

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (2): 456.doi: 10.7503/cjcu20200551
收稿日期:2020-08-11
出版日期:2021-02-10
发布日期:2021-02-05
通讯作者:
黄小青
E-mail:hxq006@xmu.edu.cn
基金资助:
PI Yecan1,2, ZHANG Ying1, CHENG Zifang2, HUANG Xiaoqing1,2( )
)
Received:2020-08-11
Online:2021-02-10
Published:2021-02-05
Contact:
HUANG Xiaoqing
E-mail:hxq006@xmu.edu.cn
Supported by:摘要:
基于各种电化学过程的能源转化技术是未来可持续能源利用和发展的关键, 而催化剂在其中扮演着非常重要的角色. 二维金属纳米材料因其独特的物理化学性质在许多电催化反应中都展现出巨大的应用潜力, 也因此受到了广泛关注. 本文介绍了二维金属纳米材料的常见合成方法与策略, 并综合评述了近年来该类材料在电催化应用领域中的研究进展, 重点探讨了材料的组分和微观结构等因素对其性能的影响机理, 最后对二维金属纳米材料目前所面临的挑战以及未来的研究方向进行了总结与展望.
中图分类号:
TrendMD:
皮业灿, 张应, 成子方, 黄小青. 二维金属纳米材料的合成及电催化应用的研究进展. 高等学校化学学报, 2021, 42(2): 456.
PI Yecan, ZHANG Ying, CHENG Zifang, HUANG Xiaoqing. Progress in Synthesis and Electrocatalysis of Two-Dimensional Metal Nanomaterials. Chem. J. Chinese Universities, 2021, 42(2): 456.
| Synthetic method | Element | Information about structure & morphology | Ref. |
|---|---|---|---|
Ligand/small molecule?mediated synthesis | Rh | Nanosheets, ca.1.3 nm in thickness | [ |
| Rh | Single?crystalline parallelogram nanosheets, (0.9±0.4) nm in thickness | [ | |
| Co | Partially oxidized 4?atomic?layers | [ | |
| Ag | Triangular sheets | [ | |
| Pd | Hexagonal nanosheets, less than 10 atomic layers thick, tunable length of 20—160 nm | [ | |
| Pd?Pt?Ag | Alloy nanosheets, 3 nm in thickness | [ | |
| Template?directed synthesis | Au | Square nanosheets, hcp phase, (5.0±0.6) nm in thickness | [ |
| Pd | Single?crystalline square nanoplates, tunable thickness of 2.4—4.5 nm | [ | |
| Pt | Single crystalline nanosheet, 9.8 nm in thickness | [ | |
| Ag | Nanoplates on MoS2 substrate | [ | |
| Space?confined growth | Au | Nanosheets with thicknesses of several to tens of nanometers, large areas(>100 μm2) | [ |
| Au | Single?crystalline nanosheets, exceeding 20 μm in lateral size | [ | |
| Ag | Nanoplate with {111} facets exposed | [ | |
| Seeded growth | Pd@Pt | Monolayer Pt grown on Pd core?shell nanoplates, less than 5 nm thick | [ |
| Pd@Ru | Sub?monolayered Ru decorated?ultrathin Pd nanosheets, 1.9 nm in thickness | [ | |
| Ag@Ag | hcp/fcc square nanosheets, (2.8±0.5) nm in thickness | [ | |
| Exfoliation method | Sb | Nanosheets with thicknesses of 3—4 nm | [ |
| As, Sb, Bi | Nanosheets | [ | |
| Ru | Nanosheet, ca. 0.6 nm in thickness | [ | |
| Pt | Monolayer nanosheets | [ | |
| Mechanical compression method | Bi | Nanosheets with thickness of ca. 2 nm and area of more than several micrometers | [ |
| Pt, Au | Nanosheets, 5—10 nm in thickness | [ | |
| Ag | Nanosheets with thicknesses of ca.1 nm and sizes of the order of micrometers | [ | |
| Al | Nanosheets, ca.3 nm in thickness | [ |
Table 1 Summary of some representative 2D metal nanomaterials with corresponding synthetic methods
| Synthetic method | Element | Information about structure & morphology | Ref. |
|---|---|---|---|
Ligand/small molecule?mediated synthesis | Rh | Nanosheets, ca.1.3 nm in thickness | [ |
| Rh | Single?crystalline parallelogram nanosheets, (0.9±0.4) nm in thickness | [ | |
| Co | Partially oxidized 4?atomic?layers | [ | |
| Ag | Triangular sheets | [ | |
| Pd | Hexagonal nanosheets, less than 10 atomic layers thick, tunable length of 20—160 nm | [ | |
| Pd?Pt?Ag | Alloy nanosheets, 3 nm in thickness | [ | |
| Template?directed synthesis | Au | Square nanosheets, hcp phase, (5.0±0.6) nm in thickness | [ |
| Pd | Single?crystalline square nanoplates, tunable thickness of 2.4—4.5 nm | [ | |
| Pt | Single crystalline nanosheet, 9.8 nm in thickness | [ | |
| Ag | Nanoplates on MoS2 substrate | [ | |
| Space?confined growth | Au | Nanosheets with thicknesses of several to tens of nanometers, large areas(>100 μm2) | [ |
| Au | Single?crystalline nanosheets, exceeding 20 μm in lateral size | [ | |
| Ag | Nanoplate with {111} facets exposed | [ | |
| Seeded growth | Pd@Pt | Monolayer Pt grown on Pd core?shell nanoplates, less than 5 nm thick | [ |
| Pd@Ru | Sub?monolayered Ru decorated?ultrathin Pd nanosheets, 1.9 nm in thickness | [ | |
| Ag@Ag | hcp/fcc square nanosheets, (2.8±0.5) nm in thickness | [ | |
| Exfoliation method | Sb | Nanosheets with thicknesses of 3—4 nm | [ |
| As, Sb, Bi | Nanosheets | [ | |
| Ru | Nanosheet, ca. 0.6 nm in thickness | [ | |
| Pt | Monolayer nanosheets | [ | |
| Mechanical compression method | Bi | Nanosheets with thickness of ca. 2 nm and area of more than several micrometers | [ |
| Pt, Au | Nanosheets, 5—10 nm in thickness | [ | |
| Ag | Nanosheets with thicknesses of ca.1 nm and sizes of the order of micrometers | [ | |
| Al | Nanosheets, ca.3 nm in thickness | [ |
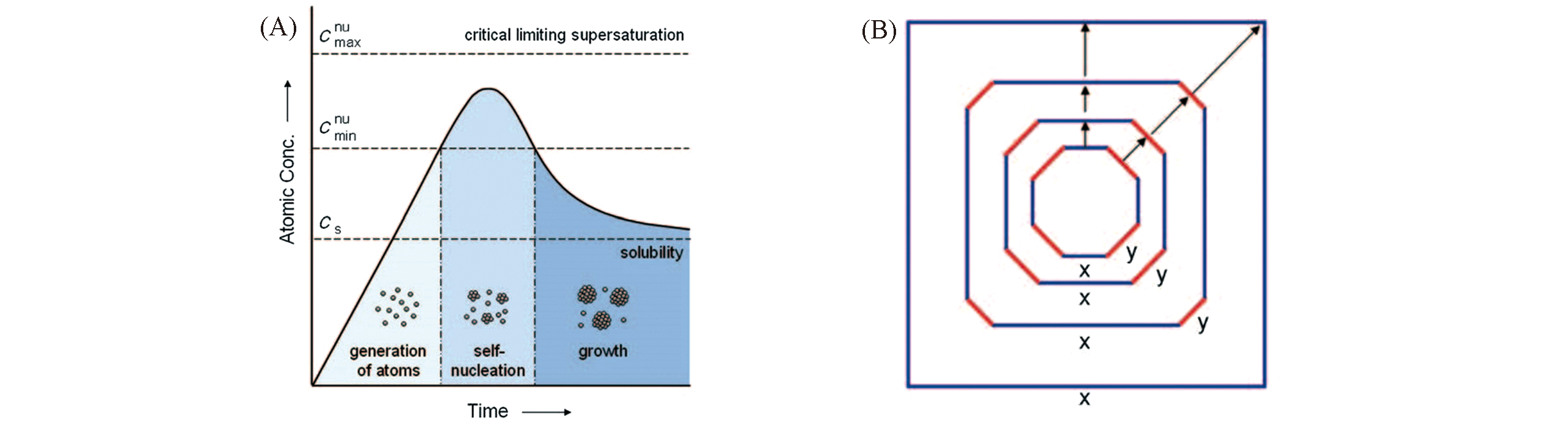
Fig.1 Plot of atomic concentration against time, illustrating the generation of atoms, nucleation and subsequent growth(A) and the shape evolution during the successive stages of growth for an imaginary 2D crystal(B)[80]Copyright 2009, Wiley‐VCH.
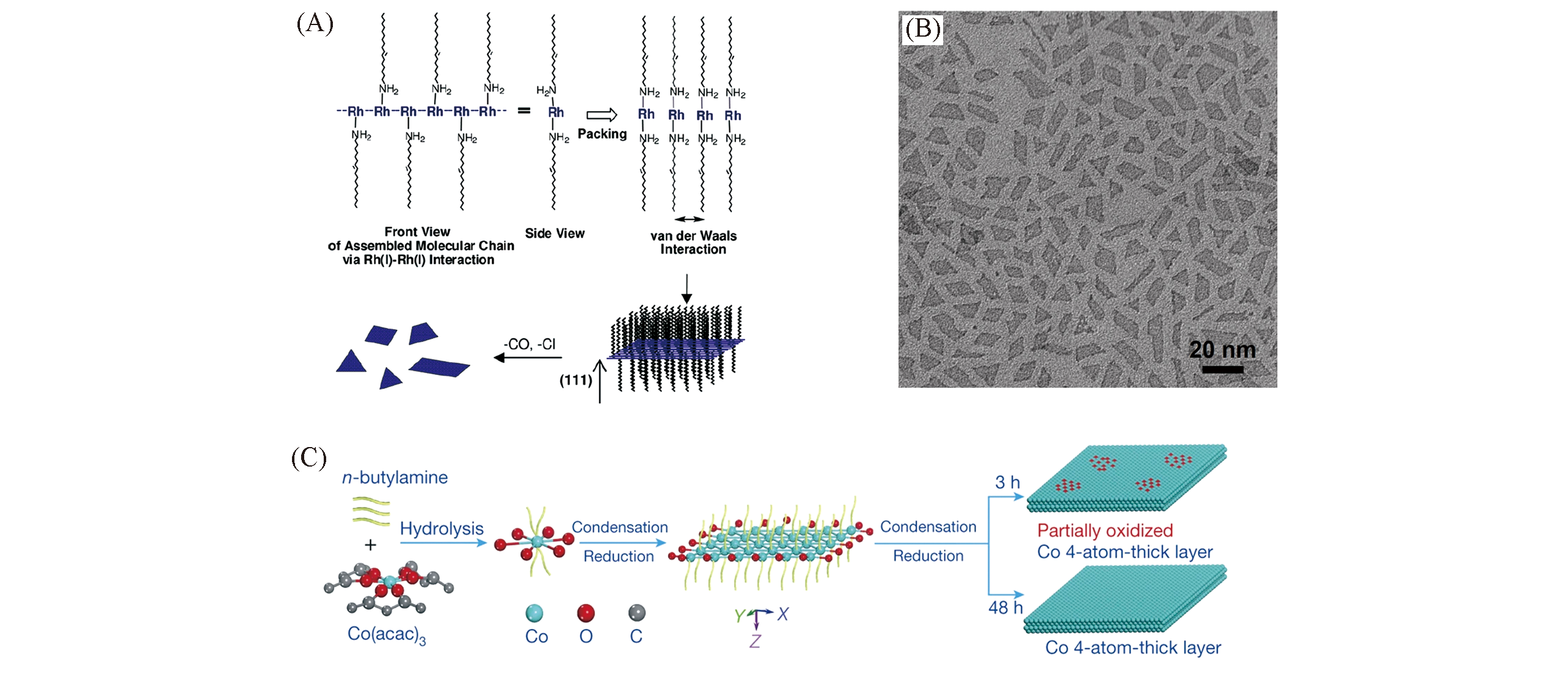
Fig.2 Growth mechanism(A) and TEM image(B) of rhodium nanoplates[84] and schematic formation process of the partially oxidized and pure Co 4?atomic?layer(C)[85](A, B) Copyright 2010, American Chemical Society; (C) Copyright 2016, Springer Nature.
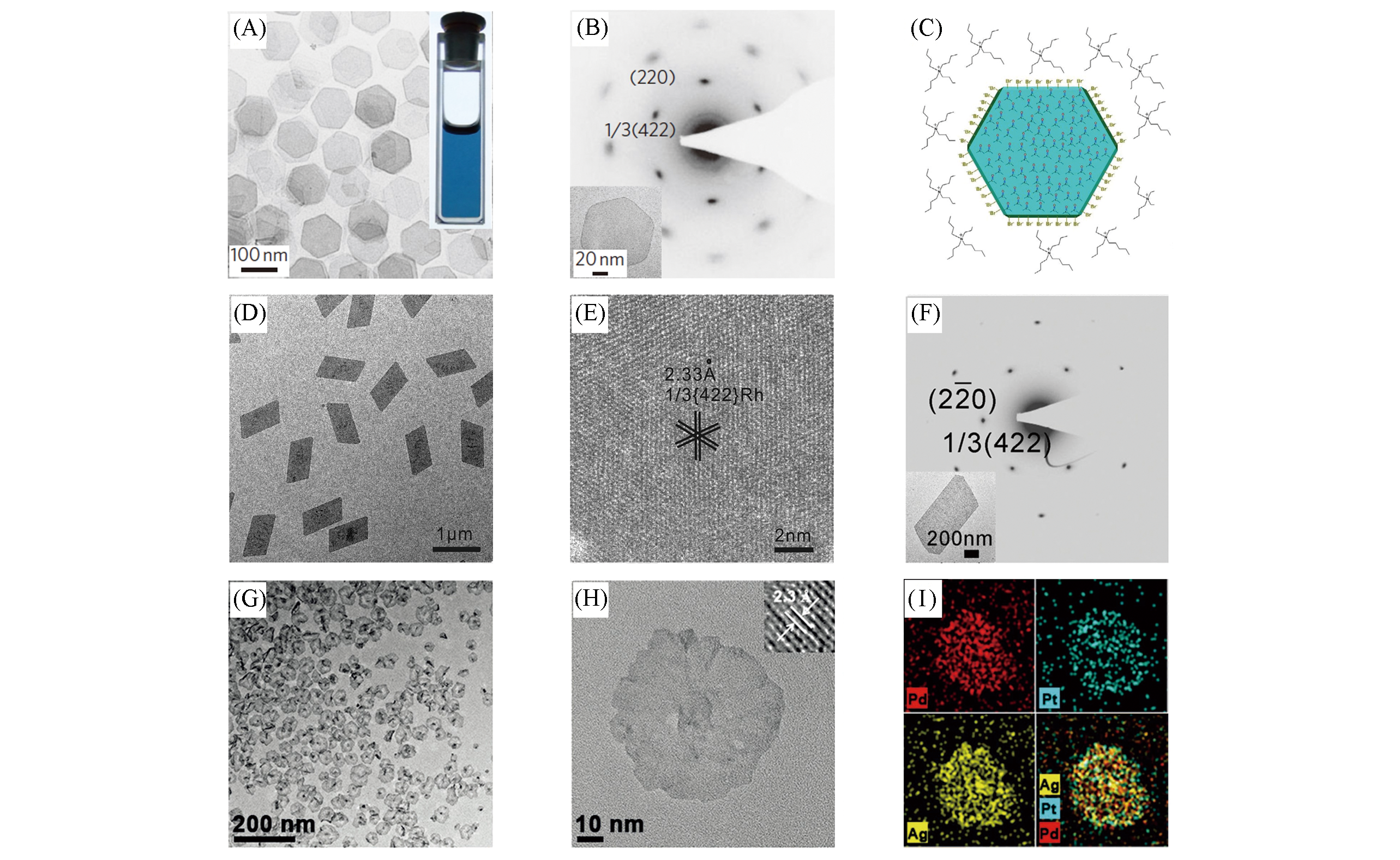
Fig.3 TEM image of Pd nanosheets(A), SAED pattern of a single Pd nanosheet(shown in the inset)(B), schematic diagram of Pd nanosheet with CO molecules on the (111) facets(C)[87], TEM image of Rh nanosheets with a long?edge length of 880 nm(D), high?resolution HAADF?STEM image projected along the [111] axis of Rh nanosheet(E), SAED pattern of a Rh nanosheet(shown in the inset)(F)[88], TEM images(G, H) and EDS elemental mappings(I) of Pd?Pt?Ag nanosheets[89]Inset of (A) is the photograph of an ethanol dispersion of Pd nanosheets in a vial. (A―C) Copyright 2010, Springer Nature; (D―F) Copyright 2015, Wiley‐VCH; (G―I) Copyright 2016, Wiley‐VCH.
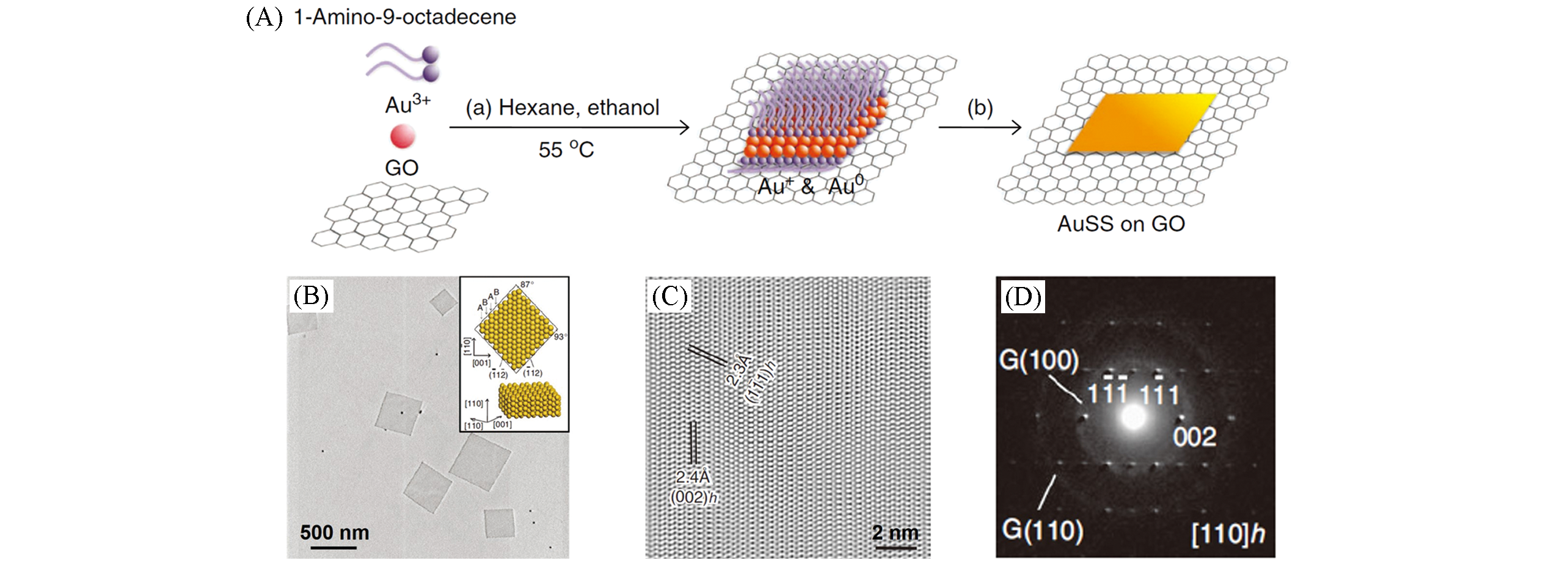
Fig.4 Schematic illustration of the formation process of Au square sheet on a GO sheet(A), TEM image(B), HRTEM image(C) and SAED pattern(D) of the Au square sheet[93]Copyright 2011, Springer Nature.
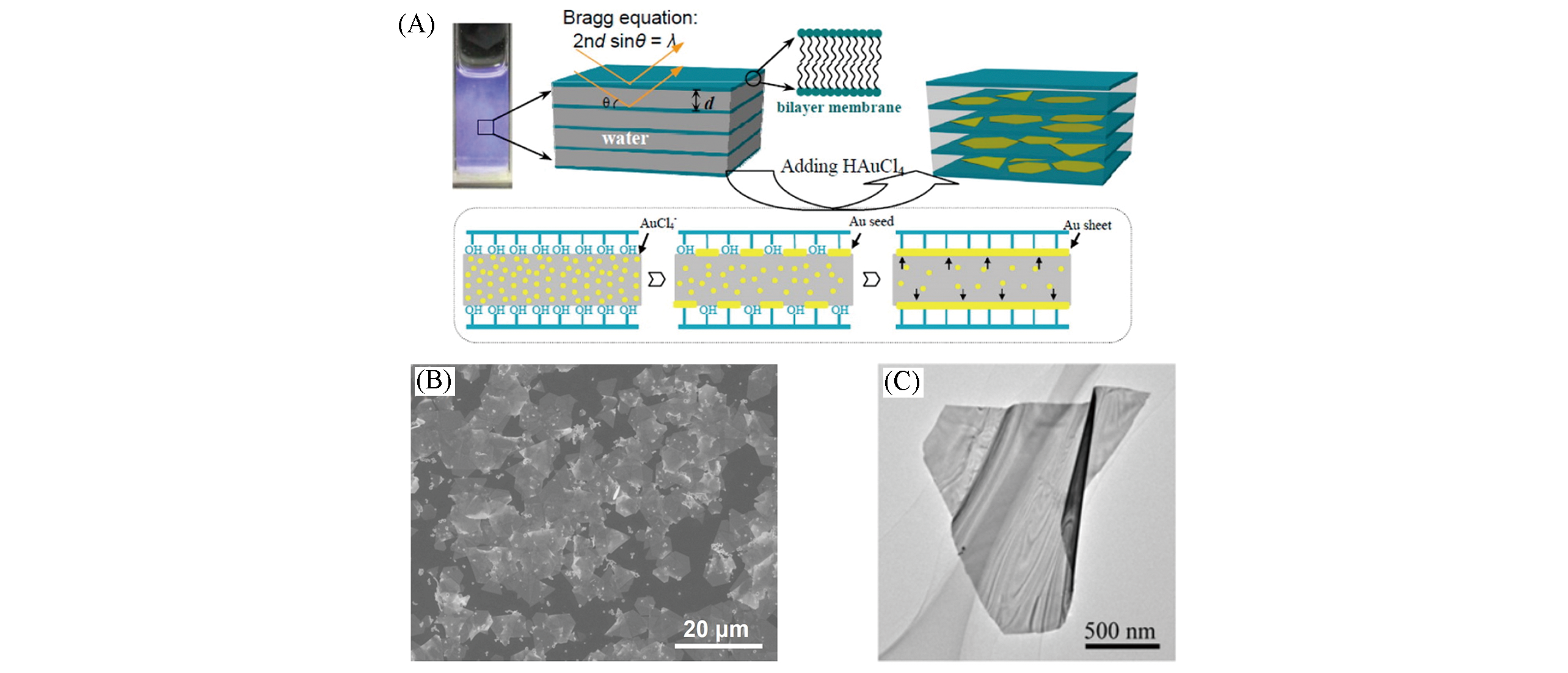
Fig.5 Schematic illustration of the Au nanosheet synthesis procedure(A), SEM image(B) and TEM image(C) of Au nanosheets[95]Copyright 2013, American Chemical Society.

Fig.6 Schematic illustration of the synthetic process of Pd@Ru nanosheets(A), aberration?corrected HAADF?STEM images of Pd@Ru nanosheets(B,C), STEM?EDS elemental mappings of a typical Pd@Ru nanosheet(D)[106], TEM image of fcc Au@Ag square sheets(E), SAED patterns of a typical fcc Au@Ag square sheet taken along the [100]f zone axes(F), HRTEM image of the fcc Au@Ag square sheets(G), HAADF?STEM image and the corresponding STEM?EDS elemental mappings of a typical fcc Au@Ag square sheet(H—J), high?resolution HAADF?STEM image and the corresponding STEM?EDS elemental mappings of the cross?section of a typical fcc Au@Ag square sheet(K)[107](A―D) Copyright 2016, Wiley‐VCH; (E―K) Copyright 2015, Springer Nature.
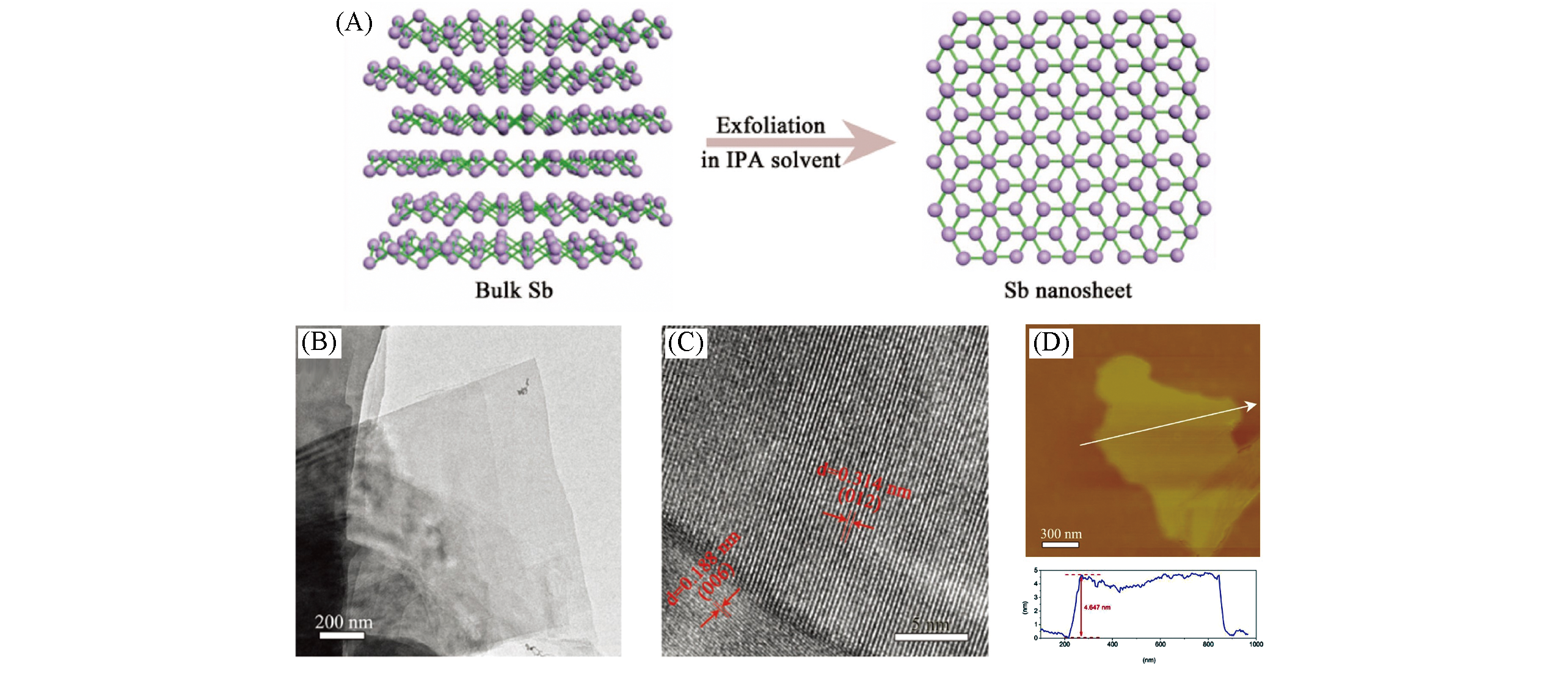
Fig.7 Schematic illustration of the fabrication process of Sb nanosheets(A), TEM image(B) and HRTEM image(C) of Sb nanosheets, typical AFM image and measured thickness of Sb nanosheet(D)[108]Copyright 2017, Wiley‐VCH.
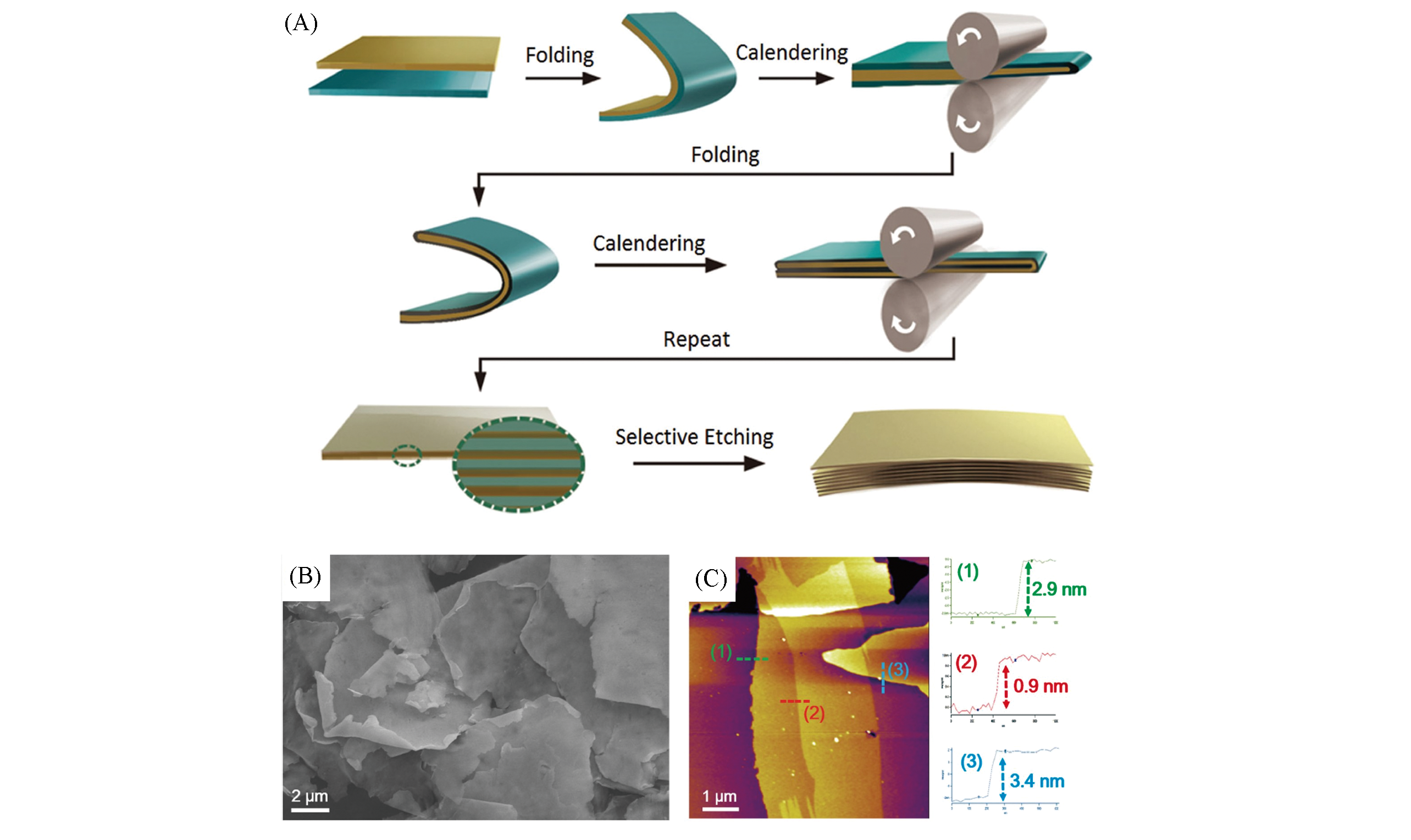
Fig.8 Schematic of the fabrication of metal nanosheets by a repeated size reduction method(A), SEM image of Ag nanosheets spread onto a Si substrate(B), AFM image of three single pieces of Ag nanosheets(C)[116]Copyright 2016, Wiley‐VCH.
| Application | Material | Electrolyte | Catalytic performance | Ref. |
|---|---|---|---|---|
ORR | PdMo bimetallene | 0.1 mol/L KOH | Mass activity:(16.37±0.60) A/mgPd Specific activity: 11.64 mA/cm2 (@ 0.9 V vs. RHE) | [ |
PtPb@Pt nanoplates | 0.1 mol/L HClO4 | Mass activity: 4.3 A/mgPt Specific activity: 7.8 mA/cm2 (@ 0.9 V vs. RHE) | [ | |
Pd@Ptmonolayer nanosheets | 0.1 mol/L HClO4 | Mass activity: 0.717 A/mgPt Specific activity: 0.438 A/cmPt2 (@ 0.9 V vs. RHE) | [ | |
5 monolayers Pd nanosheets | 0.1 mol/L KOH | Mass activity: 8.07 A/mgPd Specific activity: 10.91 mA/cm2 (@ 0.9 V vs. RHE) | [ | |
Intermetallic PtBi nanoplates | 0.1 mol/L HClO4 | Mass activity: 1.04 A/mgPt (@ 0.85 V vs. RHE) | [ | |
| MOR & EOR | PtBi nanoplatelets | 0.5 mol/L HClO4+0.5 mol/L CH3OH | 470 mA/mgPt | [ |
| Pt nanosheets/RGO | 0.5 mol/L H2SO4+0.5 mol/L CH3OH | 0.73 mA/cm2(@ 0.67 V vs. Ag/AgCl) | [ | |
| PtPb@Pt nanoplates | 0.1 mol/L HClO4+0.1 mol/L CH3OH | 1.5 A/mgPt, 2.7 mA/cm2 | [ | |
| PtCu nanosheets | 0.5 mol/L H2SO4+0.1 mol/L CH3CH2OH | 2.97 mA/cm2 | [ | |
| Pd?Pt?Ag nanosheets | 0.1 mol/L KOH+0.5 mol/L CH3CH2OH | 1.34 A/mg | [ | |
| HER & OER | Partially hydroxylated Ir nanosheets | 0.5 mol/L H2SO4 | ηOER=328 mV @ 10 mA/cm2 OER Tafel slope: 45.4 mV/dec | [ |
| 1 mol/L KOH | ηOER=266 mV @10 mA/cm2 OER Tafel slope: 29.1 mV/dec | |||
| Amorphous Ir nanosheets | 0.1 mol/L HClO4 | ηOER=255 mV @10 mA/cm2 OER Tafel slope: 40 mV/dec | [ | |
| Ir nanosheets assembly | 1 mol/L KOH | ηOER=242 mV @10 mA/cm2 | [ | |
| 0.1 mol/L HClO4 | ηOER=276 mV @10 mA/cm2 | |||
| Ru nanosheets | 0.5 mol/L H2SO4 | ηHER=20 mV @ 10 mA/mg HER Tafel slope: 46 mV/dec | [ | |
| RuCu nanosheets | 1 mol/L KOH | ηHER=20 mV @ 10 mA/cm2 ηOER=234 mV @ 10 mA/cm2 | [ | |
| 0.5 mol/L H2SO4 | ηHER=19 mV @ 10 mA/cm2 ηOER=236 mV @ 10 mA/cm2 | |||
| PtAgCo nanosheets | 0.5 mol/L H2SO4 | 705 mA/cm2(@ -0.4 V vs. RHE) | [ | |
| CO2ER | Graphene confined Sn quantum sheets | 0.1 mol/L NaHCO3 | 89% FE for formate (@ -1.8 V vs. SCE) | [ |
| Bi nanosheets | 0.5 mol/L NaHCO3 | 95% FE for formate (@ -1.5 V vs. SCE) | [ | |
| Partially oxidized Co nanosheets | 0.1 mol/L Na2SO4 | 90.1% FE for formate (@ -0.85 V vs. SCE) | [ | |
| NRR | Rhodium nanosheet | 0.1 mol/L KOH | 23.88 mg·h-1·mgcat-1 (@ -0.2 V vs. RHE) | [ |
Bi nanosheets | 0.1 mol/L Na2SO4 | (2.54±0.16) μg·h-1·cm-2 and (10.46±1.45)% FE for NH3(@ -0.8 V vs. RHE) | [ |
Table 2 Electrochemical performance of 2D metal nanomaterials for various electrocatalytic reactions*
| Application | Material | Electrolyte | Catalytic performance | Ref. |
|---|---|---|---|---|
ORR | PdMo bimetallene | 0.1 mol/L KOH | Mass activity:(16.37±0.60) A/mgPd Specific activity: 11.64 mA/cm2 (@ 0.9 V vs. RHE) | [ |
PtPb@Pt nanoplates | 0.1 mol/L HClO4 | Mass activity: 4.3 A/mgPt Specific activity: 7.8 mA/cm2 (@ 0.9 V vs. RHE) | [ | |
Pd@Ptmonolayer nanosheets | 0.1 mol/L HClO4 | Mass activity: 0.717 A/mgPt Specific activity: 0.438 A/cmPt2 (@ 0.9 V vs. RHE) | [ | |
5 monolayers Pd nanosheets | 0.1 mol/L KOH | Mass activity: 8.07 A/mgPd Specific activity: 10.91 mA/cm2 (@ 0.9 V vs. RHE) | [ | |
Intermetallic PtBi nanoplates | 0.1 mol/L HClO4 | Mass activity: 1.04 A/mgPt (@ 0.85 V vs. RHE) | [ | |
| MOR & EOR | PtBi nanoplatelets | 0.5 mol/L HClO4+0.5 mol/L CH3OH | 470 mA/mgPt | [ |
| Pt nanosheets/RGO | 0.5 mol/L H2SO4+0.5 mol/L CH3OH | 0.73 mA/cm2(@ 0.67 V vs. Ag/AgCl) | [ | |
| PtPb@Pt nanoplates | 0.1 mol/L HClO4+0.1 mol/L CH3OH | 1.5 A/mgPt, 2.7 mA/cm2 | [ | |
| PtCu nanosheets | 0.5 mol/L H2SO4+0.1 mol/L CH3CH2OH | 2.97 mA/cm2 | [ | |
| Pd?Pt?Ag nanosheets | 0.1 mol/L KOH+0.5 mol/L CH3CH2OH | 1.34 A/mg | [ | |
| HER & OER | Partially hydroxylated Ir nanosheets | 0.5 mol/L H2SO4 | ηOER=328 mV @ 10 mA/cm2 OER Tafel slope: 45.4 mV/dec | [ |
| 1 mol/L KOH | ηOER=266 mV @10 mA/cm2 OER Tafel slope: 29.1 mV/dec | |||
| Amorphous Ir nanosheets | 0.1 mol/L HClO4 | ηOER=255 mV @10 mA/cm2 OER Tafel slope: 40 mV/dec | [ | |
| Ir nanosheets assembly | 1 mol/L KOH | ηOER=242 mV @10 mA/cm2 | [ | |
| 0.1 mol/L HClO4 | ηOER=276 mV @10 mA/cm2 | |||
| Ru nanosheets | 0.5 mol/L H2SO4 | ηHER=20 mV @ 10 mA/mg HER Tafel slope: 46 mV/dec | [ | |
| RuCu nanosheets | 1 mol/L KOH | ηHER=20 mV @ 10 mA/cm2 ηOER=234 mV @ 10 mA/cm2 | [ | |
| 0.5 mol/L H2SO4 | ηHER=19 mV @ 10 mA/cm2 ηOER=236 mV @ 10 mA/cm2 | |||
| PtAgCo nanosheets | 0.5 mol/L H2SO4 | 705 mA/cm2(@ -0.4 V vs. RHE) | [ | |
| CO2ER | Graphene confined Sn quantum sheets | 0.1 mol/L NaHCO3 | 89% FE for formate (@ -1.8 V vs. SCE) | [ |
| Bi nanosheets | 0.5 mol/L NaHCO3 | 95% FE for formate (@ -1.5 V vs. SCE) | [ | |
| Partially oxidized Co nanosheets | 0.1 mol/L Na2SO4 | 90.1% FE for formate (@ -0.85 V vs. SCE) | [ | |
| NRR | Rhodium nanosheet | 0.1 mol/L KOH | 23.88 mg·h-1·mgcat-1 (@ -0.2 V vs. RHE) | [ |
Bi nanosheets | 0.1 mol/L Na2SO4 | (2.54±0.16) μg·h-1·cm-2 and (10.46±1.45)% FE for NH3(@ -0.8 V vs. RHE) | [ |
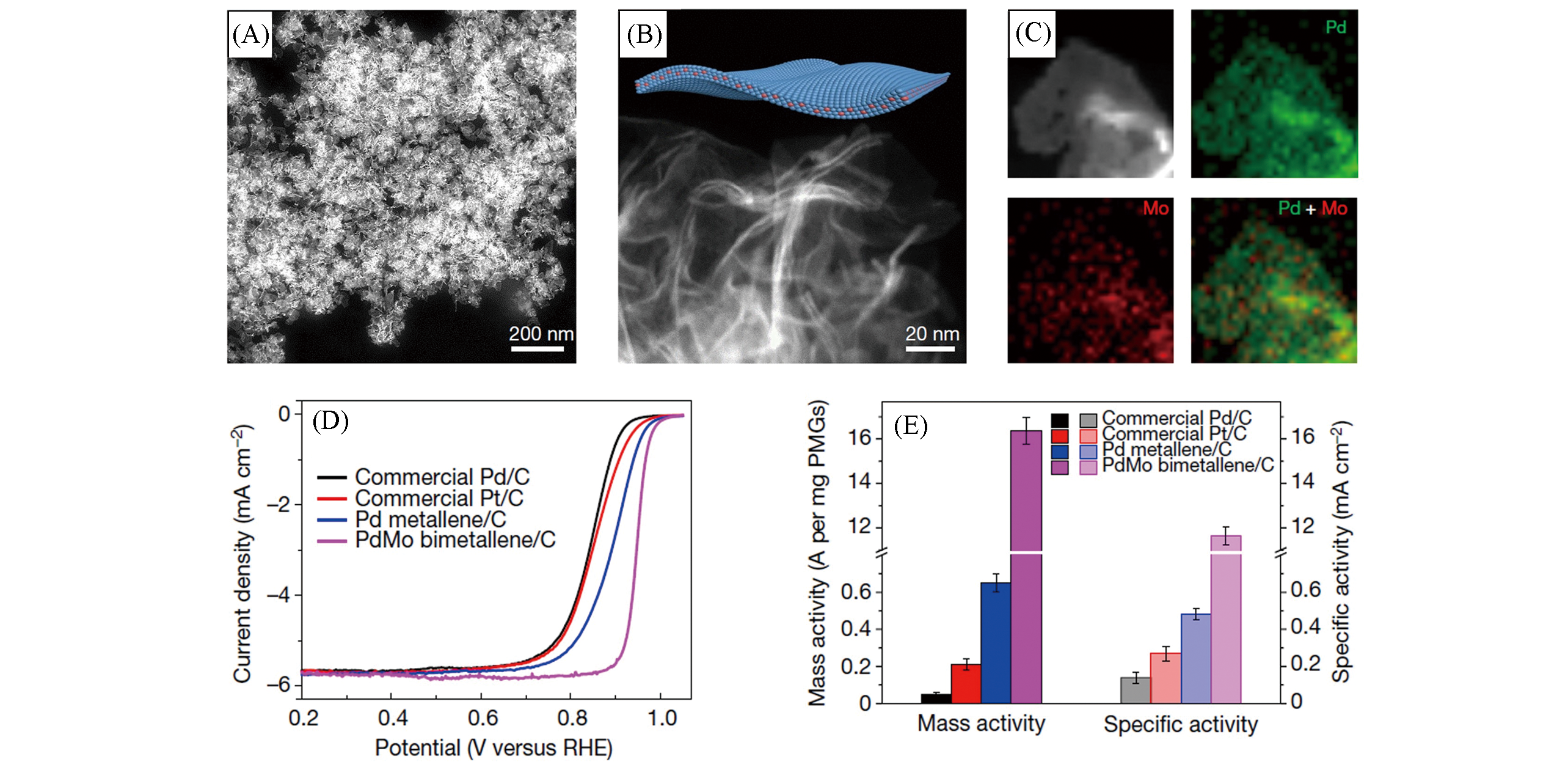
Fig.9 Low?magnification HAADF?STEM image(A), high?magnification HAADF?STEM image(B) and EDS elemental mappings of PdMo bimetallene(C), ORR polarization curves(D) and a comparison of the mass and specific activities in 0.1 mol/L KOH at 0.9 V(vs. RHE)(E) of the stated catalysts[128]Copyright 2019, Springer Nature.
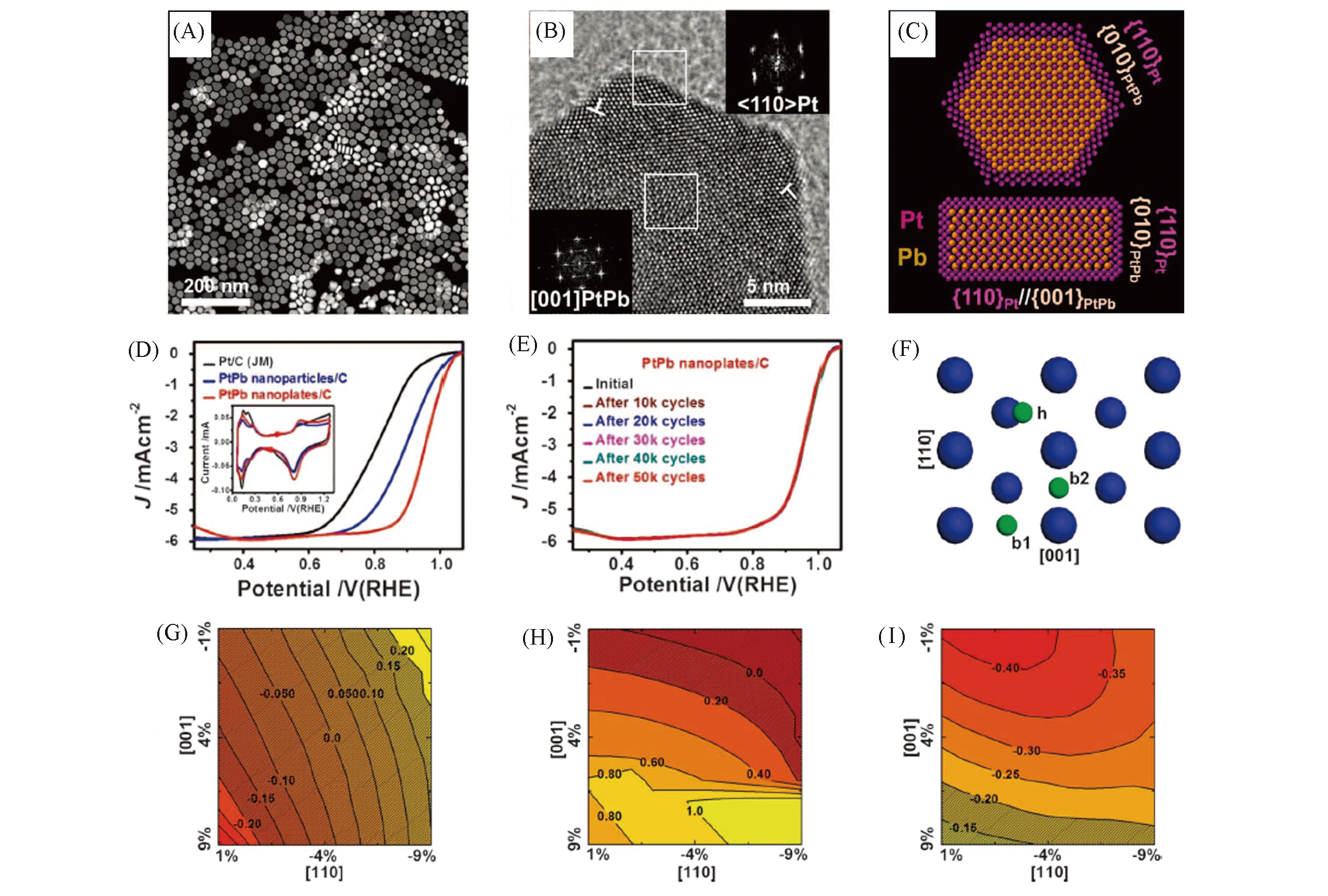
Fig.10 HAADF?STEM image(A), HRTEM image(B) and schematic atom models of the PtPb@Pt nanoplates(C), ORR polarization curves of different catalysts(D), ORR polarization curves of the PtPb@Pt nanoplates before and after different potential cycles between 0.6 and 1.1 V(vs. RHE)(E), atomic models of the Pt(110) surface(F), on the Pt(110) surface, ΔE0 as a function of biaxial strain in [110] and [001] directions for the “h” site(G) and the “b1” site(H), and the “b2” site is plotted in(I)[129](F) Three stable adsorption sites for oxygen: hollow(“h”) and two bridge sites(“b1” and “b2”). The blue and green spheres represent Pt and O atoms, respectively. (G―I) The optimal ΔE0 value is set to be 0. ΔE0 value falling into the shaded region implies a higher ORR activity than that on the flat Pt(111) surface.Copyright 2016, American Association for the Advancement of Science.
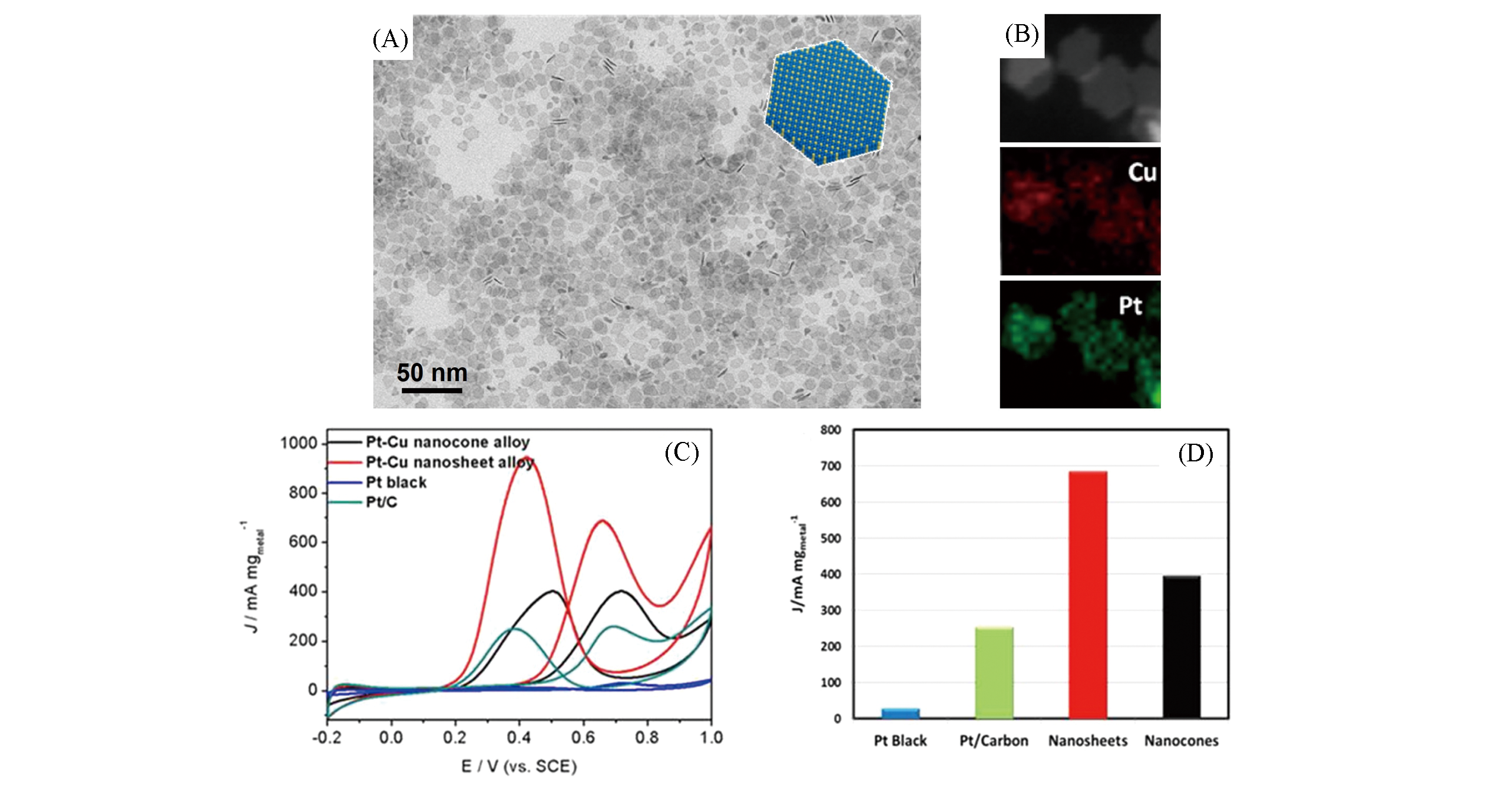
Fig.11 TEM image(A) and EDS elemental mapping(B) of Pt?Cu nanosheets, CVs obtained with various catalysts in 0.5 mol/L H2SO4 + 0.1 mol/L CH3CH2OH solution at a scan rate of 50 mV/s(C), comparison of mass activities of all catalysts(D)[67]Copyright 2013, American Chemical Society.
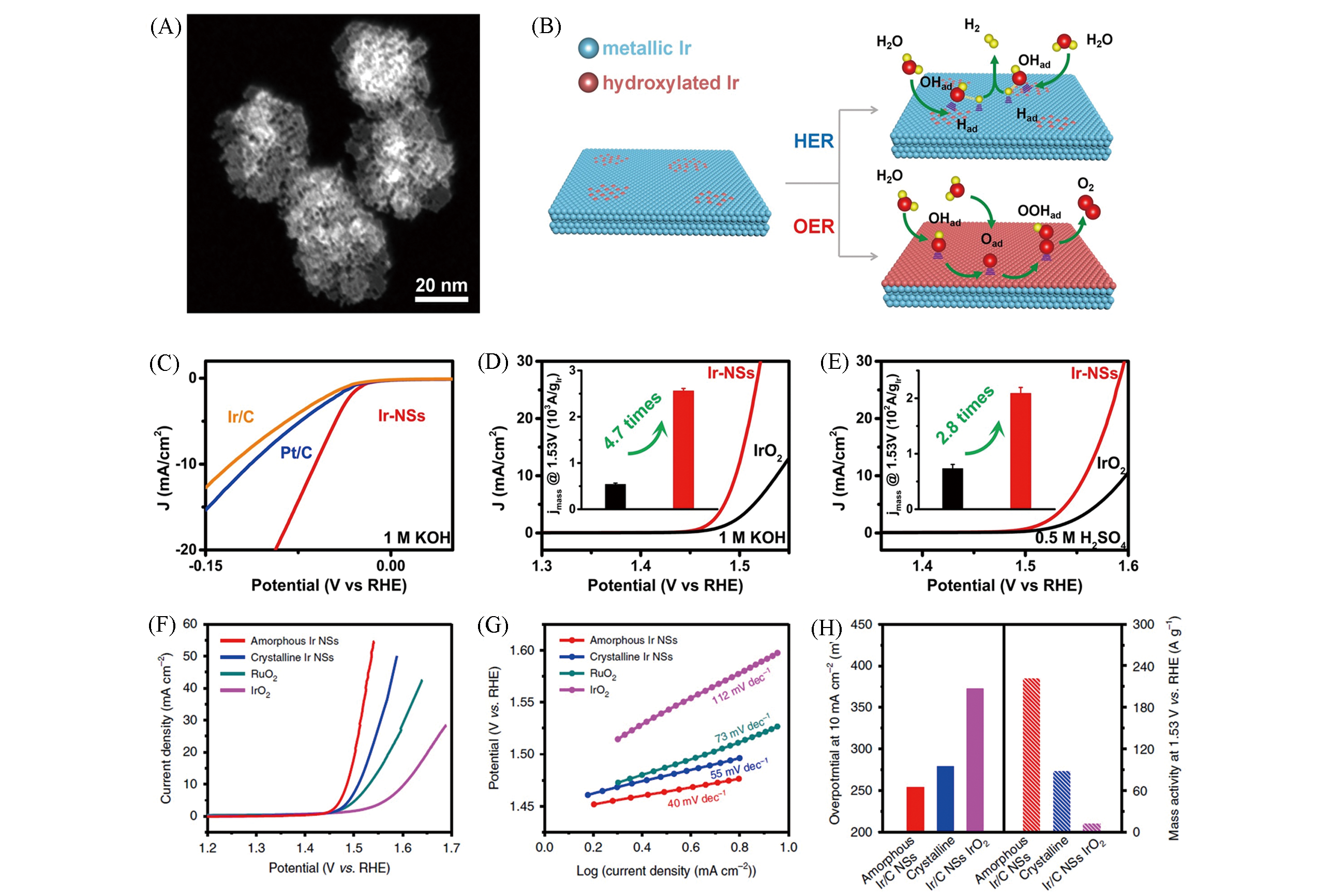
Fig.12 HAADF?STEM image of Ir?NSs(A), structural models of partially hydroxylated Ir nanosheets for OER and HER catalysis(B), polarization curves of various catalysts for HER in 1 mol/L KOH(C), polarization curves of various catalysts for OER in 1 mol/L KOH(D) and 0.5 mol/L H2SO4(E)[146]. polarization curves(F) and corresponding Tafel plots(G) of amorphous Ir nanosheets, crystalline Ir nanosheets, commercial RuO2 and IrO2 catalysts, overpotentials at 10 mA/cm2(left axis) and mass activity at 1.53 V(vs. RHE)(right axis) of various catalyst(H)[147](D,E) The insets show the corresponding mass activity at 1.53 V(vs. RHE).(A—E) Copyright 2020, Oxford University Press; (F—H) Copyright 2019, Springer Nature.
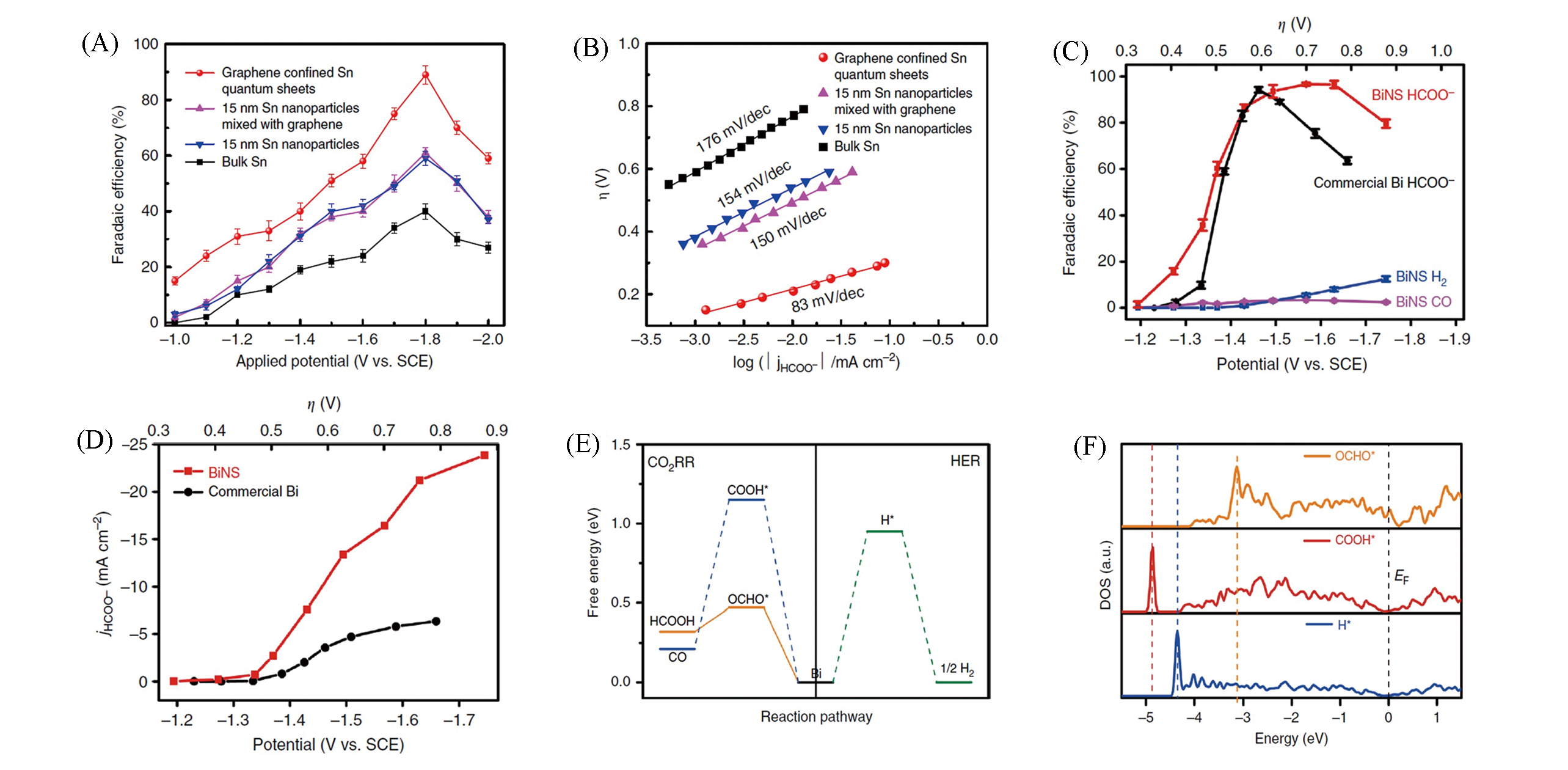
Fig.13 Faradaic efficiencies for formate at each applied potentials for 4 h(A), Tafel plots for producing formate(B) of the Sn quantum sheets confined in graphene, 15 nm Sn nanoparticles mixed with graphene, 15 nm Sn nanoparticles and bulk Sn[157], potential?dependent Faradaic efficiencies of HCOO?, CO, and H2 on BiNS in comparison with the Faradaic efficiency of HCOO- on commercial Bi nanopowder(C), potential?dependent HCOO- partial current density on BiNS and commercial Bi nanopowder(D), free?energy diagrams for HCOO-, CO, and H2 formation on Bi(001) plane(E), projected p?orbital DOS of the Bi site with OCHO*, COOH*, or H* adsorbate(F) [158](F) The Fermi level(EF) was at 0 eV, Ep in OCHO*, and COOH* and H* were highlighted with yellow, blue, and green dashed lines, respectively. (A,B) Copyright 2016, Springer Nature; (C—F) Copyright 2018, Springer Nature.
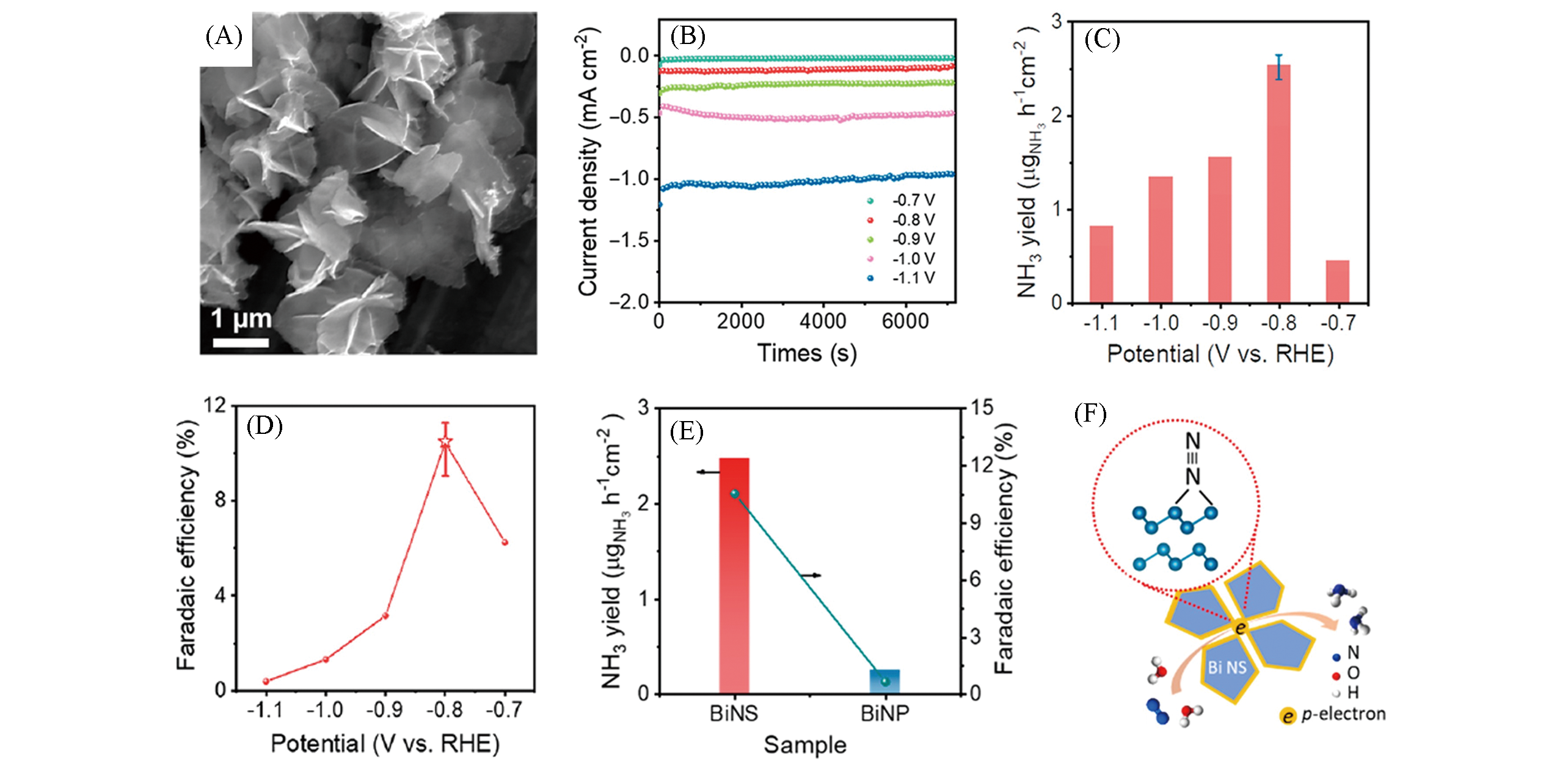
Fig.14 SEM image of Bi nanosheets(A), chronoamperometric curves of Bi nanosheets in a N2?saturated 0.1 mol/L Na2SO4 electrolyte at different applied potentials(B), NH3 formation rates(C) and NRR Faradaic efficiency(D) of Bi nanosheets at different applied potentials, comparison of NH3 yield and Faradaic efficiency of Bi nanosheets and Bi nanoparticles(E), schematic illustration of the NRR activity origin and N2 absorption mode on mosaic Bi nanosheets(F)[163]Copyright 2019, American Chemical Society.
| 1 | Dresselhaus M. S., Thomas I. L., Nature,2001, 414(6861), 332—337 |
| 2 | Chu S., Majumdar A., Nature,2012, 488(7411), 294—303 |
| 3 | Hightower M., Pierce S. A., Nature,2008, 452(7185), 285—286 |
| 4 | Lewis N. S., Science,2016, 351(6271), aad1920 |
| 5 | Dunn B., Kamath H., Tarascon J. M., Science,2011, 334(6058), 928—935 |
| 6 | Montoya J. H., Seitz L. C., Chakthranont P., Vojvodic A., Jaramillo T. F., Norskov J. K., Nat. Mater.,2017, 16(1), 70—81 |
| 7 | Yang Z., Zhang J., Kintner⁃Meyer M. C. W., Lu X., Choi D., Lemmon J. P., Liu J., Chem. Rev.,2011, 111(5), 3577—3613 |
| 8 | Winter M., Brodd R. J., Chem. Rev.,2004, 104(10), 4245—4270 |
| 9 | Xia C., Xia Y., Zhu P., Fan L., Wang H., Science,2019, 366(6462), 226—231 |
| 10 | Dai L., Qin Q., Wang P., Zhao X., Hu C., Liu P., Qin R., Chen M., Ou D., Xu C., Mo S., Wu B., Fu G., Zhang P., Zheng N., Sci. Adv.,2017, 3(9), e1701069 |
| 11 | Soloveichik G., Nat. Catal.,2019, 2(5), 377—380 |
| 12 | Lee H. K., Koh C. S. L., Lee Y. H., Liu C., Phang I. Y., Han X., Tsung C. K., Ling X. Y., Sci. Adv.,2018, 4(3), eaar3208 |
| 13 | Ren S., Joulié D., Salvatore D., Torbensen K., Wang M., Robert M., Berlinguette C. P., Science,2019, 365(6451), 367—369 |
| 14 | Ursua A., Gandia L. M., Sanchis P., Proc. IEEE,2012, 100(2), 410—426 |
| 15 | Xia C., Zhu P., Jiang Q., Pan Y., Liang W., Stavitsk E., Alshareef H. N., Wang H., Nat. Energy,2019, 4(9), 776—785 |
| 16 | Zhang X., Wu Z., Zhang X., Li L., Li Y., Xu H., Li X., Yu X., Zhang Z., Liang Y., Wang H., Nat. Commun.,2017, 8, 14675 |
| 17 | Hao Y. C., Guo Y., Chen L. W., Shu M., Wang X. Y., Bu T. A., Gao W. Y., Zhang N., Su X., Feng X., Zhou J. W., Wang B., Hu C. W., Yin A. X., Si R., Zhang Y. W., Yan C. H., Nat. Catal.,2019, 2(5), 448—456 |
| 18 | Zou X., Zhang Y., Chem. Soc. Rev.,2015, 44(15), 5148—5180 |
| 19 | Qiao J., Liu Y., Hong F., Zhang J., Chem. Soc. Rev.,2014, 43(2), 631—675 |
| 20 | Wachsman E. D., Lee K. T., Science,2011, 334(6058), 935—939 |
| 21 | Feng Y., Liu H., Yang J., Sci. Adv.,2017, 3(6), e1700580 |
| 22 | Ott S., Orfanidi A., Schmies H., Anke B., Nong H. N., Hübner J., Gernert U., Gliech M., Lerch M., Strasser P., Nat. Mater.,2019, 19(1), 77—85 |
| 23 | Wang X. X., Swihart M. T., Wu G., Nat. Catal.,2019, 2(7), 578—589 |
| 24 | Debe M. K., Nature,2012, 486(7401), 43—51 |
| 25 | Beermann V., Gocyla M., Willinger E., Rudi S., Heggen M., Dunin⁃Borkowski R. E., Willinger M. G., Strasser P., Nano Lett.,2016, 16(3), 1719—1725 |
| 26 | Carmo M., Fritz D. L., Mergel J., Stolten D., Int. J. Hydrogen Energy,2013, 38(12), 4901—4934 |
| 27 | Seh Z. W., Kibsgaard J., Dickens C. F., Chorkendorff I., Nørskov J. K., Jaramillo T. F., Science,2017, 355(6321), eaad4998 |
| 28 | Hwang J., Rao R. R., Giordano L., Katayama Y., Yu Y., Shao⁃Horn Y., Science,2017, 358(6364), 751—756 |
| 29 | Nørskov J. K., Bligaard T., Rossmeisl J., Christensen C. H., Nat. Chem.,2009, 1, 37—46 |
| 30 | Mistry H., Varela A. S., Kühl S., Strasser P., Cuenya B. R., Nat. Rev. Mater.,2016, 1(4), 16009 |
| 31 | Gao D., Arán⁃Ais R. M., Jeon H. S., Roldan Cuenya B., Nat. Catal.,2019, 2(3), 198—210 |
| 32 | Neto A. C., Guinea F., Peres N. M., Novoselov K. S., Geim A. K., Rev. Mod. Phys.,2009, 81(1), 1—23 |
| 33 | Rusponi S., Cren T., Weiss N., Epple M., Buluschek P., Claude L., Brune H., Nat. Mater.,2003, 2(8), 546—551 |
| 34 | Meng Z., Stolz R. M., Mendecki L., Mirica K. A., Chem. Rev.,2019, 119(1), 478—598 |
| 35 | Bastys V., Pastoriza⁃Santos I., Rodríguez⁃González B., Vaisnoras R., Liz⁃Marzán L. M., Adv. Funct. Mater.,2006, 16(6), 766—773 |
| 36 | Zhang H., ACS Nano,2015, 9(10), 9451—9469 |
| 37 | Tao H., Gao Y., Talreja N., Guo F., Texter J., Yan C., Sun Z., J. Mater. Chem. A,2017, 5(16), 7257—7284 |
| 38 | Chhowalla M., Shin H. S., Eda G., Li L. J., Loh K. P., Zhang H., Nat. Chem.,2013, 5(4), 263—275 |
| 39 | Novoselov K. S., Geim A. K., Morozov S. V., Jiang D., Zhang Y., Dubonos S. V., Grigorieva I. V., Firsov A. A., Science,2004, 306(5696), 666—669 |
| 40 | Wu J., Ma S., Sun J., Gold J. I., Tiwary C., Kim B., Zhu L., Chopra N., Odeh I. N., Vajtai R., Yu A. Z., Luo R., Lou J., Ding G., Kenis P. J. A., Ajayan P. M., Nat. Commun.,2016, 7, 13869 |
| 41 | Bi W., Li X., You R., Chen M., Yuan R., Huang W., Wu X., Chu W., Wu C., Xie Y., Adv. Mater., 2018, 30(18), 1706617 |
| 42 | Wang X., Li X., Zhang L., Yoon Y., Weber P. K., Wang H., Guo J., Dai H., Science,2009, 324(5928), 768—771 |
| 43 | Nistor R. A., Newns D. M., Martyna G. J., ACS Nano,2011, 5(4), 3096—3103 |
| 44 | Zhang J., Chen Y., Wang X., Energy Environ. Sci.,2015, 8(11), 3092—3108 |
| 45 | Ong W. J., Tan L. L., Ng Y. H., Yong S. T., Chai S. P., Chem. Rev.,2016, 116(12), 7159—7329 |
| 46 | Lv R., Robinson J. A., Schaak R. E., Sun D., Sun Y., Mallouk T. E., Terrones M., Acc. Chem. Res.,2015, 48(1), 56—64 |
| 47 | Tan C., Zhang H., Chem. Soc. Rev.,2015, 44(9), 2713—2731 |
| 48 | Li H., Wu J., Yin Z., Zhang H., Acc. Chem. Res.,2014, 47(4), 1067—1075 |
| 49 | Weng Q., Wang X., Wang X., Bando Y., Golberg D., Chem. Soc. Rev.,2016, 45(14), 3989—4012 |
| 50 | Li L. H., Chen Y., Adv. Funct. Mater.,2016, 26(16), 2594—2608 |
| 51 | Wang Q., O’Hare D., Chem. Rev.,2012, 112(7), 4124—4155 |
| 52 | Yin H., Tang Z., Chem. Soc. Rev.,2016, 45(18), 4873—4891 |
| 53 | Feng J., He Y., Liu Y., Du Y., Li D., Chem. Soc. Rev.,2015, 44(15), 5291—5319 |
| 54 | Naguib M., Mochalin V. N., Barsoum M. W., Gogotsi Y., Adv. Mater.,2014, 26(7), 992—1005 |
| 55 | Hong Ng V. M., Huang H., Zhou K., Lee P. S., Que W., Xu J. Z., Kong L. B., J. Mater. Chem. A,2017, 5(7), 3039—3068 |
| 56 | Fan Z., Huang X., Tan C., Zhang H., Chem. Sci.,2015, 6(1), 95—111 |
| 57 | Ling T., Wang J. J., Zhang H., Song S. T., Zhou Y. Z., Zhao J., Du X. W., Adv. Mater.,2015, 27(36), 5396—5402 |
| 58 | An B., Li M., Wang J., Li C., Front. Chem. Sci. Eng.,2016, 10(3), 360—382 |
| 59 | Ma R., Sasaki T., Acc. Chem. Res.,2015, 48(1), 136—143 |
| 60 | Peng Y., Li Y., Ban Y., Jin H., Jiao W., Liu X., Yang W., Science,2014, 346(6215), 1356—1359 |
| 61 | Zhao S., Wang Y., Dong J., He C. T., Yin H., An P., Zhao K., Zhang X., Gao C., Zhang L., Lv J., Wang J., Zhang J., Khattak A. M., Khan N. A., Wei Z., Zhang J., Liu S., Zhao H., Tang Z., Nat. Energy,2016, 1(12), 16184 |
| 62 | Rodenas T., Luz I., Prieto G., Seoane B., Miro H., Corma A., Kapteijn F., Llabrési Xamena F. X., Gascon J., Nat. Mater.,2015, 14(1), 48—55 |
| 63 | Ma Y., Li B., Yang S., Mater. Chem. Front.,2018, 2(3), 456—467 |
| 64 | Chen Y., Fan Z., Zhang Z., Niu W., Li C., Yang N., Chen B., Zhang H., Chem. Rev.,2018, 118(13), 6409—6455 |
| 65 | Luc W., Fu X., Shi J., Lv J. J., Jouny M., Ko B. H., Xu Y., Tu Q., Hu X., Wu J., Yue Q., Liu Y., Jiao F., Kang Y., Nat. Catal.,2019, 2(5), 423—430 |
| 66 | Pi Y., Zhang N., Guo S., Guo J., Huang X., Nano Lett.,2016, 16(7), 4424—4430 |
| 67 | Saleem F., Zhang Z., Xu B., Xu X., He P., Wang X., J. Am. Chem. Soc.,2013, 135(49), 18304—18307 |
| 68 | Li H., Chen G., Yang H., Wang X., Liang J., Liu P., Chen M., Zheng N., Angew. Chem. Int. Ed.,2013, 52(32), 8368—8372 |
| 69 | Yang N., Zhang Z., Chen B., Huang Y., Chen J., Lai Z., Chen Y., Sindoro M., Wang A. L., Cheng H., Fan Z., Liu X., Li B., Zong Y., Gu L., Zhang H., Adv. Mater.,2017, 29(29), 1700769 |
| 70 | Liu H. M., Han S. H., Zhao Y., Zhu Y. Y., Tian X. L., Zeng J. H., Jiang J. X., Xia B. Y., Chen Y., J. Mater. Chem. A,2018, 6(7), 3211—3217 |
| 71 | Tan C., Cao X., Wu X. J., He Q., Yang J., Zhang X., Chen J., Zhao W., Han S., Nam G. H., Sindoro M., Zhang H., Chem. Rev.,2017, 117(9), 6225—6331 |
| 72 | Nasilowski M., Mahler B., Lhuillier E., Ithurria S., Dubertret B., Chem. Rev.,2016, 116(18), 10934—10982 |
| 73 | Nicolosi V., Chhowalla M., Kanatzidis M. G., Strano M. S., Coleman J. N., Science,2013, 340(6139), 1226419 |
| 74 | Song F., Hu X., Nat. Commun.,2014, 5, 4477 |
| 75 | Duan H., Yan N., Yu R., Chang C. R., Zhou G., Hu H. S., Rong H., Niu Z., Mao J., Asakura H., Tanaka T., Dyson P. J., Li J., Li Y., Nat. Commun.,2014, 5, 3093 |
| 76 | Zhang N., Shao Q., Pi Y., Guo J., Huang X., Chem. Mater.,2017, 29(11), 5009—5015 |
| 77 | Feng Y., Shao Q., Lv F., Bu L., Guo J., Guo S., Huang X., Adv. Sci.,2020, 7(1), 1800178 |
| 78 | Feng Y., Huang B., Yang C., Shao Q., Huang X., Adv. Funct. Mater.,2019, 29(45), 1904429 |
| 79 | Yao Q., Huang B., Zhang N., Sun M., Shao Q., Huang X., Angew. Chem. Int. Ed.,2019, 58(39), 13983—13988 |
| 80 | Xia Y., Xiong Y., Lim B., Skrabalak S. E., Angew. Chem. Int. Ed.,2009, 48(1), 60—103 |
| 81 | Zeng J., Zheng Y., Rycenga M., Tao J., Li Z. Y., Zhang Q., Zhu Y., Xia Y., J. Am. Chem. Soc.,2010, 132(25), 8552—8553 |
| 82 | Niu Z., Li Y., Chem. Mater.,2014, 26(1), 72—83 |
| 83 | Yang T. H., Shi Y., Janssen A., Xia Y., Angew. Chem. Int. Ed.,2020, 59(36),15378—15401 |
| 84 | Jang K., Kim H. J., Son S. U., Chem. Mater.,2010, 22(4), 1273—1275 |
| 85 | Gao S., Lin Y., Jiao X., Sun Y., Luo Q., Zhang W., Li D., Yang J., Xie Y., Nature,2016, 529(7584), 68—71 |
| 86 | Xiong Y., Washio I., Chen J., Cai H., Li Z. Y., Xia Y., Langmuir,2006, 22(20), 8563—8570 |
| 87 | Huang X., Tang S., Mu X., Dai Y., Chen G., Zhou Z., Ruan F., Yang Z., Zheng N., Nat. Nanotechnol.,2011, 6(1), 28—32 |
| 88 | Zhao L., Xu C., Su H., Liang J., Lin S., Gu L., Wang X., Chen M., Zheng N., Adv. Sci.,2015, 2(6), 1500100 |
| 89 | Hong J. W., Kim Y., Wi D. H., Lee S., Lee S. U., Lee Y. W., Choi S. I., Han S. W., Angew. Chem. Int. Ed.,2016, 55(8), 2753—2758 |
| 90 | Huang X., Li H., Li S., Wu S., Boey F., Ma J., Zhang H., Angew. Chem. Int. Ed.,2011, 50(51), 12245—12248 |
| 91 | Jiang Y., Yan Y., Chen W., Khan Y., Wu J., Zhang H., Yang D., Chem. Commun.,2016, 52(99), 14204—14207 |
| 92 | Yang S., Qiu P., Yang G., Carbon,2014, 77, 1123—1131 |
| 93 | Huang X., Li S., Huang Y., Wu S., Zhou X., Li S., Gan C. L., Boey F., Mirkin C. A., Zhang H., Nat. Commun.,2011, 2, 292 |
| 94 | Huang X., Zeng Z., Bao S., Wang M., Qi X., Fan Z., Zhang H., Nat. Commun.,2013, 4, 1444 |
| 95 | Qin H. L., Wang D., Huang Z. L., Wu D. M., Zeng Z. C., Ren B., Xu K., Jin J., J. Am. Chem. Soc.,2013, 135(34), 12544—12547 |
| 96 | Wang L., Zhu Y., Wang J. Q., Liu F., Huang J., Meng X., Basset J. M., Han Y., Xiao F. S., Nat. Commun.,2015, 6, 6957 |
| 97 | Kida T., Langmuir,2008, 24(15), 7648—7650 |
| 98 | Banu K., Shimura T., New J. Chem.,2012, 36(10), 2112—2120 |
| 99 | Sanyal A., Sastry M., Chem. Commun.,2003, 1(11), 1236—1237 |
| 100 | Wang C. W., Ding H. P., Xin G. Q., Chen X., Lee Y. I., Hao J., Liu H. G., Colloids Surf. A,2009, 340(1), 93—98 |
| 101 | Xia Y., Gilroy K. D., Peng H. C., Xia X., Angew. Chem. Int. Ed.,2017, 56(1), 60—95 |
| 102 | Sánchez Iglesias A., Winckelmans N., Altantzis T., Bals S., Grzelczak M., Liz Marzán L. M., J. Am. Chem. Soc.,2017, 139(1), 107—110 |
| 103 | Kuo C. H., Chen C. H., Huang M., Adv. Funct. Mater.,2007, 17(18), 3773—3780 |
| 104 | Xia X., Figueroa Cosme L., Tao J., Peng H. C., Niu G., Zhu Y., Xia Y., J. Am. Chem. Soc.,2014, 136(31), 10878—10881 |
| 105 | Wang W., Zhao Y., Ding Y., Nanoscale,2015, 7(28), 11934—11939 |
| 106 | Zhang Z., Liu Y., Chen B., Gong Y., Gu L., Fan Z., Yang N., Lai Z., Chen Y., Wang J., Huang Y., Sindoro M., Niu W., Li B., Zong Y., Yang Y., Huang X., Huo F., Huang W., Zhang H., Adv. Mater.,2016, 28(46), 10282—10286 |
| 107 | Fan Z., Huang X., Han Y., Bosman M., Wang Q., Zhu Y., Liu Q., Li B., Zeng Z., Wu J., Shi W., Li S., Gan C. L., Zhang H., Nat. Commun.,2015, 6, 6571 |
| 108 | Gu J., Du Z., Zhang C., Ma J., Li B., Yang S., Adv. Energy Mater.,2017, 7(17), 1700447 |
| 109 | Gusmão R., Sofer Z., Bouša D., Pumera M., Angew. Chem. Int. Ed.,2017, 56(46), 14417—14422 |
| 110 | Gibaja C., Rodriguez San Miguel D., Ares P., Gómez Herrero J., Varela M., Gillen R., Maultzsch J., Hauke F., Hirsch A., Abellán G., Zamora F., Angew. Chem. Int. Ed.,2016, 55(46), 14345—14349 |
| 111 | Ares P., Aguilar Galindo F., Rodríguez San Miguel D., Aldave D. A., Díaz Tendero S., Alcamí M., Martín F., Gómez Herrero J., Zamora F., Adv. Mater.,2016, 28(30), 6332—6336 |
| 112 | Fukuda K., Sato J., Saida T., Sugimoto W., Ebina Y., Shibata T., Osada M., Sasaki T., Inorg. Chem.,2013, 52(5), 2280—2282 |
| 113 | Funatsu A., Tateishi H., Hatakeyama K., Fukunaga Y., Taniguchi T., Koinuma M., Matsuura H., Matsumoto Y., Chem. Commun.,2014, 50(62), 8503—8506 |
| 114 | Hussain N., Liang T., Zhang Q., Anwar T., Huang Y., Lang J., Huang K., Wu H., Small,2017, 13(36), 1701349 |
| 115 | Huang K., Hou J., Zhang Q., Ou G., Ning D., Hussain N., Xu Y., Ge B., Liu K., Wu H., Chem. Commun.,2018, 54(2), 160—163 |
| 116 | Liu H., Tang H., Fang M., Si W., Zhang Q., Huang Z., Gu L., Pan W., Yao J., Nan C., Wu H., Adv. Mater.,2016, 28(37), 8170—8176 |
| 117 | Gu J., Li B., Du Z., Zhang C., Zhang D., Yang S., Adv. Funct. Mater.,2017, 27(27), 1700840 |
| 118 | Demirdöven N., Deutch J., Science,2004, 305(5686), 974—976 |
| 119 | Schäfer A., Heywood J. B., Weiss M. A., Energy,2006, 31(12), 2064—2087 |
| 120 | Chattot R., Le Bacq O., Beermann V., Kühl S., Herranz J., Henning S., Kühn L., Asset T., Guétaz L., Renou G., Drnec J., Bordet P., Pasturel A., Eychmüller A., Schmidt T. J., Strasser P., Dubau L., Maillard F., Nat. Mater.,2018, 17(9), 827—833 |
| 121 | Shao M., Chang Q., Dodelet J. P., Chenitz R., Chem. Rev.,2016, 116(6), 3594—3657 |
| 122 | Tian X., Zhao X., Su Y. Q., Wang L., Wang H., Dang D., Chi B., Liu H., Hensen E. J. M., Lou X. W., Xia B. Y., Science,2019, 366(6467), 850—856 |
| 123 | Strasser P., Science,2015, 349(6246), 379—380 |
| 124 | Li M., Zhao Z., Cheng T., Fortunelli A., Chen C. Y., Yu R., Zhang Q., Gu L., Merinov B. V., Lin Z., Zhu E., Yu T., Jia Q., Guo J., Zhang L., Goddard W. A., Huang Y., Duan X., Science,2016, 354(6318), 1414—1419 |
| 125 | Chen C., Kang Y., Huo Z., Zhu Z., Huang W., Xin H. L., Snyder J. D., Li D., Herron J. A., Mavrikakis M., Chi M., More K. L., Li Y., Markovic N. M., Somorjai G. A., Yang P., Stamenkovic V. R., Science,2014, 343(6177), 1339—1343 |
| 126 | Bu L., Ding J., Guo S., Zhang X., Su D., Zhu X., Yao J., Guo J., Lu G., Huang X., Adv. Mater.,2015, 27(44), 7204—7212 |
| 127 | Feng Y., Shao Q., Ji Y., Cui X., Li Y., Zhu X., Huang X., Sci. Adv.,2018, 4(7), eaap8817 |
| 128 | Luo M., Zhao Z., Zhang Y., Sun Y., Xing Y., Lv F., Yang Y., Zhang X., Hwang S., Qin Y., Ma J. Y., Lin F., Su D., Lu G., Guo S., Nature,2019, 574(7776), 81—85 |
| 129 | Bu L., Zhang N., Guo S., Zhang X., Li J., Yao J., Wu T., Lu G., Ma J. Y., Su D., Huang X., Science,2016, 354(6318), 1410—1414 |
| 130 | Shih C. F., Zhang T., Li J., Bai C., Joule,2018, 2(10), 1925—1949 |
| 131 | Huang W., Wang H., Zhou J., Wang J., Duchesne P. N., Muir D., Zhang P., Han N., Zhao F., Zeng M., Zhong J., Jin C., Li Y., Lee S. T., Dai H., Nat. Commun.,2015, 6, 10035 |
| 132 | Liao H., Zhu J., Hou Y., Nanoscale,2014, 6(2), 1049—1055 |
| 133 | Zhao X., Dai L., Qin Q., Pei F., Hu C., Zheng N., Small,2017,13(12), 1602970 |
| 134 | Ge J., Wei P., Wu G., Liu Y., Yuan T., Li Z., Qu Y., Wu Y., Li H., Zhuang Z., Hong X., Li Y., Angew. Chem. Int. Ed., 2018, 130(13), 3493—3496 |
| 135 | Zhang Y., Zhu X., Guo J., Huang X., ACS Appl. Mater. Interfaces,2016, 8(32), 20642—20649 |
| 136 | Luo X., Liu C., Wang X., Shao Q., Pi Y., Zhu T., Li Y., Huang X., Nano Lett.,2020, 20(3), 1967—1973 |
| 137 | Kowal A., Li M., Shao M., Sasaki K., Vukmirovic M. B., Zhang J., Marinkovic N. S., Liu P., Frenkel A. I., Adzic R. R., Nat. Mater.,2009, 8(4), 325—330 |
| 138 | Rizo R., Arán Ais R. M., Padgett E., Muller D. A., Lázaro M. J., Solla Gullón J., Feliu J. M., Pastor E., Abruña H. D., J. Am. Chem. Soc.,2018, 140(10), 3791—3797 |
| 139 | Millet P., Mbemba N., Grigoriev S. A., Fateev V. N., Aukauloo A., Etiévant C., Int. J. Hydrogen Energy,2011, 36(6), 4134—4142 |
| 140 | Turner J. A., Science,2004, 305(5686), 972—974 |
| 141 | McCrory C. C. L., Jung S., Ferrer I. M., Chatman S. M., Peters J. C., Jaramillo T. F., J. Am. Chem. Soc.,2015, 137(13), 4347—4357 |
| 142 | Morales Guio C. G., Stern L. A., Hu X., Chem. Soc. Rev.,2014, 43(18), 6555—6569 |
| 143 | Suen N. T., Hung S. F., Quan Q., Zhang N., Xu Y. J., Chen H. M., Chem. Soc. Rev.,2017, 46(2), 337—365 |
| 144 | Song J., Wei C., Huang Z. F., Liu C., Zeng L., Wang X., Xu Z. J., Chem. Soc. Rev.,2020, 49(7), 2196—2214 |
| 145 | Wang J., Cui W., Liu Q., Xing Z., Asiri A. M., Sun X., Adv. Mater.,2016, 28(2), 215—230 |
| 146 | Cheng Z., Huang B., Pi Y., Li L., Shao Q., Huang X., Natl. Sci. Rev.,2020, 7(8), 1340—1348 |
| 147 | Wu G., Zheng X., Cui P., Jiang H., Wang X., Qu Y., Chen W., Lin Y., Li H., Han X., Hu Y., Liu P., Zhang Q., Ge J., Yao Y., Sun R., Wu Y., Gu L., Hong X., Li Y., Nat. Commun.,2019, 10, 4855 |
| 148 | Lum Y., Ager J. W., Nat. Catal.,2019, 2(1), 86—93 |
| 149 | Kuhl K. P., Hatsukade T., Cave E. R., Abram D. N., Kibsgaard J., Jaramillo T. F., J. Am. Chem. Soc.,2014, 136(40), 14107—14113 |
| 150 | Weng Z., Wu Y., Wang M., Jiang J., Yang K., Huo S., Wang X. F., Ma Q., Brudvig G. W., Batista V. S., Liang Y., Feng Z., Wang H., Nat. Commun.,2018, 9, 415 |
| 151 | Duan Y. X., Meng F. L., Liu K. H., Yi S. S., Li S. J., Yan J. M., Jiang Q., Adv. Mater., 2018, 30(14), 1706194 |
| 152 | Loiudice A., Lobaccaro P., Kamali E. A., Thao T., Huang B. H., Ager J. W., Buonsanti R., Angew. Chem. Int. Ed.,2016, 55(19), 5789—5792 |
| 153 | Kuhl K. P., Cave E. R., Abram D. N., Jaramillo T. F., Energy Environ. Sci.,2012, 5(5), 7050—7059 |
| 154 | Li M., Wang H., Luo W., Sherrell P. C., Chen J., Yang J., Adv. Mater.,2020, 32(34), 2001848 |
| 155 | Zhu D. D., Liu J. L., Qiao S. Z., Adv. Mater.,2016, 28(18), 3423—3452 |
| 156 | Sun Z., Ma T., Tao H., Fan Q., Han B., Chem.,2017, 3(4), 560—587 |
| 157 | Lei F., Liu W., Sun Y., Xu J., Liu K., Liang L., Yao T., Pan B., Wei S., Xie Y., Nat. Commun.,2016, 7, 12697 |
| 158 | Han N., Wang Y., Yang H., Deng J., Wu J., Li Y., Li Y., Nat. Commun.,2018, 9, 1320 |
| 159 | Suryanto B. H. R., Du H. L., Wang D., Chen J., Simonov A. N., MacFarlane D. R., Nat. Catal.,2019, 2(4), 290—296 |
| 160 | Xiaoyang C., Cheng T., Qiang Z., Adv. Energy Mater., 2018, 8(22), 1800369 |
| 161 | Geng Z., Liu Y., Kong X., Li P., Li K., Liu Z., Du J., Shu M., Si R., Zeng J., Adv. Mater.,2018, 30(40), 1803498 |
| 162 | Zhou F., Azofra L. M., Ali M., Kar M., Simonov A. N., McDonnell Worth C., Sun C., Zhang X., MacFarlane D. R., Energy Environ. Sci.,2017, 10(12), 2516—2520 |
| 163 | Li L., Tang C., Xia B., Jin H., Zheng Y., Qiao S. Z., ACS Catal.,2019, 9(4), 2902—2908 |
| 164 | Wang L., Zeng Z., Gao W., Maxson T., Raciti D., Giroux M., Pan X., Wang C., Greeley J., Science, 2019, 363(6429), 870—874 |
| 165 | Kong X., Xu K., Zhang C., Dai J., Norooz Oliaee S., Li L., Zeng X., Wu C., Peng Z., ACS Catalysis,2016, 6(3), 1487—1492 |
| 166 | Mahmood A., Lin H., Xie N., Wang X., Chem. Mater.,2017, 29(15), 6329—6335 |
| [1] | 秦永吉, 罗俊. 单原子催化剂在CO2转化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220300. |
| [2] | 姚青, 俞志勇, 黄小青. 单原子催化剂的合成及其能源电催化应用的研究进展[J]. 高等学校化学学报, 2022, 43(9): 20220323. |
| [3] | 范建玲, 唐灏, 秦凤娟, 许文静, 谷鸿飞, 裴加景, 陈文星. 氮掺杂超薄碳纳米片复合铂钌单原子合金催化剂的电化学析氢性能[J]. 高等学校化学学报, 2022, 43(9): 20220366. |
| [4] | 林高鑫, 王家成. 单原子掺杂二硫化钼析氢催化的进展和展望[J]. 高等学校化学学报, 2022, 43(9): 20220321. |
| [5] | 汪思聪, 庞贝贝, 刘潇康, 丁韬, 姚涛. XAFS技术在单原子电催化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220487. |
| [6] | 韩付超, 李福进, 陈良, 贺磊义, 姜玉南, 徐守冬, 张鼎, 其鲁. CoSe2/C复合电催化材料修饰隔膜对高载量锂硫电池性能的影响[J]. 高等学校化学学报, 2022, 43(8): 20220163. |
| [7] | 王茹涵, 贾顺涵, 吴丽敏, 孙晓甫, 韩布兴. CO2参与电化学构筑C—N键制备重要化学品[J]. 高等学校化学学报, 2022, 43(7): 20220395. |
| [8] | 彭奎霖, 李桂林, 江重阳, 曾少娟, 张香平. 电解液调控CO2电催化还原性能微观机制的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220238. |
| [9] | 王丽君, 李欣, 洪崧, 詹新雨, 王迪, 郝磊端, 孙振宇. 调节氧化镉-炭黑界面高效电催化CO2还原生成CO[J]. 高等学校化学学报, 2022, 43(7): 20220317. |
| [10] | 赵润瑶, 纪桂鹏, 刘志敏. 吡咯氮配位单原子铜催化剂的电催化二氧化碳还原性能[J]. 高等学校化学学报, 2022, 43(7): 20220272. |
| [11] | 龚妍熹, 王建兵, 柴歩瑜, 韩元春, 马云飞, 贾超敏. 钾掺杂g-C3N4薄膜光阳极的制备及光电催化氧化降解水中双氯芬酸钠性能[J]. 高等学校化学学报, 2022, 43(6): 20220005. |
| [12] | 杨丽君, 于洋, 张蕾. 双功能2D/3D杂化结构Co2P-CeO x 异质结一体化电极的构筑及电催化尿素氧化辅助制氢性能[J]. 高等学校化学学报, 2022, 43(6): 20220082. |
| [13] | 夏天, 万家炜, 于然波. 异原子配位结构碳基单原子电催化剂结构与性能相关性的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220162. |
| [14] | 黄汉浩, 卢湫阳, 孙明子, 黄勃龙. 石墨炔原子催化剂的崭新道路:基于自验证机器学习方法的筛选策略[J]. 高等学校化学学报, 2022, 43(5): 20220042. |
| [15] | 张宏伟, 陈雯, 赵美淇, 马超, 韩云虎. 单原子催化剂在电化学中的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220129. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||