

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (1): 149.doi: 10.7503/cjcu20140603
• Physical Chemistry • Previous Articles Next Articles
MA Peng1, SONG Jinou1,*( ), SONG Chonglin1, LÜ Gang1, CHEN Chaoxu1, YANG Chuanwang2
), SONG Chonglin1, LÜ Gang1, CHEN Chaoxu1, YANG Chuanwang2
Received:2014-07-01
Revised:2014-12-23
Online:2015-01-10
Published:2014-12-23
Contact:
SONG Jinou
E-mail:songjinou@tju.edu.cn
Supported by:CLC Number:
TrendMD:
MA Peng, SONG Jinou, SONG Chonglin, LÜ Gang, CHEN Chaoxu, YANG Chuanwang. Effect of H-atom Abstraction Reactions Among C2H3, C2H5OH and CH3HCO on the Combustion of Ethanol-Hydrocarbon Fuels†[J]. Chem. J. Chinese Universities, 2015, 36(1): 149.
| Path | Reaction | A | n | E/(J·mol-1) |
|---|---|---|---|---|
| 1 | C2H3+C2H5OH=C2H4+C2H5O | 2.61×10-1 | 3.93 | 5410 |
| 2 | C2H3+C2H5OH=C2H4+CH3CHOH | 3.99×10-1 | 3.82 | 19960 |
| 3 | C2H3+C2H5OH=C2H4+C2H4OH | 1.18×10-1 | 4.13 | 37150 |
| 4 | C2H3+CH3HCO=C2H4+CH3CO | 9.32×10-1 | 3.74 | 5060 |
| 5 | C2H3+CH3CO =C2H4+CH2HCO | 4.12×10-1 | 3.83 | 16800 |
Table 1 Rate coefficient of the new reactions[k=ATnexp(-E/RT)]
| Path | Reaction | A | n | E/(J·mol-1) |
|---|---|---|---|---|
| 1 | C2H3+C2H5OH=C2H4+C2H5O | 2.61×10-1 | 3.93 | 5410 |
| 2 | C2H3+C2H5OH=C2H4+CH3CHOH | 3.99×10-1 | 3.82 | 19960 |
| 3 | C2H3+C2H5OH=C2H4+C2H4OH | 1.18×10-1 | 4.13 | 37150 |
| 4 | C2H3+CH3HCO=C2H4+CH3CO | 9.32×10-1 | 3.74 | 5060 |
| 5 | C2H3+CH3CO =C2H4+CH2HCO | 4.12×10-1 | 3.83 | 16800 |
| Pathway | k/s-1 | |||
|---|---|---|---|---|
| C2H3 | CH3 | OH | H | |
| a | 6.81×1010 | 5.39×1010 | 2.60×1012 | 2.04×1011 |
| b | 1.03×1010 | 2.61×1011 | 1.31×1012 | 1.64×1012 |
| c | 3.09×109 | 2.39×1011 | 1.05×1012 | 1.67×1012 |
| d | 8.42×1010 | 7.52×1010 | 4.75×1011 | 9.18×1011 |
| e | 1.50×1010 | 3.47×1010 | 2.47×1012 | 1.54×1013 |
Table 2 Reaction rate constants comparison with other intermediates in similar reactions at 1000 K*
| Pathway | k/s-1 | |||
|---|---|---|---|---|
| C2H3 | CH3 | OH | H | |
| a | 6.81×1010 | 5.39×1010 | 2.60×1012 | 2.04×1011 |
| b | 1.03×1010 | 2.61×1011 | 1.31×1012 | 1.64×1012 |
| c | 3.09×109 | 2.39×1011 | 1.05×1012 | 1.67×1012 |
| d | 8.42×1010 | 7.52×1010 | 4.75×1011 | 9.18×1011 |
| e | 1.50×1010 | 3.47×1010 | 2.47×1012 | 1.54×1013 |
| Mixture | Equivalence ratio | Flow rate/(L·min-1) | Inlet temperature/K | Pressure/Pa |
|---|---|---|---|---|
| n-C7H16/O2/Ar | 1.0 | 0.0914/1/0.8 | 498 | 4000 |
| n-C7H16/C2H5OH/O2/Ar | 1.0 | 0.065/0.14/1/0.8 | 473 | 4000 |
| i-C8H18/O2/Ar | 1.0 | 0.127/1.574/0.8 | 498 | 4000 |
| i-C8H18/C2H5OH/O2/Ar | 1.0 | 0.089/0.154/1.574/0.8 | 473 | 4000 |
Table 3 Experimental parameters
| Mixture | Equivalence ratio | Flow rate/(L·min-1) | Inlet temperature/K | Pressure/Pa |
|---|---|---|---|---|
| n-C7H16/O2/Ar | 1.0 | 0.0914/1/0.8 | 498 | 4000 |
| n-C7H16/C2H5OH/O2/Ar | 1.0 | 0.065/0.14/1/0.8 | 473 | 4000 |
| i-C8H18/O2/Ar | 1.0 | 0.127/1.574/0.8 | 498 | 4000 |
| i-C8H18/C2H5OH/O2/Ar | 1.0 | 0.089/0.154/1.574/0.8 | 473 | 4000 |
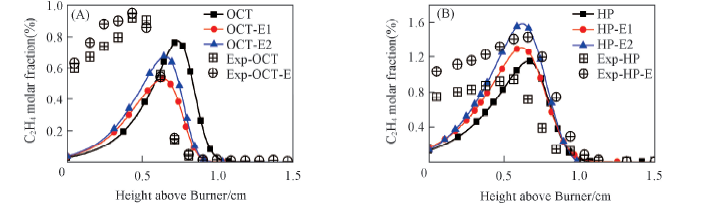
Fig.4 C2H4 molar fraction in the flames compared with experimental data (A) Octane flame and octane/ethanol flame; (B) heptane flame and heptane/ethanol flame.
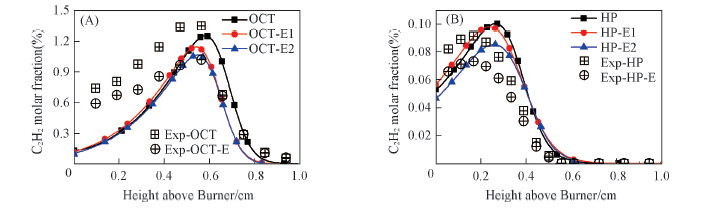
Fig.5 C2H2 molar fraction in the flames compared with experimental data(A) Octane flame and octane/ethanol flame; (B) heptane flame and heptane/ethanol flame.
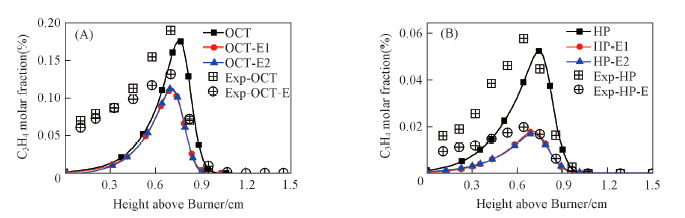
Fig.6 C3H4 molar fraction in the flames compared with experimental data(A) Octane flame and octane/ethanol flame; (B) heptane flame and heptane/ethanol flame.
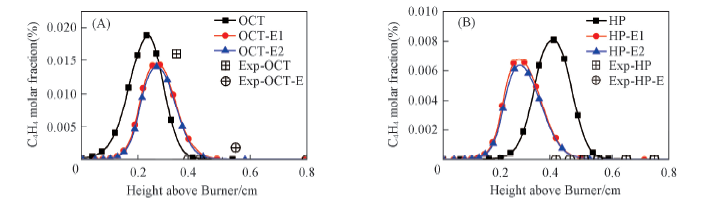
Fig.7 C4H4 molar fraction in the flames compared with experimental data (A) Octane flame and octane/ethanol flame; (B) heptane flame and heptane/ethanol flame.
| Species | r(C2H2) | r(C2H4) | r(C3H4) | r(C4H4) | ||||
|---|---|---|---|---|---|---|---|---|
| Heptane | Octane | Heptane | Octane | Heptane | Octane | Heptane | Octane | |
| Mech-3(path 3) | 1.34 | 1.12 | 0.92 | 0.56 | 0.91 | 0.84 | 0.93 | 0.90 |
| Mech-2(path 2) | 1.16 | 1.04 | 1.11 | 0.71 | 0.86 | 0.86 | 0.91 | 0.87 |
| Mech-1(path 1) | 1.10 | 0.93 | 1.22 | 0.84 | 0.90 | 0.93 | 0.88 | 0.91 |
| Expt. | 1.10[ | 0.76[ | 1.00[ | 0.78[ | 0.80[ | 0.97[ | 1.10[ | 0.86[ |
Table 4 Peak concentration ratio(r) on these species in different flames*
| Species | r(C2H2) | r(C2H4) | r(C3H4) | r(C4H4) | ||||
|---|---|---|---|---|---|---|---|---|
| Heptane | Octane | Heptane | Octane | Heptane | Octane | Heptane | Octane | |
| Mech-3(path 3) | 1.34 | 1.12 | 0.92 | 0.56 | 0.91 | 0.84 | 0.93 | 0.90 |
| Mech-2(path 2) | 1.16 | 1.04 | 1.11 | 0.71 | 0.86 | 0.86 | 0.91 | 0.87 |
| Mech-1(path 1) | 1.10 | 0.93 | 1.22 | 0.84 | 0.90 | 0.93 | 0.88 | 0.91 |
| Expt. | 1.10[ | 0.76[ | 1.00[ | 0.78[ | 0.80[ | 0.97[ | 1.10[ | 0.86[ |
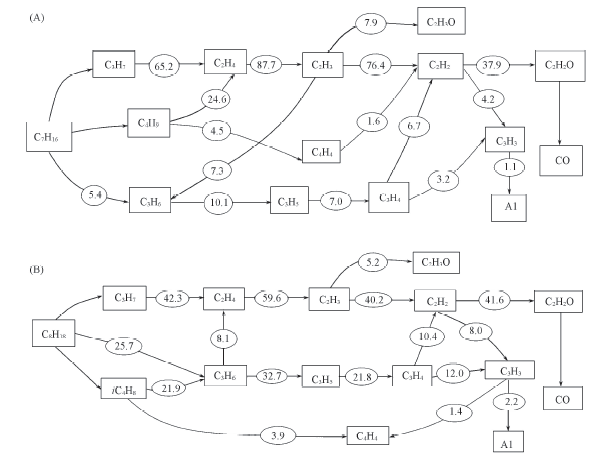
Fig.8 Pathways in n-heptane/O2/Ar(A) and iso-octane/O2/Ar(B) flames Numerical values correspond to the percent contribution to the fuel carbon conversion.
| [1] | Lemaire R., Therssen E., Desgroux P., Fuel , 2010, 89(12), 3952—3959 |
| [2] | Xing Y., Yao M. F., Zhang F. G., Zheng Z. Q., Transactions of CSISE, 2007, 25(1), 24—29 |
| (邢元, 尧命发, 张福根, 郑尊清. 内燃机学报, 2007, 25(1), 24—29) | |
| [3] | Dong S. R., Song C. L., Zhao C. P., Zhang Y. F., Guo Z. P., Xu G. P., Journal of Tianjin University, 2006, 39(1), 68—72 |
| (董素荣, 宋崇林, 赵昌普, 张延峰, 郭振鹏, 徐冠鹏. 天津大学学报, 2006, 39(1), 68—72) | |
| [4] | Song J. O., Yao C. D., Xu H. J., Liu S. Y., Journal of Engineering Thermophysics, 2009, 30(8), 1434—1436 |
| (宋金瓯, 姚春德, 许汉君, 刘士钰. 工程热物理学报, 2009, 30(8), 1434—1436) | |
| [5] | Douté C., Delfau J. L., Akrich R., Vovelle C., Combustion Science and Technology , 1997, 124(1—6), 249—276 |
| [6] | Li M. D., Wang Z., Li R. N., Zhao Y., Liu S., Qu L., Journal of Engineering Thermophysics, 2013, 34(8), 1578—1581 |
| (李铭迪, 王忠, 李瑞娜, 赵洋, 刘帅, 瞿磊. 工程热物理学报, 2013, 34(8), 1578—1581) | |
| [7] | Wu J. T., Song K. H., Litzinger T., Lee S. Y., Santoro R., Linevsky M., Colket M., Liscinsky D., Combustion and Flame , 2006, 144(4), 675—687 |
| [8] | Song K. H., Nag P., Litzinger T., Haworth D. C., Combustion and Flame , 2003, 135(3), 341—349 |
| [9] | Curran H. J., Fisher E. M., Glaude P. A., Marinov N. M., Pitz W. J., Westbrook C. K., Flynn P. F., Durrett R. P., Loye A. O., Akinyemi O. C., Dryer F. L., Sae Transactions, 2001, 110(4), 514—521 |
| [10] | Tautermann C. S., Wellenzohn B., Clary D. C., Molecular Physics, 2006, 104(1), 151—158 |
| [11] | Xie H. B., Ding Y. H., Sun C. C., Journal of Physical Chemistry A , 2005, 109(37), 8419—8423 |
| [12] | Computational Chemistry Comparison and Benchmark Database, |
| [13] | Marchal C., Delfau J. L., Vovelle C., Moréac G., Mauss F., Proceedings of the Combustion Institute , 2009, 32(1), 753—759 |
| [14] | Seiser R., Pitsch H., Seshadri K., Pitz W. J., Gurran H. J., Proceedings of the Combustion Institute , 2000, 28(2), 2029—2037 |
| [15] | Marinov N. M., Int. J. Chem. Kinet., 1999, 31183—220 |
| [16] | Yao C., Xu G., Xu H., Fuel , 2013, 111131—139 |
| [17] | Yuan T., Experimental and Kinetic Modeling Studies on Pyrolysis and Premixed Flames of n-Heptane and iso-Octane, University of Science and Technology of China, Hefei, 2010 |
| (袁涛. 正庚烷、异辛烷热解和预混火焰的实验及动力学模型研究, 合肥: 中国科学技术大学, 2010) | |
| [18] | Kee R.J., Rupley F. M., Miller J. A., Coltrin M. E., Grcar J. F., Meeks E., CHEMKIN PRO, Release 15112, Reaction Design, San Diego, 2011 |
| [19] | Feng L. X., Jin L. X., Wang W. N., Wang W. L., Acta Phys. Chim. Sin., 2012, 28(7), 1623—1629 |
| (冯丽霞, 靳玲侠, 王渭娜, 王文亮. 物理化学学报, 2012, 28(7), 1623—1629) | |
| [20] | Guo S., Wang W. N., Jin L. X., Wang S., Wang W. L., Chem. J. Chinese Universities, 2014, 35(6), 1300—1306 |
| (郭莎, 王渭娜, 靳玲侠, 王帅, 王文亮. 高等学校化学学报, 2014, 35(6), 1300—1306) | |
| [21] | Pople J. A., Head-Gordon M., Raghavachari K., J. Chem. Phys., 1987, 875968—5975 |
| [22] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., Nakatsuji H., Caricato M., Li X., Hratchian H. P., Izmaylov A. F., Bloino J., Zheng G., Sonnenberg J. L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J. A. Jr., Peralta J. E., Ogliaro F., Bearpark M., Heyd J. J., Brothers E., Kudin K. N., Staroverov V. N., KobayashiR., Normand J., Raghavachari K., Rendell A., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Rega N., Millam N. J., KleneM., Knox J. E., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Martin R. L., Morokuma K., Zakrzewski V. G., Voth G. A., Salvador P., Dannenberg J. J., Dapprich S., Daniels A. D., Farkas Ö., Foresman J. B., Ortiz J. V., Cioslowski J., Fox D. J., Gaussian 09, Wallingford CT, Gaussian Inc., 2009 |
| [23] | Canneaux S., Bohr F., Hénon E., J. Comp. Chem., 2014, 3582—93 |
| [24] | Xu H. J., Yao C. D., Xu G. L., Wang Z. D., Jin H. F., Journal of Engineering Thermophysics, 2012, 33(10), 1803—1806 |
| 许汉君, 姚春德, 徐广兰, 王占东, 金汉锋. 工程热物理学报, 2012, 33(10), 1803—1806) | |
| [25] | Bakali A. E., Delfau J. L., Vovelle C., Combustion and Flame , 1999, 118(3), 381—398 |
| [26] | Li Y. R., Pei Y. Q., Qin J., Zhang M., Acta Phys. Chim. Sin, 2014, 30(6), 1017—1026 |
| (李艳荣, 裴毅强, 秦静, 张淼. 物理化学学报, 2014, 30(6), 1017—1026) |
| [1] | WANG Jian, ZHANG Hongxing. Theoretical Study on the Structural-photophysical Relationships of Tetra-Pt Phosphorescent Emitters [J]. Chem. J. Chinese Universities, 2021, 42(7): 2245. |
| [2] | LI Xiangyuan,YAO Xiaoxia,SHENTU Jiangtao,SUN Xiaohui,LI Juanqin,LIU Mingxia,XU Shimin. Combustion Reaction Mechanism Construction by Two-parameter Rate Constant Method † [J]. Chem. J. Chinese Universities, 2020, 41(3): 512. |
| [3] | WANG Ning,ZHU Huifang,WANG Lu,ZHANG Tiantian,GU Jiali,SHU Jie. Structural Identification and Asymmetric-exchange Dynamics Study of Esomeprazole Magnesium in Specific Solution as Probed by Using 1H NMR Spectra† [J]. Chem. J. Chinese Universities, 2018, 39(9): 1919. |
| [4] | LI Yingli, WANG Jingbo, LI Xiangyuan. Kinetic Mechanism Study on Low Temperature for Decalin Combustion† [J]. Chem. J. Chinese Universities, 2018, 39(6): 1212. |
| [5] | FANG Sheng, LIU Jingjing, DUAN Xuemei, TAO Fuming, LIU Jingyao. Ab initio Calculation and Kinetic Investigation of Monacid-catalyzed Decomposition of Sulfurous Acid [J]. Chem. J. Chinese Universities, 2017, 38(8): 1390. |
| [6] | MA Qian, WANG Weina, ZHAO Qiangli, LIU Fengyi, WANG Wenliang. Theoretical Studies on the Reaction Mechanism of Criegee Intermediates RCHOO(R=H, CH3) with NCO Radical† [J]. Chem. J. Chinese Universities, 2017, 38(4): 613. |
| [7] |
WANG Rui, LI Yili, FENG Xukai, SONG Liang, ZHANG Tianlei, WANG Zhuqing, JIN Lingxia, ZHANG Qiang, XU Qiong, WANG Zhiyin.
Catalytic Effect of n(H2O)(n=1,2) on the Reaction of HO2+NO |
| [8] | GAO Zhifang, WANG Weina, MA Qian, LIU Fengyi, WANG Wenliang. Theoretical Studies on the Reaction Mechanism of Criegee Intermediates CH3CHOO with OH Radicals† [J]. Chem. J. Chinese Universities, 2016, 37(3): 513. |
| [9] | ZHU Peng, DUAN Xuemei, LIU Jingyao. Mechanism and Kinetics of the Hydrogen-abstraction Reaction of CF2ClC(O)OCH2CH3 with OH Radicals† [J]. Chem. J. Chinese Universities, 2016, 37(1): 79. |
| [10] | WANG Kuan, CHEN Jiangang, WANG Bozhou, LU Jian, WANG Wenliang, LIU Fengyi, ZHOU Cheng, LIAN Peng, LIU Zhongwen, LIU Zhaotie. Mechanisms and Kinetics of the Synthesis of FOX-12† [J]. Chem. J. Chinese Universities, 2015, 36(3): 531. |
| [11] | HU Xixi, YANG Junying, XIE Daiqian. State-to-state Quantum Dynamics of Reaction N+NH→N2+H [J]. Chem. J. Chinese Universities, 2015, 36(11): 2198. |
| [12] | LI Yue, FANG Decai. Density Functional Theory Studies on the t-Butoxyl Radical Mediated Hydrogen Atom Transfer Reactions† [J]. Chem. J. Chinese Universities, 2015, 36(10): 1954. |
| [13] | GUO Sha, WANG Weina, JIN Lingxia, WANG Shuai, WANG Wenliang. Mechanistic Studies on CH3CH2O+HCHO Reaction and Rate Constants of Major Channel† [J]. Chem. J. Chinese Universities, 2014, 35(6): 1300. |
| [14] | LI Yingli, NING Hongbo, ZHU Quan, LI Xiangyuan. Kinetics Simulation of Ethylbenzene Pyrolysis Under Supercritical Pressure† [J]. Chem. J. Chinese Universities, 2014, 35(3): 576. |
| [15] | JIN Tong-Yin, WANG Qin, LIU Jing-Yao. Mechanism and Kinetics for Reaction of CF3CH2CF2CH3(HFC-365mfc) with Cl Atom [J]. Chem. J. Chinese Universities, 2013, 34(3): 641. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||