

高等学校化学学报 ›› 2020, Vol. 41 ›› Issue (4): 604.doi: 10.7503/cjcu20190641
• 庆祝《高等学校化学学报》复刊40周年专栏 • 上一篇 下一篇
收稿日期:2019-12-09
出版日期:2020-04-10
发布日期:2020-02-24
通讯作者:
张铁锐
E-mail:tierui@mail.ipc.ac.cn
基金资助:
LI Zhenhua1,2,SHI Run1,ZHAO Jiaqi1,ZHANG Tierui1,2,*( )
)
Received:2019-12-09
Online:2020-04-10
Published:2020-02-24
Contact:
Tierui ZHANG
E-mail:tierui@mail.ipc.ac.cn
Supported by:摘要:
阐述了光驱动C1化学的最新研究进展, 分别对光驱动费托合成、 水煤气变换、 二氧化碳加氢、 甲烷重整和甲醇重整制氢的研究进行了综述, 提出了当前研究存在的问题及发展方向.
中图分类号:
TrendMD:
李振华, 施润, 赵家琦, 张铁锐. 光驱动C1转换到高附加值化学品的研究进展. 高等学校化学学报, 2020, 41(4): 604.
LI Zhenhua, SHI Run, ZHAO Jiaqi, ZHANG Tierui. Research Progress of Photo-driven C1 Conversion to Value-added Chemicals †. Chem. J. Chinese Universities, 2020, 41(4): 604.
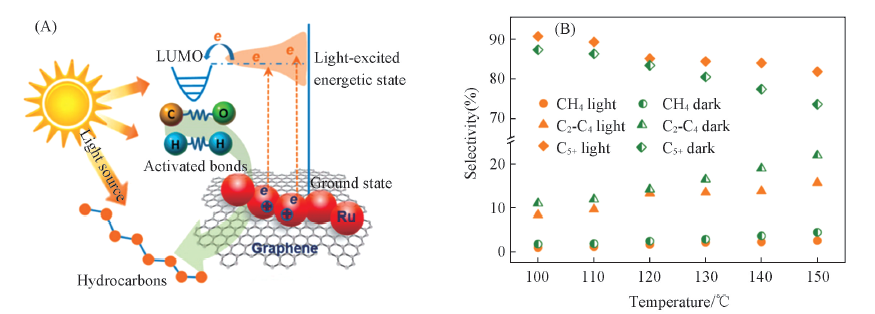
Fig.1 Schematic showing the reaction process FTS over Ru/graphene under light irradiation(A) and selectivities of photocatalytic FTS hydrocarbons with or without irradiation at different temperatures(B)[46]Copyright 2015, American Chemical Society.
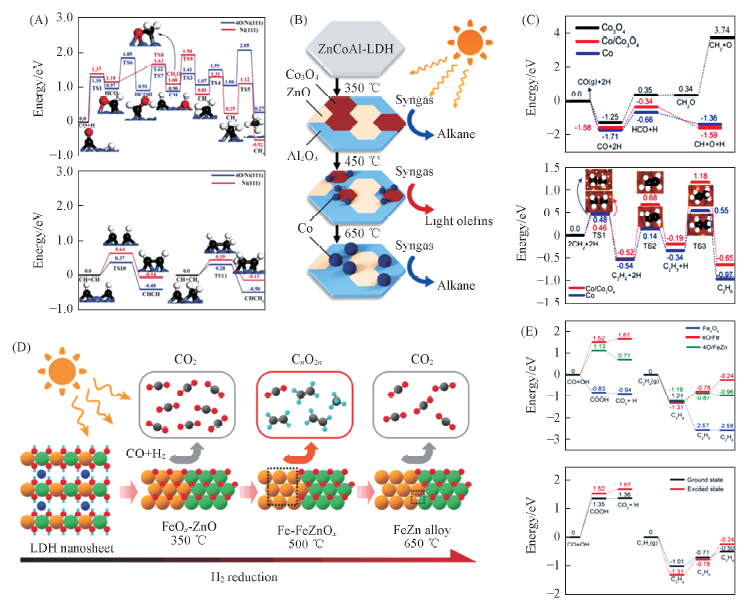
Fig.2 Potential-energy profile of the most possible reaction paths for syngas conversion on Ni(111) and 4O/Ni(111)(A)[47], fabrication of Co-x catalysts and their light-driven FTS performance(B)[48], the potential energy profile of CO dissociation on Co3O4(220), Co(111)/Co3O4(220), and Co(111), CH2 coupling and C2H4 hydrogenation on Co(111)/Co3O4(220) and Co(111)(C)[48], reductive transformation of ZnFeAl-LDH nanosheets to Fe-x catalysts and their photocatalytic behavior in FTS(D)[49] and energy profiles for CO2 formation, C2H4 adsorption and hydrogenation(E)[49](A) Copyright 2016, Wiley-VCH; (B, C) Copyright 2018, Wiley-VCH; (D, E) Copyright 2018, Wiley-VCH.
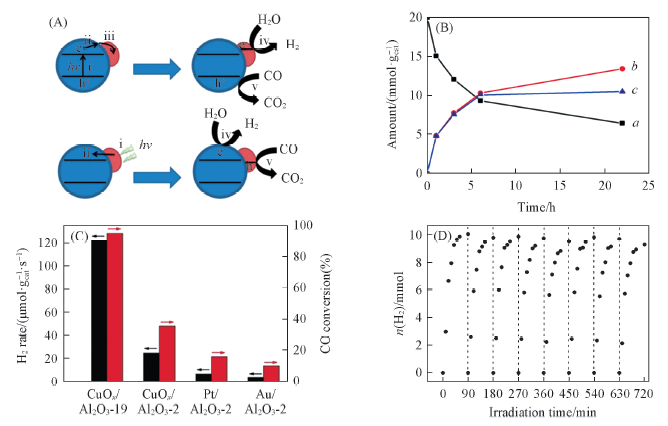
Fig.3 Two possible mechanisms to rationalize the differences in the UV-Vis or visible light photocatalytic activity of Au/TiO2(A)[52], temporal profile of CO consumption(a), CO2 evolution(b) and H2 evolution(c) over a Au/TiO2(1%) catalyst under solar light irradiation(B)[52], H2 evolution rates and CO conversion of CuOx/Al2O3-19, CuOx/Al2O3-2, Pt/Al2O3-2 and Au/Al2O3-2 catalysts(C)[53] and cycling measurements of WGS reaction under photothermal condition over CuOx/Al2O3 catalyst(D)[53](A, B) Copyright 2013, Royal Society of Chemistry; (C, D) Copyright 2019, Wiley-VCH.
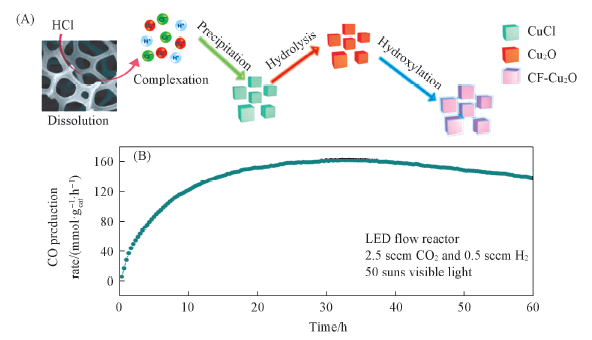
Fig.4 Schematic of CF-Cu2O growth process from the Cu foam precursor(A) and long-term stability of CF-Cu2O in an LED flow reactor(B)[57]Copyright 2019, Nature Publishing Group.
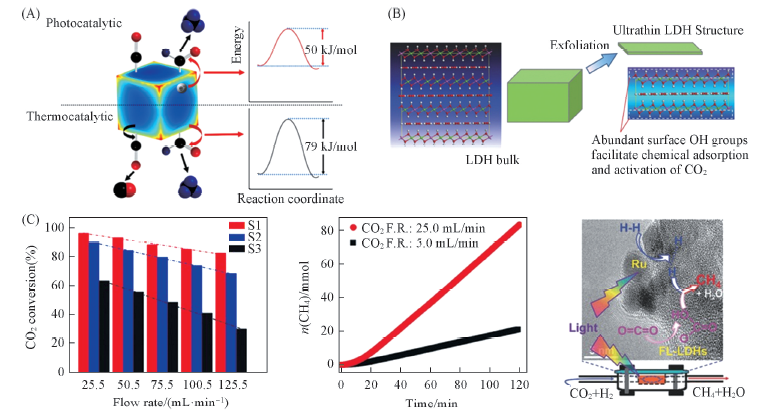
Fig.5 Reaction mechanism for CO and CH4 evolution on a rhodium nanocube(A)[58], schematic presence of the formation of ultrathin LDH structure with abundant surface hydroxyl groups(B)[60] and photothermal conversion of CO2 and cumulative yield of CH4 over Ru loaded catalysts under different flow rates(F. R. ) of CO2 and H2 mixture(C) [60](C) S1: Ru@FL-LDHs; S2: Ru@LDHs; S3: Ru@SiO2. (A) Copyright 2017,Nature Publishing Group; (B, C) Copyright 2016, Wiley-VCH.
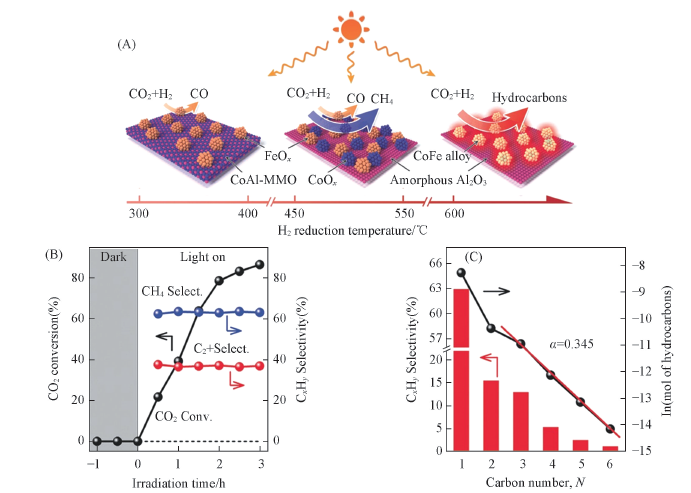
Fig.6 Illustration of the different CoFe-x catalysts formed by hydrogen reduction of CoFeAl-LDH nanosheets at different temperatures and their catalytic activities for CO2 hydrogenation(A), time course of CO2 conversion and product selectivities for CO2 hydrogenation over CoFe-650 under UV-Vis irradiation(B) and the hydrocarbon product distribution obtained over CoFe-650 under UV-Vis irradiation for 2 h(C)[61]Copyright 2018, Wiley-VCH.
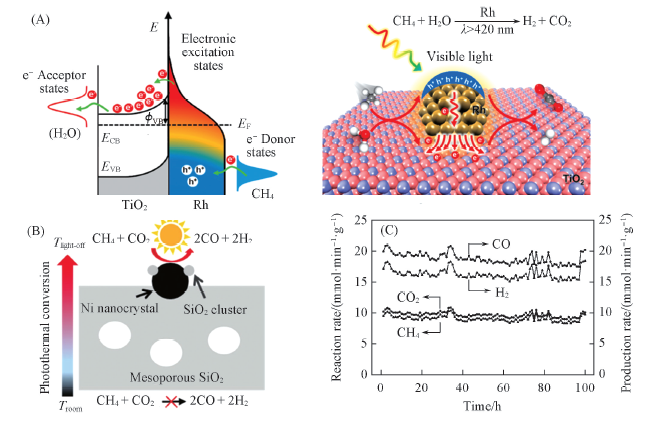
Fig.7 Schematic of energy transfer from photoexcited hot carriers to adsorbate states and proposed mechanism for SRM reaction on Rh/TiO2 under visible light illumination(A)[65], a schematic illustration of the solar-light-driven thermocatalytic(B) and the catalytic activity of SCM-Ni/SiO2 for CRM versus reaction time under the focused full-solar-spectrum irradiation from the Xe lamp(C)[66](A) Copyright 2018, American Chemical Society; (B, C) Copyright 2018, Wiley-VCH.
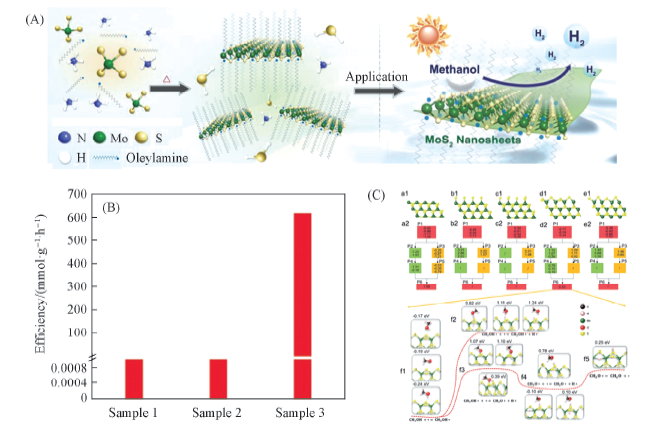
Fig.8 Synthesis and methanol photocatalytic application of single-layer MoS2 nanosheets(A), effects of surfactant, methanol, and photocatalysts on MTH efficiency between three different samples while keeping all other experimental conditions the same(B) and DFT calculations(C)[68](B) Sample 1: 10 mL(ca. 8.2 × 103 mg) pure surfactant; sample 2: 5 mL surfactant and 5 mL methanol; sample 3: 2 mg MoS2 in 10 mL methanol. Copyright 2019, Wiley-VCH.
| [1] | Li Z. J., Zhong L. S., Yu F., An Y. L., Dai Y. Y., Yang Y. Z., Lin T. J., Li S. G., Wang H., Gao P., Sun Y. H., He M. Y., ASC Catal., 2017, 7( 5), 3622— 3631 |
| [2] | Zhang Q. H., Kang J. C., Wang Y ., ChemCatChem, 2010, 2( 9), 1030— 1058 |
| [3] | Khodakov A. Y., Chu W., Fongarland P., Chem. Rev., 2007, 107( 5), 1692— 1744 |
| [4] | Leckel D., Energy Fuels, 2009, 23( 5/6), 2342— 2358 |
| [5] | Liu Y. F., de Tymowski B., Vigneron F., Florea I., Ersen O., Meny C., Nguyen P., Pham C., Luck F., Cuong P. H., ACS Catal., 2013, 3( 3), 393— 404 |
| [6] | Kudo A., Miseki Y., Chem. Soc. Rev., 2009, 38( 1), 253— 278 |
| [7] | Xu H., Ouyang S. X., Liu L. Q., Reunchan P., Umezawa N., Ye J. H., J. Mater. Chem. A, 2014, 2( 32), 12642— 12661 |
| [8] | Habisreutinger S. N., Schmidt-Mende L., Stolarczyk J. K., Angew. Chem. Int. Ed., 2013, 52( 29), 7372— 7408 |
| [9] | Wang L., Sasaki T., Chem. Rev., 2014, 114( 19), 9455— 9486 |
| [10] | Li Z. H., Liu J. J., Zhao Y. F., Shi R., Waterhouse G. I. N., Wang Y. S., Wu L. Z., Tung C. H., Zhang T. R., Nano Energy, 2019, 60, 467— 475 |
| [11] | Liu F. L., Song L. Z., Ouyang S. X., Xu H., Catal. Sci. Technol., 2019, 9( 9), 2125— 2131 |
| [12] | Liu G., Meng X., Zhang H., Zhao G., Pang H., Wang T., Li P., Kako T., Ye J., Angew. Chem. Int. Ed., 2017, 56( 20), 5570— 5574 |
| [13] | Neatu S., Macia-Agullo J. A., Concepcion P., Garcia H., J. Am. Chem. Soc., 2014, 136( 45), 15969— 15976 |
| [14] | Niu K. Y., Xu Y., Wang H. C., Ye R., Xin H. L., Lin F., Tian C. X., Lum Y. W., Bustillo K. C., Doeff M. M., Koper M. T. M., Ager J., Xu R., Zheng H. M., Sci. Adv., 2017, 3( 7), e1700921 |
| [15] | Zhao Y. F., Chen G. B., Bian T., Zhou C., Waterhouse G. I. N., Wu L. Z., Tung C. H., Smith L. J., O'Hare D., Zhang T. R., Adv. Mater., 2015, 27( 47), 7824— 7831 |
| [16] | Li N. X., Wang B. B., Si Y. T., Xue F., Zhou J. C., Lu Y. J., Liu M. C., ASC Catal., 2019, 9( 6), 5590— 5602 |
| [17] | Halabi M. H., de Croon M., van der Schaaf J., Cobden P. D., Schouten J. C., Appl. Catal. A: Gen., 2010, 389, 68— 79 |
| [18] | Kechagiopoulos P. N., Angeli S. D., Lemonidou A., Appl. Catal. B: Environ., 2017, 205, 238— 253 |
| [19] | Fang S. Y., Sun Z. X., Hu Y. H., ASC Catal., 2019, 9( 6), 5047— 5056 |
| [20] | Fischer F., Tropsch H ., Berichte Der Deutschen Chemischen Gesellschaft, 1923, 56, 2428— 2443 |
| [21] | De Klerk A., Energy Environ. Sci., 2011, 4( 4), 1177— 1205 |
| [22] | Schulz H., Appl. Catal. A: Gen., 1999, 186( 1/2), 3— 12 |
| [23] | Vannice M. A., J. Catal., 1975, 37( 3), 449— 461 |
| [24] | Vannice M. A., Garten R. L., J. Catal., 1979, 56( 2), 236— 248 |
| [25] | Enger B. C., Holmen A., Catal. Rev. Sci. Eng., 2012, 54( 4), 437— 488 |
| [26] | Kang J. C., Zhang S. L., Zhang Q. H., Wang Y., Angew. Chem. Int. Ed., 2009, 48( 14), 2565— 2568 |
| [27] | Liu Y. F., de Tymowski B., Vigneron F., Florea I., Ersen O., Meny C., Nguyen P., Pham C., Luck F., Cuong P. H., ACS Catal., 2013, 3( 3), 393— 404 |
| [28] | Leckel D., Energy Fuels, 2009, 23( 5/6), 2342— 2358 |
| [29] | Reddy G. K., Boolchand P., Smirniotis P. G., J. Phys. Chem. C, 2012, 116( 20), 11019— 11031 |
| [30] | Reddy G. K., Kim S. J., Dong J. H., Smirniotis P. G., Jasinski J. B., Appl. Catal. A: Gen., 2012, 415, 101— 110 |
| [31] | Tang Z., Kim S. J., Reddy G. K., Dong J. H., Smirniotis P., J. Membrane Sci., 2010, 354( 1/2), 114— 122 |
| [32] | Garcia-Garcia F. R., Rahman M. A., Gonzalez-Jimenez I. D., Li K., Catal. Today, 2011, 171( 1), 281— 289 |
| [33] | Zhang Z. H., Wang S. S., Song R., Cao T., Luo L. F., Chen X. Y., Gao Y. X., Lu J. Q., Li W. X., Huang W. X., Nat. Commum., 2017, 8( 1), 488 |
| [34] | Aguila G., Valenzuela A., Guerrero S., Araya P., Catal. Commum., 2013, 39, 82— 85 |
| [35] | Sun X. C., Lin J., Zhou Y., Li L., Su Y., Wang X. D., Zhang T., AIChE J., 2017, 63( 9), 4022— 4031 |
| [36] | Yao S. Y., Zhang X., Zhou W., Gao R., Xu W. Q., Ye Y. F., Lin L. L., Wen X. D., Liu P., Chen B. B., Crumlin E., Guo J. H., Zuo Z. J., Li W. Z., Xie J. L., Lu L., Kiely C. J., Gu L., Shi C., Rodriguez J. A., Ma D ., Science, 2017, 357( 6349), 389— 393 |
| [37] | Kokumai T. M., Cantane D. A., Melo G. T., Pauluccia L. B., Zanchet D., Catal. Today, 2017, 289, 249— 257 |
| [38] | D'Alessandro D. M., Smit B., Long J. R., Angew. Chem. Int. Ed., 2010, 49( 35), 6058— 6082 |
| [39] | Middleton R. S., Keating G. N., Stauffer P. H., Jordan A. B., Viswanathan H. S., Kang Q. J. J., Carey J. W., Mulkey M. L., Sullivan E. J., Chu S. P. P., Esposito R., Meckel T. A., Energy Environ. Sci., 2012, 5( 6), 7328— 7345 |
| [40] | Akri M., Zhao S., Li X. Y., Zang K. T., Lee A. F., IsaASC M. A., Xi W., Gangarajula Y., Luo J., Ren Y. J., Cui Y. T., Li L., Su Y., Pan X. L., Wen W., Pan Y., Wilson K., Li L., Qiao B. T., Ishii H., Liao Y. F., Wang A. Q., Wang X. D., Zhang T., Nat. Commum., 2019, 10( 1), 1— 10 |
| [41] | Wang L. S., Tao L. X., Xie M. S., Xu G. F., Huang J. S., Xu Y. D., Catal. Lett., 1993, 21, 35— 41 |
| [42] | Xu Y. D., Bao X. H., Lin L. W., J. Catal., 2003, 216, 386— 395 |
| [43] | Guo X., Fang G., Li G., Ma H., Fan H., Yu L., Ma C., Wu X., Deng D., Wei M., Tan D., Si R., Zhang S., Li J., Sun L., Tang Z., Pan X., Bao X ., Science, 2014, 344( 6184), 616— 619 |
| [44] | Lin L. L., Zhou W., Gao R., Yao S. Y., Zhang X., Xu W. Q., Zheng S. J., Jiang Z., Yu Q. L., Li Y. W., Shi C., Wen X. D., Ma D ., Nature, 2017, 544( 7648), 80— 83 |
| [45] | Tian P., Wei Y. X., Ye M .,Liu Z. M,ASC Catal., 2015, 5( 3), 1922— 1938 |
| [46] | Guo X. N., Jiao Z. F., Jin G. Q., Guo X. Y., ASC Catal., 2015, 5( 6), 3836— 3840 |
| [47] | Zhao Y., Zhao B., Liu J., Chen G., Gao R., Yao S., Li M., Zhang Q., Gu L., Xie J., Wen X., Wu L. Z., Tung C. H., Ma D., Zhang T. R., Angew. Chem. Int. Ed., 2016, 55( 13), 4215— 4219 |
| [48] | Li Z., Liu J., Zhao Y., Waterhouse G. I. N., Chen G., Shi R., Zhang X., Liu X., Wei Y., Wen X. D., Wu L. Z., Tung C. H., Zhang T. R., Adv. Mater., 2018, 30( 31), 1800527 |
| [49] | Zhao Y., Li Z., Li M., Liu J., Liu X., Waterhouse G. I. N., Wang Y., Zhao J., Gao W., Zhang Z., Long R., Zhang Q., Gu L., Liu X., Wen X., Ma D., Wu L. Z., Tung C. H., Zhang T. R., Adv. Mater., 2018, 30( 36), 1803127 |
| [50] | Gao W., Gao R., Zhao Y. F., Peng M., Song C. Q., Li M. Z., Li S. W., Liu J. J., Li W. Z., Deng Y. C., Zhang M. T., Xie J. L., Hu G., Zhang Z. S., Long R., Wen X. D., Ma D ., Chem., 2018, 4( 12), 2917— 2928 |
| [51] | Sastre F., Corma A., Garcia H., Angew. Chem. Int. Ed., 2013, 52( 49), 12983— 12987 |
| [52] | Sastre F., Oteri M., Corma A., Garcia H., Energy Environ. Sci., 2013, 6( 7), 2211— 2215 |
| [53] | Zhao L. K., Qi Y. H., Song L. Z., Ning S. B., Ouyang S. X., Xu H., Ye J. H., Angew. Chem. Int. Ed., 2019, 58( 23), 7708— 7712 |
| [54] | Tahir M., Amin N. S., Appl. Catal. A: Gen., 2015, 493, 90— 102 |
| [55] | Hoch L. B., Wood T. E., O'Brien P. G., Liao K., Reyes L. M., Mims C. A., Ozin G. A., Adv. Sci., 2014, 1( 1), 1400013 |
| [56] | Hoch L. B., O'Brien P. G., Jelle A., Sandhel A., Perovic D. D., Mims C. A., Ozin G. A., ACS Nano, 2016, 10( 9), 9017— 9025 |
| [57] | Wan L. L., Zhou Q. X., Wang X., Wood T. E., Wang L., Duchesne P. N., Guo J. L., Yan X. L., Xia M. K., Lie Y. F., Jelle A. A., Ulmer U., Jia J., Li T., Sun W., Ozin G. A., Nat. Catal., 2019, 2( 10), 889— 898 |
| [58] | Zhang X., Li X. Q., Zhang D., Su N. Q., Yang W. T., Everitt H. O., Liu J., Nat. Commum., 2017, 8, 1— 9 |
| [59] | O'Brien P. G., Sandhel A., Wood T. E., Jelle A. A., Hoch L. B., Perovic D. D., Mims C. A., Ozin G. A., Adv. Sci., 2014, 1( 1), 1400001 |
| [60] | Ren J., Ouyang S. X., Xu H., Meng X. G., Wang T., Wang D. F., Ye J. H., Adv. Energy Mater., 2017, 7( 5), 1601657 |
| [61] | Zhang W., Wang L., Wang K., Khan M. U., Wang M., Li H., Zeng J ., Small, 2017, 13( 7), 1602583 |
| [62] | Chen G. B., Gao R., Zhao Y. F., Li Z. H., Waterhouse G. I. N., Shi R., Zhao J. Q., Zhang M. T. , Shang L., Sheng G. Y., Zhang X. P., Wen X. D., Wu L. Z., Tung C. H., Zhang T. R., Adv. Mater., 2018, 30( 3), 1704663 |
| [63] | Taylor C. E., Catal. Today, 2003, 84, 9— 15 |
| [64] | Villa K., Murcia-Lopez S., Morante J. R., Andreu T., Appl. Catal. B: Environ., 2016, 187, 30— 36 |
| [65] | Song H., Meng X. G., Wang Z. J., Wang Z. A., Chen H. L., Weng Y. X., Ichihara F., Oshikiri M., Kako T., Ye J. H., ACS Catal., 2018, 8( 8), 7556— 7565 |
| [66] | Huang H., Mao M. Y., Zhang Q., Li Y. Z., Bai J. L., Yang Y., Zeng M., Zhao X. J., Adv. Energy Mater., 2018, 8( 10), 1702472 |
| [67] | Liu Z. Q., Yin Z. Y., Cox C., Bosman M., Qian X. F., Li N., Zhao H. Y., Du Y. P., Li J ., Nocera D. G. , Sci. Adv., 2016, 2( 9), e1501425 |
| [68] | Pang Y., Uddin M. N., Chen W., Javaid S., Barker E., Li Y., Suvorova A., Saunders M., Yin Z., Jia G., Adv. Mater., 2019, 31( 49), 1970348 |
| [1] | 韦春洪, 蒋倩, 王盼盼, 江成发, 刘岳峰. 贵金属Pt促进Co基费托合成催化剂的原子尺度结构分析[J]. 高等学校化学学报, 2022, 43(8): 20220074. |
| [2] | 郭志强, 杨博如, 席婵娟. 硼氢化试剂在二氧化碳还原官能化反应中的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220199. |
| [3] | 张昕昕, 许狄, 王艳秋, 洪昕林, 刘国亮, 杨恒权. CO2加氢制低碳醇CuFe基催化剂中的Mn助剂效应[J]. 高等学校化学学报, 2022, 43(7): 20220187. |
| [4] | 周紫璇, 杨海艳, 孙予罕, 高鹏. 二氧化碳加氢制甲醇多相催化剂研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220235. |
| [5] | 刘瀚林, 尹琳琳, 陈西凤, 李国栋. 氧化铟基纳米催化剂用于二氧化碳选择性加氢的研究进展[J]. 高等学校化学学报, 2021, 42(5): 1430. |
| [6] | 王鹏, 刘欢, 杨妲. 一氧化碳参与不饱和烃类化合物羰基化反应的研究进展[J]. 高等学校化学学报, 2021, 42(10): 3024. |
| [7] | 张维中,温月丽,宋镕鹏,王斌,张倩,黄伟. 催化剂表面Cu0含量对二氧化碳加氢合成C2+醇性能的影响[J]. 高等学校化学学报, 2020, 41(6): 1297. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||