

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (2): 539.doi: 10.7503/cjcu20200579
陈明华( ), 李宏武, 范鹤, 李誉, 刘威铎, 夏新辉, 陈庆国
), 李宏武, 范鹤, 李誉, 刘威铎, 夏新辉, 陈庆国
收稿日期:2020-08-20
出版日期:2021-02-10
发布日期:2020-12-17
通讯作者:
陈明华
E-mail:mhchen@hrbust.edu.cn
基金资助:
CHEN Minghua( ), LI Hongwu, FAN He, LI Yu, LIU Weiduo, XIA Xinhui, CHEN Qingguo
), LI Hongwu, FAN He, LI Yu, LIU Weiduo, XIA Xinhui, CHEN Qingguo
Received:2020-08-20
Online:2021-02-10
Published:2020-12-17
Contact:
CHEN Minghua
E-mail:mhchen@hrbust.edu.cn
Supported by:摘要:
近年来, 过渡金属硫族化合物(TMDs)作为一种新兴的二维材料, 因其独特的层状结构及电学特性成为超级电容器电极材料的理想候选者之一. 本文介绍了二维TMDs的常用合成方法, 阐述了钼基、 钨基和钒基等TMDs在超级电容器中的研究进展, 分析了形貌、 尺寸和改性方法等因素对TMDs材料电化学性能的影响, 并对TMDs在超级电容器领域的工业化应用和挑战进行了总结与展望.
中图分类号:
TrendMD:
陈明华, 李宏武, 范鹤, 李誉, 刘威铎, 夏新辉, 陈庆国. 二维过渡金属硫族化合物在超级电容器中的研究进展. 高等学校化学学报, 2021, 42(2): 539.
CHEN Minghua, LI Hongwu, FAN He, LI Yu, LIU Weiduo, XIA Xinhui, CHEN Qingguo. Research Progress of Two-dimensional Transition Metal Dichalcogenides in Supercapacitors. Chem. J. Chinese Universities, 2021, 42(2): 539.
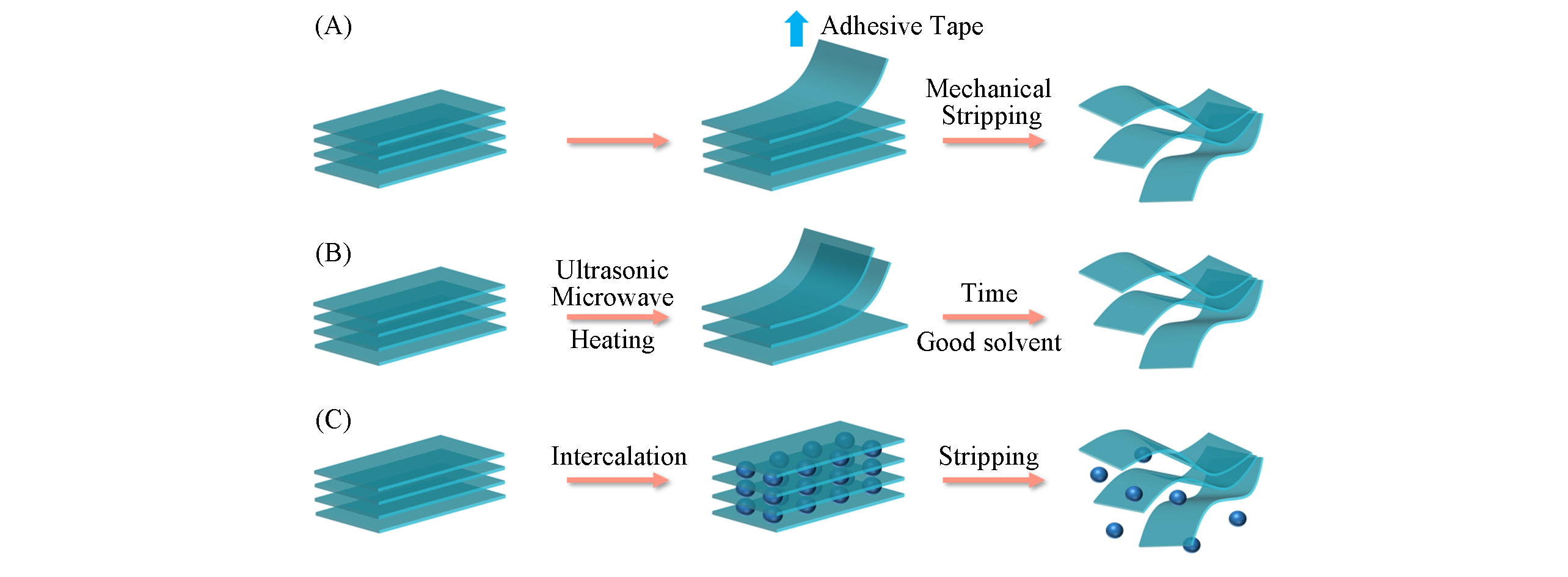
Fig.2 Schematic diagram of common stripping methods(A) Mechanical stripping; (B) sonication-assiste stripping; (C) ion intercalation assisted liquid phase stripping.
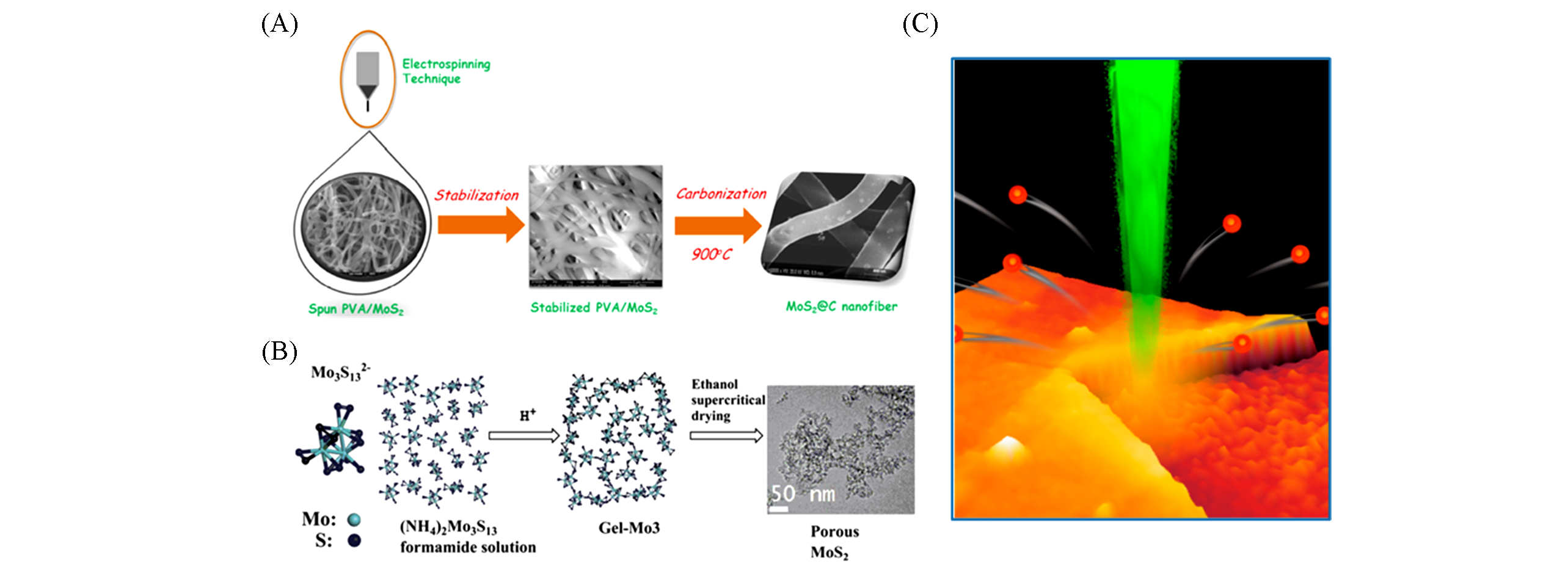
Fig.4 Other TMDs preparation methods(A) Preparation of MoS2@C nanofibers by electrospinning technology[73], Copyright 2017, Elsevier B.V.; (B) preparation of porous MoS2 by sol-gel method[74], Copyright 2012, Elsevier B.V.; (C) Laser thinning diagram[75], Copyright 2012, American Chemical Society.
| Synthesis method | Advantage | Drawback |
|---|---|---|
| Exfoliation method | High yield and simple operation | Uncontrollable product morphology and layer number |
| CVD | Good crystallinity, controllable number of layers | Low yield, high cost, high reaction temperature |
| Hydro/Solvothermal method | Large number of active sites, low cost | Poor crystallinity |
| Electrospinning | Low cost, forming soft flexible thin film | Low efficiency |
| Sol?gel method | High purity, high porosity | Longer period, high cost |
| MBE and ALD | Easy and precise control | Slow growth rate, high equipment cost and requirement |
Table 1 Preparation methods of 2D TMDs
| Synthesis method | Advantage | Drawback |
|---|---|---|
| Exfoliation method | High yield and simple operation | Uncontrollable product morphology and layer number |
| CVD | Good crystallinity, controllable number of layers | Low yield, high cost, high reaction temperature |
| Hydro/Solvothermal method | Large number of active sites, low cost | Poor crystallinity |
| Electrospinning | Low cost, forming soft flexible thin film | Low efficiency |
| Sol?gel method | High purity, high porosity | Longer period, high cost |
| MBE and ALD | Easy and precise control | Slow growth rate, high equipment cost and requirement |
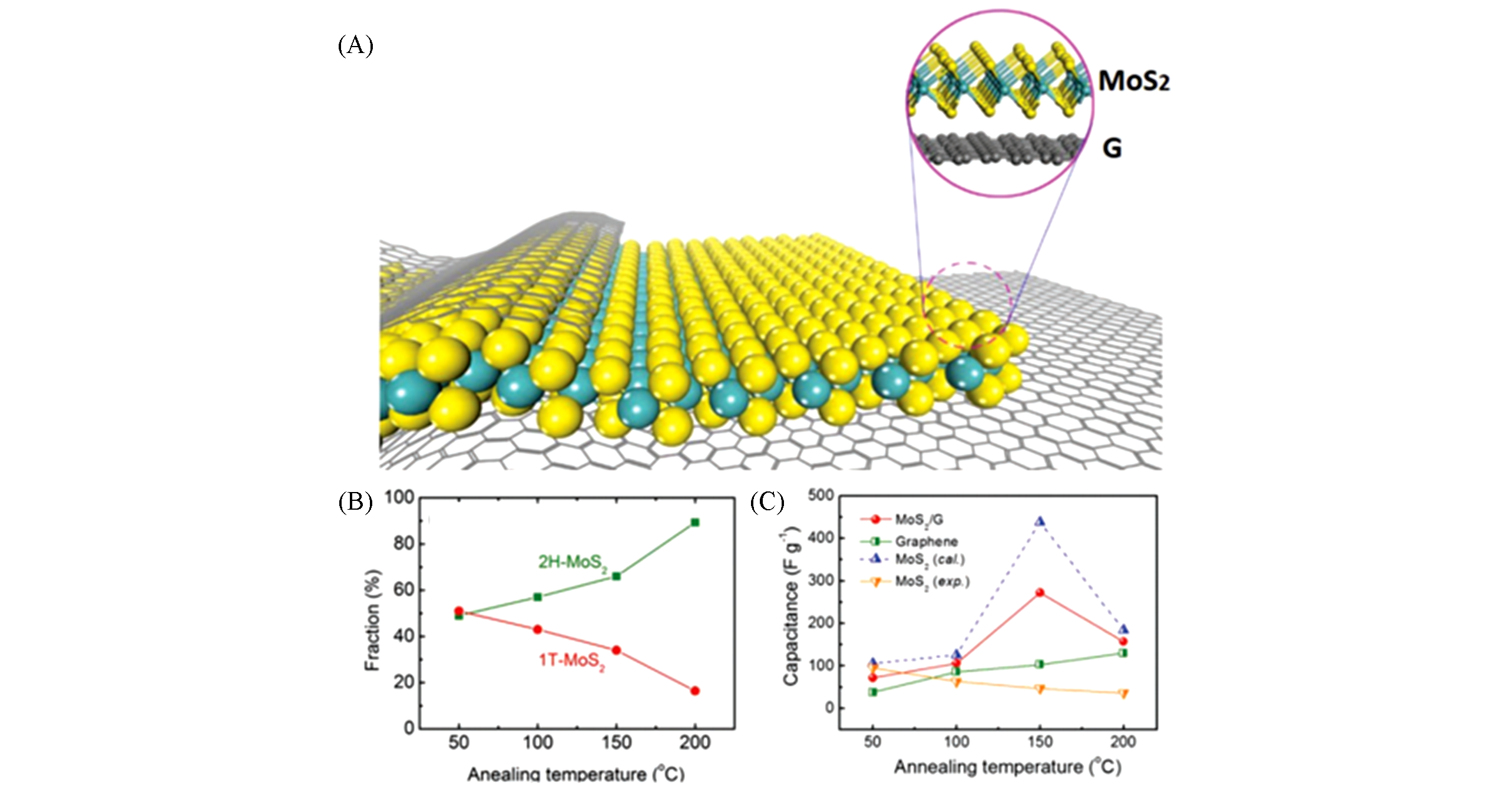
Fig.6 Schematic atomic model of the MoS2/G nanohybrid(A), relative fractions of 2H and 1T components as a function of annealing temperatures(B) and the experimental capacitance of graphene film(C)[92]MoS2/G, MoS2 film and calculated capacitance of MoS2 as a function of annealing temperatures are summarized.Copyright 2019, Wiley.
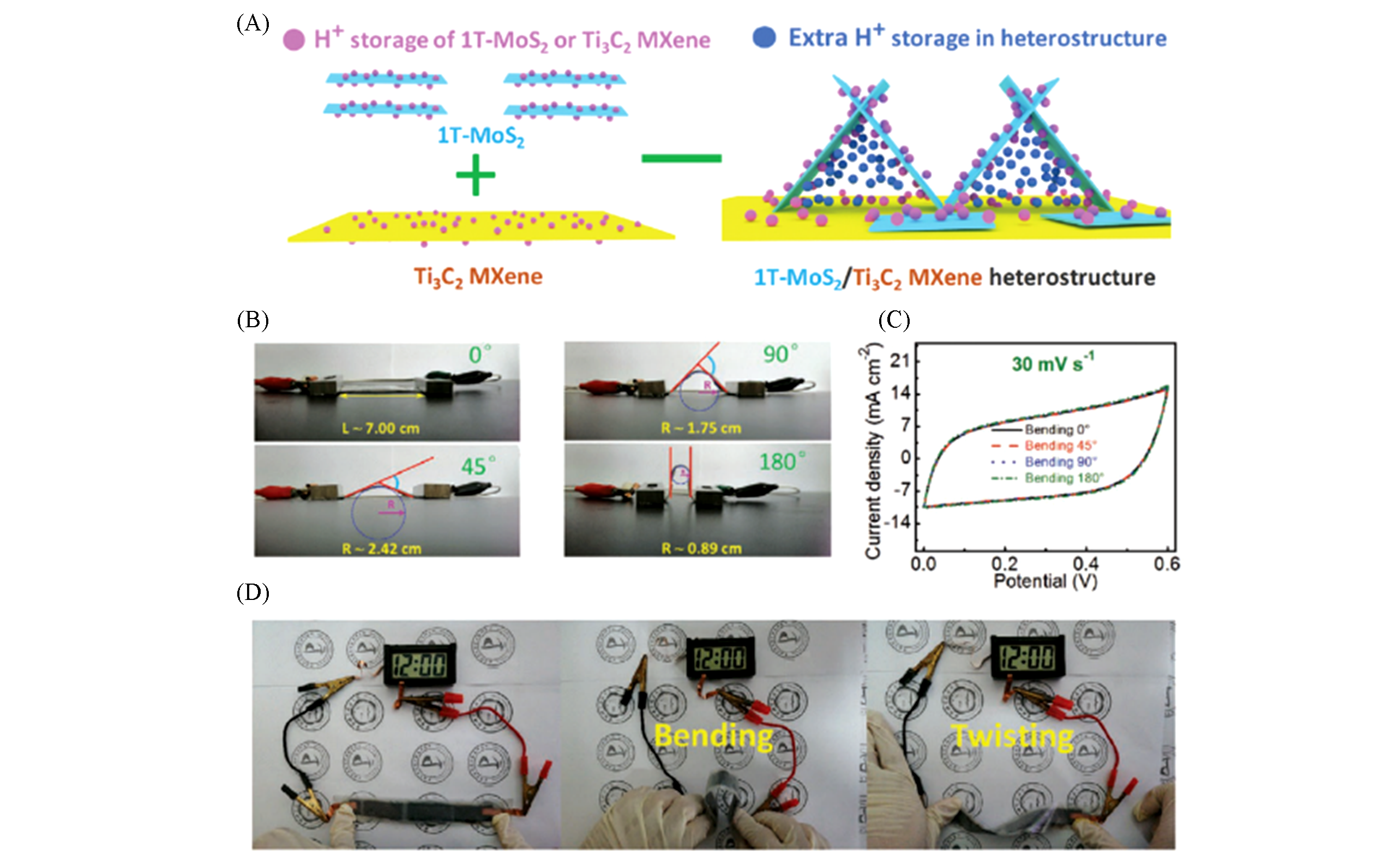
Fig.7 H+ ion storage of 1T?MoS2 or Ti3C2 MXene and extra H+ ion storage in 1T-MoS2/Ti3C2 MXene heterostucture at charged?discharged state(A), digital photographs and values of L and R of flexible 1T?MoS2/Ti3C2 MXene based flexible all?solid?state supercapacitor(ASSS) bended at different angles(B), CV curves of the ASSS device bended at different angles at 30 mV/s(C) and photographs of the three ASSS devices connected in series driving electronic watch(D)[93]Copyright 2020, Wiley-VCH.
| Electrode material | Electrolyte | Capacitance | Cycle performance | Ref. |
|---|---|---|---|---|
| Flower shape MoS2 | 1 mol/L Na2SO4 | 92.8 F/g(0.5 mA/cm2) | 93.8%(1000 cycles) | [ |
| MoS2 film | 0.5 mol/L H2SO4 | ca. 330 F/cm3(25.47 mA/cm2) | 97%(5000 cycles) | [ |
| MoS2/rGO | 1 mol/L Na2SO4 | 250%(10000 cycles) | [ | |
| MoS2/rGO@PANI | 1 mol/L H2SO4 | 1224 F/g(1 A/g) | 82.5%(3000 cycles) | [ |
| CoS@MoS2 Nanowires | 2 mol/L KOH | 1687.7 F/g(4 mA/cm2) | 96.2%(10000 cycles) | [ |
| 1T?MoS2/PVAK+ | 3 mol/L KCl | 448 F/g(1 A/g) | 96%(16000 cycles) | [ |
| 1T?2H MoS2/rGO | 1 mol/L H2SO4 | 416 F/g(1 A/g) | 50000 cycles | [ |
| MoS2/Graphene | 6 mol/L KOH | 272 F/g | [ | |
| 1T?MoS2/Ti3C2 | 1 mol/L H2SO4 | 386.7 F/g(1 A/g) | 96.8%(20000 cycles) | [ |
| MoSe2/MoS2 | 0.5 mol/L H2SO4 | 1229.6 F/g(1 A/g) | 92.8%(2000 cycles) | [ |
| MoS2/CNT | 1 mol/L Na2SO4 | 337 mF/cm2 | 97.6%(2500 cycles) | [ |
| MoSe2/Ni foam | 6 mol/L KOH | 1114.1 F/g(1 A/g) | 104.7%(1500 cycles) | [ |
| Flake MoSe2 | 1 mol/L Na2SO4 | 1467.8 F/g(4 mA/cm2) | 80%(1000 cycles) | [ |
| MoSe2/rGO | 6 mol/L KOH | 1422 F/g(1 A/g) | 100.7%(1500 cycles) | [ |
| MoSe2/Acetylene black | 6 mol/L KOH | 2020 F/g(1 A/g) | 107.5%(1500 cycles) | [ |
| 1T’?MoSe2@rGO | — | 1791 F/g(1 A/g) | 98.6%(2000 cycles) | [ |
Table 2 Molybdenum-based TMDs supercapacitor performance comparison
| Electrode material | Electrolyte | Capacitance | Cycle performance | Ref. |
|---|---|---|---|---|
| Flower shape MoS2 | 1 mol/L Na2SO4 | 92.8 F/g(0.5 mA/cm2) | 93.8%(1000 cycles) | [ |
| MoS2 film | 0.5 mol/L H2SO4 | ca. 330 F/cm3(25.47 mA/cm2) | 97%(5000 cycles) | [ |
| MoS2/rGO | 1 mol/L Na2SO4 | 250%(10000 cycles) | [ | |
| MoS2/rGO@PANI | 1 mol/L H2SO4 | 1224 F/g(1 A/g) | 82.5%(3000 cycles) | [ |
| CoS@MoS2 Nanowires | 2 mol/L KOH | 1687.7 F/g(4 mA/cm2) | 96.2%(10000 cycles) | [ |
| 1T?MoS2/PVAK+ | 3 mol/L KCl | 448 F/g(1 A/g) | 96%(16000 cycles) | [ |
| 1T?2H MoS2/rGO | 1 mol/L H2SO4 | 416 F/g(1 A/g) | 50000 cycles | [ |
| MoS2/Graphene | 6 mol/L KOH | 272 F/g | [ | |
| 1T?MoS2/Ti3C2 | 1 mol/L H2SO4 | 386.7 F/g(1 A/g) | 96.8%(20000 cycles) | [ |
| MoSe2/MoS2 | 0.5 mol/L H2SO4 | 1229.6 F/g(1 A/g) | 92.8%(2000 cycles) | [ |
| MoS2/CNT | 1 mol/L Na2SO4 | 337 mF/cm2 | 97.6%(2500 cycles) | [ |
| MoSe2/Ni foam | 6 mol/L KOH | 1114.1 F/g(1 A/g) | 104.7%(1500 cycles) | [ |
| Flake MoSe2 | 1 mol/L Na2SO4 | 1467.8 F/g(4 mA/cm2) | 80%(1000 cycles) | [ |
| MoSe2/rGO | 6 mol/L KOH | 1422 F/g(1 A/g) | 100.7%(1500 cycles) | [ |
| MoSe2/Acetylene black | 6 mol/L KOH | 2020 F/g(1 A/g) | 107.5%(1500 cycles) | [ |
| 1T’?MoSe2@rGO | — | 1791 F/g(1 A/g) | 98.6%(2000 cycles) | [ |
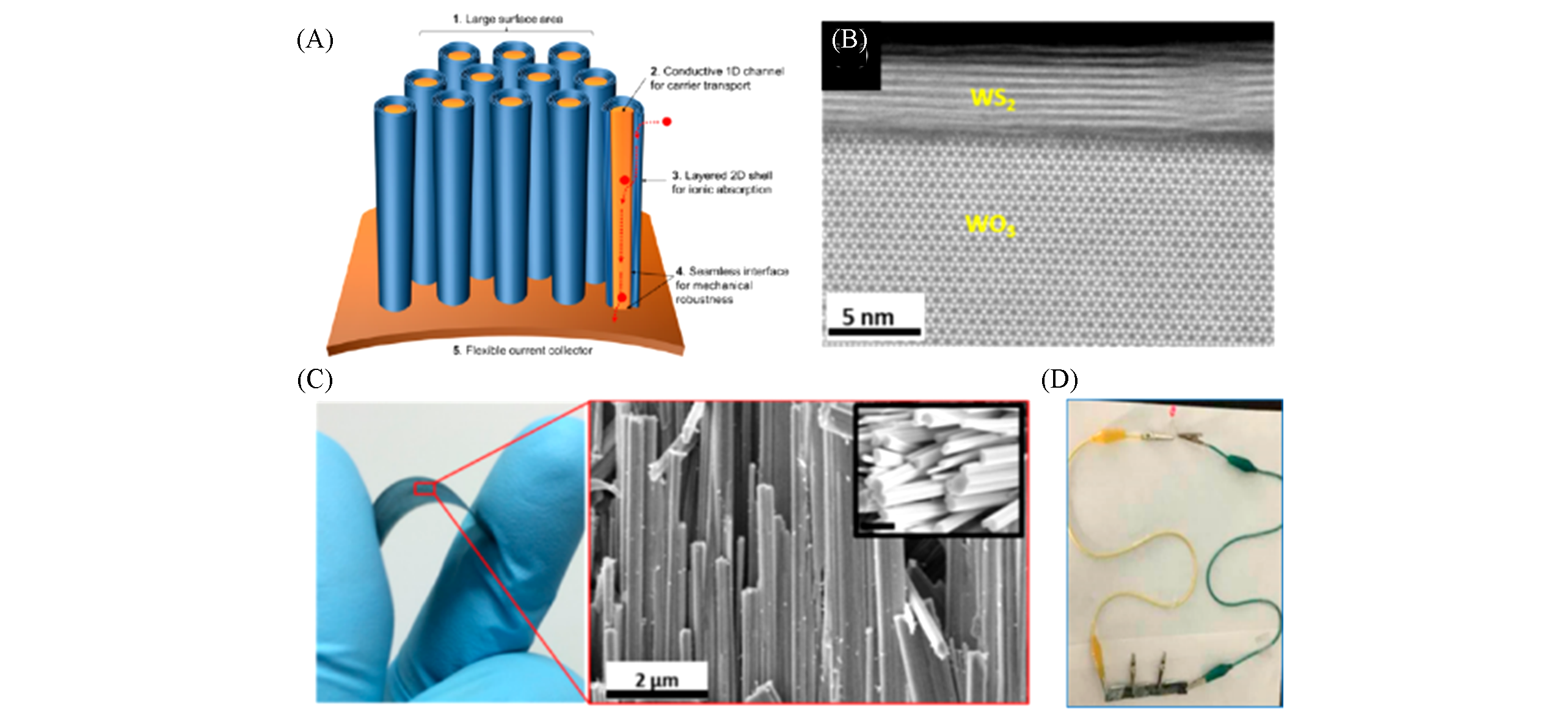
Fig.8 One?body array of core/shell nanowire supercapacitor and electrochemical performance[102](A) Schematic illustration for one-body array of core/shell nanowire supercapacitor; (B) TEM image of cross-section h-WO3/WS2 core/shell nanowire; (C) optical image of as-prepared core/shell nanowires on a W foil under mechanical bending(left), corresponding SEM image(right) shows high-density; (D) demonstration of powering a LED.Copyright 2016, American Chemical Society.
| Electrode material | Electrolyte | Capacitance | Cycle performance | Ref. |
|---|---|---|---|---|
| WS2/rGO | 1 mol/L Na2SO4 | 350 F/g(0.5 A/g) | 1000 cycles | [ |
| h?WO3/WS2 | 0.1 mol/L Na2SO4 | 47.5 mF/cm2(0.5 mA/cm2) | 30000 cycles | [ |
| WS2/Co3S4 | 1 mol/L H2SO4 | 412.7 F/g(1 A/g) | 94.3%(2000 cycles) | [ |
| WS2/PEDOT | 1 mol/L Na2SO4 | 70.64 F/g(0.2 A/g) | 90.74%(5000 cycles) | [ |
| WS2/rGO | 1 mol/L H2SO4 | 383.6 F/g(0.5 A/g) | 102.5%(10000 cycles) | [ |
| WS2 Quantum dots | PVA?H3PO4 | 28 mF/cm2(0.1 mA/cm2) | 80%(10000 cycles) | [ |
| WS2?MWCNTs/PANI | 1 mol/L H2SO4 | ca. 760.1 F/g(1 A/g) | ca. 80.2%(2000 cycles) | [ |
| WS2@NiCo2O4 | 3 mol/L KOH | 2449.9 mF/cm2(1 mA/cm) | [ | |
| WSe2 | 1 mol/L H2SO4 | 43.6 F/g(0.2 A/g) | 99%(20000 cycles) | [ |
| WTe2 | PVA?H3PO4 | 221 F/g(1 A/g) | 91%(5500 cycles) | [ |
| WSe2/rGO | 3 mol/L KOH | 389 F/g(1 A/g) | 98.7%(3000 cycles) | [ |
Table 3 Tungsten-based TMDs supercapacitor performance comparison
| Electrode material | Electrolyte | Capacitance | Cycle performance | Ref. |
|---|---|---|---|---|
| WS2/rGO | 1 mol/L Na2SO4 | 350 F/g(0.5 A/g) | 1000 cycles | [ |
| h?WO3/WS2 | 0.1 mol/L Na2SO4 | 47.5 mF/cm2(0.5 mA/cm2) | 30000 cycles | [ |
| WS2/Co3S4 | 1 mol/L H2SO4 | 412.7 F/g(1 A/g) | 94.3%(2000 cycles) | [ |
| WS2/PEDOT | 1 mol/L Na2SO4 | 70.64 F/g(0.2 A/g) | 90.74%(5000 cycles) | [ |
| WS2/rGO | 1 mol/L H2SO4 | 383.6 F/g(0.5 A/g) | 102.5%(10000 cycles) | [ |
| WS2 Quantum dots | PVA?H3PO4 | 28 mF/cm2(0.1 mA/cm2) | 80%(10000 cycles) | [ |
| WS2?MWCNTs/PANI | 1 mol/L H2SO4 | ca. 760.1 F/g(1 A/g) | ca. 80.2%(2000 cycles) | [ |
| WS2@NiCo2O4 | 3 mol/L KOH | 2449.9 mF/cm2(1 mA/cm) | [ | |
| WSe2 | 1 mol/L H2SO4 | 43.6 F/g(0.2 A/g) | 99%(20000 cycles) | [ |
| WTe2 | PVA?H3PO4 | 221 F/g(1 A/g) | 91%(5500 cycles) | [ |
| WSe2/rGO | 3 mol/L KOH | 389 F/g(1 A/g) | 98.7%(3000 cycles) | [ |
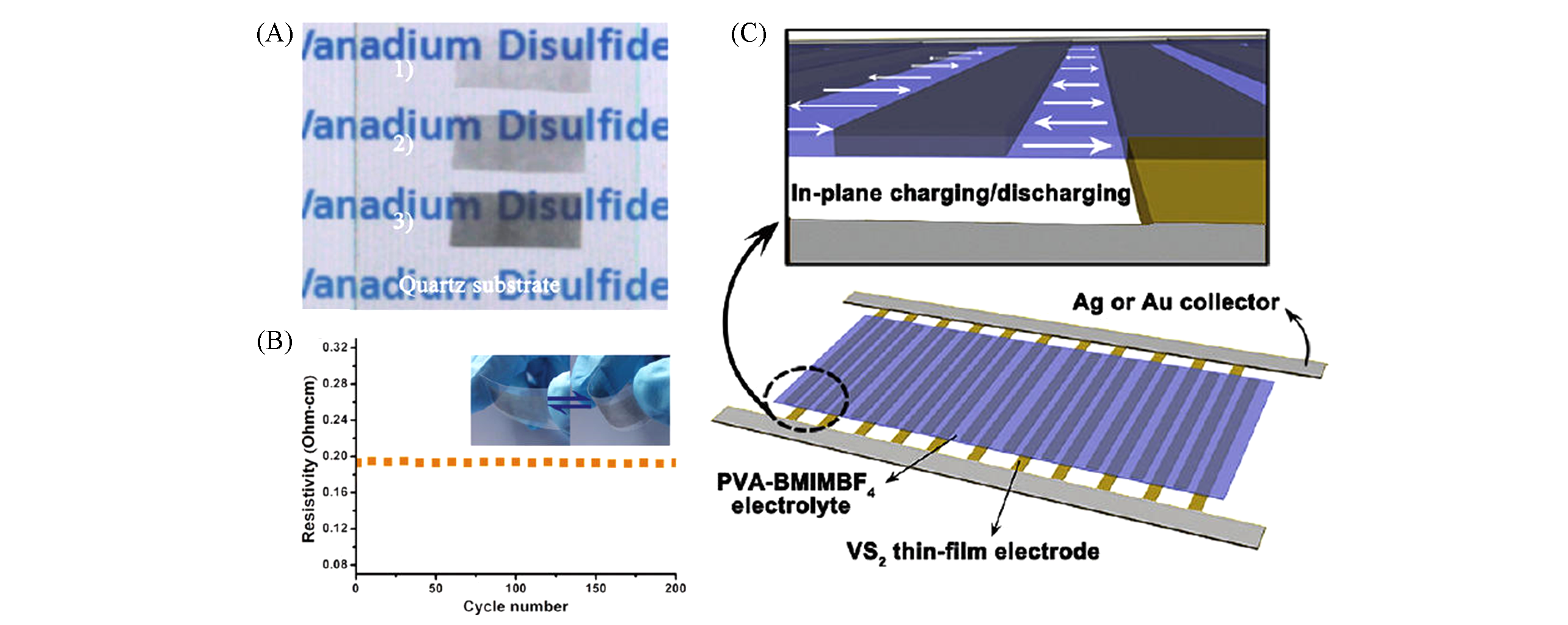
Fig.9 VS2 ultra?thin nanosheets for high two?dimensional conductivity of planar supercapacitors[112](A) VS2 thin films of different thicknesses are transferred to quartz substrate; (B) temperature dependence of planar resistivity of VS2 thin film; (C) planar ion migration pathways for the in-plane supercapacitor and schematic illustration of the in-plane configuration of the as-fabricated supercapacitor. Copyright 2011, American Chemical Society.
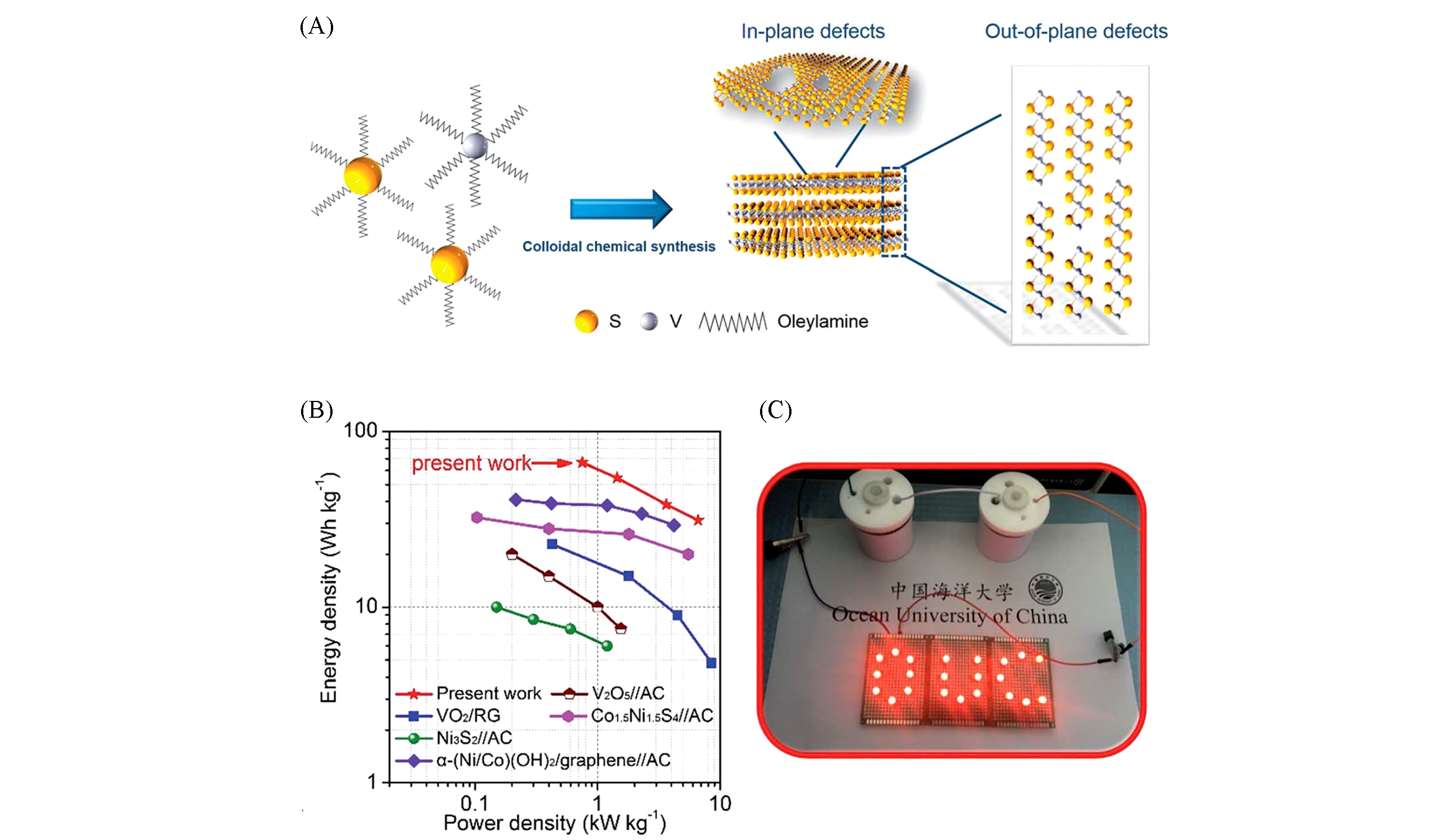
Fig.10 Synthesis and electrochemical performance of defect?rich VS2 nanoplates[115](A) Schematic illustration of the formation process of rich-defect VS2 nanoplate; (B) comparison of the energy density vs. power density curves of the full cell and other works; (C) two asymmetric supercapacitors were connected in series to light an OUC light-emitting diode(LED) panel. Copyright 2018, Royal Society of Chemistry.
| Electrode material | Electrolyte | Capacitance | Cycle performance | Ref. |
|---|---|---|---|---|
| VS2 | PVA?BMIMBF4 | 4760 μF/cm2 | >90%(1000 cycles) | [ |
| VS2/MWCNTs | 2 mol/L KCl | 830 F/g | 95.9%(10000 cycles) | [ |
| VS2/C | PVA/H2SO4 | 86.4 F/cm3(0.1 mA/cm2) | 97.7%(10000 cycles) | [ |
| VS2 | 1 mol/L KOH | 2200 F/g(1 A/g) | [ | |
| NiCo2S4@VS2 | 3 mol/L KOH | 1023.4 C/g(0.45 A/g) | 96%(2000 cycles) | [ |
| VS2/ZnO | 1 mol/L KOH | 2695.7 F/g(1 A/g) | 92.7%(5000 cycles) | [ |
| VSe2/rGO | 680 F/g(1 A/g) | ca. 81%(10000 cycles) | [ | |
| VSe2/rGO | 2 mol/L KOH | 1129 F/g(1 A/g) | 89.76%(2000 cycles) | [ |
Table 4 Vanadium-based TMDs supercapacitor performance comparison
| Electrode material | Electrolyte | Capacitance | Cycle performance | Ref. |
|---|---|---|---|---|
| VS2 | PVA?BMIMBF4 | 4760 μF/cm2 | >90%(1000 cycles) | [ |
| VS2/MWCNTs | 2 mol/L KCl | 830 F/g | 95.9%(10000 cycles) | [ |
| VS2/C | PVA/H2SO4 | 86.4 F/cm3(0.1 mA/cm2) | 97.7%(10000 cycles) | [ |
| VS2 | 1 mol/L KOH | 2200 F/g(1 A/g) | [ | |
| NiCo2S4@VS2 | 3 mol/L KOH | 1023.4 C/g(0.45 A/g) | 96%(2000 cycles) | [ |
| VS2/ZnO | 1 mol/L KOH | 2695.7 F/g(1 A/g) | 92.7%(5000 cycles) | [ |
| VSe2/rGO | 680 F/g(1 A/g) | ca. 81%(10000 cycles) | [ | |
| VSe2/rGO | 2 mol/L KOH | 1129 F/g(1 A/g) | 89.76%(2000 cycles) | [ |
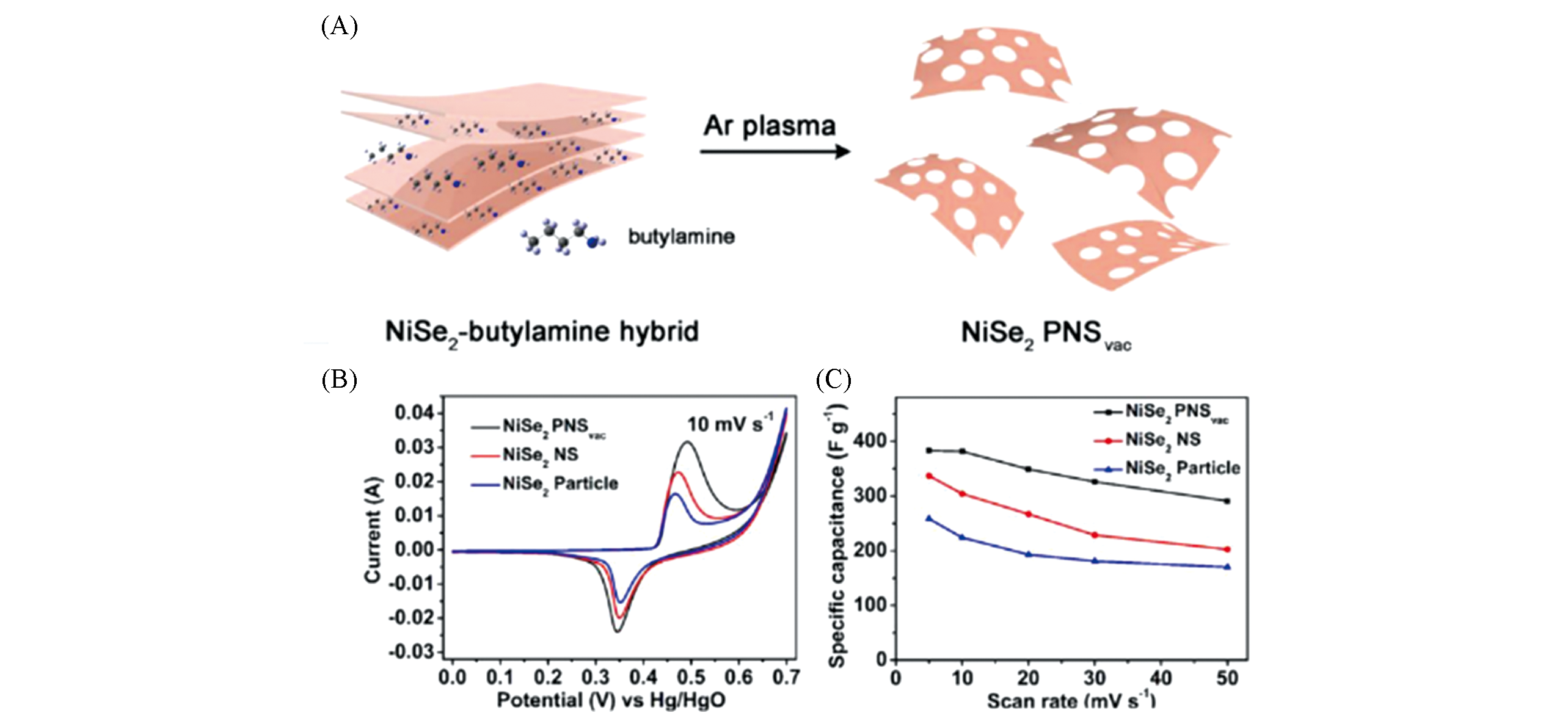
Fig.11 Preparation and electrochemical performance of NiSe2 PNSvac[123](A) Schematic procedure for the preparation of NiSe2 PNSvac; (B) CV curves of NiSe2 PNSvac, NiSe2 NS, and NiSe2 particle at a scan rate of 10 mV/s; (C) the specific capacitance of NiSe2 PNSvac, NiSe2 NS, and NiSe2 particle at different current density. Copyright 2018, American Chemical Society.
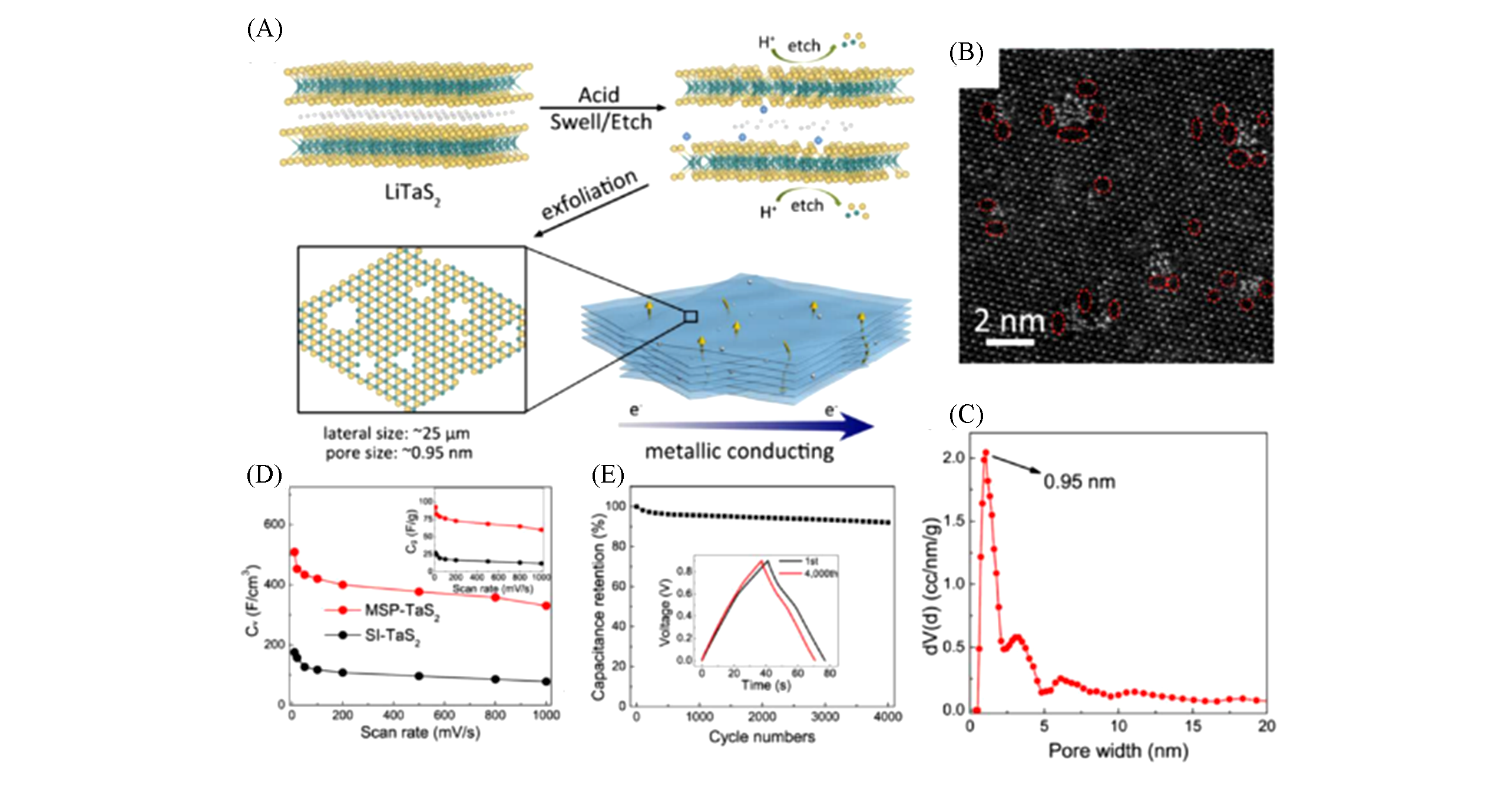
Fig.12 Preparation and electrochemical performance of acid?assisted exfoliation of TaS2 monolayer[127](A) By acid-assisted exfoliation, LiTaS2 single crystal swelled with TaS2 layer etched, achieving conductive TaS2 monolayers with large lateral size and sub-nanopore structure; (B) HAADF image of the corresponding TaS2 monolayer; (C) pore size distribution of TaS2 film by density functional theory(DFT) mode; (D) volumetric capacitance of MSP-TaS2 and SI-TaS2 based MSC as a function of scan rates; (E) cycling stability of MSP-TaS2 based MSC measured at 2 A/g. Inset: (D) the corresponding gravimetric capacitance as a function of scan rates; (E) comparison of GCD curves between the 1st cycle and the 4000th cycle for MSP-TaS2 based MSC. Copyright 2017, American Chemical Society.
| Electrode material | Electrolyte | Capacitance | Cycle performance | Ref. |
|---|---|---|---|---|
| NiSe2 | 4 mol/L KOH | 1044 F/g(3 A/g) | 67%(2000 cycles) | [ |
| NiSe2/CFC | 3 mol/L KOH | 1058 F/g(2 A/g) | [ | |
| NiSe2 PNSvac | 1 mol/L KOH | 466 F/g(3 A/g) | 81.3%(1000 cycles) | [ |
| Ni3Se2 | 3 mol/L KOH | 635 μA·h/cm2(3 mA/cm2) | [ | |
| TiS2/VACNT | 21 mol/L LiTFSI | 195 F/g | ca. 95%(10000 cycles) | [ |
| TaS2/PPy | 1 mol/L H2SO4 | 835 F/cm2 | 98.7%(10000 cycles) | [ |
| CoSe2 | 3 mol/L KOH | 544.6 F/g(1 mA/cm2) | 93.3%(5000 cycles) | [ |
| CoSe2 | 3 mol/L KOH | 759.5 F/g(1 mA/cm2) | 94.5%(5000 cycles) | [ |
| NiGa2S4 | 6 mol/L KOH | 2225 F/g(2 A/g) | 78.9%(6000 cycles) | [ |
Table 5 Performance comparison of other TMDs supercapacitors
| Electrode material | Electrolyte | Capacitance | Cycle performance | Ref. |
|---|---|---|---|---|
| NiSe2 | 4 mol/L KOH | 1044 F/g(3 A/g) | 67%(2000 cycles) | [ |
| NiSe2/CFC | 3 mol/L KOH | 1058 F/g(2 A/g) | [ | |
| NiSe2 PNSvac | 1 mol/L KOH | 466 F/g(3 A/g) | 81.3%(1000 cycles) | [ |
| Ni3Se2 | 3 mol/L KOH | 635 μA·h/cm2(3 mA/cm2) | [ | |
| TiS2/VACNT | 21 mol/L LiTFSI | 195 F/g | ca. 95%(10000 cycles) | [ |
| TaS2/PPy | 1 mol/L H2SO4 | 835 F/cm2 | 98.7%(10000 cycles) | [ |
| CoSe2 | 3 mol/L KOH | 544.6 F/g(1 mA/cm2) | 93.3%(5000 cycles) | [ |
| CoSe2 | 3 mol/L KOH | 759.5 F/g(1 mA/cm2) | 94.5%(5000 cycles) | [ |
| NiGa2S4 | 6 mol/L KOH | 2225 F/g(2 A/g) | 78.9%(6000 cycles) | [ |
| 1 | Liu C., Li F., Ma L. P., Cheng H. M., Adv. Mater., 2010, 22(8), E28—E62 |
| 2 | Chu S., Majumdar A., Nature, 2012, 488(7411), 294—303 |
| 3 | Chen M., Xia X., Yin J., Chen Q., Electrochim. Acta, 2015, 160, 15—21 |
| 4 | He X., Zhang H., Zhao X., Zhang P., Chen M., Zheng Z., Han Z., Zhu T., Tong Y., Lu X., Adv. Sci., 2019, 6(14), 1900151 |
| 5 | Zhang Y., Xia X., Liu B., Deng S., Xie D., Liu Q., Wang Y., Wu J., Wang X., Tu J., Adv. Energy Mater., 2019, 9(8), 1803342 |
| 6 | Manthiram A., Nat. Commun., 2020, 11(1), 1—9 |
| 7 | Chen Z., Chao D., Chen M., Shen Z., RSC Adv., 2020, 10(32), 18776—18783 |
| 8 | Chen M., Liang X., Wang F., Xie D., Pan G., Xia X., J. Mater. Chem. A, 2019, 7(12), 6644—6650 |
| 9 | Wang G., Zhang L., Zhang J., Chem. Soc. Rev., 2012, 41(2), 797—828 |
| 10 | Wang Y., Song Y., Xia Y., Chem. Soc. Rev., 2016, 45(21), 5925—5950 |
| 11 | Zhao X., Sánchez B. M., Dobson P. J., Grant P. S., Nanoscale, 2011, 3(3), 839—855 |
| 12 | Chen M., Zhou W., Qi M., Yin J., Xia X., Chen Q., J. Power Sources, 2017, 342, 964—969 |
| 13 | Han Y., Lu Y., Shen S., Zhong Y., Liu S., Xia X., Tong Y., Lu X., Adv. Funct. Mater., 2019, 29(7), 1806329 |
| 14 | Xia X., Zhang Y., Chao D., Xiong Q., Fan Z., Tong X., Tu J., Zhang H., Fan H. J., Energy Environ. Sci., 2015, 8(5), 1559—1568 |
| 15 | Zhang X., Deng S., Zeng Y., Yu M., Zhong Y., Xia X., Tong Y., Lu X., Adv. Funct. Mater., 2018, 28(44), 1805618 |
| 16 | Xia X., Tu J., Mai Y., Chen R., Wang X., Gu C., Zhao X., Chem. Eur. J., 2011, 17(39), 10898—10905 |
| 17 | Zhang Y., Li L., Su H., Huang W., Dong X., J. Mater. Chem. A, 2015, 3(1), 43—59 |
| 18 | Fong K. D., Wang T., Kim H. K., Kumar R. V., Smoukov S. K., ACS Energy Lett., 2017, 2(9), 2014—2020 |
| 19 | Wang Y., Ding Y., Guo X., Yu G., Nano Res., 2019, 1—10 |
| 20 | Cui H., Zhu G., Liu X., Liu F., Xie Y., Yang C., Lin T., Gu H., Huang F., Adv. Sci., 2015, 2(12), 1500126 |
| 21 | Mukherjee S., Ren Z., Singh G., Nano⁃Micro Lett., 2018, 10(4), 70 |
| 22 | Yan Y., Li B., Guo W., Pang H., Xue H., J. Power Sources, 2016, 329, 148—169 |
| 23 | Chen M., Zhang J., Xia X., Qi M., Yin J., Chen Q., Mater. Res. Bull., 2016, 76, 113—117 |
| 24 | Sheng L., Jiang H., Liu S., Chen M., Wei T., Fan Z., J. Power Sources, 2018, 397, 325—333 |
| 25 | Guo Q., Chen N., Qu L., Carbon Energy, 2020, 2(1), 54—71 |
| 26 | Hasani A., Teklagne M. A., Do H. H., Hong S. H., van Le Q., Ahn S. H., Kim S. Y., Carbon Energy, 2020, 2(2), 158—175 |
| 27 | Liu F., Wang C., Sui X., Riaz M. A., Xu M., Wei L., Chen Y., Carbon Energy, 2019, 1(2), 173—199 |
| 28 | Choi S., Kim C., Suh J. M., Jang H. W., Carbon Energy, 2019, 1(1), 85—108 |
| 29 | Chen M., Yin J., Jin R., Yao L., Su B., Lei Q., Thin Solid Films, 2015, 584, 232—237 |
| 30 | Arvas M. B., Gençten M., Sahin Y., Int. J. Energy Res., 2020, 44(3), 1624—1635 |
| 31 | Wang M., Yang J., Liu S., Hu C., Li S., Qiu J., ACS Appl. Mater. Interfaces, 2019, 11(29), 26235—26242 |
| 32 | Chen M., Liu J., Chao D., Wang J., Yin J., Lin J., Fan H. J., Shen Z. X., Nano Energy, 2014, 9, 364—372 |
| 33 | Anasori B., Lukatskaya M. R., Gogotsi Y., Nat. Rev. Mater., 2017, 2(2), 1—17 |
| 34 | Wu X., Wang Z., Yu M., Xiu L., Qiu J., Adv. Mater., 2017, 29(24), 1607017 |
| 35 | Wen Y., Rufford T. E., Chen X., Li N., Lyu M., Dai L., Wang L., Nano Energy, 2017, 38, 368—376 |
| 36 | Chen T. A., Chuu C. P., Tseng C. C., Wen C. K., Wong H. S. P., Pan S., Li R., Chao T. A., Chueh W. C., Zhang Y., Nature, 2020, 579(7798), 219—223 |
| 37 | Watts M. C., Picco L., Russell⁃Pavier F. S., Cullen P. L., Miller T. S., Bartuś S. P., Payton O. D., Skipper N. T., Tileli V., Howard C. A., Nature, 2019, 568(7751), 216—220 |
| 38 | Chen M., Chao D., Liu J., Yan J., Zhang B., Huang Y., Lin J., Shen Z. X., Adv. Funct. Mater., 2017, 27(12), 1606232 |
| 39 | Sui Z. Y., Zhang P. Y., Xu M. Y., Liu Y. W., Wei Z. X., Han B. H., ACS Appl. Mater. Interfaces, 2017, 9(49), 43171—43178 |
| 40 | Li Y., Chen M., Liu B., Zhang Y., Liang X., Xia X., Adv. Energy Mater., 2020, 10(27), 2000927 |
| 41 | Manzeli S., Ovchinnikov D., Pasquier D., Yazyev O. V., Kis A., Nat. Rev. Mater., 2017, 2(8), 17033 |
| 42 | Jiao Y., Hafez A. M., Cao D., Mukhopadhyay A., Ma Y., Zhu H., Small, 2018, 14(36), 1800640 |
| 43 | Cui Y., Zhou Z., Li T., Wang K., Li J., Wei Z., Adv. Funct. Mater., 2019, 29(24), 1900040 |
| 44 | Liang X., Chen M., Zhu H., Zhu H., Cui X., Yan J., Chen Q., Xia X., Liu Q., J. Mater. Chem. A, 2020, 8(18), 9068—9076 |
| 45 | Chang K., Hai X., Pang H., Zhang H., Shi L., Liu G., Liu H., Zhao G., Li M., Ye J., Adv. Mater., 2016, 28(45), 10033—10041 |
| 46 | Wu J., Dai J., Shao Y., Cao M., Wu X., RSC Adv., 2016, 6(81), 77999—78007 |
| 47 | Huang T., Jiang Y., Shen G., Chen D., ChemSusChem, 2020, 13(6), 1093—1113 |
| 48 | Miremadi B. K., Morrison S. R., J. Catal., 1987, 103(2), 334—345 |
| 49 | Late D. J., ACS Appl. Mater. Interfaces, 2014, 6(2), 1158—1163 |
| 50 | Huang M., Zhou Y., Guo Y., Wang H., Hu X., Xu X., Ren Z., J. Mater. Sci., 2018, 53(10), 7744—7754 |
| 51 | Coleman J. N., Lotya M., O’Neill A., Bergin S. D., King P. J., Khan U., Young K., Gaucher A., De S., Smith R. J., Science, 2011, 331(6017), 568—571 |
| 52 | Yu X., Guijarro N., Johnson M., Sivula K., Nano Lett., 2018, 18(1), 215—222 |
| 53 | Yang D., Frindt R., J. Phys. Chem. Solids, 1996, 57(6—8), 1113—1116 |
| 54 | Fan X., Xu P., Zhou D., Sun Y., Li Y. C., Nguyen M. A. T., Terrones M., Mallouk T. E., Nano Lett., 2015, 15(9), 5956—5960 |
| 55 | Lee Y. H., Zhang X. Q., Zhang W., Chang M. T., Lin C. T., Chang K. D., Yu Y. C., Wang J. T. W., Chang C. S., Li L. J., Adv. Mater., 2012, 24(17), 2320—2325 |
| 56 | Yu H., Liao M., Zhao W., Liu G., Zhou X., Wei Z., Xu X., Liu K., Hu Z., Deng K., ACS Nano, 2017, 11(12), 12001—12007 |
| 57 | Wang X., Gong Y., Shi G., Chow W. L., Keyshar K., Ye G., Vajtai R., Lou J., Liu Z., Ringe E., ACS Nano, 2014, 8(5), 5125—5131 |
| 58 | Naylor C. H., Parkin W. M., Ping J., Gao Z., Zhou Y. R., Kim Y., Streller F., Carpick R. W., Rappe A. M., Drndić M., Nano Lett., 2016, 16(7), 4297—4304 |
| 59 | Zhang Y., Zhang Y., Ji Q., Ju J., Yuan H., Shi J., Gao T., Ma D., Liu M., Chen Y., ACS Nano, 2013, 7(10), 8963—8971 |
| 60 | Chen L., Liu B., Abbas A. N., Ma Y., Fang X., Liu Y., Zhou C., Acs Nano, 2014, 8(11), 11543—11551 |
| 61 | Cui Y., Li B., Li J., Wei Z., Sci. China⁃Phys. Mech. Astron., 2018, 61(1), 016801 |
| 62 | Cai Z., Liu B., Zou X., Cheng H. M., Chem. Rev., 2018, 118(13), 6091—6133 |
| 63 | Yang T., Zheng B., Wang Z., Xu T., Pan C., Zou J., Zhang X., Qi Z., Liu H., Feng Y., Nat. Commun., 2017, 8(1), 1—9 |
| 64 | Eichfeld S. M., Hossain L., Lin Y. C., Piasecki A. F., Kupp B., Birdwell A. G., Burke R. A., Lu N., Peng X., Li J., ACS Nano, 2015, 9(2), 2080—2087 |
| 65 | Manuraj M., Nair K. K., Unni K. N., Rakhi R., J. Alloys Compd., 2020, 819, 152963 |
| 66 | Zhao B., Wang Z., Gao Y., Chen L., Lu M., Jiao Z., Jiang Y., Ding Y., Cheng L., Appl. Surf. Sci., 2016, 390, 209—215 |
| 67 | Lu P., Wang X., Wen L., Jiang X., Guo W., Wang L., Yan X., Hou F., Liang J., Cheng H. M., Small, 2019, 15(15), 1805064 |
| 68 | Li Y., Wang H., Xie L., Liang Y., Hong G., Dai H., JACS, 2011, 133(19), 7296—7299 |
| 69 | Yi J., She X., Song Y., Mao M., Xia K., Xu Y., Mo Z., Wu J., Xu H., Li H., Chem. Eng. J., 2018, 335, 282—289 |
| 70 | Fu Q., Han J., Wang X., Xu P., Yao T., Zhong J., Zhong W., Liu S., Gao T., Zhang Z., Adv. Mater., 2020, 1907818 |
| 71 | Han G. H., Duong D. L., Keum D. H., Yun S. J., Lee Y. H., Chem. Rev., 2018, 118(13), 6297—6336 |
| 72 | Xue J., Xie J., Liu W., Xia Y., Acc. Chem. Res., 2017, 50(8), 1976—1987 |
| 73 | Kumuthini R., Ramachandran R., Therese H., Wang F., J. Alloys Compd., 2017, 705, 624—630 |
| 74 | Li N., Chai Y., Dong B., Liu B., Guo H., Liu C., Mater. Lett., 2012, 88, 112—115 |
| 75 | Castellanos⁃Gomez A., Barkelid M., Goossens A., Calado V. E., van der Zant H. S., Steele G. A., Nano Lett., 2012, 12(6), 3187—3192 |
| 76 | Zang X., Hohman J. N., Yao K., Ci P., Yan A., Wei M., Hayasaka T., Zettl A., Schuck P. J., Wu J., Adv. Funct. Mater., 2019, 29(27), 1807612 |
| 77 | Li H., Chen X., Zalnezhad E., Hui K., Hui K., Ko M. J., J. Ind. Eng. Chem., 2020, 82, 309—316 |
| 78 | Chen M., Fan H., Zhang Y., Liang X., Chen Q., Xia X., Small, 2020, 16(37), 2003434 |
| 79 | Lee C., Kim S. K., Choi J. H., Chang H., Jang H. D., J. Alloys Compd., 2018, 735, 2030—2037 |
| 80 | Zheng M., Xiao X., Li L., Gu P., Dai X., Tang H., Hu Q., Xue H., Pang H., Sci. China⁃Mater., 2018, 61(2), 185—209 |
| 81 | Krishnamoorthy K., Veerasubramani G. K., Radhakrishnan S., Kim S. J., Mater. Res. Bull., 2014, 50, 499—502 |
| 82 | Choudhary N., Patel M., Ho Y. H., Dahotre N. B., Choi W., J. Mater. Chem. A, 2015, 3(47), 24049—24054 |
| 83 | Bissett M. A., Kinloch I. A., Dryfe R. A., ACS Appl. Mater. Interfaces, 2015, 7(31), 17388—17398 |
| 84 | Da Silveira Firmiano E. G., Rabelo A. C., Dalmaschio C. J., Pinheiro A. N., Pereira E. C., Schreiner W. H., Leite E. R., Adv. Energy Mater., 2014, 4(6), 1301380 |
| 85 | Li X., Zhang C., Xin S., Yang Z., Li Y., Zhang D., Yao P., ACS Appl. Mater. Interfaces, 2016, 8(33), 21373—21380 |
| 86 | Dai J., Balamurugan J., Kim N. H., Lee J. H., J. Alloys Compd., 2020, 825, 154085 |
| 87 | Eda G., Yamaguchi H., Voiry D., Fujita T., Chen M., Chhowalla M., Nano Lett., 2011, 11(12), 5111—5116 |
| 88 | Toh R. J., Sofer Z., Luxa J., Sedmidubský D., Pumera M., Chem. Commun., 2017, 53(21), 3054—3057 |
| 89 | Acerce M., Voiry D., Chhowalla M., Nat. Nanotechnol., 2015, 10(4), 313—318 |
| 90 | Feng D., Pan X., Xia Q., Qin J., Zhang Y., Chen X., J. Mater. Sci., 2020, 55(2), 713—723 |
| 91 | Gigot A., Fontana M., Serrapede M., Castellino M., Bianco S., Armandi M., Bonelli B., Pirri C. F., Tresso E., Rivolo P., ACS Appl. Mater. Interfaces, 2016, 8(48), 32842—32852 |
| 92 | Ke Q., Zhang X., Zang W., Elshahawy A. M., Hu Y., He Q., Pennycook S. J., Cai Y., Wang J., Small, 2019, 15(21), 1900131 |
| 93 | Wang X., Li H., Li H., Lin S., Ding W., Zhu X., Sheng Z., Wang H., Zhu X., Sun Y., Adv. Funct. Mater., 2020, 30(15), 0190302 |
| 94 | Li S., Zang W., Liu X., Pennycook S. J., Kou Z., Yang C., Guan C., Wang J., Chem. Eng. J., 2019, 359, 1419—1426 |
| 95 | Tiwari P., Jaiswal J., Chandra R., Electrochim. Acta, 2019, 324, 134767 |
| 96 | Huang K. J., Zhang J. Z., Fan Y., Mater. Lett., 2015, 152, 244—247 |
| 97 | Mariappan V. K., Krishnamoorthy K., Pazhamalai P., Sahoo S., Kim S. J., Electrochim. Acta, 2018, 265, 514—522 |
| 98 | Huang K. J., Zhang J. Z., Cai J. L., Electrochim. Acta, 2015, 180, 770—777 |
| 99 | Liu X., Zhang J. Z., Huang K. J., Hao P., Chem. Eng. J., 2016, 302, 437—445 |
| 100 | Liu M., Wang X., Huang Z., Guo P., Wang Z., Mater. Lett., 2017, 206, 229—232 |
| 101 | Ratha S., Rout C. S., ACS Appl. Mater. Interfaces, 2013, 5(21), 11427—11433 |
| 102 | Choudhary N., Li C., Chung H. S., Moore J., Thomas J., Jung Y., ACS Nano, 2016, 10(12), 10726—10735 |
| 103 | Shrivastav V., Sundriyal S., Goel P., Shrivastav V., Tiwari U. K., Deep A., Electrochim. Acta, 2020, 345, 136194 |
| 104 | Habib M., Khalil A., Muhammad Z., Khan R., Wang C., ur Rehman Z., Masood H. T., Xu W., Liu H., Gan W., Electrochim. Acta, 2017, 258, 71—79 |
| 105 | Yu P., Fu W., Zeng Q., Lin J., Yan C., Lai Z., Tang B., Suenaga K., Zhang H., Liu Z., Adv. Mater., 2017, 29(34), 1701909 |
| 106 | Li Y., Niu J., Xue T., Duan X., Tian Q., Wen Y., Lu X., Xu J., Lai L., Chang Y., J. Electrochem. Soc., 2020, 167(4), 047512 |
| 107 | Chen W., Yu X., Zhao Z., Ji S., Feng L., Electrochim. Acta, 2019, 298, 313—320 |
| 108 | Ghorai A., Midya A., Ray S. K., New J. Chem., 2018, 42(5), 3609—3613 |
| 109 | Gao J., Ma Y., Li J., Fan J., Shi P., Xu Q., Min Y., J. Nanopart. Res., 2018, 20(11), 298 |
| 110 | Li L., Gao J., Cecen V., Fan J., Shi P., Xu Q., Min Y., ACS Omega, 2020, 5(9), 4657—4667 |
| 111 | Gopi C. V. M., Reddy A. E., Bak J. S., Cho I. H., Kim H. J., Mater. Lett., 2018, 223, 57—60 |
| 112 | Feng J., Sun X., Wu C., Peng L., Lin C., Hu S., Yang J., Xie Y., JACS, 2011, 133(44), 17832—17838 |
| 113 | Pandit B., Karade S. S., Sankapal B. R., ACS Appl. Mater. Interfaces, 2017, 9(51), 44880—44891 |
| 114 | Haider W. A., Tahir M., He L., Yang W., Minhas⁃khan A., Owusu K. A., Chen Y., Hong X., Mai L., J. Alloys Compd., 2020, 823, 151769 |
| 115 | Guo Z., Yang L., Wang W., Cao L., Dong B., J. Mater. Chem. A, 2018, 6(30), 14681—14688 |
| 116 | Marri S. R., Ratha S., Rout C. S., Behera J., Chem. Commun., 2017, 53(1), 228—231 |
| 117 | Wang Y., Sofer Z., Luxa J., Pumera M., Adv. Mater. Interfaces, 2016, 3(23), 1600433 |
| 118 | Zhang Z., Huang X., Wang H., Teo S. H., Ma T., J. Alloys Compd., 2019, 771, 274—280 |
| 119 | Fang L., Zhang Z., Li X., Zhou H., Ma K., Ge L., Huang K., Colloid Surf. A-Physicochem. Eng. Asp., 2016, 501, 42—48 |
| 120 | Wu X., Zhai Z. B., Huang K. J., Ren R. R., Wang F., J. Power Sources, 2020, 448, 227399 |
| 121 | Wang S., Li W., Xin L., Wu M., Long Y., Huang H., Lou X., Chem. Eng. J., 2017, 330, 1334—1341 |
| 122 | Bao Q., Wu J., Fan L., Ge J., Dong J., Jia J., Zeng J., Lin J., J. Energy Chem., 2017, 26(6), 1252—1259 |
| 123 | Chang A., Zhang C., Yu Y., Yu Y., Zhang B., ACS Appl. Mater. Interfaces, 2018, 10(49), 41861—41865 |
| 124 | Shi X., Wang H., Kannan P., Ding J., Ji S., Liu F., Gai H., Wang R., J. Mater. Chem. A, 2019, 7(7), 3344—3352 |
| 125 | Bissett M. A., Worrall S. D., Kinloch I. A., Dryfe R. A., Electrochim. Acta, 2016, 201, 30—37 |
| 126 | Zang X., Shen C., Kao E., Warren R., Zhang R., Teh K. S., Zhong J., Wei M., Li B., Chu Y., Adv. Mater., 2018, 30(5), 1704754 |
| 127 | Wu J., Peng J., Yu Z., Zhou Y., Guo Y., Li Z., Lin Y., Ruan K., Wu C., Xie Y., JACS, 2018, 140(1), 493—498 |
| 128 | Wang M., Zhang L., Zhong Y., Huang M., Zhen Z., Zhu H., Nanoscale, 2018, 10(36), 17341—17346 |
| 129 | Chen T., Li S., Gui P., Wen J., Fu X., Fang G., Nanotechnology, 2018, 29(20), 205401 |
| 130 | Chen T., Li S., Wen J., Gui P., Guo Y., Guan C., Liu J., Fang G., Small, 2018, 14(5), 1700979 |
| 131 | Liu S., Kim K. H., Yun J. M., Kundu A., Sankar K. V., Patil U. M., Ray C., Jun S. C., J. Mater. Chem. A, 2017, 5(13), 6292—6298 |
| [1] | 张义超, 赵付来, 王宇, 王亚玲, 沈永涛, 冯奕钰, 封伟. 基于多层二硒化钨的高性能场效应晶体管的实验优化和理论模拟[J]. 高等学校化学学报, 2022, 43(6): 20220113. |
| [2] | 侯从聪, 王惠颖, 李婷婷, 张志明, 常春蕊, 安立宝. N-CNTs/NiCo-LDH复合材料的制备及电化学性能[J]. 高等学校化学学报, 2022, 43(10): 20220351. |
| [3] | 魏雨晨, 武婷婷, 杨磊, 金碧玉, 李宏强, 何孝军. 萘基相互连接的多孔碳纳米囊的制备及超电容性能[J]. 高等学校化学学报, 2021, 42(9): 2852. |
| [4] | 辛伟闻, 闻利平. 二维材料用于渗透能转换的研究进展[J]. 高等学校化学学报, 2021, 42(2): 445. |
| [5] | 史江维, 孟楠楠, 郭亚梅, 于一夫, 张兵 . 二维材料用于电催化析氢的研究进展[J]. 高等学校化学学报, 2021, 42(2): 492. |
| [6] | 董其政, 翟锦. 基于二维材料的仿生纳流体通道在能量转化中的应用[J]. 高等学校化学学报, 2021, 42(2): 432. |
| [7] | 黄东雪, 章颖, 曾婷, 张媛媛, 万其进, 杨年俊. 基于过渡金属硫化物/还原氧化石墨烯复合物的高性能超级电容器[J]. 高等学校化学学报, 2021, 42(2): 643. |
| [8] | 沙卉雯, 马维廷, 周晓娟, 宋卫星. 激光诱导三维网状石墨烯的一步法制备及应用[J]. 高等学校化学学报, 2021, 42(2): 607. |
| [9] | 王伟, 卢香超, 周立军, 鲁艺珍, 曹阳. 基于二维压电材料功能性器件的设计、 构筑与性能研究[J]. 高等学校化学学报, 2021, 42(2): 595. |
| [10] | 林生晃, 傅年庆, 鲍桥梁. 单元素二维材料及其衍生物作为电荷传输层在太阳能电池中应用的研究进展[J]. 高等学校化学学报, 2021, 42(2): 412. |
| [11] | 陈晓煜, 于然波. 纳米二硫化钼的掺杂及催化电解水产氢的研究进展[J]. 高等学校化学学报, 2021, 42(2): 475. |
| [12] | 邓亚茜, 吴志坦, 吕伟, 陶莹, 杨全红. 二维材料的凝胶化及电化学储能应用[J]. 高等学校化学学报, 2021, 42(2): 380. |
| [13] | 余强敏, 张致远, 罗雨婷, 李洋, 成会明, 刘碧录. 金属性二维过渡金属硫化物的溶剂热合成及电催化析氢性能[J]. 高等学校化学学报, 2021, 42(2): 654. |
| [14] | 解忱, 陈娜, 杨雁冰, 袁荃. 核酸适体功能化的二维材料场效应晶体管传感器研究进展[J]. 高等学校化学学报, 2021, 42(11): 3406. |
| [15] | 张鑫, 赵付来, 王宇, 梁雪静, 冯奕钰, 封伟. 碲化锗场效应晶体管的制备及电学性能[J]. 高等学校化学学报, 2020, 41(9): 2032. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||