

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (2): 366.doi: 10.7503/cjcu20200598
收稿日期:2020-08-24
出版日期:2021-02-10
发布日期:2020-12-25
通讯作者:
黄长水
E-mail:huangcs@qibebt.ac.cn
基金资助:
SUN Quanhu1,2, LU Tiantian1, HE Jianjiang1, HUANG Changshui1,2( )
)
Received:2020-08-24
Online:2021-02-10
Published:2020-12-25
Contact:
HUANG Changshui
E-mail:huangcs@qibebt.ac.cn
摘要:
碳材料具有价格低廉、 易制备、 环境友好、 导电性高、 比表面积大以及适合离子存储和迁移等优点, 已成为目前应用于电化学储能器件电极的重要材料之一. 石墨炔(GDY)是一种新型的二维碳同素异形体, 由sp2碳杂化形式的苯环和sp碳杂化形式的炔键构成. 这种独特的化学结构一方面保持了碳材料良好的导电特性, 另一方面形成了新颖的离子传输通道, 为碳材料带来了不同的离子传输和存储特性. 与此同时, 由于石墨炔的空间结构可调性, 可以通过引入异原子微调石墨炔电子结构, 拓展石墨炔在电极材料领域的应用. 本文重点对近几年异原子杂化石墨炔基电极材料在锂离子电池、 钠离子电池、 金属硫电池、 电容器、 金属空气电池和电极保护等储能领域的研究工作进行总结, 并对未来石墨炔类材料在储能领域的发展进行了展望.
中图分类号:
TrendMD:
孙全虎, 卢天天, 何建江, 黄长水. 含异原子石墨炔基电极材料的研究进展. 高等学校化学学报, 2021, 42(2): 366.
SUN Quanhu, LU Tiantian, HE Jianjiang, HUANG Changshui. Advances in the Study of Heteratomic Graphdiyne Electrode Materials. Chem. J. Chinese Universities, 2021, 42(2): 366.
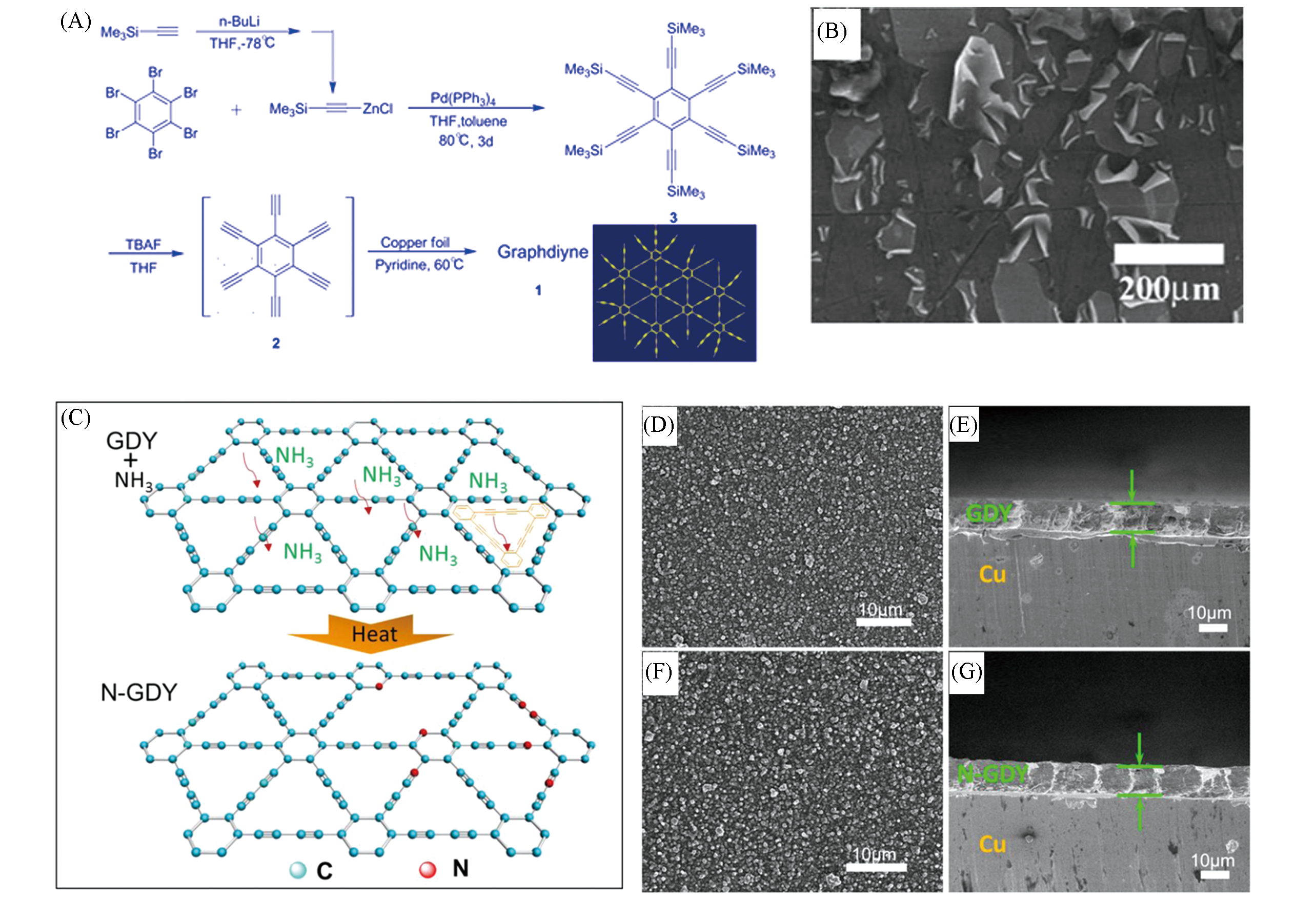
Fig.1 Synthetic route and SEM images of graphdiyne films(A) Synthetic route of graphdiyne films; (B) SEM image of cracked film on the brim of copper foil[4]; schematic representation N doping process of GDY(the unit structure of GDY molecule is shown as the inset)(C); (D—G) surface-(D, F) and cross-sectional(E, G) SEM images of GDY(D, E) and N-GDY(F, G)[13].(A, B) Copyright 2010, Royal Society of Chemistry; (C—G) Copyright 2016, American Chemical Society.
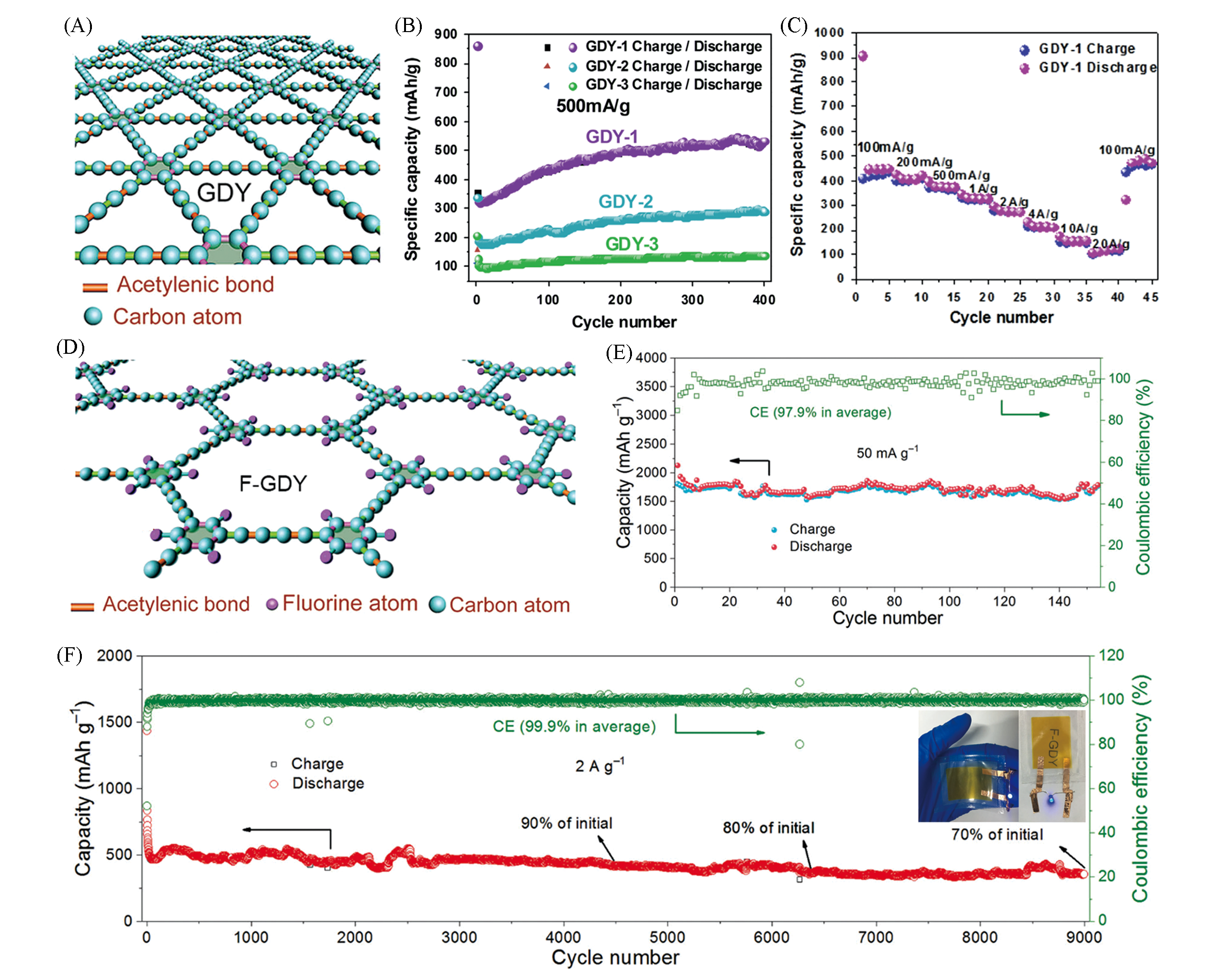
Fig.2 Structure and lithium ions storage performance of GDY and F?GDY(A) Structure of GDY[12]. Copyright 2019, The Royal Society of Chemistry. (B) Cycle performance of the GDY-1, GDY-2, and GDY-3 electrodes at a current density of 500 mA/g between 5 mV and 3 V; (C) rate performance of the GDY-1 electrode[18]. Copyright 2015, Elsevier. (D) Structure of F-GDY; (E, F) cycle performance of F-GDY at current densities of 50 mA/g(E) and 2 A/g(F)[24]. Copyright 2018, The Royal Society of Chemistry.
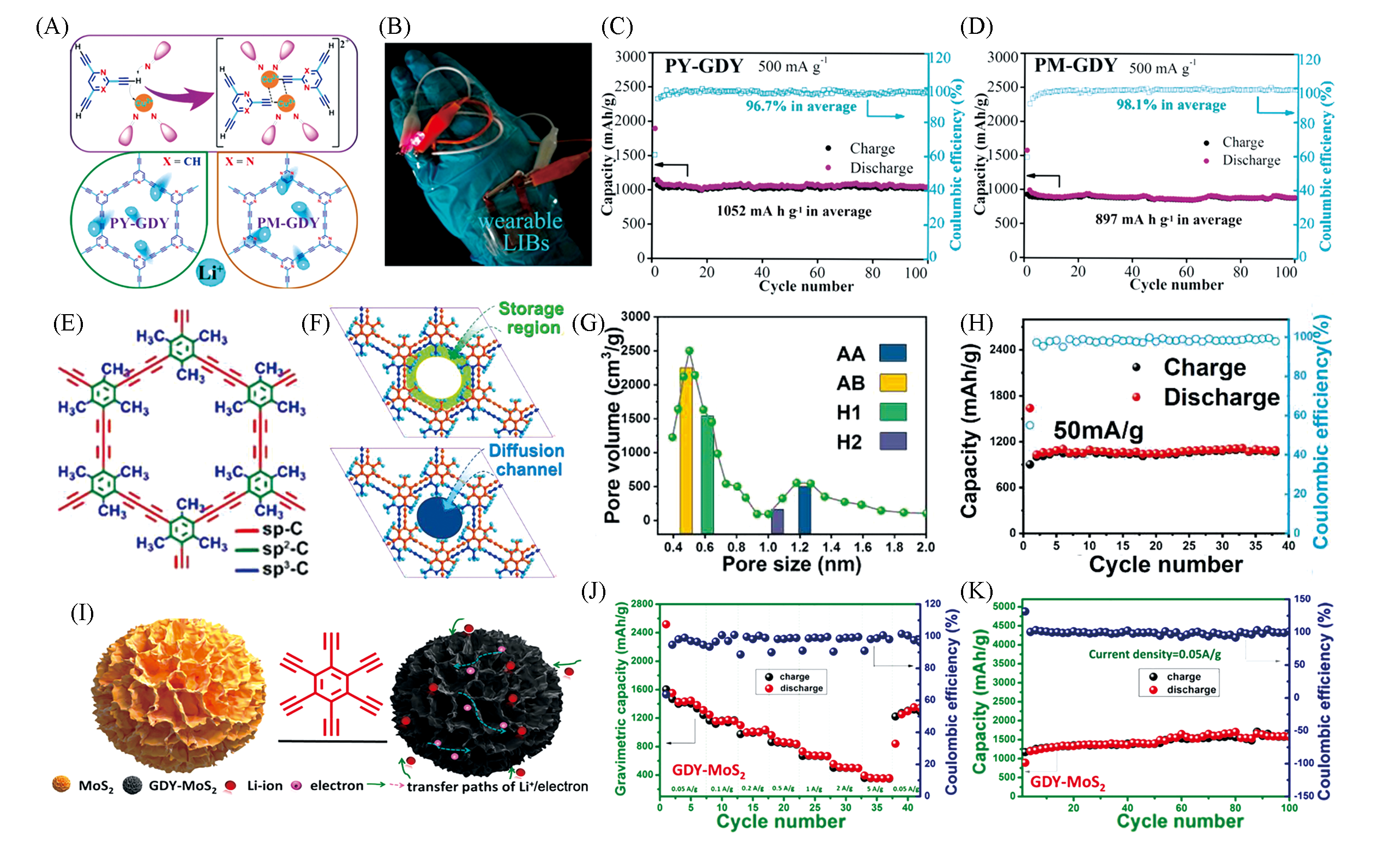
Fig.3 Structural characteristics and lithium storage performance of PY?GDY, PM?GDY, MsGDY and GDY?MoS2(A) Synthetic procedure of PY-GDY and PM-GDY; (B) photograph showing that a wearable soft package battery wrapped around a human hand can light up a LED lamp; (C, D) cycle performance of PY-GDY and PM-GDY-based electrodes at a current densities of 500 mA/g, respectively[27]. Copyright 2018, American Chemical Society. (E) Schematic diagram for the molecular fragment of MsGDY; (F) region from methyl to alkyne bond and the channel from methyl to the center; (G) experimental pore size distribution curve and simulated pore size distribution for MsGDY; (H) cycling performance of MsGDY[28]. Copyright 2020, Elsevier. (I—K) Synthetic procedure(I), rate capability(J) and cycling performance(K) of GDY-MoS2 hybrid nanomaterial[31]. Copyright 2019, Elsevier.
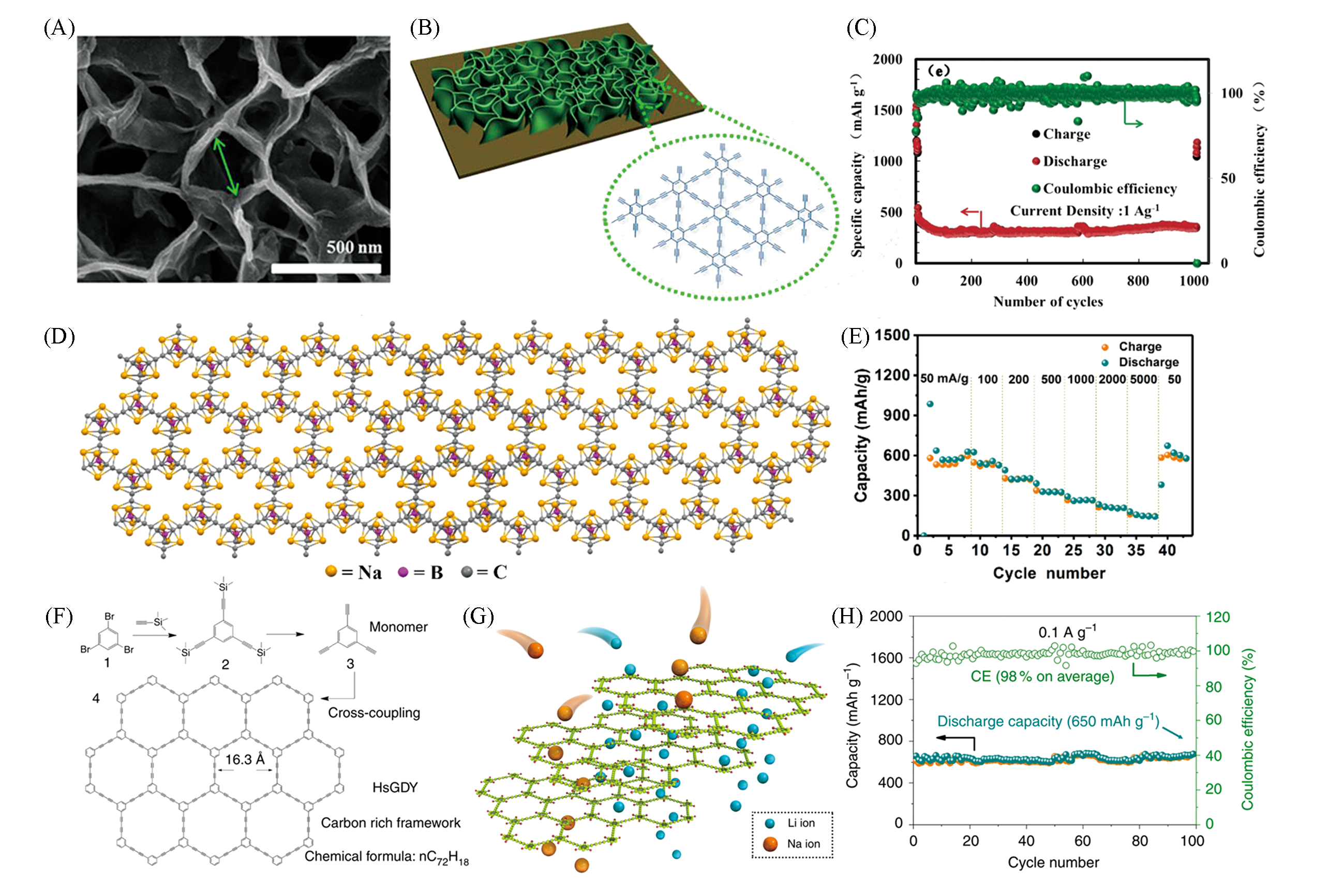
Fig.4 Sodium storage advantage of GDY?NS, HsGDY and BGDY materials(A) Top-view image of GDY-NS on the Cu substrate; (B) schematic illustration of the structure of GDY-NS on a Cu substrate; (C) cycle performance of the product-based electrodes at the current density of 1 A/g[37]. Copyright 2017, American Chemical Society. (D) The possible Na storage sites in BGDY; (E) rate performance of the electrode for NIBs[38]. Copyright 2018, Wiley-VCH. (F) Structure and appearance of HsGDY; (G) diffusion path of Li ions and Na ions in carbon-rich framework; (H) cycle performance of flexible electrode at the current density of 0.1 A/g[39]. Copyright 2017, The Author(s).
| Sample | Device | Capacity/(mA·h·g-1) | Current density/(mA·g-1) | Cycle | Ref. |
|---|---|---|---|---|---|
| Cl?GDY | LIBs | 750 | 200 | 170 | [ |
| N?GDY | LIBs | 785 | 200 | 200 | [ |
| P?GDY | LIBs | 1169 | 50 | 50 | [ |
| S?GDY | LIBs | 380 | 2000 | 1000 | [ |
| F?GDY | LIBs | 490 | 2000 | 2500 | [ |
| PY?GDY | LIBs | 764 | 5000 | 1500 | [ |
| PM?GDY | LIBs | 483 | 5000 | 4000 | [ |
| MsGDY | LIBs | 1020 | 50 | 40 | [ |
| H1F1?GDY | LIBs | 2050 | 50 | 50 | [ |
| GDY?MoS2 | LIBs | 1450 | 50 | 100 | [ |
| GDY?NS | SIBs | 405 | 1000 | 1000 | [ |
| BGDY | SIBs | 180 | 5000 | 4000 | [ |
| HsGDY | SIBs | 1050 | 100 | 100 | [ |
| HsGY | SIBs | 600 | 100 | 100 | [ |
Table 1 Summary of the performance of GDY-based electrodes in electrochemical batteries
| Sample | Device | Capacity/(mA·h·g-1) | Current density/(mA·g-1) | Cycle | Ref. |
|---|---|---|---|---|---|
| Cl?GDY | LIBs | 750 | 200 | 170 | [ |
| N?GDY | LIBs | 785 | 200 | 200 | [ |
| P?GDY | LIBs | 1169 | 50 | 50 | [ |
| S?GDY | LIBs | 380 | 2000 | 1000 | [ |
| F?GDY | LIBs | 490 | 2000 | 2500 | [ |
| PY?GDY | LIBs | 764 | 5000 | 1500 | [ |
| PM?GDY | LIBs | 483 | 5000 | 4000 | [ |
| MsGDY | LIBs | 1020 | 50 | 40 | [ |
| H1F1?GDY | LIBs | 2050 | 50 | 50 | [ |
| GDY?MoS2 | LIBs | 1450 | 50 | 100 | [ |
| GDY?NS | SIBs | 405 | 1000 | 1000 | [ |
| BGDY | SIBs | 180 | 5000 | 4000 | [ |
| HsGDY | SIBs | 1050 | 100 | 100 | [ |
| HsGY | SIBs | 600 | 100 | 100 | [ |
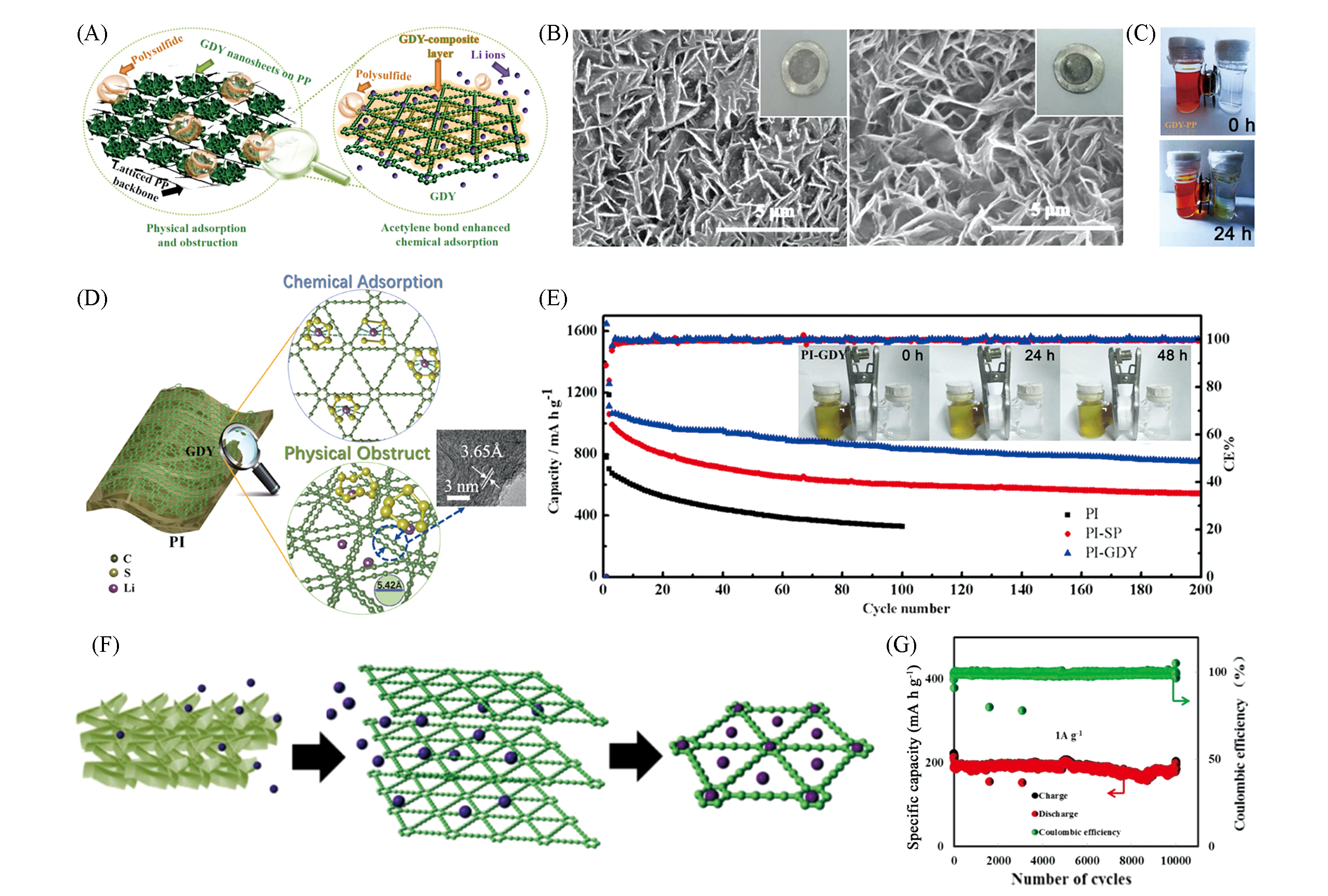
Fig.5 Application of GDY in lithium sulfur battery and lithium ion capacitor(A) Preparation of GDY-modulated PP separator; (B) as-prepared GDY-PP and separators based Li-S cells after 300 cycles; (C) color contrast of PSs solution in the left-side chambers and colourless electrolyte in the right-side chamber[47]. Copyright 2019, American Chemical Society. (D) Work mechanism between polysulfides and GDY including chemical adsorption and physical obstruction; (E) cycle performance with PI, PI-SP, and PI-GDY separators at 0.5 C[48]. Copyright 2019, American Chemical Society. (F) Suppositional lithium ions diffusion in hierarchical porous GDY-NW; (G) cycle performance of the GDY-NW at the current density of 1 A/g[49]. Copyright 2017, Elsevier.
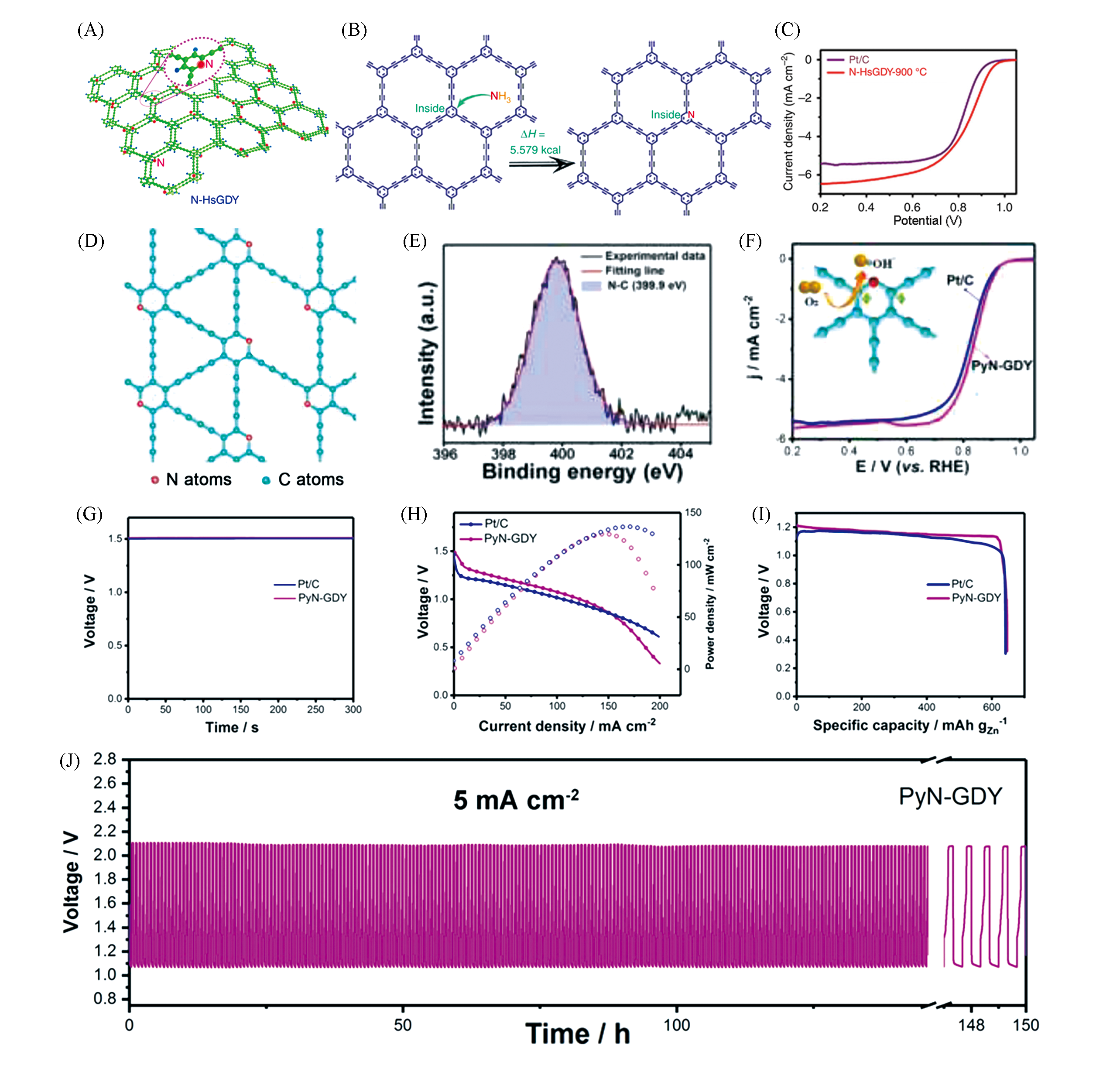
Fig.6 N?doped GDY and transition metal doped GDY applied for Zn?air batteries(A) Pyridinic nitrogen-doped hydrogen-substituted graphdiyne(N-HsGDY); (B) a reaction equation and enthalpy change for substituting C on the inside of HsGDY with pyridinic N; (C) linear sweep voltammetry curves of pyridinic nitrogen-doped hydrogen-substituted graphdiyne treated at 900 ℃[57]. Copyright 2017, the Author(s). (D) A fragment of PyN-GDY, eight possible C active sites for ORR; (E) XPS spectra of PyN-GDY; (F) RDE polarization curves of PyN-GDY and Pt/C(JM) in O2-saturated 0.1 mol/L KOH solution at a rotating speed of 1600 r/min with a scan rate of 5 mV/s; (G—I) open-circuit voltage(G), discharge polarization curve and corresponding power density curve(H) and discharge curves at a current density of 20 mA/cm2(I) of Zn-air batteries using PyN-GDY and Pt/C air electrode; (J) rechargeability cycling tests of the Zn-air batteries using PyN-GDY as cathode at current density of 5 mA/cm2[58]. Copyright 2020, Elsevier.
| 1 | Li Y. J., Xu L., Liu H. B., Li Y. L., Chem. Soc. Rev., 2014, 43(8), 2572—2586 |
| 2 | Novoselov K. S., Jiang Z., Zhang Y., Morozov S., Stormer H. L., Zeitler U., Maan J., Boebinger G., Kim P., Geim A. K., Science, 2007, 315(5817), 1379 |
| 3 | Huang C. S., Li Y. J., Wang N., Xue Y. R., Zuo Z. C., Liu H. B., Li Y. L., Chem. Rev., 2018, 118(16), 7744—7803 |
| 4 | Li G. X., Li Y. L., Liu H. B., Guo Y. B., Li Y. J., Zhu D. B., Chem. Commun.,2010, 46(19), 3256—3258 |
| 5 | Chen W. Q., Xiao P. S., Chen H. H., Zhang H. T., Zhang Q. C , Chen Y. S., Adv. Mater., 2019, 31(9), 1802403 |
| 6 | Gao X., Liu H. B., Wang D., Zhang J., Chem. Soc. Rev., 2019, 48(3), 908—936 |
| 7 | Inagaki M., Kang F. Y., J. Mater. Chem. A, 2014, 2(33), 13193—13206 |
| 8 | Pei Y., Physica B, 2012, 407(22), 4436—4439 |
| 9 | Wang N., He J. J., Wang K., Zhao Y. J., Jiu T. G., Huang C. S., Li Y. L., Adv. Mater., 2019, 31(42), 1803202 |
| 10 | Yang Z. B., Ren J., Zhang Z. T., Chen X. L., Guan G. Z., Qiu L. B., Zhang Y., Peng H. S., Chem. Rev., 2015, 115(11), 5159—5223 |
| 11 | Huang C. S., Zhao Y. L., Li Y. L., Adv. Mater., 2019, 31(42), 1904885 |
| 12 | He J. J., Li X. D., Lu T. T., Shen X. Y., Wang N., Huang C. S., Dalton Trans.,2019, 48(39), 14566—14574 |
| 13 | Zhang S. L., Du H. P., He J. J., Huang C. S., Liu H. B., Cui G. L., Li Y. L., ACS Appl. Mater. Interfaces, 2016, 8(13), 8467—8473 |
| 14 | Xie C. P., Wang N., Li X. F., Xu G. Y., Huang C. S., Chem. Eur. J.,2020, 26(3), 569—583 |
| 15 | Larcher D., Tarascon J. M., Nat. Chem.,2015, 7(1), 19 |
| 16 | Shen X. Y., He J. J., Wang N., Huang C. S., Acta Phys. Chim. Sin., 2018, 34(9), 1029—1047 |
| 17 | He J. J., Zhang C., Du H. P., Zhang S. L., Hu P., Zhang Z., Ma Y., Huang C. S., Cui G., Electrochim. Acta,2015, 178, 476—483 |
| 18 | Huang C. S., Zhang S. L., Liu H. B., Li Y. J., Cui G. L., Li Y. L., Nano Energy, 2015, 11, 481—489 |
| 19 | He J. J., Bao K. J., Cui W. W., Yu J. J., Huang C. S., Shen X. Y., Cui Z. L., Wang N., Chem. Eur. J., 2018, 24(5), 1187—1192 |
| 20 | Wang N., He J. J., Tu Z. Y., Yang Z., Zhao F. H., Li X. D., Huang C. S., Wang K., Jiu T. G., Yi Y. P., Angew. Chem. Int. Ed., 2017, 56(36), 10740—10745 |
| 21 | Lv Q., Si W. Y., Yang Z., Wang N., Tu Z. Y., Yi Y. P., Huang C. S., Jiang L., Zhang M. J., He J. J., Long Y. Z., ACS Appl. Mater. Interfaces, 2017, 9(35), 29744—29752 |
| 22 | Shen X. Y., Li X. D., Zhao F. H., Wang N., Xie C. P., He J. J., Si W. Y., Yi Y. P., Yang Z., Li X. F., 2D Mater., 2019, 6(3), 035020 |
| 23 | Yang Z., Cui W. W., Wang K., Song Y. W., Zhao F. H., Wang N., Long Y. Z., Wang H. L , Huang C. S., Chem. Eur. J., 2019, 25(22), 5643—5647 |
| 24 | He J. J., Wang N., Yang Z., Shen X. Y., Wang K., Huang C. S., Yi Y. P., Tu Z. Y., Li Y. L., Energy Environ. Sci., 2018, 11(10), 2893—2903 |
| 25 | Gao L., Ge X., Zuo Z., Wang F., Liu X., Lv M., Shi S., Xu L., Liu T., Zhou Q., Nano Letters,2020, 20(10), 7333—7341 |
| 26 | Ren X., Li X. D., Yang Z., Wang X., He J. J., Wang K., Yin J. G., Li J. Z., Huang C. S., ACS Sustain. Chem. Eng., 2020, 8(7), 2614—2621 |
| 27 | Yang Z., Shen X. Y., Wang N., He J. J., Li X. D., Wang X., Hou Z. F., Wang K., Gao J., Jiu T. G., ACS Appl. Mater. Interfaces, 2018, 11(3), 2608—2617 |
| 28 | Li X. D., Wang N., He J. J., Yang Z., Tu Z. Y., Zhao F. H., Wang K., Yi Y. P., Huang C. S., Carbon, 2020, 162, 579—585 |
| 29 | Xie C. P., Hu X. L., Guan Z. Y., Li X. D., Zhao F. H., Song Y. W., Li Y., Li X. F., Wang N., Huang C. S., Angew. Chem. Int. Ed., 2020, 26(3), 569—583 |
| 30 | Lu T. T., He J. J., Li R., Wang K., Yang Z., Shen X. Y., Li Y., Xiao J. C., Huang C. S., Energy Storage Mater., 2020, 29, 131—139 |
| 31 | Gao J., He J. J., Wang N., Li X. D., Yang Z., Wang K., Chen Y. H., Zhang Y. L., Huang C. S., Chem. Eng. J., 2019, 373, 660—667 |
| 32 | Gao J., Li J. F., Chen Y. H., Zuo Z. C., Li Y. J., Liu H. B., Li Y. L., Nano Energy, 2018, 43, 192—199 |
| 33 | Yabuuchi N., Kubota K., Dahbi M., Komaba S., Chem. Rev., 2014, 114(23), 11636—11682 |
| 34 | Hou H. S., Qiu X. Q., Wei W. F., Zhang Y., Ji X. B., Adv. EnergyMater., 2017, 7(24), 1602898 |
| 35 | Zhao F. H., Wang N., Zhang M. J., Sápi A., Yu J. J., Li X. D., Cui W. W., Yang Z., Huang C. S., Chem. Commun., 2018, 54(47), 6004—6007 |
| 36 | Zhang S. L., He J. J., Zheng J., Huang C. S., Lv Q., Wang K., Wang N., Lan Z. G., J. Mater. Chem. A, 2017, 5(5), 2045—2051 |
| 37 | Wang K., Wang N., He J. J., Yang Z., Shen X. Y., Huang C. S., ACS Appl. Mater. Interfaces, 2017, 9(46), 40604—40613 |
| 38 | Wang N., Li X. D., Tu Z. Y., Zhao F. H., He J. J., Guan Z. Y., Huang C. S., Yi Y. P., Li Y. L., Angew. Chem. Int. Ed., 2018, 130(15), 4032—4037 |
| 39 | He J. J., Wang N., Cui Z. L., Du H. P., Fu L., Huang C. S., Yang Z., Shen X., Yi Y. P., Tu Z. Y., Nat. Commun., 2017, 8(1), 1172 |
| 40 | Yang Z., Zhang C. F., Hou Z. F., Wang X., He J. J., Li X. D., Song Y. W., Wang N., Wang K., Wang H. L., Huang C. S., J. Mater. Chem. A, 2019, 7(18), 11186—11194 |
| 41 | Gao J., Wang N., He J. J., Yang Z., Huang C. S., 2D Mater., 2020, 7(2), 025032 |
| 42 | Manthiram A., Fu Y. Z., Chung S. H., Zu C. X., Su Y. S., Chem. Rev., 2014, 114(23), 11751—11787 |
| 43 | Rosenman A., Markevich E., Salitra G., Aurbach D., Garsuch A., Chesneau F. F., Adv. EnergyMater., 2015, 5(16), 1500212 |
| 44 | Du H. P., Zhang Z. H., He J. J., Cui Z. L., Chai J. C., Ma J., Yang Z., Huang C. S., Cui G. L., Small, 2017, 13(44), 1702277 |
| 45 | Zhang Y. S., Zhang P., Li B., Zhang S. J., Liu K. L., Hou R. H., Zhang X. L., Silva S., Shao G. S., Energy Storage Mater., 2020, 27, 159—168 |
| 46 | Zhang S. J., Zhang P., Hou R. H., Li B., Zhang Y. S., Liu K. L., Zhang X. L., Shao G. S., J. Energy Chem.,2020, 47, 281—290 |
| 47 | Wang J., Wang K., Yang Z., Li X. D., Gao J., He J. J., Wang N., Wang H. L., Zhang Y. L., Huang C. S., ACSSustain. Chem. Eng., 2019, 8(4), 1741—1750 |
| 48 | Wang Y. Q., He J. J., Zhang Z. Q., Liu Z. H., Huang C. S., Jin Y. Z., ACS Appl. Mater. Interfaces, 2019, 11(39), 35738—35745 |
| 49 | Wang K., Wang N., He J. J., Yang Z., Shen X. Y., Huang C. S., Electrochim. Acta, 2017, 253, 506—516 |
| 50 | Winter M., Brodd R. J., Chem. Rev., 2005, 105(3), 1021 |
| 51 | Xu J., Li J. Q., Yang Q. L., Xiong Y., Chen C. G., Electrochim. Acta, 2017, 251, 672—680 |
| 52 | Du H. P., Yang H., Huang C. S., He J. J., Liu H. B., Li Y. L., Nano Energy, 2016, 22, 615—622 |
| 53 | Shen X. Y., He J. J., Wang K., Li X. D., Wang X., Yang Z., Wang N., Zhang Y. L., Huang C. S., ChemSusChem., 2019, 12(7), 1342—1348 |
| 54 | Shen X. Y., Yang Z., Wang K., Wang N., He J. J., Du H. P., Huang C. S., ChemElectroChem., 2018, 5(11), 1435—1443 |
| 55 | Wang Y. J., Zhao N. N., Fang B. Z., Li H., Bi X. T., Wang H. J., Chem. Rev., 2015, 115(9), 3433—3467 |
| 56 | Shao M. H., Chang Q. W., Dodelet J. P., Chenitz R., Chem. Rev., 2016, 116(6), 3594—3657 |
| 57 | Lv Q., Si W. Y., He J. J., Sun L., Zhang C. F., Wang N., Yang Z., Li X. D., Wang X., Deng W. Q., Long Y. Z., Huang C. S., Li Y. L., Nat. Commun., 2018, 9(1), 3376 |
| 58 | Lv Q., Wang N., Si W. Y., Hou Z. F., Li X., Wang X., Zhao F. H., Yang Z., Zhang Y., Huang C. S., Appl. Catal. B-Environ., 2020, 261, 118234 |
| 59 | Wang K., Li X. D., Wang N., Yang Z., Gao J., He J. J., Zhang Y. L., Huang C. S., ACS Appl. Energy Mater., 2020, 3(3), 2623—2633 |
| 60 | Wang K., Li X. D., Xie Y., He J. J., Yang Z., Shen X. Y., Wang N., Huang C. S., ACS Appl. Mater. Interfaces, 2019, 11(27), 23990—23999 |
| [1] | 贾洋刚, 邵霞, 程婕, 王朋朋, 冒爱琴. 赝电容控制型钙钛矿高熵氧化物La(Co0.2Cr0.2Fe0.2Mn0.2Ni0.2)O3负极材料的制备及储锂性能[J]. 高等学校化学学报, 2022, 43(8): 20220157. |
| [2] | 黄汉浩, 卢湫阳, 孙明子, 黄勃龙. 石墨炔原子催化剂的崭新道路:基于自验证机器学习方法的筛选策略[J]. 高等学校化学学报, 2022, 43(5): 20220042. |
| [3] | 陈朝阳, 薛玉瑞, 李玉良. 石墨炔零价金属原子催化剂的合成及应用[J]. 高等学校化学学报, 2022, 43(5): 20220063. |
| [4] | 鲍俊全, 郑仕兵, 苑旭明, 史金强, 孙田将, 梁静. 有机盐PTO(KPD)2作为高性能锂离子电池正极材料的研究[J]. 高等学校化学学报, 2021, 42(9): 2911. |
| [5] | 李辉阳, 朱思颖, 李莎, 张桥保, 赵金保, 张力. 锂离子电池硅氧化物负极首次库伦效率的影响因素与提升策略[J]. 高等学校化学学报, 2021, 42(8): 2342. |
| [6] | 卓增庆, 潘锋. 基于软X射线光谱的锂电池材料的电子结构与演变的研究进展[J]. 高等学校化学学报, 2021, 42(8): 2332. |
| [7] | 吴卓彦, 李至, 赵旭东, 王倩, 陈顺鹏, 常兴华, 刘志亮. 一步法高效制备纳米Si/C复合材料及其在高性能锂离子电池中的应用[J]. 高等学校化学学报, 2021, 42(8): 2500. |
| [8] | 易聪华, 苏华坚, 钱勇, 李琼, 杨东杰. 木质素纳米炭的制备及作为锂离子电池负极的性能研究[J]. 高等学校化学学报, 2021, 42(6): 1807. |
| [9] | 高娟, 孙全虎, 黄长水. 石墨炔纳米材料的制备及在电化学能源中的应用[J]. 高等学校化学学报, 2021, 42(5): 1501. |
| [10] | 王弈艨, 刘凯, 王保国. 高镍三元正极材料的表面包覆策略[J]. 高等学校化学学报, 2021, 42(5): 1514. |
| [11] | 毛尔洋, 王莉, 孙永明. 锂离子电池高容量合金基含锂负极材料的研究进展[J]. 高等学校化学学报, 2021, 42(5): 1552. |
| [12] | 刘铁峰, 张奔, 盛欧微, 佴建威, 王垚, 刘育京, 陶新永. 硅负极黏结剂的研究进展[J]. 高等学校化学学报, 2021, 42(5): 1446. |
| [13] | 石颖, 胡广剑, 吴敏杰, 李峰. 低温等离子体在锂离子电池材料中的应用[J]. 高等学校化学学报, 2021, 42(5): 1315. |
| [14] | 王任衡, 肖哲, 李艳, 孙一翎, 范姝婷, 郑俊超, 钱正芳, 贺振江. 固相烧结法制备锂离子电池正极材料Li2FeP2O7及其电化学性能研究[J]. 高等学校化学学报, 2021, 42(4): 1299. |
| [15] | 刘志刚, 李家宝, 杨剑, 马浩, 王赪胤, 郭鑫, 汪国秀. 新型石墨化氮化碳/锡/氮掺杂碳复合物的制备及储钠性能[J]. 高等学校化学学报, 2021, 42(2): 633. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||