

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (4): 1167.doi: 10.7503/cjcu20200494
收稿日期:2020-07-27
出版日期:2021-04-10
发布日期:2020-12-14
通讯作者:
郑立炎
E-mail:zhengliyan@ynu.edu.cn
基金资助:
WANG Longjie, FAN Hongchuan, QIN Yu, CAO Qiue, ZHENG Liyan( )
)
Received:2020-07-27
Online:2021-04-10
Published:2020-12-14
Contact:
ZHENG Liyan
E-mail:zhengliyan@ynu.edu.cn
Supported by:摘要:
金属有机框架材料是由金属离子节点和有机配体通过配位键连接形成的具有序多孔骨架的材料, 因其具有比表面积大、 孔隙可调及表面性质可控等优点而备受关注. 通过对有机配体和金属离子进行选择及对金属有机框架材料进行后修饰处理, 可实现对金属有机框架材料表面性质的调控, 以提升其选择性吸附及特异性识别等性能, 进而拓展其在分离分析等领域的应用. 本文从金属有机框架材料的表面性质调控出发, 介绍了其表面性质与分离分析性能的关系, 总结了近年来该领域的代表性工作, 并展望了金属有机框架材料在分离分析领域的应用前景.
中图分类号:
TrendMD:
王隆杰, 范鸿川, 秦渝, 曹秋娥, 郑立炎. 金属有机框架材料在分离分析领域的研究进展. 高等学校化学学报, 2021, 42(4): 1167.
WANG Longjie, FAN Hongchuan, QIN Yu, CAO Qiue, ZHENG Liyan. Research Progress of Metal-organic Frameworks in the Field of Chemical Separation and Analysis. Chem. J. Chinese Universities, 2021, 42(4): 1167.
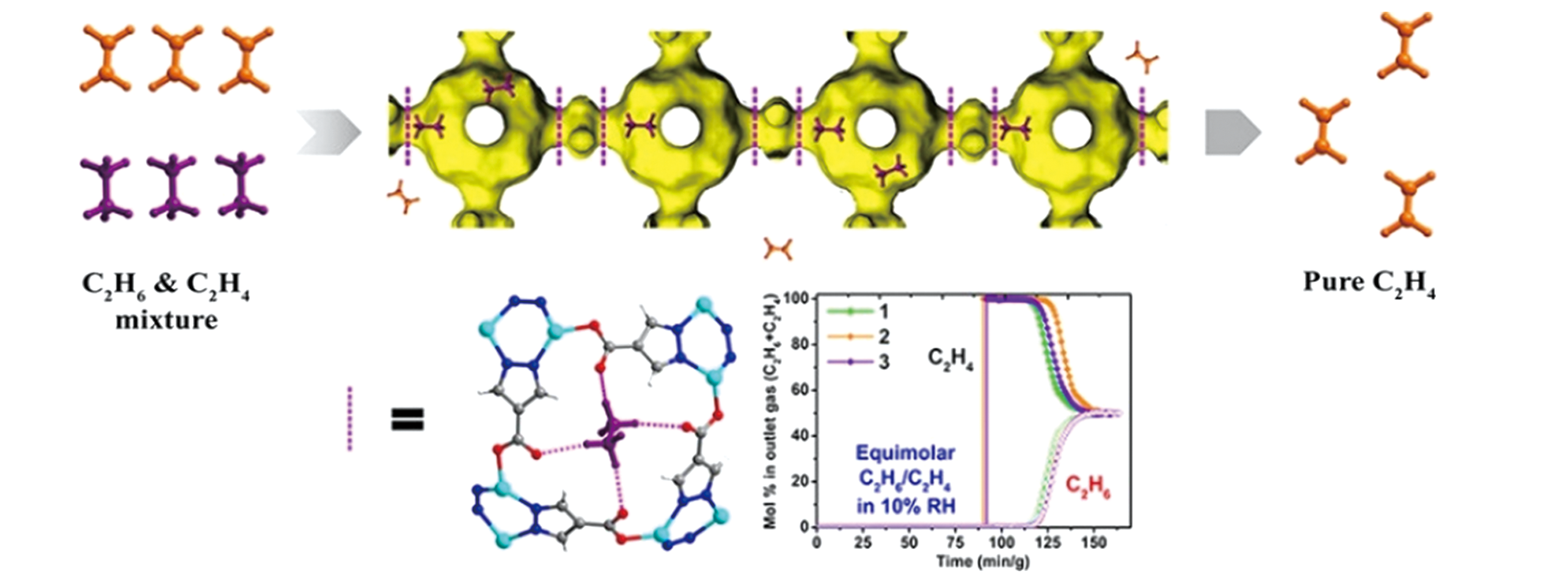
Fig.1 Schematic illustration of the multi?stage apertures in JNU?2(connolly surface)[31]Because of the differences of the host?guest interactions of C2H6 and C2H4 with JNU?2 at the aperture by DFT calculations, weaker hydrogen bonds are generated by C2H4, so it can be separated when a C2H6/C2H4 mixture pass through a packed bed of JNU?2. Zn, turquoise; C, dark grey; N, blue; O, red. Copyright 2019, American Chemical Society.
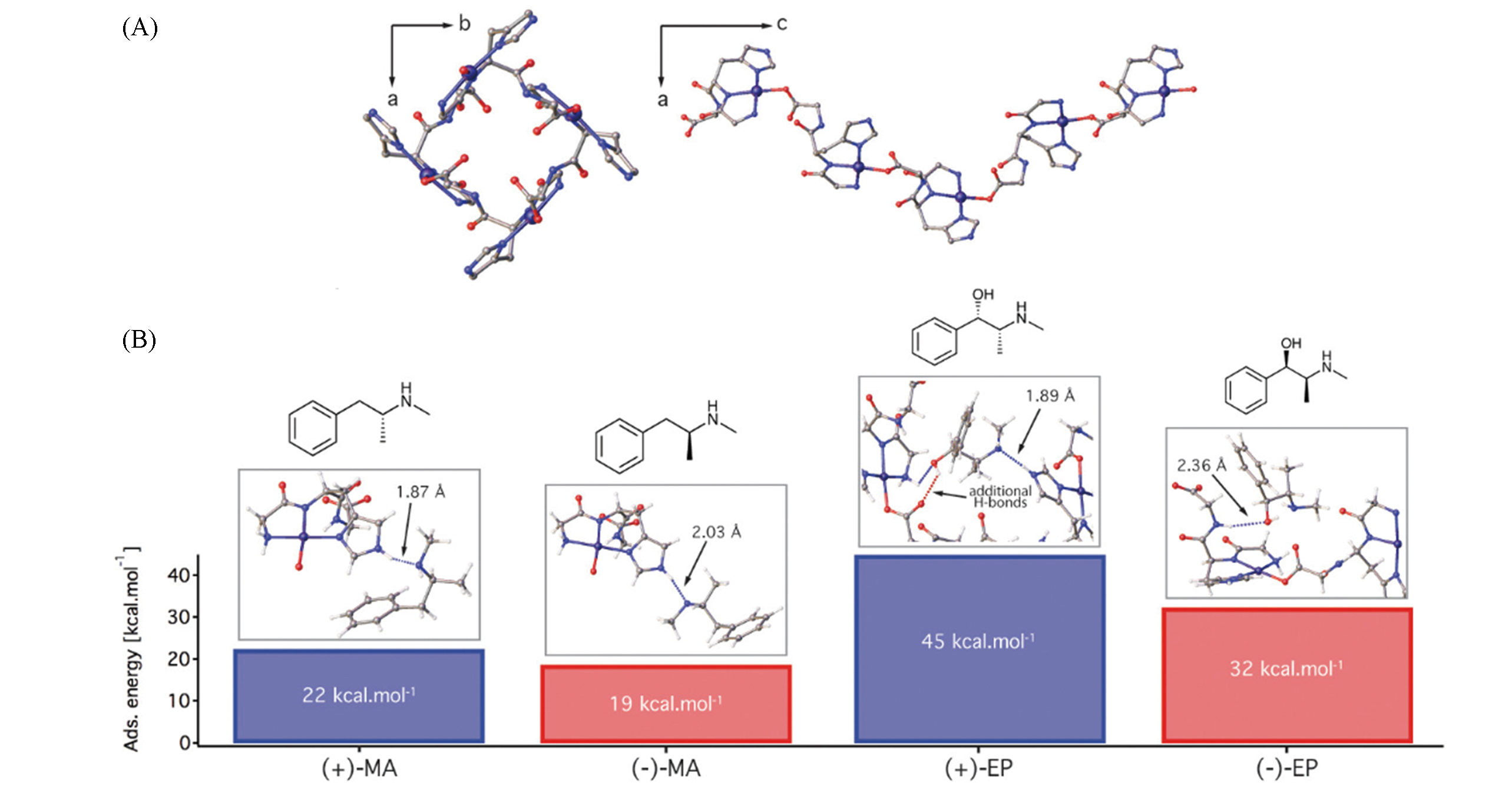
Fig.2 Structure of helicoidal chains in Cu(GHG)(A) and representative MC binding geometries of (±)?MA and EP enantiomers within the structure of Cu(GHG) and corresponding adsorption energies as absolute values calculated with respect to gas phase(B)[36]Dotted lines represent H?bonds with N―H(blue) and O―H(red) donor groups.Copyright 2017, American Chemical Society.
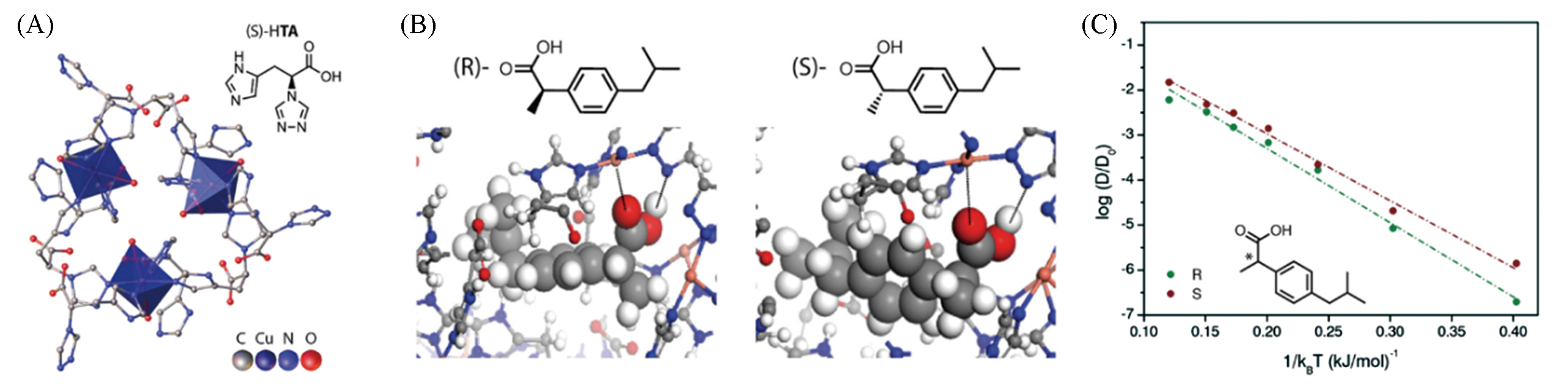
Fig.3 Crystal structure of TAMOF?1 on the [111] plane(A), binding geometries of ibuprofen upon adsorption in TAMOF?1 calculated with Monte Carlo simulations(B) and Arrhenius plot for the diffusion of ibuprofen enantiomers calculated with Molecular Dynamics(C)[37]Copyright 2019, American Chemical Society.
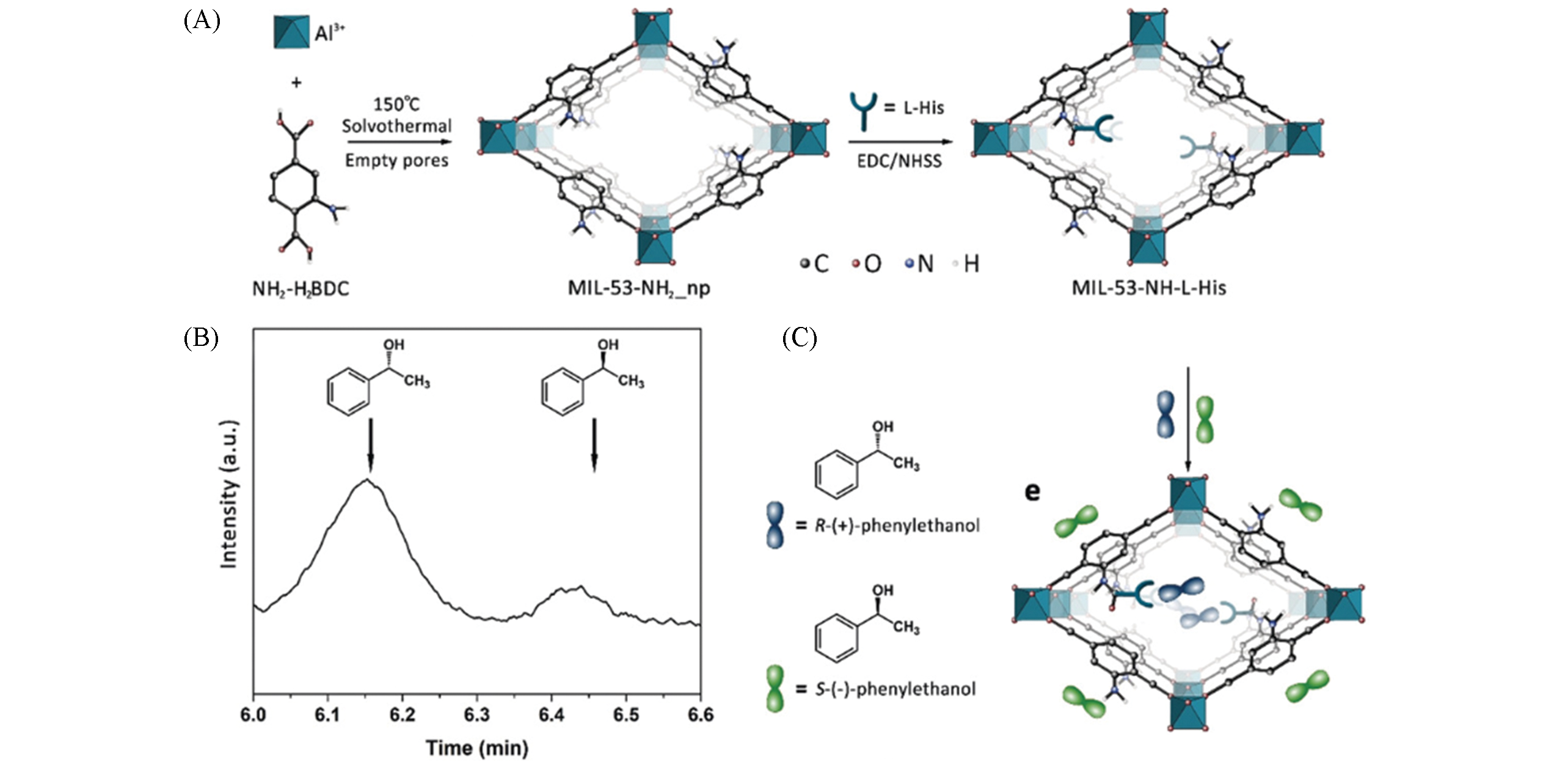
Fig.4 Schematic illustrations of L?histidine?functionalized MIL?53?NH2(A), gas chromatogram result of the (±)?1?phenylethanol adsorbed by MIL?53?NH?L?His nanocrystals(B) and schematic illustration of enantioselective adsorption of MIL?53?NH?L?His nanocrystals for (±)?1?phenylethanol(C)[39]Copyright 2019, Wiley?VCH.
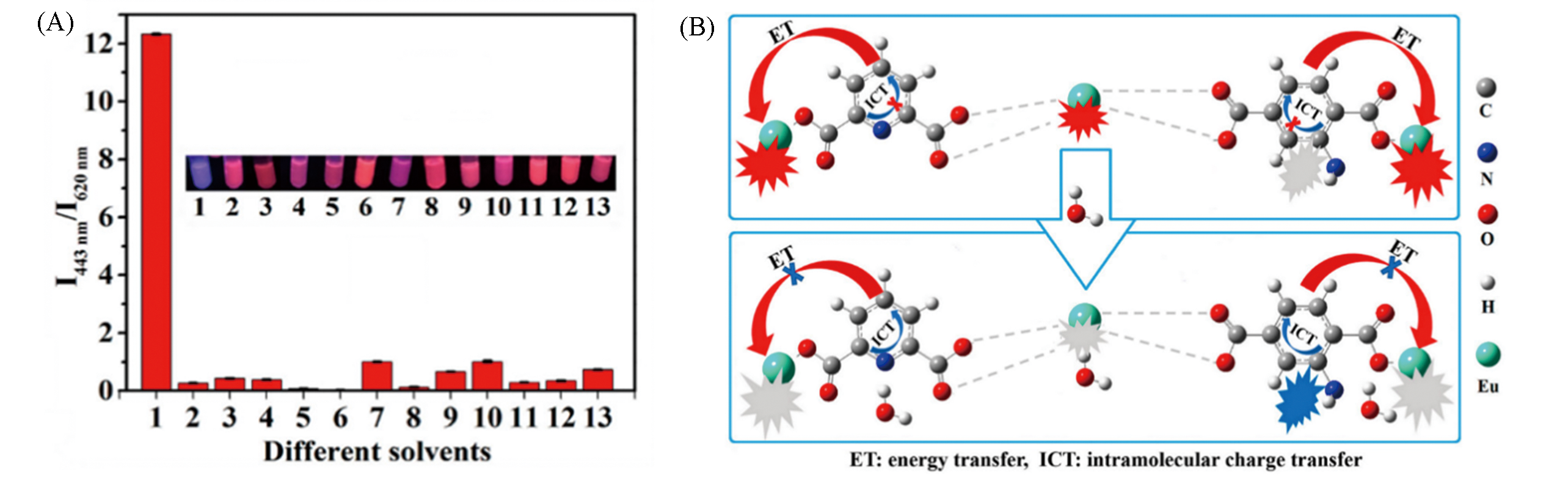
Fig.5 Response selectivity of Eu?DPA/PTA?NH2 to water(A) and water response mechanism of Eu?DPA/PTA?NH2(B)[47](A) 1―13 Represent the Eu?DPA /PTA?NH2 dispersions in water(1), acetonitrile(2), ethanol(3), hexane(4), cyclohexane(5), methanol(6), tetrahydrofuran(7), N?methylpyrrolidone(8), dimethyl sulfoxide(9), ether(10), ethyl acetate(11), isopropanol(12) and N?propanol(13), respectively.Copyright 2019, American Chemical Society.
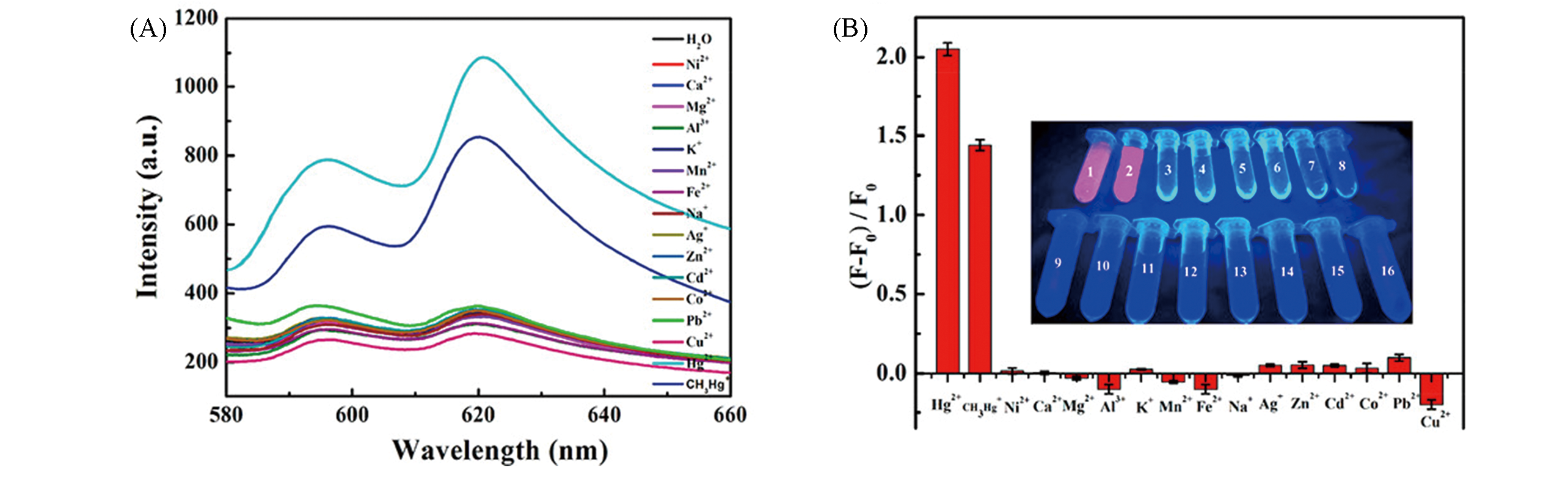
Fig.6 Fluorescence spectra of BA?Eu?MOF toward various metal ions at concentration of 120 μmol/L(A) and the detection system of BA?Eu?MOF in response to 120 μmol/L of different metal ions under UV light at 365 nm(B)[51](B) The white numbers correspond to the ions below from left to right.Copyright 2020, American Chemical Society.
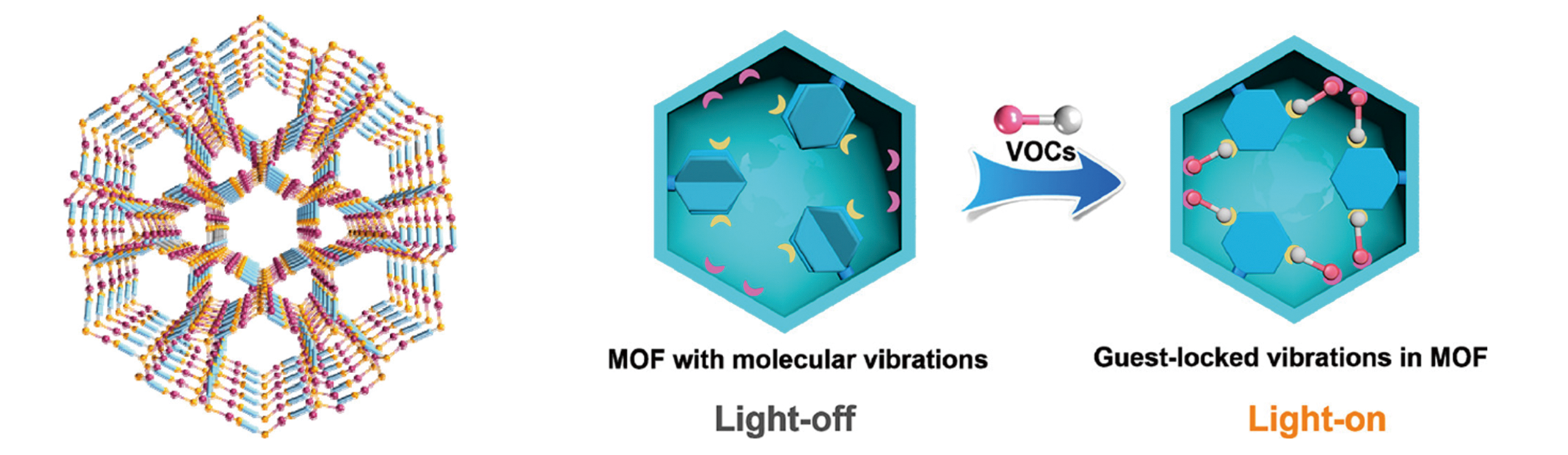
Fig.8 View of the open channels available for guest molecules along the c?axis(A) and model of light?up luminescent detection by a guest?lock process(B)[55]Copyright 2020, American Chemical Society.
| 1 | Robson R., Dalton Trans.,2008, 38, 5113―5131 |
| 2 | Yaghi O. M., Li G., Li H., Nature, 1995, 378, 703 |
| 3 | Meng J., Liu X., Niu C., Pang Q., Li J., Liu F., Liu Z., Mai L., Chem. Soc. Rev.,2020, 49(10), 3142―3186 |
| 4 | Kökçam⁃Demir Ü., Goldman A., Esrafili L., Gharib M., Morsali A., Weingart O., Janiak C., Chem. Soc. Rev.,2020, 49(9), 2751―2798 |
| 5 | Yu M. H., Space B., Franz D., Zhou W., He C., Li L., Krishna R., Chang Z., Li W., Hu T. L., J. Am. Chem. Soc.,2019, 141(44), 17703―17712 |
| 6 | Abdel⁃Mageed A. M., Rungtaweevoranit B., Parlinska⁃Wojtan M., Pei X., Yaghi O. M., Behm R. J., J. Am. Chem. Soc.,2019, 141(13), 5201―5210 |
| 7 | Li L., LI P. F., Wang B., Chem. J. Chinese Universities, 2020, 41(9), 1917―1932(李丽, 李鹏飞, 王博. 高等学校化学学报, 2020, 41(9), 1917―1932) |
| 8 | Hu F. L., Mi Y., Zhu, C., Abrahams B. F., Braunstein P., Lang J. P., Angew. Chem. Int. Ed., 2018, 130(39), 12878―12883 |
| 9 | Shi Y. X., Zhang W. H., Abrahams B. F., Braunstein P., Lang J. P., Angew. Chem. Int. Ed.,2019, 131(28), 9553―9558 |
| 10 | Gu Y., Zheng J. J., Otake K. I., Sugimoto K., Hosono N., Sakaki S., Li F., Kitagawa S., Angew. Chem. Int. Ed.,2020, 132, 15647―15651 |
| 11 | Liu D., Lang J. P., Abrahams B. F., J. Am. Chem. Soc., 2011, 133(29), 11042―11045 |
| 12 | Lang J. P., Xu Q. F., Yuan R. X., Abrahams B. F., Angew. Chem. Int. Ed., 2004, 43(36), 4741―4745 |
| 13 | Wang S. H., Hu H. Z., Chen C., Ma R. N., Zhang N., Chem. J. Chinese Universities, 2014, 35(10),(2055―2060(汪淑华, 胡汉珍, 陈超, 马润宁, 张宁. 高等学校化学学报, 2014, 35(10), 2055―2060) |
| 14 | Farha O. K., Eryazici I., Jeong N. C., Hauser B. G., Wilmer C. E., Sarjeant A. A., Snurr R. Q., Nguyen S. T., Yazaydın A. O. Z. R., Hupp J. T., J. Am. Chem. Soc.,2012, 134(36), 15016―15021 |
| 15 | Böhme U., Barth B., Paula C., Kuhnt A., Schwieger W., Mundstock A., Caro J. R., Hartmann M., Langmuir,2013, 29(27), 8592―8600 |
| 16 | Yang D., Gaggioli C. A., Ray D., Babucci M., Gagliardi L., Gates B. C., J. Am. Chem. Soc.,2020, 142(17), 8044―8056 |
| 17 | Mason J. A., Veenstra M., Long J. R., Chem. Sci.,2014, 5(1), 32―51 |
| 18 | Wang T., Peng Y. L., Lin E., Niu Z., Li P., Ma S., Zhao P., Chen Y., Cheng P., Zhang Z., Inorg. Chem.,2020, 59(7), 4868― 4873 |
| 19 | Yu L., Dong X., Gong Q., Acharya S. R., Lin Y., Wang H., Han Y., Thonhauser T., Li J., J. Am. Chem. Soc.,2020, 142(15), 6925―6929 |
| 20 | Feng L., Wang K. Y., Lv X. L., Powell J. A., Yan T. H., Willman J., Zhou H. C., J. Am. Chem. Soc.,2019, 141(37), 14524―14529 |
| 21 | He J., Xu F., Tian Y., Li C., Hou X., Chem. Commun.,2020, 56, 5803―5806 |
| 22 | He H., Du L., Guo H., An Y., Lu L., Chen Y., Wang Y., Zhong H., Shen J., Wu J., Shuai X., Small, 2020, 16(33), 2001251 |
| 23 | Haddad S., Isabel A. L., Fantham M., Mishra A., Silvestre⁃Albero J., Osterrieth J. W., Gabriele S. S., Kaminski C. F., Forgan R. S., Fairen⁃Jimenez D., J. Am. Chem. Soc.,2020, 142(14), 6661―6674 |
| 24 | Zeng H., Xie M., Huang Y. L., Zhao Y., Xie X. J., Bai J. P., Wan M. Y., Krishna R., Lu W., Li D., Angew. Chem. Int. Ed.,2019, 58(25), 8515―8519 |
| 25 | Ding M., Flaig R. W., Jiang H. L., Yaghi O. M., Chem. Soc. Rev.,2019, 48(10), 2783―2828 |
| 26 | Gao J., Qian X., Lin R. B., Krishna R., Wu H., Zhou W., Chen B., Angew. Chem. Int. Ed.,2020, 59(11), 4396―4400 |
| 27 | Kim E. J., Siegelman R. L., Jiang H. Z., Forse A. C., Lee J. H., Martell J. D., Milner P. J., Falkowski J. M., Neaton J. B., Reimer J. A., Science,2020, 369(6502), 392―396 |
| 28 | Yang S., Ramirez⁃Cuesta A. J., Newby R., Garcia⁃Sakai V., Manuel P., Callear S. K., Campbell S. I., Tang C. C., Schröder M., Nat. Chem.,2015, 7(2), 121―129 |
| 29 | Bloch E. D., Queen W. L., Krishna R., Zadrozny J. M., Brown C. M., Long J. R., Science,2012, 335(6076), 1606―1610 |
| 30 | Li L., Lin R. B., Krishna R., Li H., Xiang S., Wu H., Li J., Zhou W., Chen B., Science,2018, 362(6413), 443―446 |
| 31 | Zeng H., Xie X. J., Xie M., Huang Y. L., Luo D., Wang T., Zhao Y., Lu W., Li D., J. Am. Chem. Soc.,2019, 141(51), 20390―20396 |
| 32 | Maier N. M., Franco P., Lindner W., J. Chromatogr. A,2001, 906, 3―33 |
| 33 | Gübitz G., Schmid M. G., Biopharmaceutics & Drug Disposition, 2001, 22(7/8), 291―336 |
| 34 | Yashima E., J. Chromatogr. A,2001, 906, 105―125 |
| 35 | Das S., Xu S., Ben T., Qiu S., Angew. Chem. Int. Ed.,2018, 57(28), 8629―8633 |
| 36 | Navarro⁃Sánchez J., Argente⁃García A. I., Moliner⁃Martínez Y., Roca⁃Sanjuán D., Antypov D., Campíns⁃Falcó P., Rosseinsky M. J., Martí⁃Gastaldo C., J. Am. Chem. Soc.,2017, 139(12), 4294―4297 |
| 37 | Corella⁃Ochoa M. N., Tapia J. B., Rubin H. N., Lillo V., González⁃Cobos J., Núñez⁃Rico J. L., Balestra S. R., Almora⁃Barrios N., Lledós M., Güell⁃Bara A., J. Am. Chem. Soc.,2019, 141(36), 14306―14316 |
| 38 | Song D., Bae J., Ji H., Kim M. B., Bae Y. S., Park K. S., Moon D., Jeong N. C., J. Am. Chem. Soc.,2019, 141(19), 7853―7864 |
| 39 | Lu Y., Zhang H., Chan J. Y., Ou R., Zhu H., Forsyth M., Marijanovic E. M., Doherty C. M., Marriott P. J., Holl M. M. B., Wang H., Angew. Chem. Int. Ed.,2019, 58(47), 16928―16935 |
| 40 | Maza W. A., Padilla R., Morris A. J., J. Am. Chem. Soc.,2015, 137(25), 8161―8168 |
| 41 | Yin H. Q., Wang X. Y., Yin X. B., J. Am. Chem. Soc.,2019, 141(38), 15166―15173 |
| 42 | Wang Z., Jingjing Q., Wang X., Zhang Z., Chen Y., Huang X., Huang W., Chem. Soc. Rev.,2018, 47(16), 6128―6174 |
| 43 | Han L. J., Kong Y. J., Hou G. Z., Chen H. C., Zhang X. M., Zheng H. G., Inorg. Chem.,2020, 59(10), 7181―7187 |
| 44 | Yuan M., Tang Q., Lu Y., Zhang Z., Li X. H., Liu S. M., Sun X. W., Liu S. X., J. Chem. Educ.,2019, 96(6), 1256―1261 |
| 45 | Liu W., Jiao T., Li Y., Liu Q., Tan M., Wang H., Wang L., J. Am. Chem. Soc.,2004, 126(8), 2280―2281 |
| 46 | Zhao B., Chen X. Y., Cheng P., Liao D. Z., Yan S. P., Jiang Z. H., J. Am. Chem. Soc.,2004, 126(47), 15394―15395 |
| 47 | Yu L., Zheng Q., Wang H., Liu C., Huang X., Xiao Y., Anal. Chem.,2019, 92(1), 1402―1408 |
| 48 | Shustova N. B., Cozzolino A. F., Reineke S., Baldo M., Dincă M., J. Am. Chem. Soc.,2013, 135(36), 13326―13329 |
| 49 | Xiao J., Liu J., Liu M., Ji G., Liu Z., Inorg. Chem.,2019, 58(9), 6167―6174 |
| 50 | Chen M. M., Chen L., Li H. X., Brammer L., Lang J. P., Inorganic Chemistry Frontiers, 2016, 3(10), 1297―1305 |
| 51 | Wang H., Wang X., Liang M., Chen G., Kong R. M., Xia L., Qu F., Anal. Chem.,2020, 92(4), 3366―3372 |
| 52 | Luo J., Xie Z., Lam J. W., Cheng L., Chen H., Qiu C., Kwok H. S., Zhan X., Liu Y., Zhu D., Tang B. Z., Chem. Commun.,2001,(18), 1740―1741 |
| 53 | Lu Z., Wu M., Wu S., Yang S., Li Y., Liu X., Zheng L., Cao Q., Ding Z., Nanoscale,2016, 8(40), 17489―17495 |
| 54 | Dong J., Shen P., Ying S., Li Z. J., Yuan Y. D., Wang Y., Zheng X., Peh S. B., Yuan H., Liu G., Tang B. Z., Chem. Mater., 2020, 32(15), 6706―6720 |
| 55 | Liu C. Y., Chen X. R., Chen H. X., Niu Z., Hirao H., Braunstein P., Lang J. P., J. Am. Chem. Soc.,2020, 142(14), 6690―6697 |
| [1] | 姜宏斌, 代文臣, 张娆, 徐晓晨, 陈捷, 杨光, 杨凤林. Co3O4/UiO-66@α-Al2O3陶瓷膜对VOCs废气的分离催化性能[J]. 高等学校化学学报, 2022, 43(6): 20220025. |
| [2] | 张小玉, 薛冬萍, 杜宇, 蒋粟, 魏一帆, 闫文付, 夏会聪, 张佳楠. MOF衍生碳基电催化剂限域催化O2还原和CO2还原反应[J]. 高等学校化学学报, 2022, 43(3): 20210689. |
| [3] | 李华, 杨科, 黄俊峰, 陈凤娟. UiO-66-NH2/wood的设计构筑及高效去除水中微量重金属离子性能[J]. 高等学校化学学报, 2022, 43(3): 20210701. |
| [4] | 柳雪广, 杨晓珊, 马菁菁, 刘伟生. 铕基金属有机框架材料从混合染料中选择性分离亚甲基蓝[J]. 高等学校化学学报, 2022, 43(1): 20210715. |
| [5] | 张弛, 孙福兴, 朱广山. 双金属同构金属-有机框架材料CAU-21-Al/M的合成、 氮气吸附及复合膜性能[J]. 高等学校化学学报, 2022, 43(1): 20210578. |
| [6] | 王婕, 霍海燕, 王洋, 张仲, 刘术侠. 铜箔上原位合成NENU-n系列多酸基MOFs的通用策略[J]. 高等学校化学学报, 2022, 43(1): 20210557. |
| [7] | 莫宗文, 张学文, 周浩龙, 周东东, 张杰鹏. 一种多孔配位聚合物的氢键协同客体响应[J]. 高等学校化学学报, 2022, 43(1): 20210576. |
| [8] | 李奕川, 朱国富, 王宇, 柴永明, 刘晨光, 何盛宝. 基底表面性质与前驱液化学环境对原位定向构筑钛硅分子筛膜的影响[J]. 高等学校化学学报, 2021, 42(9): 2934. |
| [9] | 常书晴, 辛旭, 黄雅琦, 张信聪, 傅仰河, 朱伟东, 张富民, 李晓娜. Zr基金属有机框架材料的冷热驱动热释电催化性能[J]. 高等学校化学学报, 2021, 42(8): 2558. |
| [10] | 万月, 宋美娜, 赵美廷. 二维金属有机框架纳米片的合成及在超电容和电催化领域的应用[J]. 高等学校化学学报, 2021, 42(2): 575. |
| [11] | 姜沁源, 周晨晖, 蒙海兵, 韩莹, 张如范. 二维金属有机框架材料的合成及电催化应用[J]. 高等学校化学学报, 2021, 42(2): 556. |
| [12] | 吉采灵, 程兴, 谈洁, 袁荃. 功能化核酸适体的筛选及分子识别应用[J]. 高等学校化学学报, 2021, 42(11): 3457. |
| [13] | 张晓荣, 陈岚岚, 胡善文. 基于分子识别的细菌检测研究进展[J]. 高等学校化学学报, 2021, 42(11): 3468. |
| [14] | 林宁钦, 姚克, 陈祥军. 晶状体蛋白识别互作与白内障的研究进展[J]. 高等学校化学学报, 2021, 42(11): 3379. |
| [15] | 黄玲, 庄梓健, 李翔, 石沐玲, 刘高强. 基于核酸适体的外泌体分子识别研究进展[J]. 高等学校化学学报, 2021, 42(11): 3493. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||