

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (5): 932.doi: 10.7503/cjcu20180648
收稿日期:2018-09-21
出版日期:2019-05-06
发布日期:2019-01-09
作者简介:联系人简介: 王惠钢, 男, 博士, 教授, 主要从事极端条件分子间作用力的高分辨拉曼光谱原位在线研究. E-mail:
基金资助:
LIU Qiuna, XU Wenwen, LIU Maozhu, WANG Huigang*( ), ZHENG Xuming
), ZHENG Xuming
Received:2018-09-21
Online:2019-05-06
Published:2019-01-09
Contact:
WANG Huigang
E-mail:zdwhg@163.com
Supported by:摘要:
根据C=O振动的各向同性和各向异性拉曼光谱和红外光谱特点讨论研究了丙酸酐分子的局部有序排列以及振动耦合机理. 利用三级联共聚焦拉曼光谱仪测定了不同浓度丙酸酐的各向同性与各向异性拉曼光谱图, 分别采集了丙酸酐在四氯化碳和甲醇中的光谱以及不同极性溶剂中的光谱, 具体分析了丙酸酐C=O振动模的浓度效应、 溶剂效应以及拉曼光谱非一致效应(NCE). 结果表明, 丙酸酐C=O振动模的NCE效应随着浓度的降低而减小; 随着溶剂极性的减小而增加. 利用密度泛函理论的B3LYP-D3/31-311G(d,p)基组计算了丙酸酐单体和二聚体的几何稳定构型, 用聚集态理论模型解释了丙酸酐分子的NCE效应、 浓度效应与溶剂效应. 理论计算结果与实验结果相吻合.
中图分类号:
TrendMD:
刘秋娜, 许文文, 刘茂祝, 王惠钢, 郑旭明. 高分辨偏正拉曼光谱对丙酸酐C=O振动模的拉曼光谱非一致效应研究. 高等学校化学学报, 2019, 40(5): 932.
LIU Qiuna,XU Wenwen,LIU Maozhu,WANG Huigang,ZHENG Xuming. Study on Raman Spectroscopy Non-coincidence Effect of Propionic Anhydride C=O Vibration Mode†. Chem. J. Chinese Universities, 2019, 40(5): 932.
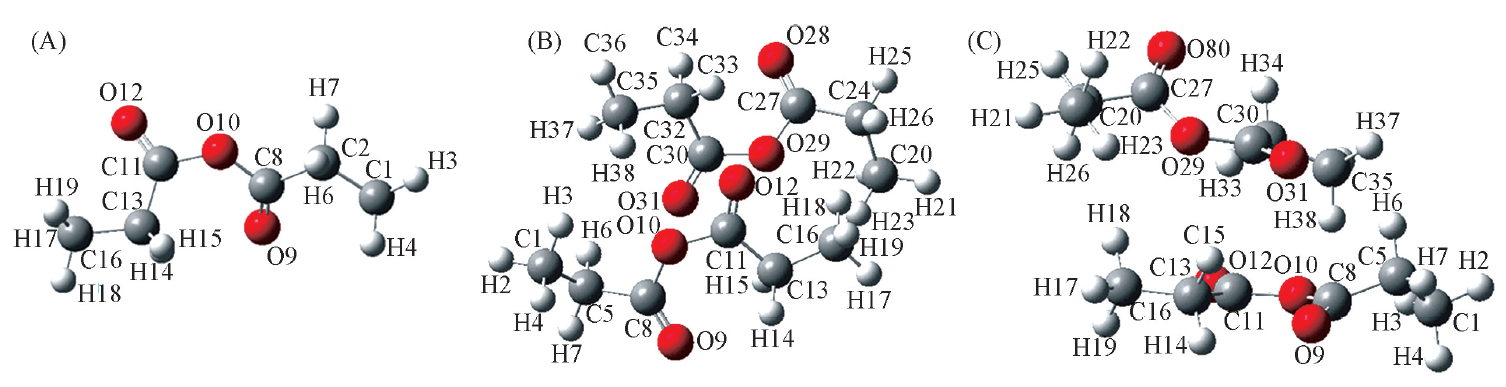
Fig.2 The most stable geometry of propionic anhydride monomer and dimer in the gas phase calculated at the B3LYP/6-311G(d, p) level (A) Monomer; (B) dimer-top view; (C) dimer-side view.
| Mode | Calculated | Experimental | Description | ||||
|---|---|---|---|---|---|---|---|
| Monomer | Dimer | Raman shiftc/ cm-1 | IR, cm-1 | ||||
| Frequency/cm-1 | D. ratio | Frequencya/cm-1 | D. ratiob | ||||
| ν1 | 3119 | 0.74 | 3132/3132 | 0.69/0.75 | Stre(C16H17) | ||
| ν2 | 3115 | 0.75 | 3123/3123 | 0.46/0.75 | Stre(C1H5) | ||
| ν3 | 3112 | 0.70 | 3132/3132 | 0.62/0.75 | Stre(C1H3) | ||
| ν4 | 3112 | 0.68 | 3108/3108 | 0.46/0.75 | Stre(C16H18) | ||
| ν5 | 3087 | 0.62 | 3103/3102 | 0.66/0.75 | Stre(C13H14) | ||
| ν6 | 3066 | 0.73 | 3076/3076 | 0.58/0.75 | Stre(C2H7) | ||
| ν7 | 3044 | 0.06 | 3048/3047 | 0.11/0.57 | Stre(C13H15) | ||
| ν8 | 3044 | 0.01 | 3045/3045 | 0.01/0.75 | Stre(C16H18) | ||
| ν9 | 3043 | 0.20 | 3043/3043 | 0.07/0.75 | Stre(C16H19) | ||
| ν10 | 3039 | 0.07 | 3043/3043 | 0.75/0.09 | Stre(C2H6) | ||
| ν11 | 1871 | 0.18 | 1850/1845 | 0.75/0.04 | Stre(O12C11) | ||
| ν12 | 1825 | 0.19 | 1822/1813 | 0.03/0.75 | 1801vs | 1797w | Stre(O9C8) |
| ν13 | 1504 | 0.75 | 1510/1510 | 0.75/0.75 | Bend(H17C16H19) | ||
| ν14 | 1503 | 0.75 | 1504/1504 | 0.75/0.75 | 1450vs | 1463w | Tore(H4C1C2C8H3C1C2C8) |
| ν15 | 1496 | 0.75 | 1498/1498 | 0.75/0.64 | Bend(H18C16H17) | ||
| ν16 | 1495 | 0.75 | 1495/1495 | 0.75/0.74 | Bend(H4C1H3) | ||
| ν17 | 1461 | 0.75 | 1452/1451 | 0.75/0.75 | Bend(H15C13H14) | ||
| ν18 | 1460 | 0.72 | 1448/1445 | 0.75/0.75 | 1428s | 1423w | Bend(H7C2H6) |
| ν19 | 1424 | 0.36 | 1426/1426 | 0.31/0.75 | Bend(H3C1H5) | ||
| ν20 | 1424 | 0.73 | 1419/1419 | 0.75/0.30 | Bend(H18C16H17) | ||
| ν21 | 1382 | 0.61 | 1387/1385 | 0.75/0.75 | Tors(H7C2C8O10) | ||
| ν22 | 1379 | 0.72 | 1383/1382 | 0.75/0.53 | Tors(H14C13C11O10) | ||
| ν23 | 1293 | 0.70 | 1297/1294 | 0.30/0.75 | 1318s | 1317w | Bend(H14C13H16) |
| ν24 | 1283 | 0.75 | 1293/1292 | 0.75/0.75 | Bend(H6C2H1,) | ||
| ν25 | 1162 | 0.50 | 1158/1157 | 0.75/0.48 | Stre(O10C8) | ||
| ν26 | 1115 | 0.75 | 1119/1119 | 0.75/0.60 | Tors(H14C13C11O10) | ||
| ν27 | 1111 | 0.34 | 1113/1111 | 0.59/0.75 | Tors(H6C2C8O10) | ||
| ν28 | 1106 | 0.44 | 1108/1107 | 0.75/0.62 | TORS(H19C16C13C11) | ||
| Mode | Calculated | Experimental | Description | ||||
| Monomer | Dimer | Raman shiftc/ cm-1 | IR, cm-1 | ||||
| Frequency/cm-1 | D. ratio | Frequencya/cm-1 | D. ratiob | ||||
| ν29 | 1094 | 0.07 | 1101/1091 | 0.75/0.07 | Stre(C1C2) | ||
| ν30 | 1044 | 0.70 | 1058/1040 | 0.75/0.41 | Stre(C13C11) | ||
| ν31 | 1025 | 0.71 | 1032/1020 | 0.75/0.47 | Stre(C1C2) | ||
| ν32 | 1000 | 0.40 | 1003/1002 | 0.75/0.32 | Stre(C16C13) | ||
| ν33 | 905 | 0.31 | 911/911 | 0.75/0.08 | Stre(O10C8) | ||
| ν34 | 824 | 0.61 | 827/827 | 0.75/0.31 | 829vs | Tors(H18C16C13C11) | |
| ν35 | 818 | 0.73 | 822/821 | 0.75/0.63 | Tors(H3C1C2C8) Out(O9C2O10C8) | ||
| ν36 | 775 | 0.12 | 777/776 | 0.06/0.75 | Stre(C11C13) | ||
| ν37 | 679 | 0.61 | 684/684 | 0.53/0.75 | Bend(O9C8O10) | ||
| ν38 | 642 | 0.75 | 636/635 | 0.75/0.75 | Bend(O12C11O10) | ||
| ν39 | 564 | 0.74 | 565/560 | 0.74/0.75 | TORS(C1C2C8O10) | ||
| ν40 | 530 | 0.59 | 534/529 | 0.75/0.40 | TORS(C16C13C11O10) | ||
| ν41 | 444 | 0.74 | 436/436 | 0.75/0.70 | Bend(O12C11O10) | ||
| ν42 | 345 | 0.29 | 347/346 | 0.75/0.22 | Bend(C1C2C8) | ||
| ν43 | 296 | 0.16 | 304/302 | 0.75/0.12 | Bend(C11O10C8) | ||
| ν44 | 251 | 0.75 | 254/249 | 0.71/0.75 | Bend(C1C2O8) | ||
| ν45 | 216 | 0.58 | 231/230 | 0.72/0.75 | TORS(H18C16C13C11) | ||
| ν46 | 204 | 0.67 | 227/223 | 0.75/0.71 | TORS(H3C1C2C8) | ||
| ν47 | 143 | 0.52 | 155/149 | 0.75/0.58 | Bend(C11O10C8) | ||
| ν48 | 100 | 0.69 | 137/127 | 0.75/0.75 | TORS(C11O10C8C2) | ||
| ν49 | 57 | 0.74 | 108/108 | 0.75/0.63 | TORS(C1C2C8O10) | ||
| ν50 | 46 | 0.54 | 106/103 | 0.75/0.53 | TORS(C11O10C8C2) | ||
| ν51 | 19 | 0.72 | 89/80 | 0.75/0.75 | TORS(C13C11O10C8) | ||
| ν52 | 72/64 | 0.74/0.75 | Relative rotation | ||||
| ν53 | 50/27 | 0.71/0.75 | Relative translation | ||||
| ν54 | 25/6.84 | 0.74/0.73 | Relative translation | ||||
Table 1 B3LYP/6-311G(d,p) calculation of the frequency of propionic anhydride monomer and dimer, depolarization ratio(D. ratio)
| Mode | Calculated | Experimental | Description | ||||
|---|---|---|---|---|---|---|---|
| Monomer | Dimer | Raman shiftc/ cm-1 | IR, cm-1 | ||||
| Frequency/cm-1 | D. ratio | Frequencya/cm-1 | D. ratiob | ||||
| ν1 | 3119 | 0.74 | 3132/3132 | 0.69/0.75 | Stre(C16H17) | ||
| ν2 | 3115 | 0.75 | 3123/3123 | 0.46/0.75 | Stre(C1H5) | ||
| ν3 | 3112 | 0.70 | 3132/3132 | 0.62/0.75 | Stre(C1H3) | ||
| ν4 | 3112 | 0.68 | 3108/3108 | 0.46/0.75 | Stre(C16H18) | ||
| ν5 | 3087 | 0.62 | 3103/3102 | 0.66/0.75 | Stre(C13H14) | ||
| ν6 | 3066 | 0.73 | 3076/3076 | 0.58/0.75 | Stre(C2H7) | ||
| ν7 | 3044 | 0.06 | 3048/3047 | 0.11/0.57 | Stre(C13H15) | ||
| ν8 | 3044 | 0.01 | 3045/3045 | 0.01/0.75 | Stre(C16H18) | ||
| ν9 | 3043 | 0.20 | 3043/3043 | 0.07/0.75 | Stre(C16H19) | ||
| ν10 | 3039 | 0.07 | 3043/3043 | 0.75/0.09 | Stre(C2H6) | ||
| ν11 | 1871 | 0.18 | 1850/1845 | 0.75/0.04 | Stre(O12C11) | ||
| ν12 | 1825 | 0.19 | 1822/1813 | 0.03/0.75 | 1801vs | 1797w | Stre(O9C8) |
| ν13 | 1504 | 0.75 | 1510/1510 | 0.75/0.75 | Bend(H17C16H19) | ||
| ν14 | 1503 | 0.75 | 1504/1504 | 0.75/0.75 | 1450vs | 1463w | Tore(H4C1C2C8H3C1C2C8) |
| ν15 | 1496 | 0.75 | 1498/1498 | 0.75/0.64 | Bend(H18C16H17) | ||
| ν16 | 1495 | 0.75 | 1495/1495 | 0.75/0.74 | Bend(H4C1H3) | ||
| ν17 | 1461 | 0.75 | 1452/1451 | 0.75/0.75 | Bend(H15C13H14) | ||
| ν18 | 1460 | 0.72 | 1448/1445 | 0.75/0.75 | 1428s | 1423w | Bend(H7C2H6) |
| ν19 | 1424 | 0.36 | 1426/1426 | 0.31/0.75 | Bend(H3C1H5) | ||
| ν20 | 1424 | 0.73 | 1419/1419 | 0.75/0.30 | Bend(H18C16H17) | ||
| ν21 | 1382 | 0.61 | 1387/1385 | 0.75/0.75 | Tors(H7C2C8O10) | ||
| ν22 | 1379 | 0.72 | 1383/1382 | 0.75/0.53 | Tors(H14C13C11O10) | ||
| ν23 | 1293 | 0.70 | 1297/1294 | 0.30/0.75 | 1318s | 1317w | Bend(H14C13H16) |
| ν24 | 1283 | 0.75 | 1293/1292 | 0.75/0.75 | Bend(H6C2H1,) | ||
| ν25 | 1162 | 0.50 | 1158/1157 | 0.75/0.48 | Stre(O10C8) | ||
| ν26 | 1115 | 0.75 | 1119/1119 | 0.75/0.60 | Tors(H14C13C11O10) | ||
| ν27 | 1111 | 0.34 | 1113/1111 | 0.59/0.75 | Tors(H6C2C8O10) | ||
| ν28 | 1106 | 0.44 | 1108/1107 | 0.75/0.62 | TORS(H19C16C13C11) | ||
| Mode | Calculated | Experimental | Description | ||||
| Monomer | Dimer | Raman shiftc/ cm-1 | IR, cm-1 | ||||
| Frequency/cm-1 | D. ratio | Frequencya/cm-1 | D. ratiob | ||||
| ν29 | 1094 | 0.07 | 1101/1091 | 0.75/0.07 | Stre(C1C2) | ||
| ν30 | 1044 | 0.70 | 1058/1040 | 0.75/0.41 | Stre(C13C11) | ||
| ν31 | 1025 | 0.71 | 1032/1020 | 0.75/0.47 | Stre(C1C2) | ||
| ν32 | 1000 | 0.40 | 1003/1002 | 0.75/0.32 | Stre(C16C13) | ||
| ν33 | 905 | 0.31 | 911/911 | 0.75/0.08 | Stre(O10C8) | ||
| ν34 | 824 | 0.61 | 827/827 | 0.75/0.31 | 829vs | Tors(H18C16C13C11) | |
| ν35 | 818 | 0.73 | 822/821 | 0.75/0.63 | Tors(H3C1C2C8) Out(O9C2O10C8) | ||
| ν36 | 775 | 0.12 | 777/776 | 0.06/0.75 | Stre(C11C13) | ||
| ν37 | 679 | 0.61 | 684/684 | 0.53/0.75 | Bend(O9C8O10) | ||
| ν38 | 642 | 0.75 | 636/635 | 0.75/0.75 | Bend(O12C11O10) | ||
| ν39 | 564 | 0.74 | 565/560 | 0.74/0.75 | TORS(C1C2C8O10) | ||
| ν40 | 530 | 0.59 | 534/529 | 0.75/0.40 | TORS(C16C13C11O10) | ||
| ν41 | 444 | 0.74 | 436/436 | 0.75/0.70 | Bend(O12C11O10) | ||
| ν42 | 345 | 0.29 | 347/346 | 0.75/0.22 | Bend(C1C2C8) | ||
| ν43 | 296 | 0.16 | 304/302 | 0.75/0.12 | Bend(C11O10C8) | ||
| ν44 | 251 | 0.75 | 254/249 | 0.71/0.75 | Bend(C1C2O8) | ||
| ν45 | 216 | 0.58 | 231/230 | 0.72/0.75 | TORS(H18C16C13C11) | ||
| ν46 | 204 | 0.67 | 227/223 | 0.75/0.71 | TORS(H3C1C2C8) | ||
| ν47 | 143 | 0.52 | 155/149 | 0.75/0.58 | Bend(C11O10C8) | ||
| ν48 | 100 | 0.69 | 137/127 | 0.75/0.75 | TORS(C11O10C8C2) | ||
| ν49 | 57 | 0.74 | 108/108 | 0.75/0.63 | TORS(C1C2C8O10) | ||
| ν50 | 46 | 0.54 | 106/103 | 0.75/0.53 | TORS(C11O10C8C2) | ||
| ν51 | 19 | 0.72 | 89/80 | 0.75/0.75 | TORS(C13C11O10C8) | ||
| ν52 | 72/64 | 0.74/0.75 | Relative rotation | ||||
| ν53 | 50/27 | 0.71/0.75 | Relative translation | ||||
| ν54 | 25/6.84 | 0.74/0.73 | Relative translation | ||||
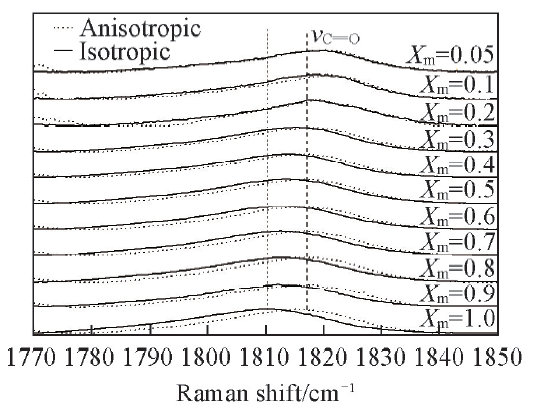
Fig.3 Isotropic and anisotropic partial Raman spectra of propionic anhydride with different volume fractions in CCl4 in a spectral region of 1770—1850 cm-1Xm is volume fraction of propionic anhydride.
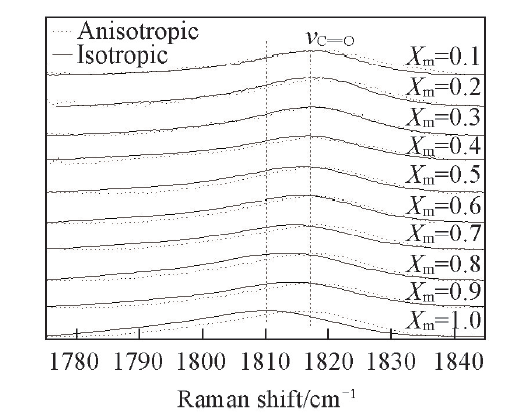
Fig.6 Isotropic and anisotropic partial Raman spectra of propionic anhydride with diffe-rent volume fractions in CH3OH in a spectral region of 1775—1845 cm-1
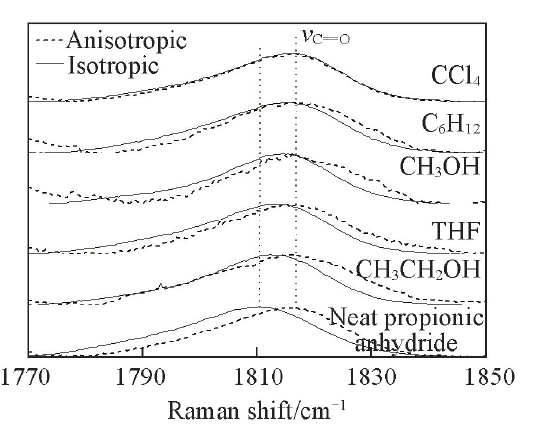
Fig.9 Isotropic and anisotropic partial Raman spectroscopy of C=O in propionic anhydride with a volume fraction of 0.4 in cyclohexane, CCl4, tetrahydrofuran(THF), ethanol and methanol, respectively
| [1] | Urban M., Hobza P., Theoreticachimica Acta., 1975, 36(3), 207—214 |
| [2] | Golding J., Macfarlane D. R., Forsyth M., Retrospective Collection,1998, 25, 146—146 |
| [3] | Pescitelli G., Bari L. D., Berova N., Chemical Society Reviews,2014, 43(15), 5211—5233 |
| [4] | Braga D., Grepioni F., Accounts of Chemical Research,2000, 33(9), 601—608 |
| [5] | Wolters L. P., Schyman P., Pavan M. J., Jorgensen W. L., Bickelhaupt F. M., Kozuch S., Wiley Interdisciplinary Reviews Computational Molecular Science,2014, 4(6), 523—540 |
| [6] | Zordan F., Brammer L., Sherwood P., Journal of America Chemistry Society,2005, 127(16), 5979—5989 |
| [7] | Pihko P. M., Angew. Chem., 2004, 43(6), 2062—2064 |
| [8] | Kosmider L., Sobczak A., Fik M., Knysak J., Zaciera M., Kurek J., Goniewicz L. M., Nicotine & Tobacco Research,2014, 16(10), 1319—1326 |
| [9] | Bekki K., Uchiyama S., Ohta K., Inaba Y., Nakagome H., Kunugita N., International Journal of Environmental Research and Public Health,2014, 11(11), 11192—11200 |
| [10] | Xu Y., Hua E., Chem. J. Chinese Universities,2018, 39(10), 1954—1960 |
| (徐宇, 花儿. 高等学校化学学报,2018, 39(10), 1954—1960) | |
| [11] | Kasyanenko V. M., Keiffer P., Rubtsov I. V., Journal of Chemical Physics,2012, 136(14), 144—503 |
| [12] | Musso M., Torii H., Giorgini M. G., Döge G., Journal of Chemical Physics,1999, 110(20), 10076—10085 |
| [13] | Fini G., Mirone P., Journal of the Chemical Society-Faraday Transactions Ⅱ,1974, 70, 1776—1782 |
| [14] | Giorgini M. G., Musso M., Asenbaum A., Molecular Physics,2000, 98(12), 783—791 |
| [15] | Musso M., Giorgini M. G., Asenbaum G. D., Molecular Physics,1997, 92(1), 97—104 |
| [16] | Upadhyay G., Devi T. G., Singh R. K., Singh A., Alapati P. R., Spectrochimica Acta Part A: Molecular and Biomolecular Spectroscopy,2013, 109(4), 239—246 |
| [17] | Giorgini M. G., Arcioni A., Polizzi C., Musso M., Ottaviani P., Journal of Chemical Physics,2004, 120(10), 4969—4979 |
| [18] | Ojha A. K., Srivastava S. K., Schlücker S., Kiefer W., Asthana B. P., Singh R. K., Journal of Raman Spectroscopy,2010, 38(12), 1656—1664 |
| [19] | Torii M. H., Musso M. G., Giorgini G. D., Molecular Physics,1998, 94(5), 821—828 |
| [20] | Logan D. E., Chemical Physics, 1986, 103(2), 215—225 |
| [21] | Giorgini M. G., Torii H., Musso M., Physical Chemistry Chemical Physics,2010, 12(1), 92—183 |
| [22] | Pádua A. A. H., Gomes M. F. C., Lopes J. N. A. C., Accounts of Chemical Research,2007, 40(11), 1087—1096 |
| [23] | Wu F. Q., Wang H. G., Zheng X. M., Journal of Raman Spectroscopy,2015, 46(6), 591—596 |
| [24] | Xu W. W., Wu F. Q.,, Zhao Y. Y., Zhou R., Wang H. G., Zheng X. M., Ni B., Sci. Rep.,2017, 7, 43835 |
| [25] | Lu T., Chen F., Journal of Computational Chemistry,2012, 33(5), 580—592 |
| [26] | Keresztury G., Holly S., Besenyei G., Varga J., Wang A., Durig J. R., Spectrochimica Acta Part A: Molecular Spectroscopy,1993, 49(13/14), 2007—2026 |
| [27] | Chen L., Yang X. G., Zheng X. M., Spectroscopy and Spectral Analysis,2013, 33(12), 3244—3248 |
| (陈林, 杨晓刚, 郑旭明. 光谱学与光谱分析,2013, 33(12), 3244—3248) | |
| [28] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Montgomery Jr. J. A., Vreven T., Kudin K. N., Burant J. C., Millam J. M., Iyengar S. S., Tomasi J., Barone V., Mennucci B., Cossi M., Scalmain G., Rega N., Petersson G. A., Nakatsuji H., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Klene M., Li X., Knox J. E., Hratchian H. P., Cross J. B., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Ayala P. Y., Morokuma K., Voth G. A., Salvador P., Dannenberg J. J., Zakrzewski V. G., Dapprich S., Daniels A. D., Strain C. M., Farkas O., Malick D. K., Rabuck A. D., Raghavachari K., Foresman J. B., Ortiz J. V., Cui Q., Baboul A. G., Clifford S., Cioslowski J., Stefanov B. B., Lui G., Liashenko A., Piskorz P., Komaromi I., Martin R. L., Fox D. J., Keith T., Peng C. Y., Nanayakkara A., Challacombe M., Gill P. M. W., Johnson B., Chen W., Wong M. W., Gonzalez C., Pople J. A., Gaussian 03, Revision C.02, Gaussian Inc., Pittsburgh, 2003 |
| [29] | Meirovitch H., Vasquez M., Scheraga H. A., Biopolymers,2010, 27(8), 1189—1204 |
| [30] | Tanabe K., Tsuzuki S., Spectrochimica Acta Part A: Molecular Spectroscopy,1986, 42(10), 1197—1199 |
| [31] | Logan D. E., Chemical Physics, 1989, 131(2), 199—207 |
| [1] | 何鸿锐, 夏文生, 张庆红, 万惠霖. 羟基氧化铟团簇与二氧化碳和甲烷作用的密度泛函理论研究[J]. 高等学校化学学报, 2022, 43(8): 20220196. |
| [2] | 黄汉浩, 卢湫阳, 孙明子, 黄勃龙. 石墨炔原子催化剂的崭新道路:基于自验证机器学习方法的筛选策略[J]. 高等学校化学学报, 2022, 43(5): 20220042. |
| [3] | 刘洋, 李旺昌, 张竹霞, 王芳, 杨文静, 郭臻, 崔鹏. Sc3C2@C80与[12]CPP纳米环之间非共价相互作用的理论研究[J]. 高等学校化学学报, 2022, 43(11): 20220457. |
| [4] | 王园月, 安梭梭, 郑旭明, 赵彦英. 5-巯基-1, 3, 4-噻二唑-2-硫酮微溶剂团簇的光谱和理论计算研究[J]. 高等学校化学学报, 2022, 43(10): 20220354. |
| [5] | 周成思, 赵远进, 韩美晨, 杨霞, 刘晨光, 贺爱华. 硅烷类外给电子体对丙烯-丁烯序贯聚合的调控作用[J]. 高等学校化学学报, 2022, 43(10): 20220290. |
| [6] | 程媛媛, 郗碧莹. ·OH自由基引发CH3SSC |
| [7] | 马丽娟, 高升启, 荣祎斐, 贾建峰, 武海顺. Sc, Ti, V修饰B/N掺杂单缺陷石墨烯的储氢研究[J]. 高等学校化学学报, 2021, 42(9): 2842. |
| [8] | 钟声广, 夏文生, 张庆红, 万惠霖. 电中性团簇MCu2Ox(M=Cu2+, Ce4+, Zr4+)上甲烷和二氧化碳直接合成乙酸的理论研究[J]. 高等学校化学学报, 2021, 42(9): 2878. |
| [9] | 黄罗仪, 翁约约, 黄旭慧, 王朝杰. 车前草中黄酮类成分结构和性质的理论研究[J]. 高等学校化学学报, 2021, 42(9): 2752. |
| [10] | 王建, 张红星. 四配位铂磷光发射体结构与光物理性质关系的理论研究[J]. 高等学校化学学报, 2021, 42(7): 2245. |
| [11] | 胡伟, 刘小峰, 李震宇, 杨金龙. 金刚石纳米线氮空位色心的表面与尺寸效应[J]. 高等学校化学学报, 2021, 42(7): 2178. |
| [12] | 杨一莹, 朱荣秀, 张冬菊, 刘成卜. 金催化炔基苯并二𫫇英环化合成8-羟基异香豆素的理论研究[J]. 高等学校化学学报, 2021, 42(7): 2299. |
| [13] | 郑若昕, 张颖, 徐昕. 低标度XYG3双杂化密度泛函的开发与测评[J]. 高等学校化学学报, 2021, 42(7): 2210. |
| [14] | 应富鸣, 计辰儒, 苏培峰, 吴玮. 基于完全活性空间自洽场的杂化多组态密度泛函方法λ-DFCAS[J]. 高等学校化学学报, 2021, 42(7): 2218. |
| [15] | 刘昌辉, 梁国俊, 李妍璐, 程秀凤, 赵显. NH3在硼纳米管表面吸附的密度泛函理论研究[J]. 高等学校化学学报, 2021, 42(7): 2263. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||