

高等学校化学学报 ›› 2026, Vol. 47 ›› Issue (5): 20250408.doi: 10.7503/cjcu20250408
王圣杰, 朱子豪, 朱羽杰, 吴春晓, 阿力木·阿卜杜热合曼(![]() )
)
收稿日期:2025-12-29
出版日期:2026-05-10
发布日期:2026-02-04
通讯作者:
阿力木·阿卜杜热合曼
E-mail:alim@jlu.edu.cn
基金资助:
WANG Shengjie, ZHU Zihao, ZHU Yujie, WU Chunxiao, ALIM Abdurahman(![]() )
)
Received:2025-12-29
Online:2026-05-10
Published:2026-02-04
Contact:
ALIM Abdurahman
E-mail:alim@jlu.edu.cn
Supported by:摘要:
稳定的开壳有机发光自由基由于其独特的电子结构及自旋允许的发光特性, 在光电器件、 量子信息和自旋相关功能材料领域展现出重要的研究价值. 然而, 该类体系在凝聚态中普遍面临聚集诱导猝灭(ACQ)问题, 严重限制了其实际应用. 与低浓度物理掺杂等策略相比, 近年来以分子层面化学结构调控为核心的研究逐渐成为突破方向. 例如, 通过构筑自由基聚合物实现自旋中心的有效分散或借助精细分子设计调控立体位阻和分子堆积方式实现稳定高效的凝聚态发光. 本文系统总结了相关体系的光物理行为、 调控机制及关键材料设计原则, 并分析了当前面临的挑战与潜在应用, 以便为发光自由基材料由基础研究迈向实际应用提供参考.
中图分类号:
TrendMD:
王圣杰, 朱子豪, 朱羽杰, 吴春晓, 阿力木·阿卜杜热合曼. 凝聚态发光稳定有机自由基的研究进展与挑战. 高等学校化学学报, 2026, 47(5): 20250408.
WANG Shengjie, ZHU Zihao, ZHU Yujie, WU Chunxiao, ALIM Abdurahman. Advances and Challenges of Stable Organic Radicals with Luminescence in the Condensed State. Chem. J. Chinese Universities, 2026, 47(5): 20250408.
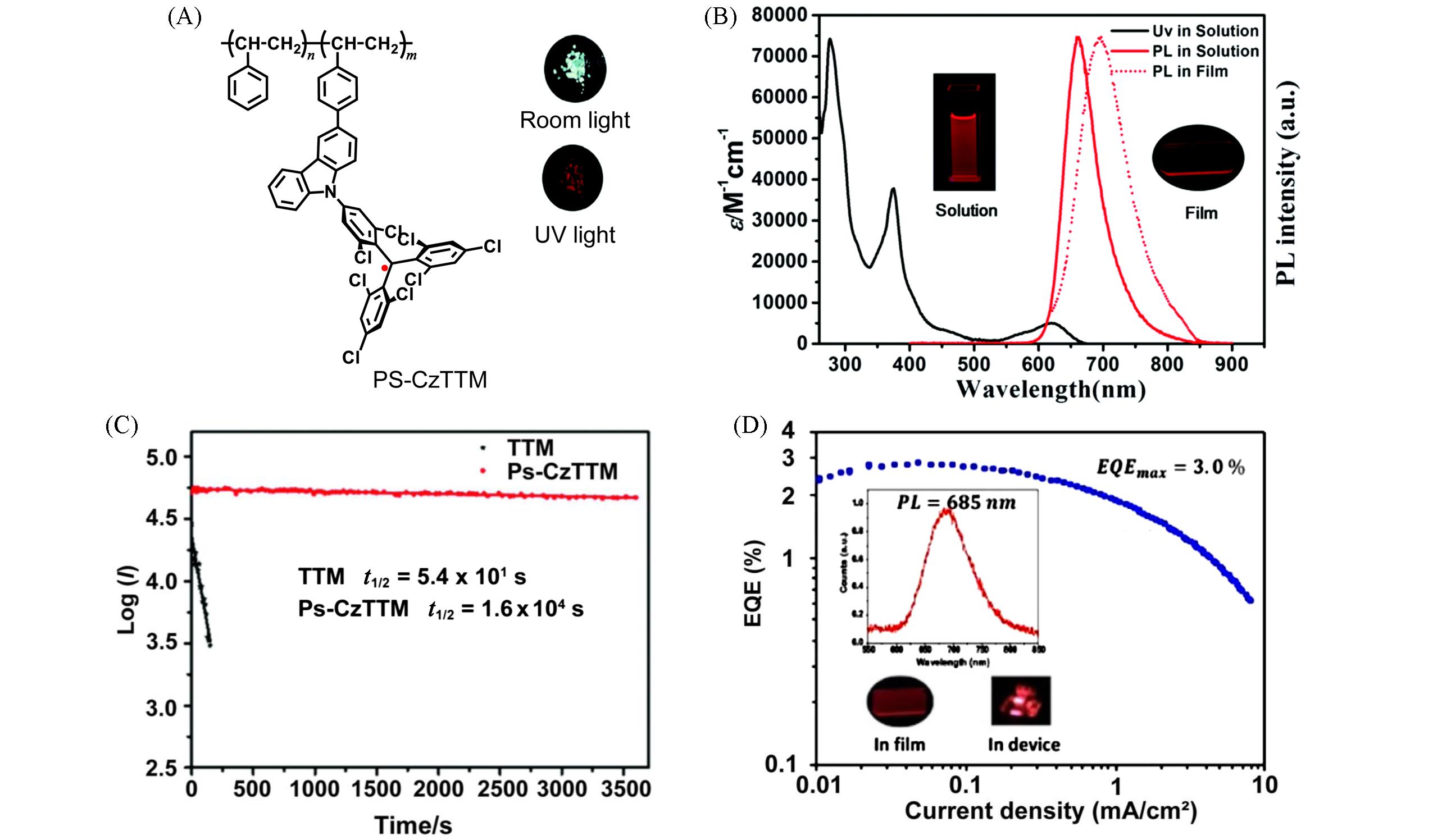
Fig.1 Photophysical and device properties of the first luminescent radical polymer PS⁃CzTTM(A) Chemical structure of PS-CzTTM and the photographs of PS-CzTTM powder under room light(up) and under UV light at 365 nm(down); (B) UV-Vis-NIR absorption spectrum(black line) and emission spectrum(red line) of PS-CzTTM in cyclohexane(1 μmol/L) and emission spectrum(red dashed line) of the PS-CzTTM in spin-coated film(100 nm, the insets); (C) comparison of the fluorescence intensity decays of TTM and PS-CzTTM in cyclohexane under irradiation with a 355 nm pulse[42]; (D) EQE of host-guest OLEDs versus current density[43]. Insets: (B) the photographs of PS-CzTTM in solution(left) and in the film state(right) under UV light(365 nm) at room temperature; (D) EL spectra of host-guest OLED at 12 V and the photographs of PS-CzTTM in the film state(left) and in the device(right).(A—C) Copyright 2019, the Royal Society of Chemistry; (D) Copyright 2020, American Chemical Society.
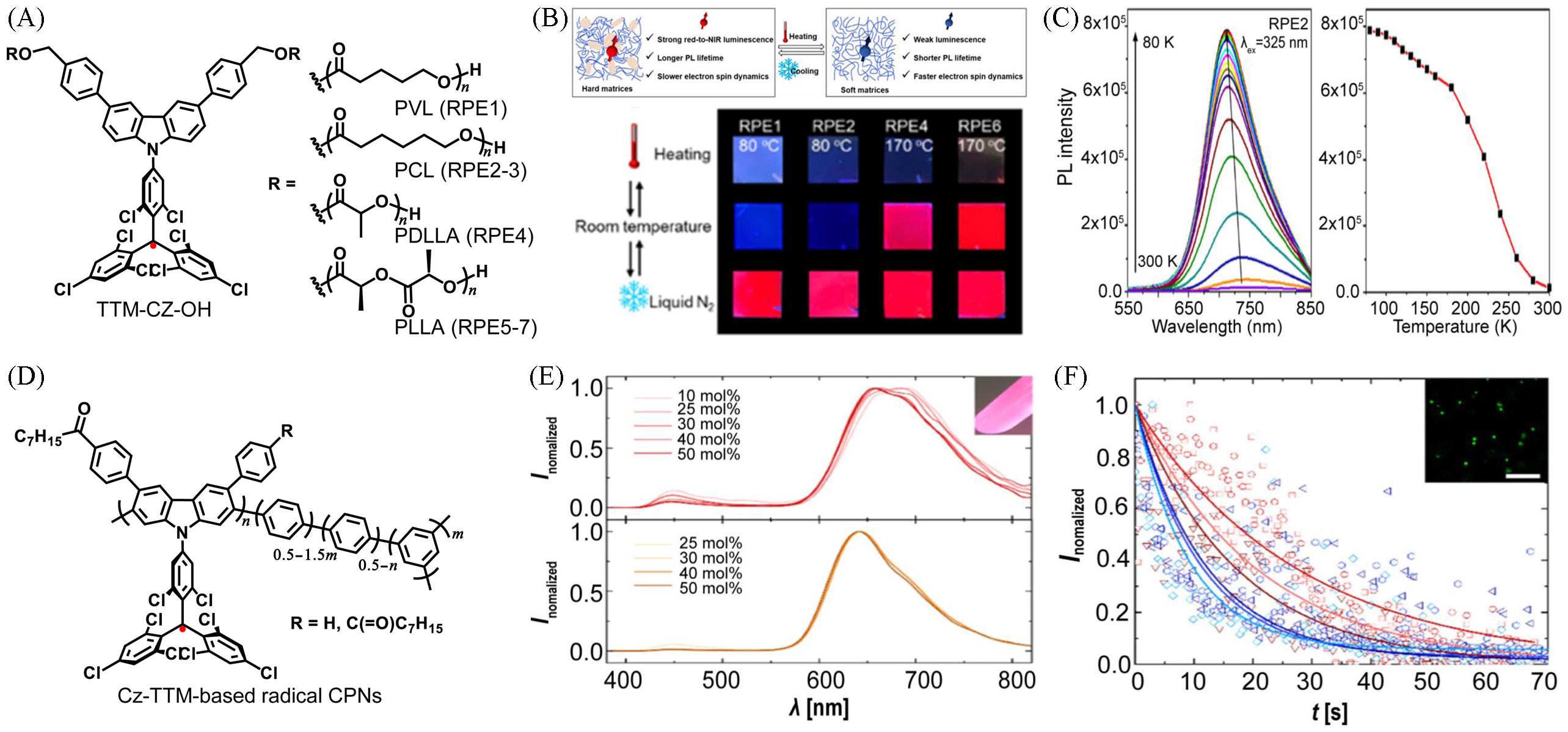
Fig.3 Luminescence properties of diverse luminescent radical⁃based polymeric materials in different conditions(A) Chemical structure of radical-centered polymers derived from TTM-CZ-OH; (B) schematic presentation to illustrate the impact of polymer rigidity on the luminescence and electron spin dynamics of the embedded radicals(up) and digital photographs of RPE1, RPE2, RPE4 and RPE6 thin films on quartz slides under UV light(365 nm, down); (C) temperature-dependent steady-state PL emission(excitation at 375 nm) spectra of the RPE2 film and the evolution of the maximal PL intensity of the RPE2 film as a function of temperature[46]; (D) chemical structure of Cz-TTM-based radical CPN; (E) PL spectra of CPN dispersions in 1-PrOH synthesized with different initial radical monomer contents of oct-CzBr2-TTM and bisoct-CzBr2-TTM in molar fraction; (F) PL intensity of radical CPNs made from 50%(molar fraction) oct-CzBr2-TTM(red) and latex reference particles loaded with commercial red dye(blue) as a function of time. Both particle dispersions are illuminated with a 5.1 mW CW-laser at 561 nm, and three distinctive particles of both batches are recorded over time; the straight lines show an exponential fit of the data, the inset shows a confocal image of the stable aqueous dispersion of the luminescent open-shell CPNs[47]. Inset of (E): photograph of a radical CPN dispersion under UV light synthesized with the same parameters as used for the kinetic study.(A—C) Copyright 2022, American Chemical Society; (D—F) Copyright 2023, the Authors. Published by American Chemical Society;
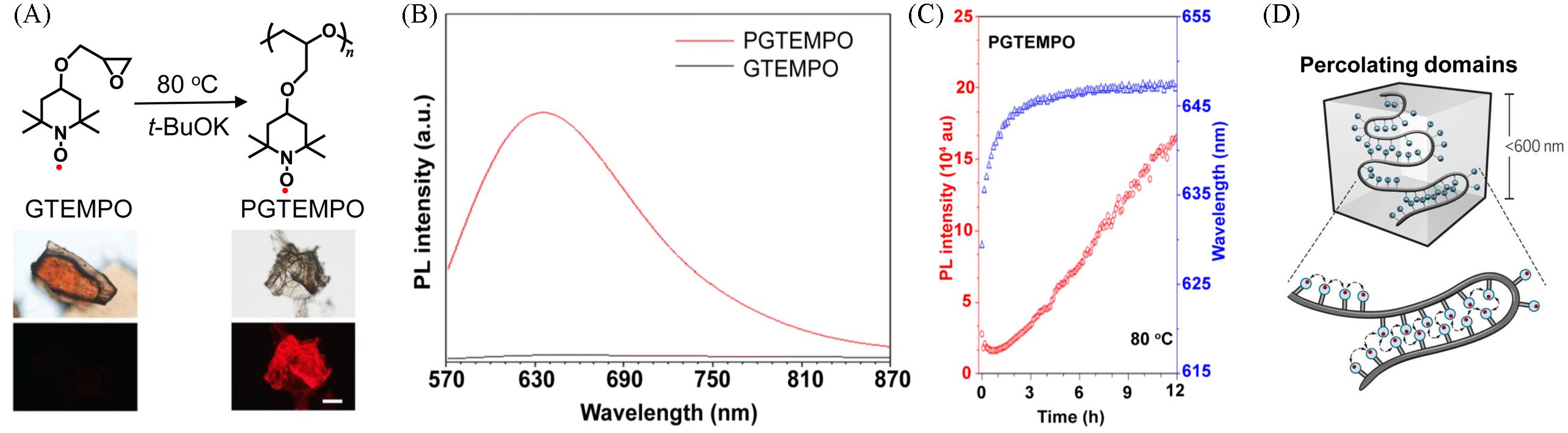
Fig.4 Synthesis and photophysical properties of radical polymer PGTEMPO(A) Synthetic route of the PGTEMPO radical polymer and its photos taken under ambient light(up) and 510—550 nm excitation(down); (B) PL spectra of PGTEMPO and GTEMPO in the solid state(excitation at 532 nm); (C) the real-time annealing of PGTEMPO at the temperature of 80 ℃ under nitrogen(excitation at 532 nm)[49]; (D) schematic of percolating domains after annealing[54].(A, B, C) Copyright 2022, the Royal Society of Chemistry; (D) Copyright 2018, the Authors, some rights reserved; exclusive licensee American Association for the Advancement of Science.
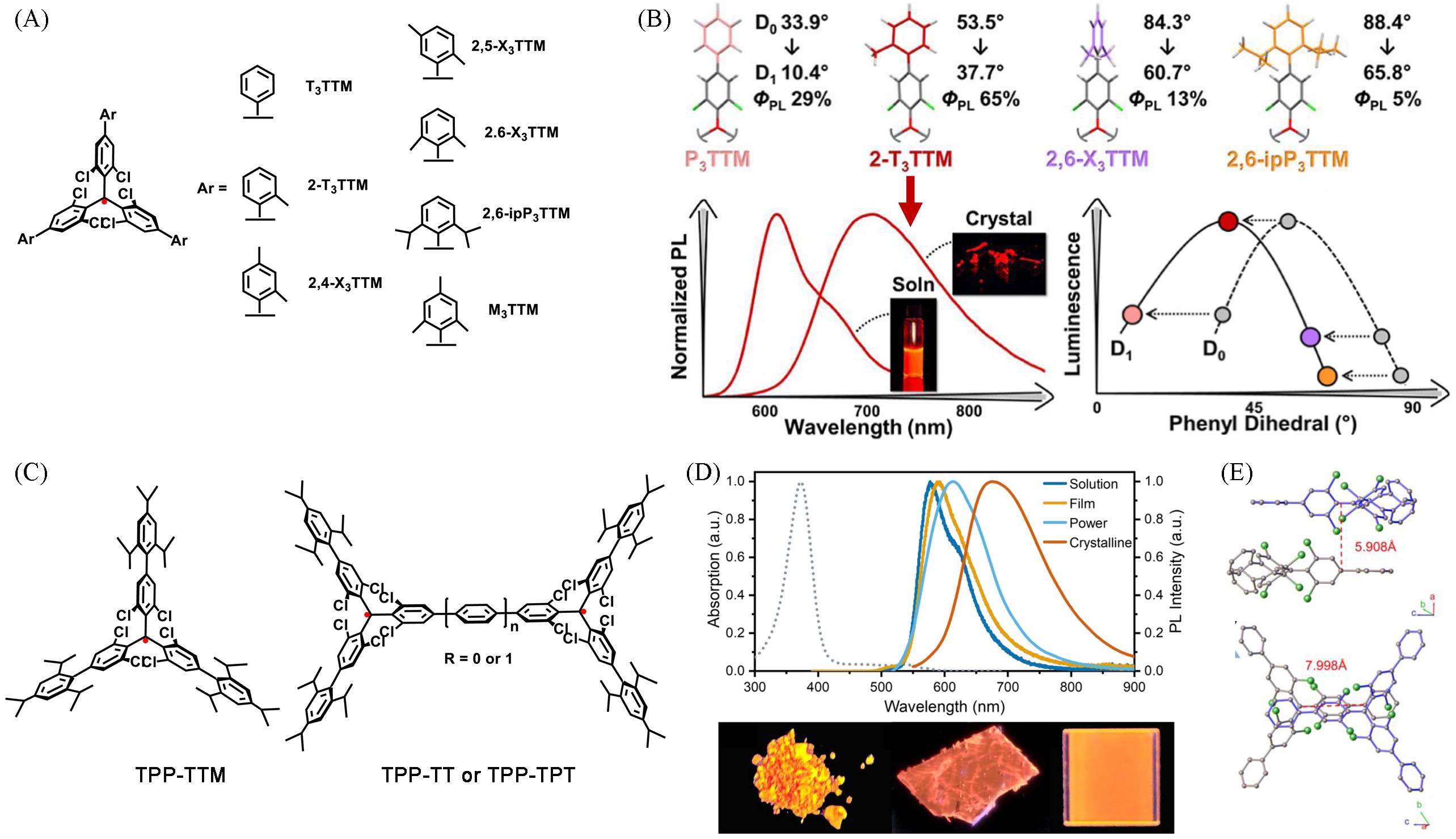
Fig.5 Luminescence of an isolated radical molecule in the condensed state(A) Chemical structures of TTM-based radicals; (B) molecular structures from X-ray crystal structures of the synthesized P3TTM, 2-T3TTM, and 2,6-X3TTM and 2,6-ipP3TTM radicals with average phenyl-phenyl dihedrals and emission spectrum of 2-T3TTM in solution and in crystal[58]; (C) chemical structure of TPP-TTM, TPP-TT and TPP-TPT; (D) normalized UV-Vis absorption and PL spectra of TPP-TTM in cyclohexane solution(1×10-5 mol/L) and aggregated states at room temperature(up); photographs of powders, a single crystal, and a spin-coated neat film of TPP-TTM under UV light(365 nm, down); (E) front(up) and top(down) view of possible intermolecular dimer of TPP-TTM in crystals[36]. Insets of (B): the photographs of 2-T3TTM in solution(left) and in the crystal(right).(A, B) Copyright 2024, the Authors, published by American Chemical Society; (C—E) Copyright 2025, Wiley-VCH.
| [1] | Mizuno A., Matsuoka R., Mibu T., Kusamoto T., Chem. Rev., 2024, 124(3), 1034—1121 |
| [2] | Abdurahman A., Peng Q. M., Chin. J. Luminesc., 2024, 45(2), 211—214 |
| [3] | Straub D., Gross M., Arnold M. E., Zolg J., Kuehne A. J. C., Beilstein J. Org. Chem., 2025, 21, 964—998 |
| [4] | Peng Q. M., Obolda A., Zhang M., Li F., Angew. Chem. Int. Ed., 2015, 54(24), 7091—7095 |
| [5] | Ai X., Evans E. W., Dong S. Z., Gillett A. J., Guo H. Q., Chen Y. X., Hele T. J. H., Friend R. H., Li F., Nature, 2018, 563, 536—540 |
| [6] | Abdurahman A., Hele T. J. H., Gu Q. Y., Zhang J. B., Peng Q. M., Zhang M., Friend R. H., Li F., Evans E. W., Nat. Mater., 2020, 19, 1224—1229 |
| [7] | Ai X., Chen Y., Feng Y., Li F., Angew. Chem. Int. Ed., 2018, 57(11), 2869—2873 |
| [8] | Qu Y. Y., Li Y. C., Tan X. L., Zhai W. X., Han G. F., Hou J. L., Liu G. Q., Song Y. G., Liu Y. P., Chem. Eur. J., 2019, 25(33), 7888—7895 |
| [9] | Zhao Y. H., Abdurahman A., Zhang Y. M., Zheng P., Zhang M., Li F., CCS Chem., 2022, 4(2), 722—731 |
| [10] | Zhou Z. B., Yang K., He L., Wang W., Lai W. M., Yang Y. H., Dong Y. G., Xie S., Yuan L., Zeng Z. B, J. Am. Chem. Soc., 2024, 146(10), 6763—6772 |
| [11] | Feng L., Tuo Y. Y., Wu Z. P., Zhang W. J., Li C. B., Yang B., Liu L. X., Gong J. Y., Jiang G. Y., Hu W., Tang B. Z., Wu L. M., Wang J. G., J. Am. Chem. Soc., 2024, 146(47), 32582—32594 |
| [12] | Chen D. J., Xu Y. X., Wang Y. T., Li X., Yin D. L., Yan L. F., ACS Appl. Mater. Interfaces, 2024, 16(44), 59907—59920 |
| [13] | Liu T., Zhu Z. H., Wang S. J., Shen L., Abdurahman A., Liu X. M., Lu G. Y., Light Sci. Appl., 2025, 14, 289 |
| [14] | Kimura S., Matsuoka R., Kimura S., Nishuhara H., Kusamoto T., J. Am. Chem. Soc., 2021, 143(15), 5610—5615 |
| [15] | Gorgon S., Ly K., Grüne J., Drummond B. H., Myers A. W. K., Londi G., Ricci G., Valverde D., Tonnellé C., Murto P., Romanov A. S., Casanova D., Dyakonov V., Sperlich A., Belionne D., Olivier Y., Li F., Friend R. H., Evans E. W., Nature, 2023, 620, 538—544 |
| [16] | Kopp S. M., Nakamura S., Phelan B. T., Poh Y. R., Tyndall S. B., Brown P. J. J., Huang Y. H., Yuen⁃Zhou J., Krzyaniak M. D., Wasielewski M. R., J. Am. Chem. Soc., 2024, 146(40), 27935—27945 |
| [17] | Poh Y. R., Morozov D., Kazmierczak N. P., Hadt R. G., Groenhof G., Yuen⁃Zhou J., J. Am. Chem. Soc., 2024, 146(22), 15549—15561 |
| [18] | Kopp S. M., Nakamura S., Poh Y. R., Peinkhofer K. W. R., Phelan B. T., Zhou J. Y., Krzyaniak M. D., Wasielewski M. R., J. Am. Chem. Soc., 2025, 147(26), 22951—22960 |
| [19] | Chowdhury R., Murto P., Panjwani N. A., Sun Y., Ghosh P., Boeije Y., Cordeiro C. D., Derkach V., Woo S., Millington O., Congrave D. G., Fu Y., Mustafa T. B. E., Monteverde A., Cerdá J., Londi G., Behrends J., Rao A., Belionne D., Chepelianskii M., Bronstein H., Friend R. H., Nat. Chem., 2025, 17, 1410—1417 |
| [20] | Schäffer D., Wischnat J., Tesi L., de Sousa J. A., Little E., McGuire J., Mas⁃Torrent M., Rovira C., Veciana J., Tuna F., Chilvers N., Van Slageren J., Adv. Mater., 2023, 35(38), 2302114 |
| [21] | Chen S. Y., Zhu Z. H., Zhou L. P., Huang H. X., Abdurahman A., Qiao X. F., Ma D. G., J. Phys. Chem. Lett., 2025, 16(36), 9401—9407 |
| [22] | Tang B. Z., Chem. J. Chinese Universities, 2019, 40(7), 1793 |
| [23] | Li L., Prindle C. R., Shi W. Z., Buckolls C., Venkataraman L., J. Am. Chem. Soc., 2023, 145(33), 18182—18204 |
| [24] | Hackney H. E., Ruchlin C., Stahle E., Perepichka D. F., Angew. Chem. Int. Ed., 2025, 64(37), e202512411 |
| [25] | Li S. Z., Zhao X. L., Shi X. L., Yang H. B., J. Am. Chem. Soc., 2025, 147(38), 34498—34507 |
| [26] | Wu C. X., Ai X., Chen Y. X., Cui Z. Y., Li F., Chem. J. Chinese Universities, 2020, 41(5), 972—980 |
| [27] | Yang Y. M., Qiu L. L., Shi X. L., Chem. Res. Chinese Universities, 2023, 39(2), 197—201 |
| [28] | Abdurahman A., Wang J. M., Zhao Y. H., Li P., Shen L., Peng Q. M., Angew. Chem. Int. Ed., 2023, 62(15), e202300772 |
| [29] | Zhu Y. J., Zhu Z. H., Wang S. X., Peng Q. M., Abdurahman A., Angew. Chem. Int. Ed., 2025, 64(10), e202423470 |
| [30] | Liu C. H., He Z. C., Ruchlin C., Che Y. X., Somers K., Perepichka D. F., J. Am. Chem. Soc., 2023, 145(29), 15702—15707 |
| [31] | Matsuoka R., Kimura S., Miura T., Ikoma T., Kusamoto T., J. Am. Chem. Soc., 2023, 145(25), 13615—13622 |
| [32] | Abdurahman A., Shen L., Wang J. M., Niu M. L, Peng Q. M., Wang J. P., Lu G. Y., Light Sci. Appl., 2023, 12, 272 |
| [33] | Mizuno A., Matsuoka R., Kimura S., Ochiai K., Kusamoto T., J. Am. Chem. Soc., 2024, 146(27), 18470—18483 |
| [34] | Wang X., Wang S. J., Ding Z. Z., Shen L., Zhu Z. H., Abdurahman A., Lu G. Y., Peng Q. M., Angew. Chem. Int. Ed., 2025, 64(40), e202515393 |
| [35] | Tong Z. K., Zhang S., Niu W. W., Yu T. X., Zhang X. F., Yao P. L., Wang J. F., Han Y. B., Li G. W., Dong S. Q., J. Am. Chem. Soc., 2025, 147(43), 39232—39246 |
| [36] | Guan J. H., Zhu Z. H., Gou Q. Q., Wang J. M., Kuang Z. Y., Zhang L. T., Zhang X. W., Ai X., Abdurahman A., Peng Q. M., Aggregate, 2025, 6(9), e70100 |
| [37] | Kimura S., Kusamoto T., Kimura S., Kato K., Teki Y., Nishihara H., Angew. Chem. Int. Ed., 2018, 57(39), 12711—12715 |
| [38] | Liu C. H., Hamzehpoor E., Otsuka Y. S., Jadhav T., Perepichka D. F., Angew. Chem. Int. Ed., 2020, 59(51), 23030—23034 |
| [39] | Zhu Z. H., Kuang Z. Y., Shen L., Wang S. J., Ai X., Abdurahman A., Peng Q. M., Angew. Chem. Int. Ed., 2024, 63(42), e202410552 |
| [40] | Wang S. J., Wang X, Zhu Z. H., Shen L, Zhu Y. J., Abdurahman A., Ma H. W., Peng Q. M., Lu G. Y., CCS Chem., 2025, e202506229 |
| [41] | Zhang Z. T., Zhang J. Y., Sun J. Z., Zhang H. K., Zhang X. H., Tang B. Z., Chem. Eur. J., 2025, 31(4), e202403493 |
| [42] | Abdurahman A., Peng Q. M., Ablikim O., Ai X., Li F., Mater. Horiz., 2019, 6, 1265—1270 |
| [43] | Gu Q. Y., Abdurahman A., Friend R. H., Li F., J. Phys. Chem. Lett., 2020, 11(14), 5638—5642 |
| [44] | Wang S. J., Wang X., Ding J. S., Zhu Z. H., Wang J. M., Shen L., Abdurahman A., Lu G. Y., Wang J. P., Peng Q. M., Macromolecules, 2024, 57(13), 6133—6139 |
| [45] | Wang S. J., Zhu Z. H., Abdurahman A., Peng Q. M., Macromolecules, 2025, 58(1), 372—378 |
| [46] | Hou L. M., Xu H. X., Zhang X. Y., Zhang Y. P., Chen R., Zhang Z. Y., Wang M. F., Macromolecules, 2022, 55(19), 8619—8628 |
| [47] | Chen L., Rudolf T., Blinder R., Suryadevara N., Dalmeida A., Welscher P. J., Lamla M., Arnold M., Herr U., Jelezko F., Ruben M., Kuehne A. J. C., Macromolecules, 2023, 56(5), 2104—2112 |
| [48] | Armet O., Veciana J., Rovira C., Riera J., Castaner J., Molins E., Rius J., Miravitlles C., Brichfeus S., J. Phys. Chem., 1987, 91(22), 5608—5616 |
| [49] | Wang Z. Y., Zou X. H., Xie Y., Zhang H. K., Hu L. R., Chan C. C. S., Zhang R. Y., Guo J., Kwok R. T. K., Lam J. W. Y., Williams I. D., Zeng Z. B., Wong K. S., Sherrill C. D., Ye R. Q., Tang B. Z., Mater. Horiz., 2022, 9, 2564—2571 |
| [50] | Chen P. Y., Zhang G. Y., Li J. G., Ma L. J., Zhou J. Y., Zhu M. G., Li S., Wang Z., Chem. Res. Chinese Universities, 2024, 40(2), 293—304 |
| [51] | Xiong J. Y., Wu M. J., Yao L. Y., Chem. Res. Chinese Universities, 2024, 40(5), 887—893 |
| [52] | Sun Z. H., Yin P. P., He S. Y., Zhang K. G., Pan X. R., Wang J. Y., Hao P. N., Zhou Z., Yang X. G., Ma L. F., Tan C. L., Chem. Res. Chinese Universities, 2025, 41(3), 519—524 |
| [53] | Joo Y., Agarkar V., Sung S. H., Savoie B. M., Boudouris B. W., Science, 2018, 359(6382), 1391—1395 |
| [54] | Lutkenhaus J., Science, 2018, 359(6382), 1334—1335 |
| [55] | Kimura S., Uejima M., Ota W., Sato T., Kusaka S., Matsuda R., Nishihara H., Kusamoto T., J. Am. Chem. Soc., 2021, 143(11), 4329—4338 |
| [56] | Kimura S., Tanushi A., Kusamoto T., Kochi S., Sato T., Nishihara H., Chem. Sci., 2018, 9, 1996—2007 |
| [57] | Matsuoka R., Kimura S., Kusamoto T., ChemPhotoChem, 2021, 5(7), 669—673 |
| [58] | Murto P., Li B., Fu Y., Walker L. E., Brown L., Bond A. D., Zeng W., Chowdhury R., Cho H. H., Yu C. P., Grey C. P., Friend R. H., Bronstein H., J. Am. Chem. Soc., 2024, 146(19) 13133—13141 |
| [59] | Murto P., Chowdhury R., Gorgon S., Guo E., Zeng W. X., Li B. W., Sun Y. Q., Francis H., Friend R. H., Bronstein H., Nat. Commun., 2023, 14, 4147 |
| [60] | Yu C. P., Chowdhury R., Fu Y., Ghosh P., Zeng W. X., Mustafa T. B. E., Grüne J., Walker L. E., Congrave D. G., Chua X. W., Murto P., Rao A., Sirringhaus H., Plasser F., Grey C. P., Friend R. H., Bronstein H., Sci. Adv., 2024, 10(30), eado3476 |
| [1] | 朱高桦, 舒菊, 耿江涛, 马夫龙, 熊玲红, 何学文. 原位激活型近红外二区聚集诱导发光探针应用于肿瘤中过氧化氢的高灵敏成像[J]. 高等学校化学学报, 2026, 47(5): 20260058. |
| [2] | 潘晶珂, 齐馨, 张露露, 王贝贝, 吕超. 主客体作用力构建的聚集诱导发光型超分子聚合物的合成与应用研究[J]. 高等学校化学学报, 2026, 47(5): 20260130. |
| [3] | 马德平 陈家兴 史志伟 张卫平 张雅珍 安虹静 武超 来华 成中军 刘宇艳. 丙烯酸酯-环氧二阶段树脂可充气展开管[J]. 高等学校化学学报, 0, (): 20250396. |
| [4] | 庞逸盈 韩建华 宗立率 王锦艳 蹇锡高. 生物基乙酰丁香酮衍生聚芳醚酮的分子设计、合成与表征[J]. 高等学校化学学报, 0, (): 20260030. |
| [5] | 苗丹丹 范学峰 罗智莱 刘灿 包春燕. 动态酰腙键水凝胶的构建与粘弹性调控[J]. 高等学校化学学报, 0, (): 20260033. |
| [6] | 刘苗苗, 傅梦然, 高蝶, 张万鹏, 梁莹, 何媛媛, 赵巧巧, 赵廷兴, 李鸿波, 丁自成, 韩艳春. 基于给/受体双重相似性“桥联”聚合物受体的侧链工程构筑高性能三元有机太阳能电池[J]. 高等学校化学学报, 2026, 47(4): 20250324. |
| [7] | 任奥成, 李青云, 吉晓帆. 荧光超分子聚合物网络[J]. 高等学校化学学报, 2026, 47(4): 20250390. |
| [8] | 孙妍, 朱东霞. 近红外激发有机发光材料的构筑及在疾病治疗中的应用[J]. 高等学校化学学报, 2026, 47(4): 20260002. |
| [9] | 汪德州 高愉淇 韩春雨 杨爽 宋文植. 负载新型多肽CFAP1的水凝胶敷料在伤口修复中的应用[J]. 高等学校化学学报, 0, (): 20260015. |
| [10] | 张雨, 吴洋, 李佳琪, 徐超, 汪少振, 李欣雨, 王存民, 张生辉, 徐欢. MOF功能化自清洁聚乳酸多级孔纤维膜的辐射冷却性能[J]. 高等学校化学学报, 2026, 47(3): 20250239. |
| [11] | 张禹, 陈龙, 陈奥, 盛全康, 王传贵, 胡思泉, 陈韶云, 胡成龙. 花状聚苯胺微纳结构的制备与电化学性能[J]. 高等学校化学学报, 2026, 47(3): 20250241. |
| [12] | 徐千惠, 徐振, 马子奥, 乔娟, 李楠, 马磊, 靳海波, 张荣月. 接枝聚N,N-二甲胺基丙基丙烯酰胺制备大孔聚合物阴离子交换层析介质及其吸附性能[J]. 高等学校化学学报, 2026, 47(3): 20250238. |
| [13] | 刘莉莉, 杨佩, 田益嘉, 黄逸瑜, 张正东, 闫伟, 张原原, 史林兴. 高密度磷酸化纳米洋葱碳/磺化聚芳醚砜复合膜的制备与性能[J]. 高等学校化学学报, 2026, 47(2): 20250256. |
| [14] | 户书达, 董丽宁, 王存民, 李欣雨, 汪少振, 李世航, 张生辉, 徐欢. 融合PM滤除和CO2吸附功能的聚乳酸活性纳纤膜[J]. 高等学校化学学报, 2026, 47(2): 20250243. |
| [15] | 赵旭, 林子瑜, 赵哲, 任强, 丁思文, 赵建盛, 申璐, 庞金辉, 姜振华. 导电聚醚醚酮/聚苯胺薄膜的制备[J]. 高等学校化学学报, 2026, 47(1): 20250326. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||