

高等学校化学学报 ›› 2020, Vol. 41 ›› Issue (9): 1917.doi: 10.7503/cjcu20200409
• 庆祝《高等学校化学学报》复刊40周年专栏 • 上一篇 下一篇
收稿日期:2020-07-01
出版日期:2020-09-10
发布日期:2020-09-02
通讯作者:
王博
E-mail:bowang@bit.edu.cn
基金资助:Received:2020-07-01
Online:2020-09-10
Published:2020-09-02
Contact:
WANG Bo
E-mail:bowang@bit.edu.cn
Supported by:摘要:
共价有机框架(COFs)材料是有机构筑基元通过共价键连接而形成的晶态有机多孔材料. COFs具有孔道结构规整、 及比表面积高等特点, 被广泛地应用于气体储存与分离、 催化、 传感、 储能及光电转化等领域. 将具有可调吸光能力的有机构筑基元引入到COFs中, 可使其展现出强大的光催化潜力. 近年来, COFs在光催化领域中发展迅猛. 本文总结了COFs在光催化产氢、 光催化二氧化碳还原、 光催化有机反应以及光催化污染物降解等方面的研究进展, 并展望了其在光催化领域的应用前景.
中图分类号:
TrendMD:
李丽, 李鹏飞, 王博. 共价有机框架材料在光催化领域中的应用. 高等学校化学学报, 2020, 41(9): 1917.
LI Li, LI Pengfei, WANG Bo. Photocatalytic Application of Covalent Organic Frameworks. Chem. J. Chinese Universities, 2020, 41(9): 1917.
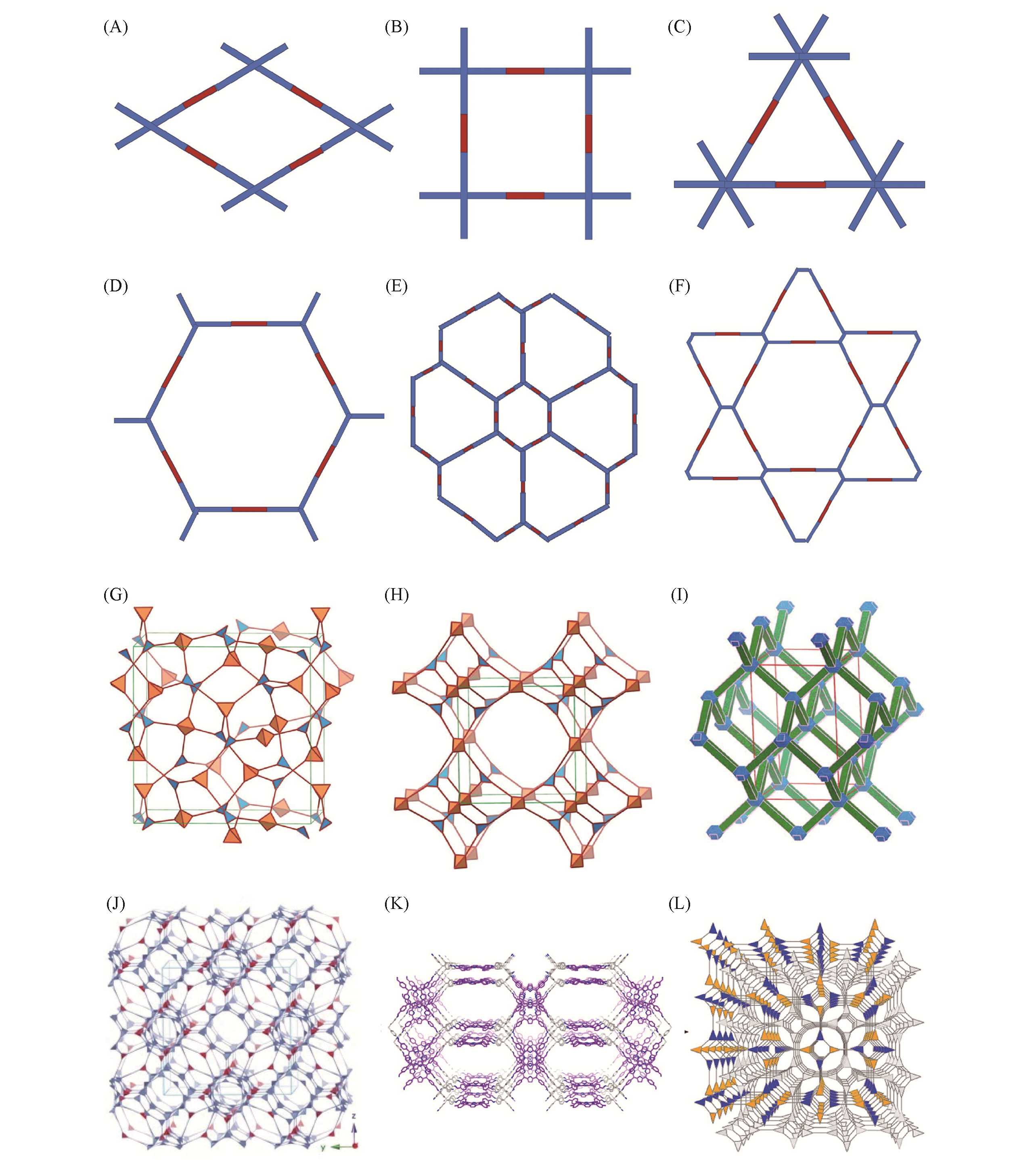
Fig.1 Topological structures of 2D(A—F) and 3D(G—L)[15—19](G)—(H) Copyright 2007, American Association for the Advancement of Science; (I) Copyright 2015, American Chemical Society; (J) Copyright 2017, Wiley-VCH; (K) Copyright 2017, American Chemical Society; (L) Copyright 2018, American Chemical Society.
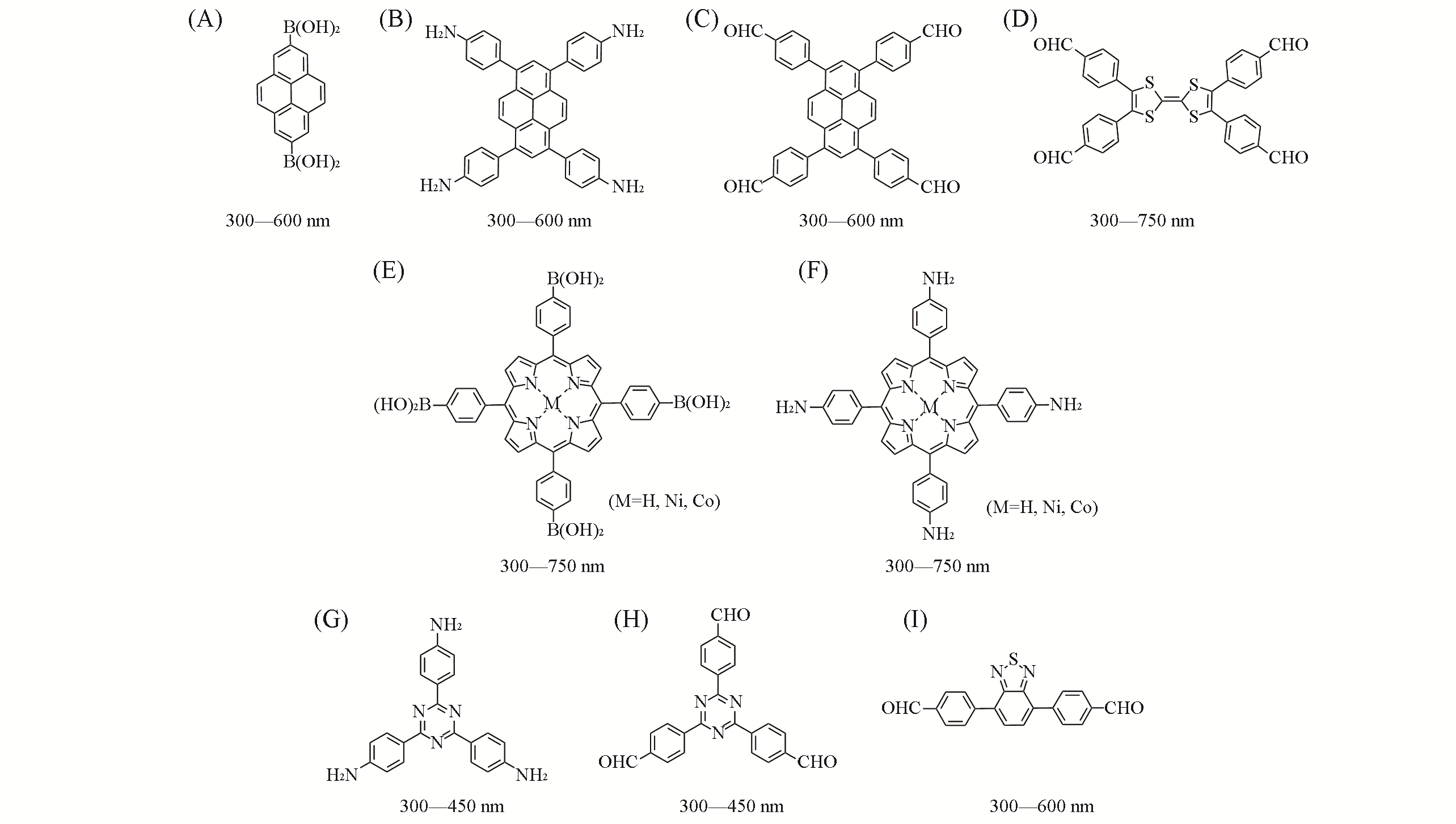
Fig.2 Common monomers with adjustable light absorption capacity and their absorption rangesThe absorption ranges are estimated from the synthesized COF. (A)—(D) Electron?rich monomers;(E)—(I) electron?deficiency monomers.
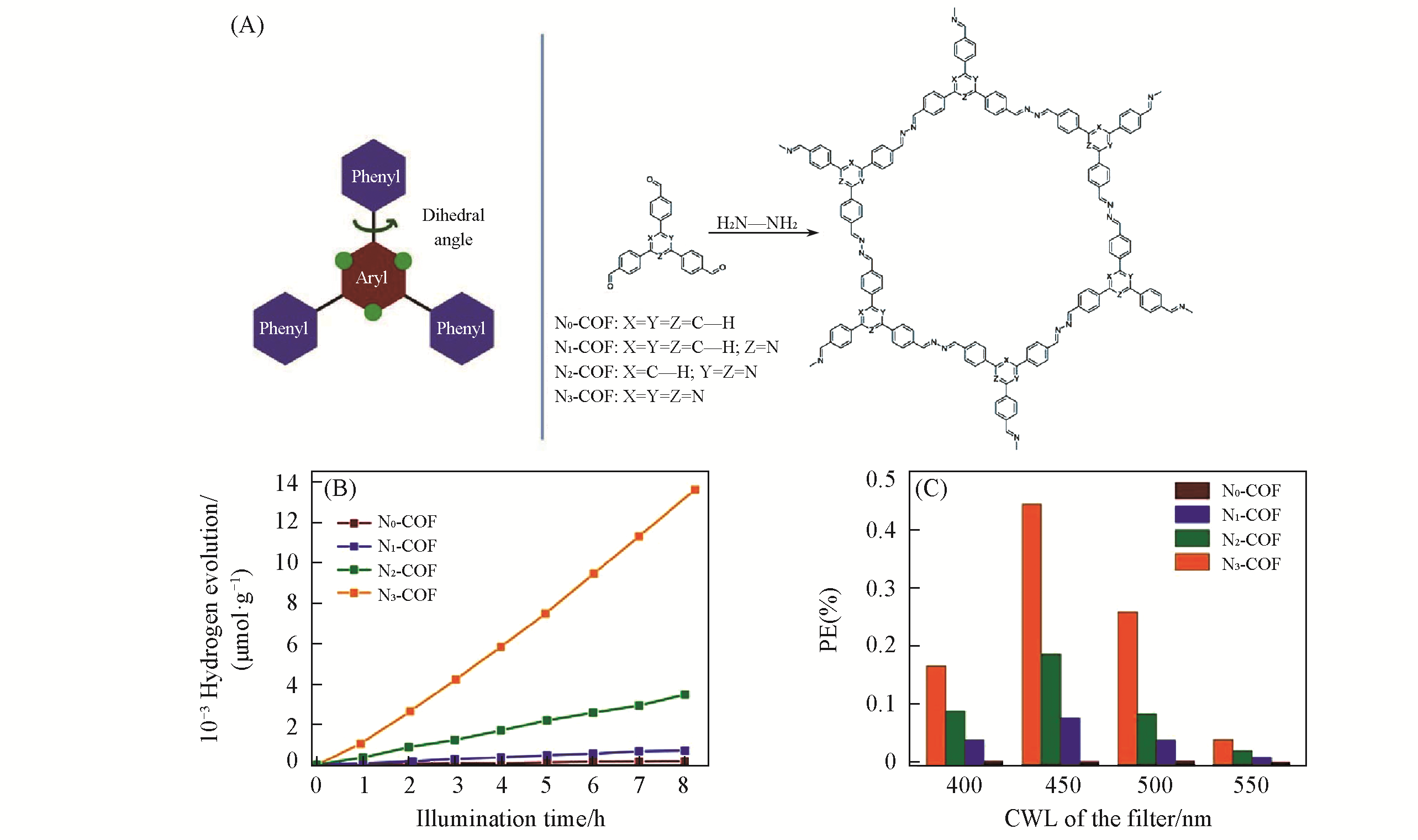
Fig.3 Synthesis and property of Nx?COF(A) Synthesis and structure of Nx-COF; (B) hydrogen evolution of Nx-COF over illumination time; (C) photonic efficiency(PE) of hydrogen evolution at different central wavelength(CWL)[34].Copyright 2015, Springer Nature.
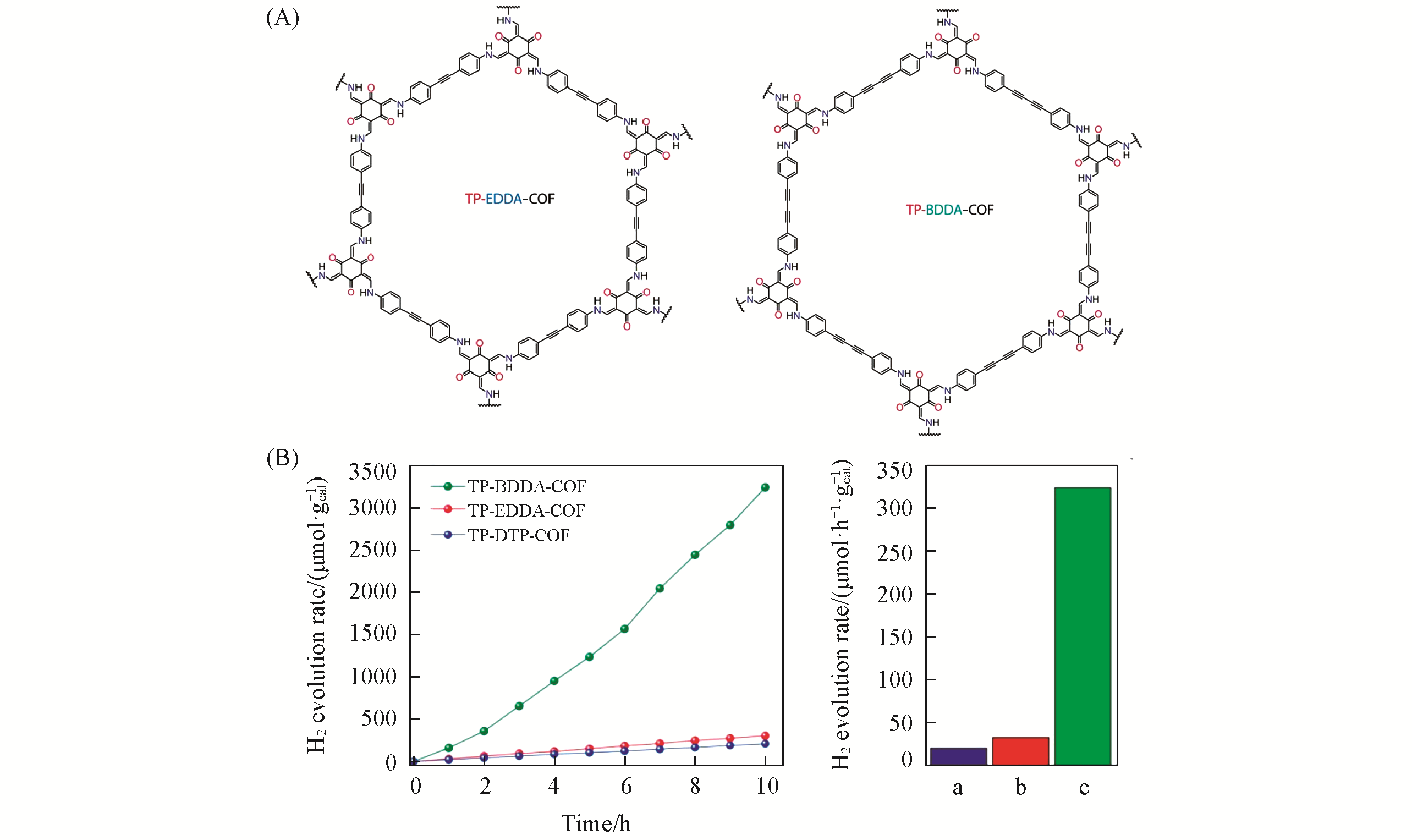
Fig.4 Synthesis and property of TP?EDDA?COF and TP?BDDA?COF[37](A) Synthesis of TP-EDDA-COF and TP-BDDA-COF; (B) photocatalytic hydrogen evolution performance of TP-BDDA-COF, TP-EDDA and TP-DTP-COF, under visible light irradiation(≥395 nm). a. TP-DTP-COF; b. TP-EDDA-COF; c. TP-BDDA-COF. Copyright 2018, American Chemical Society.
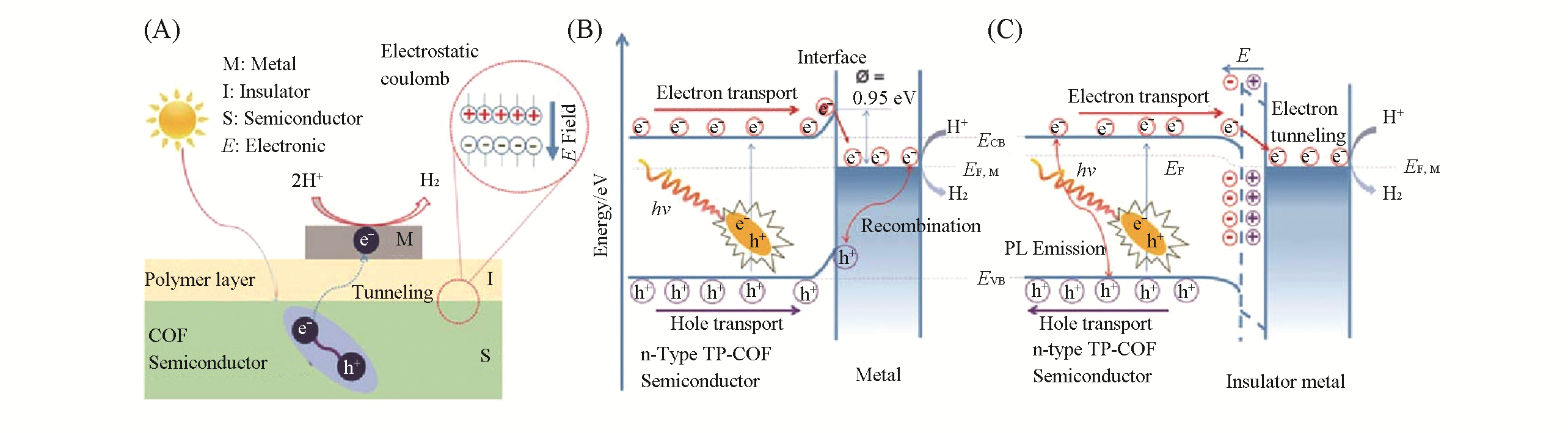
Fig.5 Synthesis and hydrogen production mechanism of metal?insulator?semiconductor[44](A) Synthesis and structure of MIS system; (B) hydrogen production mechanism on Mott-Schottky catalysts; (C) hydrogen production mechanism on MIS photocatalysts.Copyright 2019, Wiley-VCH.
| COFs | co?Catalyst | Sacrificial donor | Solvent | Light irradiation | HER/ (μmol·h-1·g-1) | AQE | Ref. |
|---|---|---|---|---|---|---|---|
| TFPT?COF | Pt | 10%(volume raction) TEOA | Water | >420 nm | 1970 | 2.2%—3.9% (500 nm) | [ |
| N0?COF | Pt | 1%(volume raction) TEOA | PBS solution | >420 nm | 23 | 0.001% (450 nm) | [ |
| N1?COF | Pt | 1%(volume raction) TEOA | PBS solution | >420 nm | 90 | 0.077% (450 nm) | [ |
| N2?COF | Pt | 1%(volume raction) TEOA | PBS solution | >420 nm | 438 | 0.19%(450 nm) | [ |
| N3?COF | Pt | 1%(volume raction) TEOA | PBS solution | >420 nm | 1703 | 0.44%(450 nm) | [ |
| PTP?COF | Pt | 1%(volume raction) TEOA | PBS solution | AM 1.5 | 83.83 | ― | [ |
| A?TEBPY?COF | Pt | 10%(volume raction) TEOA | Water | AM 1.5 | 98 | ― | [ |
| A?TENPY?COF | Pt | 10%(volume raction) TEOA | Water | AM 1.5 | 22 | ― | [ |
| A?TEPPY? COF | Pt | 10%(volume raction) TEOA | Water | AM 1.5 | 6 | ― | [ |
| TP?EDDA?COF | Pt | 10%(volume raction) TEOA | Water | >395 nm | 30 | ― | [ |
| TP?BDDA?COF | Pt | 10%(volume raction) TEOA | Water | >395 nm | 324 | 1.8%(520 nm) | [ |
| S?COF | Pt | 0.1 mol/L Ascorbic acid | Water | >420 nm | 4440 | ― | [ |
| FS?COF | Pt | 0.1 mol/L Ascorbic acid | Water | >420 nm | 10100 | ― | [ |
| FS?COF?WS5F | Pt/WS5F | 0.1 mol/L Ascorbic acid | Water | >420 nm | 16300 | 2.2%(600 nm) | [ |
| TpPa?COF | Pt | 100 mg Sodium ascorbate | PBS solution | >420 nm | 1560 | ― | [ |
| TpPa?COF?(CH3)2 | Pt | 100 mg Sodium ascorbate | PBS solution | >420 nm | 8330 | ― | [ |
| TpPa?COF?NO2 | Pt | 100 mg Sodium ascorbate | PBS solution | >420 nm | 220 | ― | [ |
| sp2c?COFERDN | Pt | 10%(volume fraction) TEOA | Water | >420 nm | 2120 | 0.48%(495 nm) | [ |
| CN?COF | Pt | 10%(volume fraction) TEOA | Water | >420 nm | 10100 | 20.7%(425 nm) | [ |
| NH2?Uio?66/TpPa?1?COF | Pt | 100 mg Sodium ascorbate | PBS solution | >420 nm | 23410 | ― | [ |
| CdS?COF(90:10) | Pt | 10%(volume fraction) Lactic acid | Water | >420 nm | 3678 | 4.2%(420 nm) | [ |
| Pt?PVP?TP?COF | Pt?PVP | 0.054 mol/L Ascorbic acid | Water | >420 nm | 8420 | 0.4%(475 nm) | [ |
| N2?COF | Co | 1%(volume fraction) TEOA | Water/ACN (volume ratio1:4) | AM 1.5 | 782 | 0.16%(400 nm) | [ |
| TpDTz?COF | Ni | 10%(volume fraction) TEOA | Water | AM 1.5 | 941 | ― | [ |
Table 1 Performance of photocatalytic H2 production by different COFs*
| COFs | co?Catalyst | Sacrificial donor | Solvent | Light irradiation | HER/ (μmol·h-1·g-1) | AQE | Ref. |
|---|---|---|---|---|---|---|---|
| TFPT?COF | Pt | 10%(volume raction) TEOA | Water | >420 nm | 1970 | 2.2%—3.9% (500 nm) | [ |
| N0?COF | Pt | 1%(volume raction) TEOA | PBS solution | >420 nm | 23 | 0.001% (450 nm) | [ |
| N1?COF | Pt | 1%(volume raction) TEOA | PBS solution | >420 nm | 90 | 0.077% (450 nm) | [ |
| N2?COF | Pt | 1%(volume raction) TEOA | PBS solution | >420 nm | 438 | 0.19%(450 nm) | [ |
| N3?COF | Pt | 1%(volume raction) TEOA | PBS solution | >420 nm | 1703 | 0.44%(450 nm) | [ |
| PTP?COF | Pt | 1%(volume raction) TEOA | PBS solution | AM 1.5 | 83.83 | ― | [ |
| A?TEBPY?COF | Pt | 10%(volume raction) TEOA | Water | AM 1.5 | 98 | ― | [ |
| A?TENPY?COF | Pt | 10%(volume raction) TEOA | Water | AM 1.5 | 22 | ― | [ |
| A?TEPPY? COF | Pt | 10%(volume raction) TEOA | Water | AM 1.5 | 6 | ― | [ |
| TP?EDDA?COF | Pt | 10%(volume raction) TEOA | Water | >395 nm | 30 | ― | [ |
| TP?BDDA?COF | Pt | 10%(volume raction) TEOA | Water | >395 nm | 324 | 1.8%(520 nm) | [ |
| S?COF | Pt | 0.1 mol/L Ascorbic acid | Water | >420 nm | 4440 | ― | [ |
| FS?COF | Pt | 0.1 mol/L Ascorbic acid | Water | >420 nm | 10100 | ― | [ |
| FS?COF?WS5F | Pt/WS5F | 0.1 mol/L Ascorbic acid | Water | >420 nm | 16300 | 2.2%(600 nm) | [ |
| TpPa?COF | Pt | 100 mg Sodium ascorbate | PBS solution | >420 nm | 1560 | ― | [ |
| TpPa?COF?(CH3)2 | Pt | 100 mg Sodium ascorbate | PBS solution | >420 nm | 8330 | ― | [ |
| TpPa?COF?NO2 | Pt | 100 mg Sodium ascorbate | PBS solution | >420 nm | 220 | ― | [ |
| sp2c?COFERDN | Pt | 10%(volume fraction) TEOA | Water | >420 nm | 2120 | 0.48%(495 nm) | [ |
| CN?COF | Pt | 10%(volume fraction) TEOA | Water | >420 nm | 10100 | 20.7%(425 nm) | [ |
| NH2?Uio?66/TpPa?1?COF | Pt | 100 mg Sodium ascorbate | PBS solution | >420 nm | 23410 | ― | [ |
| CdS?COF(90:10) | Pt | 10%(volume fraction) Lactic acid | Water | >420 nm | 3678 | 4.2%(420 nm) | [ |
| Pt?PVP?TP?COF | Pt?PVP | 0.054 mol/L Ascorbic acid | Water | >420 nm | 8420 | 0.4%(475 nm) | [ |
| N2?COF | Co | 1%(volume fraction) TEOA | Water/ACN (volume ratio1:4) | AM 1.5 | 782 | 0.16%(400 nm) | [ |
| TpDTz?COF | Ni | 10%(volume fraction) TEOA | Water | AM 1.5 | 941 | ― | [ |
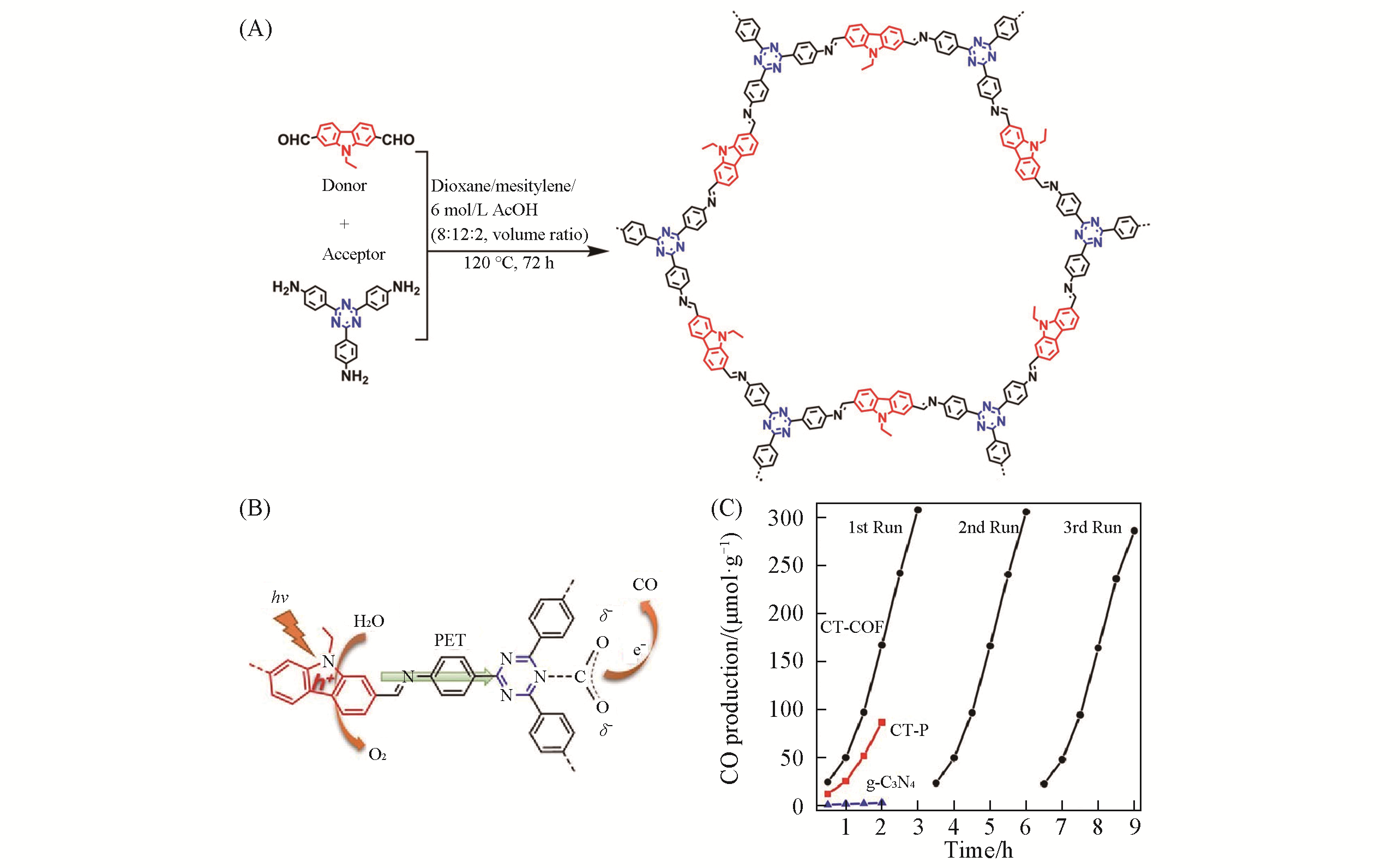
Fig.6 Synthesis and property of CT?COF[57](A) Synthesis of CT-COF; (B) photocatalytic site of CO2 reduction reaction; (C) time courses of photocatalytic activity for CO production.Copyright 2020, Wiley-VCH.
| COFs | Co?catalyst | Condition | Light irradiation/nm | Product (selectivity) | Activity/ (μmol·h-1·g-1) | TON/TOF* | Ref. |
|---|---|---|---|---|---|---|---|
| Re?CTF?py | Re | Solid?gas system | 200—1100 | CO | 353.05 | 4.8/— | [ |
| Re?COF | Re | TEOA/MeCN(volume ratio 0.2:3) | >420 | CO(98%) | 750 | 48/— | [ |
| Ni?TpBpy COF | Ni/ Ru(bpy)3Cl2 | TEOA/MeCN/H2O (volume ratio 1:3:1) | >420 | CO(96%) | 811.4 | 13.62/— | [ |
| DQTP?COF?Co | Co/Ru(bpy)3Cl2 | TEOA/MeCN(volume ratio 1:4) | >420 | CO | 1020 | —/0.55 h-1 | [ |
| DQTP?COF?Zn | Zn/Ru(bpy)3Cl2 | TEOA/MeCN(volume ratio 1:4) | >420 | HCOOH(90%) | 152.5 | —/0.08 h-1 | [ |
| TTCOF?Zn | — | H2O | 420—800 | CO(100%) | 0.2055 | —/— | [ |
| COF?367?Co NSs | Ru(bpy)3Cl2 | Ascorbic acid/0.1 mol/L KHCO3 | >420 | CO(78%) | 10162 | —/— | [ |
| ACOF?1 | — | H2O | 420—800 | CH3OH | 0.36 | —/— | [ |
| N3?COF | — | H2O | 420—800 | CH3OH | 0.57 | —/— | [ |
| CT?COF | — | H2O | >420 | CO(98%) | 102.7 | —/— | [ |
| COF?318/TiO2 | — | Solid?gas system | 380—800 | CO | 69.67 | —/— | [ |
Table 2 Performance of photocatalytic CO2 reduction by different COFs
| COFs | Co?catalyst | Condition | Light irradiation/nm | Product (selectivity) | Activity/ (μmol·h-1·g-1) | TON/TOF* | Ref. |
|---|---|---|---|---|---|---|---|
| Re?CTF?py | Re | Solid?gas system | 200—1100 | CO | 353.05 | 4.8/— | [ |
| Re?COF | Re | TEOA/MeCN(volume ratio 0.2:3) | >420 | CO(98%) | 750 | 48/— | [ |
| Ni?TpBpy COF | Ni/ Ru(bpy)3Cl2 | TEOA/MeCN/H2O (volume ratio 1:3:1) | >420 | CO(96%) | 811.4 | 13.62/— | [ |
| DQTP?COF?Co | Co/Ru(bpy)3Cl2 | TEOA/MeCN(volume ratio 1:4) | >420 | CO | 1020 | —/0.55 h-1 | [ |
| DQTP?COF?Zn | Zn/Ru(bpy)3Cl2 | TEOA/MeCN(volume ratio 1:4) | >420 | HCOOH(90%) | 152.5 | —/0.08 h-1 | [ |
| TTCOF?Zn | — | H2O | 420—800 | CO(100%) | 0.2055 | —/— | [ |
| COF?367?Co NSs | Ru(bpy)3Cl2 | Ascorbic acid/0.1 mol/L KHCO3 | >420 | CO(78%) | 10162 | —/— | [ |
| ACOF?1 | — | H2O | 420—800 | CH3OH | 0.36 | —/— | [ |
| N3?COF | — | H2O | 420—800 | CH3OH | 0.57 | —/— | [ |
| CT?COF | — | H2O | >420 | CO(98%) | 102.7 | —/— | [ |
| COF?318/TiO2 | — | Solid?gas system | 380—800 | CO | 69.67 | —/— | [ |
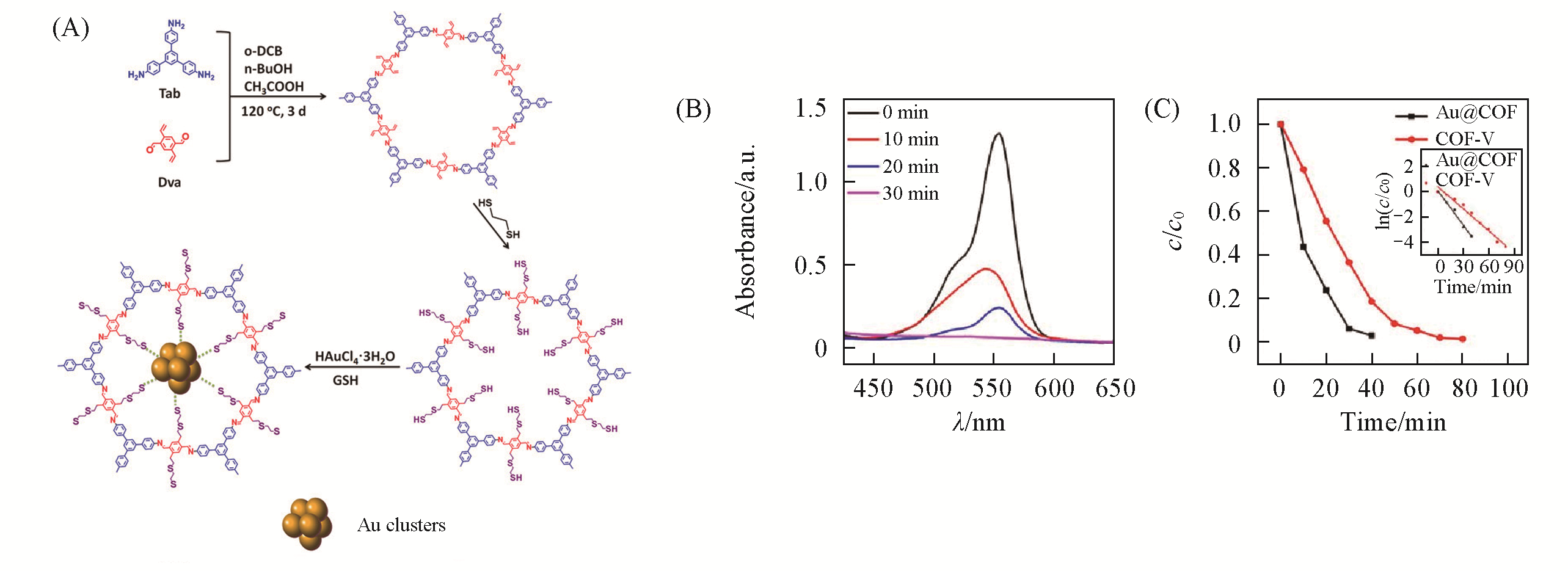
Fig.7 Synthesis and property of Au?S?COF[79](A) Synthesis of Au-S-COF; (B) UV-Vis spectra of RhB in different time under different irradiation time after Au@COF adsorption saturation; (C) degradation efficiency of RhB over Au@COF and COF-V.Copyright 2020, Wiley-VCH.
| 1 | Diercks C. S., Yaghi O. M., Science, 2017, 355, 923—931 |
| 2 | Feng X., Ding X. S., Jiang D. L., Chem. Soc. Rev., 2012, 41, 6010—6022 [3] Ding S. Y., Wang W., Chem. Soc. Rev., 2013, 42, 548—568 |
| 4 | Diercks C. S., Kalmutzki M. J., Yaghi O. M., Molecules, 2017, 22, 1575—1580 |
| 5 | Côté A. P., Benin A. I., Ockwig N. W., O’Keeffe M., Matzger A. J., Yaghi O. M., Science, 2005, 310, 1166—1170 |
| 6 | Bisbey R. P., Dichtel W. R., ACS Cent. Sci., 2017, 3, 533—543 |
| 7 | Waller P. J., Gándara F., Yaghi O. M., Acc. Chem. Res., 2015, 48, 3053—3063 |
| 8 | Huang N., Wang P., Jiang D. L., Nat. Rev. Mater., 2016, 1, 1—19 |
| 9 | Yaghi O. M., J. Am. Chem. Soc., 2016, 138, 15507—15509 |
| 10 | Nagai A., Guo Z. Q., Feng X., Jin S. B., Chen X., Ding X. S., Jiang D. L., Nat. Commun., 2011, 2, 536 |
| 11 | O’Keeffe M., Peskov M. A., Ramsden S. J., Yaghi O. M., Acc. Chem. Res., 2008, 41, 1782—1789 |
| 12 | Côté A. P., El⁃Kaderi H. M., Furukawa H., Hunt J. R., Yaghi O. M., J. Am. Chem. Soc., 2007, 129, 12914—12915 |
| 13 | Segura J. L., Mancheno M. J., Zamora F., Chem. Soc. Rev., 2016, 45, 5635—5671 |
| 14 | Jin E., Asada M., Xu Q., Dalapati S., Addicoat M. A., Brady M. A., Xu H., Nakamura T., Heine T., Chen Q. H., Jiang D. L., Science, 2017, 357, 673—676 |
| 15 | El⁃Kaderi H. M., Hunt J. R., Mendoza-Cortés J. L., Côté A. P., Taylor R. E., O’Keeffe M., Yaghi O. M., Science, 2007, 316, 268—272 |
| 16 | Fang Q. R., Wang J. H., Gu S., Kaspar R. B., Zhuang Z. B., Zheng J., Guo H. X, Qiu S. L., Yan Y. S., J. Am. Chem. Soc., 2015, 137, 8352—8355 |
| 17 | Zhang Y. Y., Duan J. Y., Ma D., Li P. F., Li S. W., Li H. W., Zhou J. W., Ma X. J., Feng X., Wang B., Angew. Chem. Int. Ed., 2017, 56, 16313—16317 |
| 18 | Lin G. Q., Ding H. M., Chen R. F., Peng Z. K., Wang B. S., Wang C., J. Am. Chem. Soc., 2017, 139, 8705—8709 |
| 19 | Yahiaoui O., Fitch A. N., Hoffmann F., Fröba M., Thomas A., Roeser J., J. Am. Chem. Soc., 2018, 140, 5330—5333 |
| 20 | Furukawa H., Yaghi O. M., J. Am. Chem. Soc., 2009, 131, 8875—8883 |
| 21 | Cao D. P., Lan J. H., Wang W. C., Smit B., Angew. Chem. Int. Ed., 2009, 48, 4730—4733 |
| 22 | Kang Z. X., Peng Y. W., Qian Y. H., Yuan D. Q., Addicoat M. A., Heine T., Hu Z. G., Tee L., Guo Z. G., Zhao D., Chem. Mater., 2016, 28, 1277—1285 |
| 23 | Rogge S. M. J., Bavykina A., Hajek J., Garcia H., Olivos-Suarez A. I., Sepulveda-Escribano A., Vimont A., Clet G., Bazin P., Kapteijn F., Daturi M., Ramos-Fernandez E. V., Xamena F. X. L. I., Speybroeck V. V., Gascon J., Chem. Soc. Rev., 2017, 46, 3134—3184 |
| 24 | Ding S. Y., Gao J., Wang Q., Zhang Y., Song W. G., Su C. Y., Wang W., J. Am. Chem. Soc., 2011, 133, 19816—19822 |
| 25 | Zhang J., Han X., Wu X. W., Liu Y., Cui Y., J. Am. Chem. Soc., 2017, 139, 8277—8285 |
| 26 | Peng Y. W., Huang Y., Zhu Y. H., Chen B., Wang L. Y., Lai Z. C., Zhang Z. C., Zhao M., Tan C. L., Yang N. L., Shao F. W., Han Y., Zhang H., J. Am. Chem. Soc., 2017, 139, 8698—8704 |
| 27 | Wang S., Wang Q. L., Shao P. P., Han Y. Z., Gao X., Ma L., Yuan S., Ma X. J., Zhou J. W., Feng X., Wang B., J. Am. Chem. Soc., 2017, 139, 4258—4261 |
| 28 | Wan S., Guo J., Kim J., Ihee H., Jiang D. L., Angew. Chem. Int. Ed., 2008, 47, 8826—8830 |
| 29 | Haldar S., Chakraborty D., Roy B., Banappanavar G., Rinku K., Mullangi D., Hazra P., Kabra D., Vaidhyanathan R., J. Am. Chem. Soc., 2018, 140, 13367—13374 |
| 30 | Bessinger D., Ascherl L., Auras F., Bein T., J. Am. Chem. Soc., 2017, 139, 12035—12042 |
| 31 | Banerjee T., Gottschling K., Savasci G., Ochsenfeld C., Lotsch B. V., ACS Energy Lett., 2018, 3, 400—409 |
| 32 | He T., Geng K., Jiang D. L., ACS Mater. Lett., 2019, 1, 203—208 |
| 33 | Stegbauer L., Schwinghammer K., Lotsch B. V., Chem. Sci., 2014, 5, 2789—2793 |
| 34 | Vyas V. S., Haase F., Stegbauer L., Savasci G., Podjaski F., Ochsenfeld C., Lotsch B. V., Nat. Commun., 2015, 6, 8508 |
| 35 | Haase F., Banerjee T., Savasci G., Ochsenfeld C., Lotsch B. V., Faraday Discuss., 2017, 201, 247—264 |
| 36 | Stegbauer L., Zech S., Savasci G., Banerjee T., Podjaski F., Schwinghammer K., Ochsenfeld C., Lotsch B. V., Adv. Energy Mater., 2018, 8, 1703278 |
| 37 | Pachfule P., Acharjya A., Roeser J., Langenhahn T., Schwarze M., Schomacker R., Thomas A., Schmidt J., J. Am. Chem. Soc., 2018, 140, 1423—1427 |
| 38 | Wang X. Y., Chen L. J., Chong S. Y., Little M. A., Wu Y. Z., Zhu W. H., Clowes R., Yan Y., Zwijnenburg M. A., Sprick R. S., Cooper A. I., Nat. Chem., 2018, 10, 1180—1189 |
| 39 | Sheng J. L., Dong H., Meng X. B., Tang H. L., Yao Y. H., Liu D. Q., Bai L. L., Zhang F. M., Wei J. Z., Sun X. J., ChemCatChem, 2019, 11, 2313—2319 |
| 40 | Jin E. Q., Lan Z. A., Jiang Q. H., Geng K. Y., Li G. S., Wang X. C., Jiang D. L., Chem., 2019, 5, 1632—1647 |
| 41 | Luo M. L., Yang Q., Liu K. W., Cao H. M., Yan H. J., Chem. Commun., 2019, 55, 5829—5832 |
| 42 | Zhang F. M., Sheng J. L., Yang Z. D., Sun X. J., Tang H. L., Lu M., Dong H., Shen F. C., Liu J., Lan Y. Q., Angew. Chem. Int. Ed., 2018, 57, 12106—12110 |
| 43 | Thote J., Aiyappa H. B., Deshpande A., Diaz D. D., Kurungot S., Banerjee R., Chem. Eur. J., 2014, 20, 15961—15965 |
| 44 | Ming J. T., Liu A., Zhao J. W., Zhang P., Huang H. W., Lin H., Xu Z. T., Zhang X. M,, Wang X. X., Hofkens J., Roeffaers M. B. J., Long J. L., Angew. Chem. Int. Ed., 2019, 58, 18290—18294 |
| 45 | Banerjee T., Haase F., Savasci G., Gottschling K., Ochsenfeld C., Lotsch B. V., J. Am. Chem. Soc., 2017, 139, 16228—16234 |
| 46 | Biswal B. P., Vignolo-Gonzalez H. A., Banerjee T., Grunenberg L., Savasci G., Gottschling K., Nuss J., Ochsenfeld C., Lotsch B. V., J. Am. Chem. Soc., 2019, 141, 11082—11092 |
| 47 | Takeda H., Koike K., Inoue H., Ishitani O., J. Am. Chem. Soc., 2008, 130, 2023—2031 |
| 48 | Morimoto T., Nakajima T., Sawa S., Nakanishi R., Imori D., Ishitani O., J. Am. Chem. Soc., 2013, 135, 16825—16828 |
| 49 | Kou Y., Nabetani Y., Masui D., Shimada T., Takagi S., Tachibana H., Inoue H., J. Am. Chem. Soc., 2014, 136, 6021—6030 |
| 50 | Xu R., Wang X. S., Zhao H., Lin H., Huang Y. B., Cao R., Catal. Sci. Technol., 2018, 8, 2224—2230 |
| 51 | Yang S. Z., Hu W. H., Zhang X., He P. L., Pattengale B., Liu C. M., Cendejas M., Hermans I., Zhang X. Y., Zhang J., Huang J. E., J. Am. Chem. Soc., 2018, 140, 14614—14618 |
| 52 | Zhong W. F., Sa R. J., Li L. Y., He Y. J., Li L. Y., Bi J. H., Zhuang Z. Y., Yu Y., Zou Z. G., J. Am. Chem. Soc., 2019, 141, 7615—7621 |
| 53 | Lu M., Li Q., Liu J., Zhang F. M., Zhang L., Wang J. L., Kang Z. H., Lan Y. Q., Appl. Catal. B: Environ., 2019, 254, 624—633 |
| 54 | Lu M., Liu J., Li Q., Zhang M., Liu M., Wang J. L., Yuan D. Q., Lan Y. Q., Angew. Chem. Int. Ed., 2019, 58, 12392—12397 |
| 55 | Liu W. B., Li X. K., Wang C. M., Pan H. H., Liu W. P., Wang K., Zeng Q. D., Wang R. M., Jiang J. Z., J. Am. Chem. Soc., 2019, 141, 17431—17440 |
| 56 | Fu Y. H., Zhu X. L., Huang L., Zhang X. C., Zhang F. M., Zhu W. D., Appl. Catal. B: Environ., 2018, 239, 46—51 |
| 57 | Lei K., Wang D., Ye L. Q., Kou M. P., Deng Y., Ma Z. Y., Wang L., Kong Y., ChemSusChem, 2020, 13, 1725—1729 |
| 58 | Zhang M., Lu M., Lang Z. L., Liu J., Liu M., Chang J. N., Li L. Y., Shang L. J., Wang M., Li S. L., Lan Y. Q., Angew. Chem. Int. Ed., 2020, 59, 6500—6506 |
| 59 | Nicewicz D. A., Nguyen T. M., ACS Cat., 2013, 4, 355—360 |
| 60 | Shaw M. H., Twilton J., MacMillan D. W. C., J. Org. Chem., 2016, 81, 6898—6926 |
| 61 | Yoon T. P., Ischay M. A., Du J., Nat. Chem., 2010, 2, 527—532 |
| 62 | Qu Y. Q., Duan X. F., Chem. Soc. Rev., 2013, 42, 2568—2580 |
| 63 | Lang X. J., Chen X. D., Zhao J. C., Chem. Soc. Rev., 2014, 43, 473—486 |
| 64 | Zhi Y. F., Li Z. P., Feng X., Xia H., Zhang Y. M., Shi Z., Mu Y., Liu X. M., J. Mater. Chem. A, 2017, 5, 22933—22938 |
| 65 | Li Z. P., Zhi Y. F., Shao P. P., Xia H., Li G. S., Feng X., Chen X., Shi Z., Liu X. M., Appl. Catal. B: Environ., 2019, 245, 334—342 |
| 66 | Liu W. T., Su Q., Ju P. Y., Guo B. X., Zhou H., Li G. H., Wu Q. L., ChemSusChem, 2017, 10, 664—669 |
| 67 | Bhadra M., Kandambeth S., Sahoo M. K., Addicoat M., Balaraman E., Banerjee R., J. Am. Chem. Soc., 2019, 141, 6152—6156 |
| 68 | Chen R. F., Shi J. L., Ma Y., Lin G. Q., Lang X. J., Wang C., Angew. Chem. Int. Ed., 2019, 58, 6430—6434 |
| 69 | Li S., Li L., Li Y. J., Dai L., Liu C. X., Liu Y. Z., Li J. N., Lv J. N., Li P. F., Wang B., ACS Catal., 2020, 10, 8717—8726 |
| 70 | Yan X. L., Liu H., Li Y. S., Chen W. B., Zhang T., Zhao Z. Q., Xing G. L., Chen L., Macromolecules, 2019, 52, 7977—7983 |
| 71 | Wei P. F., Qi M. Z., Wang Z. P., Ding S. Y., Yu W., Liu Q., Wang L. K., Wang H. Z., An W. K., Wang W., J. Am. Chem. Soc., 2018, 140, 4623—4631 |
| 72 | Jiménez‐Almarza A., López‐Magano A., Marzo L., Cabrera S., Mas‐Ballesté R., Alemán J., ChemCatChem, 2019, 11, 4916—4922 |
| 73 | Nagai A., Chen X., Feng X., Ding X. S., Guo Z. Q., Jiang D. L., Angew. Chem. Int. Ed., 2013, 52, 3770—3774 |
| 74 | Chen X., Addicoat M. A., Jin E. Q., Zhai L. P., Xu H., Huang N., Guo Z. Q., Liu L. L., Irle S., Jiang D. L., J. Am. Chem. Soc., 2015, 137, 3241—3247 |
| 75 | Liu T. T., Hu X. Y., Wang Y. F., Meng L. Y., Zhou Y. A., Zhang J. X., Chen M., Zhang X. M., J. Photochem. Photobiol. B, 2017, 175, 156—162 |
| 76 | He S. J., Rong Q. F., Niu H. Y., Cai Y. Q., Chem. Commun., 2017, 53, 9636—9639 |
| 77 | Zhou P., Yu J. G., Jaroniec M., Adv. Mater., 2014, 26, 4920—4935 |
| 78 | He S. J., Rong Q. F., Niu H. Y., Cai Y. Q., Appl. Catal. B, 2019, 247, 49—56 |
| 79 | Deng Y., Zhang Z., Du P. Y., Ning X. M., Wang Y., Zhang D. X., Liu J., Zhang S. T., Lu X. Q., Angew. Chem. Int. Ed., 2020, 59, 6082—6089 |
| 80 | Chen W. B., Yang Z. F., Xie Z., Li Y. S., Yu X., Lu F. L., Chen L., J. Mater. Chem. A, 2019, 7, 998—1004 |
| [1] | 秦永吉, 罗俊. 单原子催化剂在CO2转化中的应用[J]. 高等学校化学学报, 2022, 43(9): 20220300. |
| [2] | 林治, 彭志明, 贺韦清, 沈少华. 单原子与团簇光催化: 竞争与协同[J]. 高等学校化学学报, 2022, 43(9): 20220312. |
| [3] | 吴玉, 李轩, 杨恒攀, 何传新. 钴单原子的双重限域制备策略及高效CO2电还原性能[J]. 高等学校化学学报, 2022, 43(9): 20220343. |
| [4] | 滕镇远, 张启涛, 苏陈良. 聚合物单原子光催化剂的载流子分离和表面反应机制[J]. 高等学校化学学报, 2022, 43(9): 20220325. |
| [5] | 王新天, 李攀, 曹越, 洪文浩, 耿忠璇, 安志洋, 王昊宇, 王桦, 孙斌, 朱文磊, 周旸. 单原子材料在二氧化碳催化中的技术经济分析与产业化应用前景[J]. 高等学校化学学报, 2022, 43(9): 20220347. |
| [6] | 邱丽琪, 姚向阳, 何良年. 可见光驱动丰产金属卟啉类配合物催化的二氧化碳选择性还原反应[J]. 高等学校化学学报, 2022, 43(7): 20220064. |
| [7] | 夏雾, 任颖异, 刘京, 王锋. 壳聚糖包裹CdSe量子点组装体的水相可见光催化CO2还原[J]. 高等学校化学学报, 2022, 43(7): 20220192. |
| [8] | 赵润瑶, 纪桂鹏, 刘志敏. 吡咯氮配位单原子铜催化剂的电催化二氧化碳还原性能[J]. 高等学校化学学报, 2022, 43(7): 20220272. |
| [9] | 赵盈喆, 张建玲. 金属-有机框架基材料在二氧化碳光催化转化中的应用[J]. 高等学校化学学报, 2022, 43(7): 20220223. |
| [10] | 王丽君, 李欣, 洪崧, 詹新雨, 王迪, 郝磊端, 孙振宇. 调节氧化镉-炭黑界面高效电催化CO2还原生成CO[J]. 高等学校化学学报, 2022, 43(7): 20220317. |
| [11] | 王广琦, 毕艺洋, 王嘉博, 石洪飞, 刘群, 张钰. 非贵金属三元复合Ni(PO3)2-Ni2P/CdS NPs异质结的构建及可见光高效催化产氢性能[J]. 高等学校化学学报, 2022, 43(6): 20220050. |
| [12] | 宋颖颖, 黄琳, 李庆森, 陈立妙. CuO/BiVO4光催化剂的制备及光催化CO2还原性能[J]. 高等学校化学学报, 2022, 43(6): 20220126. |
| [13] | 龚妍熹, 王建兵, 柴歩瑜, 韩元春, 马云飞, 贾超敏. 钾掺杂g-C3N4薄膜光阳极的制备及光电催化氧化降解水中双氯芬酸钠性能[J]. 高等学校化学学报, 2022, 43(6): 20220005. |
| [14] | 金湘元, 张礼兵, 孙晓甫, 韩布兴. 单原子催化剂在电催化还原CO2领域的应用[J]. 高等学校化学学报, 2022, 43(5): 20220035. |
| [15] | 陶雨, 欧鸿辉, 雷永鹏, 熊禹. 单原子催化剂在光催化二氧化碳还原中的研究进展[J]. 高等学校化学学报, 2022, 43(5): 20220143. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
