

高等学校化学学报 ›› 2022, Vol. 43 ›› Issue (1): 20210579.doi: 10.7503/cjcu20210579
初明月1,李峰博1,高宁1,杨昕2,于婷婷2,马慧媛1,杨桂欣1,庞海军1
收稿日期:2021-08-16
出版日期:2022-01-10
发布日期:2021-09-16
基金资助:
CHU Mingyue1, LI Fengbo1, GAO Ning1, YANG Xin2, YU Tingting2, MA Huiyuan1( ), YANG Guixin1, PANG Haijun1
), YANG Guixin1, PANG Haijun1
Received:2021-08-16
Online:2022-01-10
Published:2021-09-16
Contact:
MA Huiyuan
E-mail:mahy017@163.com
Supported by:摘要:
利用自组装和电沉积交替的方法制备了基于磷钨酸盐K28Li5H7P8W48O184·92H2O(P8W48)、 碳纳米管和Ni纳米颗粒的复合膜电极, 用于NO2- 的检测. 由于复合膜中P8W48, CNTs和Ni纳米颗粒3种活性成分的协同作用, 所制备的传感器表现出低的检出限、 宽的线性范围和较高的选择性. 将该传感器用于检测果汁中的NO2- , 所得到的回收率在允许的误差范围内. 这种复合膜电极传感器有望在实际应用中高度灵敏地检测NO2- .
中图分类号:
TrendMD:
初明月, 李峰博, 高宁, 杨昕, 于婷婷, 马慧媛, 杨桂欣, 庞海军. 轮型多金属氧酸盐复合物膜的制备及在检测亚硝酸盐中的应用. 高等学校化学学报, 2022, 43(1): 20210579.
CHU Mingyue, LI Fengbo, GAO Ning, YANG Xin, YU Tingting, MA Huiyuan, YANG Guixin, PANG Haijun. Construction of a Coronal Polyoxometalate-based Composite Film for Determination of Nitrite. Chem. J. Chinese Universities, 2022, 43(1): 20210579.
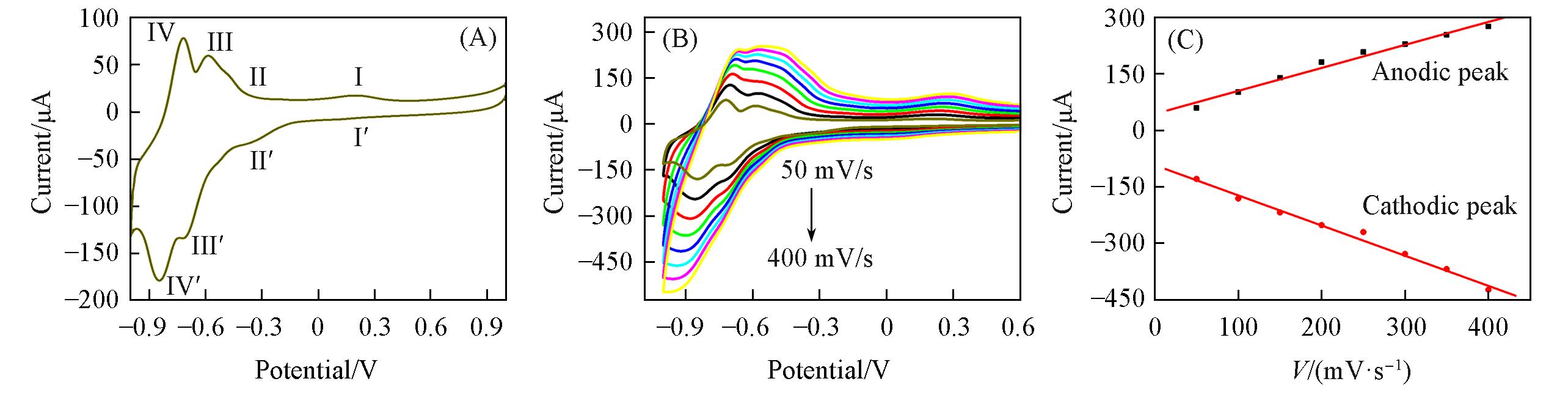
Fig.4 CVs of the {PEI/P8W48/CNTs?CS/Ni/P8W48} composite film in 0.2 mol/L PBS(pH=7.0) at 50 mV/s(A), and from 50 mV/s to 400 mV/s(step 50 mV/s)(B) and plots of the cathodic and anodic peak currents of waves against the scan rate(C)
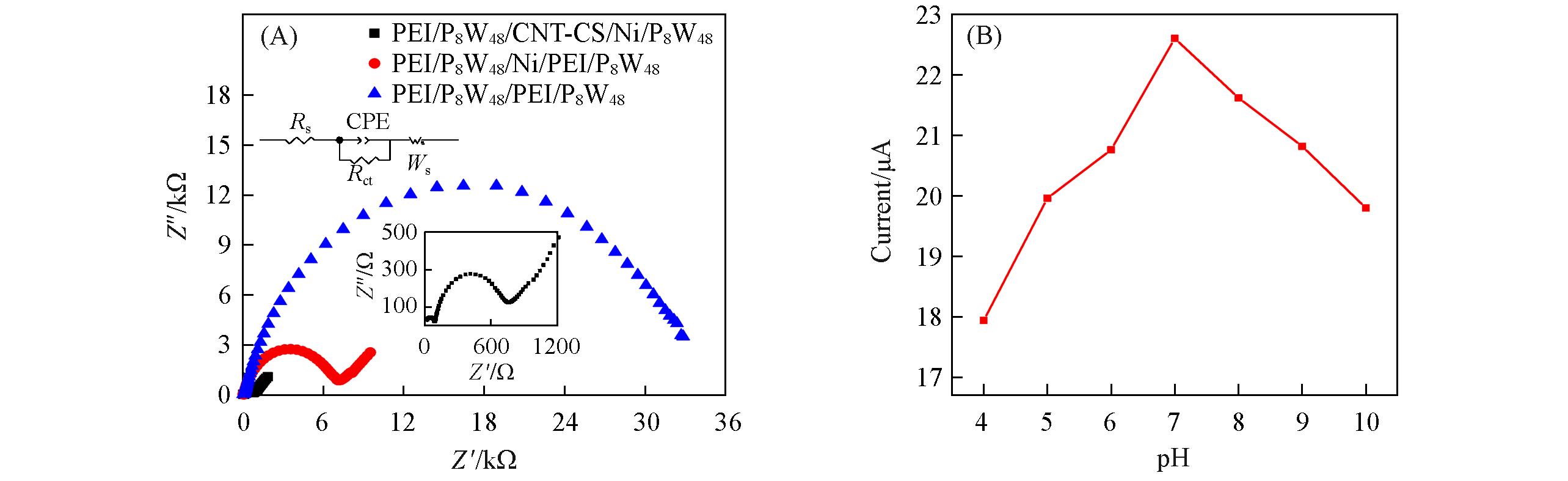
Fig.5 EIS plots of different films(A) and influence of pH on the peak current(Ipa) of {PEI/P8W48/CNTs?CS/Ni/P8W48}(B)The inset is an partially enlarged view of the Nyquist impedance plots of {PEI/P8W48/CNTs?CS/Ni/P8W48}.
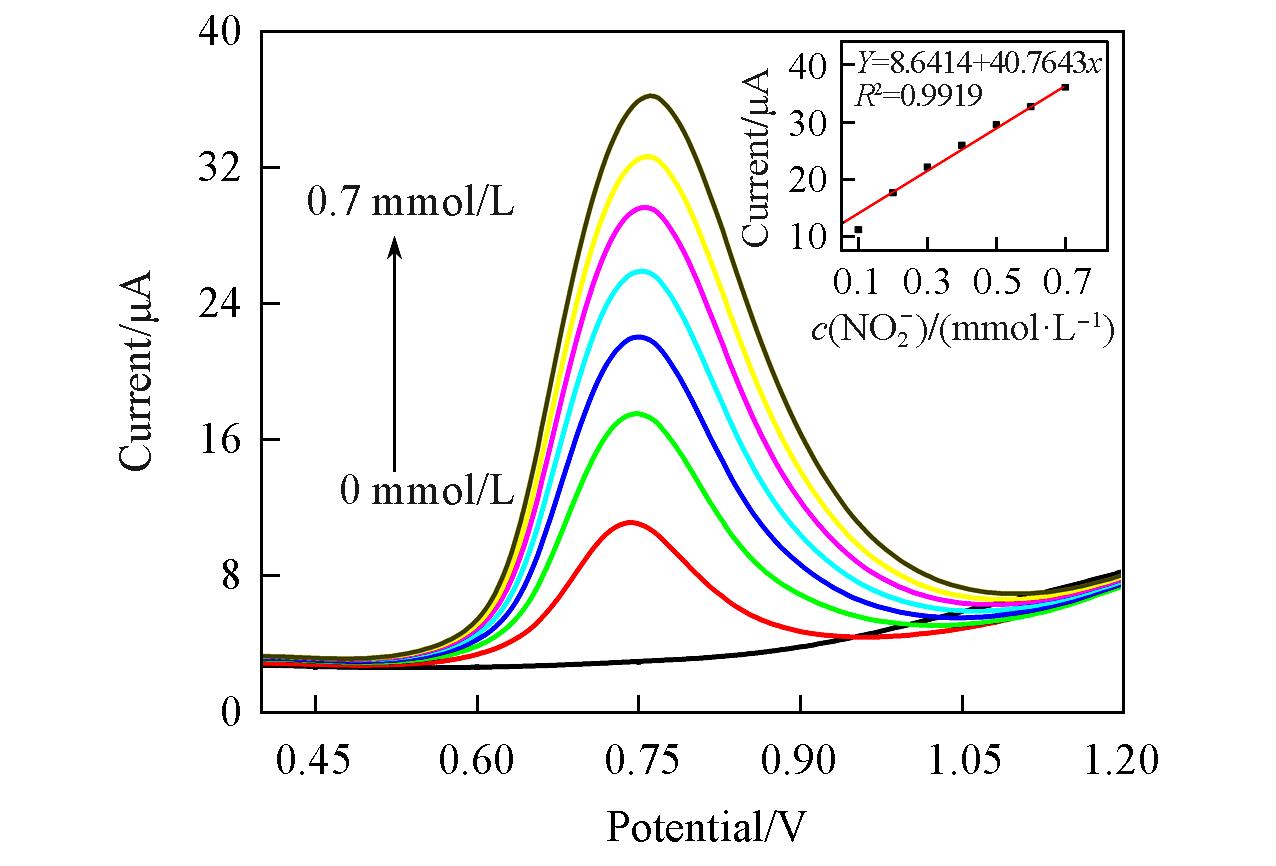
Fig.6 Differential pulse voltammograms of {PEI/P8W48/CNTs?CS/Ni/P8W48} in 0.2 mol/L PBS(pH=7.0) containing different concentrations of NO2-c(NO2- )=0—0.7 mmol/L, step 0.1 mmol/L. The inset shows the relationship between catalytic current and concentration of NO2- in 0.2 mol/L PBS(pH=7.0).
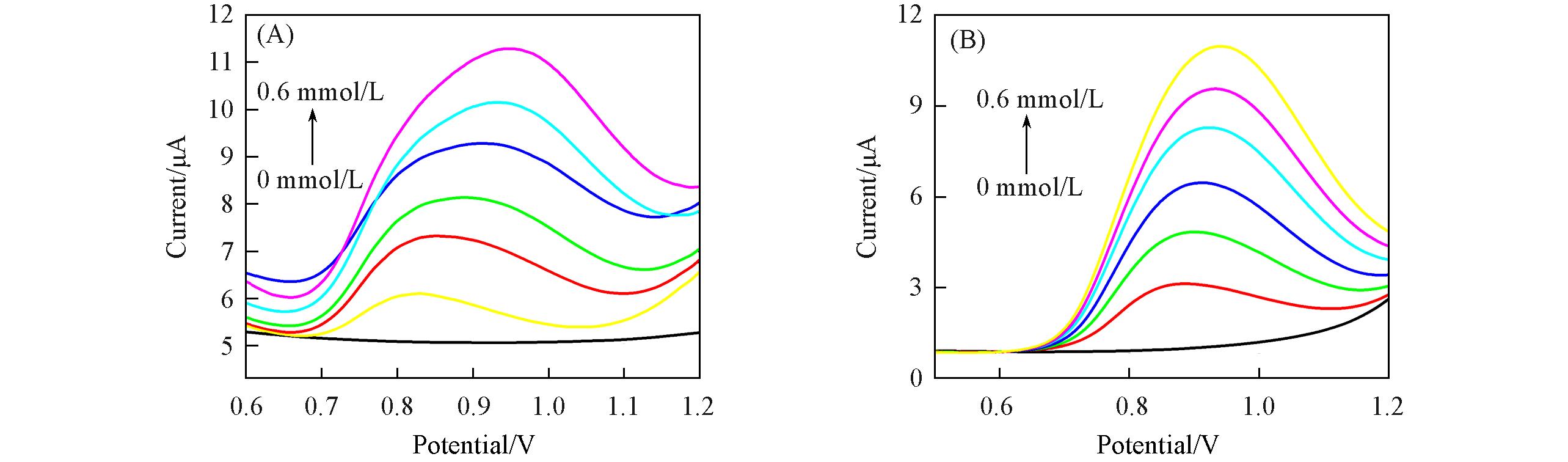
Fig.7 DPVs of the {PEI//P8W48/PEI//P8W48}(A) and {PEI//P8W48/Ni/PEI//P8W48}(B) composite films in 0.2 mol/L PBS(pH=7.0) containing different concentrations of NO2-c(NO2- )=0—0.6 mmol/L, step 0.1 mmol/L.
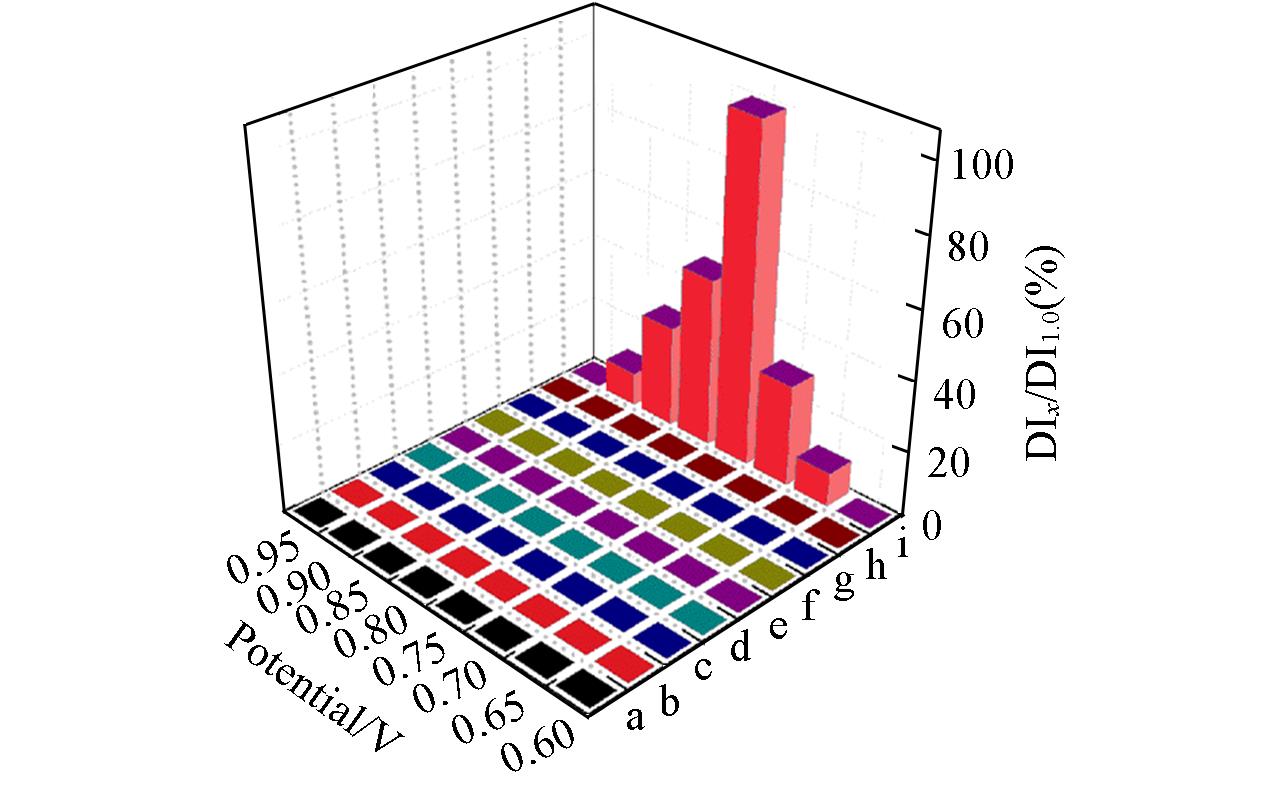
Fig.8 Anti?interference performance of the {PEI//P8W48/CNTs?CS/Ni/P8W48} composite film obtained with glucose, acetic acid, citric acid, KBrO3, KIO3, Na2CO3, KCl, fructose and NaNO2 at different applied potentials in 0.2 mol/L PBS(pH=7.0)a. Glucose; b. acetic acid; c. citric acid; d. KBrO3; e. KClO3; f. Na2CO3; g. KCl; h. fructose; i. NaNO2. DI1.0: NO2- oxidation peak current at 0.75 V; DIx: oxidation peak current values of different interferences at different voltages.
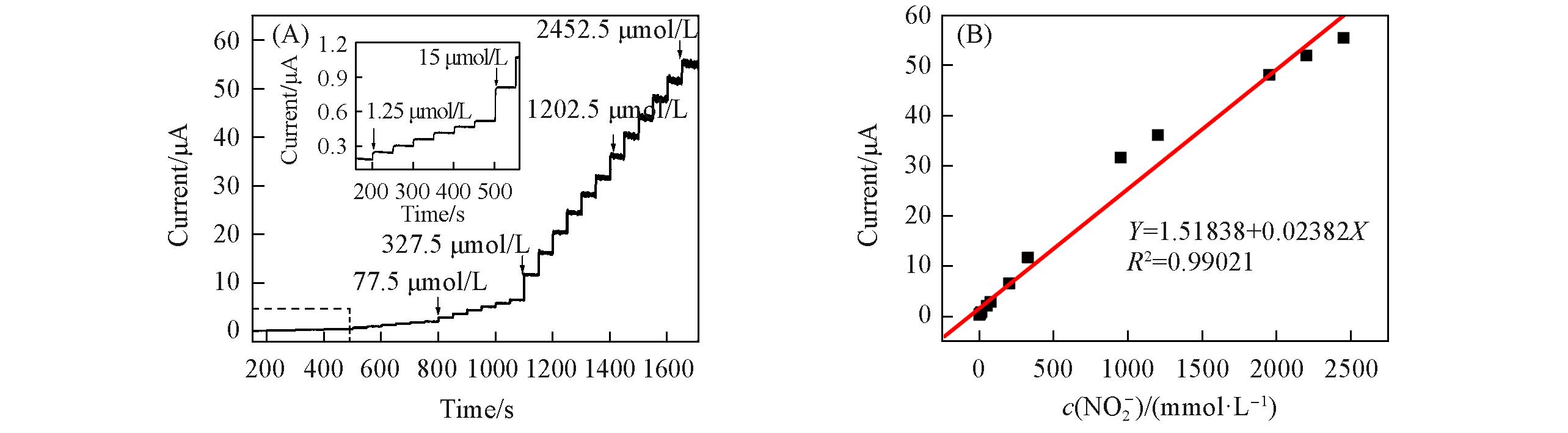
Fig.9 i?t curve of the {PEI/P8W48/CNTs?CS/Ni/P8W48} composite film during successive additions of NO2- at 0.75 V(A), and calibration plot of current and concentration of NO2- obtained at the {PEI/P8W48/CNTs?CS/Ni/P8W48} composite film(B)
| Electrode | Working potential/V | Linear range/(μmol·L-1) | LOD/(μmol·L-1) | Ref. |
|---|---|---|---|---|
| CG/Au@Ag/GCE | 0.95 | 2.5—1250 | 0.15 | [ |
| Fe3O4@Au@Cys/rGO/GCE | 0.75 | 0.03—344 | 0.008 | [ |
| 344—2215 | ||||
| LIG/f?MWCNT?AuNPs | 0.58 | 10—140 | 0.9 | [ |
| GO?PANI?AuNPs/GCE | 1.1 | 0.5—240 | 0.17 | [ |
| 240—2580 | ||||
| NPG/GCE | 0.75 | 20—400 | 0.36 | [ |
| Cu2+?Cu+/Biochar/GCE | 0.75 | 1—300 | 0.63 | [ |
| Ag/Halloysite nanotube/MoS2/CPE | 0.80 | 2—425 | 0.7 | [ |
| PEI/P8W48/CNTs?CS/Ni/P8W48/GCE | 0.75 | 1.25—2500 | 0.02 | This work |
Table 1 Comparison of the sensor performance between the prepared sensor and other sensors for the determination of nitrite
| Electrode | Working potential/V | Linear range/(μmol·L-1) | LOD/(μmol·L-1) | Ref. |
|---|---|---|---|---|
| CG/Au@Ag/GCE | 0.95 | 2.5—1250 | 0.15 | [ |
| Fe3O4@Au@Cys/rGO/GCE | 0.75 | 0.03—344 | 0.008 | [ |
| 344—2215 | ||||
| LIG/f?MWCNT?AuNPs | 0.58 | 10—140 | 0.9 | [ |
| GO?PANI?AuNPs/GCE | 1.1 | 0.5—240 | 0.17 | [ |
| 240—2580 | ||||
| NPG/GCE | 0.75 | 20—400 | 0.36 | [ |
| Cu2+?Cu+/Biochar/GCE | 0.75 | 1—300 | 0.63 | [ |
| Ag/Halloysite nanotube/MoS2/CPE | 0.80 | 2—425 | 0.7 | [ |
| PEI/P8W48/CNTs?CS/Ni/P8W48/GCE | 0.75 | 1.25—2500 | 0.02 | This work |
| Sample | cNO2- added/(μmol·L-1) | cNO2- obtained/(μmol·L-1) | Recovery(%) |
|---|---|---|---|
| 1 | 25 | 24.94 | 99.76 |
| 2 | 50 | 49.89 | 99.78 |
| 3 | 75 | 74.9 | 99.87 |
| 4 | 100 | 99.89 | 99.89 |
| 5 | 125 | 124.97 | 99.98 |
| 6 | 250 | 250.03 | 100.01 |
Table 2 Results of the recovery tests obtained for determination of NO2- in real sample
| Sample | cNO2- added/(μmol·L-1) | cNO2- obtained/(μmol·L-1) | Recovery(%) |
|---|---|---|---|
| 1 | 25 | 24.94 | 99.76 |
| 2 | 50 | 49.89 | 99.78 |
| 3 | 75 | 74.9 | 99.87 |
| 4 | 100 | 99.89 | 99.89 |
| 5 | 125 | 124.97 | 99.98 |
| 6 | 250 | 250.03 | 100.01 |
| 1 | Dou B. T., Yan J., Chen Q., Han X. J., Feng Q. M., Miao X. M., Wang P., Sensors. Actuat. B: Chem., 2021, 328(1), 129082 |
| 2 | Liu X. R., Huang X. D., Wu Y. X., Xu Q. X., Du M. T., Wang D. B., Yang Q., Liu Y. W., Ni B. J., Yang G. J., Yang F., Wang Q. L., Chem. Eng. J., 2020, 387, 124174 |
| 3 | da Silva S. M., Mazo L. H., Electroanalysis, 1998, 10(17), 1200—1203 |
| 4 | Jakszyn P., González C. A., World J. Gastroenterol.,2006, 12(27), 4296—4303 |
| 5 | Najm I., Trussell R. R., J. Am. Water Works Assoc., 2001, 93(2), 92—99 |
| 6 | Nakatani N., Kozaki D., Mori M., Tanaka K., Anal. Sci., 2012, 28(9), 845—852 |
| 7 | Jobgen W. S., Jobgen S. C., Li H., Meininger C. J., Wu G. Y., J. Chromatogr. B, 2007, 851(1/2), 71—82 |
| 8 | Vishnuvardhan V., Kala R., Rao T. P., Anal. Chim. Acta, 2008, 623(1), 53—58 |
| 9 | Filik H., Giray D., Ceylan B., Apak R., Talanta, 2011, 85(4), 1818—1824 |
| 10 | Luiz V. H. M., Pezza L., Pezza H. R., Food Chem., 2012, 134(4), 2546—2551 |
| 11 | Tsikas D., J. Chromatogr. B, 2007, 851(1/2), 51—70 |
| 12 | Senra⁃Ferreiro S., Pena⁃Pereira F., Lavilla I., Bendicho C., Anal. Chim. Acta, 2010, 668(2), 195—200 |
| 13 | Gupta S., Prakash R., RSC Adv., 2014, 4(15), 7521—7527 |
| 14 | Zhang D., Ma H. Y., Chen Y. Y., Pang H. J., Yan Y., Anal. Chim. Acta, 2013, 792, 35—44 |
| 15 | Moheimanian N., Raoof J. B., Safavi A., Ojani R., J. Iran. Chem. Soc., 2014, 11(4), 1217—1222 |
| 16 | Wei W., Jin H. H., Zhao G. C., Microchim. Acta, 2009, 164(1), 167—171 |
| 17 | Zhou L., Wang J. P., Gai L., Li D. J., Li Y. B., Sensor. Actuat. B: Chem., 2013, 181, 65—70 |
| 18 | De Marco R., Clarke G., Pejcic B., Electroanalysis, 2007, 19(19/20), 1987—2001 |
| 19 | Xiao F., Liu L., Li J., Zeng Z. Z., Zeng B. Z., Electroanalysis, 2008, 20(18), 2047—2054 |
| 20 | Manea F., Remes A., Radovan C., Pode R., Picken S., Schoonman J., Talanta, 2010, 83(1), 66—71 |
| 21 | Pal M., Ganesan V., Analyst, 2010, 135(10), 2711—2716 |
| 22 | Wang Z., Liao F., Guo T., Yang S. W., Zeng C. M., J. Electroanal. Chem., 2012, 664, 135—138 |
| 23 | Xiao F., Mo Z., Zhao F., Zeng B. Z., Electrochem. Commun., 2008, 10(11), 1740—1743 |
| 24 | Navratil T., Kopanica M., Crit. Rev. Anal. Chem., 2002, 32(2), 153—166 |
| 25 | Hill C. L., Chem. Rev., 1998, 98(1), 1—2 |
| 26 | Keita B., Zhang G. J., Dolbecq A., Mialane P., Secheresse F., Miserque F., J. Phys. Chem. C, 2007, 111, 8145—8148 |
| 27 | Tang Z. Y., Liu S. Q., Wang E., Dong S. J., Langmuir, 2000, 16(13), 5806—5813 |
| 28 | Armatas G. S., Bilis G., Louloudi M., J. Mater. Chem., 2011, 21(9), 2997—3005 |
| 29 | Kang L., Ma H. Y., Yu Y., Pang H. J., Song Y. B., Zhang D., Sensors. Actuat. B: Chem., 2013, 177, 270—278 |
| 30 | Wang Y. P., Jiang F. C., Chen J. F., Sun X. F., Xian T., Yang H., Nanomaterials, 2020, 10, 178 |
| 31 | Chen J. H., Li W. Z., Wang D. Z., Yang S. X., Wen J. G., Ren Z. F., Carbon, 2002, 40(8), 1193—1197 |
| 32 | Isari A. A., Mehregan M., Mehregan S. Hayati F., Kalantary R. R., Kakavandi B., J. Hazard. Mater., 2020, 390, 122050 |
| 33 | Goh P. S., Ismail A. F., Ng B. C., Desalination, 2013, 308, 2—14 |
| 34 | Li X., Chen S., Li L., Quan X., Zhao H. M., J. Colloid Interface Sci., 2014, 415, 159—164 |
| 35 | Li X., Chen S., Quan X., Zhang Y. B., Enviro. Sci. Technol., 2011, 45(19), 8498—8505 |
| 36 | Liu H., Zuo K., Vecitis C. D., Enviro. Sci. Technol., 2014, 48(23), 13871—13879 |
| 37 | Zhang M., Smith A., Gorski W., Anal. Chem., 2004, 76(17), 5045—5050 |
| 38 | Wu Z., Feng W., Feng Y., Liu Q., Xu Y. H., Sekino T., Fujii A., Ozaki M., Carbon, 2007, 45(6), 1212—1218 |
| 39 | Koch M., Nicolaescu R., Kamat P. V., J. Phys. Chem. C, 2009, 113(27), 11507—11513 |
| 40 | Walter E. C., Ng K., Zach M. P., Favier F., Microelectronic Eng., 2002, 61, 555—561 |
| 41 | Oliveira M. M., Castro E. G., Canestraro C. D., Zanchet D., Ugarte D., Roman L. S., Zarbin A. J. G., J. Phys. Chem. B, 2006, 110(34), 17063—17069 |
| 42 | Cordente N., Amiens C., Chaudret B., J. Appl. Phys., 2003, 94(10), 6358—6365 |
| 43 | Guskos N., Maryniak M., Typek J., Podsiadly P., Narkiewicz U., Senderek E., Roslaniec Z., J. Non⁃cryst. Solids, 2009, 355(24—27), 1400—1404 |
| 44 | Wang S. F., Xie F., Hu R. F., Anal. Bioanal. Chem., 2007, 387(3), 933—939 |
| 45 | Metin O., Mazumder V., Ozkar S., Sun S. H., J. Am. Chem. Soc., 2010, 132(5), 1468—1469 |
| 46 | Huang X. H., Tu J. P., Zhang B., Li Y., Yuan Y. F., Wu H. M., J. Power Sources, 2006, 161(1), 541—544 |
| 47 | Hosono E., Fujihara S., Honma I., Zhou H. S., Electrochem. Commun., 2006, 8(2), 284—288 |
| 48 | Zayim E. O., Turhan I., Tepehan F. Z., Ozer N., Sol. Energ. Mat. Sol. C., 2008, 92(2), 164—169 |
| 49 | Garcia⁃Miquel J. L., Zhang Q., Allen S. J., Rougier A., Blyr A., Davies H. O., Jones A. C., Leedham T. J., Williams P. A., Impey S. A., Thin Solid Films, 2003, 424(2), 165—170 |
| 50 | Ojani R., Raoof J. B., Zavvarmahalleh S. R. H., Electrochim. Acta, 2008, 53(5), 2402—2407 |
| 51 | Xu C., Hu Y., Rong J., Jiang S. P., Liu Y. L., Electrochem. Commun., 2007, 9(8), 2009—2012 |
| 52 | Niu X., Lan M., Zhao H., Chen C., Anal. Chem., 2013, 85(7), 3561—3569 |
| 53 | Sattarahmady N., Heli H., Vais R. D., Biosensors. Bioelectron., 2013, 48, 197—202 |
| 54 | Aydoğdu G., Zeybek D. K., Zeybek B., Pekyardimci S., J. Appl. Electrochem., 2013, 43(5), 523—531 |
| 55 | Lu L. M., Zhang L., Qu F. L., Lu H. X., Zhang X. B., Wu Z. S., Huan S. Y., Wang Q. A., Shen G. L., Yu R. Q., Biosensors Bioelectron., 2009, 25(1), 218—223 |
| 56 | Chen H., Zhang Z., Cai R., Rao W., Long F., Electrochim. Acta, 2014, 117, 385—392 |
| 57 | Yu H. C., Huang X. Y., Lei F. H., Tian X. C., Wei Y. C., Li H., Electrochim. Acta, 2014, 141, 45—50 |
| 58 | Couto G. G., Klein J. J., Schreiner W. H., Mosca D. H., de Oliveira A. J. A., Zarbin A. J. G., J. Coll. Inter. Sci., 2007, 311(2), 461—468 |
| 59 | Neiva E. G. C., Bergamini M. F., Oliveira M. M., Zarbin A. J. G., Sensor. Actuat. B: Chem., 2014, 196, 574—581 |
| 60 | Qiu S., Wu L., Shi G., Zhang L., Chen H. L., Gao C. J., Ind. Eng. Chem. Res., 2010, 49(22), 11667—11675 |
| 61 | Contant R., Teze A., Inorg. Chem., 1985, 24(26), 4610—4614 |
| 62 | Legrand J., Taleb A., Gota S., Guittet M. J., Petit C., Langmuir, 2002, 18(10), 4131—4137 |
| 63 | Sciortino L., Giannici F., Martorana A., Ruggirello A. M., Liveri V. T., Portale G., Casaletto M. P., Longo A., J. Phys. Chem. C, 2011, 115(14), 6360—6366 |
| 64 | Liu S., Kurth D. G., Bredenkötter B., Volkmer D.,. J. Am. Chem. Soc., 2002, 124(41), 12279—12287 |
| 65 | Bao Z. L., Zhong H., Li X. R., Zhang A. R., Liu Y. X., Chen P., Cheng Z. P., Qian H. G., Sensors. Actuat. B: Chem., 2021, 345, 130319 |
| 66 | Riahifar V., Haghnazari N., Keshavarzi F., Nasri F., Microchem. J.,2021, 166, 106217 |
| 67 | Nasraoui S., Al⁃Hamry A., Teixeira P. R., Ameur S., Paterno L. G., Air M. B., Kanoun O., J. Electroanal. Chem., 2021, 880, 114893 |
| 68 | Chen G. Z., Zheng J. B., Microchem. J., 2021, 164, 106034 |
| 69 | Wang H. M., Zhang X. L., Wang S. J., Ma H. Y., Shen Y. L., Wang X., J. Aoac. Int., 2021, 104(3), 860—866 |
| 70 | Gao L. P., Kang Z. W., Ding Q., Zhang X. H., Lin H. T., Lin M. S., Yang D. P., Sci. Total Environ., 2020, 733, 138008—138016 |
| 71 | Ghanei⁃Motlagh M., Taher M. A., Biosensors Bioelectron., 2018, 109, 279—285 |
| [1] | 赵润瑶, 纪桂鹏, 刘志敏. 吡咯氮配位单原子铜催化剂的电催化二氧化碳还原性能[J]. 高等学校化学学报, 2022, 43(7): 20220272. |
| [2] | 高京, 何文涛, 王欣欣, 向宇姝, 龙丽娟, 秦舒浩. DOPO衍生物改性碳纳米管的制备及对聚乳酸阻燃性能的影响[J]. 高等学校化学学报, 2022, 43(3): 20210670. |
| [3] | 闫文卿, 张则尧, 李彦. 碳纳米管透明导电薄膜的可控制备[J]. 高等学校化学学报, 2022, 43(3): 20210626. |
| [4] | 刘杰, 李金晟, 柏景森, 金钊, 葛君杰, 刘长鹏, 邢巍. 降低直接甲醇燃料电池浓差极化的含磺化碳管阻水夹层的构建[J]. 高等学校化学学报, 2022, 43(11): 20220420. |
| [5] | 丁钦, 张梓轩, 徐培程, 李晓宇, 段莉梅, 王寅, 刘景海. Cu, Ni, Co掺杂对Fe碳纳米管的结构及电催化性能的影响[J]. 高等学校化学学报, 2022, 43(11): 20220421. |
| [6] | 侯从聪, 王惠颖, 李婷婷, 张志明, 常春蕊, 安立宝. N-CNTs/NiCo-LDH复合材料的制备及电化学性能[J]. 高等学校化学学报, 2022, 43(10): 20220351. |
| [7] | 许晓坚, 李博, 林猛枭, 詹硕. 多孔碳基复合膜的真空冷冻干燥制备及高效太阳能水蒸发性能[J]. 高等学校化学学报, 2022, 43(10): 20220361. |
| [8] | 张太文, 郭军, 张丹, 袁常梅, 邱双艳. trz-Cl-Cu-PMo12的合成、 表征及催化氧化碘离子性能[J]. 高等学校化学学报, 2022, 43(10): 20220215. |
| [9] | 蒋君, 宫田田, 张成鹏, 刘晓倩, 赵俊伟. 吡啶二羧酸修饰的稀土嵌入碲钨酸盐的合成及电化学生物识别性质[J]. 高等学校化学学报, 2022, 43(1): 20210561. |
| [10] | 陈慧娜, 李新雄, 郑寿添. 铌多酸三维框架材料的研究进展[J]. 高等学校化学学报, 2022, 43(1): 20210625. |
| [11] | 梁宇, 刘欢, 宫丽阁, 王春晓, 王春梅, 于凯, 周百斌. 联咪唑修饰{SiW12O40}杂化物的合成及超级电容性能[J]. 高等学校化学学报, 2022, 43(1): 20210556. |
| [12] | 赵凌云, 黄汉雄, 罗杜宇, 苏逢春. 复合材料柔软性对倒金字塔微结构阵列传感器性能的影响[J]. 高等学校化学学报, 2021, 42(9): 2953. |
| [13] | 吴同华, 岳喜贵, 梅笑寒, 梁留博, 彭鑫, 马友美, 张淑玲. 三明治结构多壁碳纳米管/聚醚醚酮电磁屏蔽复合材料的制备[J]. 高等学校化学学报, 2021, 42(8): 2627. |
| [14] | 梁平平, 刘帅, 李红艺, 丁亚丹, 温晓琨, 刘俊平, 洪霞. PVDF⁃CNT自漂浮多孔微珠的制备及在高效太阳能驱动界面水蒸发中的应用[J]. 高等学校化学学报, 2021, 42(8): 2689. |
| [15] | 徐梦祎, 黄雪雯, 李小杰, 魏玮, 刘晓亚. “串珠状”复合纳米组装体修饰丝网印刷电极构建的生物传感器[J]. 高等学校化学学报, 2021, 42(6): 1768. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||