

高等学校化学学报 ›› 2023, Vol. 44 ›› Issue (5): 20220710.doi: 10.7503/cjcu20220710
收稿日期:2022-11-09
出版日期:2023-05-10
发布日期:2023-01-15
通讯作者:
刘兆清
E-mail:lzqgzu@gzhu.edu.cn
基金资助:Received:2022-11-09
Online:2023-05-10
Published:2023-01-15
Contact:
LIU Zhaoqing
E-mail:lzqgzu@gzhu.edu.cn
Supported by:摘要:
羟甲基糠醛是呋喃类化合物, 具有价格低廉及来源广泛的优点, 可作为平台化合物用于制备其它高附加值产品. 传统的热催化氧化增值方法需要高温、 高压及贵金属催化剂, 造成经济效益的下降. 而电氧化方法不需要高温、 高压条件; 同时, 通过对电催化剂的合理设计, 非贵金属催化剂表面的羟甲基糠醛选择性转化已经得以实现, 从而避免使用大量贵金属. 因此, 通过电氧化方法对羟甲基糠醛平台化合物进行高附加值转化受到了广泛关注. 在羟甲基糠醛氧化的多种产物中, 羟基和醛基被全部氧化为羧基的产物——2,5-呋喃二甲酸, 被美国能源部列为“最具有价值的12种生物质衍生化学品”之一. 鉴于此, 本文介绍了羟甲基糠醛电氧化增值生产2,5-呋喃二甲酸的重要研究价值及相关非贵金属电催化剂的最新进展, 并对催化羟甲基糠醛电氧化反应的非贵金属催化剂的发展前景进行了展望.
中图分类号:
TrendMD:
杜磊, 刘兆清. 非贵金属催化剂在羟甲基糠醛电氧化增值中的应用. 高等学校化学学报, 2023, 44(5): 20220710.
DU Lei, LIU Zhaoqing. Non-precious Metal Catalysts for Electro-oxidation Upgrading of 5-Hydroxymethy Furfural. Chem. J. Chinese Universities, 2023, 44(5): 20220710.
| Value⁃added product | Application |
|---|---|
| DFF | Synthesis of antifungal drugs, organic conductors, heterocyclic ligands, etc. |
| HMFCA | Synthesis of polyester intermediates, anti⁃tumor drugs, interleukin inhibitors, etc. |
| FFCA | Synthesis of surfactants, resins, etc. |
| FDCA | Synthesis of monomer for PEF, succinic acid, etc. |
Table 1 The value-added products derived from HMFOR and their possible applications
| Value⁃added product | Application |
|---|---|
| DFF | Synthesis of antifungal drugs, organic conductors, heterocyclic ligands, etc. |
| HMFCA | Synthesis of polyester intermediates, anti⁃tumor drugs, interleukin inhibitors, etc. |
| FFCA | Synthesis of surfactants, resins, etc. |
| FDCA | Synthesis of monomer for PEF, succinic acid, etc. |
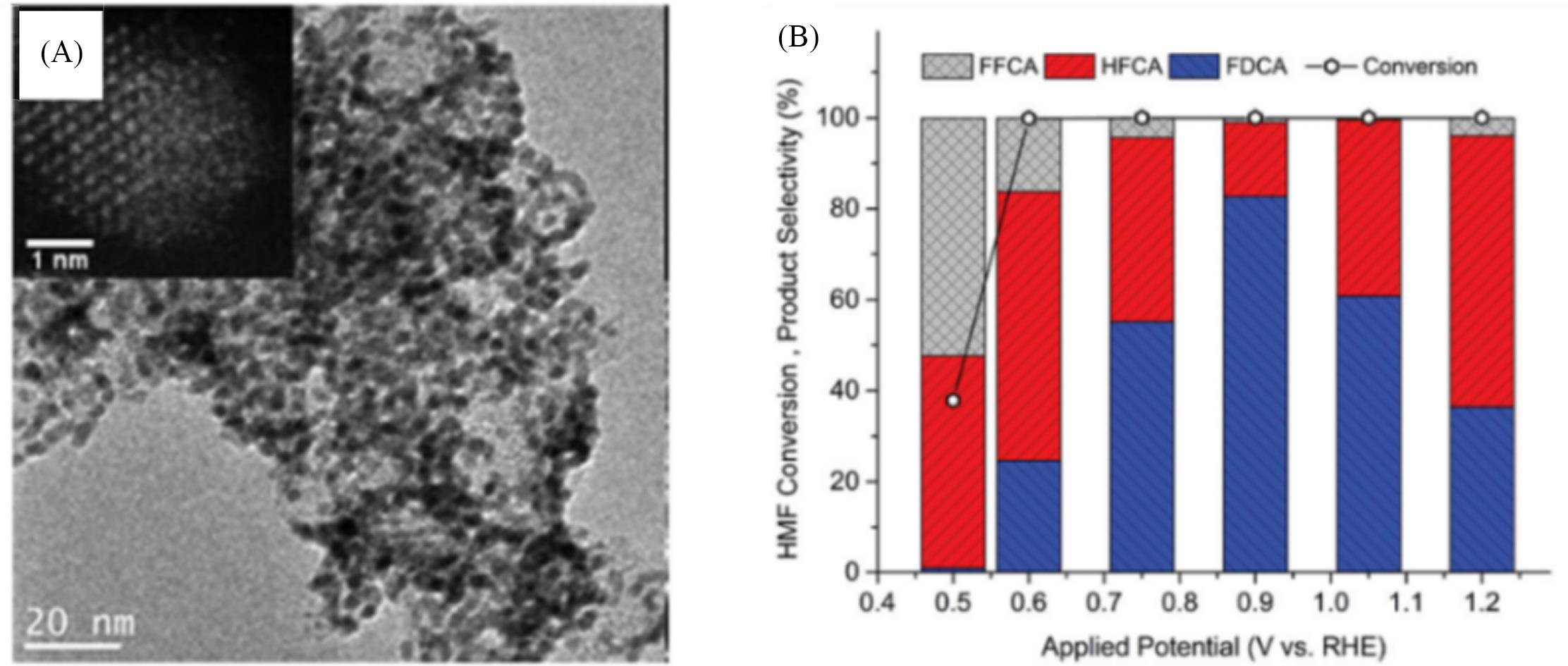
Fig.2 TEM image of Pd1Au2/C catalyst(A) and product distribution on Pd1Au2/C for the oxidation of 0.02 mol/L HMF in 0.1 mol/L KOH(B)[28]Copyright 2014, the Royal Society of Chemistry.
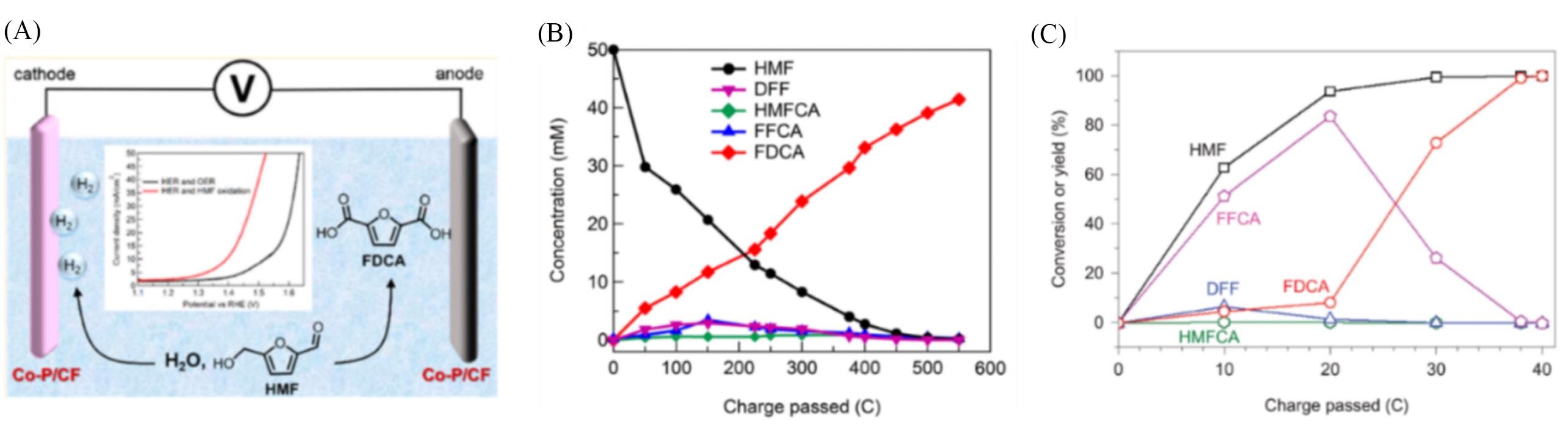
Fig.3 Schematic of the electrochemical cells with Co⁃P/CF catalyzed HMF oxidation(A), concentrations of HMF and oxidation products over passed charge during the chronoamperometry experiment conducted at 1.423 V in 1.0 mol/L KOH containing 50 mmol/L HMF(B)[47], conversion and yield(%) changes of HMF and its oxidation products during the electrochemical oxidation of HMF at 1.54 V in a 0.5 mol/L borate buffer solution containing 5 mmol/L HMF and 7.5 mmol/L TEMPO(C)[48](A, B) Copyright 2016, American Chemical Society; (C) Copyright 2015, Nature Publishing Group.
| Catalyst | c(HMF)/(mmol·L-1) | Potential/V(vs. RHE) | CD a /(mA·cm-2) | FC b (%) | FE c (%) | Ref. |
|---|---|---|---|---|---|---|
| Ni3S2 | 10 | 1.423 | >200 | 95 | 98 | [ |
| Ni2P NPA | 10 | 1.423 | >200 | 100 | 100 | [ |
| Co⁃P | 50 | 1.423 | 50 | 90 | 90 | [ |
| Ni3N@C | 10 | 1.38 | 50 | 98 | 99 | [ |
| NixB | 10 | 1.45 | 100 | 98.5 | 100 | [ |
| CoB | 10 | 1.39 | 10 | 94 | 98 | [ |
| NiO/Ni(OH)2 | 5 | 71 | 84 | [ | ||
| CoO⁃CoSe2 | 10 | 1.3 | 1 | 99 | 97.9 | [ |
| ZnCo2O4 | 50 | 1.23 | 1 | 93.7 | 94 | [ |
| NiFe⁃LDHs | 10 | 1.32 | 20 | 99.4 | 99.4 | [ |
| NiCoFe⁃LDHs | 5 | 1.51 | 20 | 95.5 | 84.9 | [ |
| NiOOH | 5 | 96 | 96 | [ | ||
| CoOOH | 5 | 35.1 | 35.1 | [ | ||
| FeOOH | 5 | 1.59 | 1.59 | [ |
Table 2 Typical non-precious metal catalysts and their HMFOR performances
| Catalyst | c(HMF)/(mmol·L-1) | Potential/V(vs. RHE) | CD a /(mA·cm-2) | FC b (%) | FE c (%) | Ref. |
|---|---|---|---|---|---|---|
| Ni3S2 | 10 | 1.423 | >200 | 95 | 98 | [ |
| Ni2P NPA | 10 | 1.423 | >200 | 100 | 100 | [ |
| Co⁃P | 50 | 1.423 | 50 | 90 | 90 | [ |
| Ni3N@C | 10 | 1.38 | 50 | 98 | 99 | [ |
| NixB | 10 | 1.45 | 100 | 98.5 | 100 | [ |
| CoB | 10 | 1.39 | 10 | 94 | 98 | [ |
| NiO/Ni(OH)2 | 5 | 71 | 84 | [ | ||
| CoO⁃CoSe2 | 10 | 1.3 | 1 | 99 | 97.9 | [ |
| ZnCo2O4 | 50 | 1.23 | 1 | 93.7 | 94 | [ |
| NiFe⁃LDHs | 10 | 1.32 | 20 | 99.4 | 99.4 | [ |
| NiCoFe⁃LDHs | 5 | 1.51 | 20 | 95.5 | 84.9 | [ |
| NiOOH | 5 | 96 | 96 | [ | ||
| CoOOH | 5 | 35.1 | 35.1 | [ | ||
| FeOOH | 5 | 1.59 | 1.59 | [ |
Table 3 The oretical ΔHfFERE and experimental ΔHfexp of several typical transition metal-based compounds[60]
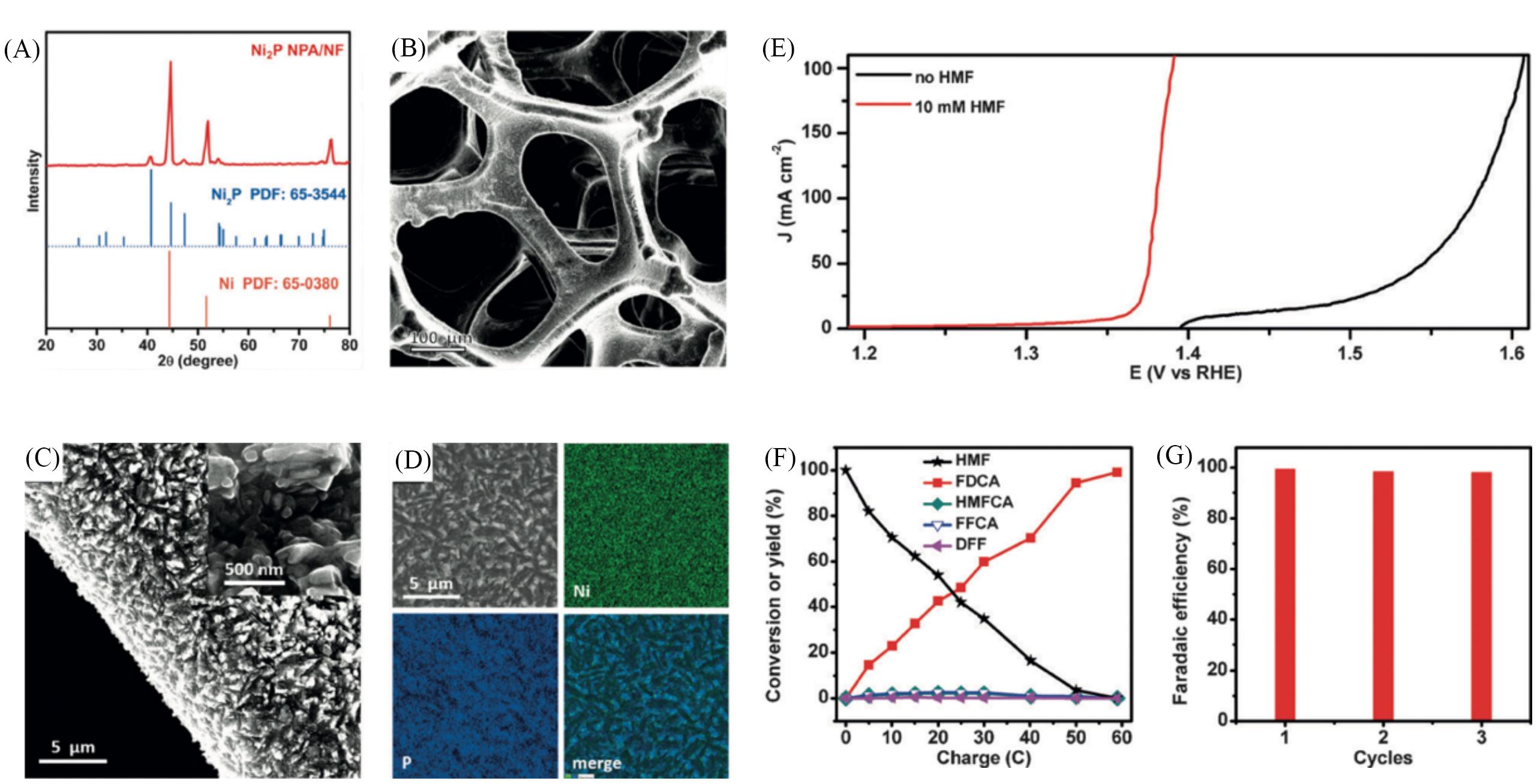
Fig.4 Morphological features of Ni2P NPA/NF and conversation of FDCA[50](A) XRD pattern of Ni2P NPA/NF with the corresponding standard patterns of Ni2P and Ni; (B, C) SEM images of Ni2P NPA/NF with different magnifications; (D) SEM and the corresponding elemental mapping images of Ni2P NPA/NF; (E) LSV curves of Ni2P NPA/NF at a scan rate of 2 mV/s in 1.0 mol/L KOH with and without 10 mmol/L HMF; (F) conversion and yield(%) changes of HMF and its oxidation products during the electrochemical oxidation of HMF at 1.423 V(vs. RHE) in 1.0 mol/L KOH with 10 mmol/L HMF; (G) faradaic efficiencies of Ni2P NPA/NF for FDCA production under three successive electrolysis cycles.Copyright 2016, Wiley-VCH GmbH.
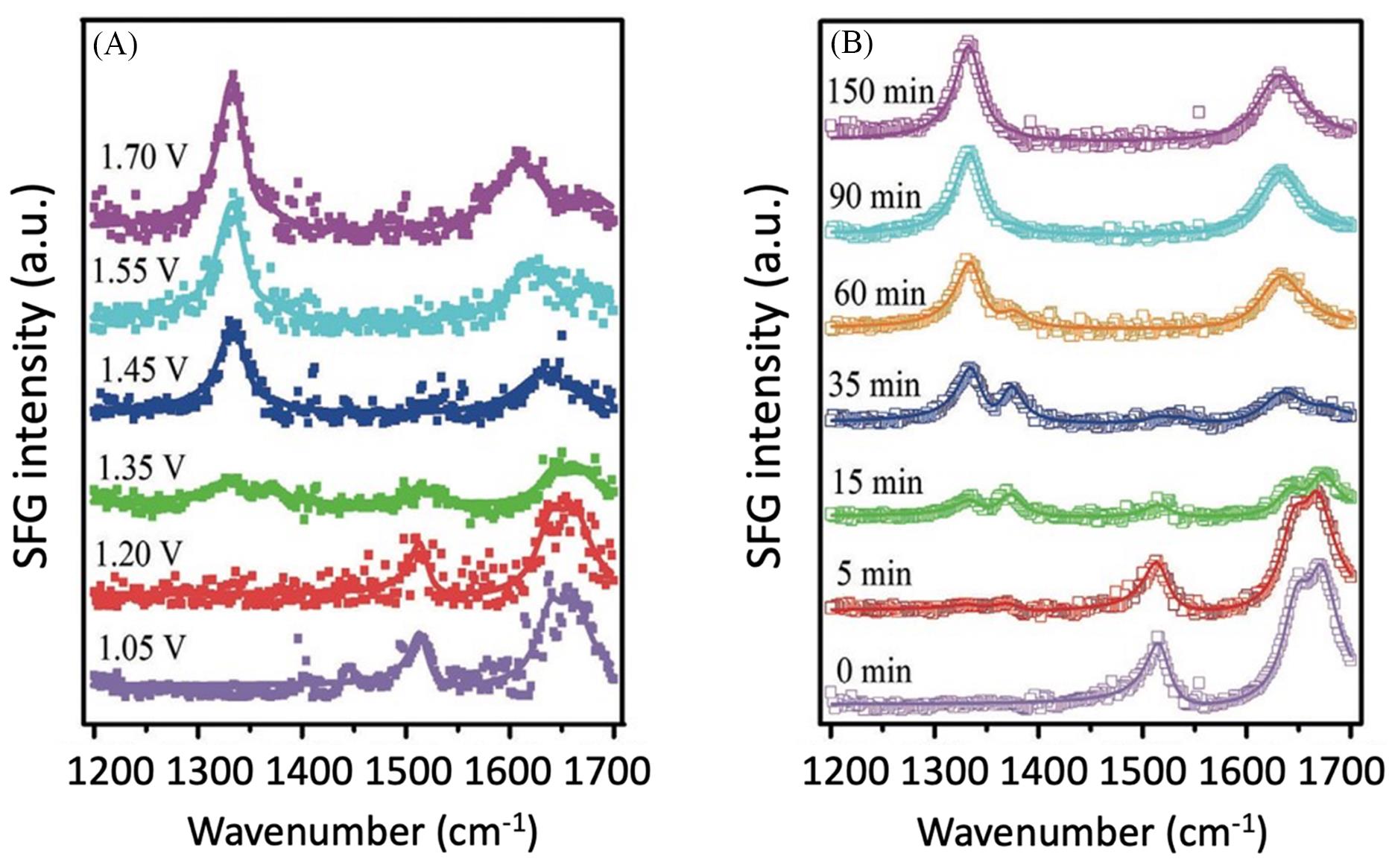
Fig.5 Sum Frequency Generation(SFG) spectra recorded at the working electrode/electrolyte interface after running the cell at various voltages for 90 min(A) and by monitoring the reaction at different times at 1.45 V(vs. RHE) in 1 mol/L KOH and 20 mmol/L HMF(B)[51]Copyright 2019, Wiley-VCH GmbH.
| Category | Initial material | Real active material | Electrolyte | Ref. |
|---|---|---|---|---|
| Sulfide | Cu2S/CM | CuO/CM | 1.0 mol/L KOH | [ |
| NiS/Ni | NiOOH | 1.0 mol/L KOH | [ | |
| CoS x | CoOOH | 1.0 mol/L KOH | [ | |
| Phosphide | Ni5P4 | NiOOH | 1.0 mol/L KOH | [ |
| Ni2P | NiO x | 1.0 mol/L KOH | [ | |
| NiFeP | NiFeOOH | 1.0 mol/L NaOH | [ | |
| Nitride | Ni3N | NiO | 1.0 mol/L KOH | [ |
| CoN | Co3O4 | 1.0 mol/L KOH | [ | |
| Co4N | Co3O4 | 1.0 mol/L KOH | [ | |
| Carbide | Co3C | CoO x | 1.0 mol/L NaOH | [ |
| β⁃Mo2C | MoO2 | 1.0 mol/L KOH | [ | |
| Ni3C/C | NiO x | 1.0 mol/L KOH | [ |
Table 4 Typical transition metal Xides and their derivates(real active materials) during anodic reactions
| Category | Initial material | Real active material | Electrolyte | Ref. |
|---|---|---|---|---|
| Sulfide | Cu2S/CM | CuO/CM | 1.0 mol/L KOH | [ |
| NiS/Ni | NiOOH | 1.0 mol/L KOH | [ | |
| CoS x | CoOOH | 1.0 mol/L KOH | [ | |
| Phosphide | Ni5P4 | NiOOH | 1.0 mol/L KOH | [ |
| Ni2P | NiO x | 1.0 mol/L KOH | [ | |
| NiFeP | NiFeOOH | 1.0 mol/L NaOH | [ | |
| Nitride | Ni3N | NiO | 1.0 mol/L KOH | [ |
| CoN | Co3O4 | 1.0 mol/L KOH | [ | |
| Co4N | Co3O4 | 1.0 mol/L KOH | [ | |
| Carbide | Co3C | CoO x | 1.0 mol/L NaOH | [ |
| β⁃Mo2C | MoO2 | 1.0 mol/L KOH | [ | |
| Ni3C/C | NiO x | 1.0 mol/L KOH | [ |
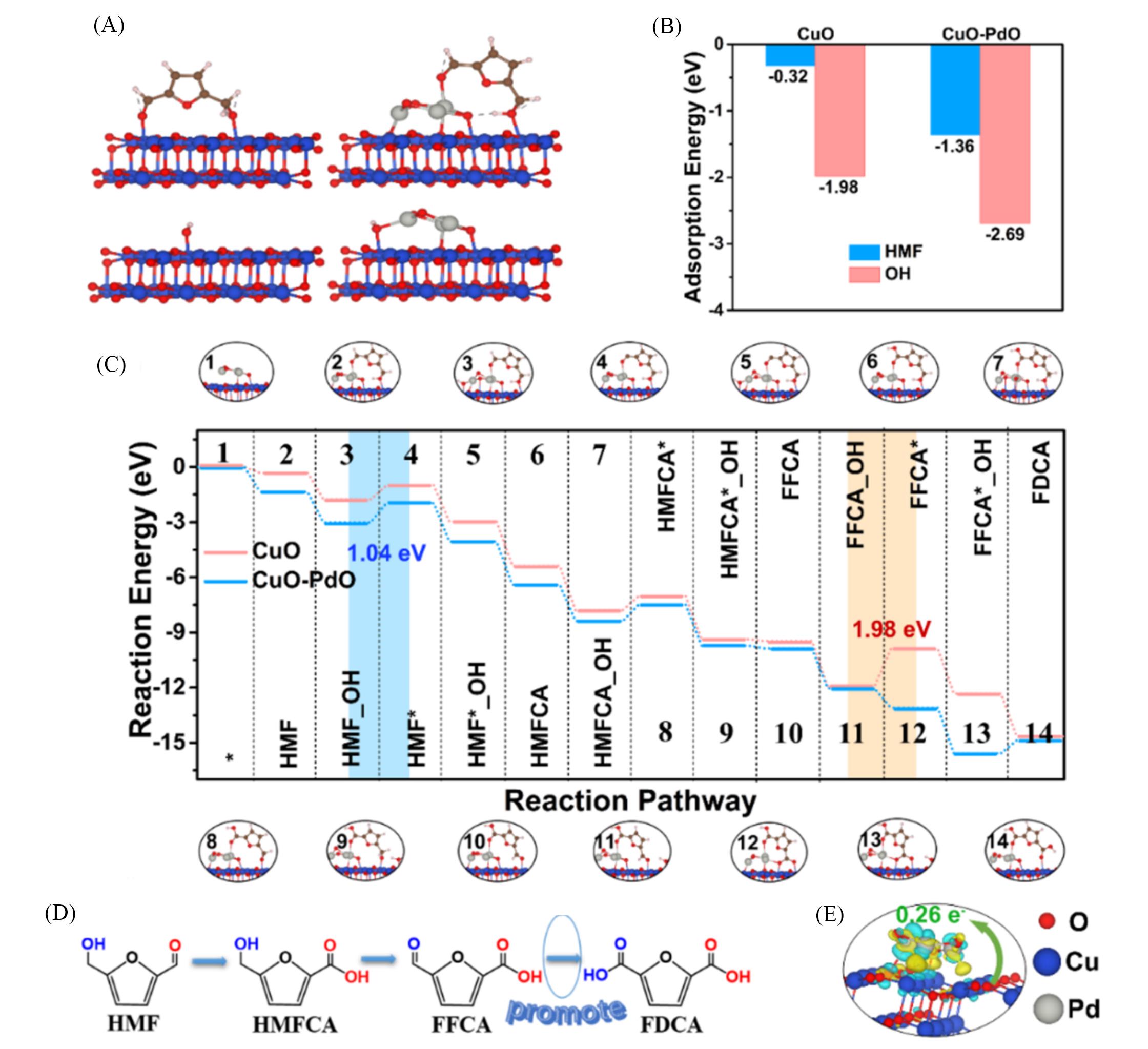
Fig.7 Theoretical calculation results to unravel the underlying HMFOR mechanisms on CuO⁃PdO catalyst[83](A) Adsorption configuration of the HMF and OH on CuO(111) surface and the as-designed CuO-PdO; (B) HMF and OH adsorption free energy; (C) free energy diagram and adsorption configurations of HMFOR intermediates on CuO(111) and CuO⁃PdO; (D) promoting mechanism of the HMFOR; (E) the charge density difference of PdO on CuO(111) surface.Copyright 2022, Wiley-VCH GmbH.
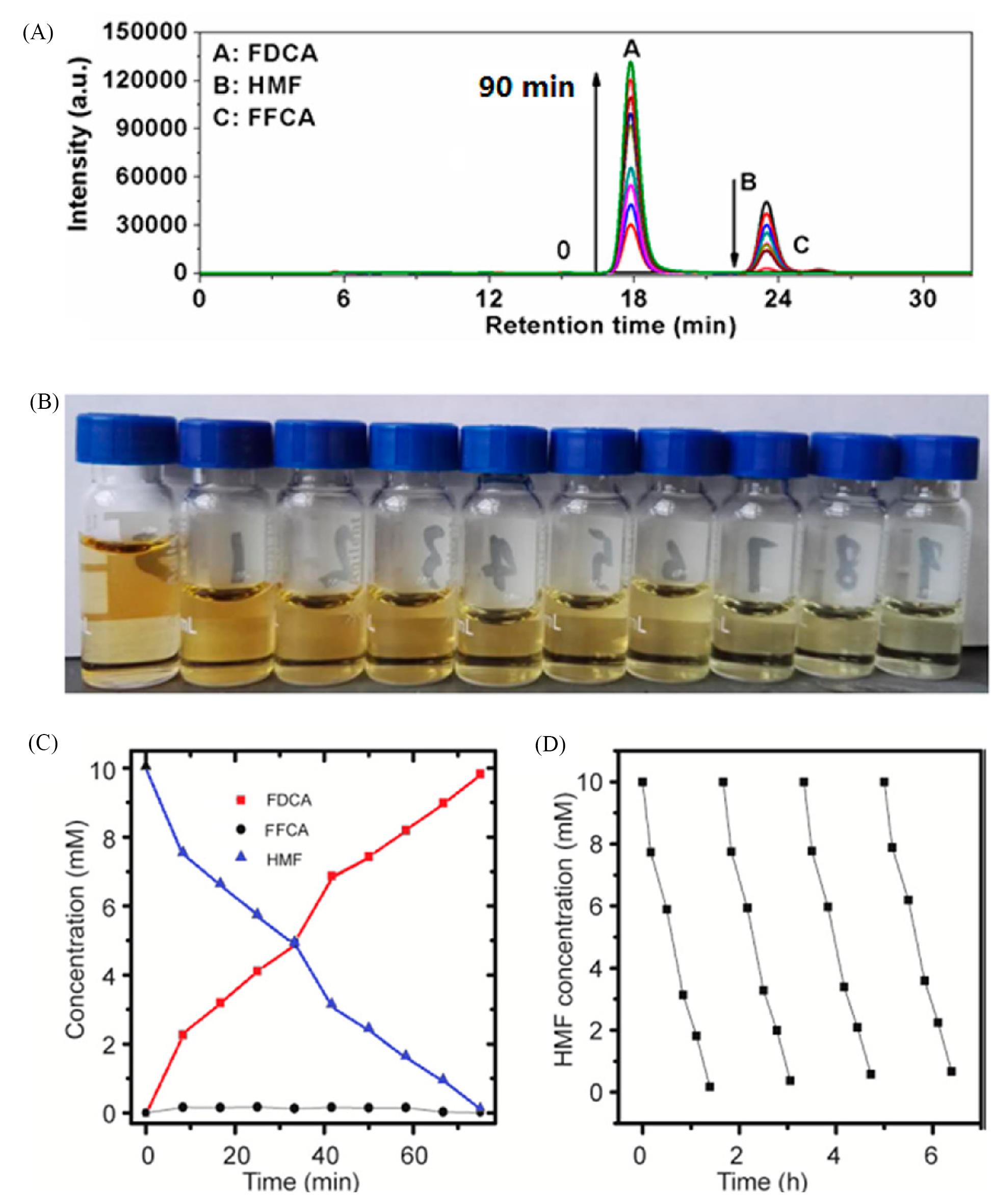
Fig.8 Electrochemical HMFOR performance using NiFe⁃LDHs catalyst[57](A) HPLC chromatogram traces of the various products at different reaction times following an HMF electrochemical oxidation reaction; (B) digital photograph showing the color change of the anodic electrolyte during the electrochemical HMF oxidation process; (C) concentration changes of HMF and its oxidation products with the time of chronoamperometric tests at 1.33 V vs. RHE; (D) HMF concentration changes during four successive cycles.Copyright 2018, American Chemical Society.
| 1 | Seh Z. W., Kibsgaard J., Dickens C. F., Chorkendorff I., Nørskov J. K., Jaramillo T. F., Science, 2017, 355, eaad4998 |
| 2 | Zeng L., Sun K., Chen Y., Liu Z., Chen Y., Pan Y., Zhao R., Liu Y., Liu C., J. Mater. Chem. A, 2019, 7, 16793—16802 |
| 3 | Hu C., Zhang L., Gong J., Energy Environ. Sci., 2019, 12, 2620—2645 |
| 4 | Walter M. G., Warren E. L., McKone J. R., Boettcher S. W., Mi Q., Santori E. A., Lewis N. S., Chem. Rev., 2010, 110, 6446—6473 |
| 5 | Guo X., Du H., Qu F., Li J., J. Mater. Chem. A, 2019, 7, 3531—3543 |
| 6 | Du L., Xing L., Zhang G., Liu X., Rawach D., Sun S., Sus. Mat., 2021, 1, 150—173 |
| 7 | Kyriakou V., Garagounis I., Vasileiou E., Vourros A., Stoukides M., Catal. Today, 2017, 286, 2—13 |
| 8 | Yang H., Lin Q., Zhang C., Yu X., Cheng Z., Li G., Hu Q., Ren X., Zhang Q., Liu J., He C., Nat. Commun., 2020, 11, 593 |
| 9 | Handoko A. D., Wei F., Jenndy Yeo B. S., Seh Z. W., Nat. Catal., 2018, 1, 922—934 |
| 10 | Sumida K., Rogow D. L., Mason J. A., McDonald T. M., Bloch E. D., Herm Z. R., Bae T. H., Long J. R., Chem. Rev., 2012, 112, 724—781 |
| 11 | Sun Y., Han G., Du L., Du C., Zhou X., Sun Q., Gao Y., Yin G., Li Y., Wang Y., Chem. Catal., 2021, 1, 1260—1272 |
| 12 | Simoes M., Baranton S., Coutanceau C., ChemSusChem, 2012, 5, 2106—2124 |
| 13 | Liu Y., Yu W., Raciti D., Gracias D. H., Wang C., J. Phys. Chem. C, 2018, 123, 426—432 |
| 14 | Lari G. M., Pastore G., Haus M., Ding Y., Papadokonstantakis S., Mondelli C., Pérez⁃Ramírez J., Energy Environ. Sci., 2018, 11, 1012—1029 |
| 15 | Dodekatos G., Schünemann S., Tüysüz H., ACS Catal., 2018, 8, 6301—6333 |
| 16 | Wang W., Jing W., Sheng L., Chai D., Kang Y., Lei Z., Appl. Catal. A, 2017, 538, 123—130 |
| 17 | Zanata C. R., Fernández P. S., Troiani H. E., Soldati A. L., Landers R., Camara G. A., Carvalho A. E., Martins C. A., Appl. Catal. B: Environ., 2016, 181, 445—455 |
| 18 | Luo R., Li Y., Xing L., Wang N., Zhong R., Qian Z., Du C., Yin G., Wang Y., Du L., Appl. Catal. B: Environ., 2022, 311, 121357 |
| 19 | Kwon Y., Schouten K. J. P., van der Waal J. C., de Jong E., Koper M. T. M., ACS Catal., 2016, 6, 6704—6717 |
| 20 | Zhong R., Wang Q., Du L., Pu Y., Ye S., Gu M., Conrad Zhang Z., Huang L., Appl. Surf. Sci., 2022, 584 |
| 21 | Chen C., Wang L., Zhu B., Zhou Z., El⁃Hout S. I., Yang J., Zhang J., J. Energy Chem., 2021, 54, 528—554 |
| 22 | Gao L., Liu Z., Ma J., Zhong L., Song Z., Xu J., Gan S., Han D., Niu L., Appl. Catal. B: Environ., 2020, 261 |
| 23 | Luo R., Li Y., Wang N., Zhong R., Xing L., Zhu L., Wang Y., Du L., Ye S., J. Phys. Chem. C, 2023, doi: 10.1021/acs.jpcc.2c08946 |
| 24 | Bozell J. J., Petersen G. R., Green Chem., 2010, 12, 539 |
| 25 | Corma A., Iborra S., Velty A., Chem. Rev., 2007, 107, 2411—2502 |
| 26 | Wang T., Ide M. S., Nolan M. R., Davis R. J., Shanks B. H., Energy Environ. Focus, 2016, 5, 13—17 |
| 27 | Nie J., Xie J., Liu H., J. Catal., 2013, 301, 83—91 |
| 28 | Chadderdon D. J., Xin L., Qi J., Qiu Y., Krishna P., More K. L., Li W., Green Chem., 2014, 16, 3778—3786 |
| 29 | Verevkin S. P., Emel'yanenko V. N., Stepurko E. N., Ralys R. V., Zaitsau D. H., Stark A., Ind. Eng. Chem. Res., 2009, 48, 10087—10093 |
| 30 | Hu L., He A. Y., Liu X. Y., Xia J., Xu J. X., Zhou S. Y., Xu J. M., ACS Sustainable Chem. Eng., 2018, 6, 15915—15935 |
| 31 | Tong X. L., Ma Y., Li Y. D., Appl. Catal. A, 2010, 385, 1—13 |
| 32 | Lu Y. X., Dong C. L., Huang Y. C., Zou Y. Q., Liu Y. B., Li Y. Y., Zhang N. N., Chen W., Zhou L., Lin H. Z., Wang S. Y., Sci. China: Chem., 2020, 63, 980—986 |
| 33 | Han X. W., Li C. Q., Guo Y., Liu X. H., Zhang Y. G., Wang Y. Q., Appl. Catal. A, 2016, 526, 1—8 |
| 34 | Gorbanev Y. Y., Kegaens S., Riisager A., Top. Catal., 2011, 54, 1318—1324 |
| 35 | Tang C., Zheng Y., Jaroniec M., Qiao S. Z., Angew. Chem., Int. Ed., 2021, 60, 19572—19590 |
| 36 | Du L., Shao Y. Y., Sun J. M., Yin G. P., Du C. Y., Wang Y., Catal. Sci. Technol., 2018, 8, 3216—3232 |
| 37 | Li R., Xiang K., Peng Z., Zou Y., Wang S., Adv. Energy Mater., 2021, 11, 2102292 |
| 38 | Horn E. J., Rosen B. R., Chen Y., Tang J., Chen K., Eastgate M. D., Baran P. S., Nature, 2016, 533, 77—81 |
| 39 | Wang T., Tao L., Zhu X., Chen C., Chen W., Du S., Zhou Y., Zhou B., Wang D., Xie C., Long P., Li W., Wang Y., Chen R., Zou Y., Fu X. Z., Li Y., Duan X., Wang S., Nat. Catal., 2021, doi: 10.1038/s41929⁃021⁃00721⁃y |
| 40 | Bellini M., Bevilacqua M., Innocenti M., Lavacchi A., Miller H. A., Filippi J., Marchionni A., Oberhauser W., Wang L., Vizza F., J. Electrochem. Soc., 2014, 161, D3032—D3043 |
| 41 | Choi S., Balamurugan M., Lee K. G., Cho K. H., Park S., Seo H., Nam K. T., J. Phys. Chem. Lett., 2020, 11, 2941—2948 |
| 42 | Zhang X., Cheng X., Zhang Q., J. Energy Chem., 2016, 25, 967—984 |
| 43 | McCrory C. C., Jung S., Ferrer I. M., Chatman S. M., Peters J. C., Jaramillo T. F., J. Am. Chem. Soc., 2015, 137, 4347—4357 |
| 44 | Du L., Luo L., Feng Z., Engelhard M., Xie X., Han B., Sun J., Zhang J., Yin G., Wang C., Wang Y., Shao Y., Nano Energy, 2017, 39, 245—252 |
| 45 | Lin R., Salehi M., Guo J., Seifitokaldani A., iScience, 2022, 25, 104744 |
| 46 | Sun Y., Wang J., Qi Y., Li W., Wang C., Adv. Sci., 2022, 9, e2200957 |
| 47 | Jiang N., You B., Boonstra R., Rodriguez I. M. T., Sun Y. J., ACS Energy Lett., 2016, 1, 386—390 |
| 48 | Cha H. G., Choi K. S., Nat. Chem., 2015, 7, 328—333 |
| 49 | You B., Liu X., Jiang N., Sun Y., J. Am. Chem. Soc., 2016, 138, 13639—13646 |
| 50 | You B., Jiang N., Liu X., Sun Y., Angew Chem. Int. Ed., 2016, 55, 9913—9917 |
| 51 | Zhang N., Zou Y., Tao L., Chen W., Zhou L., Liu Z., Zhou B., Huang G., Lin H., Wang S., Angew. Chem. Int. Ed., 2019, 58, 15895—15903 |
| 52 | Barwe S., Weidner J., Cychy S., Morales D. M., Dieckhofer S., Hiltrop D., Masa J., Muhler M., Schuhmann W., Angew. Chem., Int. Ed., 2018, 57, 11460—11464 |
| 53 | Weidner J., Barwe S., Sliozberg K., Piontek S., Masa J., Apfel U. P., Schuhmann W., Beilstein J. Org. Chem., 2018, 14, 1436—1445 |
| 54 | Grabowski G., Lewkowski J., Skowronski R., Electrochim. Acta, 1991, 36, 1995 |
| 55 | Huang X., Song J. L., Hua M. L., Xie Z. B., Liu S. S., Wu T. B., Yang G. Y., Han B. X., Green Chem., 2020, 22, 843—849 |
| 56 | Lu Y., Dong C. L., Huang Y. C., Zou Y., Liu Z., Liu Y., Li Y., He N., Shi J., Wang S., Angew. Chem. Int. Ed., 2020, 59, 19215—19221 |
| 57 | Liu W. J., Dang L. N., Xu Z. R., Yu H. Q., Jin S., Huber G. W., ACS Catal., 2018, 8, 5533—5541 |
| 58 | Zhang M., Liu Y. Q., Liu B. Y., Chen Z., Xu H., Yan K., ACS Catal., 2020, 10, 5179—5189 |
| 59 | Taitt B. J., Nam D. H., Choi K. S., ACS Catal., 2018, 9, 660—670 |
| 60 | Stevanović V., Lany S., Zhang X., Zunger A., Phy. Rev. B, 2012, 85 |
| 61 | Zhou B., Dong C. L., Huang Y. C., Zhang N., Wu Y., Lu Y., Yue X., Xiao Z., Zou Y., Wang S., J. Energy Chem., 2021, 61, 179—185 |
| 62 | Wang M., Fu W., Du L., Wei Y., Rao P., Wei L., Zhao X., Wang Y., Sun S., Appl. Surf. Sci., 2020, 515, 146059 |
| 63 | Yang C. C., Zai S. F., Zhou Y. T., Du L., Jiang Q., Adv. Funct. Mater., 2019, 29, 1901949 |
| 64 | Luo R., Qian Z., Xing L., Du C., Yin G., Zhao S., Du L., Adv. Funct. Mater., 2021, 31, 2102918 |
| 65 | Ding X., Li W., Kuang H., Qu M., Cui M., Zhao C., Qi D. C., Oropeza F. E., Zhang K. H. L., Nanoscale, 2019, 11, 23217—23225 |
| 66 | Liu P. F., Li X., Yang S., Zu M. Y., Liu P. R., Zhang B., Zheng L. R., Zhao H. J., Yang H. G., ACS Energy Lett., 2017, 2, 2257—2263 |
| 67 | Zu M. Y., Wang C. W., Zhang L., Zheng L. R., Yang H. G., Mater. Horiz., 2019, 6, 115—121 |
| 68 | Ledendecker M., Krick Calderon S., Papp C., Steinruck H. P., Antonietti M., Shalom M., Angew. Chem. Int. Ed., 2015, 54, 12361—12365 |
| 69 | Zuo Y., Liu Y. P., Li J. S., Du R. F., Han X., Zhang T., Arbiol J., Divins N. J., Llorca J., Guijarro N., Sivula K., Cabot A., Chem. Mater., 2019, 31, 7732—7743 |
| 70 | Yan C. Y., Huang J. W., Wu C. Y., Li Y. Y., Tan Y. C., Zhang L. Y., Sun Y. H., Huang X. N., Xiong J., J. Mater. Sci. Technol., 2020, 42, 10—16 |
| 71 | Fan K., Zou H., Lu Y., Chen H., Li F., Liu J., Sun L., Tong L., Toney M. F., Sui M., Yu J., ACS Nano, 2018, 12, 12369—12379 |
| 72 | Stern L. A., Feng L. G., Song F., Hu X. L., Energy Environ. Sci., 2015, 8, 2347—2351 |
| 73 | Hu F., Zhu S., Chen S., Li Y., Ma L., Wu T., Zhang Y., Wang C., Liu C., Yang X., Song L., Yang X., Xiong Y., Adv. Mater., 2017, 29, 1606570 |
| 74 | Xu K., Chen P., Li X., Tong Y., Ding H., Wu X., Chu W., Peng Z., Wu C., Xie Y., J. Am. Chem. Soc., 2015, 137, 4119—4125 |
| 75 | Zhang Y., Ouyang B., Xu J., Jia G., Chen S., Rawat R. S., Fan H. J., Angew. Chem. Int. Ed., 2016, 55, 8670—8674 |
| 76 | Chen P., Xu K., Fang Z., Tong Y., Wu J., Lu X., Peng X., Ding H., Wu C., Xie Y., Angew. Chem. Int. Ed., 2015, 54, 14710—14714 |
| 77 | Kim J. H., Kawashima K., Wygant B. R., Mabayoje O., Liu Y., Wang J. H., Mullins C. B., ACS Appl. Energy Mater., 2018, 1, 5145—5150 |
| 78 | Xing J. N., Li Y., Guo S. W., Jin T., Li H. X., Wang Y. J., Jiao L. F., Electrochim. Acta, 2019, 298, 305—312 |
| 79 | Xu K., Ding H., Lv H., Chen P., Lu X., Cheng H., Zhou T., Liu S., Wu X., Wu C., Xie Y., Adv. Mater., 2016, 28, 3326—3332 |
| 80 | Xu C., Paone E., Rodriguez⁃Padron D., Luque R., Mauriello F., Chem. Soc. Rev., 2020, 49, 4273—4306 |
| 81 | Ma X., Zhang X. Y., Yang M., Xie J. Y., Lv R. Q., Chai Y. M., Dong B., Rare Metals, 2021, 40, 1048—1055 |
| 82 | Roylance J. J., Kim T. W., Choi K. S., ACS Catal., 2016, 6, 1840—1847 |
| 83 | Zhou P., Lv X., Tao S., Wu J., Wang H., Wei X., Wang T., Zhou B., Lu Y., Frauenheim T., Fu X., Wang S., Zou Y., Adv. Mater., 2022, 34, e2204089 |
| 84 | Song H., Yu J., Tang Z., Yang B., Lu S., Adv. Energy Mater., 2022, 12, 2102573 |
| 85 | Song H., Wu M., Tang Z., Tse J. S., Yang B., Lu S., Angew. Chem. Int. Ed., 2021, 60, 7234—7244 |
| 86 | Zhang L., Liang J., Yue L., Dong K., Li J., Zhao D., Li Z., Sun S., Luo Y., Liu Q., Cui G., Ali Alshehri A., Guo X., Sun X., Nano Research Energy, 2022, 1, e9120028 |
| 87 | Shi X. F., Zhu J., Bai T. Y., Fu Z. X., Zhang J. J., Bu X. H., Chem. J. Chinese Universities, 2022, 43(1), 20210613 |
| 史潇凡, 朱剑, 白田宇, 付子萱, 张冀杰, 卜显和. 高等学校化学学报, 2022, 43(1), 20210613 | |
| 88 | Zou X., Zhang Y., Chem. Soc. Rev., 2015, 44, 5148—5180 |
| 89 | Suen N. T., Hung S. F., Quan Q., Zhang N., Xu Y J., Chen H. M., Chem. Soc. Rev., 2017, 46, 337—365 |
| 90 | Wygant B. R., Kawashima K., Mullins C. B., ACS Energy Lett., 2018, 3, 2956—2966 |
| [1] | 徐佳宁, 白文静, 楼雨寒, 于海鹏, 窦烁. 电催化氧化木质素解聚: 温和高效的生物质增值策略[J]. 高等学校化学学报, 2023, 44(5): 304. |
| [2] | 李姿若, 张红娟, 朱国勋, 夏伟, 汤静. 负载酞菁铁的氮掺杂中空碳球的电催化氧还原性能[J]. 高等学校化学学报, 2023, 44(1): 20220677. |
| [3] | 程前, 杨博龙, 吴文依, 向中华. S掺杂Fe-N-C高活性氧还原反应催化剂[J]. 高等学校化学学报, 2022, 43(9): 20220341. |
| [4] | 李晨晨, 那永. 双功能复合材料g-C3N4/CdS/Ni催化光解水产氢和5-羟甲基糠醛氧化性能[J]. 高等学校化学学报, 2021, 42(9): 2896. |
| [5] | 王跃民, 孟庆磊, 王显, 葛君杰, 刘长鹏, 邢巍. 铜,硫掺杂对铁氮碳氧还原催化剂性能的提升作用[J]. 高等学校化学学报, 2020, 41(8): 1843. |
| [6] | 孔令照,苗改,罗虎,孙予罕. 生物质水热催化制备重要含氧化学品研究进展[J]. 高等学校化学学报, 2020, 41(1): 11. |
| [7] | 殷雯婧, 刘啸, 钱汇东, 邹志青. 高活性位点密度Fe-N共掺杂碳纳米片的制备及氧还原性能[J]. 高等学校化学学报, 2019, 40(7): 1480. |
| [8] | 于彦存, 王显, 葛君杰, 刘长鹏, 邢巍. 超支化聚合物氮修饰Pd催化剂促进甲酸电催化氧化[J]. 高等学校化学学报, 2019, 40(7): 1433. |
| [9] | 杜孟孟, 孙海军, 高山, 叶小利, 乔磊, 郭芬. 自支撑镍纳米线阵列电极的制备及电催化性能[J]. 高等学校化学学报, 2018, 39(12): 2739. |
| [10] | 范仲全, 陈晨, 沈振陆, 李美超. 以六甲基二硅胺烷为氮源一锅法电氧化醇制腈[J]. 高等学校化学学报, 2018, 39(1): 78. |
| [11] | 康欢, 李赏, 刘畅, 郭伟, 潘牧. 自组装合成Fe-N-C-PANI有序介孔结构催化剂及其在酸性条件下的氧还原活性[J]. 高等学校化学学报, 2017, 38(8): 1423. |
| [12] | 闫鹏, 张栋铭, 程魁, 徐阳, 李莹莹, 叶克, 曹殿学, 王贵领. Pd-Ag/C@TiO2核壳结构纳米棒阵列的制备及对NaBH4电氧化的催化性能[J]. 高等学校化学学报, 2015, 36(9): 1801. |
| [13] | 吴锋, 谢潇怡, 张存中, 穆道斌, 吴伯荣. I-的光电化学氧化与光助充电二次锂离子电池[J]. 高等学校化学学报, 2013, 34(4): 980. |
| [14] | 刘瑶, 曾亚超, 刘冉, 王贵领, 曹殿学. 丙酮对Pd电极上异丙醇电氧化的毒化作用[J]. 高等学校化学学报, 2012, 33(05): 1017. |
| [15] | 段东红, 张忠林, 张涛, 刘世斌, 郝晓刚, 李一兵. 碱性介质中W, Ni和Sn掺杂的Pt-Ru/C电催化氧化甲醇的性能[J]. 高等学校化学学报, 2011, 32(11): 2618. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||
