
| AV | 130.4 | 188.5 |
| SD | ±0.70 | ±0.85 |
Table 1 Reproducibility of GPE/DTT electrode
| No. | Potential/mV | |
|---|---|---|
| 1.0×10-6 mol/L Hg2+ | 1.0×10-4 mol/L Hg2+ | |
| 1 | 131 | 189 |
| 2 | 130 | 188 |
| 3 | 131 | 189 |
| 4 | 131 | 189 |
| 5 | 129 | 188 |
| 6 | 130 | 187 |
| 7 | 130 | 188 |
| 8 | 131 | 188 |
| 9 | 130 | 189 |
| 10 | 131 | 190 |
| AV | 130.4 | 188.5 |
| SD | ±0.70 | ±0.85 |
| Interfering ion | Interfering ion | Interfering ion | |||
|---|---|---|---|---|---|
| Na+ | -4.95 | Cu2+ | -2.85 | Sn2+ | -3.25 |
| K+ | -3.20 | Zn2+ | -3.15 | Ni2+ | -3.48 |
| Mg2+ | -4.32 | Pb2+ | -3.65 | N | -4.69 |
| Ba2+ | -4.29 | Ag+ | -3.83 | I | -4.32 |
| Ca2+ | -3.58 | Al3+ | -3.63 | Br | -3.47 |
| Fe2+ | -3.15 | Fe3+ | -2.85 | Cl | -3.06 |
Table 2 Selectivity coefficient of GPE/DTT electrode to Hg2+
| Interfering ion | Interfering ion | Interfering ion | |||
|---|---|---|---|---|---|
| Na+ | -4.95 | Cu2+ | -2.85 | Sn2+ | -3.25 |
| K+ | -3.20 | Zn2+ | -3.15 | Ni2+ | -3.48 |
| Mg2+ | -4.32 | Pb2+ | -3.65 | N | -4.69 |
| Ba2+ | -4.29 | Ag+ | -3.83 | I | -4.32 |
| Ca2+ | -3.58 | Al3+ | -3.63 | Br | -3.47 |
| Fe2+ | -3.15 | Fe3+ | -2.85 | Cl | -3.06 |
| Sample | Concentration added/(μmol·L-1) | Spectrophotometric method with dithizone | GPE/DTT electrode | Recovery(%) | ||
|---|---|---|---|---|---|---|
| Mean/(μmol·L-1) | SD | Mean/(μmol·L-1) | SD | |||
| A | 0.400 | 0.402 | ±0.016 | 0.407 | ±0.015 | 101.75 |
| B | 0.500 | 0.510 | ±0.013 | 0.491 | ±0.019 | 98.20 |
| C | 0.600 | 0.594 | ±0.014 | 0.604 | ±0.014 | 100.67 |
| D | 0.800 | 0.794 | ±0.013 | 0.797 | ±0.022 | 99.63 |
| E | 1.200 | 1.210 | ±0.015 | 1.206 | ±0.014 | 100.50 |
| F | 1.800 | 1.798 | ±0.011 | 1.796 | ±0.016 | 99.89 |
Table 3 Recovery of the proposed GPE/DTT electrode for the determination of Hg2+ in real water samples compared with spectrophotometric method with dithizone
| Sample | Concentration added/(μmol·L-1) | Spectrophotometric method with dithizone | GPE/DTT electrode | Recovery(%) | ||
|---|---|---|---|---|---|---|
| Mean/(μmol·L-1) | SD | Mean/(μmol·L-1) | SD | |||
| A | 0.400 | 0.402 | ±0.016 | 0.407 | ±0.015 | 101.75 |
| B | 0.500 | 0.510 | ±0.013 | 0.491 | ±0.019 | 98.20 |
| C | 0.600 | 0.594 | ±0.014 | 0.604 | ±0.014 | 100.67 |
| D | 0.800 | 0.794 | ±0.013 | 0.797 | ±0.022 | 99.63 |
| E | 1.200 | 1.210 | ±0.015 | 1.206 | ±0.014 | 100.50 |
| F | 1.800 | 1.798 | ±0.011 | 1.796 | ±0.016 | 99.89 |
| [1] | Kobal A. B., Horvat M., Prezelj. M., Briski A. S., Krsnik M., Dizdarevic T., Mazej D., Falnoga I., Stibilj V., Arneric N., Kobal D., Osredkar J., J. Trace Elem. Med. Biol., 2004, 17, 261—274 |
| [2] | Hoyle I., Handy R. D., Aquatic Toxicol., 2005,72, 147—159 |
| [3] | Percy A. J., Korbas M., George G. N., Gailer J., J. Chromatogr. A, 2007,1156, 331—339 |
| [4] | Bi N., Chen Y. H., Qi H. B., Zheng X., Chen Y., Liao X., Zhang H. Q., Tian Y., Sens. Actuators B, 2012,166167, 766—771 |
| [5] | Xu J. Y., Wang T., Chen D. J., Ye L. H., Ni S. J., Chin. J. Anal. Chem, 2012,40(1), 169—172 |
| (徐进勇, 王彤, 陈杜军, 叶隆慧, 倪师军. 分析化学, 2012, 40(1), 169—172) | |
| [6] | Fong B. M. W. F., Siu T. S., Lee J. S. K., Tam S., J. Anal. Toxicol., 2007,31, 281—287 |
| [7] | Li Y. F, Chen C. Y, Li B., Sun J., Wang J. X., Gao Y.X., ZhaoY. L., Chai Z. F., J. Anal. Atomic Spectrom., 2006,21(1), 94—96 |
| [8] | Yang Q. J., Tan Q., Zhou K. Z., Xu K. L., Hou X. D., J. Anal. Atomic Spectrom., 2005,20, 760—762 |
| [9] | Hsu I. H., Hsu T. C., Sun Y. C., Biosens. Bioelectron., 2011,26, 4605—4609 |
| [10] | Bansal N., Vaughan J., Boullemant A., Leong T., Microchem. J., 2014,113, 36—41 |
| [11] | Sánchez-Rodas D., Corns W. T., Chen B., Stockwell P. B., J. Anal. Atomic Spectrom., 2010,25(7), 933—946 |
| [12] | Huang K., Xu K. L., Hou X. D., Jia Y., Zheng C. B., Yang L., J. Anal. Atomic Spectrom., 2013,28(4), 510—515 |
| [13] | Ichinoki S., Kitahata N., Fujii Y., J. Liquid Chromatogr. Relat. Technol., 2004,27, 1785—1798 |
| [14] | Hellings J., Adeloju S. B., Verheyen T. V., Microchem. J., 2013,111, 62—66 |
| [15] | Wu X. L., Lai J. P., Zhao Y. B., Chem. J. Chinese Universities, 2012,33(4), 695—699 |
| (吴小丽, 赖金平, 赵一兵. 高等学校化学学报, 2012, 33(4), 695—699 | |
| [16] | Wu Z. H., Lin J. H., Tseng W. L., Biosens. Bioelectron., 2012,34, 185—190 |
| [17] | Lei Z. J., Zhang C. Z., Hu Q. H., Liu Y., Zhang Q., Liu X. J., Chin. J. Anal. Chem., 2012,40(12), 1827—1831 |
| (雷兆静, 张存政, 胡秋辉, 刘媛, 张强, 刘贤金.分析化学, 2012, 40(12), 1827—1831) | |
| [18] | Bühlmann P., Pretsch E., Bakker E., Chem. Rev., 1998,98, 1593—1687 |
| [19] | Dalkıran B., Özel A. D., Parlayan S., Canel E., Ocak Ü., Kılıç E., Monatshefte für Chemie—Chemical Monthly, 2010,141(8), 829—839 |
| [20] | Song I., Hahm S., Jin S., Shin J. H., Nam H., Kim B. H., Sensor Lett., 2011,9(1), 179—183 |
| [21] | Lindner E., Tóth K., Pungor E., Anal. Chem., 1984,56, 1127—1131 |
| [22] | Badr I. H. A., Meyerhoff M. E., Hassan S. S. M., Anal. Chim. Acta, 1996,321, 11—19 |
| [23] | Jalali-Heravi. M, Khandar A. A., Sheikshoaie I., Spectrochim. Acta A, 1999,55, 2537—2544 |
| [24] | Mashhadizadeh M. H., Sheikhahoaie I., Talanta, 2003,60, 73—80 |
| [25] | Tyagi S., Agarwal H., Ikram S., Water Sci. Technol., 2010,61(3), 693—704 |
| [26] | Lü J. Q., Zeng X. S., Ai J., Pang D. W., Chem. J. Chinese Universities, 2005,26(2), 238—240 |
| (吕鉴泉, 曾宪顺, 艾娟, 庞代文. 高等学校化学学报, 2005, 26(2), 238—240) | |
| [27] | Gupta V. K., Chandra S., Lang H., Talanta, 2005,66, 575—580 |
| [28] | Bagheri M., Mashhadizadeh M. H., Razee S., Momeni A., Electroanalysis, 2003,15, 1824—1829 |
| [29] | Ibrahim I., Cemal Y., Humeyra B., Analyst, 1996,121, 1873—1876 |
| [30] | Mahajan R. K., Kaur I., Lobana T. S., Talanta ,2003,59, 101—105 |
| [31] | Gupta K. C., D’Arc M. J., Talanta, 2000,52, 1087—1103 |
| [32] | Akhond M., Najafi M. B., Tashkhourian J., Sens. Actuators B, 2004,99, 410—415 |
| [33] | Mazloum M., Amini M. K., Mohammadpoor-Baltork I., Sens. Actuators B, 2000,63, 80—85 |
| [34] | Mashhadizadeh M. H., Shoaei I. S., Monadi N., Talanta, 2004,64, 1048—1052 |
| [35] | Castro M., Cruz J., Otazo-Sánchez E., Perez-Marın L., J. Phys. Chem. A, 2003, 107, 9000—9007 |
| [36] | Perez-Marín L., Castro M., Otazo-Sánchez E., Cisneros G. A., Int. J. Quantum Chem., 2000,80, 609—622 |
| [37] | Gupta V. K., Singh A. K., Al Khayat M., Gupta B., Anal. Chim. Acta, 2007,590, 81—90 |
| [38] | Yu X., Zhou Z. D., Wang Y., Liu Y., Xie Q., Xiao D., Sens. Actuators B, 2007,123, 352—358 |
| [39] | Chen L., Fu X. L., Lu W. H., Chen L. X., ACS Appl. Mater. Interfaces, 2013,5, 284—290 |
| [40] | Fleissner G., Kozlowski P. M., Vargek M., Bryson J. W., O’Halloran T. V., Spiro T. G., Inorg. Chem., 1999,38(15), 3523—352 |
| [41] | Yan Z. N., Wang S. Q., Wang H. X., Wu S. Y., Mater. Sci. Eng. C, 2013,33, 2562—2568 |
| [42] | Wuhan University, Analytical Chemistry, Higher Education Press, Beijing, 2006, 62—66 |
| (武汉大学. 分析化学, 北京:高等教育出版社, 2006, 62—66) | |
| (Ed.: N, K) |
| [1] | GE Yicong, NIE Wanli, SUN Guofeng, CHEN Jiaxuan, TIAN Chong. Silver-catalyzed [5+1] Cyclization of 2-Vinylanilines with Benzisoxazoles [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220142. |
| [2] | WANG Mingzhi, ZHENG Yanping, WENG Weizheng. Catalytic Methane Combustion over CeO2 Supported PdO and Ce1‒x Pd x O2‒δ Species [J]. Chem. J. Chinese Universities, 2022, 43(4): 20210816. |
| [3] | GAO Jing, HE Wentao, WANG Xinxin, XIANG Yushu, LONG Lijuan, QIN Shuhao. Preparation of DOPO Derivative Modified Carbon Nanotubes and Their Effect on Flame Retardancy of Polylactic Acid [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210670. |
| [4] | LI Xiaohui, WEI Aijia, MU Jinping, HE Rui, ZHANG Lihui, WANG Jun, LIU Zhenfa. Effects of SmPO4 Coatingon Electrochemical Performance of High-voltage LiNi0.5Mn1.5O4 Cathode Materials [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210546. |
| [5] | ZHANG Liling, LIU Liu, ZHENG Mingqiu, FANG Wenkai, LIU Da, TANG Hongwu. Dual Signal Detection of HPV16 DNA by CRISPR/Cas12a Biosensing System Based on Upconversion Luminescent Resonance Energy Transfer [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220412. |
| [6] | LI Dan, XIAO Liping, FAN Jie. Inorganic-based Surface Materials with Anti-SARS-CoV-2 Properties and Their Mechanisms of Action [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220301. |
| [7] | WANG Yuanyue, AN Suosuo, ZHENG Xuming, ZHAO Yanying. Spectroscopic and Theoretical Studies on 5-Mercapto-1,3,4-thiadiazole-2-thione Microsolvation Clusters [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220354. |
| [8] | LIANG Yu, LIU Huan, GONG Lige, WANG Chunxiao, WANG Chunmei, YU Kai, ZHOU Baibin. Synthesis and Supercapacitor Properties of Biimidazole-modified {SiW12O40} Hybrid [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210556. |
| [9] | ZHOU Yonghui, HUANG Rujun, YAN Jianyang, LI Yajun, QIU Huanhuan, YANG Jinxuan, ZHENG Youxuan. Synthesis and Electroluminescence Properties of Two Iridium(Ⅲ) Complexes with Nitrogen Heterocycle Structures [J]. Chem. J. Chinese Universities, 2022, 43(1): 20210415. |
| [10] | LUO Qiangqiang, JIN Shaoqing, SUN Hongmin, YANG Weimin. Post-synthesis of Ti-MWW Zeolite via Titanium Incorporation in Liquid Acid Solution [J]. Chem. J. Chinese Universities, 2021, 42(9): 2742. |
| [11] | WANG Meiyin, HUANG Daofeng, CHEN Xin, ZHOU Junfu, REN Yuanhang, YE Lin, YUE Bin, HE Heyong. Liquid Phase Assembly of Mesoporous CsxH3-xPW12O40 and Characterization of Their Acidity [J]. Chem. J. Chinese Universities, 2021, 42(9): 2734. |
| [12] | LI Haibo, XIAO Changfa, JIANG Long, HUANG Yun, DAN Yi. Copolymerization of Methyl Acrylate and 1-Octene Catalyzed by the Loaded Aluminum Chloride on MCM-41 Molecular Sieve [J]. Chem. J. Chinese Universities, 2021, 42(9): 2974. |
| [13] | LIU Huazheng, PAN Xiaoguang, LI Hua, WAN Renzhong, LIU Xigong. Na2CO3-catalyzed 1,6-Conjugate Addition of Trimethylsilyl Azide to δ-CF3-δ-Aryl-disubstituted Para-Quinone Methides: Efficient Construction of Diarylmethanes Bearing CF3- and N3-Substituted Quaternary Stereocenters [J]. Chem. J. Chinese Universities, 2021, 42(9): 2772. |
| [14] | MENG Fanwei, GAO Qi, YE Qing, LI Chenxi. Potassium Poisoning Mechanism of Cu-SAPO-18 Catalyst for Selective Catalytic Reduction of NOx by Ammonia [J]. Chem. J. Chinese Universities, 2021, 42(9): 2832. |
| [15] | HU Chuanchuan, PANG Jingxiang, HE Chuangchuang, LI Wei, SUN Shutao. Sc(OTf)3 Catalyzed 1,6-Conjugate Allylation of δ-CN p-QMs: Synthesis of Allyl Substituted Diarylacetonitrile Compounds [J]. Chem. J. Chinese Universities, 2021, 42(9): 2805. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||