

高等学校化学学报 ›› 2015, Vol. 36 ›› Issue (11): 2115.doi: 10.7503/cjcu20150601
收稿日期:2015-07-31
出版日期:2015-11-10
发布日期:2015-10-13
作者简介:联系人简介: 麥松威, 男, 博士, 教授, 博士生导师, 中国科学院院士, 主要从事结构化学、 无机合成、 超分子自组装及晶体工程研究. E-mail:基金资助:
Sam S. K. HAU1,2, Ting HU1,3, Dennis Y. S. TAM1, Thomas C. W. MAK1,*( )
)
Received:2015-07-31
Online:2015-11-10
Published:2015-10-13
Contact:
Thomas C. W. MAK
E-mail:tcwmak@cuhk.edu.hk
摘要:
总结了我们在新型炔银簇化合物研究方面的最新进展. 这些化合物分为以下几种类型: (a)1,3,5-己三炔基和1,3,5,7-辛四炔基; (b)1,5-己二炔基; (c)经膦酸配体组装的乙炔基和烷基乙炔基; (d)苯乙炔基、 环烷基乙炔基和含氮杂环基乙炔基结构单元的银簇化合物; (e)经不同类型的银-碳配位键连接, 并进一步通过分子内/分子间作用力稳固其配位网络的炔银化合物. 我们还进一步讨论了溶剂、 配体的位阻大小和辅助配体类型对于多维配位网络结构的影响.
中图分类号:
TrendMD:
侯俊傑, 胡婷, 譚耀新, 麥松威. 含全碳和多炔阴离子配体的银簇化合物的超分子组装及其结构研究. 高等学校化学学报, 2015, 36(11): 2115.
Sam S. K. HAU, Ting HU, Dennis Y. S. TAM, Thomas C. W. MAK. Assembly of Organosilver(I) Networks with Multinuclear Supramolecular Synthons Containing All-carbon and Carbon-rich Anionic Ligands. Chem. J. Chinese Universities, 2015, 36(11): 2115.
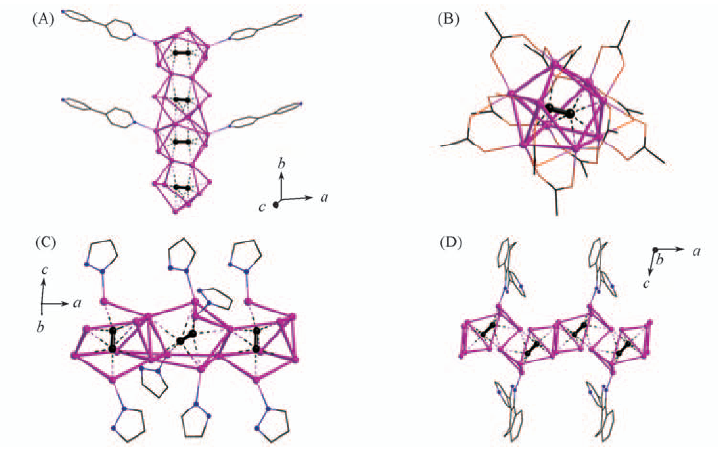
Fig.1 Coordination chain structure of Ag2C2·4AgCF3CO2·(bipyH)(CF3CO2)·H2O(A), discrete molecular structure of Ag2C2·7AgCF3CO2·(H3O)(CF3CO2)·2(bipyH2)(CF3CO2)2·3H2O(B), coordination chain structure of 2Ag2C2·8AgCF3CO2·6L1(L1=pyrazole)(C) and coordination chain structure of Ag2C2·10AgCF3CO2·2L2[L2=4,5-dihydro-3-(4-pyridinyl)-2H-benz(g)-indazole](D)
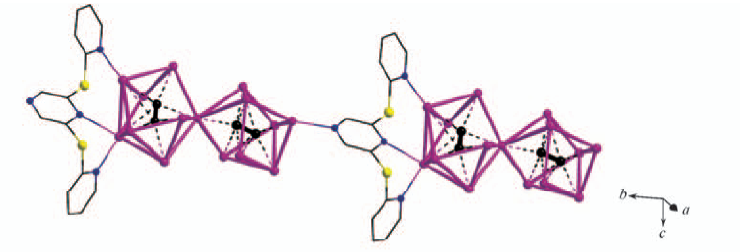
Fig.2 Ribbon-like coordination chain along b-axis formed by linkage of (C2)2@Ag15 units by bridging L3 ligands in [(Ag2C2)2(AgCF3COO)11(L3)(μ2-DMSO)3(DMSO)5]·1/4H2O
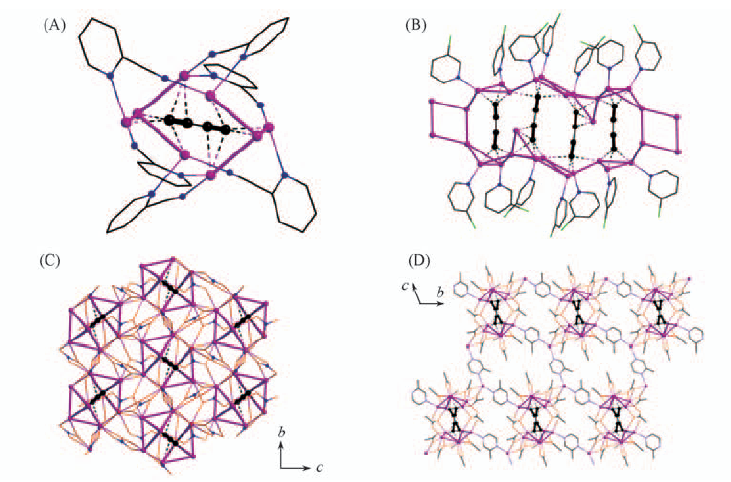
Fig.3 Discrete molecular structures of [(Ag2C4)(AgCF3CO2)6(L)6](L=2-cyanopyridine)(representative of 0D structure)(A), coordination chain structure of [(Ag2C4)2(AgCF3CO2)9(L)7(H2O)]n(L=3-chloropyridine)(representative of 1D structure)(B), coordination layer structure of [{(Ag2C4)0.5(AgNO3)3(L)}·H2O]n (L=3-chloropyridine)(representative of 2D structure)(C) and three-dimensional coordination network of [{(Ag2C4)(AgCF3CO2)5(L)1.5(H2O)2}·H2O]n(L=2-methylpyrazine)(representative of 3D structure)(D)
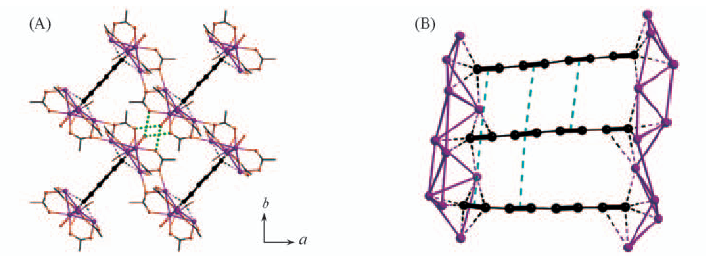
Fig.4 Coordination layer structure of Ag2C6·8AgCF3CO2·6H2O along c-axis(A) and ladder-like silver(I) double chain structure of 2.5(Ag2C8)·10AgCF3CO2·10DMSO(B)
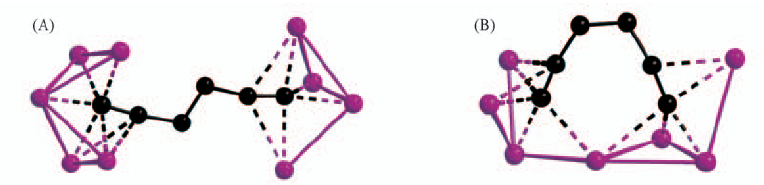
Fig.5 Two typical coordination modes of C6H42- dianion (A) Agm?C6H4?Agn, anti “stretched-Z” conformation; (B) C6H4?Agn, gauche “arch-like” conformation.
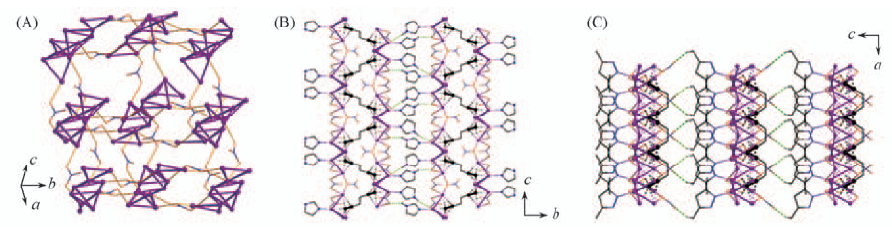
Fig.6 3D coordination network of Ag2C6H4·2AgNO3·2AgL1 constructed by linking silver layers by pillar-like nitrate groups(A) and Ag2C6H4·4AgNO3·2L5 with coordination layers linked by weak N—H…O hydrogen bonds involving imidazole ligands and nitrate groups to form a 3D supramole-cular framework(B) and 3D supramolecular framework of Ag2C6H4·4AgNO3·2L6 with each coordination layer constructed by cross-linkage of silver chains by nitrate ligands
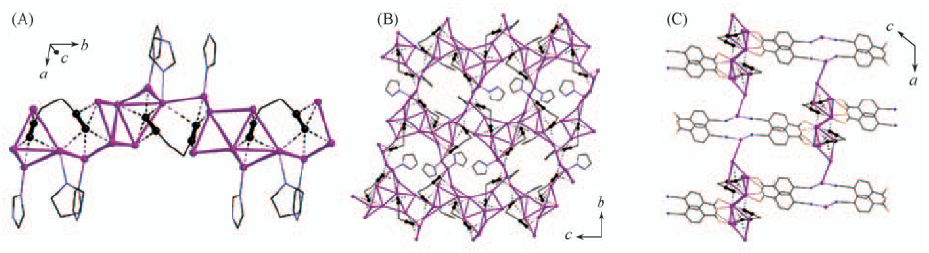
Fig.7 Infinite-chain structure of Ag2C6H4·5AgCF3CO2·3L5(A), two-dimensional coordination layer in 3Ag2C6H4·10AgCF3CO2·4L1(B) and three-dimensional coordination network of Ag2C6H4·3AgCF3CO2·2AgL7·H2O(C)

Fig.8 Top-down view of centrosymmetric Ag16 cluster in {(NO3)2@Ag16(PhC≡≡C)4[(tBuPO3)4V4O8]2·(DMF)6(NO3)2}(A) and top-down view of the core structure of pseudo-C3 Ag43 cluster in {[(O2)V2O6]3@Ag43(PhC≡≡C)19[(tBuPO3)4V4O8]3(DMF)6} For enhanced visibility, the three independent encapsulated [(O2)V2O6]4- species are represented by gray, turquoise, and blue semi-transparent polyhedra with V atoms included.
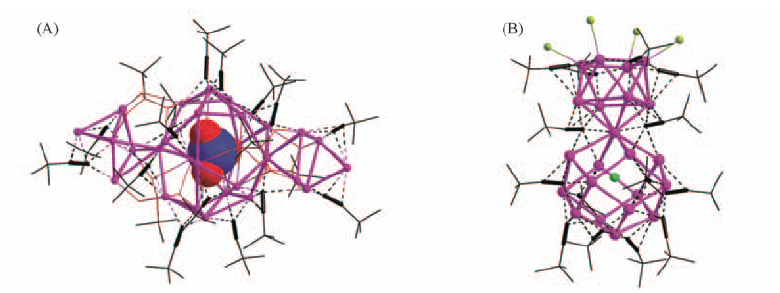
Fig.9 Core skeleton of one of the two nearly identical Ag28 clusters in [{Ag5(NO3@Ag18)Ag5}(tBuC≡≡C)16·(tBuPO3)4(H2O)4]·3SiF6·4.5H2O·3.5MeOH(A) and Cl@Ag22 cluster skeleton in[{Ag8(Cl@Ag14)}(tBuC≡≡C)14(tBuPO3)2F2·(H2O)2]BF4·3.5H2O, lying on a crystallographic C2 axis(B)
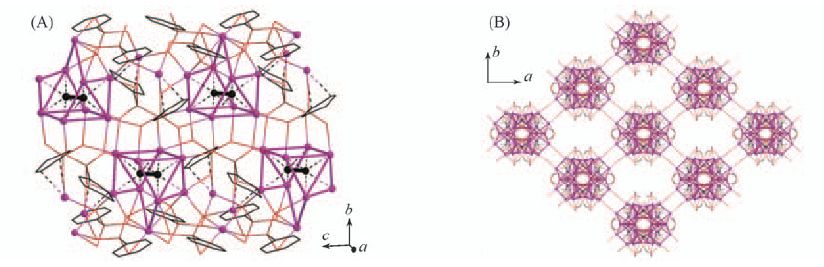
Fig.11 Layer structure of compound 1 viewed along the a-axis, in which the C2@Ag9 cages are linked by phosphonate groups(A) and three-dimensional architecture of compound 7 generated from the cross-linkage of silver chains by coordination bonding with aqua ligands(B)
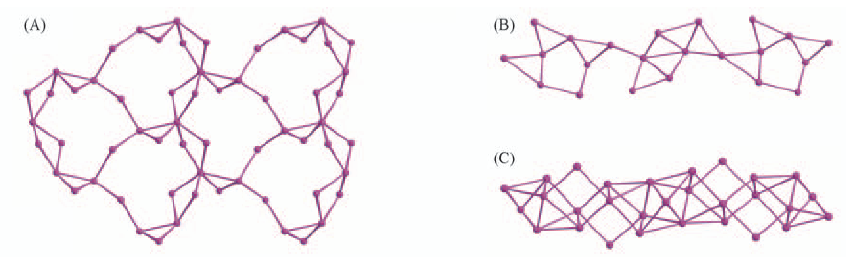
Fig.12 Perspective views of argentophilic layer structure in [AgC≡≡CPh·AgNO3](A), silver(I) chain in [2AgC≡≡CPh·5CF3CO2Ag·4DMSO](B) and thick silver coordination column in [10AgC≡≡CPh·2AgOTf·AgNO3·3DMSO](C)
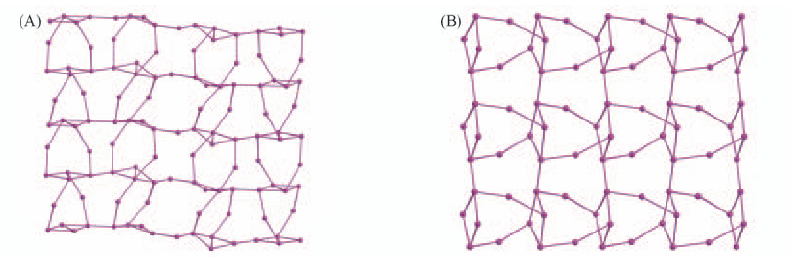
Fig.13 Perspective views of argentophilic silver(I) layer structure in 12[Ag(4-MeC6H4C≡≡C)]·7AgNO3(A) and the silver layer structure in 2[Ag(4-PhC6H4C≡≡C)]·AgNO3(B)
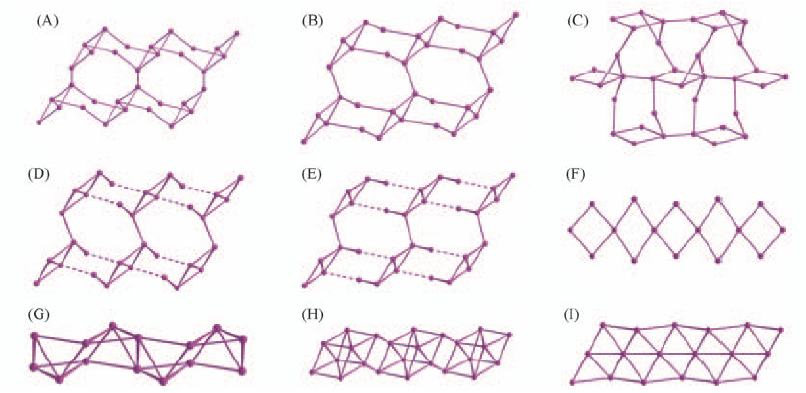
Fig.14 Argentophilic silver(I) structures in complexes generated with halophenylethynide ligands (A, D, G) Cl; (B, E, H) Br; (C, F, I) I; (A, B, C) para position; (D, E, F) meta position; (G, H, I) ortho position.
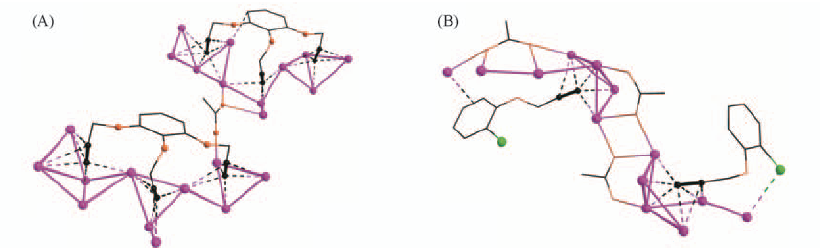
Fig.15 Different coordination modes of anionic tridentate L8 ligands in Ag11 and Ag12 aggregates(A) and coordination mode of L9 in [(AgL9)·(AgCF3CO2)5·(H2O)3][L9=1-chloro-2-(prop-2-ynyloxy)benzene]
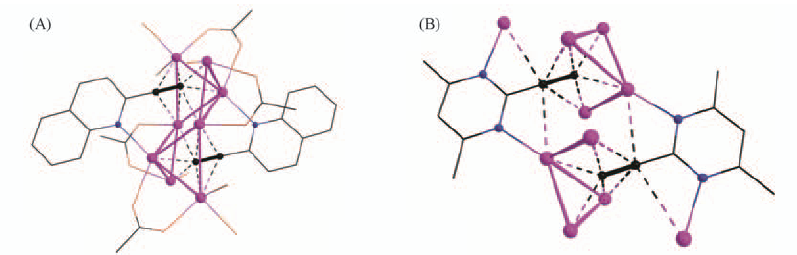
Fig.16 Perspective views of a portion of an infinite silver-organic chain in the crystal packing of AgL11·3AgCF3CO2·3H2O(L11=2-ethynylquinoline)(A) and Ag8 aggregate in [(AgL12)·(AgCF3CO2)3](L12=2-ethynyl-4,6-dimethylpyrimidine)(B)
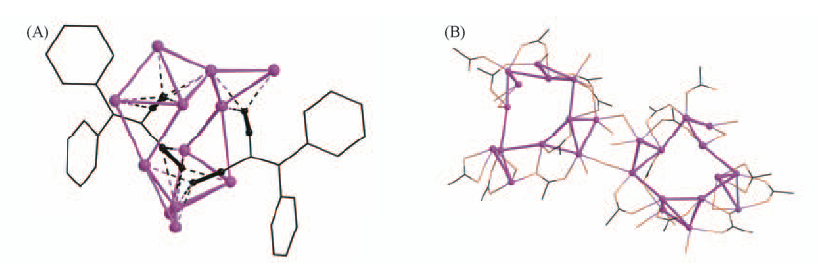
Fig.17 Coordination modes of independent L13 ligands in double salt(Ag2L13)·9AgCF3CO2·3H2O·3CH3CN(A) and perspective view of the linkage between metallocycles in the infinite-chain structure of (Ag2L13)2·9AgCF3CO2·11H2O(B)
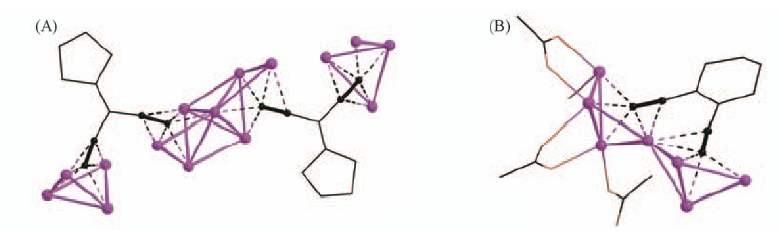
Fig.18 Perspective view of the silver(I) atoms and ethynide groups in (Ag2L15)·8AgCF3CO2·6DMSO(A) and coordination environment of the silver(I) atoms and ethynide groups in (Ag2L19)·5AgCF3CO2·2H2O(B)
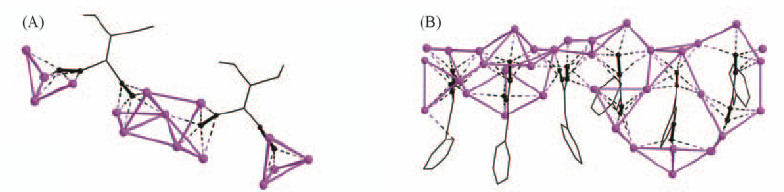
Fig.19 Coordination environment of the silver(I) atoms and ethynide groups in complex 11(A) and perspective view of the coordination silver-organic chain with L24 and Ag(I) aggregates in complex 12(B)
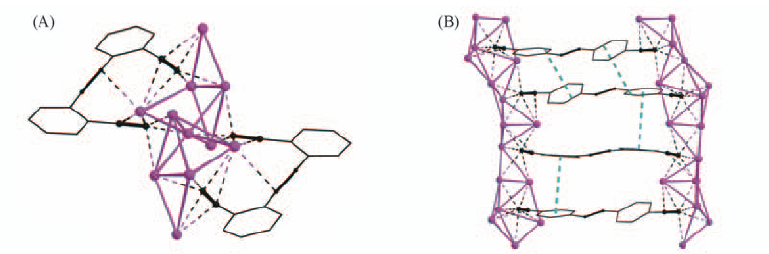
Fig.20 Perspective view of coordination geometry in double salt 2(Ag2L25)·7AgCF3CO2·3CH3CN(A) and perspective view of crystal packing in 3(Ag2L30)·14AgCF3CO2·[Ag2(CH3CN)3]·(CF3CO2)2·4H2O·6CH3CN(B)
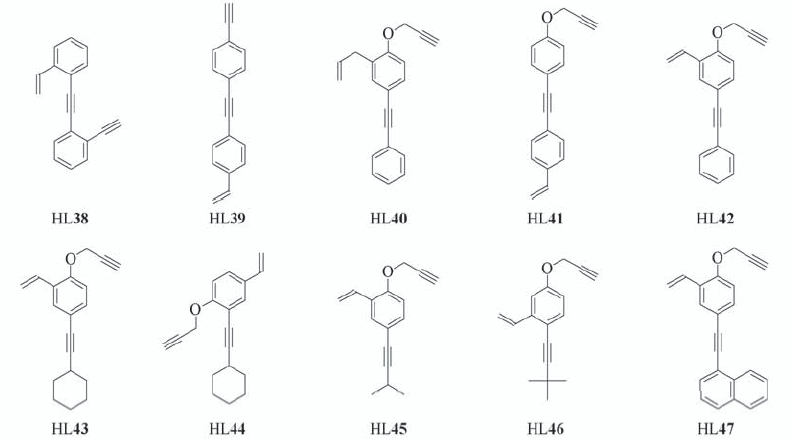
Scheme 6 A series of ten ligands containing an aromatic core with a rigid/flexible terminal ethynyl group, an internal ethynyl group, and a vinyl substituent at various positions on an aromatic ring
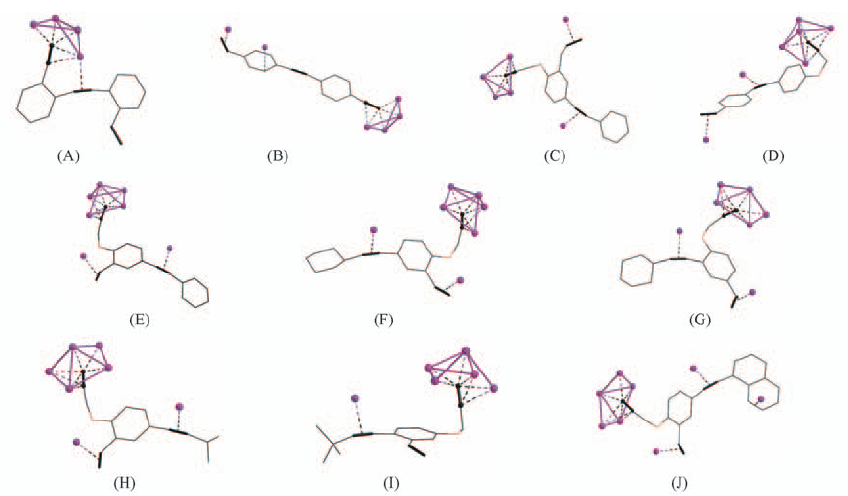
Fig.22 Coordination modes of the anionic ligands in their corresponding silver complexes, in which part (J) illustrates the presence of all the four kinds of silver-carbon binding interactions in AgL44·6AgCF3CO2·H2O·MeOH
| [1] | Mak T. C. W., Zhao L., Zhao X. L., The Importance of Pi-interactions in Crystal Engineering——Frontiers in Crystal Engineering III, Ed.: Tiekink E. R. T., Zukerman-Schpector J., Wiley Chichester, 2012, 13, 323—366 |
| [2] | Cheng P. S., Marivel S., Zang S. Q., Gao G. G., Mak T. C. W., Cryst. Growth Des., 2012, 12, 4519—4529 |
| [3] | Hau S. C. K., Cheng P. S., Mak T. C. W., J. Am. Chem. Soc., 2012, 134, 2922—2925 |
| [4] | Mak T. C. W., Zhao X. L., Wang Q. M., Guo G. C., Coord. Chem. Rev., 2007, 251, 2311—2333 |
| [5] | Wang Q. M., Mak T. C. W., J. Am. Chem. Soc., 2001, 123, 1501—1502 |
| [6] | Hu T., Mak T. C. W., Cryst. Growth Des., 2013, 13, 4957—4967 |
| [7] | Wang Q. M., Mak T. C. W., Inorg. Chem., 2013, 42, 1637—1643 |
| [8] | Hu T., Yang J., Mak T. C. W., Cryst. Eng. Comm., 2014, 16, 6316—6324 |
| [9] | Wen L. L., Wang H., Wan C. Q., Mak T. C. W., Organometallics, 2013, 32, 5144—5152 |
| [10] | Hu T., Zhao L., Mak T. C. W., Organometallics, 2012, 31, 7539—7547 |
| [11] | Hau S. C. K., Mak T. C. W., J. Am. Chem. Soc., 2014, 136, 902—905 |
| [12] | Hu T., Mak T. C. W., Organometallics, 2012, 32, 202—208 |
| [13] | Hu T., Mak T.C. W.,Eur. J. Inorg. Chem., 2013, 5476—5486 |
| [14] | Xie Y. P., Mak T. C. W., J. Am. Chem. Soc., 2011, 133, 3760—3763 |
| [15] | Xie Y. P., Mak T. C. W., Inorg. Chem., 2012, 51, 8640—8642 |
| [16] | Xie Y. P., Mak T. C. W., Dalton Trans., 2013, 42, 12869—12877 |
| [17] | Hu T., Mak T. C. W., Inorg. Chem., 2013, 52, 9066—9076 |
| [18] | Cheng P. S., Hau S. C. K., Mak T. C. W., Inorg. Chim. Acta, 2013, 403, 110—119 |
| [19] | Hau S. C. K., Mak T. C. W., J. Chin. Chem. Soc., 2013, 60, 877—896 |
| [20] | Cheng P. S., Hau S. C. K., Mak T. C. W., Aust. J. Chem., 2014, 67, 1849—1859 |
| [21] | Li B., Zang S. Q., Liang R., Wu Y. J., Mak T. C. W., Organometallics, 2011, 30, 1710—1718 |
| [22] | Li B., Zang S. Q., Ji C., Mak T. C. W., J. Organometallic Chem., 2011, 696, 2820—2828 |
| [23] | Cheng P. S., Hau S. C. K., Mak T. C. W., Aust. J. Chem., 2013, 66, 419—428 |
| [24] | Li B., Wang W. K., Zang S. Q., Mak T. C. W., J. Organometallic Chem., 2013, 745/746, 173—176 |
| [25] | Yang J., Hu T., Mak T. C. W., Cryst. Growth Des., 2014, 14, 2990—3001 |
| [26] | Braunstein P., Frison C., Oberbeckmann-Winter N., Morise X., Messaoudi A., Bénard M., Rohmer M. M., Welter R., Angew. Chem. Int. Ed., 2004, 43, 6120—6125 |
| [27] | Ronson T. K., Lazarides T., Adams H., Pope S. J. A., Sykes D., Faulkner S., Coles S. J., Hursthouse M. B., Clegg W., Harrington R. W., Ward M. D., Chem. Eur. J., 2006, 12, 9299—9313 |
| [28] | Salazar-Mendoza D., Baudron S.A., Hosseini M. W.,Chem. Commun., 2007, 2252—2254 |
| [29] | Fernádez-Cortabitarte C., García F., Morey J. V., McPartlin M., Singh S., Wheatley A. E. H., Wright D. S., Angew. Chem. Int. Ed., 2007, 46, 5425—5427 |
| [30] | Bruce M. I., Chem. Rev., 1991, 91, 197—257 |
| [31] | Lang H., George D. S. A., Rheinwald G., Coord. Chem. Rev., 2000, 206/207, 101—197 |
| [32] | Long N. J., Williams C. K., Angew. Chem. Int. Ed., 2003, 42, 2586—2617 |
| [33] | Yam V. W. W., J. Organomet. Chem., 2004, 689, 1393—1401 |
| [34] | Chui S. S. Y., Ng M. F. Y., Che C. M., Chem. Eur. J., 2005, 11, 1739—1749 |
| [35] | Wong W. Y., Coord. Chem. Rev., 2007, 251, 2400—2427 |
| [36] | McArdle C. P., Vittal J. J., Puddephatt R. J., Angew. Chem. Int. Ed., 2000, 39, 3819—3822 |
| [37] | McArdle C. P., Jennings M. C., Vittal J. J., Puddephatt R. J., Chem. Eur. J., 2001, 7, 3572—3593 |
| [38] | McArdle C. P., Irwin M. J., Jennings M. C., Vittal J. J., Puddephatt R. J., Chem. Eur. J., 2002, 8, 723—734 |
| [39] | McArdle C. P., Van S., Jennings M. C., Puddephatt R. J., J. Am. Chem. Soc., 2002, 124, 3959—3965 |
| [40] | Mohr F., Jennings M.C., Puddephatt R. J.,Eur. J. Inorg., 2003, 217—233 |
| [41] | Burchell T. J., Jennings M. C., Puddephatt R. J., Inorg. Chim. Acta, 2006, 359, 2812—2818 |
| [42] | Habermehl N. C., Eisler D. J., Kirby C. K., Yue N. L. S., Puddephatt R. J., Organometallics, 2006, 25, 2921—2928 |
| [43] | Hau S.C. K., Mak T. C. W., Polyhedron,64, 63—72 |
| [44] | Hau S. C. K., Tam D. Y. S., Mak T. C. W., Acta Crystallogr., Sect. B Struct. Sci., 2014, 70, 37—46 |
| [45] | Tam D.Y. S., Supramolecular Assembly of Multinuclear Silver(I) Complexes Containing Enediyne-functionalized Ligands, The Chinese University of Hong Kong, Hong Kong, 2014 |
| [46] | Meier H., Angew. Chem., 1992, 104, 1425—1446 |
| [47] | Meier H., Angew. Chem. Int. Ed., 1992, 31, 1437—1456 and references cited therein |
| [48] | Hau S. C. K., Mak T. C. W., Cryst. Growth Des., 2014, 14, 3567—3575 |
| [49] | Hau S. C. K., Cheng P. S., Mak T. C. W., Polyhedron, 2013, 52, 992—1008 |
| [50] | Hau S. C. K., Mak T. C. W., Chem. Eur. J., 2013, 19, 5387—5400 |
| [51] | Xie Y. P., Mak T. C. W., J. Clust. Sci., 2014, 25, 189—204 |
| [52] | Schmidbaur H., Schier A., Angew. Chem. Int. Ed., 2015, 54, 746—784 |
| [53] | Schmidbaur H., Schier A., Chem. Soc. Rev., 2012, 41, 370—412 |
| [54] | Muñiz J., Wang C., Pyykkö P., Chem. Eur. J., 2011, 17, 368—377 |
| [55] | Zhang J. P., Huang X. C., Lin Y. Y., Chen X. M., Chem. Eur. J., 2005, 11, 552—561 |
| [56] | Scherbaum F., Grohmann A., Müller G., Schmidbaur H., Angew. Chem. Int. Ed. Engl., 1989, 28, 463—465 |
| [57] | Scherbaum F., Grohmann A., Huber B., Kruger C., Schmidbaur H., Angew. Chem. Int. Ed. Engl., 1988, 27, 1544—1546 |
| [1] | 杨琳燦, 冷雪菲, 韩丽, 李超, 张松波, 雷岚, 马红卫, 李杨. 基于双锂法的炔基功能化热塑性弹性体的合成[J]. 高等学校化学学报, 2021, 42(3): 866. |
| [2] | 易卫国, 鄢东, 吴超, 兰立新. 铁(Ⅲ)催化氯代炔烃水化反应合成α-氯代甲基酮类化合物[J]. 高等学校化学学报, 2014, 35(12): 2563. |
| [3] | 唐启恒, 艾青松, 杨荣杰, 何吉宇. 主链含酰亚胺环和炔基的聚酰亚胺型聚氨酯弹性体的合成及阻燃性能[J]. 高等学校化学学报, 2014, 35(1): 199. |
| [4] | 赵宝娟, 于文全, 常俊标, 李二通, 丁群山, 李凤娟, 陈光英, 王娜. 3'-氟-2'-O,3'-C-乙烯基键联并环尿苷的合成与表征[J]. 高等学校化学学报, 2014, 35(1): 58. |
| [5] | 陶李明, 刘文奇, 周芸, 李爱桃, 刘卉. 碘催化的2-炔基苯胺与二硒醚的亲电环化反应[J]. 高等学校化学学报, 2013, 34(6): 1423. |
| [6] | 曹熙杰, 谢文兵, 潘利华, 常宇, 崔善子, 张洪杰, 党丹丹. 4,4'-二(对氨基苯乙炔基)-6,6'-二[N,N-二(乙氧基羰甲基)氨甲基]-2,2'-联吡啶的合成与表征[J]. 高等学校化学学报, 2012, 33(07): 1462. |
| [7] | 陈嘉媚, 吴传斌, 鲁统部. 超分子化学在药物共晶中的应用[J]. 高等学校化学学报, 2011, 32(9): 1996. |
| [8] | 王大明, 党国栋, 赵晓刚, 周宏伟, 王运良, 陈春海. 乙炔基封端聚酰亚胺增韧改性的相结构[J]. 高等学校化学学报, 2010, 31(5): 1051. |
| [9] | 刘文剑, 路慧哲, 肖玉梅, 王明安, 杜凤沛, 付滨, 李楠, 覃兆海. 炔丙型硫Ylide环丙烷化反应及其立体选择性[J]. 高等学校化学学报, 2009, 30(7): 1357. |
| [10] | 杜影, 党国栋, 胡南滔, 周宏伟, 陈春海. 新型乙炔封端聚酰亚胺的制备及性能[J]. 高等学校化学学报, 2009, 30(7): 1445. |
| [11] | 朱敏, 李立, 孙娜波, 李赫. 简便的β-二羰基化合物直接不对称α-苯乙炔基化反应[J]. 高等学校化学学报, 2009, 30(12): 2400. |
| [12] | 程晓红,Hoeger Sigurd . 含长链亚烷基桥的芳炔类共轭大环的合成[J]. 高等学校化学学报, 2007, 28(1): 65. |
| [13] | 赵静国, 陈振初. 苯基(苯乙炔基)对甲苯磺酸碘(Ⅲ)的应用──5-烃基-5-苯乙炔基丙二酸亚异丙酯的合成[J]. 高等学校化学学报, 1997, 18(1): 73. |
| [14] | 马春林, 马永祥, 陆熙炎. 铱配合物催化下炔基甲腙异构化反应的研究[J]. 高等学校化学学报, 1995, 16(8): 1251. |
| [15] | 还振威, 潘荫明, 唐俭生, 陈兰, 李华, 高振衡. 1,4-双(取代苯乙炔基)苯及反,反-1,4-双(β-取代苯乙烯基)苯的电子光谱的研究[J]. 高等学校化学学报, 1993, 14(11): 1543. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||