

高等学校化学学报 ›› 2019, Vol. 40 ›› Issue (3): 431.doi: 10.7503/cjcu20180717
收稿日期:2019-10-22
出版日期:2019-01-24
发布日期:2019-01-24
作者简介:联系人简介: 王 存, 女, 讲师, 主要从事纳米材料及电化学生物传感器方面的研究. E-mail:
基金资助:
WANG Cun*( ), MENG Li, HUI Junmin
), MENG Li, HUI Junmin
Received:2019-10-22
Online:2019-01-24
Published:2019-01-24
Contact:
WANG Cun
E-mail:wangcun5224@126.com
摘要:
以三聚氰胺为原料, 采用热聚合法合成了类石墨烯状二维片状氮化碳(g-C3N4)纳米材料; 通过电沉积和高电位氧化的方法制得氧化聚咪唑(PImox)/g-C3N4修饰电极(PImox/g-C3N4/GCE). 采用扫描电子显微镜(SEM)和X射线粉末衍射仪(XRD)对g-C3N4纳米材料进行了表征; 通过循环伏安法(CV)和差分脉冲伏安法(DPV)考察了尿酸(UA)、 黄嘌呤(XA)和次黄嘌呤(HX)在该电极上的电化学行为. 结果表明, UA, XA和HX的检测线性范围分别为2.0~216.0, 5.0~542.0和5.0~778.0 μmol/L; 检出限分别为0.17, 0.30和0.30 μmol/L. 将该修饰电极用于实际样品(血清和尿液)中UA, XA和HX的同时测定, 加标回收率为98.4%~105.2%.
中图分类号:
TrendMD:
王存, 孟丽, 惠俊敏. 聚咪唑/氮化碳新型纳米复合材料修饰电极对尿酸、 黄嘌呤和次黄嘌呤的同时检测. 高等学校化学学报, 2019, 40(3): 431.
WANG Cun,MENG Li,HUI Junmin. Simultaneous Determination of Uric Acid, Xanthine and Hypoxanthine Based on Polyimidazole/Carbon Nitride Novel Nanocomposite†. Chem. J. Chinese Universities, 2019, 40(3): 431.
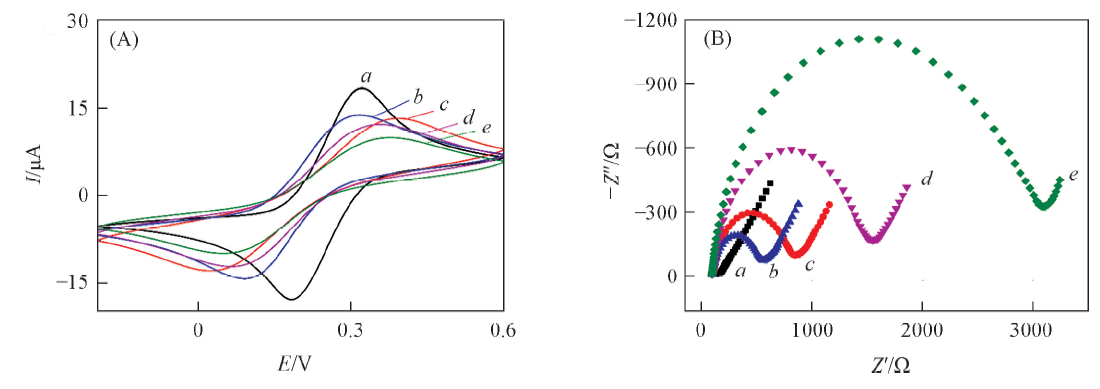
Fig.3 Cyclic voltammograms(A) and electrochemical impedance analysis(B) at GCE(a), PIm/GCE(b),g-C3N4/GCE(c), PIm/g-C3N4/GCE(d) and PImox/g-C3N4/GCE(e)
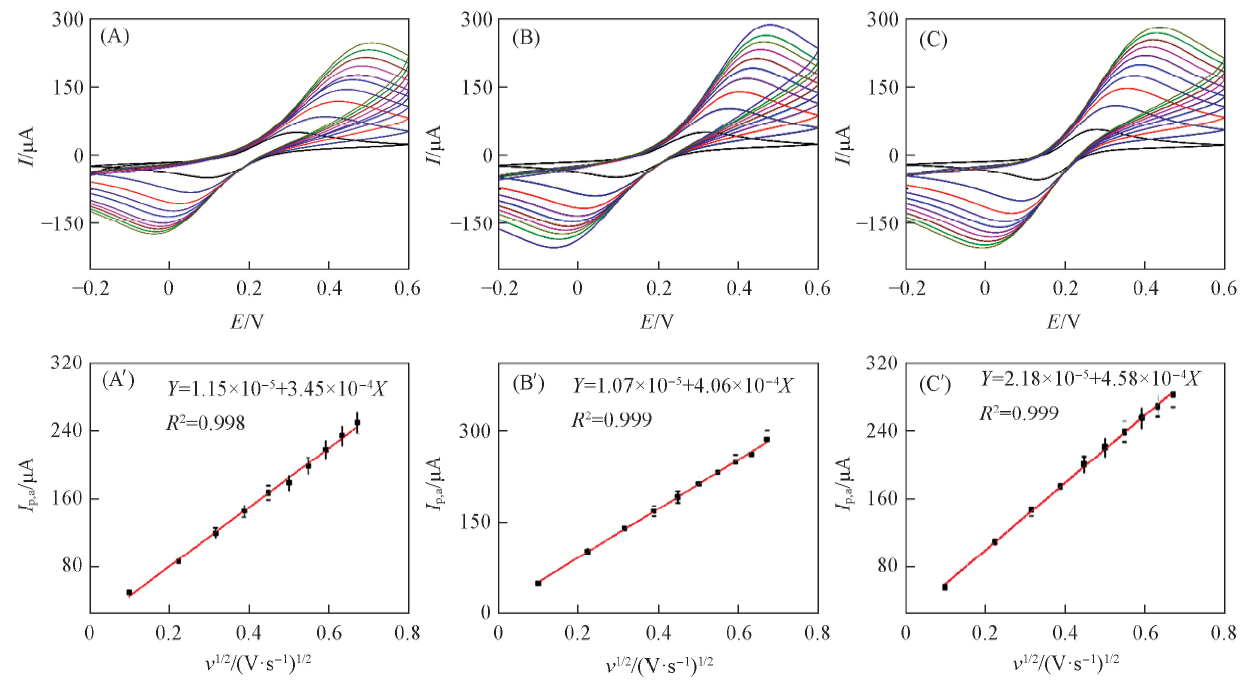
Fig.4 CVs of g-C3N4/GCE(A), PIm/GCE(B), PImox/g-C3N4/GCE(C) in 5.0 mmol/L K3[Fe(CN)6]+ 5.0 mmol/L K4[Fe(CN)6] at different scan rates and plots of anodic peak current(Ip,a) vs. square root of scan rate of g-C3N4/GCE(A'), PIm/GCE(B') and PImox/g-C3N4/GCE(C')(A)—(C) Scan rate from inner to outer: 0.01, 0.05, 0.10, 0.15, 0.20, 0.25, 0.30, 0.35, 0.40 and 0.45 V/s.
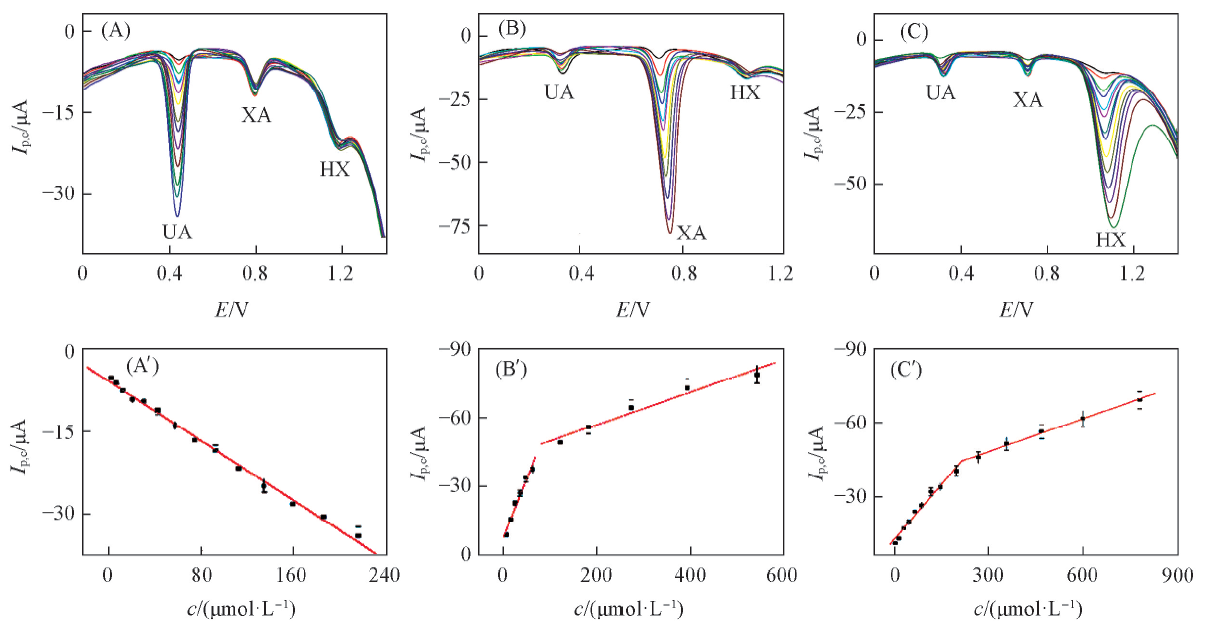
Fig.7 DPV curves at PImox/g-C3N4/GCE in 0.1 mol/L PBS solution(pH=4.0)[(A)—(C)] and plots of Ip vs. concentration for UA(A'), XA(B') and HX(C')(A) 15 μmol/L XA, 15 μmol/L HX, concentrations of UA from inner to outer: 2, 6, 12, 20, 30, 42, 57, 74, 92, 112, 134, 159, 186, 216 μmol/L; (B) 50 μmol/L UA, 15 μmol/L HX, concentrations of XA from inner to outer: 5, 15, 25, 35, 47, 62, 122, 182, 272, 392, 542 μmol/L; (C) 50 μmol/L UA, 10 μmol/L XA, concentrations of HX from inner to outer: 5, 15, 30, 47, 67, 87, 117, 147, 197, 267, 357, 468, 598, 778 μmol/L.
| Analyte | Linear range/(μmol·L-1) | Regression equation | Correlation coefficient | Detection limit/(μmol·L-1) |
|---|---|---|---|---|
| UA | 2.0—216.0 | Ip,c=-5.82-0.137c | 0.998 | 0.17 |
| XA | 5.0—62.0 | Ip,c=-8.04-0.511c | 0.987 | 0.30 |
| 122.0—542.0 | Ip,c=-42.8-0.0713c | 0.981 | ||
| HX | 5.0—197.0 | Ip,c=-12.9-0.149c | 0.992 | 0.30 |
| 267.0—778.0 | Ip,c=-35.0-0.0446c | 0.998 |
Table 1 Linear range, regression equation, correlation coefficient and detection limit for UA, XA and HX
| Analyte | Linear range/(μmol·L-1) | Regression equation | Correlation coefficient | Detection limit/(μmol·L-1) |
|---|---|---|---|---|
| UA | 2.0—216.0 | Ip,c=-5.82-0.137c | 0.998 | 0.17 |
| XA | 5.0—62.0 | Ip,c=-8.04-0.511c | 0.987 | 0.30 |
| 122.0—542.0 | Ip,c=-42.8-0.0713c | 0.981 | ||
| HX | 5.0—197.0 | Ip,c=-12.9-0.149c | 0.992 | 0.30 |
| 267.0—778.0 | Ip,c=-35.0-0.0446c | 0.998 |
| Material | Linear range/(μmol·L-1) | Detection limit/(μmol·L-1) | Ref. | ||||
|---|---|---|---|---|---|---|---|
| UA | XA | HX | UA | XA | HX | ||
| Poly(bromocresol purple) | 0.5—120 | 0.1—100 | 0.2—80 | 0.2 | 0.06 | 0.12 | [ |
| Overoxidized dopamine polymer and | 1.8—238 | 5.2—289 | 3.8—293 | 0.6 | 1.7 | 1.3 | [ |
| 3,4,9,10-perylenetetracarboxylic acid | |||||||
| Poly(2-amino-1,3,4-thiadiazole) | 5—45 | 5—45 | | 0.19 | 0.59 | | [ |
| Copper(Ⅱ)-polydopamine | 60—1680 | | | 24.6 | | | [ |
| Functional single wall carbon nanotubes | 0.1—100 | 0.2—100 | 0.8—100 | 0.08 | 0.146 | 0.562 | [ |
| Ru(DMSO)4Cl2 nano-aggregated | 100—700 | 50—500 | 50—500 | 0.372 | 2.35 | 2.37 | [ |
| nafion membrane | |||||||
| Overoxidized polyimidazole/carbon nitride | 2.0—216 | 5.0—542 | 5.0—778 | 0.17 | 0.30 | 0.30 | This work |
Table 2 Comparison of the response characteristics of different modified electrodes
| Material | Linear range/(μmol·L-1) | Detection limit/(μmol·L-1) | Ref. | ||||
|---|---|---|---|---|---|---|---|
| UA | XA | HX | UA | XA | HX | ||
| Poly(bromocresol purple) | 0.5—120 | 0.1—100 | 0.2—80 | 0.2 | 0.06 | 0.12 | [ |
| Overoxidized dopamine polymer and | 1.8—238 | 5.2—289 | 3.8—293 | 0.6 | 1.7 | 1.3 | [ |
| 3,4,9,10-perylenetetracarboxylic acid | |||||||
| Poly(2-amino-1,3,4-thiadiazole) | 5—45 | 5—45 | | 0.19 | 0.59 | | [ |
| Copper(Ⅱ)-polydopamine | 60—1680 | | | 24.6 | | | [ |
| Functional single wall carbon nanotubes | 0.1—100 | 0.2—100 | 0.8—100 | 0.08 | 0.146 | 0.562 | [ |
| Ru(DMSO)4Cl2 nano-aggregated | 100—700 | 50—500 | 50—500 | 0.372 | 2.35 | 2.37 | [ |
| nafion membrane | |||||||
| Overoxidized polyimidazole/carbon nitride | 2.0—216 | 5.0—542 | 5.0—778 | 0.17 | 0.30 | 0.30 | This work |
| Sample | Dilution factor | Analyte | Detected/(μmol·L-1) | Added/(μmol·L-1) | Found/(μmol·L-1) | Recovery(%) |
|---|---|---|---|---|---|---|
| Urine 1 | 50 | UA | 30.0 | 20.0 | 50.2 | 100.4 |
| 50 | XA | | 40.0 | 39.7 | 99.2 | |
| 50 | HX | | 30.0 | 31.1 | 103.7 | |
| Urine 2 | 100 | UA | 25.0 | 80.0 | 105.5 | 100.5 |
| 100 | XA | | 100.0 | 98.6 | 98.6 | |
| 100 | HX | | 100.0 | 105.2 | 105.2 | |
| Serum 1 | 150 | UA | | 90.0 | 89.2 | 99.1 |
| 150 | XA | | 50.0 | 49.3 | 98.6 | |
| 150 | HX | | 50.0 | 50.9 | 101.8 | |
| Serum 2 | 200 | UA | | 150.0 | 147.6 | 98.4 |
| 200 | XA | | 400.0 | 398.7 | 99.7 | |
| 200 | HX | | 500.0 | 498.9 | 99.8 |
Table 3 Determination of UA, XA and HX in human serum and urine samples
| Sample | Dilution factor | Analyte | Detected/(μmol·L-1) | Added/(μmol·L-1) | Found/(μmol·L-1) | Recovery(%) |
|---|---|---|---|---|---|---|
| Urine 1 | 50 | UA | 30.0 | 20.0 | 50.2 | 100.4 |
| 50 | XA | | 40.0 | 39.7 | 99.2 | |
| 50 | HX | | 30.0 | 31.1 | 103.7 | |
| Urine 2 | 100 | UA | 25.0 | 80.0 | 105.5 | 100.5 |
| 100 | XA | | 100.0 | 98.6 | 98.6 | |
| 100 | HX | | 100.0 | 105.2 | 105.2 | |
| Serum 1 | 150 | UA | | 90.0 | 89.2 | 99.1 |
| 150 | XA | | 50.0 | 49.3 | 98.6 | |
| 150 | HX | | 50.0 | 50.9 | 101.8 | |
| Serum 2 | 200 | UA | | 150.0 | 147.6 | 98.4 |
| 200 | XA | | 400.0 | 398.7 | 99.7 | |
| 200 | HX | | 500.0 | 498.9 | 99.8 |
| [1] | Kalimuthu P., Leimkühler S., Bernhardt P. V., Anal. Chem., 2012, 84(23), 10359—10365 |
| [2] | Emrana M. Y., Shenashen M. A., Morita H., El-Safty S. A., Biosens. Bioelectron., 2019, 109, 237—245 |
| [3] | Emran M.Y.., Shenashen M. A., Abdelwah A. A., Khalif H., Mekawy M., Akhtar N., Abdelmottale M., El-Safty S. A.,J. Appl. Electrochem., 2019, 48(5), 529—542 |
| [4] | Wang Y., Colloid. Surface. B, 2011, 88(2), 614—621 |
| [5] | Ibrahim H., Temerk Y., Sensor. Actuat.B: Chem., 2016, 232, 125—137 |
| [6] | Zhang F. Y., Wang Z. H., Zhang Y. Z., Zheng Z. X., Wang C. M., Du Y. L., Ye W. C., Talanta, 2012, 93, 320—325 |
| [7] | Cooper N., Khosravan R., Erdmann C., Fiene J., Lee J. W. J., Chromatogr. B, 2006, 837(1), 1—10 |
| [8] | Causse E., Pradelles A., Dirat B., Negre-Salvayre A., Salvayre R., Couderc F., Electrophoresis, 2007, 28(3), 381—387 |
| [9] | Burt J. R., Process Biochem., 1977, 12(1), 32—37 |
| [10] | Ibrahim H., Temerk Y., Sensor.Actuat. B: Chem., 2016, 232, 125—137 |
| [11] | Lavany N., Sekar C., Murugan R., Ravi G., Mat. Sci. Eng. C, 2016, 65, 278—286 |
| [12] | Wang X. L., Zheng K. Y., Feng X., Xu C. H., Song W. B., Sensor. Actuat. B: Chem., 2015, 219, 361—369 |
| [13] | Wang Y., Tong L. L., Sensor. Actuat. B: Chem., 2010, 150, 43—49 |
| [14] | Emran M. Y., Mekawy M., Akhtar N., Shenashen M. A., EL-Sewify I. M., Faheem A., El-Safty S. A., Biosens. Bioelectron., 2019, 100, 122—131 |
| [15] | Zhu D., Guo D. X., Zhang L. L., Tan L. C., Pang H. J., Ma H. Y., Zha M., Sensor. Actuat. B: Chem., 2019, 281, 893—904 |
| [16] | Dalkiran B., Kacar C., Erden P. E., Kilic E., Sensor. Actuat. B: Chem., 2014, 200, 83—91 |
| [17] | Wang Y., Colloid. Surface. B, 2011, 88(2), 614—621 |
| [18] | Ojani R., Alinezhad A., Abedi Z., Sensor. Actuat. B: Chem., 2013, 188, 621—630 |
| [19] | Pierini G. D., Robledo S. N., Zon M. A., Nezio M. S. D., Granero A. M., Fernández H., Microchem. J., 2019, 138, 58—64 |
| [20] | Wen Y.P.., Chang J., Xu L. J., Liao X. N., Bai L., Lan Y. D., Li M. F.,J. Electroanal. Chem., 2017, 805, 159—170 |
| [21] | Wang Y. G., Wang Y. Z., Chen Y. T., Yin C. C., Zuo Y. H., Cui L. F., Mater. Lett., 2015, 139(1), 70—72 |
| [22] | Xing Z., Chen Z. X., Zong X., Wang L. Z., Chem. Commun., 2014, 50, 6762—6764 |
| [23] | Liu X. F., Ou X., Lu Q. Y., Zhang J. J., Chen S. H., Wei S. P., RSC Adv., 2014, 4, 42632—42637 |
| [24] | Wang C., Xiong Z.W.., Sun P., Wang R., Zhao X., Wang Q.,J. Electroanal. Chem., 2017, 801, 395—402 |
| [25] | Wang C., Li J., Luo X.Y.., Hui J. M., Liu X., Tan J., Zhao X.,J. Electroanal. Chem., 2016, 780, 147—152 |
| [26] | Wang Y. G., Wang Y. Z., Chen Y. T., Yin C. C., Zuo Y. H., Cui L. F., Mater. Lett., 2015, 139(1), 70—72 |
| [27] | Wang W. J., Li G. Y., An T. C., Chan D. K. L., Yu J. M. C., Wong P. K., Appl. Catal. B: Environ., 2019, 238, 126—135 |
| [28] | An H., Lin B., Xue C., Yan X. Q., Dai Y. Z., Wei J. J., Yang G. D., Chinese. J. Catal., 2019, 39(4), 654—663 |
| (安华, 林波, 薛超, 严孝清, 代艳竹, 魏进家, 杨贵东. 催化学报, 2019, 39(4), 654—663) | |
| [29] | Lamouroux C., Foglia G., Le Rouzo G., J. Chromatogr. A, 2011, 1218(20), 3022—3028 |
| [30] | Zhang Y., Yuan R., Chai Y. Q., Li W. J., Zhong X., Zhong H. A., Biosens. Bioelectron., 2011, 26(9), 3977—3980 |
| [31] | Wang C., Yuan R., Chai Y. Q., Chen S. H., Zhang Y., Hu F. X., Zhang M. H., Electrochim. Acta, 2012, 62, 109—115 |
| [32] | Kalimuthu P., John S. A., Talanta, 2010, 80(5), 1686—1691 |
| [33] | Sahin M., Sahin Y., Ozcan A., Sensor. Actuat.B: Chem., 2008, 133(1), 5—14 |
| [34] | Ulubay S., Dursun Z., Talanta, 2010, 80(3), 1461—1466 |
| [35] | Tu X. M., Xie Q. J., Jiang S. Y., Yao S. Z., Biosens. Bioelectron., 2007, 22(12), 2819—2826 |
| [36] | Lian Q. W., He Z. F., He Q., Luo A., Yan K. W., Zhang D. X., Lu X. Q., Zhou X. B., Anal. Chim. Acta, 2014, 823, 32—39 |
| [37] | Huang L., Jiao S. F., Li M. G., Electrochim. Acta, 2014, 121, 233—239 |
| [38] | Wang Z., Dong X., Li. J., Sensor. Actuat.B: Chem., 2008, 131(2), 411—416 |
| [39] | Kumar A.S.., Swetha P.,J. Electroanal. Chem., 2010, 642(2), 135—142 |
| [1] | 龚妍熹, 王建兵, 柴歩瑜, 韩元春, 马云飞, 贾超敏. 钾掺杂g-C3N4薄膜光阳极的制备及光电催化氧化降解水中双氯芬酸钠性能[J]. 高等学校化学学报, 2022, 43(6): 20220005. |
| [2] | 魏闯宇, 陈艳丽, 姜建壮. 基于乙硫基取代的三层酞菁铕二聚体修饰ITO电极构筑电化学多巴胺和尿酸传感器[J]. 高等学校化学学报, 2022, 43(1): 20210582. |
| [3] | 王鹏, 阳敏, 汤森培, 陈飞台, 李佑稷. 蜂窝状C3N4/CoSe2/GA复合光催化剂的制备及CO2还原性能[J]. 高等学校化学学报, 2021, 42(6): 1924. |
| [4] | 李冬平, 李彬, 李长恒, 于薛刚, 单妍, 陈克正. Ni5P4/g-C3N4复合光催化剂的制备及光催化性能[J]. 高等学校化学学报, 2021, 42(4): 1292. |
| [5] | 齐人睿, 李明昊, 常浩, 付学奇, 高波, 韩葳葳, 韩璐, 李婉南. 基于拉伸分子动力学模拟的黄嘌呤氧化酶抑制剂解离途径的理论研究[J]. 高等学校化学学报, 2021, 42(3): 758. |
| [6] | 刘志刚, 李家宝, 杨剑, 马浩, 王赪胤, 郭鑫, 汪国秀. 新型石墨化氮化碳/锡/氮掺杂碳复合物的制备及储钠性能[J]. 高等学校化学学报, 2021, 42(2): 633. |
| [7] | 王乙舒, 李雪, 闫丽, 徐红赟, 祝玉鑫, 宋艳华, 崔言娟. 二维Z型BCN/Sn3O4复合材料的光催化还原性能[J]. 高等学校化学学报, 2021, 42(12): 3722. |
| [8] | 孙亚光, 张含烟, 明涛, 徐宝彤, 高雨, 丁茯, 徐振和. ZnIn2S4/g-C3N4复合材料的制备及可见光催化制氢性能[J]. 高等学校化学学报, 2021, 42(10): 3160. |
| [9] | 廖妮, 张捷源, 黄梓阳, 赵晏汐, 柴雅琴, 袁若, 卓颖. 共晶诱导增强的电化学发光9,10-二苯基蒽-苝微晶构建高效尿酸传感器[J]. 高等学校化学学报, 2020, 41(9): 1989. |
| [10] | 祝玉鑫, 欧阳杰, 宋艳华, 唐盛, 崔言娟. 硼碘共掺杂氮化碳的制备及光解水制氢性能[J]. 高等学校化学学报, 2020, 41(7): 1645. |
| [11] | 马祥英, 廖艳娟, 覃方红, 尹源浩, 黄在银, 陈其锋. 碳自掺杂g-C3N4光催化性能的原位光微量热-荧光光谱研究[J]. 高等学校化学学报, 2020, 41(11): 2526. |
| [12] | 赵艳锋,孙效龙,胡绍争,王辉,王菲,李萍. 氧掺杂对VN-g-C3N4催化剂光催化固氮性能的影响[J]. 高等学校化学学报, 2020, 41(1): 132. |
| [13] | 殷雯婧, 刘啸, 钱汇东, 邹志青. 高活性位点密度Fe-N共掺杂碳纳米片的制备及氧还原性能[J]. 高等学校化学学报, 2019, 40(7): 1480. |
| [14] | 贾宏亮, 赵建伟, 秦丽溶, 赵敏. 基于镍丝负载氧化镍纳米片的尿酸生物传感器[J]. 高等学校化学学报, 2019, 40(2): 240. |
| [15] | 梁芷珊, 倪爽, 代梦娇, 韩方杰, 韩立鹏, 牛利, 韩冬雪. 基于g-C3N4/P25复合光电敏感体系的中草药抗氧化容量测定[J]. 高等学校化学学报, 2019, 40(10): 2081. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||