

高等学校化学学报 ›› 2015, Vol. 36 ›› Issue (12): 2435.doi: 10.7503/cjcu20150397
收稿日期:2015-05-18
出版日期:2015-12-10
发布日期:2015-10-28
作者简介:联系人简介: 方 浩, 男, 博士, 教授, 博士生导师, 主要从事药物化学中抗肿瘤药物的研究. E-mail:基金资助:
WANG Gang, HAN Leiqiang, FANG Hao*( )
)
Received:2015-05-18
Online:2015-12-10
Published:2015-10-28
Contact:
FANG Hao
E-mail:haofangcn@sdu.edu.cn
Supported by:摘要:
合成了一系列结构全新的苯基哌嗪类衍生物, 采用溴化噻唑蓝四氮唑(MTT)法进行了初步体外抗肿瘤细胞增殖活性研究. 结果表明, 当哌嗪环4位连有苯磺酰胺结构时, 其1位引入苯并呋喃结构的目标化合物4t的活性最好. 化合物4t对多种瘤株具有明显抑制作用, 特别是对人乳腺癌细胞MDA-MB-231的增殖抑制强度与阳性对照药棉酚相近.
中图分类号:
TrendMD:
王刚, 韩雷强, 方浩. 苯基哌嗪衍生物的合成及抗肿瘤活性. 高等学校化学学报, 2015, 36(12): 2435.
WANG Gang, HAN Leiqiang, FANG Hao. Syntheses and Antitumor Activities of Phenylpiperazine Derivatives†. Chem. J. Chinese Universities, 2015, 36(12): 2435.
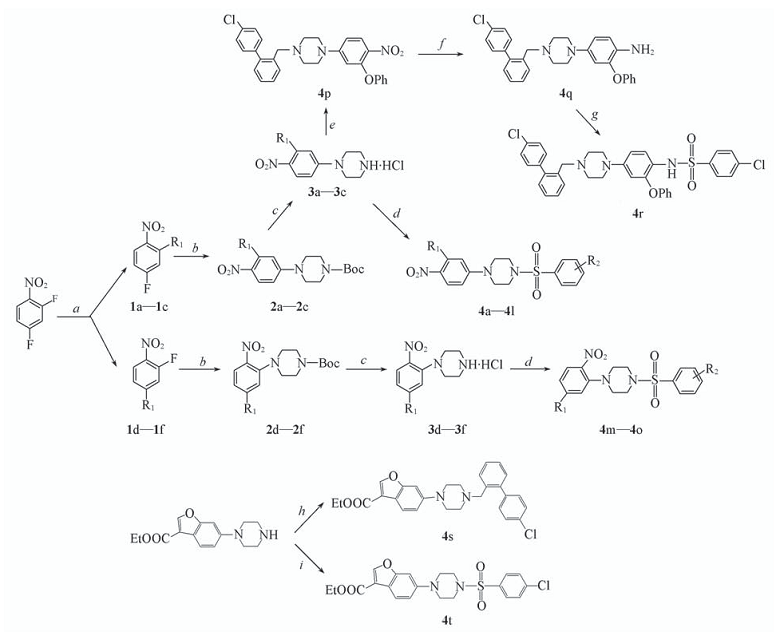
Scheme 1 Synthetic routes of target compounds 4a—4t 1a—3a, 1d—3d: R1=4-nitrophenoxyl; 1b—3b, 1e—3e: R1=phenoxyl; 1c—3c, 1f—3f: R1=8-quinolinoxyl; 4a, 4m: R1=4-nitrophenoxyl, R2=3-nitro; 4b: R1=4-nitrophenoxyl, R2=4-nitro; 4c: R1=4-nitrophenoxyl, R2=4-chloro; 4d: R1=4-nitrophenoxyl, R2=4-methoxyl; 4e, 4n: R1=phenoxyl, R2=3-nitro; 4f: R1=phenoxyl, R2=4-nitro; 4g: R1=phenoxyl, R2=4-chloro; 4h: R1=phenoxyl, R2=4-methoxyl; 4i, 4o: R1=8-quinolinoxyl, R2=3-nitro; 4j: R1=8-quinolino-xyl, R2=4-nitro; 4k: R1=8-quinolino-xyl, R2=4-chloro; 4l: R1=8-quinolinoxyl, R2=4-methoxyl a. Substituted phenol, K2CO3, DMF, r.t.; b. 1-Boc-piperazine, K2CO3, DMSO, 90 ℃; c. HCl gas, MeOH, r.t.; d. arylsulfonyl chloride, TEA, DCM, r.t.; e. R1=phenoxyl, 4'-chlorobiphenyl-2-carbaldehyde, sodium triacetoxyborohydride, DCM, r.t.; f. Zn, NH4Cl, EtOH, HOAc, reflux; g. 4-chlorobenzenesulfonyl chloride, TEA, DCM, r.t.; h. 4'-Chlorobiphenyl-2-carbaldehyde, sodium triacetoxyborohydride, DCM, r.t.; i. 4-chlorobenzenesulfonyl chloride, TEA, DCM, r.t..
| Compd. | Appearance | Yield(%) | m. p./℃ | HRMS(calcd.), m/z [M+H]+ |
|---|---|---|---|---|
| 4a | Yellow solid | 72 | >250 | 530.0977(530.0976) |
| 4b | Yellow solid | 72 | 248—250 | 530.0979(530.0976) |
| 4c | Yellow solid | 88 | 220—221 | 519.0736(519.0736) |
| 4d | Slightly yellow solid | 74 | >250 | 515.1231(515.1231) |
| 4e | Yellow solid | 69 | 164—166 | 485.1125(485.1125) |
| 4f | Yellow solid | 83 | 214—216 | 485.1125(485.1125) |
| 4g | Yellow solid | 78 | 174—176 | 474.0884(474.0885) |
| 4h | Slightly yellow solid | 95 | 182—184 | 470.1384(470.1380) |
| 4i | Yellow solid | 43 | 238—240 | 536.1234(536.1234) |
| 4j | Yellow solid | 67 | 202—204 | 536.1237(536.1234) |
| 4k | Slightly yellow solid | 89 | 212—214 | 525.0994(525.0994) |
| 4l | Off-white solid | 90 | 224—226 | 521.1486(521.1489) |
| 4m | Yellow solid | 72 | 242—244 | 530.0974(530.0976) |
| 4n | Yellow solid | 98 | 164—166 | 485.1121(485.1125) |
| 4o | Slightly yellow solid | 77 | 188—189 | 536.1236(536.1234) |
| 4q | Slightly brown solid | 73 | 64—66 | 470.1992(470.1994) |
| 4r | Slightly brown solid | 44 | 84—86 | 644.1536(644.1540) |
| 4s | Slightly yellow solid | 67 | 84—86 | 475.1786(475.1783) |
| 4t | Off-white solid | 56 | 202—204 | 449.0930(449.0932) |
Table 1 Appearances, yields, melting points and HRMS data of target compounds
| Compd. | Appearance | Yield(%) | m. p./℃ | HRMS(calcd.), m/z [M+H]+ |
|---|---|---|---|---|
| 4a | Yellow solid | 72 | >250 | 530.0977(530.0976) |
| 4b | Yellow solid | 72 | 248—250 | 530.0979(530.0976) |
| 4c | Yellow solid | 88 | 220—221 | 519.0736(519.0736) |
| 4d | Slightly yellow solid | 74 | >250 | 515.1231(515.1231) |
| 4e | Yellow solid | 69 | 164—166 | 485.1125(485.1125) |
| 4f | Yellow solid | 83 | 214—216 | 485.1125(485.1125) |
| 4g | Yellow solid | 78 | 174—176 | 474.0884(474.0885) |
| 4h | Slightly yellow solid | 95 | 182—184 | 470.1384(470.1380) |
| 4i | Yellow solid | 43 | 238—240 | 536.1234(536.1234) |
| 4j | Yellow solid | 67 | 202—204 | 536.1237(536.1234) |
| 4k | Slightly yellow solid | 89 | 212—214 | 525.0994(525.0994) |
| 4l | Off-white solid | 90 | 224—226 | 521.1486(521.1489) |
| 4m | Yellow solid | 72 | 242—244 | 530.0974(530.0976) |
| 4n | Yellow solid | 98 | 164—166 | 485.1121(485.1125) |
| 4o | Slightly yellow solid | 77 | 188—189 | 536.1236(536.1234) |
| 4q | Slightly brown solid | 73 | 64—66 | 470.1992(470.1994) |
| 4r | Slightly brown solid | 44 | 84—86 | 644.1536(644.1540) |
| 4s | Slightly yellow solid | 67 | 84—86 | 475.1786(475.1783) |
| 4t | Off-white solid | 56 | 202—204 | 449.0930(449.0932) |
| Compd. | 1H NMR(300 MHz), δ |
|---|---|
| 4a | 8.57(d, J=8.8 Hz, 1H, ArH), 8.39(s, 1H, ArH), 8.20—8.24(m, 3H, ArH), 8.10(d, J=9.2 Hz, 1H, ArH), 7.96(t, J=8.4 Hz, 1H, ArH), 7.07(d, J=9.2 Hz, 2H, ArH), 6.96(dd, J=2.4, 9.6 Hz, 1H, ArH), 6.87(d, J=2.4 Hz, 1H, ArH), 3.60(t, J=4.4 Hz, 4H, piperazine), 3.11(t, J=4.4 Hz, 4H, piperazine) |
| 4b | 8.44(d, J=5.6 Hz, 2H, ArH), 8.22(d, J=5.6 Hz, 2H, ArH), 8.10(d, J=6.4 Hz, 1H, ArH), 8.04(d, J=5.6 Hz, 2H, ArH), 7.07(d, J=5.6 Hz, 2H, ArH), 6.96(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.87(d, J=1.6 Hz, 1H, ArH), 3.59(d, J=2.8 Hz, 4H, piperazine), 3.10(d, J=2.8 Hz, 4H, piperazine) |
| 4c | 8.22(d, J=6.0 Hz, 2H, ArH), 8.10(d, J=6.4 Hz, 1H, ArH), 7.75(dd, J=6.0, 17.6 Hz, 4H, ArH), 7.08(d, J=6.0 Hz, 2H, ArH), 6.97(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.87(d, J=1.6 Hz, 1H, ArH), 3.58(s, 4H, piperazine), 3.07(s, 4H, piperazine) |
| 4d | 8.23(d, J=9.2 Hz, 2H, ArH), 8.10(d, J=9.6 Hz, 1H, ArH), 7.69(d, J=8.8 Hz, 2H, ArH), 7.17(d, J=8.8 Hz, 2H, ArH), 7.08(d, J=9.2 Hz, 2H, ArH), 6.96(dd, J=2.8, 9.6 Hz, 1H, ArH), 6.87(d, J=2.8 Hz, 1H, ArH), 3.85(s, 3H, OCH3), 3.58(t, J=4.8 Hz, 4H, piperazine), 2.95(t, J=4.8 Hz, 4H, piperazine) |
| 4e | 8.55(dd, J=0.8, 5.6 Hz, 1H, ArH), 8.37(s, 1H, ArH), 8.19(d, J=6.0 Hz, 1H, ArH), 8.02(d, J=6.4 Hz, 1H, ArH), 7.95(t, J=5.2 Hz, 1H, ArH), 7.34(t, J=5.2 Hz, 2H, ArH), 7.10(t, J=5.2 Hz, 1H, ArH), 6.90(d, J=5.2 Hz, 2H, ArH), 6.84(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.57(d, J=1.6 Hz, 1H, ArH), 3.50(t, J=3.2 Hz, 4H, piperazine), 3.08(t, J=3.2 Hz, 4H, piperazine) |
| 4f | 8.43(d, J=5.6 Hz, 2H, ArH), 8.02(dd, J=1.2, 6.0 Hz, 3H, ArH), 7.34(t, J=5.6 Hz, 2H, ArH), 7.10(t, J=4.8 Hz, 1H, ArH), 6.90(d, J=4.8 Hz, 2H, ArH), 6.85(dd, J=2.0, 6.4 Hz, 1H, ArH), 6.58(d, J=1.6 Hz, 1H, ArH), 3.49(t, J=3.2 Hz, 4H, piperazine), 3.08(t, J=3.2 Hz, 4H, piperazine) |
| 4g | 8.03(d, J=6.4 Hz, 1H, ArH), 7.75(dd, J=5.6, 14.8 Hz, 4H, ArH), 7.35(t, J=5.6 Hz, 2H, ArH), 7.10(t, J=5.2 Hz, 1H, ArH), 6.91(d, J=5.2 Hz, 2H, ArH), 6.85(dd, J=2.0, 6.0 Hz, 1H, ArH), 6.59(d, J=2.0 Hz, 1H, ArH), 3.48(t, J=3.2 Hz, 4H, piperazine), 3.00(t, J=3.2 Hz, 4H, piperazine) |
| 4h | 8.03(d, J=9.2 Hz, 1H, ArH), 7.68(d, J=8.8 Hz, 2H, ArH), 7.35(t, J=7.6 Hz, 2H, ArH), 7.17(d, J=8.8 Hz, 2H, ArH), 7.10(t, J=7.2 Hz, 1H, ArH), 6.91(d, J=8.4 Hz, 2H, ArH), 6.86(dd, J=2.4, 9.2 Hz, 1H, ArH), 6.59(d, J=2.4 Hz, 1H, ArH), 3.85(s, 3H, OCH3), 3.48(t, J=4.8 Hz, 4H, piperazine), 2.94(t, J=4.8 Hz, 4H, piperazine) |
| 4i | 8.85(dd, J=1.6, 4.4 Hz, 1H, ArH), 8.55(td, J=0.8, 8.4 Hz, 1H, ArH), 8.44(dd, J=1.2, 8.4 Hz, 1H, ArH), 8.34(d, J=1.2 Hz, 1H, ArH), 8.16(d, J=7.6 Hz, 1H, ArH), 8.06(d, J=9.6 Hz, 1H, ArH), 7.94(t, J=8.0 Hz, 1H, ArH), 7.79(d, J=8.0 Hz, 1H, ArH), 7.60(dd, J=4.0, 8.0 Hz, 1H, ArH), 7.54(t, J=8.0 Hz, 1H, ArH), 7.17(d, J=8.0 Hz, 1H, ArH), 6.81(dd, J=2.4, 9.2 Hz, 1H, ArH), 6.36(d, J=2.4 Hz, 1H, ArH), 3.38(t, J=4.8 Hz, 4H, piperazine), 3.02(t, J=4.8 Hz, 4H, piperazine) |
| 4j | 8.84(dd, J=0.8, 2.4 Hz, 1H, ArH), 8.41—8.44(m, 3H, ArH), 8.05(d, J=6.4 Hz, 1H, ArH), 7.98(d, J=5.6 Hz, 2H, ArH), 7.78(d, J=5.6 Hz, 1H, ArH), 7.60(dd, J=2.8, 5.6 Hz, 1H, ArH), 7.53(t, J=5.2 Hz, 1H, ArH), 7.16(d, J=4.8 Hz, 1H, ArH), 6.80(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.37(d, J=1.6 Hz, 1H, ArH), 3.37(t, J=3.2 Hz, 4H, piperazine), 3.00(t, J=3.2 Hz, 4H, piperazine) |
| 4k | 8.85(dd, J=0.8, 2.4 Hz, 1H, ArH), 8.44(dd, J=0.8, 5.2 Hz, 1H, ArH), 8.06(d, J=6.0 Hz, 1H, ArH), 7.79(d, J=5.2 Hz, 2H, ArH), 7.72(d, J=0.8 Hz, 4H, ArH), 7.60(dd, J=2.8, 5.6 Hz, 1H, ArH), 7.54(t, J=5.2 Hz, 1H, ArH), 7.16(d, J=5.2 Hz, 1H, ArH), 6.81(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.38(d, J=1.6 Hz, 1H, ArH), 2.93(t, J=3.2 Hz, 8H, piperazine) |
| 4l | 8.84(dd, J=0.8, 2.4 Hz, 1H, ArH), 8.43(dd, J=1.2, 5.6 Hz, 1H, ArH), 8.06(d, J=6.4 Hz, 1H, ArH), 7.79(d, J=5.2 Hz, 1H, ArH), 7.64(d, J=6.0 Hz, 2H, ArH), 7.60(dd, J=2.8, 5.6 Hz, 1H, ArH), 7.53(t, J=5.2 Hz, 1H, ArH), 7.13—7.17(m, 3H, ArH), 6.81(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.37(d, J=2.0 Hz, 1H, ArH), 3.84(s, 3H, OCH3), 3.34(t, J=3.2 Hz, 4H, piperazine), 2.86(t, J=3.2 Hz, 4H, piperazine) |
| 4m | 8.58(d, J=5.6 Hz, 1H, ArH), 8.41(s, 1H, ArH), 8.29(d, J=6.0 Hz, 2H, ArH), 8.23(d, J=5.2 Hz, 1H, ArH), 7.96—8.01(m, 2H), 7.30(d, J=5.6 Hz, 2H, ArH), 7.09(d, J=0.8 Hz, 1H, ArH), 6.84(dd, J=0.8, 6.0 Hz, 1H, ArH), 3.12(s, 8H, piperazine) |
| 4n | 8.58(d, J=5.6 Hz, 1H, ArH), 8.41(s, 1H, ArH), 8.23(d, J=4.8 Hz, 1H, ArH), 8.00(t, J=5.6 Hz, 1H, ArH), 7.92(d, J=6.0 Hz, 1H, ArH), 7.47(t, J=5.6 Hz, 2H, ArH), 7.28(t, J=5.2 Hz, 1H, ArH), 7.15(d, J=5.6 Hz, 2H, ArH), 6.89(d, J=1.6 Hz, 1H, ArH), 6.54(dd, J=1.6, 6.0 Hz, 1H, ArH), 3.11(d, J=6.4 Hz, 8H, piperazine) |
| 4o | 8.84(d, J=2.4 Hz, 1H, ArH), 8.59(d, J=5.2 Hz, 1H, ArH), 8.48(d, J=5.2 Hz, 1H, ArH), 8.42(s, 1H, ArH), 8.23(d, J=5.2 Hz, 1H, ArH), 8.00(t, J=5.2 Hz, 1H, ArH), 7.97(d, J=5.6 Hz, 1H, ArH), 7.82(d, J=6.4 Hz, 1H, ArH), 7.68(t, J=5.6 Hz, 1H, ArH), 7.60—7.62(m, 2H, ArH), 6.94(d, J=1.2 Hz, 1H, ArH), 6.27(dd, J=1.6, 6.0 Hz, 1H, ArH), 3.11(d, J=2.4 Hz, 8H, piperazine) |
| 4q | 7.46—7.50(m, 5H, ArH), 7.28—7.36(m, 4H, ArH), 7.22(dd, J=0.8, 4.8 Hz, 1H, ArH), 7.01(t, J=4.8 Hz, 1H, ArH), 6.86(d, J=5.2 Hz, 2H, ArH), 6.73(d, J=5.6 Hz, 1H, ArH), 6.59(dd, J=1.6, 5.6 Hz, 1H, ArH), 6.42(d, J=1.6 Hz, 1H, ArH), 4.39(s, 2H, NH2), 3.35(s, 2H, CH2), 2.84(s, 4H, piperazine), 2.36(s, 4H, piperazine) |
| 4r | 7.57(dd, J=2.4, 8.8 Hz, 3H, ArH), 7.33—7.36(m, 6H, ArH), 7.22—7.30(m, 5H, ArH), 7.09(t, J=6.4 Hz, 1H, ArH), 6.63—6.66(m, 2H, ArH), 6.50(d, J=8.0 Hz, 2H, ArH), 6.20(d, J=2.0 Hz, 1H, ArH), 3.39(s, 2H, CH2), 3.00(s, 4H, piperazine), 2.45(s, 4H, piperazine) |
| 4s | 7.60(d, J=0.8 Hz, 1H, ArH), 7.55(d, J=6.4 Hz, 1H, ArH), 7.52(d, J=4.8 Hz, 1H, ArH), 7.49(s, 4H, ArH), 7.36—7.40(m, 2H, ArH), 7.22—7.26(m, 2H, ArH), 7.14(d, J=1.2 Hz, 1H, ArH), 4.34(q, J=4.8 Hz, 2H, OCH2), 3.40(s, 2H, ArCH2), 3.07(s, 4H, piperazine), 2.45(s, 4H, piperazine), 1.33(t, J=4.8 Hz, 3H, CH3) |
| 4t | 7.78(dd, J=5.6, 18.0 Hz, 4H, ArH), 7.62(s, 1H, ArH), 7.58(d, J=6.4 Hz, 1H, ArH), 7.22(dd, J=1.2, 6.0 Hz, 1H, ArH), 7.18(d, J=1.2 Hz, 1H, ArH), 4.34(q, J=4.8 Hz, 2H, OCH2), 3.19(s, 4H, piperazine), 3.07(s, 4H, piperazine), 1.33(t, J=4.8 Hz, 3H, CH3) |
Table 2 1H NMR data of all target compounds*
| Compd. | 1H NMR(300 MHz), δ |
|---|---|
| 4a | 8.57(d, J=8.8 Hz, 1H, ArH), 8.39(s, 1H, ArH), 8.20—8.24(m, 3H, ArH), 8.10(d, J=9.2 Hz, 1H, ArH), 7.96(t, J=8.4 Hz, 1H, ArH), 7.07(d, J=9.2 Hz, 2H, ArH), 6.96(dd, J=2.4, 9.6 Hz, 1H, ArH), 6.87(d, J=2.4 Hz, 1H, ArH), 3.60(t, J=4.4 Hz, 4H, piperazine), 3.11(t, J=4.4 Hz, 4H, piperazine) |
| 4b | 8.44(d, J=5.6 Hz, 2H, ArH), 8.22(d, J=5.6 Hz, 2H, ArH), 8.10(d, J=6.4 Hz, 1H, ArH), 8.04(d, J=5.6 Hz, 2H, ArH), 7.07(d, J=5.6 Hz, 2H, ArH), 6.96(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.87(d, J=1.6 Hz, 1H, ArH), 3.59(d, J=2.8 Hz, 4H, piperazine), 3.10(d, J=2.8 Hz, 4H, piperazine) |
| 4c | 8.22(d, J=6.0 Hz, 2H, ArH), 8.10(d, J=6.4 Hz, 1H, ArH), 7.75(dd, J=6.0, 17.6 Hz, 4H, ArH), 7.08(d, J=6.0 Hz, 2H, ArH), 6.97(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.87(d, J=1.6 Hz, 1H, ArH), 3.58(s, 4H, piperazine), 3.07(s, 4H, piperazine) |
| 4d | 8.23(d, J=9.2 Hz, 2H, ArH), 8.10(d, J=9.6 Hz, 1H, ArH), 7.69(d, J=8.8 Hz, 2H, ArH), 7.17(d, J=8.8 Hz, 2H, ArH), 7.08(d, J=9.2 Hz, 2H, ArH), 6.96(dd, J=2.8, 9.6 Hz, 1H, ArH), 6.87(d, J=2.8 Hz, 1H, ArH), 3.85(s, 3H, OCH3), 3.58(t, J=4.8 Hz, 4H, piperazine), 2.95(t, J=4.8 Hz, 4H, piperazine) |
| 4e | 8.55(dd, J=0.8, 5.6 Hz, 1H, ArH), 8.37(s, 1H, ArH), 8.19(d, J=6.0 Hz, 1H, ArH), 8.02(d, J=6.4 Hz, 1H, ArH), 7.95(t, J=5.2 Hz, 1H, ArH), 7.34(t, J=5.2 Hz, 2H, ArH), 7.10(t, J=5.2 Hz, 1H, ArH), 6.90(d, J=5.2 Hz, 2H, ArH), 6.84(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.57(d, J=1.6 Hz, 1H, ArH), 3.50(t, J=3.2 Hz, 4H, piperazine), 3.08(t, J=3.2 Hz, 4H, piperazine) |
| 4f | 8.43(d, J=5.6 Hz, 2H, ArH), 8.02(dd, J=1.2, 6.0 Hz, 3H, ArH), 7.34(t, J=5.6 Hz, 2H, ArH), 7.10(t, J=4.8 Hz, 1H, ArH), 6.90(d, J=4.8 Hz, 2H, ArH), 6.85(dd, J=2.0, 6.4 Hz, 1H, ArH), 6.58(d, J=1.6 Hz, 1H, ArH), 3.49(t, J=3.2 Hz, 4H, piperazine), 3.08(t, J=3.2 Hz, 4H, piperazine) |
| 4g | 8.03(d, J=6.4 Hz, 1H, ArH), 7.75(dd, J=5.6, 14.8 Hz, 4H, ArH), 7.35(t, J=5.6 Hz, 2H, ArH), 7.10(t, J=5.2 Hz, 1H, ArH), 6.91(d, J=5.2 Hz, 2H, ArH), 6.85(dd, J=2.0, 6.0 Hz, 1H, ArH), 6.59(d, J=2.0 Hz, 1H, ArH), 3.48(t, J=3.2 Hz, 4H, piperazine), 3.00(t, J=3.2 Hz, 4H, piperazine) |
| 4h | 8.03(d, J=9.2 Hz, 1H, ArH), 7.68(d, J=8.8 Hz, 2H, ArH), 7.35(t, J=7.6 Hz, 2H, ArH), 7.17(d, J=8.8 Hz, 2H, ArH), 7.10(t, J=7.2 Hz, 1H, ArH), 6.91(d, J=8.4 Hz, 2H, ArH), 6.86(dd, J=2.4, 9.2 Hz, 1H, ArH), 6.59(d, J=2.4 Hz, 1H, ArH), 3.85(s, 3H, OCH3), 3.48(t, J=4.8 Hz, 4H, piperazine), 2.94(t, J=4.8 Hz, 4H, piperazine) |
| 4i | 8.85(dd, J=1.6, 4.4 Hz, 1H, ArH), 8.55(td, J=0.8, 8.4 Hz, 1H, ArH), 8.44(dd, J=1.2, 8.4 Hz, 1H, ArH), 8.34(d, J=1.2 Hz, 1H, ArH), 8.16(d, J=7.6 Hz, 1H, ArH), 8.06(d, J=9.6 Hz, 1H, ArH), 7.94(t, J=8.0 Hz, 1H, ArH), 7.79(d, J=8.0 Hz, 1H, ArH), 7.60(dd, J=4.0, 8.0 Hz, 1H, ArH), 7.54(t, J=8.0 Hz, 1H, ArH), 7.17(d, J=8.0 Hz, 1H, ArH), 6.81(dd, J=2.4, 9.2 Hz, 1H, ArH), 6.36(d, J=2.4 Hz, 1H, ArH), 3.38(t, J=4.8 Hz, 4H, piperazine), 3.02(t, J=4.8 Hz, 4H, piperazine) |
| 4j | 8.84(dd, J=0.8, 2.4 Hz, 1H, ArH), 8.41—8.44(m, 3H, ArH), 8.05(d, J=6.4 Hz, 1H, ArH), 7.98(d, J=5.6 Hz, 2H, ArH), 7.78(d, J=5.6 Hz, 1H, ArH), 7.60(dd, J=2.8, 5.6 Hz, 1H, ArH), 7.53(t, J=5.2 Hz, 1H, ArH), 7.16(d, J=4.8 Hz, 1H, ArH), 6.80(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.37(d, J=1.6 Hz, 1H, ArH), 3.37(t, J=3.2 Hz, 4H, piperazine), 3.00(t, J=3.2 Hz, 4H, piperazine) |
| 4k | 8.85(dd, J=0.8, 2.4 Hz, 1H, ArH), 8.44(dd, J=0.8, 5.2 Hz, 1H, ArH), 8.06(d, J=6.0 Hz, 1H, ArH), 7.79(d, J=5.2 Hz, 2H, ArH), 7.72(d, J=0.8 Hz, 4H, ArH), 7.60(dd, J=2.8, 5.6 Hz, 1H, ArH), 7.54(t, J=5.2 Hz, 1H, ArH), 7.16(d, J=5.2 Hz, 1H, ArH), 6.81(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.38(d, J=1.6 Hz, 1H, ArH), 2.93(t, J=3.2 Hz, 8H, piperazine) |
| 4l | 8.84(dd, J=0.8, 2.4 Hz, 1H, ArH), 8.43(dd, J=1.2, 5.6 Hz, 1H, ArH), 8.06(d, J=6.4 Hz, 1H, ArH), 7.79(d, J=5.2 Hz, 1H, ArH), 7.64(d, J=6.0 Hz, 2H, ArH), 7.60(dd, J=2.8, 5.6 Hz, 1H, ArH), 7.53(t, J=5.2 Hz, 1H, ArH), 7.13—7.17(m, 3H, ArH), 6.81(dd, J=1.6, 6.4 Hz, 1H, ArH), 6.37(d, J=2.0 Hz, 1H, ArH), 3.84(s, 3H, OCH3), 3.34(t, J=3.2 Hz, 4H, piperazine), 2.86(t, J=3.2 Hz, 4H, piperazine) |
| 4m | 8.58(d, J=5.6 Hz, 1H, ArH), 8.41(s, 1H, ArH), 8.29(d, J=6.0 Hz, 2H, ArH), 8.23(d, J=5.2 Hz, 1H, ArH), 7.96—8.01(m, 2H), 7.30(d, J=5.6 Hz, 2H, ArH), 7.09(d, J=0.8 Hz, 1H, ArH), 6.84(dd, J=0.8, 6.0 Hz, 1H, ArH), 3.12(s, 8H, piperazine) |
| 4n | 8.58(d, J=5.6 Hz, 1H, ArH), 8.41(s, 1H, ArH), 8.23(d, J=4.8 Hz, 1H, ArH), 8.00(t, J=5.6 Hz, 1H, ArH), 7.92(d, J=6.0 Hz, 1H, ArH), 7.47(t, J=5.6 Hz, 2H, ArH), 7.28(t, J=5.2 Hz, 1H, ArH), 7.15(d, J=5.6 Hz, 2H, ArH), 6.89(d, J=1.6 Hz, 1H, ArH), 6.54(dd, J=1.6, 6.0 Hz, 1H, ArH), 3.11(d, J=6.4 Hz, 8H, piperazine) |
| 4o | 8.84(d, J=2.4 Hz, 1H, ArH), 8.59(d, J=5.2 Hz, 1H, ArH), 8.48(d, J=5.2 Hz, 1H, ArH), 8.42(s, 1H, ArH), 8.23(d, J=5.2 Hz, 1H, ArH), 8.00(t, J=5.2 Hz, 1H, ArH), 7.97(d, J=5.6 Hz, 1H, ArH), 7.82(d, J=6.4 Hz, 1H, ArH), 7.68(t, J=5.6 Hz, 1H, ArH), 7.60—7.62(m, 2H, ArH), 6.94(d, J=1.2 Hz, 1H, ArH), 6.27(dd, J=1.6, 6.0 Hz, 1H, ArH), 3.11(d, J=2.4 Hz, 8H, piperazine) |
| 4q | 7.46—7.50(m, 5H, ArH), 7.28—7.36(m, 4H, ArH), 7.22(dd, J=0.8, 4.8 Hz, 1H, ArH), 7.01(t, J=4.8 Hz, 1H, ArH), 6.86(d, J=5.2 Hz, 2H, ArH), 6.73(d, J=5.6 Hz, 1H, ArH), 6.59(dd, J=1.6, 5.6 Hz, 1H, ArH), 6.42(d, J=1.6 Hz, 1H, ArH), 4.39(s, 2H, NH2), 3.35(s, 2H, CH2), 2.84(s, 4H, piperazine), 2.36(s, 4H, piperazine) |
| 4r | 7.57(dd, J=2.4, 8.8 Hz, 3H, ArH), 7.33—7.36(m, 6H, ArH), 7.22—7.30(m, 5H, ArH), 7.09(t, J=6.4 Hz, 1H, ArH), 6.63—6.66(m, 2H, ArH), 6.50(d, J=8.0 Hz, 2H, ArH), 6.20(d, J=2.0 Hz, 1H, ArH), 3.39(s, 2H, CH2), 3.00(s, 4H, piperazine), 2.45(s, 4H, piperazine) |
| 4s | 7.60(d, J=0.8 Hz, 1H, ArH), 7.55(d, J=6.4 Hz, 1H, ArH), 7.52(d, J=4.8 Hz, 1H, ArH), 7.49(s, 4H, ArH), 7.36—7.40(m, 2H, ArH), 7.22—7.26(m, 2H, ArH), 7.14(d, J=1.2 Hz, 1H, ArH), 4.34(q, J=4.8 Hz, 2H, OCH2), 3.40(s, 2H, ArCH2), 3.07(s, 4H, piperazine), 2.45(s, 4H, piperazine), 1.33(t, J=4.8 Hz, 3H, CH3) |
| 4t | 7.78(dd, J=5.6, 18.0 Hz, 4H, ArH), 7.62(s, 1H, ArH), 7.58(d, J=6.4 Hz, 1H, ArH), 7.22(dd, J=1.2, 6.0 Hz, 1H, ArH), 7.18(d, J=1.2 Hz, 1H, ArH), 4.34(q, J=4.8 Hz, 2H, OCH2), 3.19(s, 4H, piperazine), 3.07(s, 4H, piperazine), 1.33(t, J=4.8 Hz, 3H, CH3) |
| Compd. | IC50/(μmol·L-1) | ||||
|---|---|---|---|---|---|
| MDA-MB-231 | A549 | MCF-7 | Hela | KG1 | |
| 4t | 3.50±0.68 | 25.9±7.9 | 20.9±3.9 | > 42 | 17.3±0.4 |
| Gossypol | 1.40±0.24 | 4.02±0.41 | 3.00±0.87 | 8.81±1.22 | 2.87±0.77 |
Table 3 Cytotoxicity of compound 4t and gossypol
| Compd. | IC50/(μmol·L-1) | ||||
|---|---|---|---|---|---|
| MDA-MB-231 | A549 | MCF-7 | Hela | KG1 | |
| 4t | 3.50±0.68 | 25.9±7.9 | 20.9±3.9 | > 42 | 17.3±0.4 |
| Gossypol | 1.40±0.24 | 4.02±0.41 | 3.00±0.87 | 8.81±1.22 | 2.87±0.77 |
| [1] | Yang W. S., Shimada K., Delva D., Patel M., Ode E., Skouta R., Stockwell B., ACS Med. Chem. Lett., 2012, 3(1), 35—38 |
| [2] | Hamer P. C. D. W., Mir S. E., Noske D., Noorden C. J. F. V., Wudinger T., Clin. Cancer Res., 2011, 17(13), 4200—4207 |
| [3] | Tong Y. S., Torrent M., Florjancic A. S., Bromberg K. D., Buchanan F. G., Ferguson D. C., Johnson E. F., Lasko L. M., Maag D., Merta P. J., Olson A. M., Osterling D. J., Soni N., Shoemaker A. R., Penning T. D., ACS Med. Chem. Lett., 2015, 6(1), 58—62 |
| [4] | Ma Y., Lahue B. R., Gibeau C. R., Shipps G. W., Bogen S. L., Wang Y., Guo Z., Guzi T. J., ACS Med. Chem. Lett., 2014, 5(5), 572—575 |
| [5] | Deng X. M., Yang Q. K., Kwiatkowski N., Sim T., McDermott U., Settleman J. E., Lee J., Gray N. S., ACS Med. Chem. Lett., 2011, 2(3), 195—200 |
| [6] | Toogood P. L., Harvey P. J., Repine J. T., Sheehan D. J., van der Wel S. N., Zhou H., Keller P. R., McNamara D. J., Sherry D., Zhu T., Brodfuehrer J., Choi C., Barvian M. R., Fry D. W., J. Med. Chem., 2005, 48(7), 2388—2406 |
| [7] | Liu Q. S., Chang J. W., Wang J., Kang S. A., Thoreen C. C., Markhard A., Hur W., Zhang J., Sim T., Sabatini D. M., Gray N. S., J. Med. Chem., 2010, 53(19), 7146—7155 |
| [8] | Lee S., Hsu E., Chou C., Chuang H., Bai L., Kulp S. K., Chen C., J. Med. Chem., 2011, 54(18), 6364—6374 |
| [9] | Luo H. M., Yang C. L., Zhang X. Y., Zhao M. M., Jiang D., Xiao J. H., Yang X. H., Li S., Chem. Res. Chinese Universities,2013, 29(5), 906—910 |
| [10] | Chen K. X., Jiang Z. Z., Chen W. H., Xi B. M., J. South Med. Univ., 2014, 34(12), 1830—1833 |
| (陈凯旋, 江振洲, 陈文华, 习保民. 南方医科大学学报, 2014, 34(12), 1830—1833) | |
| [11] | Gong H. W., Qi H., Sun W., Jiang D., Xiao J. H., Yang X. H., Wang Y., Li S., Chem. J. Chinese Universities,2013, 34(9), 2131—2138 |
| (巩宏伟, 綦辉, 孙薇, 姜丹, 肖军海, 杨晓虹, 王应, 李松. 高等学校化学学报,2013, 34(9), 2131—2138) | |
| [12] | Shah S. S. A., Rivera G., Ashfaq M., Mini-Rev. Med. Chem., 2013, 13(1), 70—86 |
| [13] | Wang J. H., Wang Q. D., Dun Y. Y., Fang H., Chem. J. Chinese Universities,2014, 35(6), 1189—1198 |
| (王军华, 王泉德, 顿艳艳, 方浩. 高等学校化学学报,2014, 35(6), 1189—1198) | |
| [14] | Hann C. L., Daniel V. C., Sugar E. A., Dobromilskaya I., Murphy S. C., Cope L., Lin X., Hierman J. S., Wilburn D. L., Watkins D. N., Rudin C. M., Cancer Res., 2008, 68(7), 2321—2328 |
| [15] | Souers A. J., Leverson J. D., Boghaert E. R., Ackler S. L., Catron N. D., Chen J., Dayton B. D., Ding H., Enschede S. H., Fairbrother W. J., Huang D. C. S., Hymowitz S. G., Jin S., Khaw S. L., Kovar P. J., Lam L. T., Lee J., Maecker H. L., Marsh K. C., Mason K. D., Mitten M. J., Nimmer P. M., Oleksijew A., Park C. H., Park C. M., Phillips D. C., Roberts A. W., Sampath D., Seymour J. F., Smith M. L., Sullivan G. M., Tahir S. K., Tse C., Wendt M. D., Xiao Y., Xue J. C., Zhang H., Humerickhouse R. A., Rosenberg S. H., Elmore S. W., Nat. Med., 2013, 19(2), 202—208 |
| [16] | Wang X. L., Salaski E. J., Berger D. M., Powell D., Org. Lett., 2009, 11(24), 5662—5664 |
| [17] | Bourrain S., Collins I., Neduvelil J. G., Rowley M., Leeson P. D., Patel S., Patel S., Emms F., Marwood R., Chapman K. L., Fletcher A. E., Showell G. A., Bioorg. Med. Chem., 1998, 6(10), 1731—1743 |
| [18] | Klioze S. S., Allen R. C., J. Med. Chem., 1980, 23(6), 677—679 |
| [19] | Ullah N., Med. Chem., 2014, 10(5), 484—496 |
| [20] | Martin E. L., Org. Synth., 1943, 2, 501—503 |
| [1] | 赵盈喆, 张建玲. 金属-有机框架基材料在二氧化碳光催化转化中的应用[J]. 高等学校化学学报, 2022, 43(7): 20220223. |
| [2] | 杨丹, 刘旭, 戴翼虎, 祝艳, 杨艳辉. 金团簇电催化二氧化碳还原反应的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220198. |
| [3] | 孙海珠, 杨国夺, 杨柏. 碳点的设计合成、 结构调控及应用[J]. 高等学校化学学报, 2021, 42(2): 349. |
| [4] | 侯华, 王宝山. 六氟化硫替代气体绝缘强度的官能团加和理论方法[J]. 高等学校化学学报, 2021, 42(12): 3709. |
| [5] | 叶晓栋, 齐国栋, 徐君, 邓风. Au负载SBA-15分子筛上葡萄糖氧化反应[J]. 高等学校化学学报, 2020, 41(5): 960. |
| [6] | 常俊朋, 赵佳瑞, 陈思佳, 孟凯, 石微妮, 李瑞芳. 抗菌肽SAMP1及其类似肽的构效关系[J]. 高等学校化学学报, 2019, 40(4): 705. |
| [7] | 余敏, 黄晶晶, 马敏, 付瑞燕, 鄢嫣, 张福生, 殷俊峰, 谢宁宁. 三肽的锌螯合活性及定量构效关系分析[J]. 高等学校化学学报, 2018, 39(2): 234. |
| [8] | 侯华, 余小娟, 周文俊, 罗运柏, 王宝山. 绝缘气体介电强度的构效关系[J]. 高等学校化学学报, 2018, 39(11): 2477. |
| [9] | 王磊, 郑国钧, 季奇, 陈博, 巩龙龙, 高聪敏, 杜镇建, 张兴民. PI3K/mTOR抑制剂的合成及生物活性[J]. 高等学校化学学报, 2017, 38(9): 1590. |
| [10] | 刘玉明, 田丽珺, 胡栋, 聂建兵. 4-N-苯胺基喹啉衍生物的合成及胆碱酯酶抑制活性[J]. 高等学校化学学报, 2017, 38(3): 392. |
| [11] | 刘本国, 刘江伟, 李嘉琪, 耿升, 莫海珍, 梁桂兆. 类黄酮抑制P糖蛋白的三维定量构效关系与作用模式[J]. 高等学校化学学报, 2017, 38(1): 41. |
| [12] | 王美怡, 马翼, 王海英, 曹刚, 李正名. 苯环5位取代磺酰脲类化合物的水解动力学及三维定量构效关系初步研究[J]. 高等学校化学学报, 2016, 37(9): 1636. |
| [13] | 郭亮, 曹日晖, 范文玺, 甘紫云, 马芹. 双-β-咔啉衍生物的设计、合成及抗肿瘤活性[J]. 高等学校化学学报, 2016, 37(6): 1093. |
| [14] | 张洁, 周昌健, 谢建伟, 代斌. 大黄酸-缬氨酸加合物的合成及初步抗肿瘤活性[J]. 高等学校化学学报, 2016, 37(12): 2159. |
| [15] | 张静, 穆博帅, 吴萌, 边艳青, 李媛. 4-氟苯基-2,3-二氢-1,5-苯并[b]硫氮杂䓬类化合物的合成、结构和抑真菌活性[J]. 高等学校化学学报, 2015, 36(4): 687. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||