

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (5): 1514.doi: 10.7503/cjcu20200489
收稿日期:2020-07-23
出版日期:2021-05-10
发布日期:2020-11-27
通讯作者:
刘凯
E-mail:liukai2019@tsinghua.edu.cn
基金资助:
WANG Yimeng, LIU Kai( ), WANG Baoguo
), WANG Baoguo
Received:2020-07-23
Online:2021-05-10
Published:2020-11-27
Contact:
LIU Kai
E-mail:liukai2019@tsinghua.edu.cn
Supported by:摘要:
锂离子电池(LIBs)因具有更高的重量/体积能量密度、 更长的使用寿命、 更低的自放电率等优点而逐渐被广泛应用. 相比于已经广泛使用的钴酸锂和磷酸铁锂等正极材料, 高镍三元正极材料Li[Ni1-x-yCoxMny]O2(NCM)以其高电压和高容量等优点, 逐渐成为下一代高能锂离子电池的首选正极材料之一. 尽管高镍NCM正极材料具有上述优点, 但在进一步的实际应用前还需解决其循环稳定性、 倍率性能和安全性等问题, 这些问题主要源于NCM材料本身的晶体结构不稳定、 正极-电解液间界面副反应及高界面电阻等. 针对这些问题, 目前对高镍NCM正极电化学性能优化的大量研究都与电极-电解液界面有关, 如何通过改善界面稳定性、 增加离子在固液界面的迁移率、 抑制界面副反应、 提高正极材料的稳定性进而改善电池性能成为了关注焦点. 本文总结了目前对于其电化学性能衰减的机理解释, 分类概括了包括电化学惰性包覆锂、 残积物清除剂包覆和锂离子良导体包覆等对于高镍NCM正极材料的颗粒表面包覆策略, 简述了一些新兴的包覆策略, 并对高镍NCM正极材料的发展方向和前景提出了展望.
中图分类号:
TrendMD:
王弈艨, 刘凯, 王保国. 高镍三元正极材料的表面包覆策略. 高等学校化学学报, 2021, 42(5): 1514.
WANG Yimeng, LIU Kai, WANG Baoguo. Coating Strategies of Ni-rich Layered Cathode in LIBs. Chem. J. Chinese Universities, 2021, 42(5): 1514.
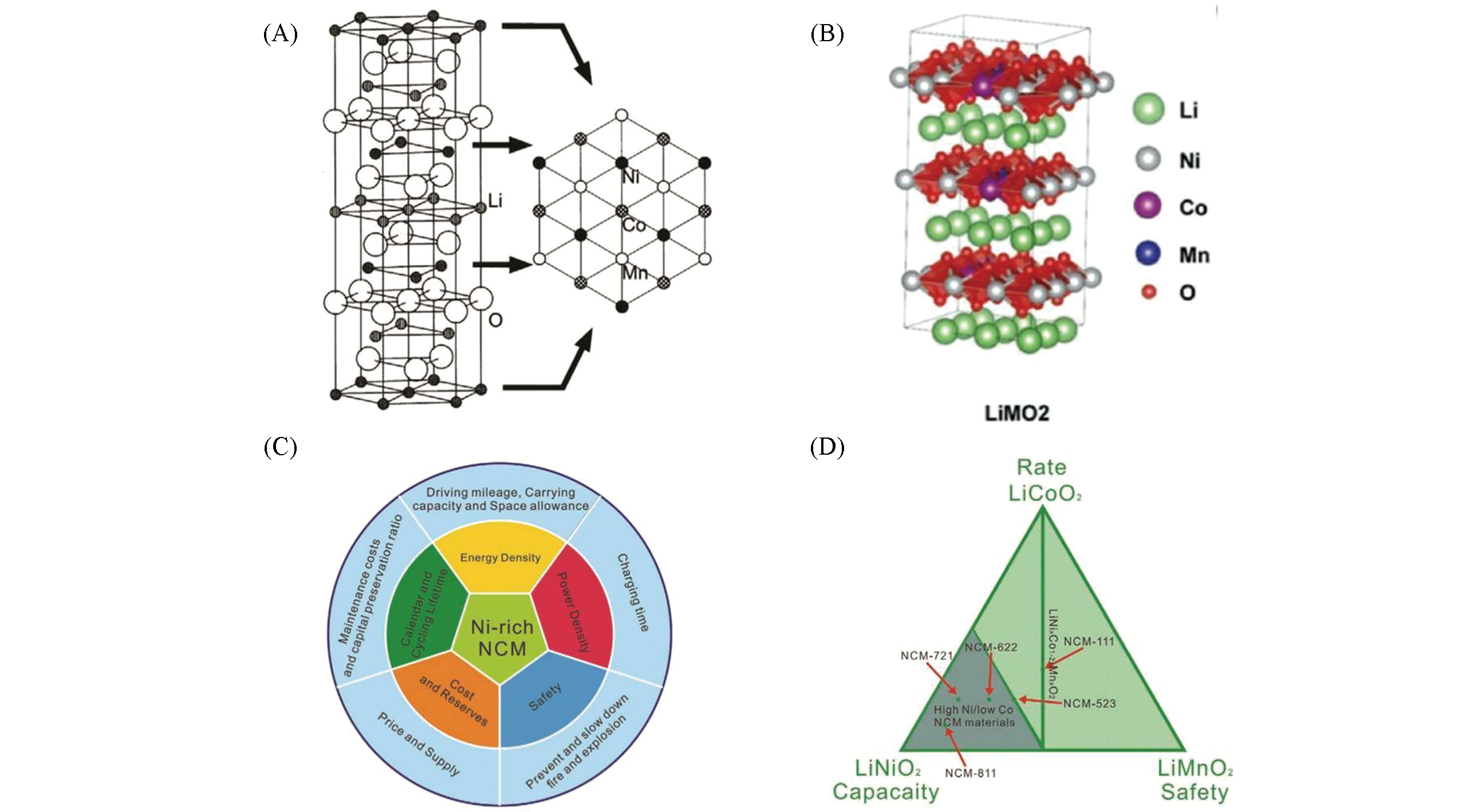
Fig.2 Ni?rich layered cathode(A), (B) Atomic configuration for the layered NCM cathode material[11]. Copyright 2019, Wiley‐VCH; (C) key parameters of Ni-rich NCM cathode in LIBs for EV application; (D) compositional phase diagrams of layered lithium transition metal oxide[13]. Copyright 2020, Wiley‐VCH.
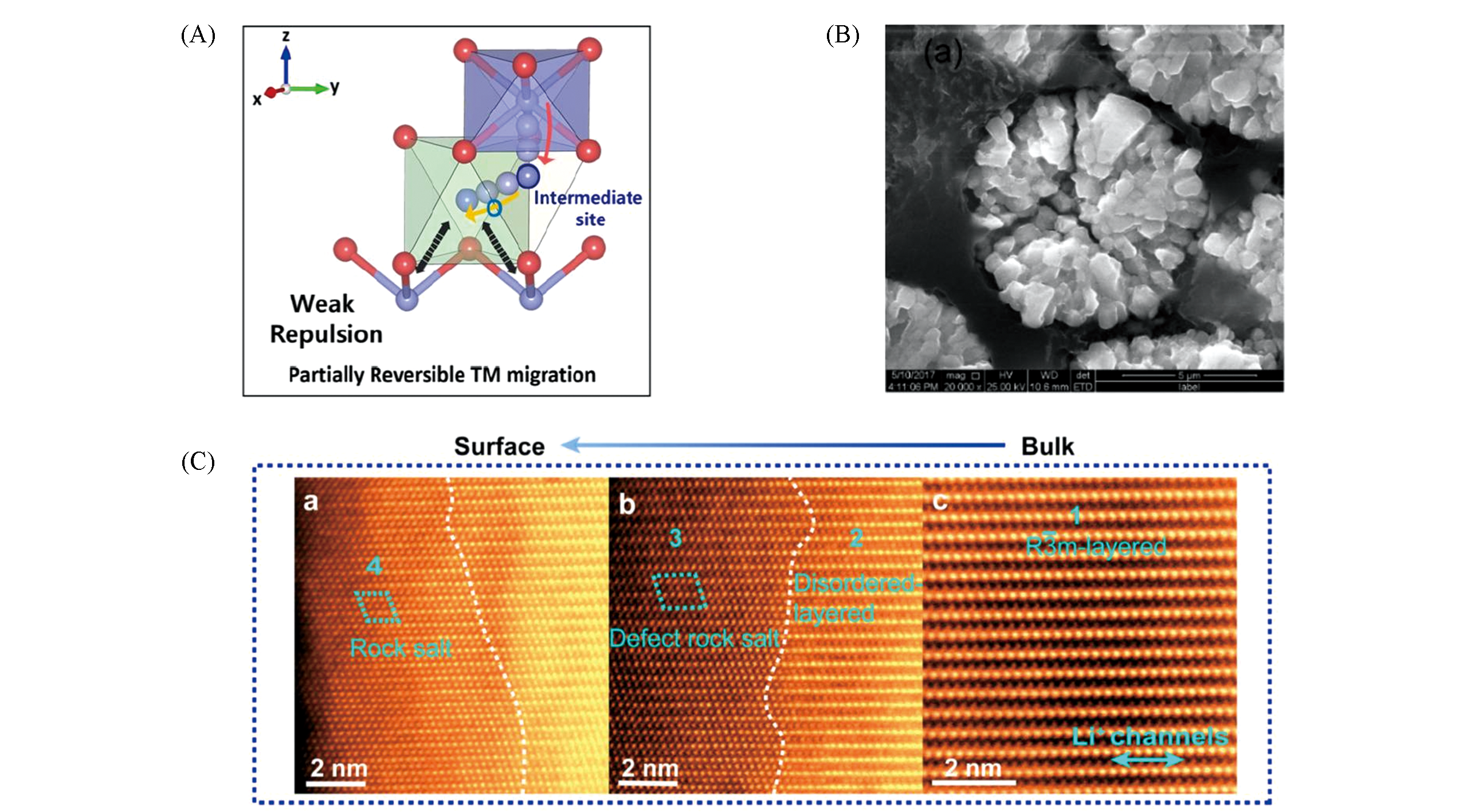
Fig.3 Surface/interface structure degradation of Ni?rich layered oxide cathodes(A) Partially cation mixed process with TM ions in Li slab[16]. Copyright 2020, Springer Nature; (B) SEM images of cross section for bare NCM811 cathode materials after 50 cycles[21]. Copyright 2017, American Chemical Society; (C) atomic-resolution STEM-HAADF(high angle annular dark field scanning transmission electron microscopy) images at different regions from bulk to surface. Detailed structural information from the layered region to disordered layered region, defect rock-salt region, and rock-salt region[19]. Copyright 2018, Elsevier.
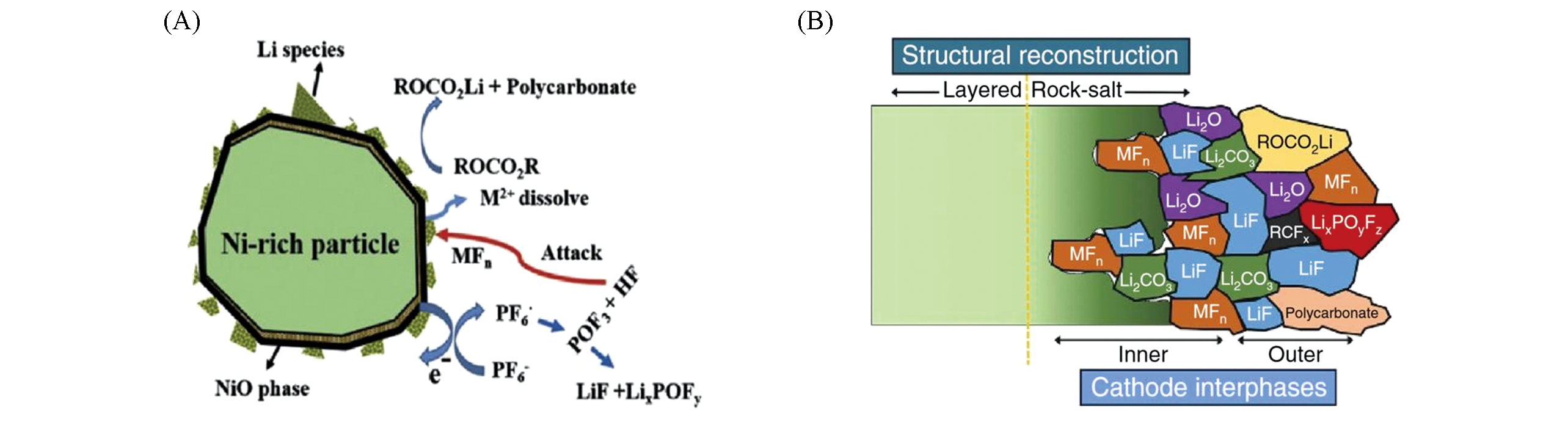
Fig.4 Schematic view of the intricate cathode?electrolyte interfacial(A)[23], structural and chemical reconstruction upon cycling(B)[24](A) Copyright 2016, Wiley‐VCH; (B) Copyright 2017, Springer Nature.
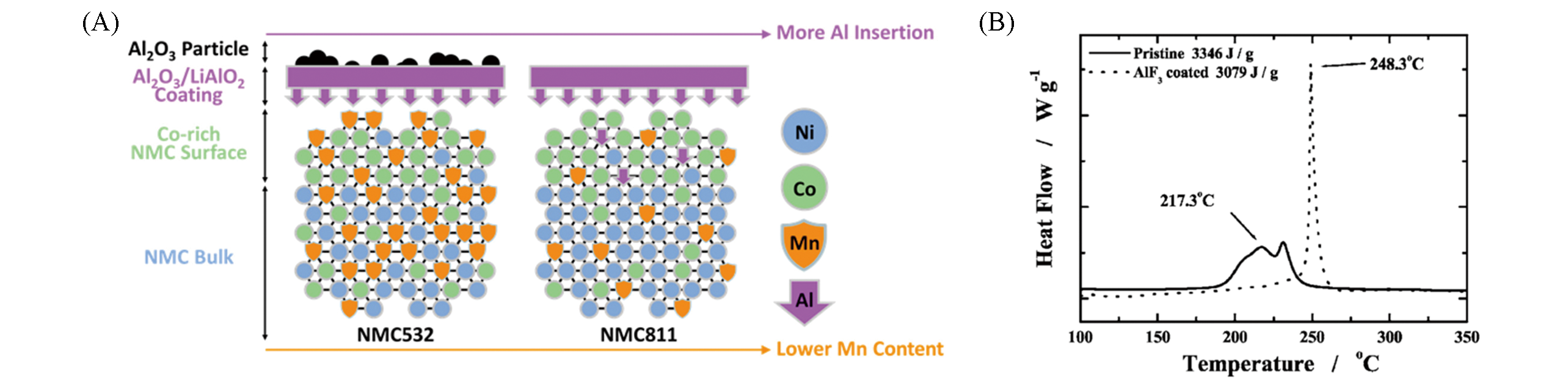
Fig.6 How the transition tetal composition affects alumina coatings on Ni?Rich cathodes(A)[34] and DSC profiles of pristine NCM811 and AlF3?coated NCM811 cells charged at state to 4.3 V(B)[37](A) Copyright 2017, American Chemical Society. (B) Copyright 2017, Ionics.
| Electrode material | Voltage range/V | Rate | Cycle | Specific capacity/(mA·h·g-1) | Capacity retention(%) | Ref. |
|---|---|---|---|---|---|---|
| Li?Zr?O@NCM811 | 3.0—4.3 | 0.5C | 50 | 175 | 94.3 | [ |
| MgO@NCM622 | 3.0—4.3 | 1C | 100 | 130 | 82 | [ |
| Al2O3@NCM523 | 3.0—4.5 | 1C | 100 | 140 | 85 | [ |
| Y2O3@NCM811 | 3.0—4.3 | 1C | 100 | 136 | 91.4 | [ |
| Al?doped ZnO@NCM523 | 3.5—4.5 | 1C | 200 | 134 | 90.2 | [ |
| YF3@NCM111 | 2.8—4.3 | 5C | 100 | 125 | 93 | [ |
| AlF3@NCM811 | 3.0—4.3 | 0.5C | 60 | — | 93 | [ |
| CeF3@NCM111 | 2.8—4.5 | 0.5C | 100 | 140 | 88 | [ |
| Na2SO4@NCM9 | 2.8—4.3 | 5C | 100 | — | 85.2 | [ |
| LixBOyFz@NCM622 | 42.8—4.3 | 1C | 50 | 174 | 97.4 | [ |
Table 1 Comparison of the improvement on the cycle performance of different inert-Coated Ni-rich NCM cathode materials
| Electrode material | Voltage range/V | Rate | Cycle | Specific capacity/(mA·h·g-1) | Capacity retention(%) | Ref. |
|---|---|---|---|---|---|---|
| Li?Zr?O@NCM811 | 3.0—4.3 | 0.5C | 50 | 175 | 94.3 | [ |
| MgO@NCM622 | 3.0—4.3 | 1C | 100 | 130 | 82 | [ |
| Al2O3@NCM523 | 3.0—4.5 | 1C | 100 | 140 | 85 | [ |
| Y2O3@NCM811 | 3.0—4.3 | 1C | 100 | 136 | 91.4 | [ |
| Al?doped ZnO@NCM523 | 3.5—4.5 | 1C | 200 | 134 | 90.2 | [ |
| YF3@NCM111 | 2.8—4.3 | 5C | 100 | 125 | 93 | [ |
| AlF3@NCM811 | 3.0—4.3 | 0.5C | 60 | — | 93 | [ |
| CeF3@NCM111 | 2.8—4.5 | 0.5C | 100 | 140 | 88 | [ |
| Na2SO4@NCM9 | 2.8—4.3 | 5C | 100 | — | 85.2 | [ |
| LixBOyFz@NCM622 | 42.8—4.3 | 1C | 50 | 174 | 97.4 | [ |
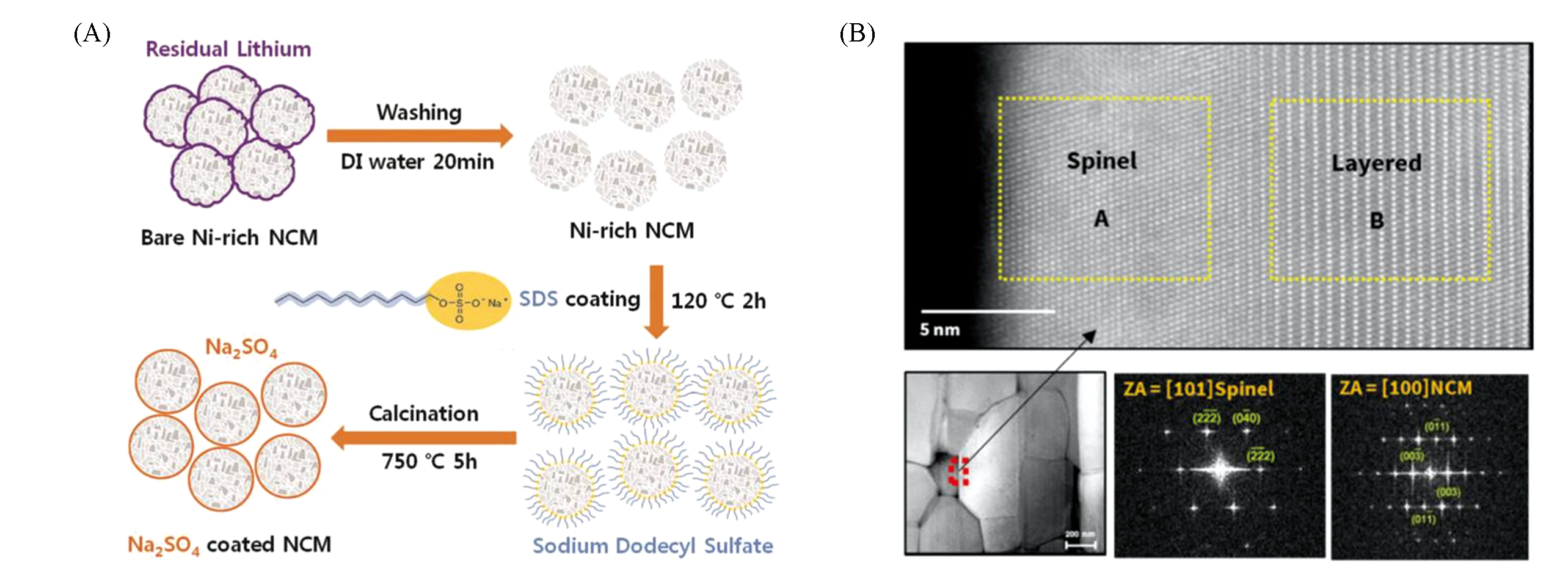
Fig.7 Key steps for the synthesis of Na2SO4‐coated NCM(A)[39], atomic?resolution STEM?HAADF image (top) from the corresponding region(bottom?left) in HAADF image taken at zone axis in NCM and indexed FFT images(bottom?middle and bottom?right) marked A and B(yellow dotted box), respec? tively(B)[40](A) Copyright 2016, Wiley‐VCH; (B) Copyright 2018, American Chemical Society.
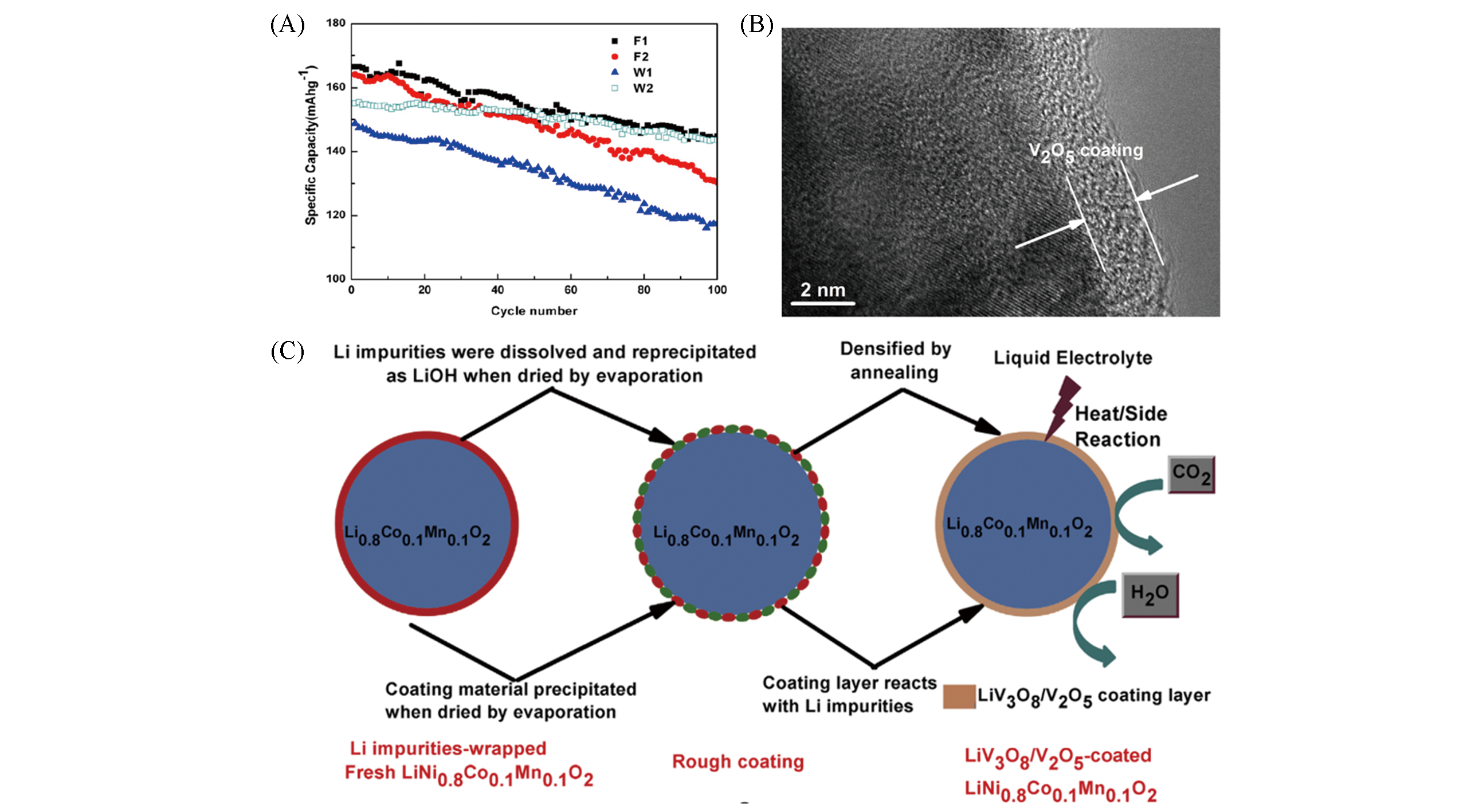
Fig.8 Cycle performance at 2C rate of sample F1, W1, W2 and F2(A)[42], schematic illustrations of LiV3O8/V2O5 coating layer formed onto NCM811 and its function as a protection skin to suppress the unwanted interfacial side reactions(B) and HRTEM images of the selected area in V2O5?coated electrodes(C)[43](A) Copyright 2012, Elsevier; (B,C) Copyright 2015, Elsevier.
| Electrode material | Voltage range/V | Rate | Rate performance | Ref. |
|---|---|---|---|---|
| Li2Si2O5@NCM622 | 2.75—4.5 | 0.1C, 1C, 2C, 3C, 5C, 10C | 213, 185, 170, 148, 122 | [ |
| Li2SO4@Li1.2[Ni0.54Co0.13Mn0.13]O2 | 2.0—4.8 | 0.2C, 0.5C, 1C, 3C | 245, 234, 223, 176 | [ |
| LiF@NCM811 | 2.8—4.3 | 0.1C, 0.5C, 1C, 2C, 5C, 10C | 204, 190, 182, 171, 148, 135 | [ |
| MoO3@NCM811 | 2.8—4.3 | 0.1C, 0.5C, 1C, 2C, 5C, 10C | 204, 196, 188, 182, 170, 160, 131 | [ |
Table 2 Comparison of the improvement on the cycle performance of different Li-reactive matter-Coated Ni-rich NCM cathode materials
| Electrode material | Voltage range/V | Rate | Rate performance | Ref. |
|---|---|---|---|---|
| Li2Si2O5@NCM622 | 2.75—4.5 | 0.1C, 1C, 2C, 3C, 5C, 10C | 213, 185, 170, 148, 122 | [ |
| Li2SO4@Li1.2[Ni0.54Co0.13Mn0.13]O2 | 2.0—4.8 | 0.2C, 0.5C, 1C, 3C | 245, 234, 223, 176 | [ |
| LiF@NCM811 | 2.8—4.3 | 0.1C, 0.5C, 1C, 2C, 5C, 10C | 204, 190, 182, 171, 148, 135 | [ |
| MoO3@NCM811 | 2.8—4.3 | 0.1C, 0.5C, 1C, 2C, 5C, 10C | 204, 196, 188, 182, 170, 160, 131 | [ |
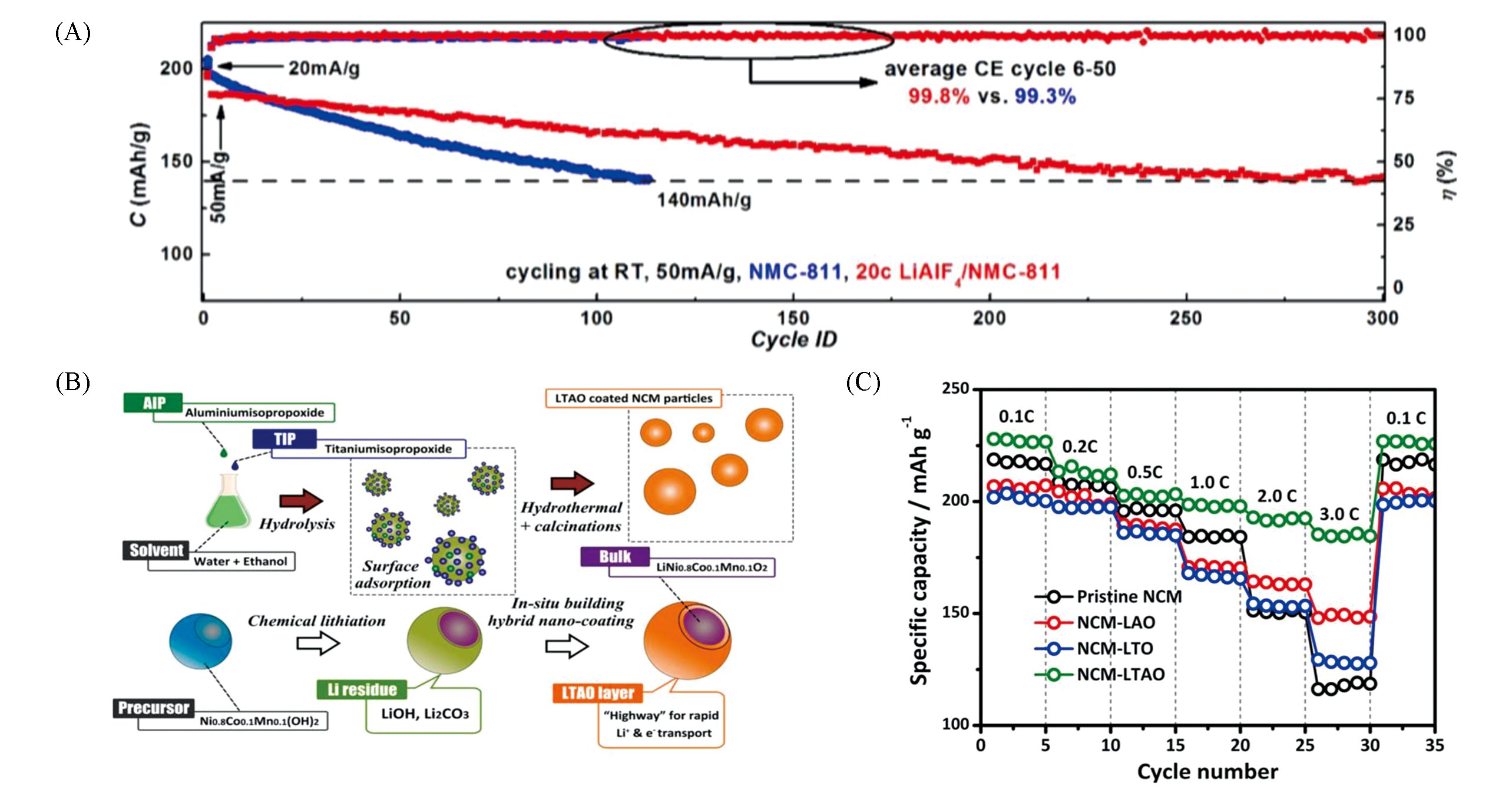
Fig.11 Cycle performance of pristine and 20c ALD LiAlF4 coated NMC811 electrodes at room temperature with an electrochemical window of 2.75—4.50 V(vs. Li+/Li)(A)[47], diagram for the design and synthesis of hybrid nano?membrane encapsulated NCM cathode material(B) and rate capability of pristine NCM, NCM?LAO, NCM?LTO, and NCM?LTAO cathode materials tested from 0.1C to 3.0C rate(C)[48](A) Copyright 2017, American Chemical Society; (B,C) Copyright 2018, American Chemical Society.
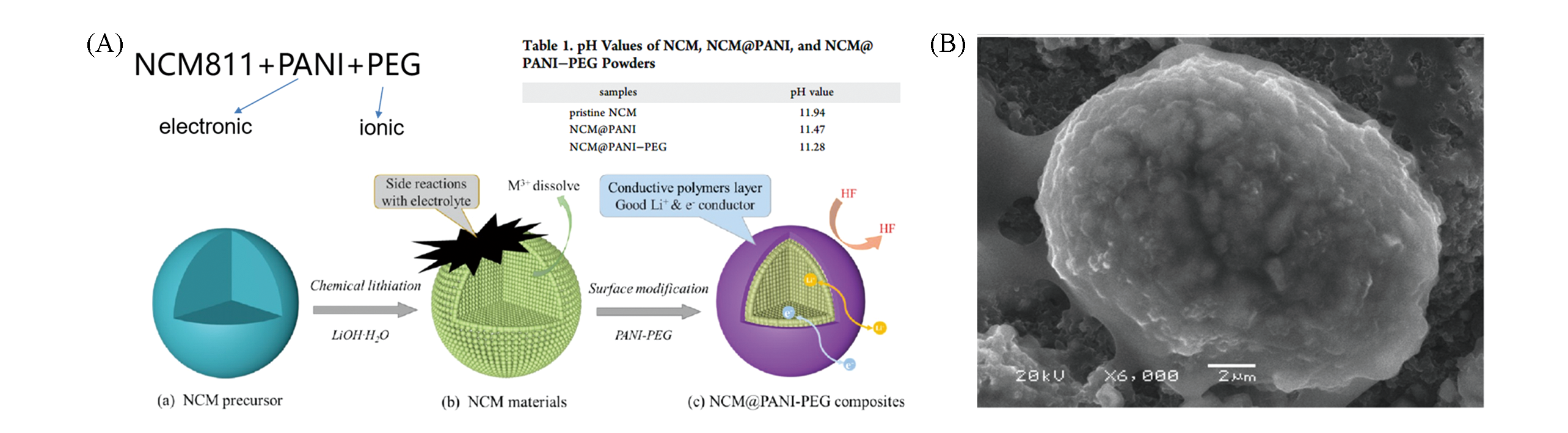
Fig.12 Schematic illustration of the synthesis of NCM@PANI?PEG composites(A) and SEM image of NCM@PANI?PEG sample(B)[54]Copyright 2018, American Chemical Society.
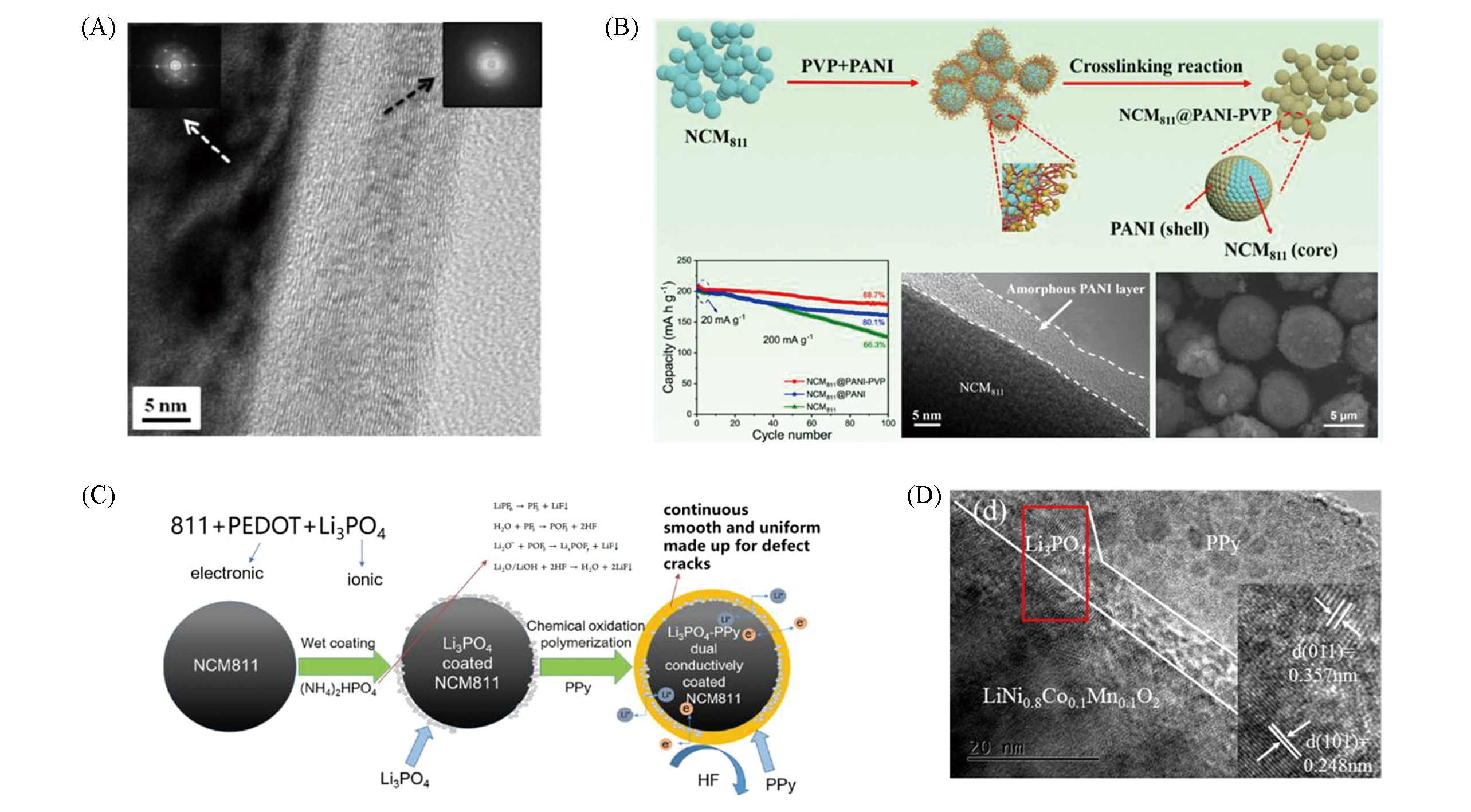
Fig.13 TEM image and SAED patterns of the surface?modi?ed NCM622 particle(left SAED: NCM622 particle, right SAED: coating layer)(A)[57], schematic illustration of the preparation of NCM811@PANI?PVP(B)[58] and schematic of dual conductively coating of NCM811 cathode material with Li3PO4 and PPy(C) and TEM images of PPy?LP samples(D)[59](A) Copyright 2017, American Chemical Society; (B) Copyright 2017, American Chemical Society; (C,D) Copyright 2017, American Chemical Society.
| Electrode material | Li+ diffusion conductivity, D+Li/(cm2·s-1) | Voltage range/V | Rate/C | Rate performance | Ref. |
|---|---|---|---|---|---|
| Li2SiO3@NCM622 | 1.49×10-10 | 2.8—4.4 | 0.2, 0.5, 1, 2, 5 | 196, 195, 191, 168, 158 | [ |
| LATO@NCM811 | 5.40×10-11 | 3.0—4.6 | 0.5, 1, 2, 3, 6 | 172, 161, 140, 130, 100 | [ |
| LaPO4@NCM811 | 1.42×10-11 | 3.0—4.3 | 0.1, 1, 2, 5, 10 | 197, 179, 166, 144, 124 | [ |
| PEDOT?PEG@NCM622 | 4.20×10-3 | 2.8—4.5 | 0.1, 0.5, 1, 2, 5 | 185, 183, 179, 175, 169 | [ |
| PANI?PEG@NCM811 | 2.85×10-2 | 2.8—4.3 | 0.2, 0.5, 1, 2, 5, 10 | 200, 195, 186, 177, 167, 156 | [ |
| PANI?PVP@NCM811 | — | 2.8—4.3 | 20, 40, 100, 200, 400, 1000 | 225, 218, 208, 190, 180, 150 | [ |
| PPy?LPO@NVM811 | 2.40×10-10 | 2.8—4.5 | 0.1, 0.5, 1, 2, 5, 10 | 205, 204, 198, 190, 177, 159 | [ |
Table 3 Comparison of the improvement on the cycle performance of different Li+ conductive material-Coated Ni-rich NCM cathode
| Electrode material | Li+ diffusion conductivity, D+Li/(cm2·s-1) | Voltage range/V | Rate/C | Rate performance | Ref. |
|---|---|---|---|---|---|
| Li2SiO3@NCM622 | 1.49×10-10 | 2.8—4.4 | 0.2, 0.5, 1, 2, 5 | 196, 195, 191, 168, 158 | [ |
| LATO@NCM811 | 5.40×10-11 | 3.0—4.6 | 0.5, 1, 2, 3, 6 | 172, 161, 140, 130, 100 | [ |
| LaPO4@NCM811 | 1.42×10-11 | 3.0—4.3 | 0.1, 1, 2, 5, 10 | 197, 179, 166, 144, 124 | [ |
| PEDOT?PEG@NCM622 | 4.20×10-3 | 2.8—4.5 | 0.1, 0.5, 1, 2, 5 | 185, 183, 179, 175, 169 | [ |
| PANI?PEG@NCM811 | 2.85×10-2 | 2.8—4.3 | 0.2, 0.5, 1, 2, 5, 10 | 200, 195, 186, 177, 167, 156 | [ |
| PANI?PVP@NCM811 | — | 2.8—4.3 | 20, 40, 100, 200, 400, 1000 | 225, 218, 208, 190, 180, 150 | [ |
| PPy?LPO@NVM811 | 2.40×10-10 | 2.8—4.5 | 0.1, 0.5, 1, 2, 5, 10 | 205, 204, 198, 190, 177, 159 | [ |
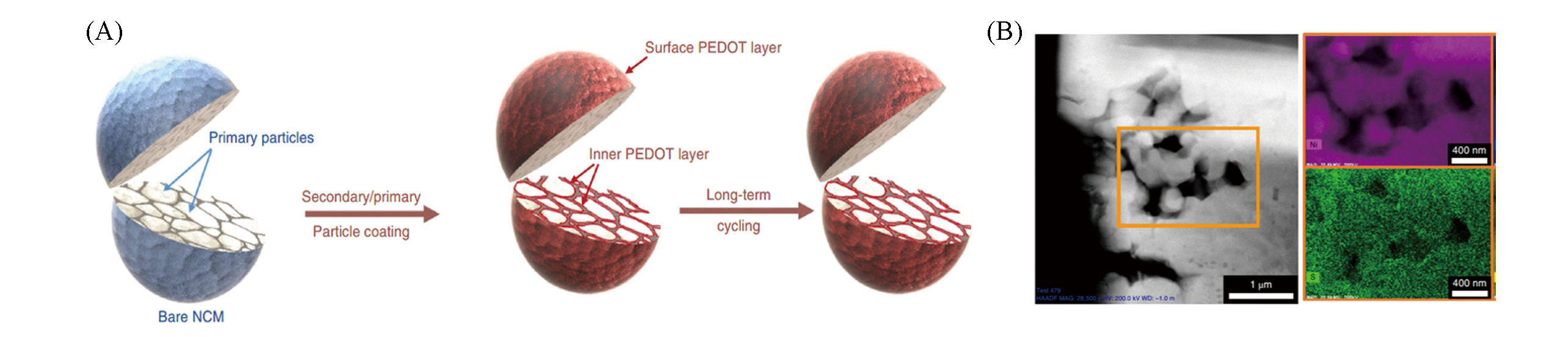
Fig.14 Illustration of the structural stability of both secondary/primary particle coating and secondary particle coating only after long?term cycling(A), STEM-HAADF image of the inner primary particles of 60?PEDOT@NCM111, and the corresponding EDS element mappings of S and Ni taken from the selected section(B)[62]Copyright 2019, Springer Nature.
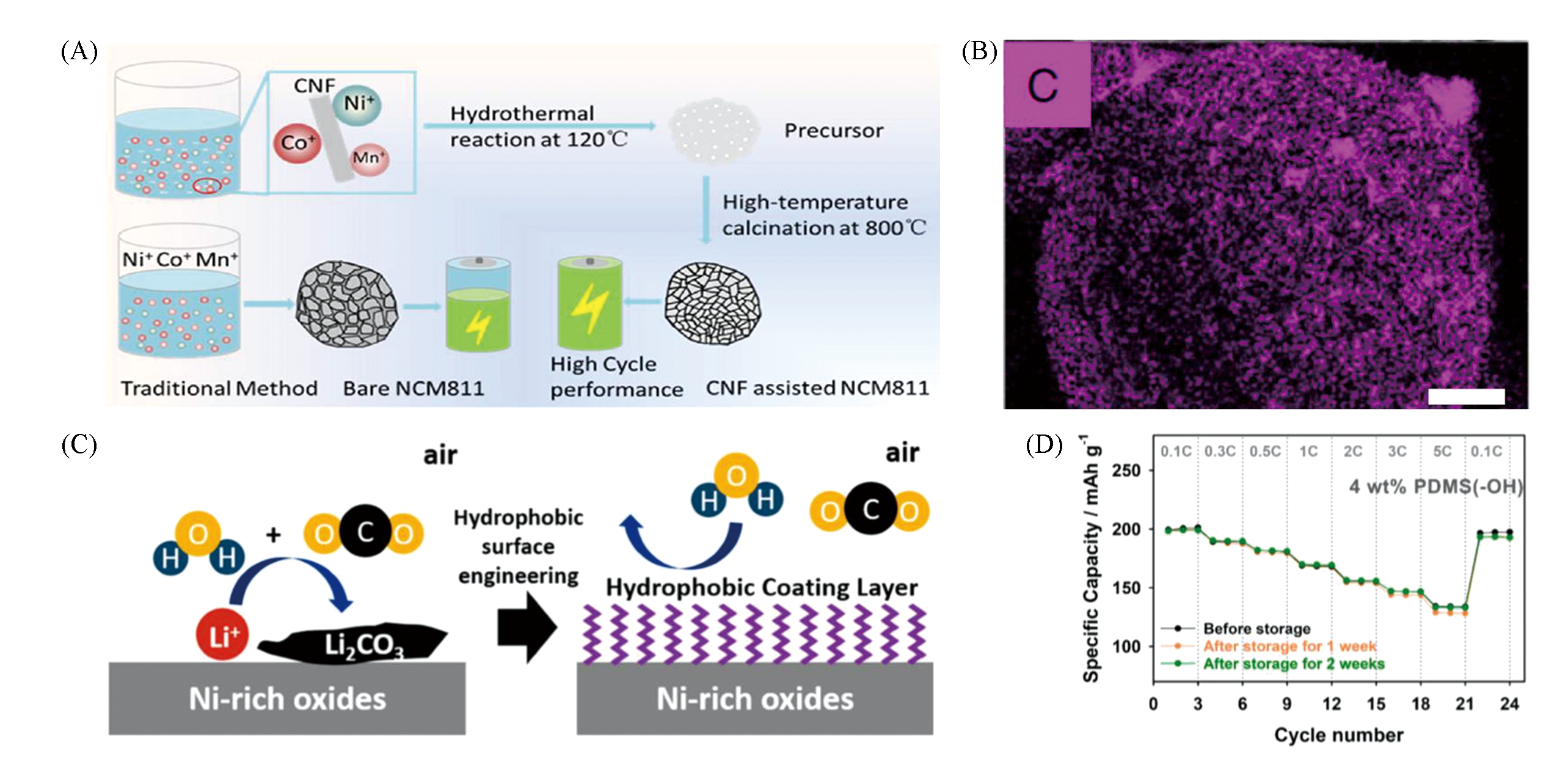
Fig.15 Schematic of the preparation procedure and related NCM811 particles(A) and EDX image of the CNF on NCM811 particles(B)[65], schematic concept of hydrophobic Ni?rich layered oxides(C), and rate performances of 4% PDMS(—OH)?grafted NCM811 electrodes before and after storage in a humidity chamber at 50% RH and 25 °C(D)[58](A, B) Copyright 2019, American Chemical Society; (C, D) Copyright 2019, American Chemical Society.
| 1 | John B. G., Youngsik K., Chem. Mater, 2010, 132(35), 1168—1175 |
| 2 | Xu K., Chem. Rev., 2014, 114(23), 11503—11618 |
| 3 | Ohzuku T., Ueda A., J. Electrochem. Soc., 1993, 110, 4—9 |
| 4 | Rougier A., Gravereau P., Delmas C., J. Electrochem. Soc., 1996, 402, 77—83 |
| 5 | Zhan C., Chen W., Energy Environ. Sci., 2018. 54, 313—321 |
| 6 | Harper G., Sommerville R., Kendrick E., Nature, 2019, 575, 75—86 |
| 7 | David L., Mohanty D., Geng L., Ruther R. E., Sefat A. S., Chem. Electro. Chem., 2019, 6, 5571—5580 |
| 8 | Liu Z., Yu A., Lee J., J. Power Sources, 1999, 81, 416—419 |
| 9 | Ohzuku T., Makimura Y., Chem. Lett., 2001, 1(7), 642—643 |
| 10 | Kim M. H., Shin H. S., Shin D., Sun Y. K., J. Power Sources, 2006, 159, 1328—1333 |
| 11 | Maleki Kheimeh Sari H., Li X., Adv. Energy Mater., 2019, 9, 1901597 |
| 12 | Choi J., Manthiram A., J. Electrochem. Soc., 2016, 327, 145—150 |
| 13 | Wang X., Ding Y. L., Deng Y. P., Chen Z., Adv. Energy Mater., 2020, 10, 1903864 |
| 14 | Noh H. J., Youn S., Yoon C. S., Sun Y. K., J. Power Sources, 2013, 233, 121—130 |
| 15 | Ma L., Nie M., Xia J., Dahn J. R., J. Power Sources, 2016, 327, 145—150 |
| 16 | Eum D., Kim B., Kim S. J., Park H., Wu J., Cho S. P., Yoon G., Lee M. H., Jung S. K., Yang W., Seong W. M., Ku K., Tamwattana O., Park S. K., Hwang I., Kang K., Nat. Mater., 2020, 19, 419—427 |
| 17 | Li Y., Hou X. H., Zhou Y., Han W. Q., Liang C., Wu X., Wang S. F., Energy Technol., 2018, 6, 391—396 |
| 18 | Bak S. M., Hu E., Zhou Y., Yu X., Senanayake S. D., Cho S. J., Kim K. B., Chung K. Y., Yang X. Q., Nam K. W., ACS Appl. Mater. Interface, 2014, 6, 22594—22601 |
| 19 | Lin Q., Guan W., Meng J., Huang W., Wei X., Zeng Y., Li J., Zhang Z., Nano Energy, 2018, 54, 313—321 |
| 20 | Kondrakov A. O., Schmidt A., Xu J., Geßwein H., Mönig R., Hartmann P., Sommer H., Brezesinski T., Janek J., J. Phys. Chem., 2017, 121, 3286—3294 |
| 21 | Chen S., He T., Su Y., Lu Y., Bao L., Chen L., Zhang Q., Wang J., Chen R., Wu F., ACS Appl. Mater. Interface, 2017, 9, 29732—29743 |
| 22 | Yan P., Zheng J., Zheng J., Wang Z., Teng G., Kuppan S., Xiao J., Chen G., Pan F., Zhang J. G., Wang C. M., Adv. Energy Mater., 2016, 6, 1502455 |
| 23 | Kim H., Lee S., Cho H., Kim J., Lee J., Park S., Joo S. H., Kim S. H., Cho Y. G., Song H. K., Kwak S. K., Cho J., Adv. Mater., 2016, 28, 4705—4712 |
| 24 | Li W., Dolocan A., Oh P., Nat. Comm., 2017, 8(1), 33546—33552 |
| 25 | Aurbach D., Markovsky B., Salitra G., Markevich E., Talyossef Y., Koltypin M., Nazar L., Ellis B., Kovacheva D., J. Power Sources, 2007, 165, 491—499 |
| 26 | Nicholas P., Zhongyi L., Peng L., J. Phys. Chem. C, 2013, 117, 15947—15957 |
| 27 | Zhu Z. Q., Tang Y. X., Zhang Y., Wang R., Zhang W., Angew. Chem. Int. Ed., 2018, 57, 3656—3660 |
| 28 | Biasi L., Schwarz B., Brezesinski T., Hartmann P., Janek J., Adv. Mater., 2019, 31, 1900985 |
| 29 | Yoon C., Jun D., Myung S., ACS Energy Lett., 2017, 2(5), 1150—1155 |
| 30 | Renfrew S. E., McCloskey B. D., J. Am. Chem. Soc., 2017, 139, 17853—17860 |
| 31 | Salitra G., Mccloskey D., RSC Adv., 2016, 6(23), 19233—19237 |
| 32 | Woo S., Yoon C., Amine K., J. Electrochem. Soc., 2007, (3), 447—454 |
| 33 | Myung S. T., Maglia F., Park K. J., Yoon C. S., Lamp P., Kim S. J., Sun Y. K., ACS Energy Lett., 2016, 2, 196—223 |
| 34 | Han B., Key B., Lapidus S., ACS Appl. Mater. Interface, 2017, 9(47), 41291—41302 |
| 35 | Hu G., Zhang M., Wu L., Peng Z., Du K., Cao Y., ACS Appl. Mater. Interface, 2016, 8, 33546—33552 |
| 36 | Kraytsberg A., Drezner H., Auinat M., Shapira A., Solomatin N., Axmann P., Wohlfahrt M., Ein⁃Eli Y., Chem. Nano. Mat., 2015, 1, 577—585 |
| 37 | Liu B., Zhang Z., Wan J., Ionics, 2017, 23(6), 1365—1374 |
| 38 | Woo S., Yoon C., Amine K., J. Electrochem. Soc., 2009, (6), 224—232 |
| 39 | Choi J., Kim J., Lee K. T., Lim J., Lee J., Yun Y. S., Adv. Mater. Interfaces, 2016, 3, 1600784 |
| 40 | Min K., Jung C., Ko D. S., Kim K., Jang J., Park K., Cho E., ACS Appl. Mater. Interface, 2018, 10, 20599—20610 |
| 41 | Zhuang G. V., Chen G., Shim J., Song X., Ross P. N., Richardson T. J., J. Power Sources, 2004, 134, 293—297 |
| 42 | Xiong X., Wang Z., Yue P., Guo H., Wu F., Wang J., Li X., J. Power Sources, 2013, 222, 318—325 |
| 43 | Xiong X., Wang Z., Guo C., Wu F., Guo H., Li X., J. Power. Sources, 2014, 245, 183—193 |
| 44 | Liu S., Wu H., Huang L., Xiang M., Liu H., Zhang Y., J. Alloys. Compd., 2016, 674, 447—454 |
| 45 | Ban L., Yin Y., Zhuang W., Electrochemical Acta, 2015, 180, 218—226 |
| 46 | Lee W., Kim M. S., Jeong J. H., Kim D. H., J. Power. Sources, 2016, 245, 137—145 |
| 47 | Xie J., Sendek A., Cubuk D., ACS Nano, 2017, 11(7), 7019 |
| 48 | Li L., Xu M., Yao Q., Chen Z., Song L., Zhang Z., Gao C., Wang P., Yu Z., Lai Y., ACS Appl. Mater. Interfaces, 2016, 8, 30879—30889 |
| 49 | Hou Q., Cao G., Wang P., Zhao D., Cui X., Li S., Li C., J. Alloys. Compd., 2018, 747, 796—802 |
| 50 | Cho W., Kim S. , Lee K W., Electrochimica. Acta, 2016, 402, 77—83 |
| 51 | Chen Z., Kim G. T., Guang Y., Bresser D., Diemant T., Huang Y., Copley M., Behm R. J., Passerini S., Shen Z., J. Power Sources, 2018, 402, 263—271 |
| 52 | Park K. S., Schougaard S. B., Goodenough J. B., Adv. Mater., 2007, 19, 848—851 |
| 53 | Song L. B., Tian F. L., Xue Z. L., J. Electron. Mater., 2018, 9(6),455—462 |
| 54 | Cao Y., Qi X., Hu K., Wang Y., Gan Z., Li Y., Hu G., Peng Z., Du K., ACS Appl. Mater. Interface, 2018, 10, 18270—18280 |
| 55 | Xiong X., Ding D., Wang Z., J. Solid State Electron., 2014, 18(9), 2619—2624 |
| 56 | Sun Z., Li Z., Gao L., Adv. Energy Mater., 2019, 9(6), 1802946 |
| 57 | Ju S. H., Kang I. S., Lee Y. S., Shin W. K., Kim S., Shin K., Kim D. W., ACS Appl. Mater. Interface, 2014, 6, 2546—2552 |
| 58 | Doo S. W., Lee S., Kim H., Choi J. H., Lee K. T., ACS Appl. Energy Mater., 2019, 2, 6246—6253 |
| 59 | Chen S., He T., Chen L., Lu Y., Wu F., ACS Appl. Mater. Interface, 2017, 9, 29732—29743 |
| 60 | Zhang J. P., Xue L., Li Y., Lei T., Deng S., Chen Y., Zhu J., Guo J., Chem. Electro. Chem., 2019, 6, 3125—3131 |
| 61 | Sun Y. K., Chen Z., Lee D. J., Nat. Mater., 2012, 11, 942—947 |
| 62 | Xu G. L., Liu Q., Lau K. K. S., Liu Y., Liu X., Gao H., Zhou X., Zhuang M., Ren Y., Li J., Shao M., Ouyang M., Pan F., Chen Z., Amine K., Chen G., Nat. Energy, 2019, 4, 484—494 |
| 63 | Yan P., Zheng J., Liu J., Wang B., Cheng X., Zhang Y., Sun X., Wang C., Zhang J. G., Nat. Energy, 2018, 3, 600—605 |
| 64 | Zhang M., Zhao H., Tan M., Liu J., Hu Y., Liu S., Shu X., Li H., Ran Q., Cai J., Liu X., J. Alloys. Compd., 2019, 774, 82—92 |
| 65 | Wu H., Pang X., Bi J., Wang L., Li Z., Guo L., Liu H., Meng Q., Jiang H., Liu C., Wang L., J. Alloys. Compd., 2020, 829, 154571 |
| 66 | Gan Q., Qin N., Zhu Y., ACS Appl. Mater. Interface, 2019, 11, 12594—12604 |
| 67 | Lee S. W., Kim M. S., Jeong J. H., Kim D. H., Chung K. Y., Roh K. C., Kim K. B., J. Power. Sources, 2017, 360, 206—214 |
| 68 | Kumar A., Nazzario R., Torres⁃Castro L., Pena⁃Duarte A., Tomar M. S., Inter. J. Hydrogen Energy, 2015, 40, 4931—4935 |
| 69 | Dai S., Yuan M., Wang L., Luo L., Chen Q., Xie T., Li Y., Yang Y., Ceramics Inter., 2019, 45, 674—680 |
| 70 | Dorai A. K., Subramanian S., Hellar N., Leiro J., Mater. Chem. Phys., 2014, 143, 765—772 |
| 71 | Song G., Kim B., Park J., J. Electro., 2012, 29(2), 163—169 |
| 72 | Huang J., Fang X., Wu Y., J. Electro. Chem., 2018, 45, 359—367 |
| 73 | Zhu J., Li Y., Xue L., Chen Y., Lei T., Deng S., Cao G., J. Alloys. Compd., 2019, 773, 112—120 |
| [1] | 贾洋刚, 邵霞, 程婕, 王朋朋, 冒爱琴. 赝电容控制型钙钛矿高熵氧化物La(Co0.2Cr0.2Fe0.2Mn0.2Ni0.2)O3负极材料的制备及储锂性能[J]. 高等学校化学学报, 2022, 43(8): 20220157. |
| [2] | 鲍俊全, 郑仕兵, 苑旭明, 史金强, 孙田将, 梁静. 有机盐PTO(KPD)2作为高性能锂离子电池正极材料的研究[J]. 高等学校化学学报, 2021, 42(9): 2911. |
| [3] | 李辉阳, 朱思颖, 李莎, 张桥保, 赵金保, 张力. 锂离子电池硅氧化物负极首次库伦效率的影响因素与提升策略[J]. 高等学校化学学报, 2021, 42(8): 2342. |
| [4] | 卓增庆, 潘锋. 基于软X射线光谱的锂电池材料的电子结构与演变的研究进展[J]. 高等学校化学学报, 2021, 42(8): 2332. |
| [5] | 吴卓彦, 李至, 赵旭东, 王倩, 陈顺鹏, 常兴华, 刘志亮. 一步法高效制备纳米Si/C复合材料及其在高性能锂离子电池中的应用[J]. 高等学校化学学报, 2021, 42(8): 2500. |
| [6] | 易聪华, 苏华坚, 钱勇, 李琼, 杨东杰. 木质素纳米炭的制备及作为锂离子电池负极的性能研究[J]. 高等学校化学学报, 2021, 42(6): 1807. |
| [7] | 毛尔洋, 王莉, 孙永明. 锂离子电池高容量合金基含锂负极材料的研究进展[J]. 高等学校化学学报, 2021, 42(5): 1552. |
| [8] | 刘铁峰, 张奔, 盛欧微, 佴建威, 王垚, 刘育京, 陶新永. 硅负极黏结剂的研究进展[J]. 高等学校化学学报, 2021, 42(5): 1446. |
| [9] | 石颖, 胡广剑, 吴敏杰, 李峰. 低温等离子体在锂离子电池材料中的应用[J]. 高等学校化学学报, 2021, 42(5): 1315. |
| [10] | 王任衡, 肖哲, 李艳, 孙一翎, 范姝婷, 郑俊超, 钱正芳, 贺振江. 固相烧结法制备锂离子电池正极材料Li2FeP2O7及其电化学性能研究[J]. 高等学校化学学报, 2021, 42(4): 1299. |
| [11] | 孙全虎, 卢天天, 何建江, 黄长水. 含异原子石墨炔基电极材料的研究进展[J]. 高等学校化学学报, 2021, 42(2): 366. |
| [12] | 高小雅, 左自成, 李玉良. 石墨炔电化学电池界面构筑[J]. 高等学校化学学报, 2021, 42(2): 321. |
| [13] | 周战, 马录芳, 谭超良. 层状钒青铜纳米片的制备及其锂离子电池阳极材料性能[J]. 高等学校化学学报, 2021, 42(2): 662. |
| [14] | 韩慕瑶, 赵丽娜, 孙洁. 硅及硅基负极材料的研究进展[J]. 高等学校化学学报, 2021, 42(12): 3547. |
| [15] | 巩珊珊, 吴彤, 王官格, 黄擎, 苏岳锋, 吴锋. 基于高效回收废旧锂离子电池正极材料的低共熔溶剂的筛选[J]. 高等学校化学学报, 2021, 42(10): 3151. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||