

高等学校化学学报 ›› 2026, Vol. 47 ›› Issue (3): 20250333.doi: 10.7503/cjcu20250333
何雨桐1, 李涵希2, 范晓燕3, 于美慧2( ), 张冀杰2
), 张冀杰2
收稿日期:2025-11-04
出版日期:2026-03-10
发布日期:2025-12-25
通讯作者:
于美慧
E-mail:mh@nankai.edu.cn
基金资助:
HE Yutong1, LI Hanxi2, FAN Xiaoyan3, YU Meihui2( ), ZHANG Jijie2
), ZHANG Jijie2
Received:2025-11-04
Online:2026-03-10
Published:2025-12-25
Contact:
YU Meihui
E-mail:mh@nankai.edu.cn
Supported by:摘要:
光催化水分解制氢和电催化水分解制氢是未来绿氢经济的关键技术, 但低成本、 高效且稳定的催化剂的开发仍是亟待解决的核心问题. 单原子催化剂凭借独特的电子结构和极致的原子利用率, 在催化领域展现出了巨大应用潜力. 金属有机框架材料(MOFs)具备超高比表面积、 可调变的孔隙结构及丰富的活性位点等特点, 是理想的单原子(SACs)锚定载体; 而其独特的热解特性, 又可作为SACs构筑前驱体. MOFs与单原子的复合体系(MOF-SACs)充分利用两者的协同效应, 能够显著提升催化制氢活性. 本文综合评述了近年来此类复合催化剂在光催化/电催化水分解领域的应用和研究进展, 总结了复合材料提升催化活性的策略和方法, 并对MOF-SACs催化剂的未来发展方向和研究热点进行了展望.
中图分类号:
TrendMD:
何雨桐, 李涵希, 范晓燕, 于美慧, 张冀杰. MOF基单原子催化剂在水分解制氢领域的研究进展. 高等学校化学学报, 2026, 47(3): 20250333.
HE Yutong, LI Hanxi, FAN Xiaoyan, YU Meihui, ZHANG Jijie. Research Progress of MOF-SACs in Water Splitting for Hydrogen Evolution Reaction. Chem. J. Chinese Universities, 2026, 47(3): 20250333.
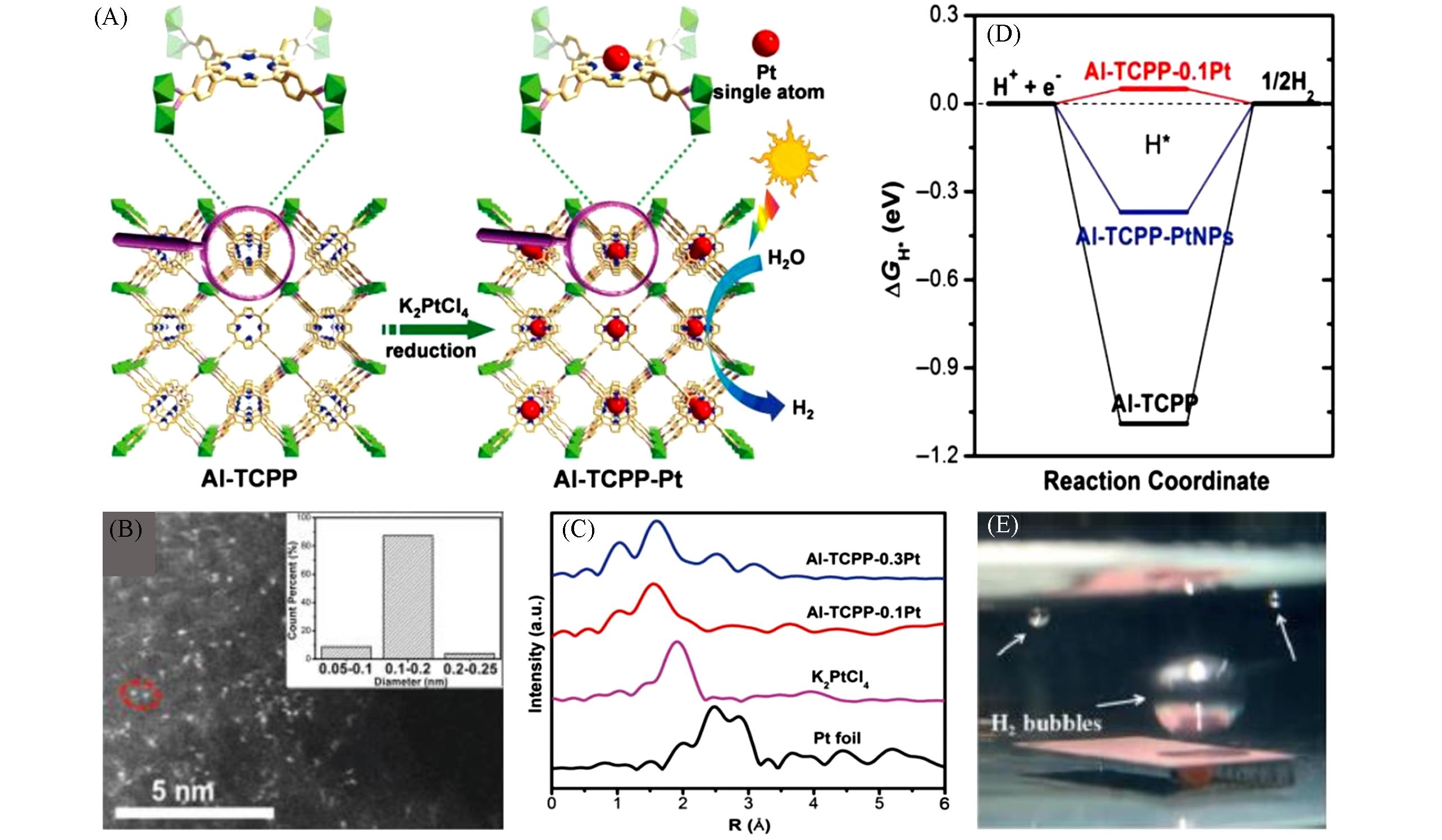
Fig.2 Schematic illustration showing the synthesis of Al⁃TCPP⁃Pt for photocatalytic hydrogen production(A), aberration⁃corrected HAADF⁃STEM image of Al⁃TCPP⁃0.1Pt(B), Fourier transformed(FT) k3⁃weighted χ(k)⁃function of the XAFS spectra for Al⁃TCPP⁃0.3Pt, Al⁃TCPP⁃0.1Pt, K2PtCl4 and Pt foil(C), calculated free energy diagram for photocatalytic H2 production(D)[23], photograph of the film producing hydrogen gas(E)[31](A—D) Copyright 2018, John Wiley and Sons; (E) Copyright 2019, John Wiley and Sons.
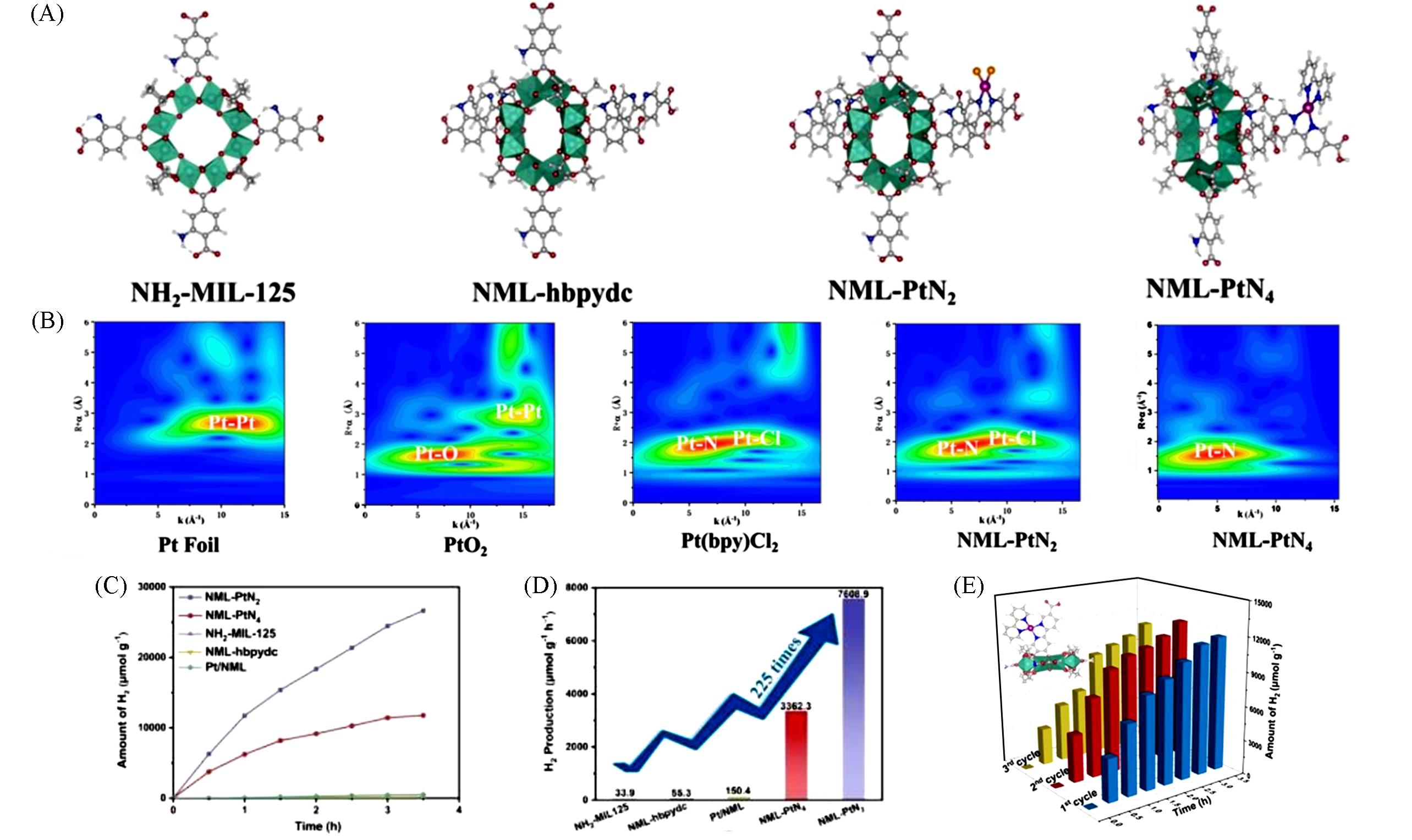
Fig.3 Structure of NML⁃hbpydc, NML⁃PtN2 and NML⁃PtN4(A), wavelet transform Pt L3⁃edge EXAFS spectra for all samples and Pt foil and PtO2(B), photocatalytic H2 production amounts of NH2⁃MIL⁃125, NML⁃hbpydc, Pt/NML, NML⁃PtN2, NML⁃PtN4(C), the comparison of photocatalytic H2 activity(D), the photocatalytic cycle stability of NML⁃PtN2(E)[32]Copyright 2024, John Wiley and Sons.
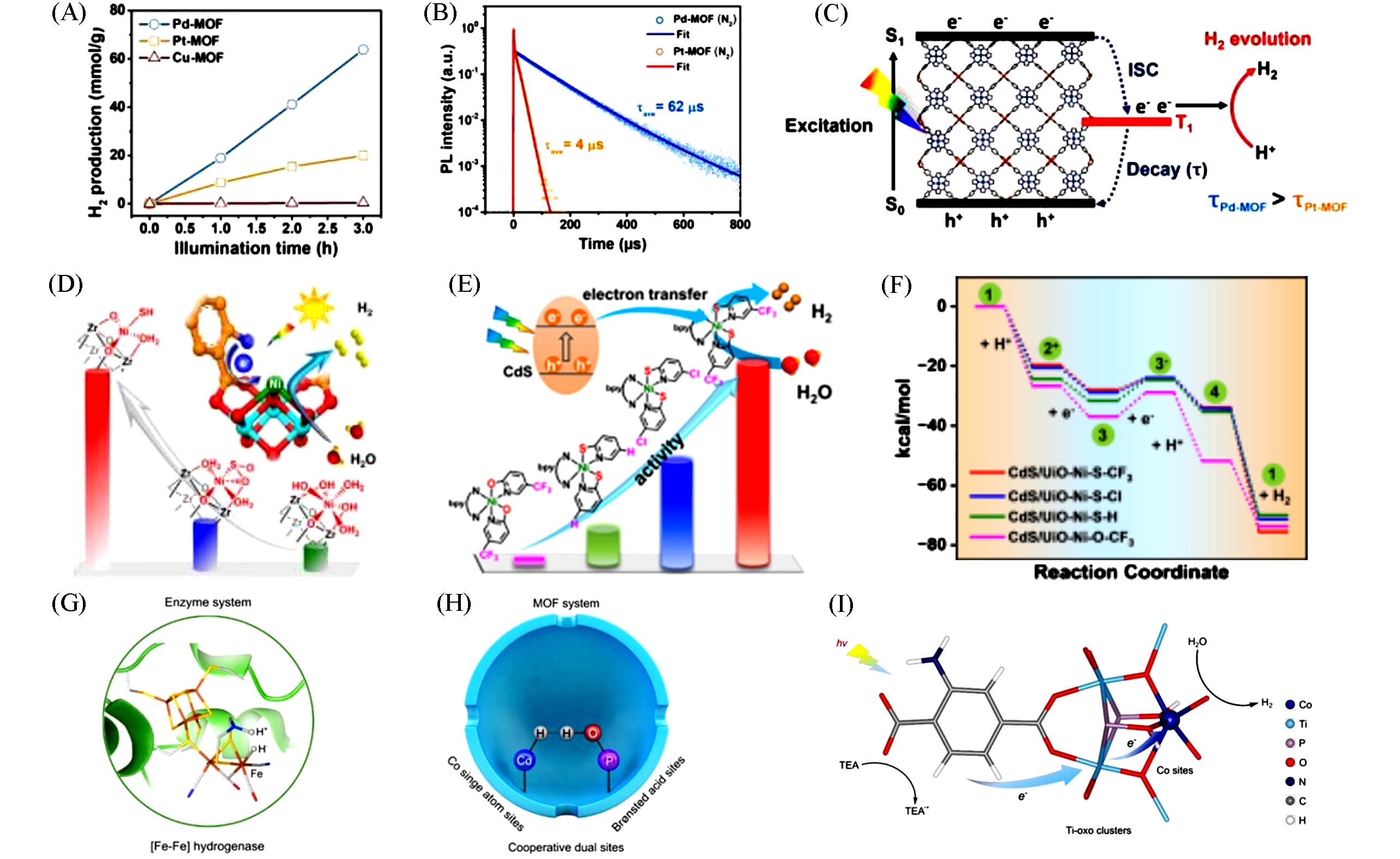
Fig.4 Photocatalytic H2 evolution under visible light irradiation(A), time⁃resolved PL spectra of Pd⁃MOF and Pt⁃MOF collected at the corresponding PL maxima(λex=355 nm) under N2 atmosphere(B), schematic illustration of the photocatalytic H2 production mechanism on Pd⁃MOF and Pt⁃MOF(C)[40], photocatalytic H2 evolution rate of Ni1⁃X/MOF(D)[44], hydrogen production yield of CdS/UiO⁃Ni⁃X⁃Y(E), calculated free energy diagrams of hydrogen production over CdS/UiO⁃Ni⁃X⁃Y(F)[45], key intermediate of cooperative dual sites in enzymatic(G) and MOF system(H), schematic illustration of the CoPOH/MIL photocatalytic system(I)[46](A—C) Copyright 2024, John Wiley and Sons; (D) Copyright 2021, American Chemical Society; (E, F) Copyright 2024, American Chemical Society; (G—I) Copyright 2025, American Chemical Society.
| Photocatalyst | Single⁃atom | Loading | Light source | Sacrificial reagent | Photocatalytic activity | Stability | Ref. |
|---|---|---|---|---|---|---|---|
| Al⁃TCPP⁃Pt | Pt | 0.07% (mass fraction) | 300 W Xe lamp (>380 nm) | TEOA | 129 µmol∙g-1∙h-1 | No Noticeable change during four runs | [ |
| MOF⁃808⁃EDTA⁃Pt | Pt | 0.98% (mass fraction) | 300 W Xe lamp (>420 nm) | TEOA Eosin Y | 68.33 mmol∙g-1∙h-1 | The Activity is kept after five cycles | [ |
| PCN⁃222(Pt) | Pt | 4.67%(mass loading) | 300 W Xe lamp (≥420 nm) | TEOA | 614 µmol∙g-1∙h-1 | Photoactivity remains at 99%, 95%, and 93% in subsequent second, third and fourth cycles | [ |
| TMF⁃Pt | Pt | 8.9% (mass fraction) | 300 W Xe lamp (>420 nm) | Ascorbic acid(AH2, 1 mol/L) | 15456 µmol∙g-1∙h-1 | Photocatalytic hydrogen release rate hardly decreased after three cycles(15 h) | [ |
| M⁃SAs@Pd⁃PCN⁃222⁃NH2 | Pt | 0.2% (mass fraction) | 300 W Xe lamp (≥420 nm) | Triisopropanolamine | 16591 µmol∙g-1∙h-1 | The high reactivity is well⁃maintained for seven runs | [ |
| PtSA⁃MNSs | Pt | 12.0% (mass fraction) | 300 W Xe lamp (>420 nm) | Ascorbic acid | 11320 µmol∙g-1∙h-1 | 93% of the initial activity retained after four cycles with 5 h for each cycle | [ |
| Ti⁃MOF@P⁃Pt₁ | Pt | 1.20% (mass fraction) | 300 W Xenon lamp (≥400 nm) | TEOA | 4193 µmol∙g-1∙h-1 | A slight activity decrease after six cycles | [ |
| TMF⁃Pt | Pt | — | 300 W Xenon lamp (>420 nm) | 1 mol/L Ascorbic acid | 33185.66 µmol∙g-1∙h-1 | The three⁃cycle test results were basically the same | [ |
| Pt@ MIL⁃125⁃NH2 ⁃TiO2nanotubes | Pt | 0.54% (atomic fraction) | 150 W Xe arc lamp with a Cornerstone motorized 1/8m monochromator | Water/ethanol=50∶50 Nafion slurry that placed on a carbon electrode | 0.943 mL/h | 100% Retention of the stability over the entire test period of 168 h | [ |
| 0.45%(atomic fraction) | 1.472 mL/h | 80% of the activity has been lost after 168 h | [ | ||||
| NH2⁃MIL⁃125⁃PtN2 | Pt | 0.78% (mass fraction) | Xe lamp with full⁃spectrum light (290 mW/cm2) | Triethanolamine | 7.608 mmol∙g-1∙h-1 | In three stability tests lasting up to 12 h each, the activity exhibited minimal decline | [ |
| CdS@PCN⁃222(Pt) | Pt | — | 300 W Xe lamp (λ≥420 nm) | Lactic acid | 71645 μmol∙g | A slight activity decrease during 25 h | [ |
| Photocatalyst | Single⁃atom | Loading | Light source | Sacrificial reagent | Photocatalytic activity | Stability | Ref. |
| TiO2/Ti⁃BPDC⁃Pt | Pt | 1.8% (mass fraction) | 300 W Xe lamp (420—760 nm) | TEOA | 12.4 mmol∙g-1∙h-1 | The efficiency is almost identical in the three cycles | [ |
| M1/SnO2/UiO⁃66⁃NH2 | Pt | 1.35% (mass fraction) | 300 W Xenon lamp(>380 nm) | TEA | 2167 µmol∙g-1∙h-1 | No noticeable activity drop occurs in 6 h | [ |
| Pd⁃TCPP | Pd | 0.89%(atomicfraction) | 450 nm LED (500 mW/cm2) | Ascorbic acid (0.1 mol/L) | 21.3 mmol∙g-1∙h-1 | High stability after 24 h illumination for all wavelengths of 405, 450, 520, and 532 nm | [ |
| Ultra⁃thin 2D Ti⁃MOF⁃Pd | Pd | 2.79% (mass fraction) | 300 W Xenon lamp(>420 nm) | Ascorbic acid | 1.32 mmol∙g-1∙h-1 | The hydrogen evolution rate only slightly decreased after five cycles(25 h) | [ |
| Ru1/UiO⁃67⁃o⁃(NH2)2 | Ru | 1.1% (mass fraction) | 300 W Xenon lamp(>380 nm) | Ethanol 5% (mass fraction) Nafion solution | 20.52 mmol∙g-1∙h-1 | No noticeable degradation is observed in the three consecutive cycles | [ |
| CdS/Fe⁃MOF⁃525 | Fe | 0.17% (mass ratio) | 300 W Xenon lamp(>420 nm) | TEOA or lactic acid | 3638.6 µmol∙g-1∙h-1 | Retained the H2 evolution amount of ca. 90.1% after five cycles(25 h) | [ |
| UiO⁃66⁃NH2MOF⁃Ni SACs⁃PDA | Ni | 2.8% (mass fraction) | Xenon lamp of 300 W power | TEA | 0.63 mmol∙g-1∙h-1 | The catalytic activity and crystallinity were retained for at least 3 cycles | [ |
| Ni1⁃S/UiO⁃66⁃NH2 | Ni | >4% (mass fraction) | Xe lamp (>380 nm) | TEA | 1360 µmol∙g-1∙h-1 | The good activity can be well maintained for at least six cycles with intact crystallinity(12 h) | [ |
| CdS/UiO⁃Ni⁃S⁃CF3 | Ni | ca. 5% (mass fraction) | Xe lamp (>380 nm) | Lactic acid | 1.87 mmol∙g-1∙h-1 | That activity can be maintained in at least three cycles with retained crystallinity(12 h) | [ |
| CoPOH/MIL | Co | 1.422% (mass fraction) | Xe lamp (>385 nm) | TEA | 6.6 mmol∙g-1∙h-1 | Maintaining its activity over six cycles(12 h) | [ |
| HNTM⁃Ir/Pt | Ir/Pt | Ir:1.05%(mass loading) Pt:2.54%(mass loading) | 300 W Xe lamp (>400 nm) | TEOA | 201.9 µmol∙g-1∙h-1 | No noticeable activity⁃decreasing occurs during the three catalytic runs | [ |
| Pd10@Pt1/UiO⁃66⁃NH2 | Pd10@Pt1 | — | Xe lamp (>380 nm) | Triethylamine | 1200.5 µmol∙g-1∙h-1 | Its activity does not present obvious decay in five consecutive runs and a continued test for 25 h | [ |
| Au@PCN⁃222(Pt) | Pt | — | Solar simulator(Abet 103 with light intensity fixed at 150 mW/cm2) | TEOA | 1015 µmol∙g-1∙h-1 | No significant change after 4 cycles | [ |
Table 1 Photocatalytic water splitting performance of MOF-SACs in different reaction environments
| Photocatalyst | Single⁃atom | Loading | Light source | Sacrificial reagent | Photocatalytic activity | Stability | Ref. |
|---|---|---|---|---|---|---|---|
| Al⁃TCPP⁃Pt | Pt | 0.07% (mass fraction) | 300 W Xe lamp (>380 nm) | TEOA | 129 µmol∙g-1∙h-1 | No Noticeable change during four runs | [ |
| MOF⁃808⁃EDTA⁃Pt | Pt | 0.98% (mass fraction) | 300 W Xe lamp (>420 nm) | TEOA Eosin Y | 68.33 mmol∙g-1∙h-1 | The Activity is kept after five cycles | [ |
| PCN⁃222(Pt) | Pt | 4.67%(mass loading) | 300 W Xe lamp (≥420 nm) | TEOA | 614 µmol∙g-1∙h-1 | Photoactivity remains at 99%, 95%, and 93% in subsequent second, third and fourth cycles | [ |
| TMF⁃Pt | Pt | 8.9% (mass fraction) | 300 W Xe lamp (>420 nm) | Ascorbic acid(AH2, 1 mol/L) | 15456 µmol∙g-1∙h-1 | Photocatalytic hydrogen release rate hardly decreased after three cycles(15 h) | [ |
| M⁃SAs@Pd⁃PCN⁃222⁃NH2 | Pt | 0.2% (mass fraction) | 300 W Xe lamp (≥420 nm) | Triisopropanolamine | 16591 µmol∙g-1∙h-1 | The high reactivity is well⁃maintained for seven runs | [ |
| PtSA⁃MNSs | Pt | 12.0% (mass fraction) | 300 W Xe lamp (>420 nm) | Ascorbic acid | 11320 µmol∙g-1∙h-1 | 93% of the initial activity retained after four cycles with 5 h for each cycle | [ |
| Ti⁃MOF@P⁃Pt₁ | Pt | 1.20% (mass fraction) | 300 W Xenon lamp (≥400 nm) | TEOA | 4193 µmol∙g-1∙h-1 | A slight activity decrease after six cycles | [ |
| TMF⁃Pt | Pt | — | 300 W Xenon lamp (>420 nm) | 1 mol/L Ascorbic acid | 33185.66 µmol∙g-1∙h-1 | The three⁃cycle test results were basically the same | [ |
| Pt@ MIL⁃125⁃NH2 ⁃TiO2nanotubes | Pt | 0.54% (atomic fraction) | 150 W Xe arc lamp with a Cornerstone motorized 1/8m monochromator | Water/ethanol=50∶50 Nafion slurry that placed on a carbon electrode | 0.943 mL/h | 100% Retention of the stability over the entire test period of 168 h | [ |
| 0.45%(atomic fraction) | 1.472 mL/h | 80% of the activity has been lost after 168 h | [ | ||||
| NH2⁃MIL⁃125⁃PtN2 | Pt | 0.78% (mass fraction) | Xe lamp with full⁃spectrum light (290 mW/cm2) | Triethanolamine | 7.608 mmol∙g-1∙h-1 | In three stability tests lasting up to 12 h each, the activity exhibited minimal decline | [ |
| CdS@PCN⁃222(Pt) | Pt | — | 300 W Xe lamp (λ≥420 nm) | Lactic acid | 71645 μmol∙g | A slight activity decrease during 25 h | [ |
| Photocatalyst | Single⁃atom | Loading | Light source | Sacrificial reagent | Photocatalytic activity | Stability | Ref. |
| TiO2/Ti⁃BPDC⁃Pt | Pt | 1.8% (mass fraction) | 300 W Xe lamp (420—760 nm) | TEOA | 12.4 mmol∙g-1∙h-1 | The efficiency is almost identical in the three cycles | [ |
| M1/SnO2/UiO⁃66⁃NH2 | Pt | 1.35% (mass fraction) | 300 W Xenon lamp(>380 nm) | TEA | 2167 µmol∙g-1∙h-1 | No noticeable activity drop occurs in 6 h | [ |
| Pd⁃TCPP | Pd | 0.89%(atomicfraction) | 450 nm LED (500 mW/cm2) | Ascorbic acid (0.1 mol/L) | 21.3 mmol∙g-1∙h-1 | High stability after 24 h illumination for all wavelengths of 405, 450, 520, and 532 nm | [ |
| Ultra⁃thin 2D Ti⁃MOF⁃Pd | Pd | 2.79% (mass fraction) | 300 W Xenon lamp(>420 nm) | Ascorbic acid | 1.32 mmol∙g-1∙h-1 | The hydrogen evolution rate only slightly decreased after five cycles(25 h) | [ |
| Ru1/UiO⁃67⁃o⁃(NH2)2 | Ru | 1.1% (mass fraction) | 300 W Xenon lamp(>380 nm) | Ethanol 5% (mass fraction) Nafion solution | 20.52 mmol∙g-1∙h-1 | No noticeable degradation is observed in the three consecutive cycles | [ |
| CdS/Fe⁃MOF⁃525 | Fe | 0.17% (mass ratio) | 300 W Xenon lamp(>420 nm) | TEOA or lactic acid | 3638.6 µmol∙g-1∙h-1 | Retained the H2 evolution amount of ca. 90.1% after five cycles(25 h) | [ |
| UiO⁃66⁃NH2MOF⁃Ni SACs⁃PDA | Ni | 2.8% (mass fraction) | Xenon lamp of 300 W power | TEA | 0.63 mmol∙g-1∙h-1 | The catalytic activity and crystallinity were retained for at least 3 cycles | [ |
| Ni1⁃S/UiO⁃66⁃NH2 | Ni | >4% (mass fraction) | Xe lamp (>380 nm) | TEA | 1360 µmol∙g-1∙h-1 | The good activity can be well maintained for at least six cycles with intact crystallinity(12 h) | [ |
| CdS/UiO⁃Ni⁃S⁃CF3 | Ni | ca. 5% (mass fraction) | Xe lamp (>380 nm) | Lactic acid | 1.87 mmol∙g-1∙h-1 | That activity can be maintained in at least three cycles with retained crystallinity(12 h) | [ |
| CoPOH/MIL | Co | 1.422% (mass fraction) | Xe lamp (>385 nm) | TEA | 6.6 mmol∙g-1∙h-1 | Maintaining its activity over six cycles(12 h) | [ |
| HNTM⁃Ir/Pt | Ir/Pt | Ir:1.05%(mass loading) Pt:2.54%(mass loading) | 300 W Xe lamp (>400 nm) | TEOA | 201.9 µmol∙g-1∙h-1 | No noticeable activity⁃decreasing occurs during the three catalytic runs | [ |
| Pd10@Pt1/UiO⁃66⁃NH2 | Pd10@Pt1 | — | Xe lamp (>380 nm) | Triethylamine | 1200.5 µmol∙g-1∙h-1 | Its activity does not present obvious decay in five consecutive runs and a continued test for 25 h | [ |
| Au@PCN⁃222(Pt) | Pt | — | Solar simulator(Abet 103 with light intensity fixed at 150 mW/cm2) | TEOA | 1015 µmol∙g-1∙h-1 | No significant change after 4 cycles | [ |
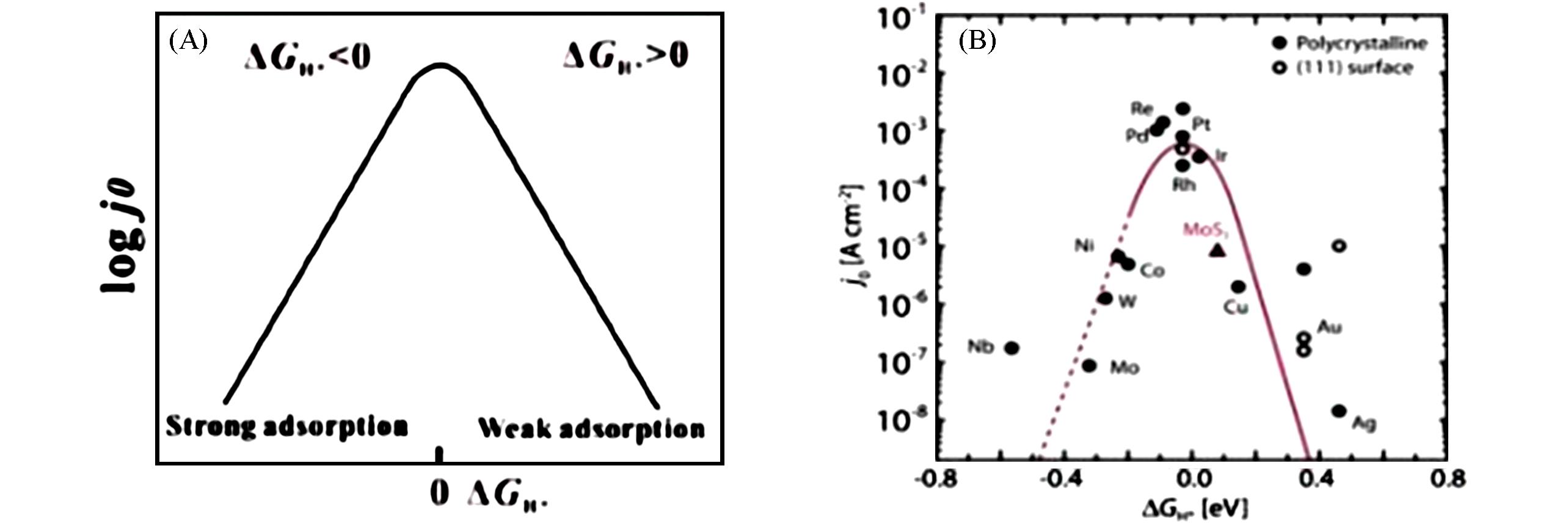
Fig.5 Relationship between j0 and ΔGH* under the assumption of a Langmuir adsorption model(A), HER volcano plot for metals⁃supported MoS2(B)[52]Copyright 2022, John Wiley and Sons.
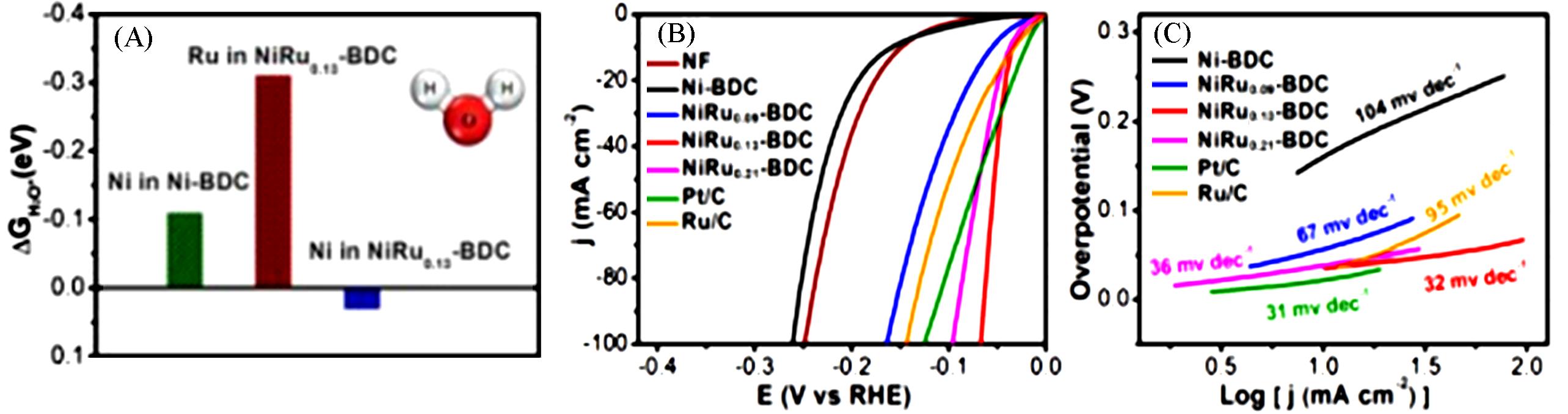
Fig.6 Calculated adsorption free energy of water on Ni⁃BDC and NiRu0.13⁃BDC(A), LSV curves toward HER(B) and Tafel plots(C) of Ni⁃BDC, NiRu0.09⁃BDC, NiRu0.13⁃BDC, NiRu0.21⁃BDC in 1 mol/L KOH[57]Copyright 2021, Spring Nature.
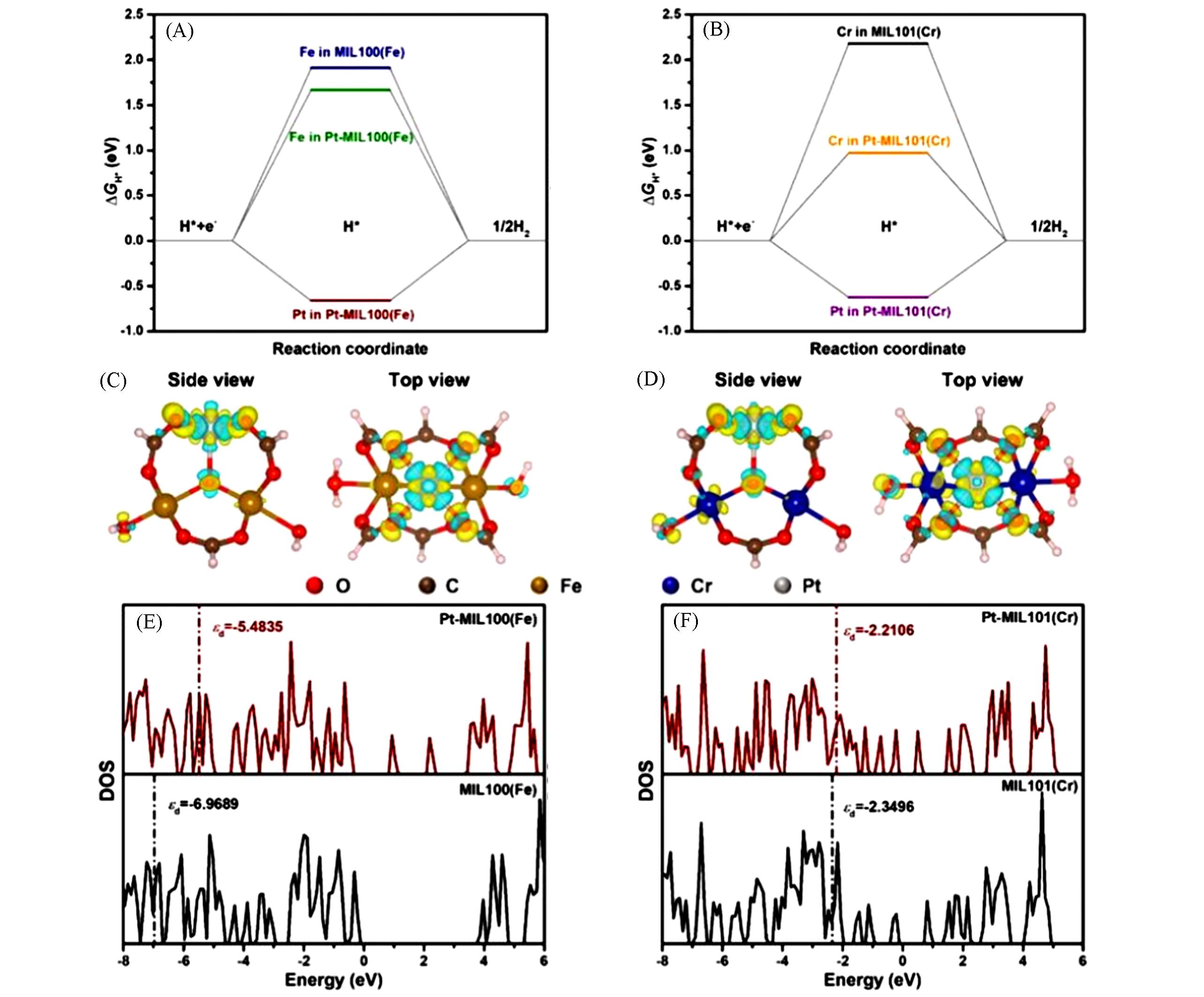
Fig.7 Free⁃energy diagram for the HER on Fe in MIL100(Fe), Fe in Pt⁃MIL100(Fe), and Pt in Pt⁃MIL100(Fe)(A), Cr in MIL101(Cr), Cr in Pt⁃MIL101(Cr), and Pt in Pt⁃MIL101(Cr)(B), difference charge density of Pt⁃MIL100(Fe)(C) and Pt⁃MIL101(Cr)(D), comparison of calculated DOS of MIL100(Fe) and Pt⁃MIL100(Fe)(E) and MIL101(Cr) and Pt⁃MIL101(Cr)(F)[61](C) and (D) Charge accumulation and depletion are shown in yellow and blue, respectively. Copyright 2023, the Royal Society of Chemistry.
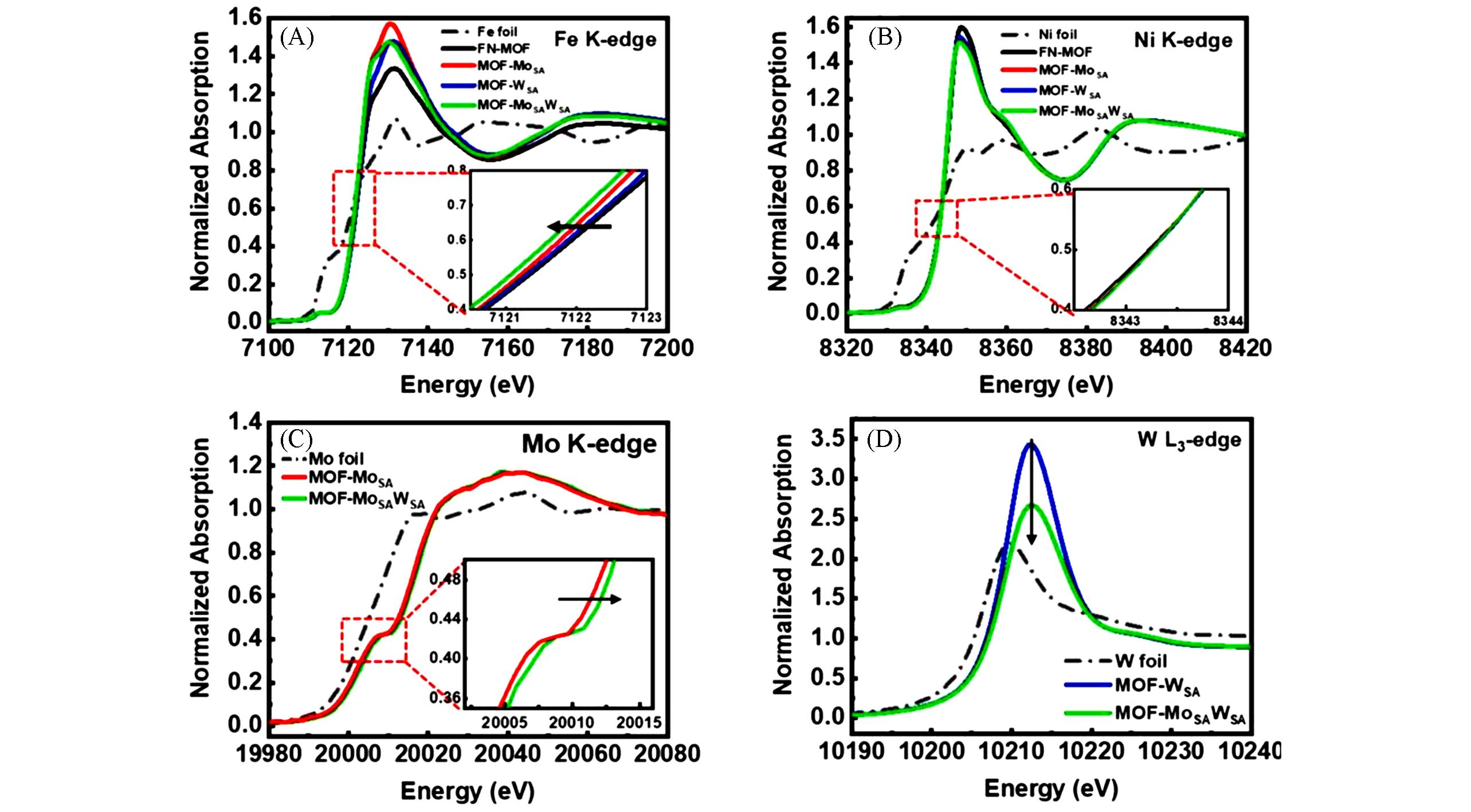
Fig.8 XANES spectra at Fe K⁃edge(A), Ni K⁃edge(B), Mo K⁃edge(C) and W L3⁃edge(D) of FN⁃MOF, MOF⁃MoSA, MOF⁃WSA, MOF⁃MoSAWSA, and corresponding reference metal foils[65]Insets show locally enlarged absorption edges. Copyright 2023, Elsevier.
| Electrocatalyst | Single⁃ atom | Loading (mass fraction) | Electrolyte | Overpotential (10 mA/cm2) | Tafel slope | Stability | Ref. |
|---|---|---|---|---|---|---|---|
| NiRu0.13⁃BDC | Ru | 0.13% | 1 mol/L Phosphate buffered saline solution | 36 mV | 32 mV/dec | Good stability with a negligible current decrease after 30 h test | [ |
| Co⁃BPDC⁃Ru Sas | Ru | — | 1.0 mol/L KOH | 37 mV | 73.22 mV/dec | Steadily work for 300 h at a current density of 10 mA/cm2 | [ |
| RuCo⁃CAT | Ru doping | 0.9% | 1.0 mol/L KOH | 38 mV | 32.1 mV/dec | Current retention of 93.1% at 10 mA/cm2 for 100 h | [ |
| CoIr⁃BDC(terephthalic acid) | Ir doping | — | 0.5 mol/L H2SO4 | 39 mV | 35 mV/dec | — | [ |
| MOF⁃MoSAWSA | Mo W | Mo: 0.43%, W: 0.90% | 1.0 mol/L KOH | 57 mV | 82.4 mV/dec | Only 1% decay in current densities after a 50 h operation | [ |
| {Cu(NH4)2NiMo6O24H6}@HKUST⁃1 | Ni | 2.93% | 1.0 mol/L KOH | 100.03 mV | 76.9 mV/dec | Slight increase in overpotential and slight decrease in the activity after 48 h test | [ |
Table 2 HER performance of hybrid catalysts in different reaction environments
| Electrocatalyst | Single⁃ atom | Loading (mass fraction) | Electrolyte | Overpotential (10 mA/cm2) | Tafel slope | Stability | Ref. |
|---|---|---|---|---|---|---|---|
| NiRu0.13⁃BDC | Ru | 0.13% | 1 mol/L Phosphate buffered saline solution | 36 mV | 32 mV/dec | Good stability with a negligible current decrease after 30 h test | [ |
| Co⁃BPDC⁃Ru Sas | Ru | — | 1.0 mol/L KOH | 37 mV | 73.22 mV/dec | Steadily work for 300 h at a current density of 10 mA/cm2 | [ |
| RuCo⁃CAT | Ru doping | 0.9% | 1.0 mol/L KOH | 38 mV | 32.1 mV/dec | Current retention of 93.1% at 10 mA/cm2 for 100 h | [ |
| CoIr⁃BDC(terephthalic acid) | Ir doping | — | 0.5 mol/L H2SO4 | 39 mV | 35 mV/dec | — | [ |
| MOF⁃MoSAWSA | Mo W | Mo: 0.43%, W: 0.90% | 1.0 mol/L KOH | 57 mV | 82.4 mV/dec | Only 1% decay in current densities after a 50 h operation | [ |
| {Cu(NH4)2NiMo6O24H6}@HKUST⁃1 | Ni | 2.93% | 1.0 mol/L KOH | 100.03 mV | 76.9 mV/dec | Slight increase in overpotential and slight decrease in the activity after 48 h test | [ |
| [1] | Molaei M. J., Fuel, 2024, 365, 131159 |
| [2] | Zhao C., Chen Z., Shi R., Yang X., Zhang T., Adv. Mater., 2020, 32(28), e1907296 |
| [3] | Shi X. F., Zhu J., Bai T. Y., Fu Z. X., Zhang J. J., Bu X. H., Chem. J. Chinese Universities, 2022, 43(1), 20210613 |
| 史潇凡, 朱剑, 白田宇, 付子萱, 张冀杰, 卜显和. 高等学校化学学报, 2022, 43(1), 20210613 | |
| [4] | Zhang Y., Ma D., Li J., Zhi C., Zhang Y., Liang L., Mao S., Shi J. W., Coord. Chem. Rev., 2024, 517, 215995 |
| [5] | Yang J., Yang S., An L., Zhu J., Xiao J., Zhao X., Wang D., ACS Catal., 2024, 14(5), 3466—3474 |
| [6] | Hanan A., Awan H. T. A., Bibi F., Sulaiman R. R. R., Wong W. Y., Walvekar R., Singh S., Khalid M., J. Energy Chem., 2024, 92, 176—206 |
| [7] | Zhao D., Zhuang Z., Cao X., Zhang C., Peng Q., Chen C., Li Y., Chem. Soc. Rev., 2020, 49(7), 2215—2264 |
| [8] | Zhang X., Lian X., Jiao H., Wang Y., Li H., Chang X., Li Y., Zhang J., Bu X. H., Adv. Funct. Mater., 2025, 2508730 |
| [9] | Wang H., Li X., Deng Y., Jiang J., Ma H., Zou J., Coordin. Chem. Rev., 2025, 529, 216462 |
| [10] | Liu H., Cheng M., Liu Y., Wang J., Zhang G., Li L., Du L., Wang G., Yang S., Wang X., Energy Environ. Sci., 2022, 15(9), 3722—3749 |
| [11] | Vallem S., Sibi M. G., Patil R., Goyal V., Giridhar Babu A., Lohith E. A., Keerthi K., Umer M., Jyothi N. V. V., Vandichel M., Energy Environ. Sci., 2025, 18, 9632—9712 |
| [12] | Yuan Z., Yu Y., Xie Q., Ding L., Lei W., Zhang H., Yao Y., Wang Y., Adv. Mater. Interfaces, 2025, 12, 2400916 |
| [13] | Sun N., Shah S. S. A., Lin Z., Zheng Y. Z., Jiao L., Jiang H. L., Chem. Rev., 2025, 125(5), 2703—2792 |
| [14] | Fujishima A., Honda K., Nature, 1972, 238(5358), 37—38 |
| [15] | Bard A. J., Science, 1980, 207(4427), 139—144 |
| [16] | Chen X., Shen S., Guo L., Mao S. S., Chem. Rev., 2010, 110(11), 6503—6570 |
| [17] | Kataoka Y., Sato K., Miyazaki Y., Masuda K., Tanaka H., Naito S., Mori W., Energy Environ. Sci., 2009, 2(4), 344—345 |
| [18] | Zhang J., Bai T., Huang H., Yu M. H., Fan X., Chang Z., Bu X. H., Adv. Mater., 2020, 32(49), e2004747 |
| [19] | Remiro⁃Buenamañana S., Cabrero⁃Antonino M., Martínez⁃Guanter M., Álvaro M., Navalón S., García H., Appl. Catal. B: Environ., 2019, 254, 677—684 |
| [20] | Zhang H., Zuo S., Qiu M., Wang S., Zhang Y., Zhang J., Lou X. W. D., Sci. Adv., 2020, 6(39), eabb9823 |
| [21] | Gao M., Tian F., Guo Z., Zhang X., Li Z., Zhou J., Zhou X., Yu Y., Yang W., Chem. Eng. J., 2022, 446, 137127 |
| [22] | Li W., Chu X., Wang F., Dang Y., Liu X., Wang X., Wang C., Appl. Catal. B: Environ., 2021, 288, 120034 |
| [23] | Fang X., Shang Q., Wang Y., Jiao L., Yao T., Li Y., Zhang Q., Luo Y., Jiang H. L., Adv. Mater., 2018, 30(7), 1705112 |
| [24] | Li J., Huang H., Liu P., Song X., Mei D., Tang Y., Wang X., Zhong C., J. Catal., 2019, 375, 351—360 |
| [25] | Zhang H., Li Q., Li B., Weng B., Tian Z., Yang J., Hofkens J., Lai F., Liu T., J. Catal., 2022, 407, 1—9 |
| [26] | Feng H., Li H., Liu X., Huang Y., Pan Q., Peng R., Du R., Zheng X., Yin Z., Li S., He Y., Chem. Eng. J., 2022, 428, 132045 |
| [27] | Mo Q., Zhang L., Li S., Song H., Fan Y., Su C. Y., J. Am. Chem. Soc., 2022, 144(49), 22747—22758 |
| [28] | Zhu Y., Song N., Liu S., Zhang K., Liu B., Wang Y., Green Chem., 2025, 27(26), 7973—7981 |
| [29] | Li H., Liu X., He Y., Feng H., Zhang Y., Liu C., Wu Z., Fuel, 2023, 338, 127369 |
| [30] | Qin S., Guo J., Chen X., Cao R., Denisov N., Song Y. Y., Schmuki P., J. Mater. Chem. A, 2023, 11(33), 17759—17768 |
| [31] | Zuo Q., Liu T., Chen C., Ji Y., Gong X., Mai Y., Zhou Y., Angew. Chem. Int. Ed., 2019, 58(30), 10198—10203 |
| [32] | Zhang X., Li Z., Li H., Yang D., Ren Z., Zhang Y., Zhang J., Bu X. H., Angew. Chem. Int. Ed., 2024, 63(30), e202404386 |
| [33] | Guan G. W., Zheng S. T., Xia M., Li K. X., Ouyang Y. S., Yang G., Yang Q. Y., Chem. Eng. J., 2023, 464, 142530 |
| [34] | Dash S., Tripathy S. P., Subudhi S., Behera P., Mishra B. P., Panda J., Parida K., Langmuir, 2024, 40(8), 4514—4530 |
| [35] | Zhou G., Wu M. F., Xing Q. J., Li F., Liu H., Luo X. B., Zou J. P., Luo J. M., Zhang A. Q., Appl. Catal. B: Environ., 2018, 220, 607—614 |
| [36] | Navalon S., Dhakshinamoorthy A., Alvaro M., Ferrer B., Garcia H., Chem. Rev., 2023, 123(1), 445—490 |
| [37] | He X., Ding Y., Huang Z., Liu M., Chi M., Wu Z., Segre C. U., Song C., Wang X., Guo X., Angew. Chem. Int. Ed., 2023, 62(25), e202217439 |
| [38] | Sui J., Liu H., Hu S., Sun K., Wan G., Zhou H., Zheng X., Jiang H. L., Adv. Mater., 2022, 34(6), e2109203 |
| [39] | Liu X., Jia C., Jiang G., Zhang C., Chen M., Zhao X., Zhang X., Fu M., Li S., Wu J., Jia Y., He Y., Chin. Chem. Lett., 2024, 35(9), 109455 |
| [40] | Kim J. H., Wu S., Zdrazil L., Denisov N., Schmuki P., Angew. Chem. Int. Ed., 2024, 63(7), e202319255 |
| [41] | Hu S., Gao M. L., Huang J., Wang H., Wang Q., Yang W., Sun Z., Zheng X., Jiang H. L., J. Am. Chem. Soc., 2024, 146(29), 20391—20400 |
| [42] | Chiromo H., Nyakuchena J., Streater D., Ma Q., Turchiano C., Zhang X., Gu J., Huang J., J. Phys. Chem. C, 2023, 127(41), 20354—20359 |
| [43] | Wu Y., Qu Y., Su C., Yang X., Yang Y., Zhang Y., Huang W., Inorg. Chem., 2023, 62(51), 21290—21298 |
| [44] | Ma X., Liu H., Yang W., Mao G., Zheng L., Jiang H. L., J. Am. Chem. Soc., 2021, 143(31), 12220—12229 |
| [45] | Yang G., Wang D., Wang Y., Hu W., Hu S., Jiang J., Huang J., Jiang H. L., J. Am. Chem. Soc., 2024, 146(15), 10798—10805 |
| [46] | Jung Y., Lee C. W., Lee B. H., Yu Y., Moon J., Lee H. S., Ko W., Bok J., Lee K., Lee J., Bootharaju M. S., Ryu J., Kim M., Hyeon T., J. Am. Chem. Soc., 2025, 147(2), 1740—1748 |
| [47] | He T., Chen S., Ni B., Gong Y., Wu Z., Song L., Gu L., Hu W., Wang X., Angew. Chem. Int. Ed., 2018, 57(13), 3493—3498 |
| [48] | Pan Y., Qian Y., Zheng X., Chu S. Q., Yang Y., Ding C., Wang X., Yu S. H., Jiang H. L., Natl. Sci. Rev., 2021, 8(1), nwaa224 |
| [49] | Pena E., Becerra J., Gopalakrishnan V. N., Do T. O., Mol. Catal., 2023, 548, 113470 |
| [50] | Quan L., Jiang H., Mei G., Sun Y., You B., Chem. Rev., 2024, 124(7), 3694—3812 |
| [51] | Chen H., Liang X., Liu Y., Ai X., Asefa T., Zou X., Adv. Mater., 2020, 32(44), e2002435 |
| [52] | Zhang C., Wang H., Yu H., Yi K., Zhang W., Yuan X., Huang J., Deng Y., Zeng G., Adv. Energy Mater., 2022, 12(27), 2200875 |
| [53] | Li C., Zhang H., Liu M., Lang F. F., Pang J., Bu X. H., Ind. Chem. Mater., 2023, 1(1), 9—38 |
| [54] | Zhang Z., Liu G., Cui X., Chen B., Zhu Y., Gong Y., Saleem F., Xi S., Du Y., Borgna A., Lai Z., Zhang Q., Li B., Zong Y., Han Y., Gu L., Zhang H., Adv. Mater., 2018, 30(30), e1801741 |
| [55] | Yao R., Sun K., Zhang K., Wu Y., Du Y., Zhao Q., Liu G., Chen C., Sun Y., Li J., Nat. Commun., 2024, 15(1), 2218 |
| [56] | Ding H., Su C., Wu J., Lv H., Tan Y., Tai X., Wang W., Zhou T., Lin Y., Chu W., Wu X., Xie Y., Wu C., J. Am. Chem. Soc., 2024, 146(11), 7858—7867 |
| [57] | Sun Y., Xue Z., Liu Q., Jia Y., Li Y., Liu K., Lin Y., Liu M., Li G., Su C. Y., Nat. Commun., 2021, 12(1), 1369 |
| [58] | Li X., Luo D., Jiang F., Zhang K., Wang S., Li S., Zha Q., Huang Y., Ni Y., Small, 2023, 19(28), e2301850 |
| [59] | He Y., Yan F., Zhang X., Zhu C., Zhao Y., Geng B., Chou S., Xie Y., Chen Y., Adv. Energy Mater., 2023, 13(20), 2204177 |
| [60] | Li G., Li J., Liu X., Guo J., Ionics, 2023, 29(5), 1963—1973 |
| [61] | Zhu J., Cen Y., Ma H., Lian W., Liu J., Ou H., Ouyang F., Zhang L., Zhang W., Nanoscale Horiz., 2023, 8(9), 1273—1281 |
| [62] | Hanan A., Lakhan M. N., Bibi F., Khan A., Soomro I. A., Hussain A., Aftab U., Chem. Eng. J., 2024, 482, 148776 |
| [63] | Luo Y., Zhang Y., Zhu J., Tian X., Liu G., Feng Z., Pan L., Liu X., Han N., Tan R., Small Methods, 2024, 8(12), e2400158 |
| [64] | Liang Y., Jiang H., Lin H., Wang C., Yu K., Wang C., Lv J., Zhou B., Chem. Eng. J., 2023, 466, 143220 |
| [65] | Cheng C. C., Lin T. Y., Ting Y. C., Lin S. H., Choi Y., Lu S. Y., Nano Energy, 2023, 112, 108450 |
| [66] | Fan Y., Feng Z., Cao X., Peng Z., Chen J., Wang L., Small, 2025, e12470 |
| [67] | Miao L., Jia W., Cao X., Jiao L., Chem. Soc. Rev., 2024, 53(6), 2771—2807 |
| [1] | 张雨, 吴洋, 李佳琪, 徐超, 汪少振, 李欣雨, 王存民, 张生辉, 徐欢. MOF功能化自清洁聚乳酸多级孔纤维膜的辐射冷却性能[J]. 高等学校化学学报, 2026, 47(3): 20250239. |
| [2] | 葛佳, 王仁颖, 孙贤迪, 刘航, 郑亚荣. 双金属氧化物协同效应增强电催化丙烯氧化性能[J]. 高等学校化学学报, 2026, 47(2): 20250168. |
| [3] | 秦彩翼, 李娟, 李莹, 张九文, 龙含意, 李信宇, 米楠, 刘金炜, 李华. 原位构筑Co-MOFs/碳纤维复合电芬顿阴极及其高效降解四环素[J]. 高等学校化学学报, 2025, 46(8): 20250118. |
| [4] | 惠辰洋, 徐晨晖, 杨洋, 刘佳伦, 李亚太, 郭振国, 杨真真, 张根磊. P-NiCoMo-LDH电催化5-羟甲基糠醛氧化制备2,5-呋喃二甲酸的性能[J]. 高等学校化学学报, 2025, 46(8): 20250126. |
| [5] | 张小会, 赵冬冬, 张俊杰, 庄金亮. D-A型共价有机框架纳米棒用于可见光催化苄胺偶联反应[J]. 高等学校化学学报, 2025, 46(7): 20250020. |
| [6] | 李承育, 尚露, 张铁锐. 缺陷改性商业碳纳米管用于耐毒化二氧化碳电还原[J]. 高等学校化学学报, 2025, 46(5): 20250043. |
| [7] | 刘兆镇, 姚燕芳, 房坤, 吕昀叡, 叶勇, 刘海洋. 金属卟啉/多壁碳纳米管复合材料的电催化析氢反应[J]. 高等学校化学学报, 2025, 46(5): 20240529. |
| [8] | 卢净宇, 刘志萍, 盛夏, 封心建. 二氧化钛表面浸润性调控增强光催化氧化反应性能[J]. 高等学校化学学报, 2025, 46(5): 20240571. |
| [9] | 杨思伟, 黄旭日. B, N共掺杂富勒烯C70作为氧还原和氧析出非金属电催化剂的理论研究[J]. 高等学校化学学报, 2025, 46(4): 20240490. |
| [10] | 康莎, 章柯, 卫亚静, 王传义. N缺陷g-C3N5/CdS/Ti3C2异质结光催化剂的制备及高效去除NO性能[J]. 高等学校化学学报, 2025, 46(4): 20240488. |
| [11] | 郑娜, 聂丽君, 高宇航, 薛坤坤, 韩晓蓓, 马跃宇, 任丽蓉, 苏王潮, 石建惠. H3PO4质子化改性的g-C3N4及其光催化产H2O2性能[J]. 高等学校化学学报, 2025, 46(4): 20240485. |
| [12] | 张望, 鲁新环, 董妍红, 郭昊天, 严姗, 周丹, 夏清华. 梭形含钴MOFs材料协同催化双烯烃空气环氧化反应[J]. 高等学校化学学报, 2025, 46(4): 20240555. |
| [13] | 温俊青, 王嘉辉, 张建民. 掺杂和点缺陷调控MoS2/ZnO异质结光解水性能的第一性原理研究[J]. 高等学校化学学报, 2025, 46(2): 20240380. |
| [14] | 郭新, 刘淼. 改性碳纳米管-钛电极的制备及电催化氧化应用[J]. 高等学校化学学报, 2025, 46(12): 20250252. |
| [15] | 王德龙, 邢世禄, 李春丽, 周楠, 郝亚玲, 霍蓉, 李秀华, 刘惠锋. 纳米结构Co-Nd-MOF/GO电极材料的制备及电化学性能[J]. 高等学校化学学报, 2025, 46(11): 20250191. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||