

高等学校化学学报 ›› 2018, Vol. 39 ›› Issue (7): 1475.doi: 10.7503/cjcu20180117
收稿日期:2018-02-09
出版日期:2018-07-10
发布日期:2018-06-14
作者简介:联系人简介: 王梅艳, 女, 博士, 讲师, 主要从事催化反应微观机理的理论研究. E-mail:基金资助:
FANG Sheng, WANG Meiyan*( ), LIU Jingjing, LIU Jingyao
), LIU Jingjing, LIU Jingyao
Received:2018-02-09
Online:2018-07-10
Published:2018-06-14
Contact:
WANG Meiyan
E-mail:mywang858@jlu.edu.cn
Supported by:摘要:
采用密度泛函理论(DFT)方法, 对镍配合物Ni(PPh3)2催化N-烯丙基酰胺异构化生成N-丙烯基酰胺的微观反应机理进行了计算. 反应涉及了C—H键活化、 异构化及还原消除生成新的C—H键等步骤. 对C—H键活化和异构化步骤, 分别考虑了Ni(PPh3)2和Ni(PPh3)的催化活性, 发现均为前者对应的能垒更低; 对异构化步骤, 分别考虑了π-烯丙基和σ-烯丙基机理, 发现前者能垒更低. 在整个反应路径中, 生成产物E异构体的决速能垒为141.8 kJ/mol, 与生成Z异构体的决速能垒(141.1 kJ/mol)仅差0.7 kJ/mol, 与实验上E/Z选择性不高(56/44)一致. Pd(PPh3)2催化的决速中间体和过渡态的计算表明, 生成E和Z异构体的决速能垒较高, 均超过175 kJ/mol, 与实验上Pd(PPh3)4没有催化活性一致. Ni(PPh3)2和Pd(PPh3)2催化活性不同, 可由Ni的d电子对烯丙基阴离子π*反键的反馈作用较Pd更强来解释. 此外, 通过反应物中不同取代基对产物E/Z选择性影响的分析, 发现E/Z选择性不同是由各取代基在生成E和Z异构体的决速过渡态中所受空间位阻不同所导致.
中图分类号:
TrendMD:
方升, 王梅艳, 刘静静, 刘靖尧. 镍配合物催化N-烯丙基酰胺异构化反应机理. 高等学校化学学报, 2018, 39(7): 1475.
FANG Sheng, WANG Meiyan, LIU Jingjing, LIU Jingyao. Reaction Mechanism of Nickel Complex Catalyzed Isomerization of N-Allylamides†. Chem. J. Chinese Universities, 2018, 39(7): 1475.
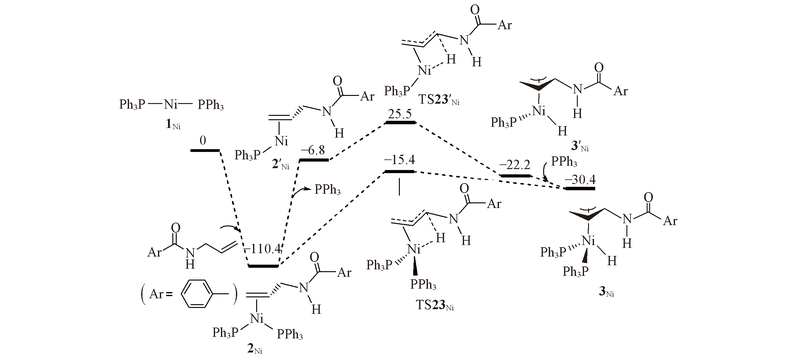
Fig.1 Energy profiles calculated for C—H bond activation by Ni(PPh3)2(1Ni) to form complex 3Ni^ The calculated free energies relative to 1Ni+N-allyl-4-methylbenzamide are given in kJ/mol.
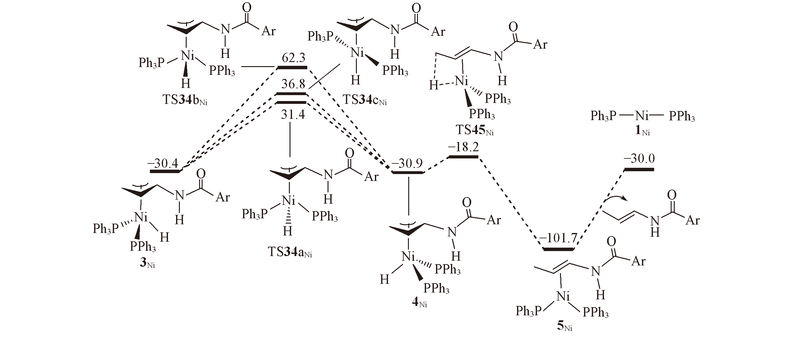
Fig.2 Energy profiles calculated for isomerization of π-allyl complex 3Ni via the π-allyl mechanism to generate E isomer^ The calculated free energies relative to 1Ni+N-allyl-4-methylbenzamide are given in kJ/mol.
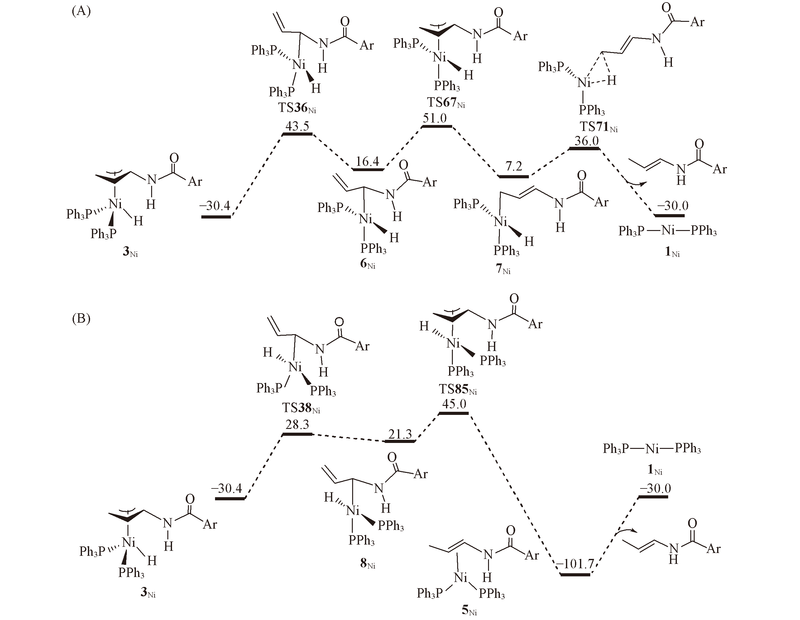
Fig.3 Energy profiles calculated for isomerization of π-allyl complex 3Ni via the σ-allyl mechanism to generate E isomer^ The H atom bonded to Ni is directed outwards(A) and inwards(B). The calculated free energies relative to 1Ni+N-allyl-4-methylbenzamide are given in kJ/mol.
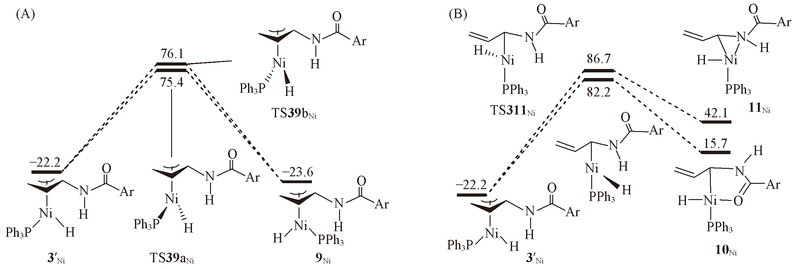
Fig.4 Energy profiles calculated for isomerization of π-allyl complex 3'Ni via the π-allyl(A) and σ-allyl(B) mechanism^ The calculated free energies relative to 1Ni+N-allyl-4-methylbenzamide are given in kJ/mol.
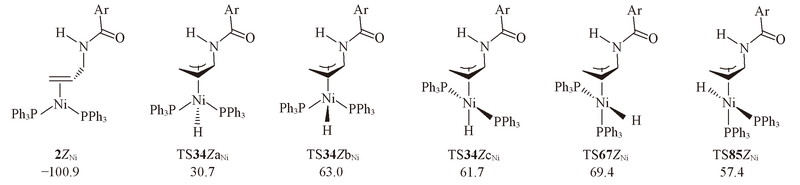
Fig.5 Structures and energies of the rate-determining intermediate 2ZNi and five lowest transition states on the energy profiles to generate Z isomer^The calculated free energies relative to 1Ni+N-allyl-4-methylbenzamide are given in kJ/mol.
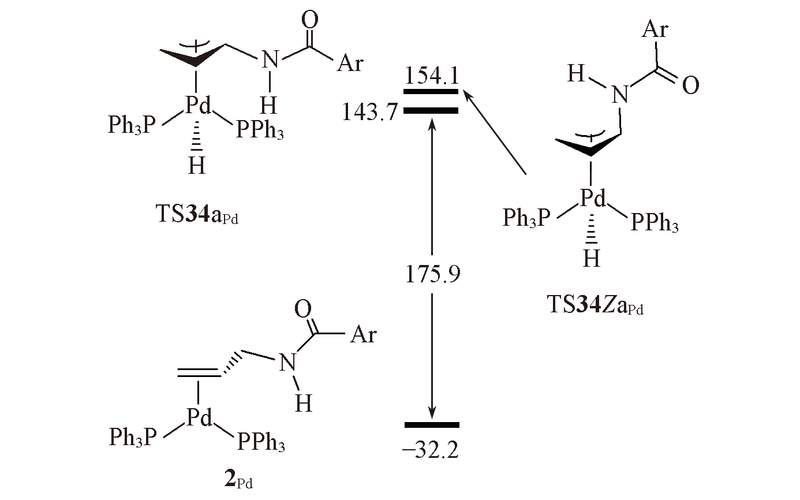
Fig.6 Structures and energies of the rate-determining intermediate 2Pd and transition states TS34aPd and TS34ZaPd on the energy profiles to generate E and Z isomer^The calculated free energies relative to 1Pd+N-allyl-4-methylbenzamide are given in kJ/mol.
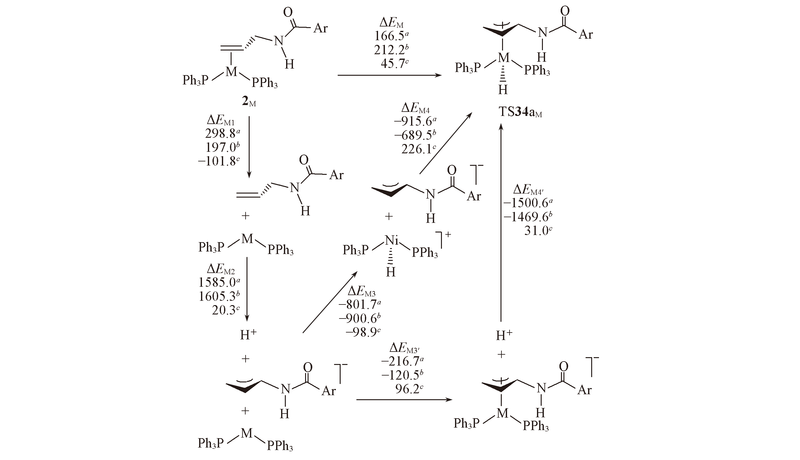
Fig.7 Energy decomposition analyses of the energy barriers(kJ/mol) from rate-determining intermediates 2M to transition states TS34aM^ a. The energy of Ni; b. the energy of Pd; c. the energy difference of Ni and Pd.
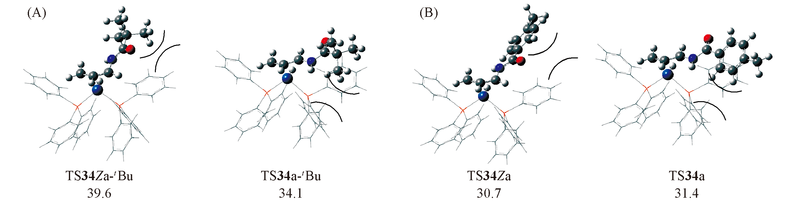
Fig.8 Structures and energies(kJ/mol) of two rate-determining transition states TS34a-tBu and TS34Za-tBu(A) for reactant with R=tBu (relative to 1Ni+N-allyl-pivaloylamide), as well as TS34a and TS34Za(B) for reactant with R=p-MeC6H4 (relative to 1Ni+N-allyl-4-methylbenzamide)
| [1] | Van Santen R.A.,van Leeuwen P. W. N. M.,Moulijn J A.,Averill B. A., Catalysis: An Integrated Approach, Elsevier,Amsterdam, 1999 |
| [2] | Mirza-Aghayan M., Boukherroub R., Bolourtchian M., Hoseini M., Tabar-Hydar K., J. Organomet. Chem., 2003, 678, 1—4 |
| [3] | Donohoe T. J., O'Riordan T. J. C., Rosa C. P., Angew. Chem. Int. Ed., 2009, 48, 1014—1017 |
| [4] | Krompiec S., Krompiec M., Penczek H., Ignasiak R., Coord. Chem. Rev., 2008, 252, 1819—1841 |
| [5] | Escoubet S., Gastaldi S., Bertrand M.,Eur. J. Org. Chem., 2005, 3855—3873 |
| [6] | Kramer S., Mielby J., Buss K., Kasama T., Kegnæs S., Chem. Cat. Chem., 2017, 9, 2930—2934 |
| [7] | Wu Q., Wang L., Jin R., Kang C., Bian Z., Du Z., Ma X., Guo H., Gao L.,Eur. J. Org. Chem., 2016, 5415—5422 |
| [8] | Kocen A. L., Brookhart M., Daugulis O., Chem. Commun., 2017, 53, 10010—10013 |
| [9] | Zhuo L. G., Yao Z. K., Yu Z. X., Org. Lett., 2013, 15, 4634—4637 |
| [10] | Stille J.K., Becker Y., J. Org. Chem., 1980, 45, 2139—2145 |
| [11] | Sergeyev S. A., Hesse M., Helvetica Chimica Acta, 2003, 86, 750—755 |
| [12] | Yamada H., Sodeoka M., Shibasaki M., J. Org. Chem., 1991, 56, 4569—4574 |
| [13] | Zacuto M. J., Xu F., J. Org. Chem., 2007, 72, 6298—6300 |
| [14] | Krompiec S., Pigulla M., Kuz'nik N., Krompiec M., Marciniec B., Chadyniak D., Kasperczyk J., J. Mol. Cat. A: Chem., 2005, 225, 91—101 |
| [15] | Krompiec S., Pigulla M., Bieg T., Szczepankiewicz W., Kuz'nik N., Krompiec M., Kubicki M., J. Mol. Cat. A: Chem., 2002, 189, 169—185 |
| [16] | Krompiec S., Pigulla M., Szczepankiewicz W., Bieg T., Kuznik N., Leszczynska-Sejda K., Kubicki M., Borowiak T., Tetrahedron Lett., 2001, 42, 7095—7098 |
| [17] | Neugnot B., Cintrat J. C., Rousseau B., Tetrahedron, 2004, 60, 3575—3579 |
| [18] | Nakanishi S., Otsuji Y., Itoh K., Hayashi N., Bull. Chem. Soc. Jpn., 1990, 63, 3595—3600 |
| [19] | Couture A., Deniau E., Grandclaudon P., Lebrun S., Tetrahedron Lett., 1996, 37, 7749—7752 |
| [20] | Naito T., Yuumoto Y., Kiguchi T., Ninomiya I., J. Chem. Soc., Perkin Trans., 1996, 1, 281—288 |
| [21] | Wang L., Liu C., Bai R., Pan Y., Lei A., Chem. Commun., 2013, 49, 7923—7925 |
| [22] | Kozuch S., Shaik S., J. Am. Chem. Soc., 2017, 11, 4075—4086 |
| [23] | Zhang X., Tutkowski B., Oliver A., Helquist P., Wiest O., ACS Catal., 2018, 8, 1740—1747 |
| [24] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., Nakatsuji H., Caricato M., Li X., Hratchian H. P., Izmaylov A. F., Bloino J., Zheng G., Sonnenberg J. L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J. A. Jr., Peralta J. E., Ogliaro F., Bearpark M., Heyd J. J., Brothers E., Kudin K. N., Staroverov V. N., Keith T., Kobayashi R., Normand J., Raghavachari K., Rendell A., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Rega N., Millam J. M., Klene M., Knox J. E., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Martin R. L., Morokuma K., Zakrzewski V. G., Voth G. A., Salvador P., Dannenberg J. J., Dapprich S., Daniels A. D., Farkas O., Foresman J. B., Ortiz J. V., Cioslowski J., Fox D. J., Gaussian 09, Revision E. 01, Gaussian Inc., Wallingford CT, 2013 |
| [25] | Becke A. D., Phys. Rev.A, 1988, 38, 3098—3100 |
| [26] | Lee C., Yang W., Parr R. G., Phys. Rev. B, 1988, 37, 785—789 |
| [27] | Becke A. D., J. Chem.Phys., 1993, 98, 5648—5652 |
| [28] | Hay P. J., Wadt W. R., J. Chem. Phys., 1985, 82, 270—283 |
| [29] | Hay P. J., Wadt W. R., J. Chem. Phys., 1985, 82, 299—310 |
| [30] | Ehlers A. W., Böhme M., Dapprich S., Gobbi A., Höllwarth A., Jonas V., Köhler K. F., Stegmann R., Veldkamp A., Frenking G., Chem. Phys. Lett., 1993, 208, 111—114 |
| [31] | Fukui K., Acc. Chem.Res., 1981, 14, 363—368 |
| [32] | Zhao Y., Truhlar D. G., J. Chem. Phys., 2006, 125, 194101 |
| [33] | Dolg M., Wedig U., Stoll H., Preuss H., J. Chem.Phys., 1987, 86, 866—872 |
| [34] | Andrae D., Haeussermann U., Dolg M., Stoll H., Preuss H., Theor. Chem. Acc., 1990, 77, 123—141 |
| [35] | Marenich A. V., Cramer C. J., Truhlar D. G., J. Phys. Chem. B, 2009, 113, 6378—6396 |
| [36] | Kozuch S., Shaik S., Acc. Chem. Res., 2011, 44, 101—110 |
| [37] | Xie H., Kuang J., Wang L., Li Y., Huang L., Fan T., Lei Q., Fang W., Organometallics, 2017, 36, 3371—3381 |
| [1] | 张咪, 田亚锋, 高克利, 侯华, 王宝山. 三氟甲基磺酰氟绝缘介质理化特性的分子动力学模拟[J]. 高等学校化学学报, 2022, 43(11): 20220424. |
| [2] | 刘洋, 李旺昌, 张竹霞, 王芳, 杨文静, 郭臻, 崔鹏. Sc3C2@C80与[12]CPP纳米环之间非共价相互作用的理论研究[J]. 高等学校化学学报, 2022, 43(11): 20220457. |
| [3] | 王思佳 侯璐 李成龙 李文翠 陆安慧. 空腔型纳米炭的制备与应用[J]. 高等学校化学学报, 0, (): 20220637. |
| [4] | 武晴滢, 祝震予, 吴剑鸣, 徐昕. 泛Kennard-Stone算法的数据集代表性度量与分块采样策略[J]. 高等学校化学学报, 2022, 43(10): 20220397. |
| [5] | 王园月, 安梭梭, 郑旭明, 赵彦英. 5-巯基-1, 3, 4-噻二唑-2-硫酮微溶剂团簇的光谱和理论计算研究[J]. 高等学校化学学报, 2022, 43(10): 20220354. |
| [6] | 张伶育, 张继龙, 曲泽星. RDX分子内振动能量重分配的动力学研究[J]. 高等学校化学学报, 2022, 43(10): 20220393. |
| [7] | 沈琦 陈海瑶 高登辉 赵 熹 那日松 刘佳 黄旭日. 天然产物法卡林二醇与人类 GABAA 受体的相互作用机制研究[J]. 高等学校化学学报, 0, (): 0. |
| [8] | 陈少臣 程敏 王诗慧 吴金奎 罗磊 薛小雨 吉旭 张长春 周利. 预测金属有机骨架的甲烷和氢气输送能力的迁移学习建模[J]. 高等学校化学学报, 0, (): 20220459. |
| [9] | 彭辛哲, 葛娇阳, 王访丽, 余国静, 冉雪芹, 周栋, 杨磊, 解令海. 一种基于苯并噻吩平面格的张力与重组能的理论研究[J]. 高等学校化学学报, 0, (): 20220313. |
| [10] | 郭程, 张威, 唐云. 有序介孔材料: 历史、 现状与发展趋势[J]. 高等学校化学学报, 2022, 43(8): 20220167. |
| [11] | 汤乔伟 蔡小青 李江 诸颖 王丽华 田阳 樊春海 胡钧. 同步辐射X射线成像技术在脑成像研究中的应用[J]. 高等学校化学学报, 0, (): 20220379. |
| [12] | 杨丹, 刘旭, 戴翼虎, 祝艳, 杨艳辉. 金团簇电催化二氧化碳还原反应的研究进展[J]. 高等学校化学学报, 2022, 43(7): 20220198. |
| [13] | 戴卫, 侯华, 王宝山. 七氟异丁腈负离子结构与反应活性的理论研究[J]. 高等学校化学学报, 2022, 43(6): 20220044. |
| [14] | 施耐克, 张娅, SANSON Andrea, 王蕾, 陈骏. Zn(NCN)单轴的负热膨胀性及机理研究[J]. 高等学校化学学报, 2022, 43(6): 20220124. |
| [15] | 任娜娜, 薛洁, 王治钒, 姚晓霞, 王繁. 热力学数据对1, 3-丁二烯燃烧特性的影响[J]. 高等学校化学学报, 2022, 43(6): 20220151. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||