

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (7): 1396.doi: 10.7503/cjcu20140168
• Analytical Chemistry • Previous Articles Next Articles
DAI Yulin1, YU Shanshan1, ZHANG Ying1, HAO Ying1, ZHONG Wei1, YUE Hao1, LIU Shuying1,2,*( )
)
Received:2014-03-04
Online:2014-07-10
Published:2014-05-26
Contact:
LIU Shuying
E-mail:syliu@ciac.jl.cn
Supported by:CLC Number:
TrendMD:
DAI Yulin, YU Shanshan, ZHANG Ying, HAO Ying, ZHONG Wei, YUE Hao, LIU Shuying. Studies on the Isoflavone in Extract of the Flower of Pueraria Lobata by RRLC-Q-TOF MS/MS†[J]. Chem. J. Chinese Universities, 2014, 35(7): 1396.
| Compd. | Structure | R1 | R2 | R3 | R4 | MF | Mw |
|---|---|---|---|---|---|---|---|
| Irisolidone | OH | OMe | OH | OMe | C17H14O6 | 314.0790 | |
| Genistin | Oglc | H | OH | OH | C21H20O10 | 432.1057 | |
| Tectoridin | Oglc | OMe | OH | OH | C22H22O11 | 462.1162 | |
| Daidzein | OH | H | H | OH | C15H10O4 | 254.0579 | |
| Tectorigenin | OH | OMe | OH | OH | C16H12O6 | 300.0634 | |
| Genistein | OH | H | OH | OH | C15H10O5 | 270.0528 | |
| Biocanin A | OH | H | OH | OMe | C16H12O5 | 284.0685 | |
| Kakkatin | OMe | OH | H | OH | C16H12O5 | 284.0685 | |
| Glycitein | OH | OMe | H | OH | C16H12O5 | 284.0685 | |
| Daidzin | Oglc | H | H | OH | C21H20O9 | 416.1107 | |
| Kakkalide | Oglc-6xyl | OMe | OH | OMe | C28H32O15 | 608.1741 | |
| 6-O-Xylosyl-tectoridin | Oglc-6xyl | OMe | H | OH | C27H30O15 | 594.1585 |
Table 1 Possible isoflavone compounds in Pueraria lobata(Willd.) Ohwi. flower
| Compd. | Structure | R1 | R2 | R3 | R4 | MF | Mw |
|---|---|---|---|---|---|---|---|
| Irisolidone | OH | OMe | OH | OMe | C17H14O6 | 314.0790 | |
| Genistin | Oglc | H | OH | OH | C21H20O10 | 432.1057 | |
| Tectoridin | Oglc | OMe | OH | OH | C22H22O11 | 462.1162 | |
| Daidzein | OH | H | H | OH | C15H10O4 | 254.0579 | |
| Tectorigenin | OH | OMe | OH | OH | C16H12O6 | 300.0634 | |
| Genistein | OH | H | OH | OH | C15H10O5 | 270.0528 | |
| Biocanin A | OH | H | OH | OMe | C16H12O5 | 284.0685 | |
| Kakkatin | OMe | OH | H | OH | C16H12O5 | 284.0685 | |
| Glycitein | OH | OMe | H | OH | C16H12O5 | 284.0685 | |
| Daidzin | Oglc | H | H | OH | C21H20O9 | 416.1107 | |
| Kakkalide | Oglc-6xyl | OMe | OH | OMe | C28H32O15 | 608.1741 | |
| 6-O-Xylosyl-tectoridin | Oglc-6xyl | OMe | H | OH | C27H30O15 | 594.1585 |
| Compd. | Structure | R1 | R2 | MF | Mw |
|---|---|---|---|---|---|
| Quercetin | OH | OH | C15H10O7 | 302.0427 | |
| Rutin | Oglc-6glc | OH | C27H30O16 | 610.1534 | |
| Nicotiflorin | Oglc-6glc | H | C27H30O15 | 594.1580 |
Table 2 Possible flavonoid compounds in Pueraria lobata(Willd.) Ohwi. flower
| Compd. | Structure | R1 | R2 | MF | Mw |
|---|---|---|---|---|---|
| Quercetin | OH | OH | C15H10O7 | 302.0427 | |
| Rutin | Oglc-6glc | OH | C27H30O16 | 610.1534 | |
| Nicotiflorin | Oglc-6glc | H | C27H30O15 | 594.1580 |
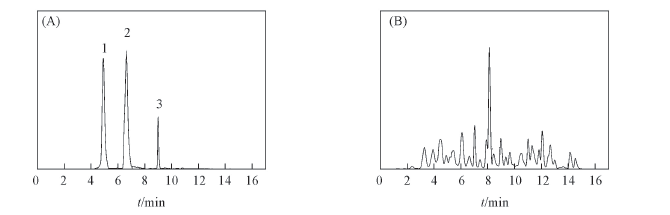
Fig.1 RRLC profiles of the three reference compounds(A) and the water extracts of Pueraria lobata(Willd.) Ohwi. flower(B) on the molecular ions in negative ion mode (A) 1. Genistin; 2. tectoridin; 3. kakkalide.
| tR/min | [M-H]-, m/z | MS/MS, m/z | Compd. | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean measured mass | Theoretical exact mass | 106 Mass accuracy | |||||||||
| 2.245 | 417.1770 | 417.1707 | 15.1 | 152[M-H-C11H20]-, 108[M-H-C13H25]- | Puerarol | ||||||
| 3.373 | 461.1240 | 461.1242 | 0.4 | 301[M-H-C10H8O2]- | Alopecuroides A | ||||||
| 3.919 | 623.1607 | 623.1618 | -1.7 | 283[M-H-C12H20O11]- , | 6″-O-Xylosyl-glycitin | ||||||
| [M+HCOOH-H]- | [M+HCOOH-H]- | 268[M-H-C12H20O11-CH3]- | |||||||||
| 4.598 | 491.1180 | 491.1195 | -3.0 | 445[M-H-HCOOH]-, | Sissotrin, glycitin, | ||||||
| [M+HCOOH-H]- | [M+HCOOH-H]- | 283[M-H-HCOOH-C6H10O5]- | 3'-oxymethy-puerarin | ||||||||
| 4.911 | 431.0953 | 431.0984 | 7.1 | 311[M-H-C4H804]- , | Genistin | ||||||
| 283[M-H-C4H804-CO]- | |||||||||||
| 5.224 | 593.1508 | 593.1512 | -0.6 | 299[M-H-C17H18O9]- , 284[M-H-C17H18O9-CH3]- | 6″-O-Xylosyl-tectoridin | ||||||
| 5.658 | 609.1353 | 609.1461 | 17.7 | 285[M-H-C12H20010]-, | Rutin | ||||||
| 269[M-H-C12H20010-H2O]- | |||||||||||
| 6.014 | 283.0610 | 283.0612 | -0.7 | 268[M-H-CH3]-, 240[M-H-CH3-CO]-, | BiocaninA, | ||||||
| 196[M-H-CH3-CO-OH-COH]- | Kakkatin, Glycitein | ||||||||||
| 6.603 | 461.1106 | 461.1089 | -3.6 | 299[M-H-C6H10O5]-, | Tectoridin | ||||||
| 284[M-H-C6H10O5-CH3]-, 255[M-H- C6H10O5 -CH3-CO]- | |||||||||||
| 7.025 | 269.0466 | 269.0455 | -4 | 240[M-H-CO2H]- | Genistein | ||||||
| 7.461 | 491.1180 [M+HCOOH-H]- | 491.1195 [M+HCOOH-H]- | -3 | 283[M-H-C6H10O5]- | Sissotrin, glycitin, 3'-oxymethy-puerarin | ||||||
| 8.425 | 298.0844 | 298.0841 | 1.0 | 282[M-H-CH3]- , 267[M-H-CH3-CH3]-, | 3'-Oxymethy-daidzein-7-O-methylether | ||||||
| 254[M-H-CH3-CH3-H2O]-, | |||||||||||
| 239[M-H-CH3-CH3-CO]-, | |||||||||||
| 211[M-H-CH3-CH3-CO-H2O]-, 195[M-H-CH3-CH3-CO-H2O-OH]- | |||||||||||
| 8.937 | 607.1639 | 607.1668 | -4.7 | 313[M-H-C11H18O9]-, 298[M-H-C11H18O9-CH3] - | Kakkalide | ||||||
| 9.336 | 253.0522 | 253.0506 | -6.3 | 224[M-H-COH]-, 209[M-H-CO2]-, 135[M-H-C7H2O2]- | Daidzein | ||||||
| 9.783 | 313.0725 | 313.0718 | -2.2 | 298[M-H-CH3]- , 283[M-H-C2H6]-, | Irisolidone | ||||||
| 270[M-H-CH3-CO]-, | |||||||||||
| 255[M-H-CH3-CO-H2O]-, | |||||||||||
| 239[M-H-CH3-CO2-H2O]-, | |||||||||||
| 211[M-H-CH3-CO2-H2O-CH3O]- | |||||||||||
| 10.598 | 475.1246 | 475.1246 | 0 | 313[M-H-C6H10O5]-, | 7-O-Glc-Irisolidone | ||||||
| 298[M-H-C6H10O5-CH3]- | |||||||||||
| 11.072 | 299.0557 | 299.0561 | 1.3 | 284[M-H-CH3]-, 255[M-H-CH3-CO]- | Tectorigenin | ||||||
| 11.354 | 329.2734 | 329.2697 | -11.2 | 211[M-H-C4H6O4]- | 2,3-Dihydroxypropyl- palmitate | ||||||
| 12.004 | 301.0350 | 301.0354 | 1.3 | 283[M-H-H2O]-, 257[M-H-C2O]- | Quercetin | ||||||
Table 3 RRLC-ESI-MS/MS data of the water extract of Pueraria lobata(Willd.) Ohwi. flower in negative ion mode
| tR/min | [M-H]-, m/z | MS/MS, m/z | Compd. | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean measured mass | Theoretical exact mass | 106 Mass accuracy | |||||||||
| 2.245 | 417.1770 | 417.1707 | 15.1 | 152[M-H-C11H20]-, 108[M-H-C13H25]- | Puerarol | ||||||
| 3.373 | 461.1240 | 461.1242 | 0.4 | 301[M-H-C10H8O2]- | Alopecuroides A | ||||||
| 3.919 | 623.1607 | 623.1618 | -1.7 | 283[M-H-C12H20O11]- , | 6″-O-Xylosyl-glycitin | ||||||
| [M+HCOOH-H]- | [M+HCOOH-H]- | 268[M-H-C12H20O11-CH3]- | |||||||||
| 4.598 | 491.1180 | 491.1195 | -3.0 | 445[M-H-HCOOH]-, | Sissotrin, glycitin, | ||||||
| [M+HCOOH-H]- | [M+HCOOH-H]- | 283[M-H-HCOOH-C6H10O5]- | 3'-oxymethy-puerarin | ||||||||
| 4.911 | 431.0953 | 431.0984 | 7.1 | 311[M-H-C4H804]- , | Genistin | ||||||
| 283[M-H-C4H804-CO]- | |||||||||||
| 5.224 | 593.1508 | 593.1512 | -0.6 | 299[M-H-C17H18O9]- , 284[M-H-C17H18O9-CH3]- | 6″-O-Xylosyl-tectoridin | ||||||
| 5.658 | 609.1353 | 609.1461 | 17.7 | 285[M-H-C12H20010]-, | Rutin | ||||||
| 269[M-H-C12H20010-H2O]- | |||||||||||
| 6.014 | 283.0610 | 283.0612 | -0.7 | 268[M-H-CH3]-, 240[M-H-CH3-CO]-, | BiocaninA, | ||||||
| 196[M-H-CH3-CO-OH-COH]- | Kakkatin, Glycitein | ||||||||||
| 6.603 | 461.1106 | 461.1089 | -3.6 | 299[M-H-C6H10O5]-, | Tectoridin | ||||||
| 284[M-H-C6H10O5-CH3]-, 255[M-H- C6H10O5 -CH3-CO]- | |||||||||||
| 7.025 | 269.0466 | 269.0455 | -4 | 240[M-H-CO2H]- | Genistein | ||||||
| 7.461 | 491.1180 [M+HCOOH-H]- | 491.1195 [M+HCOOH-H]- | -3 | 283[M-H-C6H10O5]- | Sissotrin, glycitin, 3'-oxymethy-puerarin | ||||||
| 8.425 | 298.0844 | 298.0841 | 1.0 | 282[M-H-CH3]- , 267[M-H-CH3-CH3]-, | 3'-Oxymethy-daidzein-7-O-methylether | ||||||
| 254[M-H-CH3-CH3-H2O]-, | |||||||||||
| 239[M-H-CH3-CH3-CO]-, | |||||||||||
| 211[M-H-CH3-CH3-CO-H2O]-, 195[M-H-CH3-CH3-CO-H2O-OH]- | |||||||||||
| 8.937 | 607.1639 | 607.1668 | -4.7 | 313[M-H-C11H18O9]-, 298[M-H-C11H18O9-CH3] - | Kakkalide | ||||||
| 9.336 | 253.0522 | 253.0506 | -6.3 | 224[M-H-COH]-, 209[M-H-CO2]-, 135[M-H-C7H2O2]- | Daidzein | ||||||
| 9.783 | 313.0725 | 313.0718 | -2.2 | 298[M-H-CH3]- , 283[M-H-C2H6]-, | Irisolidone | ||||||
| 270[M-H-CH3-CO]-, | |||||||||||
| 255[M-H-CH3-CO-H2O]-, | |||||||||||
| 239[M-H-CH3-CO2-H2O]-, | |||||||||||
| 211[M-H-CH3-CO2-H2O-CH3O]- | |||||||||||
| 10.598 | 475.1246 | 475.1246 | 0 | 313[M-H-C6H10O5]-, | 7-O-Glc-Irisolidone | ||||||
| 298[M-H-C6H10O5-CH3]- | |||||||||||
| 11.072 | 299.0557 | 299.0561 | 1.3 | 284[M-H-CH3]-, 255[M-H-CH3-CO]- | Tectorigenin | ||||||
| 11.354 | 329.2734 | 329.2697 | -11.2 | 211[M-H-C4H6O4]- | 2,3-Dihydroxypropyl- palmitate | ||||||
| 12.004 | 301.0350 | 301.0354 | 1.3 | 283[M-H-H2O]-, 257[M-H-C2O]- | Quercetin | ||||||
| [1] | Editorial Committee of State Administration of Traditional Chinese Medicine of People’s Republic of China, The Chinese Materia Medica, Shanghai Science and Technology Press, Shanghai, 1999, 4, 619 |
| (国家中医药管理局《中华本草》编委会. 中华本草, 上海: 上海科技出版社, 1999, 4, 619) | |
| [2] | Han Y. O., Han M. J., Park S. H., Pharmacol. Sci., 2003, 93(3), 331—336 |
| [3] | Lee H. W., Choo M. K., Bea E. A., Liver Int., 2003, 23(4),221—226 |
| [4] | Choi J., Shin M. H., Park K. Y., Med. Food,2004, 7(1),31—37 |
| [5] | Wang S. P., Chen M. W., Wang Y. T., Pharmacology and Clinics of Chinese Materia Medica,2012, 28(2), 193—195 |
| (王胜鹏, 陈美婉, 王一涛. 中药药理与临床,2012, 28(2), 193—195) | |
| [6] | Han Y. O., Han M. J., Park S. H., J. Pharmacol. Sci., 2003, 93(3), 331—336 |
| [7] | Li L., Liu C. M., Wu W., Yue H., Liu Z. Q., Liu S. Y., Tian C., Chin. J. Anal. Chem,2005, 33, 1087—1090 |
| (李丽, 刘春明, 吴巍, 越皓, 刘志强, 刘淑莹, 田成. 分析化学, 2005, 33, 1087—1090) | |
| [8] | Cui M., Liu Z. Q., Song F. R., Liu S. Y., Chem. J. Chinese Universities,2001, 22(8), 1323—1325 |
| (崔勐, 刘志强, 宋凤瑞, 刘淑莹. 高等学校化学学报,2001, 22(8), 1323—1325) | |
| [9] | Chen M. L., Song F. R., Guo M. Q., Liu Z. Q., Liu S. Y., Chem. J. Chinese Universities,2002, 23(5), 805—808 |
| (陈貌连, 宋凤瑞, 郭明全, 刘志强, 刘淑莹. 高等学校化学学报,2002, 23(5), 805—808) | |
| [10] | Wu Q. L., Yang Y. H., Simon J., Am. J. Anal. Chem., 2011, 2, 665—674 |
| [11] | Tian L., Gao X. L., Chen X. Y., Zhong D. F., Zhang Y. F., Dai X. J., Chem. Res. Chinese Universities,2011, 27(2), 185—189 |
| [12] | Bai J., Shi X. W., Du Y. F., Xiang B., Wang S., Cao D. Y., Chem. Res. Chinese Universities,2012, 28(3), 399—405 |
| [13] | Zhang Y. C., Pi Z. F., Liu C. M., Song F. R., Liu Z. Q., Liu S. Y., Chem. Res. Chinese Universities,2012, 28(1), 31—36 |
| [14] | Huang X., Song F. R., Liu Z. Q., Liu S. Y., Acta Chim. Sinica,2008, 66(9), 1059—1066 |
| (黄鑫, 宋凤瑞, 刘志强, 刘淑莹. 化学学报,2008, 66(9), 1059—1066) | |
| [15] | Fabre N., Rustan I., de Hoffmann. E., Quetin L. J., Am. Soc. Mass Spectr., 2001, 12, 707—715 |
| [16] | Klejdus B., Vacek J., Benešová L., Kopecky J., Lapcík O., Kubáň V., Anal. Bioanal. Chem., 2007, 389(7), 2277—2285 |
| [17] | Liu S.Y., Song F. R., Liu Z. Q., Chinese Herb Mass Spectrometry, Science Press, Beijing, 2012, 103—113 |
| (刘淑莹, 宋凤瑞, 刘志强. 中药质谱分析, 北京: 科学出版社, 2012, 103—113) | |
| [18] | Justesena U., Arrigonia E., Larsenb B. R., Amadoa R., Food Sci. Technol., 2000, 33(6), 424—430 |
| [19] | Justesen U., J. Mass Spectrom, 2001, 36(2), 169—178 |
| [20] | Li Y. H., Dai H. X., Wang M. M., Li X. R., Wang L. J., Xue M., Journal of Chinese Mass Spectrometry Society,2009, 30(6), 374—375 |
| (李宇航, 戴海学, 汪明明, 李晓蓉, 王丽娟, 薛明. 质谱学报,2009, 30(6), 374—375) | |
| [21] | Yu Y. L., Liao Y. T., Li X., Ye Y., Ke C. Q., Li X. Q., Yang X. Z., Yao M. C., Journal of Asian Natural Products Research,2011, 13(4), 284—289 |
| [22] | Yang H., Isolation and Antioxidantion of Isoflavonoids in Pueraria Lobata and Effect of Light Quality on Isoflavone Biosynthesis, Anhui Agricultural University, Hefei, 2009 |
| (杨华. 野葛异黄酮的分离纯化抗氧化性及光质对异黄酮生物合成的影响, 合肥: 安徽农业大学, 2009) | |
| [23] | Zhen Z., Liu G. M., Wang Y. H., Li C. S., Ren Y. L., Xiao H., Hao X. J., He H. P., Chinese Chemical Letters,2007, 18, 297—299 |
| (Ed.: I, K) |
| [1] | LI Wenhong,WANG Danyang,CAO Jinjin,WEI Yongju. Comparative Study of Absorption and Fluorescence Spectra of Glycitein and Glycitin† [J]. Chem. J. Chinese Universities, 2019, 40(1): 47. |
| [2] | CHEN Zhan-Guo*, ZHAO Hai-Xia, WEI Jun-Fa, LIU Bo. Total Synthesis of 2-Methyl-7-[ω-(1H-imidazol-1-yl)ethoxy]-Isoflavone Derivatives and Their Antioxidative Activity [J]. Chem. J. Chinese Universities, 2009, 30(1): 82. |
| [3] | LI Hui1, WAN Le-Ren2, WANG Hong1, Hashi Yuki2*, CHEN Shi-Zhong1*. Identification and Mass Spectrometric Characterization of Isomeric Isoflavone Aglycones by ESI-IT-TOF Mass Spectrometry [J]. Chem. J. Chinese Universities, 2007, 28(12): 2284. |
| [4] | ZHANG Zun-Ting, WANG Qiu-Ya, HE Yun, WANG Xiao-Bing, XUE Dong, ZHENG Jian-Bin. Syntheses,Crystal Structures and Biological Activity of Bimethylation Daidzein Sulfonates [J]. Chem. J. Chinese Universities, 2005, 26(12): 2247. |
| [5] | ZHANG Zun-Ting, WANG Qiu-Ya, HE Yun, WANG Xiao-Bing, XUE Dong, ZHENG Jian-Bin. Syntheses,Crystal Structures and Biological Activity of Bimethylation Daidzein Sulfonates [J]. Chem. J. Chinese Universities, 2005, 26(12): 2247. |
| [6] | LIU Qian-Guang, ZHANG Zun-Ting, XUE Dong. Synthesis, Crystal Structure and Activity of Sul fated Daidzein [J]. Chem. J. Chinese Universities, 2003, 24(5): 820. |
| [7] | LIU Peng, CHEN Rong-Feng, CHANG Jun-Biao, XIE Jing-Xi . 1H NMR Studies on Synthetic Isoflavones with p-Substituents on B Ring [J]. Chem. J. Chinese Universities, 2000, 21(11): 1671. |
| [8] | Li Liang-zhu, Rui Yuan-jin . Oxido-Rearrangement of Silyl Enol Ethers of Flavanones to Isoflavones [J]. Chem. J. Chinese Universities, 1991, 12(6): 777. |
| [9] | Yue Baozhen, Zhou Zewei, Cai Mengsben . Studies on Flavonoids(XIV)— Debromination Reaction in the Synthesis of Bromoisoflavones [J]. Chem. J. Chinese Universities, 1990, 11(1): 99. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||