

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (6): 1189.doi: 10.7503/cjcu20131048
• Organic Chemistry • Previous Articles Next Articles
WANG Junhua, WANG Quande, DUN Yanyan, FANG Hao*( )
)
Received:2013-10-28
Online:2014-06-10
Published:2014-01-13
Contact:
FANG Hao
E-mail:haofangcn@sdu.edu.cn
Supported by:CLC Number:
TrendMD:
WANG Junhua, WANG Quande, DUN Yanyan, FANG Hao. Syntheses and Antitumor Activities of Purine-sulfonamides Derivatives†[J]. Chem. J. Chinese Universities, 2014, 35(6): 1189.
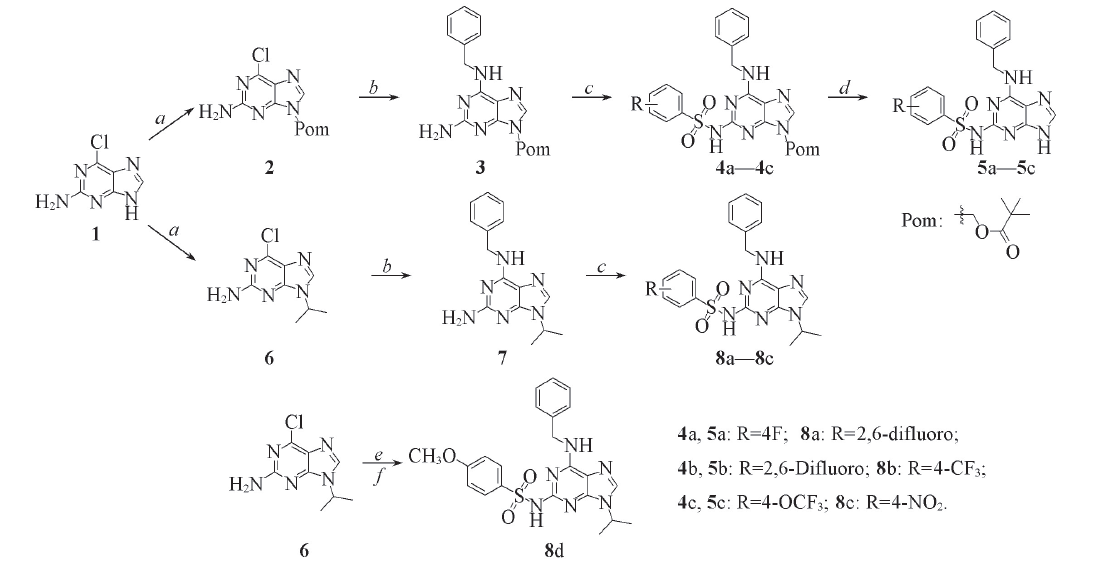
Scheme 1 Synthesis routes of target compounds 5a—5c and 8a—8da. Chloromethyl pivalate(Pom-Cl), K2CO3, DMSO, r. t. or 2-bromopropane, K2CO3, DMSO, 80 ℃; b. benzyl amine, TEA, n-butanol, reflux; c. arylsulfonyl chloride, pyridine, r. t.; d. 3 mol/L NaOH, THF, r. t.; e. 4-methoxybenzene sulfonyl chloride, pyridine, r. t.; f. benzyl amine, TEA, n-butanol, reflux.
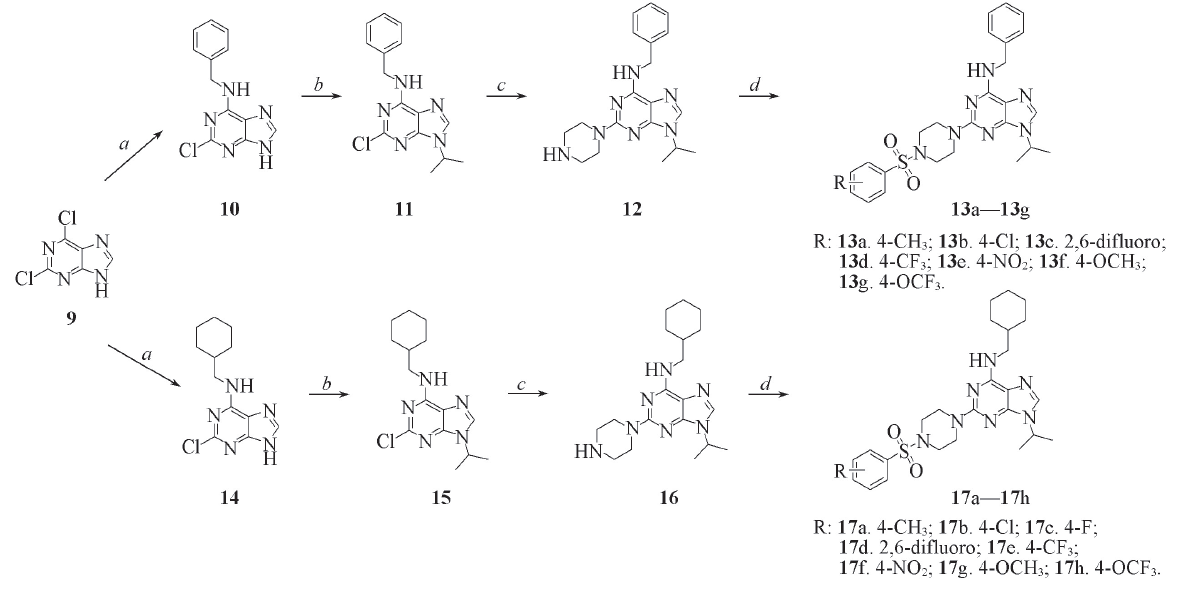
Scheme 2 Synthesis routes of target compounds 13a—13g and 17a—17ha. Benzyl amine or aminomethylcyclohexane, TEA, n-butanol, reflux; b. 2-bromopropane, K2CO3, DMSO, 80 ℃; c. piperazine, n-butanol, N2, 110 ℃; d. arylsulfonyl chloride, pyridine, r. t..
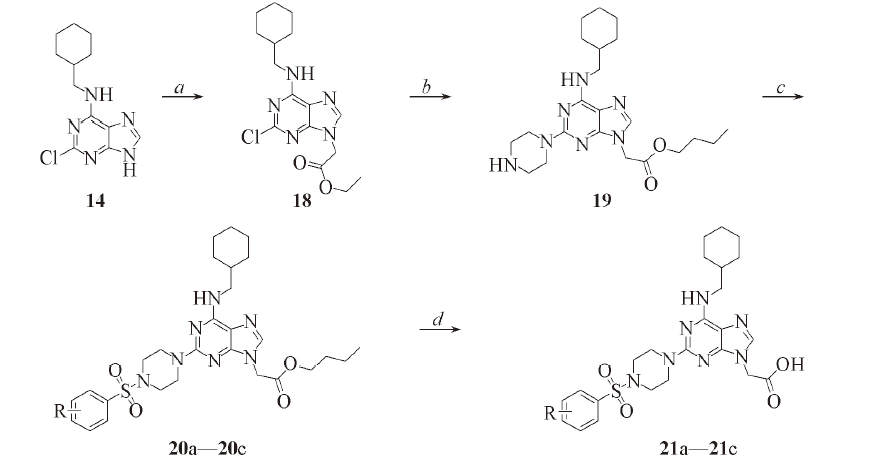
Scheme 3 Synthesis routes of target compounds 21a—21ca. Ethyl chloroacetate, K2CO3, DMSO, 90 ℃; b. piperazine, n-butanol, N2, 110 ℃; c. arylsulfonyl chloride, pyridine, r. t.; d. 2 mol/L NaOH, THF, r. t.. R: a. 4-F; b. 2,6-difluoro; c. 4-OCH3.
| Compd. | m.p./℃ | Yield(%) | Appearance | HRMS(calcd.)[M+H]+ |
|---|---|---|---|---|
| 5a | 254(Dec.) | 77 | White solid | 399.1034(399.1034) |
| 5b | 258(Dec.) | 65 | White solid | 417.0940(417.0940) |
| 5c | 286(Dec.) | 82 | White solid | 465.0955(465.0951) |
| 8a | 241—244 | 32 | Light pink solid | 459.1406(459.1409) |
| 8b | 215—218 | 24 | White solid | 491.1468(491.1472) |
| 8c | 245—246 | 60 | Light yellow solid | 468.1444(468.1449) |
| 8d | 206—208 | 5.5 | White solid | 453.1703(453.1703) |
| 13a | 86—90 | 39 | Light yellow solid | 506.2329(506.2333) |
| 13b | 80—83 | 42 | Light yellow solid | 526.1784(526.1786) |
| 13c | 108—111 | 59 | White solid | 528.1984(528.1988) |
| 13d | 86—88 | 65 | Gray solid | 560.2050(560.2050) |
| 13e | 226—228 | 28 | Brick red solid | 537.2023(537.2027) |
| 13f | 81—84 | 42 | Light yellow solid | 522.2279(522.2282) |
| 13g | 76—78 | 38 | Light yellow solid | 576.2000(576.1999) |
| 17a | 146—150 | 11 | White solid | 512.2803(512.2802) |
| 17b | 157—160 | 19 | White solid | 532.2254(532.2256) |
| 17c | 119—121 | 16 | White solid | 516.2548(516.2552) |
| 17d | 186—190 | 42 | White solid | 534.2454(534.2457) |
| 17e | 192—194 | 67 | White solid | 566.2520(566.2520) |
| 17f | 198—200 | 48 | Yellow solid | 543.2494(543.2496) |
| 17g | 148—150 | 15 | White solid | 528.2748(528.2751) |
| 17h | 167—168 | 59 | White solid | 582.2468(582.2469) |
| 21a | 256(Dec.) | 78 | White solid | 532.2130(532.2137) |
| 21b | 275(Dec.) | 47 | White solid | 550.2044(550.2043) |
| 21c | 228(Dec.) | 91 | White solid | 544.2326(544.2337) |
Table 1 Physical data and HRMS results of all target compounds
| Compd. | m.p./℃ | Yield(%) | Appearance | HRMS(calcd.)[M+H]+ |
|---|---|---|---|---|
| 5a | 254(Dec.) | 77 | White solid | 399.1034(399.1034) |
| 5b | 258(Dec.) | 65 | White solid | 417.0940(417.0940) |
| 5c | 286(Dec.) | 82 | White solid | 465.0955(465.0951) |
| 8a | 241—244 | 32 | Light pink solid | 459.1406(459.1409) |
| 8b | 215—218 | 24 | White solid | 491.1468(491.1472) |
| 8c | 245—246 | 60 | Light yellow solid | 468.1444(468.1449) |
| 8d | 206—208 | 5.5 | White solid | 453.1703(453.1703) |
| 13a | 86—90 | 39 | Light yellow solid | 506.2329(506.2333) |
| 13b | 80—83 | 42 | Light yellow solid | 526.1784(526.1786) |
| 13c | 108—111 | 59 | White solid | 528.1984(528.1988) |
| 13d | 86—88 | 65 | Gray solid | 560.2050(560.2050) |
| 13e | 226—228 | 28 | Brick red solid | 537.2023(537.2027) |
| 13f | 81—84 | 42 | Light yellow solid | 522.2279(522.2282) |
| 13g | 76—78 | 38 | Light yellow solid | 576.2000(576.1999) |
| 17a | 146—150 | 11 | White solid | 512.2803(512.2802) |
| 17b | 157—160 | 19 | White solid | 532.2254(532.2256) |
| 17c | 119—121 | 16 | White solid | 516.2548(516.2552) |
| 17d | 186—190 | 42 | White solid | 534.2454(534.2457) |
| 17e | 192—194 | 67 | White solid | 566.2520(566.2520) |
| 17f | 198—200 | 48 | Yellow solid | 543.2494(543.2496) |
| 17g | 148—150 | 15 | White solid | 528.2748(528.2751) |
| 17h | 167—168 | 59 | White solid | 582.2468(582.2469) |
| 21a | 256(Dec.) | 78 | White solid | 532.2130(532.2137) |
| 21b | 275(Dec.) | 47 | White solid | 550.2044(550.2043) |
| 21c | 228(Dec.) | 91 | White solid | 544.2326(544.2337) |
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
|---|---|---|---|
| 5aa,d | 4.53(br s, 2H, CH2), 7.15—7.18(m, 2H, ArH), 7.25—7.30(m, 5H, ArH), 7.90—7.95(m, 3H, ArH, purine C8), 8.25(br s, 1H, NH), 11.08(br s, 1H, SO2NH), 12.76(br s, 1H, purine C9) | 163.78(d,J=249 Hz), 153.33, 152.43, 150.55, 139.54, 138.64, 138.21, 129.80(d, J=5.25 Hz), 128.15, 127.34, 126.71, 115.60(d, J=22.5 Hz), 42.80 | |
| 5ba,d | 4.38(br s, 2H, CH2), 6.98—7.08(m, 2H, ArH), 7.20—7.27(m, 5H, ArH), 7.41—7.48(m, 1H, ArH), 7.99(s, 1H, purine C8), 8.32(br s, 1H, NH), 11.72(br s, 1H, SO2NH), 12.84(br s, 1H, purine C9) | 158.72(dd,J=4.5, 252.75 Hz), 153.14, 152.40, 149.49, 139.67, 138.64, 133.34, 128.25, 127.68, 126.98, 112,72(d, J=24 Hz), 42.76 | |
| 5ca,d | 4.49(br s, 2H, CH2), 7.25—7.31(m, 7H, ArH), 7.89—7.99(m, 3H, ArH, purine C8), 8.35(br s, 1H, NH) | 153.00, 152.84, 150.13, 141.68, 139.29, 129.19, 128.15, 127.32, 126.72, 120.54, 119.80(q, J=256.5 Hz), 111.54, 42.84 | |
| 8ab,d | 1.36[d, J=6.3 Hz, 6H, (CH3)2], 4.47—4.53(m, 3H, CH, CH2), 7.16(t, J=9.0 Hz, 2H, ArH), 7.20—7.29(m, 5H, ArH), 7.54—7.64(m, 1H, ArH), 8.21(s, 1H, purine C8), 8.46(br s, 1H, NH), 11.74(br s, 1H, SO2NH) | 158.87(d,J=255 Hz), 153.30, 152.63, 148.26, 139.12, 137.45, 134.91, 128.17, 127.52, 126.81, 118.94, 113.06(d, J=24 Hz), 113.05(t, J=11.25 Hz), 47.33, 42.73, 21.56 | |
| 8b b,d | 1.37[d, J=6.6 Hz, 6H, (CH3)2], 4.48—4.54(m, 3H, CH, CH2), 7.18—7.28(m, 5H, ArH), 7.81(d, J=7.2 Hz, 2H, ArH), 8.05(s, 1H, purine C8), 8.10(d, J=7.2 Hz, 2H, ArH), 8.36(br s, 1H, NH), 11.39(br s, 1H, SO2NH) | 154.12, 151.77, 148.66, 145.29, 139.66, 137.74, 131.94(q, J=31.5 Hz), 128.11, 128.01, 127.25, 126.64, 125.92, 123.43(q, J=271.5 Hz), 115.53, 46.58, 42.71, 21.73 | |
| 8cb,d | 1.41[d, J=6.3 Hz, 6H, (CH3)2], 4.50—4.52(m, 3H, CH, CH2), 7.16—7.22(m, 5H, ArH), 8.05(s, 1H, purine C8), 8.09(d, J=7.2 Hz, 2H, ArH), 8.18(d, J=7.2 Hz, 2H, ArH), 8.36(br s, 1H, NH), 11.49(br s, 1H, SO2NH) | 154.14, 151.62, 149.08, 147.06, 139.51, 137.71, 128.29, 127.95, 126.93, 126.48, 123.97, 115.78, 46.39, 42.59, 21.85 | |
| 8d c,d | 1.51[d, J=6.6 Hz, 6H, (CH3)2], 3.80(s, 3H, OCH3), 4.63(hept, J=6.6 Hz, 1H, CH), 4.72(br s, 2H, CH2), 6.52(br s, 1H, NH), 6.81(d, J=9.0 Hz, 2H, ArH), 7.26—7.32(m, 5H, ArH), 7.61(s, 1H, purine C8), 8.01(d, J=9.0 Hz, 2H, ArH), 8.24(br s, 1H, SO2NH) | 162.03, 154.29, 151.94, 149.12, 139.94, 137.41, 132.69, 129.44, 128.01, 127.37, 126.50, 115.84, 113.61, 59.64, 46.24, 42.58, 21.88 | |
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
| 13a a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.37(s, 3H, CH3), 2.75—2.78(m, 4H, piperazine), 3.74—3.82(m, 4H, piperazine), 4.50—4.52(m, 3H, CH, CH2), 7.22—7.32(m, 5H, ArH), 7.42(d, J=7.8 Hz, 2H, ArH), 7.58(d, J=7.8 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.11(br s, 1H, NH) | 158.80, 152.94, 150.20, 143.77, 139.13, 133.86, 132.47, 129.70, 128.31, 127.82, 127.61, 126.91, 109.51, 47.47, 45.85, 44.42, 43.72, 22.12, 21.04 | |
| 13b a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.81—2.88(m, 4H, piperazine), 3.75—3.80(m, 4H, piperazine), 4.50—4.55(m, 3H, CH, CH2), 7.21—7.32(m, 5H, ArH), 7.70(d, J=8.4 Hz, 2H, ArH), 7.72(d, J=8.4 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.09(br s, 1H, NH) | 158.74, 153.35, 150.50, 139.53, 139.08, 134.13, 133.89, 129.41, 129.15, 128.40, 127.60, 127.04, 111.01, 47.11, 45.79, 44.50, 43.77, 22.30 | |
| 13c a,d | 1.43[d, J=6.6 Hz, 6H, (CH3)2], 2.99—3.13(m, 4H, piperazine), 3.76—3.85(m, 4H, piperazine), 4.49—4.57(m, 3H, CH, CH2), 7.18—7.35(m, 7H, ArH), 7.75—7.77(m, 1H, ArH), 7.87(s, 1H, purine C8), 8.11(br s, 1H, NH) | 158.80(dd,J=3.75, 255 Hz), 157.34, 153.92, 150.21, 140.52, 136.21(t, J=11.25 Hz), 135.99, 127.90, 127.35, 126.37, 113.68(dd, J=3, 23.25 Hz), 113.35, 113.13, 45.67, 44.86, 43.40, 42.62, 21.97 | |
| 13d a,d | 1.43[d, J=6.6 Hz, 6H, (CH3)2], 2.79—3.03(m, 4H, piperazine), 3.71—3.89(m, 4H, piperazine), 4.45—4.61(m, 3H, CH, CH2), 7.19—7.32(m, 5H, ArH), 7.90(s, 1H, purine C8), 7.94(d, J=7.8 Hz, 2H, ArH), 8.02(d, J=7.8 Hz, 2H, ArH), 8.10(br s, 1H, NH) | 157.31, 153.85, 150.20, 140.50, 138.88, 135.99, 132.82(q, J=32.25 Hz), 128.47, 127.99, 127.37, 126.59(q, J=3.75 Hz), 126.40, 123.35(q, J=271.5 Hz), 113.45, 45.77, 45.45, 43.24, 42.65, 21.97 | |
| 13ea,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.87—2.93(m, 4H, piperazine), 3.76—3.80(m, 4H, piperazine), 4.48—4.55(m, 3H, CH, CH2), 7.21—7.31(m, 5H, ArH), 7.85(s, 1H, purine C8), 7.98(d, J=8.4 Hz, 2H, ArH), 8.10(br s, 1H, NH), 8.41(d, J=8.4 Hz, 2H, ArH) | 158.04, 154.34, 150.87, 150.22, 141.82, 139.21, 135.16, 128.89, 128.51, 127.51, 127.18, 124.31, 114.23, 46.42, 45.79, 44.45, 43.86, 22.48 | |
| 13f a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.75—2.78(m, 4H, piperazine), 3.75—3.80(m, 4H, piperazine), 4.49—4.52(m, 3H, CH, CH2), 7.13(d, J=8.4 Hz, 2H, ArH), 7.21—7.33(m, 5H, ArH), 7.64(d, J=8.4 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.12(br s, 1H, NH) | 163.11, 158.73, 153.25, 150.31, 139.27, 133.92, 129.90, 128.30, 127.62, 127.02, 126.87, 114.24, 110.44, 55.62, 47.16, 45.87, 44.38, 43.72, 22.21 | |
| 13g a,e | 1.42[d,J=6.6 Hz, 6H, (CH3)2], 2.84—2.90(m, 4H, piperazine), 3.76—3.81(m, 4H, piperazine), 4.50—4.56(m, 3H, CH, CH2), 7.19—7.32(m, 5H, ArH), 7.62(d, J=8.4 Hz, 2H, ArH), 7.86—7.88(m, 3H, purine C8, ArH), 8.10(br s, 1H, NH) | 158.68, 153.30, 152.36, 150.30, 139.24, 134.04, 133.96, 129.88, 128.31, 127.59, 126.90, 120.89, 120.19(q, J=258 Hz), 110.60, 47.19, 45.81, 44.40, 43.72, 22.22 | |
| 17ac,e | 0.96—1.01(m, 2H, cyclohexyl), 1.14—1.25(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.57—1.79(m, 6H, cyclohexyl), 2.41(s, 3H, CH3), 3.04—3.06(m, 4H, piperazine CH2), 3.35(br s, 2H, CH2), 3.90—3.92(m, 4H, piperazine CH2), 4.62(hept, J=6.6 Hz, 1H, CH), 5.68(br s, 1H, NH), 7.30(d, J=8.4 Hz, 2H, ArH), 7.53(s, 1H, purine C8), 7.65(d, J=8.4 Hz, 2H, ArH) | 158.30, 154.72, 150.65, 143.67, 134.70, 132.53, 129.65, 127.87, 114.02, 46.65, 46.33, 46.02, 43.85, 38.34, 31.00, 26.45, 25.94, 22.50, 21.50 | |
| 17b a,e | 0.88—0.90(m, 2H, cyclohexyl), 1.07—1.15(m, 3H, cyclohexyl), 1.46[d, J=6.6 Hz, 6H, (CH3)2], 1.57—1.68(m, 6H, cyclohexyl), 2.92—2.98(m, 4H, piperazine), 3.24(br s, 2H, CH2), 3.78—3.88(m, 4H, piperazine), 4.57(hept, J=6.6 Hz, 1H, CH), 7.45(br s, 1H, NH), 7.71(d, J=8.4 Hz, 2H, ArH), 7.76(d, J=8.4 Hz, 2H, ArH), 7.87(s, 1H, purine C8) | 158.22, 154.82, 150.63, 139.47, 134.81, 134.21, 129.37, 129.19, 114.25, 46.68, 46.33, 45.95, 43.84, 38.35, 31.01, 26.45, 25.94, 22.51 | |
| 17c a,e | 0.88—0.92(m, 2H, cyclohexyl), 1.12—1.19(m, 3H, cyclohexyl), 1.44[d, J=6.6 Hz, 6H, (CH3)2], 1.59—1.67(m, 6H, cyclohexyl), 2.91—2.97(m, 4H, piperazine), 3.22(br s, 2H, CH2), 3.77—3.83(m, 4H, piperazine), 4.54(hept, J=6.6 Hz, 1H, CH), 7.44—7.48(m, 3H, NH, ArH), 7.81—7.84(m, 3H, ArH, purine C8) | 165.28(d, J=253.5 Hz), 158.18, 154.74, 134.75, 131.82(d, J=3 Hz), 130.47(d, J=9 Hz), 116.33(d, J=22.5 Hz), 114.07, 46.65, 46.39, 45.97, 43.88, 38.36, 31.02, 26.46, 25.95, 22.50 | |
| 17dc,e | 0.97—1.03(m, 2H, cyclohexyl), 1.15—1.22(m, 3H, cyclohexyl), 1.54[d, J=6.6 Hz, 6H, (CH3)2], 1.62—1.81(m, 6H, cyclohexyl), 3.29—3.34(m, 4H, piperazine), 3.37(br s, 2H, CH2), 3.93—4.02(m, 4H, piperazine), 4.66(hept, J=6.6 Hz, 1H, CH), 6.22(br s, 1H, NH), 7.04(t, J=9.0 Hz, 2H, ArH), 7.50—7.54(m, 1H, ArH), 7.62(s, 1H, purine C8) | 159.88(dd,J=3.75, 258 Hz), 154.25, 152.36, 150.53, 134.68(t, J=11.25 Hz), 115.05(t, J=16.5 Hz), 113.23(dd, J=3.75, 24 Hz), 112.54, 47.76, 46.80, 45.54, 44.09, 38.22, 31.01, 26.43, 25.92, 22.42 | |
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
| 17ec,e | 0.97—1.01(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.51[d,J=6.6 Hz, 6H,(CH3)2], 1.57—1.79(m, 6H, cyclohexyl), 3.08—3.14(m, 4H, piperazine), 3.35(br s, 2H, CH2), 3.92—3.96(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.66(br s, 1H, NH), 7.52(s, 1H, purine C8), 7.79(d, J=8.4 Hz, 2H, ArH), 7.90(d, J=8.4 Hz, 2H, ArH) | 158.19, 154.88, 150.69, 139.47, 134.88, 134.57(q, J=33 Hz), 128.27, 126.22(q, J=3.75 Hz), 123.20(q, J=271.5 Hz), 114.35, 46.71, 46.33, 45.98, 43.85, 38.35, 31.02, 26.45, 25.95, 22.51 | |
| 17f c,e | 0.94—0.99(m, 2H, cyclohexyl), 1.14—1.26(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.53—1.77(m, 6H, cyclohexyl), 3.11—3.13(m, 4H, piperazine), 3.34(br s, 2H, CH2), 3.93—3.97(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.90(br s, 1H, NH), 7.51(s, 1H, purine C8), 7.95(d, J=9.0 Hz, 2H, ArH), 8.35(d, J=9.0 Hz, 2H, ArH) | 158.11, 154.88, 150.60, 150.22, 141.87, 134.92, 128.91, 124.31, 114.38, 46.71, 46.33, 45.89, 43.87, 38.34, 31.00, 26.44, 25.94, 22.50 | |
| 17g c,e | 0.96—1.02(m, 2H, cyclohexyl), 1.16—1.22(m, 3H, cyclohexyl), 1.52[d, J=6.6 Hz, 6H, (CH3)2], 1.67—1.80(m, 6H, cyclohexyl), 3.01—3.11(m, 4H, piperazine), 3.37(br s, 2H, CH2), 3.87(s, 3H, OCH3), 3.88—3.99(m, 4H, piperazine), 4.63(hept, J=6.6 Hz, 1H, CH), 5.72(br s, 1H, NH), 6.99(d, J=7.8 Hz, 2H, ArH), 7.51(s, 1H, purine C8), 7.72(d, J=7.8 Hz, 2H, ArH) | 163.07, 158.31, 154.80, 150.67, 134.75, 129.93, 127.15, 114.20, 55.59, 46.74, 46.29, 46.02, 43.84, 38.35, 31.00, 26.46, 25.95, 22.51 | |
| 17h c,e | 0.95—1.01(m, 2H, cyclohexyl), 1.14—1.24(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.55—1.78(m, 6H, cyclohexyl), 3.07—3.13(m, 4H, piperazine), 3.35(br s, 2H, CH2), 3.91—3.95(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.68(br s, 1H, NH), 7.34(d, J=8.4 Hz, 2H, ArH), 7.51(s, 1H, purine C8), 7.82(d, J=8.4 Hz, 2H, ArH) | 158.18, 154.81, 150.67, 139.45, 134.83, 134.14(q, J=33 Hz), 128.26, 126.22(q, J=3.75 Hz), 123.19(q, J=271.5 Hz), 114.25, 46.70, 46.35, 45.97, 43.85, 38.34, 31.01, 26.45, 25.94, 22.51 | |
| 21a a,d | 0.87—0.94(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.62—1.70(m, 6H, cyclohexyl), 2.85—3.05(m, 4H, piperazine), 3.24(br s, 2H, CH2), 3.70—3.89(m, 4H, piperazine), 4.77(s, 2H, CH2), 7.46—7.49(m, 3H, ArH, NH), 7.75(s, 1H, purine C8), 7.83(dd, J=7.2, 5.4 Hz, 2H, ArH), 13.12(br s, 1H, COOH) | 169.49, 164.64(d, J=250.5 Hz), 157.83, 154.44, 150.71, 138.31, 131.23, 130.58(d, J=9.75 Hz), 116.59(d, J=22.5 Hz), 112.66, 45.79, 45.63, 43.35, 43.18, 37.57, 30.53, 26.08, 25.45 | |
| 21ba,d | 0.85—0.98(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.62—1.71(m, 6H, cyclohexyl), 3.11—3.20(m, 4H, piperazine), 3.25(br s, 2H, CH2), 3.74—3.94(m, 4H, piperazine), 4.79(s, 2H, CH2), 7.33(t, J=9.0 Hz, 2H, ArH), 7.50(br s, 1H, NH), 7.70—7.86(m, 2H, ArH, purine C8), 13.12(br s, 1H, COOH) | 169.51, 158.92(dd, J=3.75, 255 Hz), 157.87, 154.48, 150.78, 138.34, 136.26(t, J=10.5 Hz), 113.71(dd, J=3, 24 Hz), 113.33, 112.77, 45.81, 45.00, 43.44, 43.35, 37.62, 30.54, 26.08, 25.46 | |
| 21ca,d | 0.86—0.94(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.61—1.67(m, 6H, cyclohexyl), 2.78—2.96(m, 4H, piperazine), 3.23(br s, 2H, CH2), 3.72—3.81(m, 4H, piperazine), 3.83(s, 3H, OCH3), 4.76(s, 2H, CH2), 7.13(d, J=7.8 Hz, 2H, ArH), 7.47(br s, 1H, NH), 7.68(d, J=7.8 Hz, 2H, ArH), 7.73(s, 1H, purine C8), 13.06(br s, 1H, COOH) | 169.50, 162.77, 157.79, 154.48, 150.83, 138.29, 129.74, 126.18, 114.50, 112.76, 55.62, 45.75, 45.61, 43.34, 43.21, 37.56, 30.54, 26.09, 25.47 | |
Table 2 Spectral date of all target compounds*
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
|---|---|---|---|
| 5aa,d | 4.53(br s, 2H, CH2), 7.15—7.18(m, 2H, ArH), 7.25—7.30(m, 5H, ArH), 7.90—7.95(m, 3H, ArH, purine C8), 8.25(br s, 1H, NH), 11.08(br s, 1H, SO2NH), 12.76(br s, 1H, purine C9) | 163.78(d,J=249 Hz), 153.33, 152.43, 150.55, 139.54, 138.64, 138.21, 129.80(d, J=5.25 Hz), 128.15, 127.34, 126.71, 115.60(d, J=22.5 Hz), 42.80 | |
| 5ba,d | 4.38(br s, 2H, CH2), 6.98—7.08(m, 2H, ArH), 7.20—7.27(m, 5H, ArH), 7.41—7.48(m, 1H, ArH), 7.99(s, 1H, purine C8), 8.32(br s, 1H, NH), 11.72(br s, 1H, SO2NH), 12.84(br s, 1H, purine C9) | 158.72(dd,J=4.5, 252.75 Hz), 153.14, 152.40, 149.49, 139.67, 138.64, 133.34, 128.25, 127.68, 126.98, 112,72(d, J=24 Hz), 42.76 | |
| 5ca,d | 4.49(br s, 2H, CH2), 7.25—7.31(m, 7H, ArH), 7.89—7.99(m, 3H, ArH, purine C8), 8.35(br s, 1H, NH) | 153.00, 152.84, 150.13, 141.68, 139.29, 129.19, 128.15, 127.32, 126.72, 120.54, 119.80(q, J=256.5 Hz), 111.54, 42.84 | |
| 8ab,d | 1.36[d, J=6.3 Hz, 6H, (CH3)2], 4.47—4.53(m, 3H, CH, CH2), 7.16(t, J=9.0 Hz, 2H, ArH), 7.20—7.29(m, 5H, ArH), 7.54—7.64(m, 1H, ArH), 8.21(s, 1H, purine C8), 8.46(br s, 1H, NH), 11.74(br s, 1H, SO2NH) | 158.87(d,J=255 Hz), 153.30, 152.63, 148.26, 139.12, 137.45, 134.91, 128.17, 127.52, 126.81, 118.94, 113.06(d, J=24 Hz), 113.05(t, J=11.25 Hz), 47.33, 42.73, 21.56 | |
| 8b b,d | 1.37[d, J=6.6 Hz, 6H, (CH3)2], 4.48—4.54(m, 3H, CH, CH2), 7.18—7.28(m, 5H, ArH), 7.81(d, J=7.2 Hz, 2H, ArH), 8.05(s, 1H, purine C8), 8.10(d, J=7.2 Hz, 2H, ArH), 8.36(br s, 1H, NH), 11.39(br s, 1H, SO2NH) | 154.12, 151.77, 148.66, 145.29, 139.66, 137.74, 131.94(q, J=31.5 Hz), 128.11, 128.01, 127.25, 126.64, 125.92, 123.43(q, J=271.5 Hz), 115.53, 46.58, 42.71, 21.73 | |
| 8cb,d | 1.41[d, J=6.3 Hz, 6H, (CH3)2], 4.50—4.52(m, 3H, CH, CH2), 7.16—7.22(m, 5H, ArH), 8.05(s, 1H, purine C8), 8.09(d, J=7.2 Hz, 2H, ArH), 8.18(d, J=7.2 Hz, 2H, ArH), 8.36(br s, 1H, NH), 11.49(br s, 1H, SO2NH) | 154.14, 151.62, 149.08, 147.06, 139.51, 137.71, 128.29, 127.95, 126.93, 126.48, 123.97, 115.78, 46.39, 42.59, 21.85 | |
| 8d c,d | 1.51[d, J=6.6 Hz, 6H, (CH3)2], 3.80(s, 3H, OCH3), 4.63(hept, J=6.6 Hz, 1H, CH), 4.72(br s, 2H, CH2), 6.52(br s, 1H, NH), 6.81(d, J=9.0 Hz, 2H, ArH), 7.26—7.32(m, 5H, ArH), 7.61(s, 1H, purine C8), 8.01(d, J=9.0 Hz, 2H, ArH), 8.24(br s, 1H, SO2NH) | 162.03, 154.29, 151.94, 149.12, 139.94, 137.41, 132.69, 129.44, 128.01, 127.37, 126.50, 115.84, 113.61, 59.64, 46.24, 42.58, 21.88 | |
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
| 13a a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.37(s, 3H, CH3), 2.75—2.78(m, 4H, piperazine), 3.74—3.82(m, 4H, piperazine), 4.50—4.52(m, 3H, CH, CH2), 7.22—7.32(m, 5H, ArH), 7.42(d, J=7.8 Hz, 2H, ArH), 7.58(d, J=7.8 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.11(br s, 1H, NH) | 158.80, 152.94, 150.20, 143.77, 139.13, 133.86, 132.47, 129.70, 128.31, 127.82, 127.61, 126.91, 109.51, 47.47, 45.85, 44.42, 43.72, 22.12, 21.04 | |
| 13b a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.81—2.88(m, 4H, piperazine), 3.75—3.80(m, 4H, piperazine), 4.50—4.55(m, 3H, CH, CH2), 7.21—7.32(m, 5H, ArH), 7.70(d, J=8.4 Hz, 2H, ArH), 7.72(d, J=8.4 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.09(br s, 1H, NH) | 158.74, 153.35, 150.50, 139.53, 139.08, 134.13, 133.89, 129.41, 129.15, 128.40, 127.60, 127.04, 111.01, 47.11, 45.79, 44.50, 43.77, 22.30 | |
| 13c a,d | 1.43[d, J=6.6 Hz, 6H, (CH3)2], 2.99—3.13(m, 4H, piperazine), 3.76—3.85(m, 4H, piperazine), 4.49—4.57(m, 3H, CH, CH2), 7.18—7.35(m, 7H, ArH), 7.75—7.77(m, 1H, ArH), 7.87(s, 1H, purine C8), 8.11(br s, 1H, NH) | 158.80(dd,J=3.75, 255 Hz), 157.34, 153.92, 150.21, 140.52, 136.21(t, J=11.25 Hz), 135.99, 127.90, 127.35, 126.37, 113.68(dd, J=3, 23.25 Hz), 113.35, 113.13, 45.67, 44.86, 43.40, 42.62, 21.97 | |
| 13d a,d | 1.43[d, J=6.6 Hz, 6H, (CH3)2], 2.79—3.03(m, 4H, piperazine), 3.71—3.89(m, 4H, piperazine), 4.45—4.61(m, 3H, CH, CH2), 7.19—7.32(m, 5H, ArH), 7.90(s, 1H, purine C8), 7.94(d, J=7.8 Hz, 2H, ArH), 8.02(d, J=7.8 Hz, 2H, ArH), 8.10(br s, 1H, NH) | 157.31, 153.85, 150.20, 140.50, 138.88, 135.99, 132.82(q, J=32.25 Hz), 128.47, 127.99, 127.37, 126.59(q, J=3.75 Hz), 126.40, 123.35(q, J=271.5 Hz), 113.45, 45.77, 45.45, 43.24, 42.65, 21.97 | |
| 13ea,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.87—2.93(m, 4H, piperazine), 3.76—3.80(m, 4H, piperazine), 4.48—4.55(m, 3H, CH, CH2), 7.21—7.31(m, 5H, ArH), 7.85(s, 1H, purine C8), 7.98(d, J=8.4 Hz, 2H, ArH), 8.10(br s, 1H, NH), 8.41(d, J=8.4 Hz, 2H, ArH) | 158.04, 154.34, 150.87, 150.22, 141.82, 139.21, 135.16, 128.89, 128.51, 127.51, 127.18, 124.31, 114.23, 46.42, 45.79, 44.45, 43.86, 22.48 | |
| 13f a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.75—2.78(m, 4H, piperazine), 3.75—3.80(m, 4H, piperazine), 4.49—4.52(m, 3H, CH, CH2), 7.13(d, J=8.4 Hz, 2H, ArH), 7.21—7.33(m, 5H, ArH), 7.64(d, J=8.4 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.12(br s, 1H, NH) | 163.11, 158.73, 153.25, 150.31, 139.27, 133.92, 129.90, 128.30, 127.62, 127.02, 126.87, 114.24, 110.44, 55.62, 47.16, 45.87, 44.38, 43.72, 22.21 | |
| 13g a,e | 1.42[d,J=6.6 Hz, 6H, (CH3)2], 2.84—2.90(m, 4H, piperazine), 3.76—3.81(m, 4H, piperazine), 4.50—4.56(m, 3H, CH, CH2), 7.19—7.32(m, 5H, ArH), 7.62(d, J=8.4 Hz, 2H, ArH), 7.86—7.88(m, 3H, purine C8, ArH), 8.10(br s, 1H, NH) | 158.68, 153.30, 152.36, 150.30, 139.24, 134.04, 133.96, 129.88, 128.31, 127.59, 126.90, 120.89, 120.19(q, J=258 Hz), 110.60, 47.19, 45.81, 44.40, 43.72, 22.22 | |
| 17ac,e | 0.96—1.01(m, 2H, cyclohexyl), 1.14—1.25(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.57—1.79(m, 6H, cyclohexyl), 2.41(s, 3H, CH3), 3.04—3.06(m, 4H, piperazine CH2), 3.35(br s, 2H, CH2), 3.90—3.92(m, 4H, piperazine CH2), 4.62(hept, J=6.6 Hz, 1H, CH), 5.68(br s, 1H, NH), 7.30(d, J=8.4 Hz, 2H, ArH), 7.53(s, 1H, purine C8), 7.65(d, J=8.4 Hz, 2H, ArH) | 158.30, 154.72, 150.65, 143.67, 134.70, 132.53, 129.65, 127.87, 114.02, 46.65, 46.33, 46.02, 43.85, 38.34, 31.00, 26.45, 25.94, 22.50, 21.50 | |
| 17b a,e | 0.88—0.90(m, 2H, cyclohexyl), 1.07—1.15(m, 3H, cyclohexyl), 1.46[d, J=6.6 Hz, 6H, (CH3)2], 1.57—1.68(m, 6H, cyclohexyl), 2.92—2.98(m, 4H, piperazine), 3.24(br s, 2H, CH2), 3.78—3.88(m, 4H, piperazine), 4.57(hept, J=6.6 Hz, 1H, CH), 7.45(br s, 1H, NH), 7.71(d, J=8.4 Hz, 2H, ArH), 7.76(d, J=8.4 Hz, 2H, ArH), 7.87(s, 1H, purine C8) | 158.22, 154.82, 150.63, 139.47, 134.81, 134.21, 129.37, 129.19, 114.25, 46.68, 46.33, 45.95, 43.84, 38.35, 31.01, 26.45, 25.94, 22.51 | |
| 17c a,e | 0.88—0.92(m, 2H, cyclohexyl), 1.12—1.19(m, 3H, cyclohexyl), 1.44[d, J=6.6 Hz, 6H, (CH3)2], 1.59—1.67(m, 6H, cyclohexyl), 2.91—2.97(m, 4H, piperazine), 3.22(br s, 2H, CH2), 3.77—3.83(m, 4H, piperazine), 4.54(hept, J=6.6 Hz, 1H, CH), 7.44—7.48(m, 3H, NH, ArH), 7.81—7.84(m, 3H, ArH, purine C8) | 165.28(d, J=253.5 Hz), 158.18, 154.74, 134.75, 131.82(d, J=3 Hz), 130.47(d, J=9 Hz), 116.33(d, J=22.5 Hz), 114.07, 46.65, 46.39, 45.97, 43.88, 38.36, 31.02, 26.46, 25.95, 22.50 | |
| 17dc,e | 0.97—1.03(m, 2H, cyclohexyl), 1.15—1.22(m, 3H, cyclohexyl), 1.54[d, J=6.6 Hz, 6H, (CH3)2], 1.62—1.81(m, 6H, cyclohexyl), 3.29—3.34(m, 4H, piperazine), 3.37(br s, 2H, CH2), 3.93—4.02(m, 4H, piperazine), 4.66(hept, J=6.6 Hz, 1H, CH), 6.22(br s, 1H, NH), 7.04(t, J=9.0 Hz, 2H, ArH), 7.50—7.54(m, 1H, ArH), 7.62(s, 1H, purine C8) | 159.88(dd,J=3.75, 258 Hz), 154.25, 152.36, 150.53, 134.68(t, J=11.25 Hz), 115.05(t, J=16.5 Hz), 113.23(dd, J=3.75, 24 Hz), 112.54, 47.76, 46.80, 45.54, 44.09, 38.22, 31.01, 26.43, 25.92, 22.42 | |
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
| 17ec,e | 0.97—1.01(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.51[d,J=6.6 Hz, 6H,(CH3)2], 1.57—1.79(m, 6H, cyclohexyl), 3.08—3.14(m, 4H, piperazine), 3.35(br s, 2H, CH2), 3.92—3.96(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.66(br s, 1H, NH), 7.52(s, 1H, purine C8), 7.79(d, J=8.4 Hz, 2H, ArH), 7.90(d, J=8.4 Hz, 2H, ArH) | 158.19, 154.88, 150.69, 139.47, 134.88, 134.57(q, J=33 Hz), 128.27, 126.22(q, J=3.75 Hz), 123.20(q, J=271.5 Hz), 114.35, 46.71, 46.33, 45.98, 43.85, 38.35, 31.02, 26.45, 25.95, 22.51 | |
| 17f c,e | 0.94—0.99(m, 2H, cyclohexyl), 1.14—1.26(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.53—1.77(m, 6H, cyclohexyl), 3.11—3.13(m, 4H, piperazine), 3.34(br s, 2H, CH2), 3.93—3.97(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.90(br s, 1H, NH), 7.51(s, 1H, purine C8), 7.95(d, J=9.0 Hz, 2H, ArH), 8.35(d, J=9.0 Hz, 2H, ArH) | 158.11, 154.88, 150.60, 150.22, 141.87, 134.92, 128.91, 124.31, 114.38, 46.71, 46.33, 45.89, 43.87, 38.34, 31.00, 26.44, 25.94, 22.50 | |
| 17g c,e | 0.96—1.02(m, 2H, cyclohexyl), 1.16—1.22(m, 3H, cyclohexyl), 1.52[d, J=6.6 Hz, 6H, (CH3)2], 1.67—1.80(m, 6H, cyclohexyl), 3.01—3.11(m, 4H, piperazine), 3.37(br s, 2H, CH2), 3.87(s, 3H, OCH3), 3.88—3.99(m, 4H, piperazine), 4.63(hept, J=6.6 Hz, 1H, CH), 5.72(br s, 1H, NH), 6.99(d, J=7.8 Hz, 2H, ArH), 7.51(s, 1H, purine C8), 7.72(d, J=7.8 Hz, 2H, ArH) | 163.07, 158.31, 154.80, 150.67, 134.75, 129.93, 127.15, 114.20, 55.59, 46.74, 46.29, 46.02, 43.84, 38.35, 31.00, 26.46, 25.95, 22.51 | |
| 17h c,e | 0.95—1.01(m, 2H, cyclohexyl), 1.14—1.24(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.55—1.78(m, 6H, cyclohexyl), 3.07—3.13(m, 4H, piperazine), 3.35(br s, 2H, CH2), 3.91—3.95(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.68(br s, 1H, NH), 7.34(d, J=8.4 Hz, 2H, ArH), 7.51(s, 1H, purine C8), 7.82(d, J=8.4 Hz, 2H, ArH) | 158.18, 154.81, 150.67, 139.45, 134.83, 134.14(q, J=33 Hz), 128.26, 126.22(q, J=3.75 Hz), 123.19(q, J=271.5 Hz), 114.25, 46.70, 46.35, 45.97, 43.85, 38.34, 31.01, 26.45, 25.94, 22.51 | |
| 21a a,d | 0.87—0.94(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.62—1.70(m, 6H, cyclohexyl), 2.85—3.05(m, 4H, piperazine), 3.24(br s, 2H, CH2), 3.70—3.89(m, 4H, piperazine), 4.77(s, 2H, CH2), 7.46—7.49(m, 3H, ArH, NH), 7.75(s, 1H, purine C8), 7.83(dd, J=7.2, 5.4 Hz, 2H, ArH), 13.12(br s, 1H, COOH) | 169.49, 164.64(d, J=250.5 Hz), 157.83, 154.44, 150.71, 138.31, 131.23, 130.58(d, J=9.75 Hz), 116.59(d, J=22.5 Hz), 112.66, 45.79, 45.63, 43.35, 43.18, 37.57, 30.53, 26.08, 25.45 | |
| 21ba,d | 0.85—0.98(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.62—1.71(m, 6H, cyclohexyl), 3.11—3.20(m, 4H, piperazine), 3.25(br s, 2H, CH2), 3.74—3.94(m, 4H, piperazine), 4.79(s, 2H, CH2), 7.33(t, J=9.0 Hz, 2H, ArH), 7.50(br s, 1H, NH), 7.70—7.86(m, 2H, ArH, purine C8), 13.12(br s, 1H, COOH) | 169.51, 158.92(dd, J=3.75, 255 Hz), 157.87, 154.48, 150.78, 138.34, 136.26(t, J=10.5 Hz), 113.71(dd, J=3, 24 Hz), 113.33, 112.77, 45.81, 45.00, 43.44, 43.35, 37.62, 30.54, 26.08, 25.46 | |
| 21ca,d | 0.86—0.94(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.61—1.67(m, 6H, cyclohexyl), 2.78—2.96(m, 4H, piperazine), 3.23(br s, 2H, CH2), 3.72—3.81(m, 4H, piperazine), 3.83(s, 3H, OCH3), 4.76(s, 2H, CH2), 7.13(d, J=7.8 Hz, 2H, ArH), 7.47(br s, 1H, NH), 7.68(d, J=7.8 Hz, 2H, ArH), 7.73(s, 1H, purine C8), 13.06(br s, 1H, COOH) | 169.50, 162.77, 157.79, 154.48, 150.83, 138.29, 129.74, 126.18, 114.50, 112.76, 55.62, 45.75, 45.61, 43.34, 43.21, 37.56, 30.54, 26.09, 25.47 | |
| Compd. | IC50 /(μmol·L-1) | Compd. | IC50 /(μmol·L-1) | ||||
|---|---|---|---|---|---|---|---|
| PC-3 | HCT116 | K562 | PC-3 | HCT116 | K562 | ||
| 13d | 23.4 | > 50 | > 100 | 17d | 19.1 | 27.3 | 33.1 |
| 13g | 39.7 | > 50 | > 100 | Roscovitine | 12.1 | 21.2 | 26.6 |
| 17c | > 50 | 26.7 | 47.6 | ||||
Table 3 Cytotoxicity of representative compounds in vitro
| Compd. | IC50 /(μmol·L-1) | Compd. | IC50 /(μmol·L-1) | ||||
|---|---|---|---|---|---|---|---|
| PC-3 | HCT116 | K562 | PC-3 | HCT116 | K562 | ||
| 13d | 23.4 | > 50 | > 100 | 17d | 19.1 | 27.3 | 33.1 |
| 13g | 39.7 | > 50 | > 100 | Roscovitine | 12.1 | 21.2 | 26.6 |
| 17c | > 50 | 26.7 | 47.6 | ||||
| [1] | Di Virgilio F., Cancer Res., 2012, 72(21), 5441—5447 |
| [2] | Legraverend M., Grierson D. S., Bioorg. Med. Chem., 2006, 14(12), 3987—4006 |
| [3] | Chen Z., Kwong A. K. Y., Yang Z. J., Zhang L. R., Lee H. C., Zhang L. H., Chem. J. Chinese Universities, 2012, 33(6), 1226—1232 |
| (陈哲, KWONG Anna Ka-Yee, 杨振军, 张亮仁, LEE Hon Cheung, 张礼和.高等学校化学学报, 2012,33(6), 1226—1232) | |
| [4] | Huang Y., Wang J., Guo G. Y., Tao Z., Xue S. F., Zhu Q. J., Zhou Q. D., Chem. J. Chinese Universities, 2013, 34(2), 375—380 |
| (黄英, 王娟, 郭改英, 陶朱, 薛赛凤, 祝黔江, 周清娣.高等学校化学学报, 2013,34(2), 375—380) | |
| [5] | Lakshman M. K., Kumar A., Balachandran R., Day B. W., Andrei G., Snoeck R., Balzarini J., J. Org. Chem., 2012, 77(14), 5870—5883 |
| [6] | Vijay Kumar D., Hoarau C., Bursavich M., Slattum P., Gerrish D., Yager K., Saunders M., Shenderovich M., Roth B. L., McKinnon R., Chan A., Cimbora D. M., Bradford C., Reeves L., Patton S., Papac D. I., Williams B. L., Carlson R. O., Bioorg. Med. Chem. Lett., 2012, 22(13), 4377—4385 |
| [7] | Blanchard S., Soh C. K., Lee C. P., Poulsen A., Bonday Z., Goh K. L., Goh K. C., Goh M. K., Pasha M. K., Wang H., Williams M., Wood J. M., Ethirajulu K., Dymock B. W., Bioorg. Med. Chem. Lett., 2012, 22(8), 2880—2884 |
| [8] | Piccoli M., Palazzolo G., Conforti E., Lamorte G., Papini N., Creo P., Fania C., Scaringi R., Bergante S., Tringali C., Roncoroni L., Mazzoleni S., Doneda L., Galli R., Venerando B., Tettamanti G., Gelfi C., Anastasia L., J. Cell Biochem., 2012, 113(10), 3207—3217 |
| [9] | Kim S. H., Bajji A., Tangallapally R., Markovitz B., Trovato R., Shenderovich M., Baichwal V., Bartel P., Cimbora D., McKinnon R., J. Med. Chem., 2012, 55(17), 7480—7501 |
| [10] | Yenugonda V. M., Deb T. B., Grindrod S. C., Dakshanamurthy S., Yang Y., Paige M., Brown M. L., Bioorg. Med. Chem., 2011, 19(8), 2714—2725 |
| [11] | Korycka A., Lech-Maranda E., Robak T., Recent Pat. Anti-Cancer Drug Discovery, 2008, 3(2), 123—136 |
| [12] | Meijer L., Borgne A., Mulner O., Chong J. P., Blow J. J., Inagaki N., Inagaki M., Delcros J. G., Moulinoux J. P., Eur. J. Biochem., 1997, 243(1/2), 527—536 |
| [13] | Mgbonyebi O., Russo J., Russo I., Anticancer Res., 1998, 18(2A), 751—755 |
| [14] | Popowycz F., Fournet G., Schneider C., Bettayeb K., Ferandin Y., Lamigeon C., Tirado O. M., Mateo-Lozano S., Notario V., Colas P., J. Med. Chem., 2009, 52(3), 655—663 |
| [15] | Jorda R., Paruch K., Krystof V., Curr. Pharm. Des., 2012, 18(20), 2974—2980 |
| [16] | Wang Q., Su L., Liu N., Zhang L., Xu W., Fang H., Curr. Med. Chem., 2011, 18(13), 2025—2043 |
| [17] | Syed S. A. S., Rivera G., Ashfaq M., Mini-Rev. Med. Chem., 2013, 13(1), 70—86 |
| [18] | Kim H. S., Ohno M., Xu B., Kim H. O., Choi Y., Ji X. D., Maddileti S., Marquez V. E., Harden T. K., Jacobson K. A., J. Med. Chem., 2003, 46(23), 4974—4987 |
| [19] | Nair N., Kudo W., Smith M. A., Abrol R., Goddard W. A., Reddy V. P., Bioorg. Med. Chem. Lett., 2011, 21(13), 3957—3961 |
| [20] | Oumata N., Ferandin Y., Meijer L., Galons H., Org. Process Res. Dev., 2009, 13(3), 641—644 |
| [21] | Wang J., Wang Q., Zhang L., Fang H., Chin. J. Chem., 2013, 31(9), 1181—1191 |
| [1] | XIAO Yanhua, ZHANG Guangjie, ZONG Liang, LIU Guohong, REN Lijun, DONG Junxing. Chemical Constituents and Antitumor Activity of Tupistra chinensis † [J]. Chem. J. Chinese Universities, 2019, 40(9): 1897. |
| [2] | LÜ Mingjun,LI Wen,YANG Xinying,FANG Hao. Synthesis and Antitumor Activity of N9 Position Aromatic Substituted Purine-8-one Derivatives† [J]. Chem. J. Chinese Universities, 2019, 40(2): 254. |
| [3] | FANG Fang,XUE Liangmin,CONG Jing,TIAN Chao,WANG Xiaowei,LIU Junyi,ZHANG Zhili. Synthesis and Anti-tumor Activity Evaluation of a Series of 2- or 4-Substituted Pyrido[3,2-d]pyrimidines as Nonclassical Antifolates † [J]. Chem. J. Chinese Universities, 2019, 40(10): 2111. |
| [4] | ZHANG Peiquan,YANG Qianqian,LONG Huidan,CHEN Xin. Synthesis and Antitumor Activity of Auranofin Derivatives † [J]. Chem. J. Chinese Universities, 2019, 40(10): 2097. |
| [5] | BAI Xinfa, MA Xuan, XIE Xiaoxia, SHAO Mingsha, GUO Ningning, YAN Ning, YAO Lei. Synthesis and Anti-tumor Activity of Tubulysins Analogues† [J]. Chem. J. Chinese Universities, 2017, 38(1): 47. |
| [6] | GUO Liang, CAO Rihui, FAN Wenxi, GAN Ziyun, MA Qin. Design, Synthesis and in vitro Antitumor Activities of Novel Bivalent β-Carbolines† [J]. Chem. J. Chinese Universities, 2016, 37(6): 1093. |
| [7] | ZHANG Jie, ZHOU Changjian, XIE Jianwei, DAI Bin. Synthesis and Antitumor Activities of Rhein-Valine Adducts† [J]. Chem. J. Chinese Universities, 2016, 37(12): 2159. |
| [8] | ZHOU Hao, DUAN Zhigang, ZHAO Shuang, BAO Meiying, LI Zhiwei, PEI Yazhong. Design and Synthesis of Phenylpyrimidine and Their Anticancer Activity† [J]. Chem. J. Chinese Universities, 2015, 36(9): 1694. |
| [9] | XU Jian, WEI Mengxue, LI Guoming, LI Xueqiang. Synthesis and Anti-tumor Activities of Novel Artemisone-piperazine-sulfonamide Derivatives† [J]. Chem. J. Chinese Universities, 2015, 36(5): 919. |
| [10] | WANG Gang, HAN Leiqiang, FANG Hao. Syntheses and Antitumor Activities of Phenylpiperazine Derivatives† [J]. Chem. J. Chinese Universities, 2015, 36(12): 2435. |
| [11] | GUO Hua, YANG Chengling, WANG Wei, LAI Quanyong, YUAN Zhi. Preparation of Liver-targeted Nano-prodrug Based on Sodium Alginate Derivative and the Study on Antitumor Activity† [J]. Chem. J. Chinese Universities, 2014, 35(8): 1835. |
| [12] | SONG Xiudao, HE Jun, MA Jin, LIU Yunmei, ZHENG Xing, LEI Xiaoyong, GUO Yu. Syntheses and Anticancer Activities of Glycine Derivatives of Chrysin† [J]. Chem. J. Chinese Universities, 2014, 35(7): 1465. |
| [13] | GUO Liang, CAO Rihui, FAN Wenxi, MA Qin. Synthesis and Biological Evaluation of 1,2,7,9-Tetrasubstituted Harmine Derivatives as Potential Antitumor Agents† [J]. Chem. J. Chinese Universities, 2014, 35(3): 518. |
| [14] | YANG Haikui, XU Wanfu, DUAN Anna, YOU Wenwei, ZHAO Peiliang. Syntheses and Biological Activities of Novel Imine and Imide Derivatives Bearing 1,2,4-Triazole Moiety† [J]. Chem. J. Chinese Universities, 2014, 35(3): 555. |
| [15] | YANG Hongliang, XU Guoxing, BAO Meiying, ZHANG Dapeng, LI Zhiwei, PEI Yazhong. Design and Synthesis of Pyridinylisoxazoles and Their Anticancer Activities† [J]. Chem. J. Chinese Universities, 2014, 35(12): 2584. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||