

高等学校化学学报 ›› 2015, Vol. 36 ›› Issue (9): 1771.doi: 10.7503/cjcu20150144
收稿日期:2015-02-11
出版日期:2015-09-10
发布日期:2015-08-21
作者简介:联系人简介: 吴德印, 男, 博士, 教授, 主要从事物理化学和谱学电化学理论研究. E-mail:基金资助:
PAN Wenbo, LI Mingxue, SU Yaqiong, WU Deyin*( ), TIAN Zhongqun
), TIAN Zhongqun
Received:2015-02-11
Online:2015-09-10
Published:2015-08-21
Contact:
WU Deyin
E-mail:dywu@xmu.edu.cn
Supported by:摘要:
通过密度泛函理论(DFT)计算了锂硫电池中多硫阴离子 (Sn2-)和碳酸乙烯酯(EC)的2种反应机理, 并对其反应物和主要产物的拉曼光谱进行了理论分析. 计算结果表明, 2种反应路径中, (Sn2-)进攻EC分子烷基碳所需活化能和Gibbs自由能均较低. 多硫基团有很强的拉曼特征谱峰, 与EC分子相关的部分拉曼谱亦发生显著变化. 锂离子的存在影响反应产物的结构和拉曼光谱. 密度泛函理论计算提供了锂硫电池中锂、 多硫阴离子和电解液反应前后的结构变化和反应信息.
中图分类号:
TrendMD:
潘文博, 李明雪, 苏亚琼, 吴德印, 田中群. 锂硫电池中碳酸乙烯酯与硫簇反应拉曼光谱的理论研究. 高等学校化学学报, 2015, 36(9): 1771.
PAN Wenbo, LI Mingxue, SU Yaqiong, WU Deyin, TIAN Zhongqun. Theoretical Study of Reactions Between Polysulfides and Ethylene Carbonate and Raman Spectra in Lithium-sulfur Battery†. Chem. J. Chinese Universities, 2015, 36(9): 1771.
| Parameter | EC | Li+(EC) | ||||
|---|---|---|---|---|---|---|
| 6-311++G(d,p) | Expt.(crys)[ | Expt.(MW)[ | aug-cc-pVDZ[ | 6-311++G(d,p) | ||
| Bond length/nm | C=O | 0.1188 | 0.1203 | 0.1200 | 0.1203 | 0.1208 |
| C3—O | 0.1361 | 0.1342 | 0.1358 | 0.1370 | 0.1333 | |
| C—O | 0.1437 | 0.1457 | 0.1428 | 0.1440 | 0.1456 | |
| C—C | 0.1529 | 0.1522 | 0.1540 | 0.1520 | 0.1531 | |
| C—H | 0.1089 | 0.1091 | 0.1095 | 0.1090 | 0.1089 | |
| Bending angle/(°) | O=C—O | 124.9 | 124.2 | 124.2 | 124.0 | |
| C—O—C | 109.4 | 108.7 | 109.5 | 108.4 | 109.3 | |
| O—C—O | 110.1 | 111.7 | 110.4 | 110.4 | 112.4 | |
| C—C—O | 102.6 | 102.2 | 102.4 | 102.5 | 102.6 | |
| H—C—H | 109.9 | 110.8 | 114.0 | 110.6 | ||
| Dihedral angle/(°) | O=C—O—C | -171.7 | -171.3 | -171.5 | 170.6 | 179.9 |
| C—O—C—C | -20.0 | -21.3 | -20.2 | 22.9 | -16.3 | |
| O—C—C—O | 23.5 | 24.8 | 23.9 | 27.2 | 19.1 | |
| O—C—O—C | 8.3 | 8.7 | 8.5 | 9.4 | 6.7 | |
Table 1 Comparison of theoretical and experimental geometrical parameters for EC and Li+(EC)
| Parameter | EC | Li+(EC) | ||||
|---|---|---|---|---|---|---|
| 6-311++G(d,p) | Expt.(crys)[ | Expt.(MW)[ | aug-cc-pVDZ[ | 6-311++G(d,p) | ||
| Bond length/nm | C=O | 0.1188 | 0.1203 | 0.1200 | 0.1203 | 0.1208 |
| C3—O | 0.1361 | 0.1342 | 0.1358 | 0.1370 | 0.1333 | |
| C—O | 0.1437 | 0.1457 | 0.1428 | 0.1440 | 0.1456 | |
| C—C | 0.1529 | 0.1522 | 0.1540 | 0.1520 | 0.1531 | |
| C—H | 0.1089 | 0.1091 | 0.1095 | 0.1090 | 0.1089 | |
| Bending angle/(°) | O=C—O | 124.9 | 124.2 | 124.2 | 124.0 | |
| C—O—C | 109.4 | 108.7 | 109.5 | 108.4 | 109.3 | |
| O—C—O | 110.1 | 111.7 | 110.4 | 110.4 | 112.4 | |
| C—C—O | 102.6 | 102.2 | 102.4 | 102.5 | 102.6 | |
| H—C—H | 109.9 | 110.8 | 114.0 | 110.6 | ||
| Dihedral angle/(°) | O=C—O—C | -171.7 | -171.3 | -171.5 | 170.6 | 179.9 |
| C—O—C—C | -20.0 | -21.3 | -20.2 | 22.9 | -16.3 | |
| O—C—C—O | 23.5 | 24.8 | 23.9 | 27.2 | 19.1 | |
| O—C—O—C | 8.3 | 8.7 | 8.5 | 9.4 | 6.7 | |
| Modea | 6-311++G(d,p) | Scaled | Expt.[ | aug-cc-pVDZ[ | 1019AR/(nm4·kg-1) | PED(%)b |
|---|---|---|---|---|---|---|
| ν1(b) | 3135 | 3031 | 3011 | 3205 | 46.6 | νCH2(100) |
| ν2(a) | 3123 | 3020 | 3004 | 3194 | 57.6 | νCH2(86) |
| ν3(b) | 3058 | 2957 | 3000 | 3112 | 18.3 | νCH2(100) |
| ν4(a) | 3054 | 2953 | 2990 | 3109 | 132.3 | νCH2(86) |
| ν5(a) | 1895 | 1860 | 1868 | 1898 | 10.4 | ν(C=O) (86) |
| ν6(a) | 1529 | 1500 | 1570 | 1546 | 2.5 | sciCH2(99) |
| ν7(b) | 1521 | 1492 | 1480 | 1539 | 6.3 | sciCH2(98) |
| ν8(b) | 1400 | 1373 | 1421 | 1421 | 0.6 | ωCH2(81) |
| ν9(a) | 1386 | 1360 | 1386 | 1420 | 2.6 | ωCH2(87) |
| ν10(a) | 1245 | 1222 | 1223 | 1271 | 4.4 | τCH2(91) |
| ν11(a) | 1238 | 1214 | 1218 | 1267 | 4.6 | τCH2(79) |
| ν12(a) | 1154 | 1132 | 1157 | 1175 | 0.2 | rockCH2(95) |
| ν13(b) | 1115 | 1070 | 1125 | 1138 | 0.4 | ν(C—O)(59) |
| β(C=O)(12) | ||||||
| ν14(a) | 1090 | 1094 | 1090 | 1123 | 1.4 | ν(C—O)(76) |
| ν(C—C)(14) | ||||||
| ν15(b) | 1051 | 1031 | 1087 | 1079 | 0.1 | ν(C—O)(72) |
| ν16(a) | 964 | 946 | 960 | 991 | 4.5 | ν(C—C)(62) |
| ν17(a) | 891 | 874 | 891 | 895 | 0.9 | R breathing |
| ν18(b) | 889 | 872 | 881 | 919 | 8.3 | rockCH2(52) |
| ν19(b) | 772 | 758 | 768 | 779 | 0.3 | β(C=O)(97) |
| ν20(a) | 718 | 704 | 715 | 719 | 3.7 | νR(68) |
| ν21(b) | 691 | 678 | 673 | 673 | 1.6 | τR(62) |
| ν22(b) | 523 | 513 | 527 | 527 | 0.3 | γ(C=O)(70) |
| ν23(a) | 187 | 184 | 227 | 227 | 0.2 | τR(97) |
| ν24(b) | 178 | 175 | 184 | 184 | 0.2 | τR(97.3) |
Table 2 Calculated frequencies, Raman activity(AR) and assignment of EC along with the reference dataa
| Modea | 6-311++G(d,p) | Scaled | Expt.[ | aug-cc-pVDZ[ | 1019AR/(nm4·kg-1) | PED(%)b |
|---|---|---|---|---|---|---|
| ν1(b) | 3135 | 3031 | 3011 | 3205 | 46.6 | νCH2(100) |
| ν2(a) | 3123 | 3020 | 3004 | 3194 | 57.6 | νCH2(86) |
| ν3(b) | 3058 | 2957 | 3000 | 3112 | 18.3 | νCH2(100) |
| ν4(a) | 3054 | 2953 | 2990 | 3109 | 132.3 | νCH2(86) |
| ν5(a) | 1895 | 1860 | 1868 | 1898 | 10.4 | ν(C=O) (86) |
| ν6(a) | 1529 | 1500 | 1570 | 1546 | 2.5 | sciCH2(99) |
| ν7(b) | 1521 | 1492 | 1480 | 1539 | 6.3 | sciCH2(98) |
| ν8(b) | 1400 | 1373 | 1421 | 1421 | 0.6 | ωCH2(81) |
| ν9(a) | 1386 | 1360 | 1386 | 1420 | 2.6 | ωCH2(87) |
| ν10(a) | 1245 | 1222 | 1223 | 1271 | 4.4 | τCH2(91) |
| ν11(a) | 1238 | 1214 | 1218 | 1267 | 4.6 | τCH2(79) |
| ν12(a) | 1154 | 1132 | 1157 | 1175 | 0.2 | rockCH2(95) |
| ν13(b) | 1115 | 1070 | 1125 | 1138 | 0.4 | ν(C—O)(59) |
| β(C=O)(12) | ||||||
| ν14(a) | 1090 | 1094 | 1090 | 1123 | 1.4 | ν(C—O)(76) |
| ν(C—C)(14) | ||||||
| ν15(b) | 1051 | 1031 | 1087 | 1079 | 0.1 | ν(C—O)(72) |
| ν16(a) | 964 | 946 | 960 | 991 | 4.5 | ν(C—C)(62) |
| ν17(a) | 891 | 874 | 891 | 895 | 0.9 | R breathing |
| ν18(b) | 889 | 872 | 881 | 919 | 8.3 | rockCH2(52) |
| ν19(b) | 772 | 758 | 768 | 779 | 0.3 | β(C=O)(97) |
| ν20(a) | 718 | 704 | 715 | 719 | 3.7 | νR(68) |
| ν21(b) | 691 | 678 | 673 | 673 | 1.6 | τR(62) |
| ν22(b) | 523 | 513 | 527 | 527 | 0.3 | γ(C=O)(70) |
| ν23(a) | 187 | 184 | 227 | 227 | 0.2 | τR(97) |
| ν24(b) | 178 | 175 | 184 | 184 | 0.2 | τR(97.3) |
| Species | Structure | Charge distribution | ||||
|---|---|---|---|---|---|---|
| Bond length/nm | Bending angle/(°) | S1 | S2 | S3 | S4 | |
| 0.2220 | -1.00 | -1.00 | ||||
| 0.2151 | 110.5 | -1.05 | -0.34 | -0.61 | ||
| 0.2127 | 111.4 | -0.61 | -0.39 | -0.39 | -0.61 | |
Table 3 Structure parameters and NBO charge distribution of polysulfide anions at the PCM-B3LYP/6-311++G(d,p) level
| Species | Structure | Charge distribution | ||||
|---|---|---|---|---|---|---|
| Bond length/nm | Bending angle/(°) | S1 | S2 | S3 | S4 | |
| 0.2220 | -1.00 | -1.00 | ||||
| 0.2151 | 110.5 | -1.05 | -0.34 | -0.61 | ||
| 0.2127 | 111.4 | -0.61 | -0.39 | -0.39 | -0.61 | |
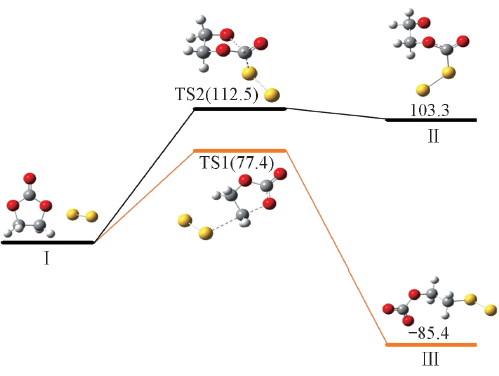
Fig.3 Reaction activation energies and Gibbs free energies(kJ/mol) for nucleophilic attack of S22- at the carbonyl and ethyl carbon atoms of EC calculated at the PCM-B3LYP/6-311++G(d,p) level
| Species | Attacking ethyl carbon | Attacking carbonyl carbon | ||||||
|---|---|---|---|---|---|---|---|---|
| ΔHact,1 | ΔGact,1 | ΔHr,1 | ΔGr,1 | ΔHact,2 | ΔGact,2 | ΔHr,2 | ΔGr,2 | |
| 40.9 | 77.4 | -116.0 | -85.4 | 71.3 | 112.5 | 68.8 | 103.3 | |
| 53.7 | 93.3 | -80.0 | -43.5 | 116.6 | 163.5 | 116.2 | 156.6 | |
| 62.4 | 100.9 | -60.4 | -24.9 | 135.6 | 187.3 | 141.0 | 178.2 | |
Table 4 Reaction activation energies, enthalpies and Gibbs free energies(kJ/mol) for nucleophilic attack of polysulfide anions S2-n(n=2—4) at the carbonyl and ethyl carbon atoms of EC calculated at the PCM-B3LYP/6-311++G(d,p) level
| Species | Attacking ethyl carbon | Attacking carbonyl carbon | ||||||
|---|---|---|---|---|---|---|---|---|
| ΔHact,1 | ΔGact,1 | ΔHr,1 | ΔGr,1 | ΔHact,2 | ΔGact,2 | ΔHr,2 | ΔGr,2 | |
| 40.9 | 77.4 | -116.0 | -85.4 | 71.3 | 112.5 | 68.8 | 103.3 | |
| 53.7 | 93.3 | -80.0 | -43.5 | 116.6 | 163.5 | 116.2 | 156.6 | |
| 62.4 | 100.9 | -60.4 | -24.9 | 135.6 | 187.3 | 141.0 | 178.2 | |
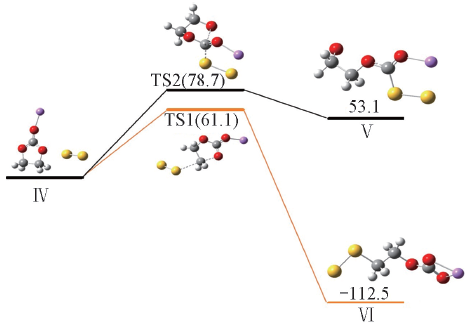
Fig.5 Reaction activation energies and Gibbs free energies(kJ/mol) for nucleophilic attack of S22- at the carbonyl and ethyl carbon atoms of Li+EC calculated at the PCM-B3LYP/6-311++G(d,p) level
| Species | Attacking ethyl carbon | Attacking carbonyl carbon | ||||||
|---|---|---|---|---|---|---|---|---|
| ΔHact,1 | ΔGact,1 | ΔHr,1 | ΔGr,1 | ΔHact,2 | ΔHr,2 | ΔGact,2 | ΔGr,2 | |
| 21.8 | 61.1 | -168.0 | -112.5 | 28.9 | -41.2 | 78.7 | 53.1 | |
| 34.3 | 78.5 | -131.6 | -81.7 | 77.7 | 203.1 | 132.6 | 62.5 | |
| 42.8 | 86.7 | -256.1 | -57.5 | 114.2 | 41.2 | 165.8 | 91.5 | |
Table 5 Reaction activation energies, enthalpies and Gibbs free energies(kJ/mol) for nucleophilic attack of polysulfides anions S2-n(n=2—4) at the carbonyl and ethyl carbon atoms of Li+(EC) calculated at the PCM-B3LYP/6-311++G(d,p) level
| Species | Attacking ethyl carbon | Attacking carbonyl carbon | ||||||
|---|---|---|---|---|---|---|---|---|
| ΔHact,1 | ΔGact,1 | ΔHr,1 | ΔGr,1 | ΔHact,2 | ΔHr,2 | ΔGact,2 | ΔGr,2 | |
| 21.8 | 61.1 | -168.0 | -112.5 | 28.9 | -41.2 | 78.7 | 53.1 | |
| 34.3 | 78.5 | -131.6 | -81.7 | 77.7 | 203.1 | 132.6 | 62.5 | |
| 42.8 | 86.7 | -256.1 | -57.5 | 114.2 | 41.2 | 165.8 | 91.5 | |
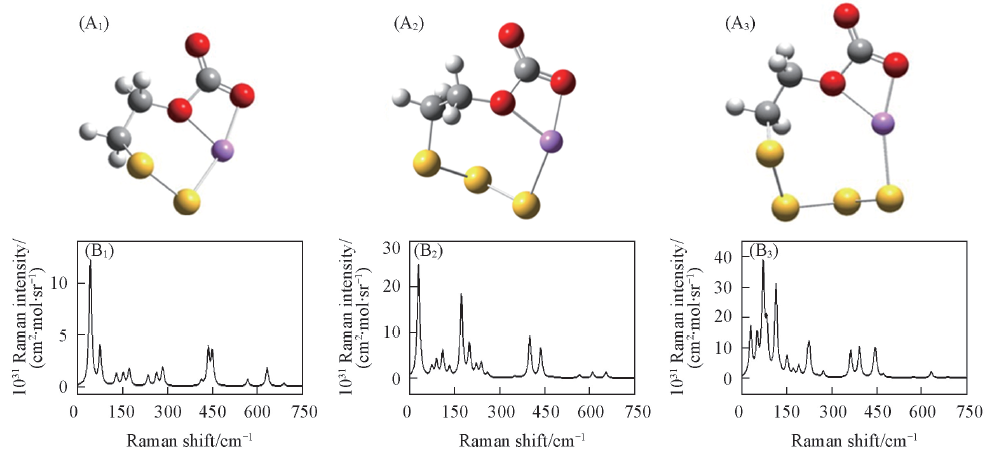
Fig.7 Optimized structures(A1—A3) and simulated Raman spectra(B1—B3) of isomerization products calculated at the PCM-B3LYP/6-311++G(d,p) level(A1), (B1) Ring-Li+(EC)S22-; (A2), (B2) ring-Li+(EC)S32-; (A3), (B3) ring-Li+(EC)S42-.
| Reaction | ΔH/(kJ·mol-1) | ΔS/(J·mol-1·K-1) | ΔG/(kJ·mol-1) |
|---|---|---|---|
| Li+(EC) | -18.1 | -17.8 | -12.8 |
| Li+(EC) | -18.6 | -26.5 | -10.7 |
| Li+(EC) | -14.3 | -37.9 | -3.0 |
Table 6 Enthalpies and Gibbs free energies for isomerization products calculated at the PCM-B3LYP/6-311++G(d,p) level
| Reaction | ΔH/(kJ·mol-1) | ΔS/(J·mol-1·K-1) | ΔG/(kJ·mol-1) |
|---|---|---|---|
| Li+(EC) | -18.1 | -17.8 | -12.8 |
| Li+(EC) | -18.6 | -26.5 | -10.7 |
| Li+(EC) | -14.3 | -37.9 | -3.0 |
| [1] | Yuan K. G., Wang A. B., Yu Z. B., Wang W. K., Yang Y. S., Chem. J. Chinese Universities, 2006, 27(9), 1738—1741 |
| (苑克国, 王安邦, 余仲宝, 王维坤, 杨裕生. 高等学校化学学报, 2006, 27(9), 1738—1741) | |
| [2] | Ji X., Nazar L. F., J. Mater. Chem., 2010, 20(44), 9821—9826 |
| [3] | Yin Y. X., Xin S., Guo Y. G., Wan L. J., Angew. Chem.Int. Ed., 2013, 52(50), 13186—13200 |
| [4] | Chen R. J., Zhao T., Li L., Chen J. Z., Wu F., Sci.China Chem., 2014, 8, 8 |
| (陈人杰, 赵腾, 李丽, 陈君政, 吴锋. 中国科学: 化学, 2014, 8, 8) | |
| [5] | Manthiram A., Fu Y., Su Y. S., Accounts. Chem. Res., 2012, 46(5), 1125—1134 |
| [6] | Manthiram A., Fu Y., Chung S. H., Zu C., Su Y. S., Chem. Rev., 2014, 114(23), 11751—11787 |
| [7] | Zhang S. S., J. Power Sources, 2013, 231, 153—162 |
| [8] | Scheers J., Fantini S., Johansson P., J. Power Sources, 2014, 255, 204—218 |
| [9] | Yamin H., Peled E., J. Power Sources, 1983, 9(3), 281—287 |
| [10] | Peled E., Gorenshtein A., Segal M., Sternberg Y., J. Power Sources, 1989, 26, 269—271 |
| [11] | Nelson J., Misra S., Yang Y., Jackson A., Liu Y., Wang H., Dai H., Andrews J. C., Cui Y., Toney M. F., J. Am. Chem. Soc., 2012, 134(14), 6337—6343 |
| [12] | Hagen M., Schiffels P., Hammer M., Dörfler S., Tübke J., Hoffmann M., Althues H., Kaskel S., J. Electrochem. Soc., 2013, 160(8), A1205—A1214 |
| [13] | Kim S., Jung Y., Park S. J., J. Power Sources, 2005, 152, 272—277 |
| [14] | Kim S., Jung Y., Park S. J., Electrochim. Acta, 2007, 52(5), 2116—2122 |
| [15] | Dong Q. F., Wang C., Zheng M. S., Process Chem., 2011, 23(2), 533—539 |
| (董全峰, 王翀, 郑明森. 化学进展, 2011, 23(2), 533—539) | |
| [16] | Wang L., Zhang T., Yang S., Cheng F., Liang J., Chen J., J. Energ. Chem., 2013, 22(1), 72—77 |
| [17] | Assary R. S., Curtiss L. A., Moore J. S., J. Phys. Chem. C, 2014, 118(22), 11545—11558 |
| [18] | Gao J., Lowe M.a H. C. D., J. Phys. Chem. C, 2011, 115(50), 25132—25137 |
| [19] | Yim T., Park M. S., Yu J. S., Kim K. J., Im K. Y., Kim J. H., Jeong G., Jo Y. N., Woo S. G., Kang K. S., Electrochim. Acta, 2013, 107, 454—460 |
| [20] | Wang J., Chew S. Y., Zhao Z. W., Ashraf S., Wexler D., Chen J., Ng S. H., Chou S. L., Liu H. K., Carbon, 2008, 46(2), 229—235 |
| [21] | Liang X., Wen Z., Liu Y., Zhang H., Jin J., Wu M., Wu X., J. Power Sources, 2012, 206(0), 409—413 |
| [22] | Li T., Balbuena P. B., J. Electrochem. Soc., 1999, 146(10), 3613—3622 |
| [23] | Allen J. L., Borodin O., Seo D. M., Henderson W. A., J. Power Sources, 2014, 267, 821—830 |
| [24] | Masia M., Probst M., Rey R., J. Phys. Chem. B, 2004, 108(6), 2016—2027 |
| [25] | Wang Y., Nakamura S., Ue M., Balbuena P. B., J. Am. Chem. Soc., 2001, 123(47), 11708—11718 |
| [26] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., et al., Gaussian 09, Revision A. 01; Gaussian Inc., Wallingford, CT, 2009 |
| [27] | Krishnan R., Binkley J. S., Seeger R., Pople J. A., J. Chem. Phys., 1980, 72(1), 650—654 |
| [28] | McLean A., Chandler G., J. Chem. Phys., 1980, 72(10), 5639—5648 |
| [29] | Pulay P., Fogarasi G., Pang F., Boggs J. E., J. Am. Chem. Soc., 1979, 101(10), 2550—2560 |
| [30] | Wu D. Y., Liu X. M., Huang Y. F., Ren B., Xu X., Tian Z. Q., J. Phys. Chem. C, 2009, 113(42), 18212—18222 |
| [31] | Pulay P., Fogarasi G., Pongor G., Boggs J. E., Vargha A., J. Am. Chem. Soc., 1983, 105(24), 7037—7047 |
| [32] | Angell C., Trans. Faraday Soc., 1956, 52, 1178—1183 |
| [33] | Wang I., Britt C. O., Boggs J. E., J. Am. Chem. Soc., 1965, 87(21), 4950—4951 |
| [34] | Fortunato B., Mirone P., Fini G., Spectrochim. Acta Part A: Mol. Spectro., 1971, 27(9), 1917—1927 |
| [35] | Alonso J. L., Cervellati R., Degli Esposti A., Lister D. G., Palmieri P., J. Chem. Soc. Faraday Transactions 2: Molecular and Chemical Physics, 1986, 82(3), 337—356 |
| [36] | Matias P. M., Jeffrey G., Wingert L. M., Ruble J. R., J. Mol. Struct: Theochem., 1989, 184(3), 247—260 |
| [37] | Cremer D., Pople J. A., J. Am. Chem. Soc., 1975, 97(6), 1354—1358 |
| [38] | Blint R. J., J. Electrochem. Soc., 1995, 142(3), 696—702 |
| [39] | Klassen B., Aroca R., Nazri M., Nazri G. A., J. Phys. Chem. B, 1998, 102(24), 4795—4801 |
| [40] | Brown C., Acta Crystallographica, 1954, 7(1), 92—96 |
| [41] | Berghof V., Sommerfeld T., Cederbaum L., J. Phys. Chem. A, 1998, 102(26), 5100—5105 |
| [42] | Meyer B., Chem. Rev., 1976, 76(3), 367—388 |
| [43] | Yu H. J., Liu Z. J., Yin Y. F., Fu J., Ding L., Mo Y. J., Spectrosc.Spect.Anal., 2009, 29(11), 2975—2979 |
| (余红静, 刘照军, 尹延峰, 符娟, 丁丽, 莫育俊. 光谱学与光谱分析, 2009, 29(11), 2975—2979) |
| [1] | 韩付超, 李福进, 陈良, 贺磊义, 姜玉南, 徐守冬, 张鼎, 其鲁. CoSe2/C复合电催化材料修饰隔膜对高载量锂硫电池性能的影响[J]. 高等学校化学学报, 2022, 43(8): 20220163. |
| [2] | 何鸿锐, 夏文生, 张庆红, 万惠霖. 羟基氧化铟团簇与二氧化碳和甲烷作用的密度泛函理论研究[J]. 高等学校化学学报, 2022, 43(8): 20220196. |
| [3] | 陈嘉敏, 曲孝章, 齐国华, 徐蔚青, 金永东, 徐抒平. SERS纳米探针对电刺激过程中细胞内产生活性氧的检测[J]. 高等学校化学学报, 2022, 43(6): 20220033. |
| [4] | 黄汉浩, 卢湫阳, 孙明子, 黄勃龙. 石墨炔原子催化剂的崭新道路:基于自验证机器学习方法的筛选策略[J]. 高等学校化学学报, 2022, 43(5): 20220042. |
| [5] | 尹肖菊, 孙逊, 赵程浩, 姜波, 赵晨阳, 张乃庆. 单原子催化剂在锂硫电池中的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220076. |
| [6] | 张诗昱, 何润合, 李永兵, 魏士俊, 张兴祥. 辐照交联制备低分子量聚丙烯腈纤维锂硫电池正极材料及其储硫机理[J]. 高等学校化学学报, 2022, 43(3): 20210632. |
| [7] | 刘洋, 李旺昌, 张竹霞, 王芳, 杨文静, 郭臻, 崔鹏. Sc3C2@C80与[12]CPP纳米环之间非共价相互作用的理论研究[J]. 高等学校化学学报, 2022, 43(11): 20220457. |
| [8] | 王园月, 安梭梭, 郑旭明, 赵彦英. 5-巯基-1, 3, 4-噻二唑-2-硫酮微溶剂团簇的光谱和理论计算研究[J]. 高等学校化学学报, 2022, 43(10): 20220354. |
| [9] | 程媛媛, 郗碧莹. ·OH自由基引发CH3SSC |
| [10] | 周成思, 赵远进, 韩美晨, 杨霞, 刘晨光, 贺爱华. 硅烷类外给电子体对丙烯-丁烯序贯聚合的调控作用[J]. 高等学校化学学报, 2022, 43(10): 20220290. |
| [11] | 马丽娟, 高升启, 荣祎斐, 贾建峰, 武海顺. Sc, Ti, V修饰B/N掺杂单缺陷石墨烯的储氢研究[J]. 高等学校化学学报, 2021, 42(9): 2842. |
| [12] | 钟声广, 夏文生, 张庆红, 万惠霖. 电中性团簇MCu2Ox(M=Cu2+, Ce4+, Zr4+)上甲烷和二氧化碳直接合成乙酸的理论研究[J]. 高等学校化学学报, 2021, 42(9): 2878. |
| [13] | 黄罗仪, 翁约约, 黄旭慧, 王朝杰. 车前草中黄酮类成分结构和性质的理论研究[J]. 高等学校化学学报, 2021, 42(9): 2752. |
| [14] | 陈铭苏, 张会茹, 张琪, 刘家琴, 吴玉程. 锂硫电池中钴磷共掺杂MoS2催化性能的第一性原理研究[J]. 高等学校化学学报, 2021, 42(8): 2540. |
| [15] | 薛谨, 曹小卫, 刘依帆, 王敏. 纸质空心金纳米笼SERS传感器的制备及对非小细胞肺癌患者痰液中miRNAs的快速高灵敏检测[J]. 高等学校化学学报, 2021, 42(8): 2393. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||