

高等学校化学学报 ›› 2015, Vol. 36 ›› Issue (11): 2099.doi: 10.7503/cjcu20150741
收稿日期:2015-09-21
出版日期:2015-11-10
发布日期:2015-10-23
作者简介:联系人简介: 黄富强, 男, 博士, 教授, 主要从事新化合物设计和新能源材料应用. E-mail:基金资助:
ZHU Guilian1,2, LIN Tianquan1, HUANG Fuqiang1,2,*( )
)
Received:2015-09-21
Online:2015-11-10
Published:2015-10-23
Contact:
HUANG Fuqiang
E-mail:huangfq@mail.sic.ac.cn
摘要:
二氧化钛是最重要的光电半导体材料之一, 纳米二氧化钛作为最重要的光催化剂所面临的巨大挑战是无法吸收可见光而导致太阳能利用效率低下. 拓展二氧化钛的光谱响应范围是提高其太阳能利用效率的关键. 最近发展的可以有效吸收可见光的黑色二氧化钛受到广泛关注, 我们针对黑色纳米二氧化钛材料展开了一系列工作, 发展出多种黑色二氧化钛材料的制备方法, 获得了太阳能高吸收、 高效光催化的材料. 本文综合了我们的部分研究工作, 概述了所制备黑色纳米二氧化钛的物化性质、 制备方法及其应用, 并提出了该材料的研究方向.
中图分类号:
TrendMD:
朱桂莲, 林天全, 黄富强. 高性能黑色纳米二氧化钛材料研究. 高等学校化学学报, 2015, 36(11): 2099.
ZHU Guilian, LIN Tianquan, HUANG Fuqiang. Black Nano Titania for Efficient Solar Energy Utilization. Chem. J. Chinese Universities, 2015, 36(11): 2099.
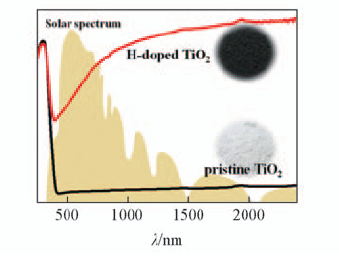
Fig.1 Diffuse reflectance spectra of H-doped black titania(TiO2-xHx) and pristine TiO2(P25, starting material)[26] The backgroud is the total solar spectrum. Copyright from Wiley-VCH.
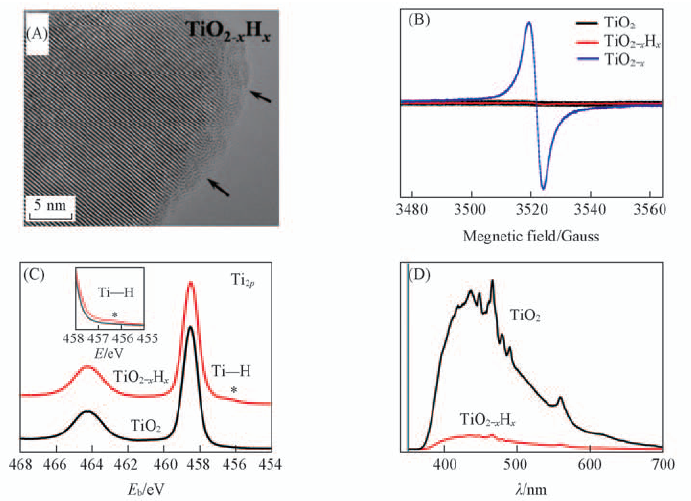
Fig.2 HRTEM micrograph of black TiO2-xHx(A), EPR spectrum(B), Ti2p XPS spectra(C) and photoluminescent spectra(D) of black TiO2-xHx and prisitine TiO2[26] Copyright from Wiley-VCH.
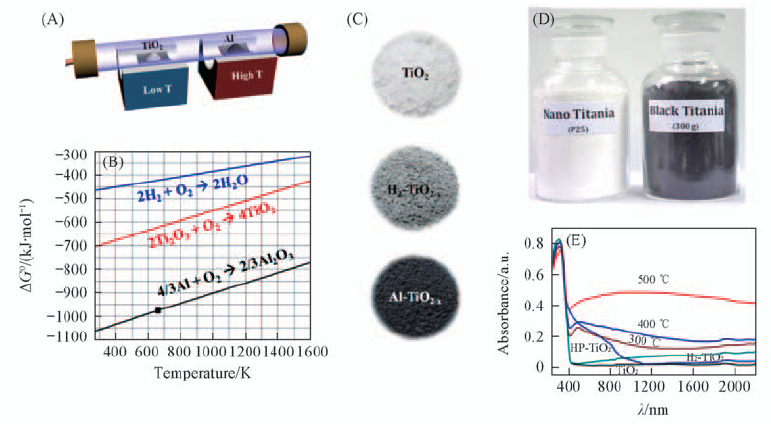
Fig.3 Schematic low-temperature reduction of TiO2 in a two-zone furnace(A), Ellingham diagram of ΔG vs. temperature(B), photographs of pristine TiO2, gray TiO2-x obtained by H2 anneal(H2-TiO2-x), and black TiO2-x obtained by Al reduction(Al-TiO2-x)(C), mass production of black titania(TiO2-x) using our Al-reduction method(D) and absorption spectra of TiO2-x samples reduced at different temperatures, the H2 annealed H2-TiO2-x and high-pressure hydrogenated black titania(HP-TiO2, from ref.[12])(D)[27] Copyright from the Royal Society of Chemistry.
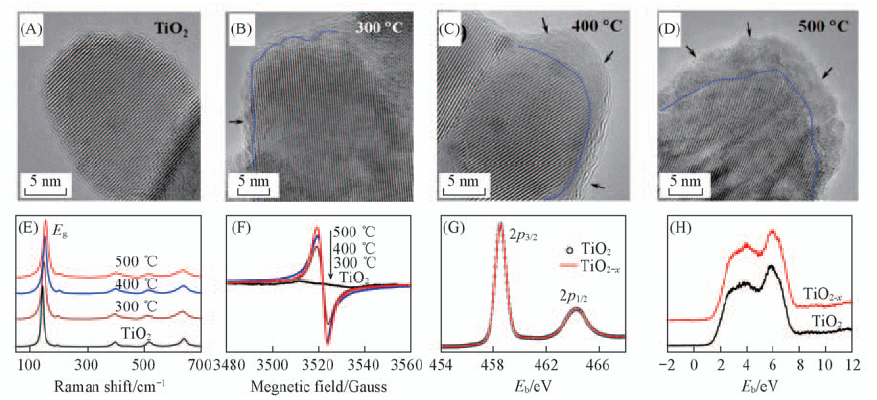
Fig.4 HRTEM images of TiO2 nanocrystals before(A) and after(B—D) Al reduction at different temperatures for 6 h, Raman spectra(E), EPR spectra of black TiO2-x and pristine TiO2 as a reference sample(F), Ti2p XPS spectra(G) and XPS valence band spectra(H) of pristine TiO2 and TiO2-x[27] Copyright from the Royal Society of Chemistry.
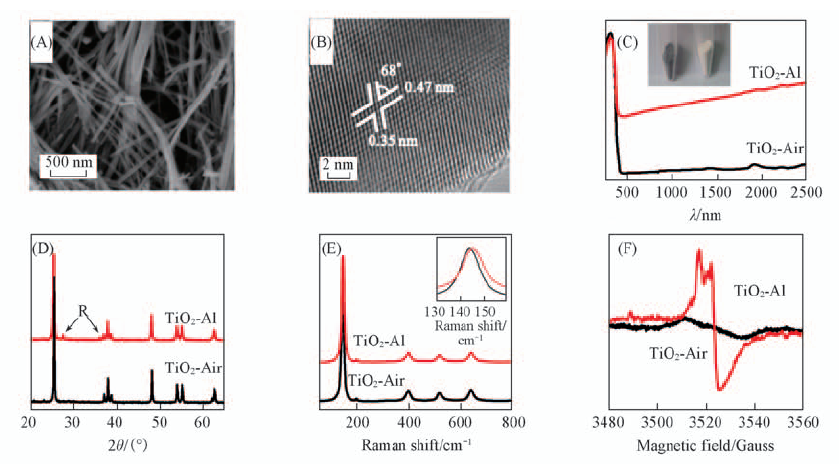
Fig.5 SEM(A), HRTEM images of TiO2-Al(B), UV-Vis absorption spectra(C), XRD patterns(D), Raman spectra(E) and EPR spectra(F) of TiO2-Al and TiO2-air nanowires[37] Copyright from Wiley-VCH.
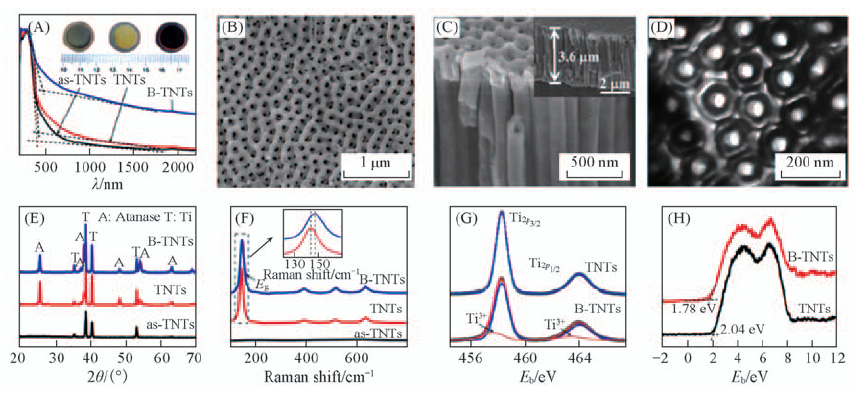
Fig.6 Absorption spectra and photographs(inset) of as-TNTs, TNTs, and B-TNTs(A), typical top view FE-SEM of B-TNTs(B), sideview FE-SEM of B-TNTs(inset is the full side view)(C), typical top view of B-TNTS(D), XRD patterns(E) and Raman spectra of TNTs with different annealing treatment(inset is the magnification of Eg peak)(F), Ti2p XPS(G) and XPS valence band spectra(H) of TNTs and B-TNTs[29]
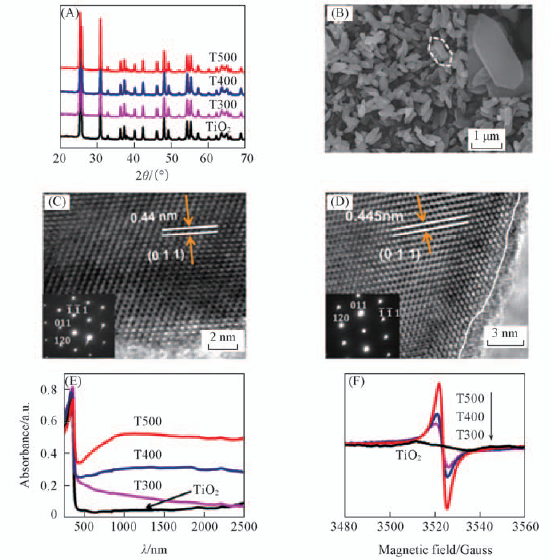
Fig.7 XRD patterns of pristine TiO2 and black TiO2-x after Al reduction at different temperatures for 4 h(A), SEM images of synthesized TiO2(B), HRTEM images of TiO2(C), T500(D), UV-Vis-NIR diffuse reflectance(E) and EPR spectra(F) of the pristine TiO2 and the TiO2-x after Al reduction at different temperatures for 4 h[32] Copyright from the Royal Society of Chemistry.
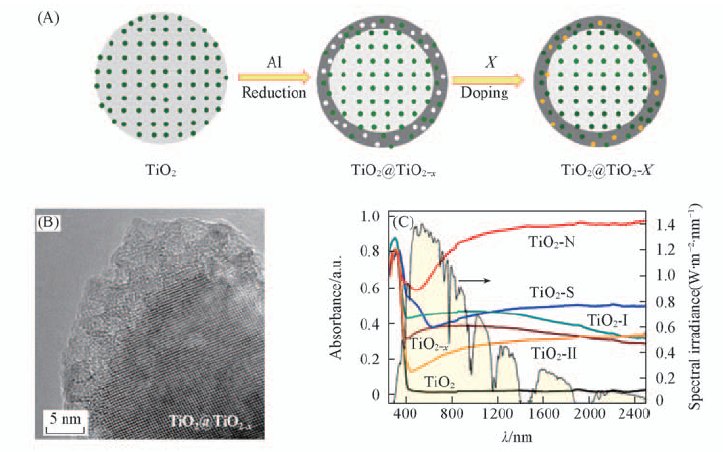
Fig.9 Schematic core/shell structures of TiO2@TiO2-x(denoted as TiO2-x) and TiO2@TiO2-X(denoted as TiO2-X) with Ti4+(oxygen vacancies and X sites in green, white and orange, respectively)(A), HRTEM image of black TiO2-x nanocrystals(B) and diffuse reflectance spectra of TiO2-X(X=H, N, S, I) and solar spectral irradiance(right)(C)[30] Copyright from the Royal Society of Chemistry.
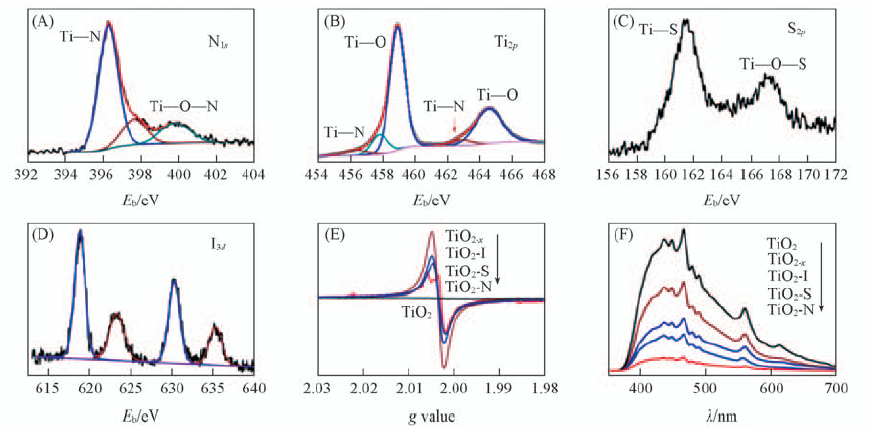
Fig.10 High resolution XPS spectra of N1s and Ti2p of TiO2-N(A, B), S2pXPS spectrum of TiO2-S(C), I3d XPS spectrum acquired from TiO2-I(D), EPR spectra(E) and PL spectra(F) of TiO2-x, TiO2-I, TiO2-N, TiO2-S and pristine TiO2[30] Copyright from the Royal Society of Chemistry.
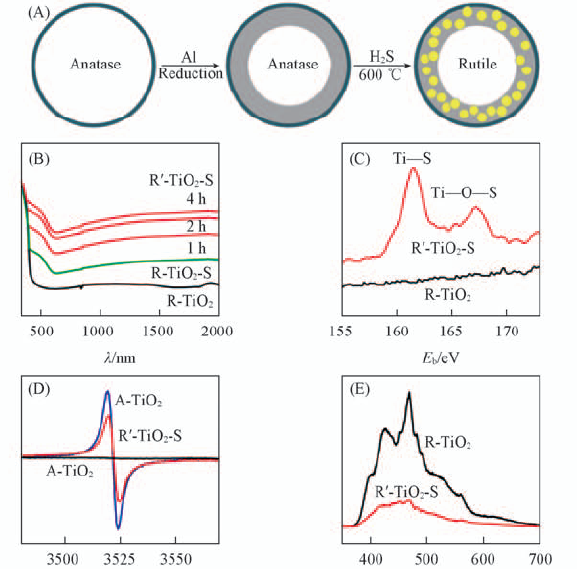
Fig.11 Schematic synthetic route of rutile TiO2 with sulfided surface(R'-TiO2-S)(A), diffuse reflectance spectra of R-TiO2, R-TiO2-S and R'-TiO2-S with different sulfidation time(B), S2p XPS spectra of R'-TiO2-S and R-TiO2(C), EPR spectra of R'-TiO2-S, A-TiO2-x and pristine A-TiO2(D) and photoluminescent spectra of R'-TiO2-S and R-TiO2(E)[28] Copyright from the American Chemical Society.
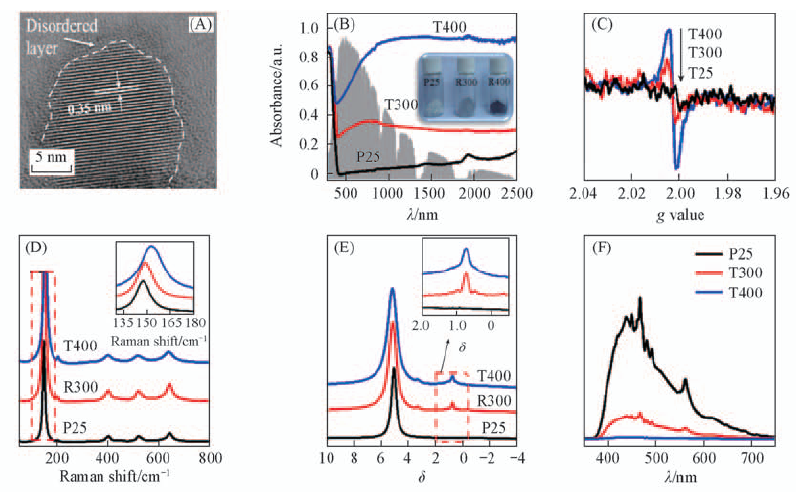
Fig.12 HRTEM image of T400(A), UV-Vis absorption spectra(B), EPR spectra(C), Raman spectra(D), 1H NMR spectra(E) and PL spectra(F) of reference P25 and the reduced samples[31] The insets in (D) and (E) are partial magnification of the marked region with a red border. Copyright from Wiley-VCH.
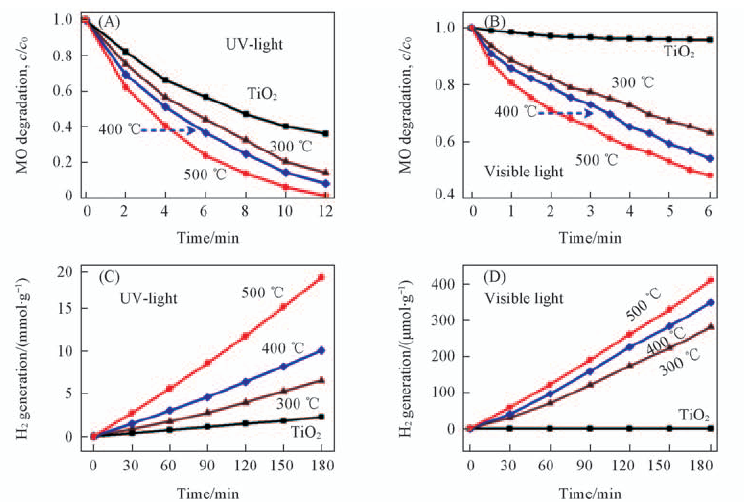
Fig.13 Evaluation of the photocatalytic activities of TiO2-x[27] (A) UV light photo-catalytic degradation of methyl orange; (B) visible light photocatalytic degradation of methyl orange; (C) H2 generation of black TiO2-x under UV light; (D) visible light irradiation. Copyright from the Royal Society of Chemistry.
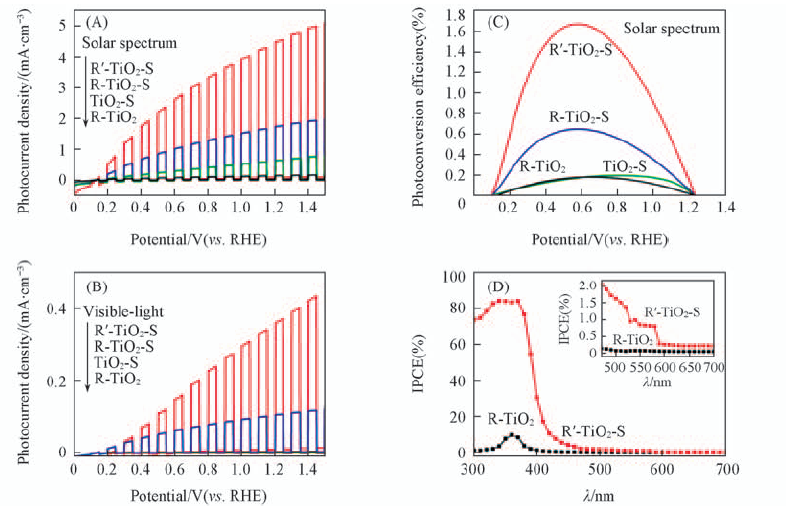
Fig.15 Photoelectrochemical properties of R'-TiO2-S, R-TiO2-S, TiO2-S, and R-TiO2 electrodes (A) Chopped J-V curves under simulated solar light illumination; (B) visible-light illumination via a three-electrode setup(TiO2 working, Pt counter, Ag/AgCl reference electrode, scan rate of 10 mV/s) in 1 mol/L NaOH electrolyte(pH=13.6); (C) photoconversion efficiencies as a function of applied potential; (D) IPCE spectra in the region of 300—700 nm at 0.65 VRHE. Inset of (D) IPCE spectra in the region of 420—700 nm[28]. Copyright from the American Chemical Society.
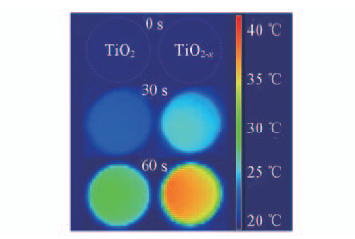
Fig.16 Thermal image map of cool-pressed disks after irradiation under AM 1.5 G Xe lamp solar simulator for different time[27] Copyright from the Royal Society of Chemistry.
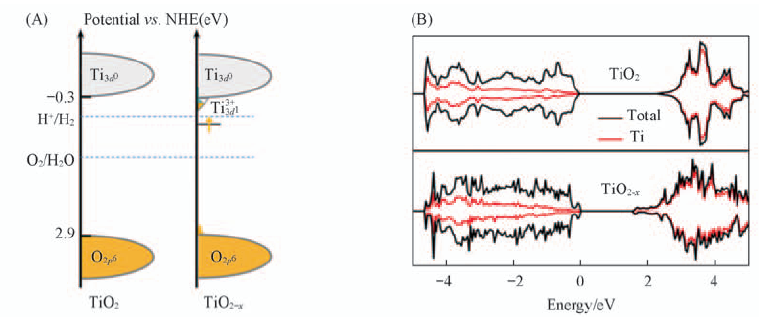
Fig.17 Schematic electronic structures for TiO2 and TiO2-x(A) and total DOS and partial DOS(Ti) for TiO2 and TiO2-x(B)[27] Copyright from the Royal Society of Chemistry.
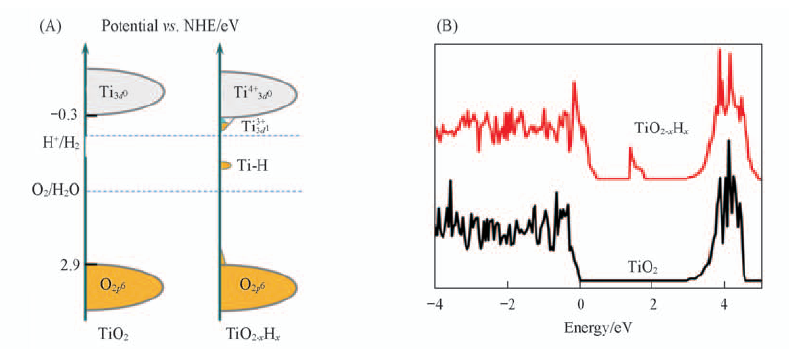
Fig.18 Schematic electronic structures for hydrogenated titania(TiO2-xHx) and pristine TiO2(A) and calculated DOS of pristine TiO2 and black TiO2-xHx(B)[26] Copyright from Wiley-VCH.
| [1] | Kubacka A., Fernandez-Garcia M., Colon G., Chem. Rev., 2012, 112, 1555—1614 |
| [2] | Kudo A., Miseki Y., Chem. Soc. Rev., 2009, 38, 253—278 |
| [3] | Chen X., Mao S. S., Chem. Rev., 2007, 107, 2891—2959 |
| [4] | Lü X., Mou X., Wu J., Zhang D., Zhang L., Huang F., Xu F., Huang S., Adv. Funct. Mater., 2010, 20, 509—515 |
| [5] | Lu X., Huang F., Mou X., Wang Y., Xu F., Adv. Mater., 2010, 22, 3719—3722 |
| [6] | Gratzel M., Nature, 2001, 414, 338—344 |
| [7] | Batzill M., Morales E., Diebold U., Phys. Rev. Lett., 2006, 96, 02613 |
| [8] | Subramanian V., Wolf E. E., Kamat P. V., Langmuir, 2003, 19, 469—474 |
| [9] | Schneider J., Matsuoka M., Takeuchi M., Zhang J., Horiuchi Y., Anpo M., Bahnemann D. W., Chem. Rev., 2014, 114, 9919—9986 |
| [10] | Zhou H., Qu Y., Zeid T., Duan X., Energy Environ. Sci., 2012, 5, 6732—6743 |
| [11] | Yi Z., Ye J., Kikugawa N., Kako T., Ouyang S., Stuart-Williams H., Yang H., Cao J., Luo W., Li Z., Liu Y., Withers R. L., Nat. Mater., 2010, 9, 559—564 |
| [12] | Chen X., Liu L., Yu P. Y., Mao S. S., Science, 2011, 331, 746—750 |
| [13] | Hoang S., Berglund S. P., Hahn N. T., Bard A. J., Mullins C. B., J. Am. Chem. Soc., 2012, 134, 3659—3662 |
| [14] | Jiang X., Zhang Y., Jiang J., Rong Y., Wang Y., Wu Y., Pan C., J. Phys. Chem. C, 2012, 116, 22619—22624 |
| [15] | Liu L., Yu P. Y., Chen X., Mao S. S., Shen D. Z., Phys. Rev. Lett., 2013, 111, 0065505 |
| [16] | Yang Y., Ling Y. C., Wang G. M., Li Y., Eur. J. Inorg. Chem., 2014, 4, 760—766 |
| [17] | Zhu W. D., Wang C. W., Chen J. B., Li D. S., Zhou F., Zhang H. L., Nanotechnology, 2012, 23, 455204—455209 |
| [18] | Xia T., Zhang C., Oyler N. A., Chen X. B., Adv. Mater., 2013, 25, 6905—6910 |
| [19] | Chen X., Liu L., Huang F., Chem. Soc. Rev., 2015, 46, 1861—1885 |
| [20] | Zheng Z., Huang B., Lu J., Wang Z., Qin X., Zhang X., Dai Y., Whangbo M. H., Chem. Commun., 2012, 48, 5733—5735 |
| [21] | Lu J., Dai Y., Jin H., Huang B., Phys. Chem. Chem. Phys., 2011, 13, 18063—18068 |
| [22] | Wang G., Wang H., Ling Y., Tang Y., Yang X., Fitzmorris R. C., Wang C., Zhang J. Z., Li Y., Nano Lett., 2011, 11, 3026—3033 |
| [23] | Wei W., Yaru N., Chunhua L., Zhongzi X., RSC Adv., 2012, 2, 8286—8288 |
| [24] | Leshuk T., Parviz R., Everett P., Krishnakumar H., Varin R. A., Gu F., ACS Appl. Mater. Interfaces, 2013, 5, 1892—1895 |
| [25] | Zhang S., Zhang S., Peng B., Wang H., Yu H., Wang H., Peng F., Electrochem. Commun., 2014, 40, 24—27 |
| [26] | Wang Z., Yang C., Lin T., Yin H., Chen P., Wan D., Xu F., Huang F., Lin J., Xie X., Jiang M., Adv. Funct. Mater., 2013, 23, 5444—5450 |
| [27] | Wang Z., Yang C., Lin T., Yin H., Chen P., Wan D., Xu F., Huang F., Lin J., Xie X., Jiang M., Energy Environ. Sci., 2013, 6, 3007—3014 |
| [28] | Yang C., Wang Z., Lin T., Yin H., Lu X., Wan D., Xu T., Zheng C., Lin J., Huang F., Xie X., Jiang M., J. Am. Chem. Soc., 2013, 135, 17831—17838 |
| [29] | Cui H., Zhao W., Yang C., Yin H., Lin T., Shan Y., Xie Y., Gu H., Huang F., J. Mater. Chem. A, 2014, 2, 8612—8616 |
| [30] | Lin T. Q., Yang C. Y., Wang Z., Yin H., Lu X. J., Huang F. Q., Lin J. H., Xie X. M., Jiang M. H., Energy Environ. Sci., 2014, 7, 967—972 |
| [31] | Zhu G., Yin H., Yang C., Cui H., Wang Z., Xu J., Lin T., Huang F., ChemCatChem, 2015, 7, 2614—2619 |
| [32] | Zhu G., Lin T., Lü X., Zhao W., Yang C., Wang Z., Yin H., Liu Z., Huang F., Lin J., J. Mater. Chem. A, 2013, 1, 9650—9653 |
| [33] | Yu J., Dai G., Huang B., J. Phys. Chem. C, 2009, 113, 16394—16401 |
| [34] | Yu J., Tao H., Cheng B., ChemPhysChem, 2010, 11, 1617—1618 |
| [35] | Prokes S. M., Gole J. L., Chen X., Burda C., Carlos W. E., Adv. Funct. Mater., 2005, 15, 161—167 |
| [36] | Lazarus M. S., Sham T. K., Chem. Phys. Lett., 1982, 92, 670—674 |
| [37] | Yin H., Lin T., Yang C., Wang Z., Zhu G., Xu T., Xie X., Huang F., Jiang M., Chemistry Eur. J., 2013, 19, 13313—13316 |
| [38] | Iskandar F., Nandiyanto A. B. D., Yun K. M., Hogan C. J., Okuyama K., Biswas P., Adv. Mater., 2007, 19, 1408—1412 |
| [39] | Kandiel T.A., Feldhoff A., Robben L., Dillert R., Bahnemann D. W., Chem. Mater., 2010, 22, 2050—2060 |
| [40] | Zhao W., Zhao W., Zhu G., Lin T., Xu F., Huang F., CrystEngComm, 2015, 17, 7528—7534 |
| [41] | Henderson M. A., Epling W. S., Peden C. H. F., Perkins C. L., J. Phys. Chem. B, 2003, 107, 534—545 |
| [42] | Finazzi E., Di Valentin C., Pacchioni G., Selloni A., J. Chem. Phys., 2008, 129, 154113 |
| [43] | Tojo S., Tachikawa T., Fujitsuka M., Majima T., J. Phys. Chem. C, 2008, 112, 14948—14954 |
| [44] | Morgan W. E., van Wazer J. R., Stec W. J., J. Am. Chem. Soc., 1973, 95, 751—755 |
| [45] | Wang J., Tafen D. N., Lewis J. P., Hong Z., Manivannan A., Zhi M., Li M., Wu N., J. Am. Chem. Soc., 2009, 131, 12290—12297 |
| [1] | 秦永吉, 罗俊. 单原子催化剂在CO2转化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220300. |
| [2] | 林治, 彭志明, 贺韦清, 沈少华. 单原子与团簇光催化: 竞争与协同[J]. 高等学校化学学报, 2022, 43(9): 20220312. |
| [3] | 滕镇远, 张启涛, 苏陈良. 聚合物单原子光催化剂的载流子分离和表面反应机制[J]. 高等学校化学学报, 2022, 43(9): 20220325. |
| [4] | 刘苏毓, 丁飞, 李茜, 樊春海, 冯景. 偶氮苯类DNA纳米机器[J]. 高等学校化学学报, 2022, 43(8): 20220122. |
| [5] | 邱丽琪, 姚向阳, 何良年. 可见光驱动丰产金属卟啉类配合物催化的二氧化碳选择性还原反应[J]. 高等学校化学学报, 2022, 43(7): 20220064. |
| [6] | 赵盈喆, 张建玲. 金属-有机框架基材料在二氧化碳光催化转化中的应用[J]. 高等学校化学学报, 2022, 43(7): 20220223. |
| [7] | 张振, 邓煜, 张琴芳, 余达刚. 可见光促进二氧化碳参与的羧基化反应[J]. 高等学校化学学报, 2022, 43(7): 20220255. |
| [8] | 夏雾, 任颖异, 刘京, 王锋. 壳聚糖包裹CdSe量子点组装体的水相可见光催化CO2还原[J]. 高等学校化学学报, 2022, 43(7): 20220192. |
| [9] | 龚妍熹, 王建兵, 柴歩瑜, 韩元春, 马云飞, 贾超敏. 钾掺杂g-C3N4薄膜光阳极的制备及光电催化氧化降解水中双氯芬酸钠性能[J]. 高等学校化学学报, 2022, 43(6): 20220005. |
| [10] | 王广琦, 毕艺洋, 王嘉博, 石洪飞, 刘群, 张钰. 非贵金属三元复合Ni(PO3)2-Ni2P/CdS NPs异质结的构建及可见光高效催化产氢性能[J]. 高等学校化学学报, 2022, 43(6): 20220050. |
| [11] | 宋颖颖, 黄琳, 李庆森, 陈立妙. CuO/BiVO4光催化剂的制备及光催化CO2还原性能[J]. 高等学校化学学报, 2022, 43(6): 20220126. |
| [12] | 陶雨, 欧鸿辉, 雷永鹏, 熊禹. 单原子催化剂在光催化二氧化碳还原中的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220143. |
| [13] | 冯丽, 邵兰兴, 李思骏, 全文选, 庄金亮. 超薄Sm-MOF纳米片的合成及可见光催化降解芥子气模拟剂性能[J]. 高等学校化学学报, 2022, 43(4): 20210867. |
| [14] | 孟祥钰, 詹琦, 武亚南, 马晓双, 姜靖逸, 孙岳明, 代云茜. 光热效应增强的Au/RGO/Na2Ti3O7光催化加氢性能[J]. 高等学校化学学报, 2022, 43(3): 20210655. |
| [15] | 郭彪, 赵晨灿, 刘芯辛, 于洲, 周丽景, 袁宏明, 赵震. 表面水热碳层对磁性NiFe2O4八面体光催化活性的影响[J]. 高等学校化学学报, 2022, 43(11): 20220472. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||