

高等学校化学学报 ›› 2014, Vol. 35 ›› Issue (6): 1189.doi: 10.7503/cjcu20131048
收稿日期:2013-10-28
出版日期:2014-06-10
发布日期:2014-01-13
作者简介:联系人简介: 方 浩, 男, 博士, 教授, 博士生导师, 主要从事药物化学中抗肿瘤药物研究. E-mail:基金资助:
WANG Junhua, WANG Quande, DUN Yanyan, FANG Hao*( )
)
Received:2013-10-28
Online:2014-06-10
Published:2014-01-13
Contact:
FANG Hao
E-mail:haofangcn@sdu.edu.cn
Supported by:摘要:
合成了一系列结构全新的嘌呤磺胺类衍生物, 并应用溴化噻唑蓝四氮唑(MTT)法进行了初步体外抗肿瘤细胞增殖活性研究. 结果表明, 嘌呤环C2位、 N6位和N9位的取代对活性均有较大影响, C2位引入苯磺酰基哌嗪片段后有利于提高抗肿瘤活性. 化合物17d对3株肿瘤细胞PC-3, HCT116和K562 的增殖均有明显抑制作用, 且强度与阳性对照药Roscovitine相近.
中图分类号:
TrendMD:
王军华, 王泉德, 顿艳艳, 方浩. 嘌呤磺胺衍生物的合成及抗肿瘤活性. 高等学校化学学报, 2014, 35(6): 1189.
WANG Junhua, WANG Quande, DUN Yanyan, FANG Hao. Syntheses and Antitumor Activities of Purine-sulfonamides Derivatives†. Chem. J. Chinese Universities, 2014, 35(6): 1189.
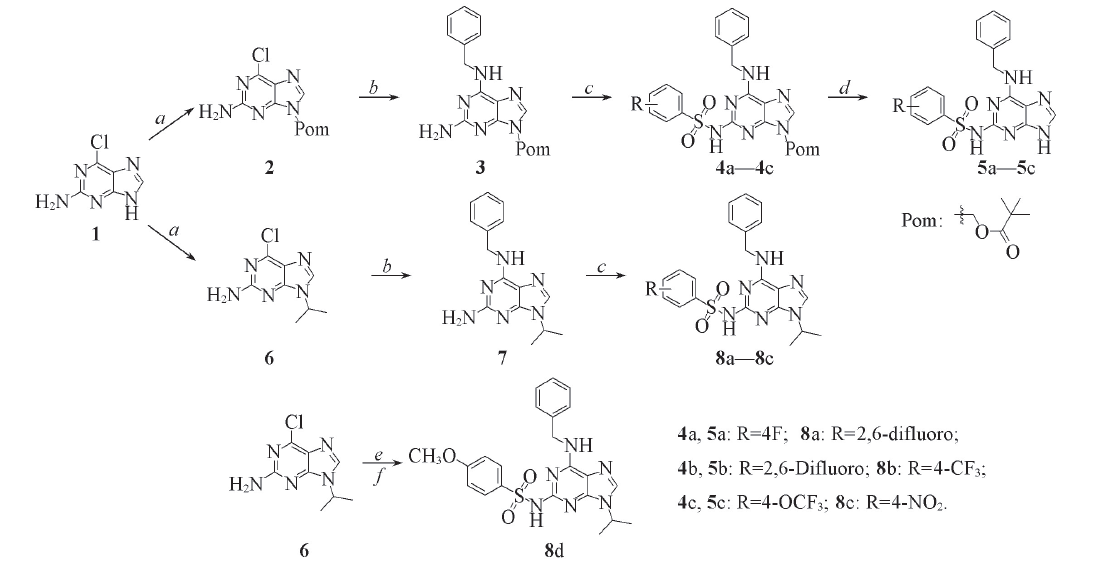
Scheme 1 Synthesis routes of target compounds 5a—5c and 8a—8da. Chloromethyl pivalate(Pom-Cl), K2CO3, DMSO, r. t. or 2-bromopropane, K2CO3, DMSO, 80 ℃; b. benzyl amine, TEA, n-butanol, reflux; c. arylsulfonyl chloride, pyridine, r. t.; d. 3 mol/L NaOH, THF, r. t.; e. 4-methoxybenzene sulfonyl chloride, pyridine, r. t.; f. benzyl amine, TEA, n-butanol, reflux.
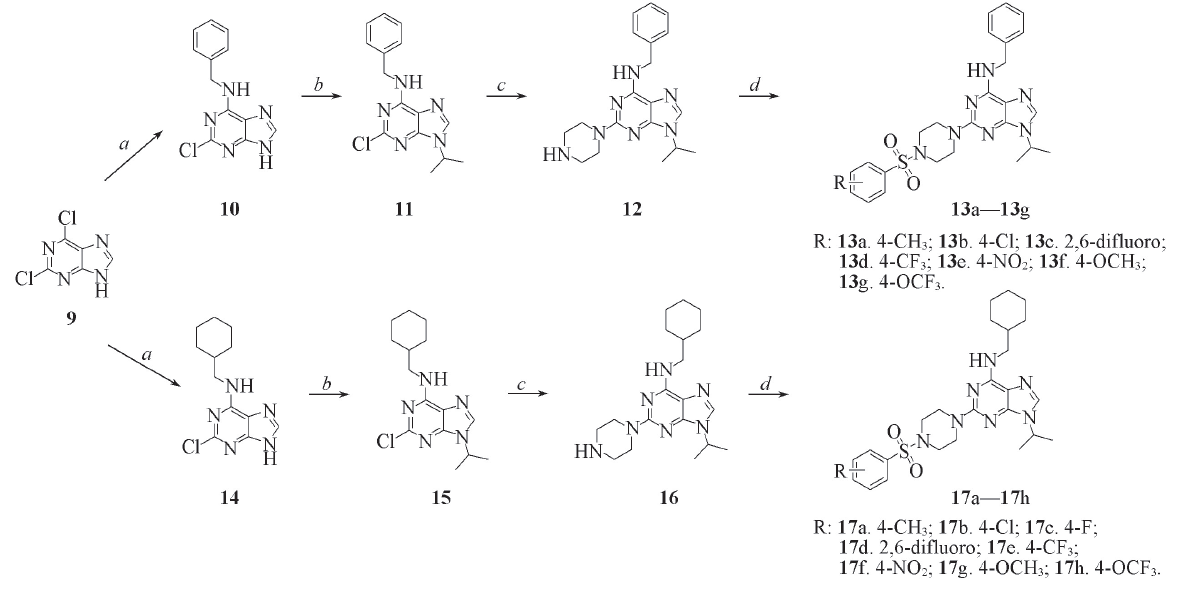
Scheme 2 Synthesis routes of target compounds 13a—13g and 17a—17ha. Benzyl amine or aminomethylcyclohexane, TEA, n-butanol, reflux; b. 2-bromopropane, K2CO3, DMSO, 80 ℃; c. piperazine, n-butanol, N2, 110 ℃; d. arylsulfonyl chloride, pyridine, r. t..
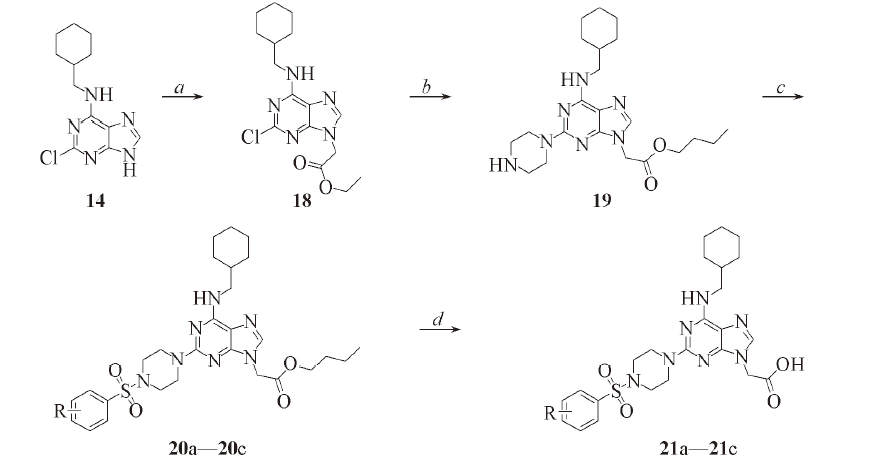
Scheme 3 Synthesis routes of target compounds 21a—21ca. Ethyl chloroacetate, K2CO3, DMSO, 90 ℃; b. piperazine, n-butanol, N2, 110 ℃; c. arylsulfonyl chloride, pyridine, r. t.; d. 2 mol/L NaOH, THF, r. t.. R: a. 4-F; b. 2,6-difluoro; c. 4-OCH3.
| Compd. | m.p./℃ | Yield(%) | Appearance | HRMS(calcd.)[M+H]+ |
|---|---|---|---|---|
| 5a | 254(Dec.) | 77 | White solid | 399.1034(399.1034) |
| 5b | 258(Dec.) | 65 | White solid | 417.0940(417.0940) |
| 5c | 286(Dec.) | 82 | White solid | 465.0955(465.0951) |
| 8a | 241—244 | 32 | Light pink solid | 459.1406(459.1409) |
| 8b | 215—218 | 24 | White solid | 491.1468(491.1472) |
| 8c | 245—246 | 60 | Light yellow solid | 468.1444(468.1449) |
| 8d | 206—208 | 5.5 | White solid | 453.1703(453.1703) |
| 13a | 86—90 | 39 | Light yellow solid | 506.2329(506.2333) |
| 13b | 80—83 | 42 | Light yellow solid | 526.1784(526.1786) |
| 13c | 108—111 | 59 | White solid | 528.1984(528.1988) |
| 13d | 86—88 | 65 | Gray solid | 560.2050(560.2050) |
| 13e | 226—228 | 28 | Brick red solid | 537.2023(537.2027) |
| 13f | 81—84 | 42 | Light yellow solid | 522.2279(522.2282) |
| 13g | 76—78 | 38 | Light yellow solid | 576.2000(576.1999) |
| 17a | 146—150 | 11 | White solid | 512.2803(512.2802) |
| 17b | 157—160 | 19 | White solid | 532.2254(532.2256) |
| 17c | 119—121 | 16 | White solid | 516.2548(516.2552) |
| 17d | 186—190 | 42 | White solid | 534.2454(534.2457) |
| 17e | 192—194 | 67 | White solid | 566.2520(566.2520) |
| 17f | 198—200 | 48 | Yellow solid | 543.2494(543.2496) |
| 17g | 148—150 | 15 | White solid | 528.2748(528.2751) |
| 17h | 167—168 | 59 | White solid | 582.2468(582.2469) |
| 21a | 256(Dec.) | 78 | White solid | 532.2130(532.2137) |
| 21b | 275(Dec.) | 47 | White solid | 550.2044(550.2043) |
| 21c | 228(Dec.) | 91 | White solid | 544.2326(544.2337) |
Table 1 Physical data and HRMS results of all target compounds
| Compd. | m.p./℃ | Yield(%) | Appearance | HRMS(calcd.)[M+H]+ |
|---|---|---|---|---|
| 5a | 254(Dec.) | 77 | White solid | 399.1034(399.1034) |
| 5b | 258(Dec.) | 65 | White solid | 417.0940(417.0940) |
| 5c | 286(Dec.) | 82 | White solid | 465.0955(465.0951) |
| 8a | 241—244 | 32 | Light pink solid | 459.1406(459.1409) |
| 8b | 215—218 | 24 | White solid | 491.1468(491.1472) |
| 8c | 245—246 | 60 | Light yellow solid | 468.1444(468.1449) |
| 8d | 206—208 | 5.5 | White solid | 453.1703(453.1703) |
| 13a | 86—90 | 39 | Light yellow solid | 506.2329(506.2333) |
| 13b | 80—83 | 42 | Light yellow solid | 526.1784(526.1786) |
| 13c | 108—111 | 59 | White solid | 528.1984(528.1988) |
| 13d | 86—88 | 65 | Gray solid | 560.2050(560.2050) |
| 13e | 226—228 | 28 | Brick red solid | 537.2023(537.2027) |
| 13f | 81—84 | 42 | Light yellow solid | 522.2279(522.2282) |
| 13g | 76—78 | 38 | Light yellow solid | 576.2000(576.1999) |
| 17a | 146—150 | 11 | White solid | 512.2803(512.2802) |
| 17b | 157—160 | 19 | White solid | 532.2254(532.2256) |
| 17c | 119—121 | 16 | White solid | 516.2548(516.2552) |
| 17d | 186—190 | 42 | White solid | 534.2454(534.2457) |
| 17e | 192—194 | 67 | White solid | 566.2520(566.2520) |
| 17f | 198—200 | 48 | Yellow solid | 543.2494(543.2496) |
| 17g | 148—150 | 15 | White solid | 528.2748(528.2751) |
| 17h | 167—168 | 59 | White solid | 582.2468(582.2469) |
| 21a | 256(Dec.) | 78 | White solid | 532.2130(532.2137) |
| 21b | 275(Dec.) | 47 | White solid | 550.2044(550.2043) |
| 21c | 228(Dec.) | 91 | White solid | 544.2326(544.2337) |
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
|---|---|---|---|
| 5aa,d | 4.53(br s, 2H, CH2), 7.15—7.18(m, 2H, ArH), 7.25—7.30(m, 5H, ArH), 7.90—7.95(m, 3H, ArH, purine C8), 8.25(br s, 1H, NH), 11.08(br s, 1H, SO2NH), 12.76(br s, 1H, purine C9) | 163.78(d,J=249 Hz), 153.33, 152.43, 150.55, 139.54, 138.64, 138.21, 129.80(d, J=5.25 Hz), 128.15, 127.34, 126.71, 115.60(d, J=22.5 Hz), 42.80 | |
| 5ba,d | 4.38(br s, 2H, CH2), 6.98—7.08(m, 2H, ArH), 7.20—7.27(m, 5H, ArH), 7.41—7.48(m, 1H, ArH), 7.99(s, 1H, purine C8), 8.32(br s, 1H, NH), 11.72(br s, 1H, SO2NH), 12.84(br s, 1H, purine C9) | 158.72(dd,J=4.5, 252.75 Hz), 153.14, 152.40, 149.49, 139.67, 138.64, 133.34, 128.25, 127.68, 126.98, 112,72(d, J=24 Hz), 42.76 | |
| 5ca,d | 4.49(br s, 2H, CH2), 7.25—7.31(m, 7H, ArH), 7.89—7.99(m, 3H, ArH, purine C8), 8.35(br s, 1H, NH) | 153.00, 152.84, 150.13, 141.68, 139.29, 129.19, 128.15, 127.32, 126.72, 120.54, 119.80(q, J=256.5 Hz), 111.54, 42.84 | |
| 8ab,d | 1.36[d, J=6.3 Hz, 6H, (CH3)2], 4.47—4.53(m, 3H, CH, CH2), 7.16(t, J=9.0 Hz, 2H, ArH), 7.20—7.29(m, 5H, ArH), 7.54—7.64(m, 1H, ArH), 8.21(s, 1H, purine C8), 8.46(br s, 1H, NH), 11.74(br s, 1H, SO2NH) | 158.87(d,J=255 Hz), 153.30, 152.63, 148.26, 139.12, 137.45, 134.91, 128.17, 127.52, 126.81, 118.94, 113.06(d, J=24 Hz), 113.05(t, J=11.25 Hz), 47.33, 42.73, 21.56 | |
| 8b b,d | 1.37[d, J=6.6 Hz, 6H, (CH3)2], 4.48—4.54(m, 3H, CH, CH2), 7.18—7.28(m, 5H, ArH), 7.81(d, J=7.2 Hz, 2H, ArH), 8.05(s, 1H, purine C8), 8.10(d, J=7.2 Hz, 2H, ArH), 8.36(br s, 1H, NH), 11.39(br s, 1H, SO2NH) | 154.12, 151.77, 148.66, 145.29, 139.66, 137.74, 131.94(q, J=31.5 Hz), 128.11, 128.01, 127.25, 126.64, 125.92, 123.43(q, J=271.5 Hz), 115.53, 46.58, 42.71, 21.73 | |
| 8cb,d | 1.41[d, J=6.3 Hz, 6H, (CH3)2], 4.50—4.52(m, 3H, CH, CH2), 7.16—7.22(m, 5H, ArH), 8.05(s, 1H, purine C8), 8.09(d, J=7.2 Hz, 2H, ArH), 8.18(d, J=7.2 Hz, 2H, ArH), 8.36(br s, 1H, NH), 11.49(br s, 1H, SO2NH) | 154.14, 151.62, 149.08, 147.06, 139.51, 137.71, 128.29, 127.95, 126.93, 126.48, 123.97, 115.78, 46.39, 42.59, 21.85 | |
| 8d c,d | 1.51[d, J=6.6 Hz, 6H, (CH3)2], 3.80(s, 3H, OCH3), 4.63(hept, J=6.6 Hz, 1H, CH), 4.72(br s, 2H, CH2), 6.52(br s, 1H, NH), 6.81(d, J=9.0 Hz, 2H, ArH), 7.26—7.32(m, 5H, ArH), 7.61(s, 1H, purine C8), 8.01(d, J=9.0 Hz, 2H, ArH), 8.24(br s, 1H, SO2NH) | 162.03, 154.29, 151.94, 149.12, 139.94, 137.41, 132.69, 129.44, 128.01, 127.37, 126.50, 115.84, 113.61, 59.64, 46.24, 42.58, 21.88 | |
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
| 13a a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.37(s, 3H, CH3), 2.75—2.78(m, 4H, piperazine), 3.74—3.82(m, 4H, piperazine), 4.50—4.52(m, 3H, CH, CH2), 7.22—7.32(m, 5H, ArH), 7.42(d, J=7.8 Hz, 2H, ArH), 7.58(d, J=7.8 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.11(br s, 1H, NH) | 158.80, 152.94, 150.20, 143.77, 139.13, 133.86, 132.47, 129.70, 128.31, 127.82, 127.61, 126.91, 109.51, 47.47, 45.85, 44.42, 43.72, 22.12, 21.04 | |
| 13b a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.81—2.88(m, 4H, piperazine), 3.75—3.80(m, 4H, piperazine), 4.50—4.55(m, 3H, CH, CH2), 7.21—7.32(m, 5H, ArH), 7.70(d, J=8.4 Hz, 2H, ArH), 7.72(d, J=8.4 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.09(br s, 1H, NH) | 158.74, 153.35, 150.50, 139.53, 139.08, 134.13, 133.89, 129.41, 129.15, 128.40, 127.60, 127.04, 111.01, 47.11, 45.79, 44.50, 43.77, 22.30 | |
| 13c a,d | 1.43[d, J=6.6 Hz, 6H, (CH3)2], 2.99—3.13(m, 4H, piperazine), 3.76—3.85(m, 4H, piperazine), 4.49—4.57(m, 3H, CH, CH2), 7.18—7.35(m, 7H, ArH), 7.75—7.77(m, 1H, ArH), 7.87(s, 1H, purine C8), 8.11(br s, 1H, NH) | 158.80(dd,J=3.75, 255 Hz), 157.34, 153.92, 150.21, 140.52, 136.21(t, J=11.25 Hz), 135.99, 127.90, 127.35, 126.37, 113.68(dd, J=3, 23.25 Hz), 113.35, 113.13, 45.67, 44.86, 43.40, 42.62, 21.97 | |
| 13d a,d | 1.43[d, J=6.6 Hz, 6H, (CH3)2], 2.79—3.03(m, 4H, piperazine), 3.71—3.89(m, 4H, piperazine), 4.45—4.61(m, 3H, CH, CH2), 7.19—7.32(m, 5H, ArH), 7.90(s, 1H, purine C8), 7.94(d, J=7.8 Hz, 2H, ArH), 8.02(d, J=7.8 Hz, 2H, ArH), 8.10(br s, 1H, NH) | 157.31, 153.85, 150.20, 140.50, 138.88, 135.99, 132.82(q, J=32.25 Hz), 128.47, 127.99, 127.37, 126.59(q, J=3.75 Hz), 126.40, 123.35(q, J=271.5 Hz), 113.45, 45.77, 45.45, 43.24, 42.65, 21.97 | |
| 13ea,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.87—2.93(m, 4H, piperazine), 3.76—3.80(m, 4H, piperazine), 4.48—4.55(m, 3H, CH, CH2), 7.21—7.31(m, 5H, ArH), 7.85(s, 1H, purine C8), 7.98(d, J=8.4 Hz, 2H, ArH), 8.10(br s, 1H, NH), 8.41(d, J=8.4 Hz, 2H, ArH) | 158.04, 154.34, 150.87, 150.22, 141.82, 139.21, 135.16, 128.89, 128.51, 127.51, 127.18, 124.31, 114.23, 46.42, 45.79, 44.45, 43.86, 22.48 | |
| 13f a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.75—2.78(m, 4H, piperazine), 3.75—3.80(m, 4H, piperazine), 4.49—4.52(m, 3H, CH, CH2), 7.13(d, J=8.4 Hz, 2H, ArH), 7.21—7.33(m, 5H, ArH), 7.64(d, J=8.4 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.12(br s, 1H, NH) | 163.11, 158.73, 153.25, 150.31, 139.27, 133.92, 129.90, 128.30, 127.62, 127.02, 126.87, 114.24, 110.44, 55.62, 47.16, 45.87, 44.38, 43.72, 22.21 | |
| 13g a,e | 1.42[d,J=6.6 Hz, 6H, (CH3)2], 2.84—2.90(m, 4H, piperazine), 3.76—3.81(m, 4H, piperazine), 4.50—4.56(m, 3H, CH, CH2), 7.19—7.32(m, 5H, ArH), 7.62(d, J=8.4 Hz, 2H, ArH), 7.86—7.88(m, 3H, purine C8, ArH), 8.10(br s, 1H, NH) | 158.68, 153.30, 152.36, 150.30, 139.24, 134.04, 133.96, 129.88, 128.31, 127.59, 126.90, 120.89, 120.19(q, J=258 Hz), 110.60, 47.19, 45.81, 44.40, 43.72, 22.22 | |
| 17ac,e | 0.96—1.01(m, 2H, cyclohexyl), 1.14—1.25(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.57—1.79(m, 6H, cyclohexyl), 2.41(s, 3H, CH3), 3.04—3.06(m, 4H, piperazine CH2), 3.35(br s, 2H, CH2), 3.90—3.92(m, 4H, piperazine CH2), 4.62(hept, J=6.6 Hz, 1H, CH), 5.68(br s, 1H, NH), 7.30(d, J=8.4 Hz, 2H, ArH), 7.53(s, 1H, purine C8), 7.65(d, J=8.4 Hz, 2H, ArH) | 158.30, 154.72, 150.65, 143.67, 134.70, 132.53, 129.65, 127.87, 114.02, 46.65, 46.33, 46.02, 43.85, 38.34, 31.00, 26.45, 25.94, 22.50, 21.50 | |
| 17b a,e | 0.88—0.90(m, 2H, cyclohexyl), 1.07—1.15(m, 3H, cyclohexyl), 1.46[d, J=6.6 Hz, 6H, (CH3)2], 1.57—1.68(m, 6H, cyclohexyl), 2.92—2.98(m, 4H, piperazine), 3.24(br s, 2H, CH2), 3.78—3.88(m, 4H, piperazine), 4.57(hept, J=6.6 Hz, 1H, CH), 7.45(br s, 1H, NH), 7.71(d, J=8.4 Hz, 2H, ArH), 7.76(d, J=8.4 Hz, 2H, ArH), 7.87(s, 1H, purine C8) | 158.22, 154.82, 150.63, 139.47, 134.81, 134.21, 129.37, 129.19, 114.25, 46.68, 46.33, 45.95, 43.84, 38.35, 31.01, 26.45, 25.94, 22.51 | |
| 17c a,e | 0.88—0.92(m, 2H, cyclohexyl), 1.12—1.19(m, 3H, cyclohexyl), 1.44[d, J=6.6 Hz, 6H, (CH3)2], 1.59—1.67(m, 6H, cyclohexyl), 2.91—2.97(m, 4H, piperazine), 3.22(br s, 2H, CH2), 3.77—3.83(m, 4H, piperazine), 4.54(hept, J=6.6 Hz, 1H, CH), 7.44—7.48(m, 3H, NH, ArH), 7.81—7.84(m, 3H, ArH, purine C8) | 165.28(d, J=253.5 Hz), 158.18, 154.74, 134.75, 131.82(d, J=3 Hz), 130.47(d, J=9 Hz), 116.33(d, J=22.5 Hz), 114.07, 46.65, 46.39, 45.97, 43.88, 38.36, 31.02, 26.46, 25.95, 22.50 | |
| 17dc,e | 0.97—1.03(m, 2H, cyclohexyl), 1.15—1.22(m, 3H, cyclohexyl), 1.54[d, J=6.6 Hz, 6H, (CH3)2], 1.62—1.81(m, 6H, cyclohexyl), 3.29—3.34(m, 4H, piperazine), 3.37(br s, 2H, CH2), 3.93—4.02(m, 4H, piperazine), 4.66(hept, J=6.6 Hz, 1H, CH), 6.22(br s, 1H, NH), 7.04(t, J=9.0 Hz, 2H, ArH), 7.50—7.54(m, 1H, ArH), 7.62(s, 1H, purine C8) | 159.88(dd,J=3.75, 258 Hz), 154.25, 152.36, 150.53, 134.68(t, J=11.25 Hz), 115.05(t, J=16.5 Hz), 113.23(dd, J=3.75, 24 Hz), 112.54, 47.76, 46.80, 45.54, 44.09, 38.22, 31.01, 26.43, 25.92, 22.42 | |
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
| 17ec,e | 0.97—1.01(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.51[d,J=6.6 Hz, 6H,(CH3)2], 1.57—1.79(m, 6H, cyclohexyl), 3.08—3.14(m, 4H, piperazine), 3.35(br s, 2H, CH2), 3.92—3.96(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.66(br s, 1H, NH), 7.52(s, 1H, purine C8), 7.79(d, J=8.4 Hz, 2H, ArH), 7.90(d, J=8.4 Hz, 2H, ArH) | 158.19, 154.88, 150.69, 139.47, 134.88, 134.57(q, J=33 Hz), 128.27, 126.22(q, J=3.75 Hz), 123.20(q, J=271.5 Hz), 114.35, 46.71, 46.33, 45.98, 43.85, 38.35, 31.02, 26.45, 25.95, 22.51 | |
| 17f c,e | 0.94—0.99(m, 2H, cyclohexyl), 1.14—1.26(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.53—1.77(m, 6H, cyclohexyl), 3.11—3.13(m, 4H, piperazine), 3.34(br s, 2H, CH2), 3.93—3.97(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.90(br s, 1H, NH), 7.51(s, 1H, purine C8), 7.95(d, J=9.0 Hz, 2H, ArH), 8.35(d, J=9.0 Hz, 2H, ArH) | 158.11, 154.88, 150.60, 150.22, 141.87, 134.92, 128.91, 124.31, 114.38, 46.71, 46.33, 45.89, 43.87, 38.34, 31.00, 26.44, 25.94, 22.50 | |
| 17g c,e | 0.96—1.02(m, 2H, cyclohexyl), 1.16—1.22(m, 3H, cyclohexyl), 1.52[d, J=6.6 Hz, 6H, (CH3)2], 1.67—1.80(m, 6H, cyclohexyl), 3.01—3.11(m, 4H, piperazine), 3.37(br s, 2H, CH2), 3.87(s, 3H, OCH3), 3.88—3.99(m, 4H, piperazine), 4.63(hept, J=6.6 Hz, 1H, CH), 5.72(br s, 1H, NH), 6.99(d, J=7.8 Hz, 2H, ArH), 7.51(s, 1H, purine C8), 7.72(d, J=7.8 Hz, 2H, ArH) | 163.07, 158.31, 154.80, 150.67, 134.75, 129.93, 127.15, 114.20, 55.59, 46.74, 46.29, 46.02, 43.84, 38.35, 31.00, 26.46, 25.95, 22.51 | |
| 17h c,e | 0.95—1.01(m, 2H, cyclohexyl), 1.14—1.24(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.55—1.78(m, 6H, cyclohexyl), 3.07—3.13(m, 4H, piperazine), 3.35(br s, 2H, CH2), 3.91—3.95(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.68(br s, 1H, NH), 7.34(d, J=8.4 Hz, 2H, ArH), 7.51(s, 1H, purine C8), 7.82(d, J=8.4 Hz, 2H, ArH) | 158.18, 154.81, 150.67, 139.45, 134.83, 134.14(q, J=33 Hz), 128.26, 126.22(q, J=3.75 Hz), 123.19(q, J=271.5 Hz), 114.25, 46.70, 46.35, 45.97, 43.85, 38.34, 31.01, 26.45, 25.94, 22.51 | |
| 21a a,d | 0.87—0.94(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.62—1.70(m, 6H, cyclohexyl), 2.85—3.05(m, 4H, piperazine), 3.24(br s, 2H, CH2), 3.70—3.89(m, 4H, piperazine), 4.77(s, 2H, CH2), 7.46—7.49(m, 3H, ArH, NH), 7.75(s, 1H, purine C8), 7.83(dd, J=7.2, 5.4 Hz, 2H, ArH), 13.12(br s, 1H, COOH) | 169.49, 164.64(d, J=250.5 Hz), 157.83, 154.44, 150.71, 138.31, 131.23, 130.58(d, J=9.75 Hz), 116.59(d, J=22.5 Hz), 112.66, 45.79, 45.63, 43.35, 43.18, 37.57, 30.53, 26.08, 25.45 | |
| 21ba,d | 0.85—0.98(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.62—1.71(m, 6H, cyclohexyl), 3.11—3.20(m, 4H, piperazine), 3.25(br s, 2H, CH2), 3.74—3.94(m, 4H, piperazine), 4.79(s, 2H, CH2), 7.33(t, J=9.0 Hz, 2H, ArH), 7.50(br s, 1H, NH), 7.70—7.86(m, 2H, ArH, purine C8), 13.12(br s, 1H, COOH) | 169.51, 158.92(dd, J=3.75, 255 Hz), 157.87, 154.48, 150.78, 138.34, 136.26(t, J=10.5 Hz), 113.71(dd, J=3, 24 Hz), 113.33, 112.77, 45.81, 45.00, 43.44, 43.35, 37.62, 30.54, 26.08, 25.46 | |
| 21ca,d | 0.86—0.94(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.61—1.67(m, 6H, cyclohexyl), 2.78—2.96(m, 4H, piperazine), 3.23(br s, 2H, CH2), 3.72—3.81(m, 4H, piperazine), 3.83(s, 3H, OCH3), 4.76(s, 2H, CH2), 7.13(d, J=7.8 Hz, 2H, ArH), 7.47(br s, 1H, NH), 7.68(d, J=7.8 Hz, 2H, ArH), 7.73(s, 1H, purine C8), 13.06(br s, 1H, COOH) | 169.50, 162.77, 157.79, 154.48, 150.83, 138.29, 129.74, 126.18, 114.50, 112.76, 55.62, 45.75, 45.61, 43.34, 43.21, 37.56, 30.54, 26.09, 25.47 | |
Table 2 Spectral date of all target compounds*
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
|---|---|---|---|
| 5aa,d | 4.53(br s, 2H, CH2), 7.15—7.18(m, 2H, ArH), 7.25—7.30(m, 5H, ArH), 7.90—7.95(m, 3H, ArH, purine C8), 8.25(br s, 1H, NH), 11.08(br s, 1H, SO2NH), 12.76(br s, 1H, purine C9) | 163.78(d,J=249 Hz), 153.33, 152.43, 150.55, 139.54, 138.64, 138.21, 129.80(d, J=5.25 Hz), 128.15, 127.34, 126.71, 115.60(d, J=22.5 Hz), 42.80 | |
| 5ba,d | 4.38(br s, 2H, CH2), 6.98—7.08(m, 2H, ArH), 7.20—7.27(m, 5H, ArH), 7.41—7.48(m, 1H, ArH), 7.99(s, 1H, purine C8), 8.32(br s, 1H, NH), 11.72(br s, 1H, SO2NH), 12.84(br s, 1H, purine C9) | 158.72(dd,J=4.5, 252.75 Hz), 153.14, 152.40, 149.49, 139.67, 138.64, 133.34, 128.25, 127.68, 126.98, 112,72(d, J=24 Hz), 42.76 | |
| 5ca,d | 4.49(br s, 2H, CH2), 7.25—7.31(m, 7H, ArH), 7.89—7.99(m, 3H, ArH, purine C8), 8.35(br s, 1H, NH) | 153.00, 152.84, 150.13, 141.68, 139.29, 129.19, 128.15, 127.32, 126.72, 120.54, 119.80(q, J=256.5 Hz), 111.54, 42.84 | |
| 8ab,d | 1.36[d, J=6.3 Hz, 6H, (CH3)2], 4.47—4.53(m, 3H, CH, CH2), 7.16(t, J=9.0 Hz, 2H, ArH), 7.20—7.29(m, 5H, ArH), 7.54—7.64(m, 1H, ArH), 8.21(s, 1H, purine C8), 8.46(br s, 1H, NH), 11.74(br s, 1H, SO2NH) | 158.87(d,J=255 Hz), 153.30, 152.63, 148.26, 139.12, 137.45, 134.91, 128.17, 127.52, 126.81, 118.94, 113.06(d, J=24 Hz), 113.05(t, J=11.25 Hz), 47.33, 42.73, 21.56 | |
| 8b b,d | 1.37[d, J=6.6 Hz, 6H, (CH3)2], 4.48—4.54(m, 3H, CH, CH2), 7.18—7.28(m, 5H, ArH), 7.81(d, J=7.2 Hz, 2H, ArH), 8.05(s, 1H, purine C8), 8.10(d, J=7.2 Hz, 2H, ArH), 8.36(br s, 1H, NH), 11.39(br s, 1H, SO2NH) | 154.12, 151.77, 148.66, 145.29, 139.66, 137.74, 131.94(q, J=31.5 Hz), 128.11, 128.01, 127.25, 126.64, 125.92, 123.43(q, J=271.5 Hz), 115.53, 46.58, 42.71, 21.73 | |
| 8cb,d | 1.41[d, J=6.3 Hz, 6H, (CH3)2], 4.50—4.52(m, 3H, CH, CH2), 7.16—7.22(m, 5H, ArH), 8.05(s, 1H, purine C8), 8.09(d, J=7.2 Hz, 2H, ArH), 8.18(d, J=7.2 Hz, 2H, ArH), 8.36(br s, 1H, NH), 11.49(br s, 1H, SO2NH) | 154.14, 151.62, 149.08, 147.06, 139.51, 137.71, 128.29, 127.95, 126.93, 126.48, 123.97, 115.78, 46.39, 42.59, 21.85 | |
| 8d c,d | 1.51[d, J=6.6 Hz, 6H, (CH3)2], 3.80(s, 3H, OCH3), 4.63(hept, J=6.6 Hz, 1H, CH), 4.72(br s, 2H, CH2), 6.52(br s, 1H, NH), 6.81(d, J=9.0 Hz, 2H, ArH), 7.26—7.32(m, 5H, ArH), 7.61(s, 1H, purine C8), 8.01(d, J=9.0 Hz, 2H, ArH), 8.24(br s, 1H, SO2NH) | 162.03, 154.29, 151.94, 149.12, 139.94, 137.41, 132.69, 129.44, 128.01, 127.37, 126.50, 115.84, 113.61, 59.64, 46.24, 42.58, 21.88 | |
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
| 13a a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.37(s, 3H, CH3), 2.75—2.78(m, 4H, piperazine), 3.74—3.82(m, 4H, piperazine), 4.50—4.52(m, 3H, CH, CH2), 7.22—7.32(m, 5H, ArH), 7.42(d, J=7.8 Hz, 2H, ArH), 7.58(d, J=7.8 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.11(br s, 1H, NH) | 158.80, 152.94, 150.20, 143.77, 139.13, 133.86, 132.47, 129.70, 128.31, 127.82, 127.61, 126.91, 109.51, 47.47, 45.85, 44.42, 43.72, 22.12, 21.04 | |
| 13b a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.81—2.88(m, 4H, piperazine), 3.75—3.80(m, 4H, piperazine), 4.50—4.55(m, 3H, CH, CH2), 7.21—7.32(m, 5H, ArH), 7.70(d, J=8.4 Hz, 2H, ArH), 7.72(d, J=8.4 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.09(br s, 1H, NH) | 158.74, 153.35, 150.50, 139.53, 139.08, 134.13, 133.89, 129.41, 129.15, 128.40, 127.60, 127.04, 111.01, 47.11, 45.79, 44.50, 43.77, 22.30 | |
| 13c a,d | 1.43[d, J=6.6 Hz, 6H, (CH3)2], 2.99—3.13(m, 4H, piperazine), 3.76—3.85(m, 4H, piperazine), 4.49—4.57(m, 3H, CH, CH2), 7.18—7.35(m, 7H, ArH), 7.75—7.77(m, 1H, ArH), 7.87(s, 1H, purine C8), 8.11(br s, 1H, NH) | 158.80(dd,J=3.75, 255 Hz), 157.34, 153.92, 150.21, 140.52, 136.21(t, J=11.25 Hz), 135.99, 127.90, 127.35, 126.37, 113.68(dd, J=3, 23.25 Hz), 113.35, 113.13, 45.67, 44.86, 43.40, 42.62, 21.97 | |
| 13d a,d | 1.43[d, J=6.6 Hz, 6H, (CH3)2], 2.79—3.03(m, 4H, piperazine), 3.71—3.89(m, 4H, piperazine), 4.45—4.61(m, 3H, CH, CH2), 7.19—7.32(m, 5H, ArH), 7.90(s, 1H, purine C8), 7.94(d, J=7.8 Hz, 2H, ArH), 8.02(d, J=7.8 Hz, 2H, ArH), 8.10(br s, 1H, NH) | 157.31, 153.85, 150.20, 140.50, 138.88, 135.99, 132.82(q, J=32.25 Hz), 128.47, 127.99, 127.37, 126.59(q, J=3.75 Hz), 126.40, 123.35(q, J=271.5 Hz), 113.45, 45.77, 45.45, 43.24, 42.65, 21.97 | |
| 13ea,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.87—2.93(m, 4H, piperazine), 3.76—3.80(m, 4H, piperazine), 4.48—4.55(m, 3H, CH, CH2), 7.21—7.31(m, 5H, ArH), 7.85(s, 1H, purine C8), 7.98(d, J=8.4 Hz, 2H, ArH), 8.10(br s, 1H, NH), 8.41(d, J=8.4 Hz, 2H, ArH) | 158.04, 154.34, 150.87, 150.22, 141.82, 139.21, 135.16, 128.89, 128.51, 127.51, 127.18, 124.31, 114.23, 46.42, 45.79, 44.45, 43.86, 22.48 | |
| 13f a,e | 1.42[d, J=6.6 Hz, 6H, (CH3)2], 2.75—2.78(m, 4H, piperazine), 3.75—3.80(m, 4H, piperazine), 4.49—4.52(m, 3H, CH, CH2), 7.13(d, J=8.4 Hz, 2H, ArH), 7.21—7.33(m, 5H, ArH), 7.64(d, J=8.4 Hz, 2H, ArH), 7.86(s, 1H, purine C8), 8.12(br s, 1H, NH) | 163.11, 158.73, 153.25, 150.31, 139.27, 133.92, 129.90, 128.30, 127.62, 127.02, 126.87, 114.24, 110.44, 55.62, 47.16, 45.87, 44.38, 43.72, 22.21 | |
| 13g a,e | 1.42[d,J=6.6 Hz, 6H, (CH3)2], 2.84—2.90(m, 4H, piperazine), 3.76—3.81(m, 4H, piperazine), 4.50—4.56(m, 3H, CH, CH2), 7.19—7.32(m, 5H, ArH), 7.62(d, J=8.4 Hz, 2H, ArH), 7.86—7.88(m, 3H, purine C8, ArH), 8.10(br s, 1H, NH) | 158.68, 153.30, 152.36, 150.30, 139.24, 134.04, 133.96, 129.88, 128.31, 127.59, 126.90, 120.89, 120.19(q, J=258 Hz), 110.60, 47.19, 45.81, 44.40, 43.72, 22.22 | |
| 17ac,e | 0.96—1.01(m, 2H, cyclohexyl), 1.14—1.25(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.57—1.79(m, 6H, cyclohexyl), 2.41(s, 3H, CH3), 3.04—3.06(m, 4H, piperazine CH2), 3.35(br s, 2H, CH2), 3.90—3.92(m, 4H, piperazine CH2), 4.62(hept, J=6.6 Hz, 1H, CH), 5.68(br s, 1H, NH), 7.30(d, J=8.4 Hz, 2H, ArH), 7.53(s, 1H, purine C8), 7.65(d, J=8.4 Hz, 2H, ArH) | 158.30, 154.72, 150.65, 143.67, 134.70, 132.53, 129.65, 127.87, 114.02, 46.65, 46.33, 46.02, 43.85, 38.34, 31.00, 26.45, 25.94, 22.50, 21.50 | |
| 17b a,e | 0.88—0.90(m, 2H, cyclohexyl), 1.07—1.15(m, 3H, cyclohexyl), 1.46[d, J=6.6 Hz, 6H, (CH3)2], 1.57—1.68(m, 6H, cyclohexyl), 2.92—2.98(m, 4H, piperazine), 3.24(br s, 2H, CH2), 3.78—3.88(m, 4H, piperazine), 4.57(hept, J=6.6 Hz, 1H, CH), 7.45(br s, 1H, NH), 7.71(d, J=8.4 Hz, 2H, ArH), 7.76(d, J=8.4 Hz, 2H, ArH), 7.87(s, 1H, purine C8) | 158.22, 154.82, 150.63, 139.47, 134.81, 134.21, 129.37, 129.19, 114.25, 46.68, 46.33, 45.95, 43.84, 38.35, 31.01, 26.45, 25.94, 22.51 | |
| 17c a,e | 0.88—0.92(m, 2H, cyclohexyl), 1.12—1.19(m, 3H, cyclohexyl), 1.44[d, J=6.6 Hz, 6H, (CH3)2], 1.59—1.67(m, 6H, cyclohexyl), 2.91—2.97(m, 4H, piperazine), 3.22(br s, 2H, CH2), 3.77—3.83(m, 4H, piperazine), 4.54(hept, J=6.6 Hz, 1H, CH), 7.44—7.48(m, 3H, NH, ArH), 7.81—7.84(m, 3H, ArH, purine C8) | 165.28(d, J=253.5 Hz), 158.18, 154.74, 134.75, 131.82(d, J=3 Hz), 130.47(d, J=9 Hz), 116.33(d, J=22.5 Hz), 114.07, 46.65, 46.39, 45.97, 43.88, 38.36, 31.02, 26.46, 25.95, 22.50 | |
| 17dc,e | 0.97—1.03(m, 2H, cyclohexyl), 1.15—1.22(m, 3H, cyclohexyl), 1.54[d, J=6.6 Hz, 6H, (CH3)2], 1.62—1.81(m, 6H, cyclohexyl), 3.29—3.34(m, 4H, piperazine), 3.37(br s, 2H, CH2), 3.93—4.02(m, 4H, piperazine), 4.66(hept, J=6.6 Hz, 1H, CH), 6.22(br s, 1H, NH), 7.04(t, J=9.0 Hz, 2H, ArH), 7.50—7.54(m, 1H, ArH), 7.62(s, 1H, purine C8) | 159.88(dd,J=3.75, 258 Hz), 154.25, 152.36, 150.53, 134.68(t, J=11.25 Hz), 115.05(t, J=16.5 Hz), 113.23(dd, J=3.75, 24 Hz), 112.54, 47.76, 46.80, 45.54, 44.09, 38.22, 31.01, 26.43, 25.92, 22.42 | |
| Compd. | 1H NMR, δa,b,c | 13C NMR, δd,e | |
| 17ec,e | 0.97—1.01(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.51[d,J=6.6 Hz, 6H,(CH3)2], 1.57—1.79(m, 6H, cyclohexyl), 3.08—3.14(m, 4H, piperazine), 3.35(br s, 2H, CH2), 3.92—3.96(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.66(br s, 1H, NH), 7.52(s, 1H, purine C8), 7.79(d, J=8.4 Hz, 2H, ArH), 7.90(d, J=8.4 Hz, 2H, ArH) | 158.19, 154.88, 150.69, 139.47, 134.88, 134.57(q, J=33 Hz), 128.27, 126.22(q, J=3.75 Hz), 123.20(q, J=271.5 Hz), 114.35, 46.71, 46.33, 45.98, 43.85, 38.35, 31.02, 26.45, 25.95, 22.51 | |
| 17f c,e | 0.94—0.99(m, 2H, cyclohexyl), 1.14—1.26(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.53—1.77(m, 6H, cyclohexyl), 3.11—3.13(m, 4H, piperazine), 3.34(br s, 2H, CH2), 3.93—3.97(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.90(br s, 1H, NH), 7.51(s, 1H, purine C8), 7.95(d, J=9.0 Hz, 2H, ArH), 8.35(d, J=9.0 Hz, 2H, ArH) | 158.11, 154.88, 150.60, 150.22, 141.87, 134.92, 128.91, 124.31, 114.38, 46.71, 46.33, 45.89, 43.87, 38.34, 31.00, 26.44, 25.94, 22.50 | |
| 17g c,e | 0.96—1.02(m, 2H, cyclohexyl), 1.16—1.22(m, 3H, cyclohexyl), 1.52[d, J=6.6 Hz, 6H, (CH3)2], 1.67—1.80(m, 6H, cyclohexyl), 3.01—3.11(m, 4H, piperazine), 3.37(br s, 2H, CH2), 3.87(s, 3H, OCH3), 3.88—3.99(m, 4H, piperazine), 4.63(hept, J=6.6 Hz, 1H, CH), 5.72(br s, 1H, NH), 6.99(d, J=7.8 Hz, 2H, ArH), 7.51(s, 1H, purine C8), 7.72(d, J=7.8 Hz, 2H, ArH) | 163.07, 158.31, 154.80, 150.67, 134.75, 129.93, 127.15, 114.20, 55.59, 46.74, 46.29, 46.02, 43.84, 38.35, 31.00, 26.46, 25.95, 22.51 | |
| 17h c,e | 0.95—1.01(m, 2H, cyclohexyl), 1.14—1.24(m, 3H, cyclohexyl), 1.51[d, J=6.6 Hz, 6H, (CH3)2], 1.55—1.78(m, 6H, cyclohexyl), 3.07—3.13(m, 4H, piperazine), 3.35(br s, 2H, CH2), 3.91—3.95(m, 4H, piperazine), 4.61(hept, J=6.6 Hz, 1H, CH), 5.68(br s, 1H, NH), 7.34(d, J=8.4 Hz, 2H, ArH), 7.51(s, 1H, purine C8), 7.82(d, J=8.4 Hz, 2H, ArH) | 158.18, 154.81, 150.67, 139.45, 134.83, 134.14(q, J=33 Hz), 128.26, 126.22(q, J=3.75 Hz), 123.19(q, J=271.5 Hz), 114.25, 46.70, 46.35, 45.97, 43.85, 38.34, 31.01, 26.45, 25.94, 22.51 | |
| 21a a,d | 0.87—0.94(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.62—1.70(m, 6H, cyclohexyl), 2.85—3.05(m, 4H, piperazine), 3.24(br s, 2H, CH2), 3.70—3.89(m, 4H, piperazine), 4.77(s, 2H, CH2), 7.46—7.49(m, 3H, ArH, NH), 7.75(s, 1H, purine C8), 7.83(dd, J=7.2, 5.4 Hz, 2H, ArH), 13.12(br s, 1H, COOH) | 169.49, 164.64(d, J=250.5 Hz), 157.83, 154.44, 150.71, 138.31, 131.23, 130.58(d, J=9.75 Hz), 116.59(d, J=22.5 Hz), 112.66, 45.79, 45.63, 43.35, 43.18, 37.57, 30.53, 26.08, 25.45 | |
| 21ba,d | 0.85—0.98(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.62—1.71(m, 6H, cyclohexyl), 3.11—3.20(m, 4H, piperazine), 3.25(br s, 2H, CH2), 3.74—3.94(m, 4H, piperazine), 4.79(s, 2H, CH2), 7.33(t, J=9.0 Hz, 2H, ArH), 7.50(br s, 1H, NH), 7.70—7.86(m, 2H, ArH, purine C8), 13.12(br s, 1H, COOH) | 169.51, 158.92(dd, J=3.75, 255 Hz), 157.87, 154.48, 150.78, 138.34, 136.26(t, J=10.5 Hz), 113.71(dd, J=3, 24 Hz), 113.33, 112.77, 45.81, 45.00, 43.44, 43.35, 37.62, 30.54, 26.08, 25.46 | |
| 21ca,d | 0.86—0.94(m, 2H, cyclohexyl), 1.15—1.25(m, 3H, cyclohexyl), 1.61—1.67(m, 6H, cyclohexyl), 2.78—2.96(m, 4H, piperazine), 3.23(br s, 2H, CH2), 3.72—3.81(m, 4H, piperazine), 3.83(s, 3H, OCH3), 4.76(s, 2H, CH2), 7.13(d, J=7.8 Hz, 2H, ArH), 7.47(br s, 1H, NH), 7.68(d, J=7.8 Hz, 2H, ArH), 7.73(s, 1H, purine C8), 13.06(br s, 1H, COOH) | 169.50, 162.77, 157.79, 154.48, 150.83, 138.29, 129.74, 126.18, 114.50, 112.76, 55.62, 45.75, 45.61, 43.34, 43.21, 37.56, 30.54, 26.09, 25.47 | |
| Compd. | IC50 /(μmol·L-1) | Compd. | IC50 /(μmol·L-1) | ||||
|---|---|---|---|---|---|---|---|
| PC-3 | HCT116 | K562 | PC-3 | HCT116 | K562 | ||
| 13d | 23.4 | > 50 | > 100 | 17d | 19.1 | 27.3 | 33.1 |
| 13g | 39.7 | > 50 | > 100 | Roscovitine | 12.1 | 21.2 | 26.6 |
| 17c | > 50 | 26.7 | 47.6 | ||||
Table 3 Cytotoxicity of representative compounds in vitro
| Compd. | IC50 /(μmol·L-1) | Compd. | IC50 /(μmol·L-1) | ||||
|---|---|---|---|---|---|---|---|
| PC-3 | HCT116 | K562 | PC-3 | HCT116 | K562 | ||
| 13d | 23.4 | > 50 | > 100 | 17d | 19.1 | 27.3 | 33.1 |
| 13g | 39.7 | > 50 | > 100 | Roscovitine | 12.1 | 21.2 | 26.6 |
| 17c | > 50 | 26.7 | 47.6 | ||||
| [1] | Di Virgilio F., Cancer Res., 2012, 72(21), 5441—5447 |
| [2] | Legraverend M., Grierson D. S., Bioorg. Med. Chem., 2006, 14(12), 3987—4006 |
| [3] | Chen Z., Kwong A. K. Y., Yang Z. J., Zhang L. R., Lee H. C., Zhang L. H., Chem. J. Chinese Universities, 2012, 33(6), 1226—1232 |
| (陈哲, KWONG Anna Ka-Yee, 杨振军, 张亮仁, LEE Hon Cheung, 张礼和.高等学校化学学报, 2012,33(6), 1226—1232) | |
| [4] | Huang Y., Wang J., Guo G. Y., Tao Z., Xue S. F., Zhu Q. J., Zhou Q. D., Chem. J. Chinese Universities, 2013, 34(2), 375—380 |
| (黄英, 王娟, 郭改英, 陶朱, 薛赛凤, 祝黔江, 周清娣.高等学校化学学报, 2013,34(2), 375—380) | |
| [5] | Lakshman M. K., Kumar A., Balachandran R., Day B. W., Andrei G., Snoeck R., Balzarini J., J. Org. Chem., 2012, 77(14), 5870—5883 |
| [6] | Vijay Kumar D., Hoarau C., Bursavich M., Slattum P., Gerrish D., Yager K., Saunders M., Shenderovich M., Roth B. L., McKinnon R., Chan A., Cimbora D. M., Bradford C., Reeves L., Patton S., Papac D. I., Williams B. L., Carlson R. O., Bioorg. Med. Chem. Lett., 2012, 22(13), 4377—4385 |
| [7] | Blanchard S., Soh C. K., Lee C. P., Poulsen A., Bonday Z., Goh K. L., Goh K. C., Goh M. K., Pasha M. K., Wang H., Williams M., Wood J. M., Ethirajulu K., Dymock B. W., Bioorg. Med. Chem. Lett., 2012, 22(8), 2880—2884 |
| [8] | Piccoli M., Palazzolo G., Conforti E., Lamorte G., Papini N., Creo P., Fania C., Scaringi R., Bergante S., Tringali C., Roncoroni L., Mazzoleni S., Doneda L., Galli R., Venerando B., Tettamanti G., Gelfi C., Anastasia L., J. Cell Biochem., 2012, 113(10), 3207—3217 |
| [9] | Kim S. H., Bajji A., Tangallapally R., Markovitz B., Trovato R., Shenderovich M., Baichwal V., Bartel P., Cimbora D., McKinnon R., J. Med. Chem., 2012, 55(17), 7480—7501 |
| [10] | Yenugonda V. M., Deb T. B., Grindrod S. C., Dakshanamurthy S., Yang Y., Paige M., Brown M. L., Bioorg. Med. Chem., 2011, 19(8), 2714—2725 |
| [11] | Korycka A., Lech-Maranda E., Robak T., Recent Pat. Anti-Cancer Drug Discovery, 2008, 3(2), 123—136 |
| [12] | Meijer L., Borgne A., Mulner O., Chong J. P., Blow J. J., Inagaki N., Inagaki M., Delcros J. G., Moulinoux J. P., Eur. J. Biochem., 1997, 243(1/2), 527—536 |
| [13] | Mgbonyebi O., Russo J., Russo I., Anticancer Res., 1998, 18(2A), 751—755 |
| [14] | Popowycz F., Fournet G., Schneider C., Bettayeb K., Ferandin Y., Lamigeon C., Tirado O. M., Mateo-Lozano S., Notario V., Colas P., J. Med. Chem., 2009, 52(3), 655—663 |
| [15] | Jorda R., Paruch K., Krystof V., Curr. Pharm. Des., 2012, 18(20), 2974—2980 |
| [16] | Wang Q., Su L., Liu N., Zhang L., Xu W., Fang H., Curr. Med. Chem., 2011, 18(13), 2025—2043 |
| [17] | Syed S. A. S., Rivera G., Ashfaq M., Mini-Rev. Med. Chem., 2013, 13(1), 70—86 |
| [18] | Kim H. S., Ohno M., Xu B., Kim H. O., Choi Y., Ji X. D., Maddileti S., Marquez V. E., Harden T. K., Jacobson K. A., J. Med. Chem., 2003, 46(23), 4974—4987 |
| [19] | Nair N., Kudo W., Smith M. A., Abrol R., Goddard W. A., Reddy V. P., Bioorg. Med. Chem. Lett., 2011, 21(13), 3957—3961 |
| [20] | Oumata N., Ferandin Y., Meijer L., Galons H., Org. Process Res. Dev., 2009, 13(3), 641—644 |
| [21] | Wang J., Wang Q., Zhang L., Fang H., Chin. J. Chem., 2013, 31(9), 1181—1191 |
| [1] | 肖艳华, 张广杰, 宗良, 刘国宏, 任丽君, 董俊兴. 开口箭化学成分及抗肿瘤活性[J]. 高等学校化学学报, 2019, 40(9): 1897. |
| [2] | 吕明君, 李雯, 杨新颖, 方浩. N9位芳基取代嘌呤-8-酮类衍生物的合成及抗肿瘤活性[J]. 高等学校化学学报, 2019, 40(2): 254. |
| [3] | 方芳,薛良敏,丛婧,田超,王孝伟,刘俊义,张志丽. 2-位或4-位取代吡啶并嘧啶类非经典叶酸拮抗剂的合成及抗肿瘤活性[J]. 高等学校化学学报, 2019, 40(10): 2111. |
| [4] | 张培全,杨倩倩,龙惠丹,陈鑫. 金诺芬衍生物的合成及抗肿瘤活性[J]. 高等学校化学学报, 2019, 40(10): 2097. |
| [5] | 刘莉, 马洋洋, 王宽, 贾云静, 李婉, 朱华结. β-咔啉衍生物的抗肿瘤及抗菌活性[J]. 高等学校化学学报, 2018, 39(4): 674. |
| [6] | 陈小娟, 刘根起, 任宸锐, 高敏君, 范晓东. 磺胺二甲嘧啶分子印迹二维光子晶体水凝胶传感器的研究[J]. 高等学校化学学报, 2018, 39(2): 212. |
| [7] | 王磊, 郑国钧, 季奇, 陈博, 巩龙龙, 高聪敏, 杜镇建, 张兴民. PI3K/mTOR抑制剂的合成及生物活性[J]. 高等学校化学学报, 2017, 38(9): 1590. |
| [8] | 白信法, 马宣, 解晓霞, 邵明莎, 郭宁宁, 严宁, 姚雷. 微管菌素类似物的合成及抗肿瘤活性[J]. 高等学校化学学报, 2017, 38(1): 47. |
| [9] | 史蕾, 杨文聪, 曾淑莹, 莫婷婷, 张召, 曹曼丽, 刘海洋. 咔咯钴(Ⅲ)配合物与DNA的相互作用及抗肿瘤活性[J]. 高等学校化学学报, 2016, 37(6): 1059. |
| [10] | 郭亮, 曹日晖, 范文玺, 甘紫云, 马芹. 双-β-咔啉衍生物的设计、合成及抗肿瘤活性[J]. 高等学校化学学报, 2016, 37(6): 1093. |
| [11] | 张洁, 周昌健, 谢建伟, 代斌. 大黄酸-缬氨酸加合物的合成及初步抗肿瘤活性[J]. 高等学校化学学报, 2016, 37(12): 2159. |
| [12] | 周皓, 段志刚, 赵爽, 宝梅英, 李志伟, 裴亚中. 嘧啶联苯类化合物的设计、 合成及抗肿瘤活性[J]. 高等学校化学学报, 2015, 36(9): 1694. |
| [13] | 赵晓霞, 高晓亚, 张小超, 李瑞, 王雅文, 王韵芳, 樊彩梅. 多级结构BiOCl的可控制备及对药物废水的有效降解[J]. 高等学校化学学报, 2015, 36(5): 955. |
| [14] | 王刚, 韩雷强, 方浩. 苯基哌嗪衍生物的合成及抗肿瘤活性[J]. 高等学校化学学报, 2015, 36(12): 2435. |
| [15] | 郭华, 杨承玲, 王蔚, 赖全勇, 袁直. 基于海藻酸钠衍生物的肝靶向纳米前药的构建及抗肿瘤活性研究[J]. 高等学校化学学报, 2014, 35(8): 1835. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||