

Chem. J. Chinese Universities ›› 2015, Vol. 36 ›› Issue (3): 531.doi: 10.7503/cjcu20140873
• Physical Chemistry • Previous Articles Next Articles
WANG Kuan1,2, CHEN Jiangang1,2,*( ), WANG Bozhou3, LU Jian3, WANG Wenliang2, LIU Fengyi2, ZHOU Cheng3, LIAN Peng3, LIU Zhongwen1,2, LIU Zhaotie1,2
), WANG Bozhou3, LU Jian3, WANG Wenliang2, LIU Fengyi2, ZHOU Cheng3, LIAN Peng3, LIU Zhongwen1,2, LIU Zhaotie1,2
Received:2014-09-26
Online:2015-03-10
Published:2015-01-30
Contact:
CHEN Jiangang
E-mail:jgchen@snnu.edu.cn
Supported by:CLC Number:
TrendMD:
WANG Kuan, CHEN Jiangang, WANG Bozhou, LU Jian, WANG Wenliang, LIU Fengyi, ZHOU Cheng, LIAN Peng, LIU Zhongwen, LIU Zhaotie. Mechanisms and Kinetics of the Synthesis of FOX-12†[J]. Chem. J. Chinese Universities, 2015, 36(3): 531.
| Route | Species | ZPE/ (kJ·mol-1) | ΔG(298 K)/ (kJ·mol-1) | ΔH(298 K)/ (kJ·mol-1) | (ΔE+ZPE)/(kJ·mol-1) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| B3LYP | MP2 | |||||||||
| A | NH2S | 234.11 | 0 | 0 | 0 | 0 | ||||
| A-IM1+N | 236.58 | -617.13 | -659.09 | -658.12 | -579.15 | |||||
| A-TS1+N | 235.83 | -527.28 | -574.70 | -571.52 | -517.61 | |||||
| A-IM2+N | 238.95 | -662.48 | -693.19 | -693.50 | -615.60 | |||||
| A1-IM3+SO3 | 242.54 | -1231.37 | -1307.64 | -1306.55 | -1161.46 | |||||
| A1-TS2+SO3 | 234.08 | -1163.15 | -1249.01 | -1245.34 | -1126.87 | |||||
| A1-IM4+SO3 | 242.56 | -1270.19 | -1352.04 | -1349.98 | -1220.69 | |||||
| A2-IM3+SO3+HS | 234.47 | -695.28 | -720.00 | -719.45 | -608.41 | |||||
| A2-TS2+SO3+HS | 222.85 | -548.53 | -578.98 | -576.74 | -459.56 | |||||
| A2-IM4+SO3+HS | 235.61 | -667.95 | -701.59 | -698.96 | -555.50 | |||||
| B | B-IM1+N | 239.72 | -691.83 | -783.65 | -781.62 | -694.39 | ||||
| B-TS1+N | 239.06 | -595.52 | -691.98 | -688.35 | -629.50 | |||||
| B-IM2+N | 247.11 | -794.49 | -886.47 | -883.68 | -812.49 | |||||
| B-IM3+H2SO4 | 243.02 | -1221.70 | -1309.81 | -1306.36 | -1174.61 | |||||
| B-TS2+H2SO4 | 239.73 | -1146.81 | -1238.48 | -1234.21 | -1132.00 | |||||
| B-IM4+H2SO4 | 240.14 | -1250.95 | -1323.35 | -1322.95 | -1192.61 | |||||
| HDN+SO3+H2SO4 | 237.91 | -1279.82 | -1315.89 | -1312.71 | -1167.02 | |||||
| HDN→FOX-12 | R+HDN | 379.06 | 0 | 0 | 0 | 0 | ||||
| Route | Species | ZPE/ (kJ·mol-1) | ΔG(298 K)/ (kJ·mol-1) | ΔH(298 K)/ (kJ·mol-1) | (ΔE+ZPE)/(kJ·mol-1) | |||||
| B3LYP | MP2 | |||||||||
| HDN→FOX-12 | IM1 | 378.82 | -30.79 | -74.93 | -76.38 | -75.23 | ||||
| TS1 | 372.62 | -34.22 | -82.20 | -82.08 | -79.37 | |||||
| IM2(FOX-12) | 379.85 | -58.76 | -103.05 | -104.68 | -96.80 | |||||
Table 1 Zero-point energies(ZPE), relative energies[ΔE and(ΔE+ZPE)], enthalpies(ΔH), and free energies(ΔG) of reactants, products, transition states, and intermediates during the synthesis process of FOX-12
| Route | Species | ZPE/ (kJ·mol-1) | ΔG(298 K)/ (kJ·mol-1) | ΔH(298 K)/ (kJ·mol-1) | (ΔE+ZPE)/(kJ·mol-1) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| B3LYP | MP2 | |||||||||
| A | NH2S | 234.11 | 0 | 0 | 0 | 0 | ||||
| A-IM1+N | 236.58 | -617.13 | -659.09 | -658.12 | -579.15 | |||||
| A-TS1+N | 235.83 | -527.28 | -574.70 | -571.52 | -517.61 | |||||
| A-IM2+N | 238.95 | -662.48 | -693.19 | -693.50 | -615.60 | |||||
| A1-IM3+SO3 | 242.54 | -1231.37 | -1307.64 | -1306.55 | -1161.46 | |||||
| A1-TS2+SO3 | 234.08 | -1163.15 | -1249.01 | -1245.34 | -1126.87 | |||||
| A1-IM4+SO3 | 242.56 | -1270.19 | -1352.04 | -1349.98 | -1220.69 | |||||
| A2-IM3+SO3+HS | 234.47 | -695.28 | -720.00 | -719.45 | -608.41 | |||||
| A2-TS2+SO3+HS | 222.85 | -548.53 | -578.98 | -576.74 | -459.56 | |||||
| A2-IM4+SO3+HS | 235.61 | -667.95 | -701.59 | -698.96 | -555.50 | |||||
| B | B-IM1+N | 239.72 | -691.83 | -783.65 | -781.62 | -694.39 | ||||
| B-TS1+N | 239.06 | -595.52 | -691.98 | -688.35 | -629.50 | |||||
| B-IM2+N | 247.11 | -794.49 | -886.47 | -883.68 | -812.49 | |||||
| B-IM3+H2SO4 | 243.02 | -1221.70 | -1309.81 | -1306.36 | -1174.61 | |||||
| B-TS2+H2SO4 | 239.73 | -1146.81 | -1238.48 | -1234.21 | -1132.00 | |||||
| B-IM4+H2SO4 | 240.14 | -1250.95 | -1323.35 | -1322.95 | -1192.61 | |||||
| HDN+SO3+H2SO4 | 237.91 | -1279.82 | -1315.89 | -1312.71 | -1167.02 | |||||
| HDN→FOX-12 | R+HDN | 379.06 | 0 | 0 | 0 | 0 | ||||
| Route | Species | ZPE/ (kJ·mol-1) | ΔG(298 K)/ (kJ·mol-1) | ΔH(298 K)/ (kJ·mol-1) | (ΔE+ZPE)/(kJ·mol-1) | |||||
| B3LYP | MP2 | |||||||||
| HDN→FOX-12 | IM1 | 378.82 | -30.79 | -74.93 | -76.38 | -75.23 | ||||
| TS1 | 372.62 | -34.22 | -82.20 | -82.08 | -79.37 | |||||
| IM2(FOX-12) | 379.85 | -58.76 | -103.05 | -104.68 | -96.80 | |||||
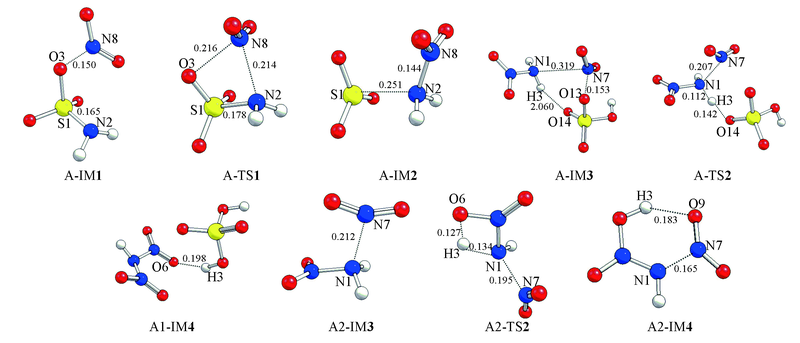
Fig.1 Optimized geometries of species of the channels in replacing-SO3 first calculated at B3LYP/6-311G(d,p) level of theory Bond length units are in nm.
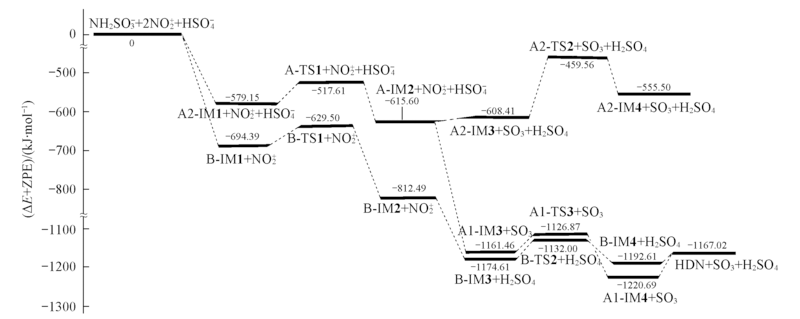
Fig.2 Energy diagram for the potential energy surface of each nitration path of NH2SO3- in mixed nitric-sulfuric acid predicted at the MP2/6-311++G(3df,3pd)//B3LYP/6-311G(d,p) level of theory
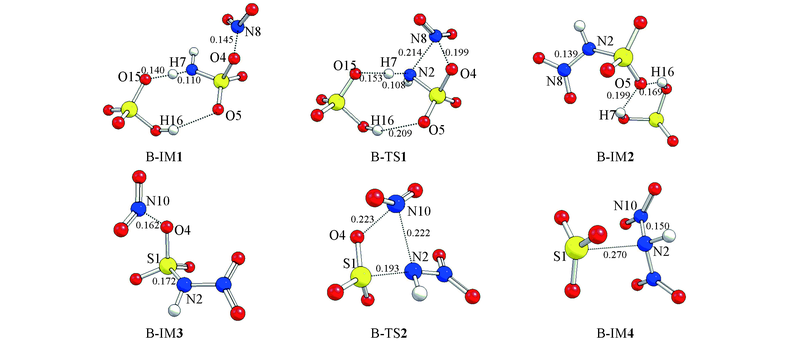
Fig.3 Optimized geometries of species of priority to replacing H channels calculated at B3LYP/6-311G(d,p) level of theory Bond length units are in nm.
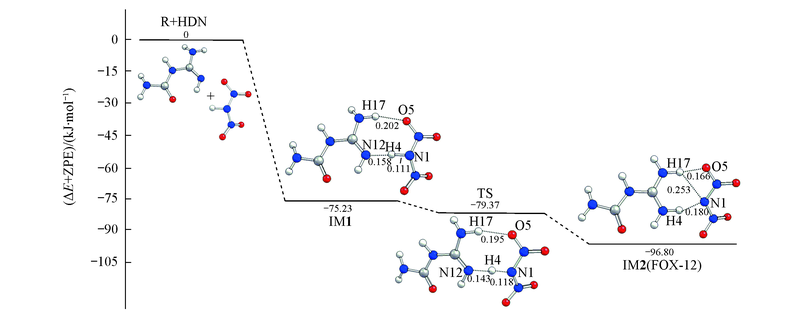
Fig.5 Schematic energy diagram for the potential energy surface of the formation of FOX-12, which directly synthesized from HDN, obtained at the MP2/6-311++G(3df,3pd)//B3LYP/6-311G(d,p) level of theory Bond length units are in nm.
| T/K | |||||
|---|---|---|---|---|---|
| 200 | 6.56×10-5 | 4.64×10-3 | 1.01×10-10 | 1.09×10-5 | 5.98×1011 |
| 220 | 1.13×10-3 | 5.08×10-2 | 9.87×10-10 | 7.39×10-4 | 5.23×1011 |
| 233 | 5.38×10-3 | 1.92×10-1 | 3.67×10-9 | 2.87×10-3 | 4.82×1011 |
| 255 | 5.43×10-2 | 1.32×100 | 2.65×10-8 | 2.21×10-2 | 4.25×1011 |
| 265 | 1.39×10-1 | 2.85×100 | 6.00×10-8 | 5.11×10-2 | 4.03×1011 |
| 270 | 2.17×10-1 | 4.09×100 | 8.87×10-8 | 7.61×10-2 | 3.94×1011 |
| 273 | 2.81×10-1 | 5.05×100 | 1.12×10-7 | 9.62×10-2 | 3.88×1011 |
| 276 | 3.63×10-1 | 6.20×100 | 1.40×10-7 | 1.21×10-1 | 3.83×1011 |
| 280 | 5.07×10-1 | 8.10×100 | 1.88×10-7 | 1.64×10-1 | 3.76×1011 |
| 285 | 7.60×10-1 | 1.12×101 | 2.71×10-7 | 2.36×10-1 | 3.68×1011 |
| 295 | 1.65×100 | 2.06×101 | 5.45×10-7 | 4.75×10-1 | 3.54×1011 |
| 300 | 2.38×100 | 2.76×101 | 7.64×10-7 | 6.64×10-1 | 3.47×1011 |
| 305 | 3.41×100 | 3.65×101 | 1.07×10-6 | 9.21×10-1 | 3.41×1011 |
| 310 | 4.83×100 | 4.79×101 | 1.47×10-6 | 1.27×100 | 3.36×1011 |
| 315 | 6.78×100 | 6.22×101 | 2.03×10-6 | 1.73×100 | 3.30×1011 |
| 320 | 9.42×100 | 8.02×101 | 2.78×10-6 | 2.33×100 | 3.25×1011 |
| 325 | 1.30×101 | 1.02×102 | 3.78×10-6 | 3.13×100 | 3.21×1011 |
| 330 | 1.77×101 | 1.30×102 | 5.11×10-6 | 4.17×100 | 3.17×1011 |
| 335 | 2.40×101 | 1.63×102 | 6.89×10-6 | 5.52×100 | 3.13×1011 |
| 340 | 3.22×101 | 2.04×102 | 9.25×10-6 | 7.25×100 | 3.09×1011 |
| 350 | 5.68×101 | 3.13×102 | 1.65×10-5 | 1.23×101 | 3.03×1011 |
| 360 | 9.74×101 | 4.67×102 | 2.88×10-5 | 2.03×101 | 2.97×1011 |
| 370 | 1.63×102 | 6.81×102 | 4.99×10-5 | 3.28×101 | 2.93×1011 |
| 380 | 2.65×102 | 9.74×102 | 8.52×10-5 | 5.19×101 | 2.89×1011 |
| 400 | 6.58×102 | 1.88×103 | 2.41×10-4 | 1.23×101 | 2.84×1011 |
Table 2 Calculated rate constants(cm3·molecule-1·s-1) of the elementary reactions during the nitration of NH2SO3- in mixed nitric-sulfuric acid at temperature ranges of 200—400 K
| T/K | |||||
|---|---|---|---|---|---|
| 200 | 6.56×10-5 | 4.64×10-3 | 1.01×10-10 | 1.09×10-5 | 5.98×1011 |
| 220 | 1.13×10-3 | 5.08×10-2 | 9.87×10-10 | 7.39×10-4 | 5.23×1011 |
| 233 | 5.38×10-3 | 1.92×10-1 | 3.67×10-9 | 2.87×10-3 | 4.82×1011 |
| 255 | 5.43×10-2 | 1.32×100 | 2.65×10-8 | 2.21×10-2 | 4.25×1011 |
| 265 | 1.39×10-1 | 2.85×100 | 6.00×10-8 | 5.11×10-2 | 4.03×1011 |
| 270 | 2.17×10-1 | 4.09×100 | 8.87×10-8 | 7.61×10-2 | 3.94×1011 |
| 273 | 2.81×10-1 | 5.05×100 | 1.12×10-7 | 9.62×10-2 | 3.88×1011 |
| 276 | 3.63×10-1 | 6.20×100 | 1.40×10-7 | 1.21×10-1 | 3.83×1011 |
| 280 | 5.07×10-1 | 8.10×100 | 1.88×10-7 | 1.64×10-1 | 3.76×1011 |
| 285 | 7.60×10-1 | 1.12×101 | 2.71×10-7 | 2.36×10-1 | 3.68×1011 |
| 295 | 1.65×100 | 2.06×101 | 5.45×10-7 | 4.75×10-1 | 3.54×1011 |
| 300 | 2.38×100 | 2.76×101 | 7.64×10-7 | 6.64×10-1 | 3.47×1011 |
| 305 | 3.41×100 | 3.65×101 | 1.07×10-6 | 9.21×10-1 | 3.41×1011 |
| 310 | 4.83×100 | 4.79×101 | 1.47×10-6 | 1.27×100 | 3.36×1011 |
| 315 | 6.78×100 | 6.22×101 | 2.03×10-6 | 1.73×100 | 3.30×1011 |
| 320 | 9.42×100 | 8.02×101 | 2.78×10-6 | 2.33×100 | 3.25×1011 |
| 325 | 1.30×101 | 1.02×102 | 3.78×10-6 | 3.13×100 | 3.21×1011 |
| 330 | 1.77×101 | 1.30×102 | 5.11×10-6 | 4.17×100 | 3.17×1011 |
| 335 | 2.40×101 | 1.63×102 | 6.89×10-6 | 5.52×100 | 3.13×1011 |
| 340 | 3.22×101 | 2.04×102 | 9.25×10-6 | 7.25×100 | 3.09×1011 |
| 350 | 5.68×101 | 3.13×102 | 1.65×10-5 | 1.23×101 | 3.03×1011 |
| 360 | 9.74×101 | 4.67×102 | 2.88×10-5 | 2.03×101 | 2.97×1011 |
| 370 | 1.63×102 | 6.81×102 | 4.99×10-5 | 3.28×101 | 2.93×1011 |
| 380 | 2.65×102 | 9.74×102 | 8.52×10-5 | 5.19×101 | 2.89×1011 |
| 400 | 6.58×102 | 1.88×103 | 2.41×10-4 | 1.23×101 | 2.84×1011 |
| [1] | Bottaro J. C., Penwell P. E., Schmitt R. J., J. Am. Chem. Soc., 1997, 119(40), 9405—9410 |
| [2] | Brill T. B., Brush P. J., Patil D. G., Combust. Flame,1993, 92(1/2), 178—186 |
| [3] | Pace M. D., J. Phys. Chem., 1994, 98(25), 6251—6257 |
| [4] | Rossi M. J., Bottaro J. C., McMillen D. F., Int. J. Chem. Kinet., 1993, 25(7), 549—570 |
| [5] | Vyazovkin S., Wight A. C., J. Phys. Chem. A,1997, 101(31), 5653—5658 |
| [6] | Ostmark H., Bemm U., Bergman H., Langlet A., Thermochim. Acta,2002, 384(1/2), 253—259 |
| [7] | Wang B. Z., Liu Q., Zhang Z. Z., Ji Y. P., Zhu C. H., Chin. J. Energ. Mater., 2004, 12(1), 38—39 |
| (王伯周, 刘愆, 张志忠, 姬月萍, 朱春华. 含能材料, 2004, 12(1), 38—39 | |
| [8] | Liu Q., Wang B. Z., Zhang Z. Z., Zhu C. H., Lian P., Chin. J. Explos. Propellants,2006, 29(1), 29—31 |
| (刘愆, 王伯周, 张志忠, 朱春华, 廉鹏. 火炸药学报, 2006, 29(1), 29—31) | |
| [9] | Venkatachalam S., Gopalakrishnan S., Ninan K. N., Propellants Explos., Pyrotech., 2004, 29(3), 178—187 |
| [10] | Zhao F. Q., Chen P., Yuan H. A., Gao S. L., Hu R. Z., Shi Q. Z., Chin. J. Chem., 2004, 22(2), 136—141 |
| [11] | Chen X. F., Yang K., Wang B. Z., J. Phys. Chem. A,2013, 117, 5007—5014 |
| [12] | Cheng K. F., Liu M. H., Yang P. H., Int. J. Quantum Chem., 2012, 112, 703—712 |
| [13] | Lian P., Lai W. P., Wang B. Z., Ge Z. X., Xue Y. Q., Int. J. Quantum Chem., 2011, 111, 2332—2339 |
| [14] | Perreault N. N., Halasz A., Thiboutot S., Ampleman G., Hawari J., Environ. Sci. Technol., 2013, 47(10), 5193—5198 |
| [15] | Carin V., Henrik S., Production of Dinitramidic Acid Salts, WO 2005070823,2005-08-04 |
| [16] | Nikolai L., Abraham L., Manufacture of Dinitramide Salts for Propellants, WO9946202,1999-09-16 |
| [17] | Frisch M.J., Trucks G. W., Schlegel H. B., Scuseria G. E., Robb M. A., Cheeseman J. R., Scalmani G., Barone V., Mennucci B., Petersson G. A., Nakatsuji H., Caricato M., Li X., Hratchian H. P., Izmaylov A. F., Bloino J., Zheng G., Sonnenberg J. L., Hada M., Ehara M., Toyota K., Fukuda R., Hasegawa J., Ishida M., Nakajima T., Honda Y., Kitao O., Nakai H., Vreven T., Montgomery J. A. Jr., Peralta J. E., Ogliaro F., Bearpark M., Heyd J. J., Brothers E., Kudin K. N., Staroverov V. N., Keith T., Kobayashi R., Normand J., Raghavachari K., Rendell A., Burant J. C., Iyengar S. S., Tomasi J., Cossi M., Rega N., Millam J. M., Klene M., Knox J. E., Cross J. B., Bakken V., Adamo C., Jaramillo J., Gomperts R., Stratmann R. E., Yazyev O., Austin A. J., Cammi R., Pomelli C., Ochterski J. W., Martin R. L., Morokuma K., Zakrzewski V. G., Voth G. A., Salvador P., Dannenberg J. J., Dapprich S., Daniels A. D., Farkas O., Foresman J. B., Ortiz J. V., Cioslowski J., Fox D. J., Gaussian 09, Revision B.01, Gaussian Inc., Wallingford CT, 2010 |
| [18] | Jin X. H., Hu B. C., Jia H. Q., Lv C. X., Chem. J. Chinese Universities,2013, 34(7), 1685—1690 |
| (金兴辉, 胡炳成, 贾欢庆, 吕春绪. 高等学校化学学报, 2013, 34(7), 1685—1690) | |
| [19] | Zhang S. L., Xiong X. F., Yu T., Wang Y. B , Wang B. Z., Ge Z. X., Zhai G. H., Li H., Chem. J. Chinese Universities,2012, 33(7), 1444—1449 |
| (张淑利, 熊贤峰, 尉涛, 王友兵, 王伯周, 葛忠学, 翟高红, 李华. 高等学校化学学报, 2012, 33(7), 1444—1449) | |
| [20] | Lee C., Yang W., Parr R. G., Phys. Rev. B,1988, 37(2), 785—789 |
| [21] | McLean A. D., Chandler G. S., J. Chem. Phys., 1980, 72(10), 5639—5648 |
| [22] | Gonzales C., Schlegel H. B., J. Chem. Phys., 1989, 90(4), 2154—2161 |
| [23] | Lu D. H., Truong T. N., Melissas V. S., Lynch G. C., Liu Y. P., Garrett B. C., Steckler R., Isaacson A. D., Rai S. N., Hancock G. C., Lauderdale J. G., Joseph T., Truhlar D. G., Comput. Phys. Commun., 1992, 71(3), 235—262 |
| [24] | Liu Y. P., Lynch T. N., Truong T. N., Lu D. H., Truhlar D. G., Garret B. C., J. Am. Chem. Soc., 1993, 115(6), 2408—2415 |
| [25] | Garrett B. C., Truhlar D. G., J. Chem. Phys., 1979, 70(4), 1593—1598 |
| [26] | Garrett B. C., Truhlar D. G., J. Am. Chem. Soc., 1979, 101(16), 4534—4548 |
| [27] | Garrett B. C., Truhlar D. G., Grev R. S., Magnuson A. W., J. Phys. Chem., 1980, 84(13), 1730—1748 |
| [28] | Zhang S.W., Truong T. N., VKLab, Version 1.0, University of Utah, Salt Lake City, 2001 |
| [29] | Fulvio C., Marina A., Giulia D. P., Maurizio S., J. Am. Chem. Soc., 1990, 112(3), 1014—1018 |
| [30] | Blackall E. L., Hughes E. D., Nature,1952, 170, 972—973 |
| [31] | Hughes E.D., Ingold C., Pearson R. B.,J. Chem. Soc., 1958, 4357—4365 |
| [32] | Blackall E.L., Hughes E. D., Ingold C.,J. Chem. Soc., 1958, 4366—4374 |
| [33] | Lee T. J., Rice J. E., J. Am. Chem. Soc., 1992, 114, 8247 |
| [34] | Swinarski A., Bialozynski G., Roczniki Chemii., 1959, 33, 907—918 |
| [35] | Parker V. D., Kar T., Bethell D., J. Org. Chem., 2013, 78(18), 9522—9525 |
| [36] | Bottaro J. C., Schmitt R. J., Penwell P. E., Ross S., Alto P., Dinitramide Salts ADN Method of Making Same, US 5254324,1993-12-26 |
| [1] | PENG Kuilin, LI Guilin, JIANG Chongyang, ZENG Shaojuan, ZHANG Xiangping. Research Progress for the Role of Electrolytes in the CO2 Electrochemical Reduction [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220238. |
| [2] | ZHOU Zixuan, YANG Haiyan, SUN Yuhan, GAO Peng. Recent Progress in Heterogeneous Catalysts for the Hydrogenation of Carbon Dioxide to Methanol [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220235. |
| [3] | YANG Dan, LIU Xu, DAI Yihu, ZHU Yan, YANG Yanhui. Research Progress in Electrocatalytic CO2 Reduction Reaction over Gold Clusters [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220198. |
| [4] | ZHOU Leilei, CHENG Haiyang, ZHAO Fengyu. Research Progress of CO2 Hydrogenation over Pd-based Heterogeneous Catalysts [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220279. |
| [5] | CHANG Yunfei, LIAO Mingyi, WEN Jiaming. Reduction Performance and Mechanism of Liquid Terminated-carboxyl Fluoroelastomers Using NaBH4/MCl x Reduction System [J]. Chem. J. Chinese Universities, 2022, 43(6): 20210835. |
| [6] | REN Nana, XUE Jie, WANG Zhifan, YAO Xiaoxia, WANG Fan. Effects of Thermodynamic Data on Combustion Characters of 1,3-Butadiene [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220151. |
| [7] | GAO Zhiwei, LI Junwei, SHI Sai, FU Qiang, JIA Junru, AN Hailong. Analysis of Gating Characteristics of TRPM8 Channel Based on Molecular Dynamics [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220080. |
| [8] | ZHANG Shiyu, HE Runhe, LI Yongbing, WEI Shijun, ZHANG Xingxiang. Fabrication of Lithium-sulfur Battery Cathode with Radiation Crosslinked Low Molecular Weight of Polyacrylonitrile and the Mechanism of Sulfur Storage [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210632. |
| [9] | BI Gening, XIAO Xiaohua, LI Gongke. Development and Validation of Multiple Physical Fields Coupling Model for Microwave-assisted Extraction [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210739. |
| [10] | SUN Cuihong, LYU Liqiang, LIU Ying, WANG Yan, YANG Jing, ZHANG Shaowen. Mechanism and Kinetics on the Reaction of Isopropyl Nitrate with Cl, OH and NO3 Radicals [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210591. |
| [11] | WEN Zhiguo, QIAO Zaiyin, TIAN Chong, MAXIM Borzov, NIE Wanli. Catalytic Activity and Reaction Mechanism of FLPs for the Reduction of Enamine [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220555. |
| [12] | CHANG Sihui, CHEN Tao, ZHAO Liming, QIU Yongjun. Thermal Degradation Mechanism of Bio-based Polybutylactam Plasticized by Ionic Liquids [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220353. |
| [13] | CHENG Yuanyuan, XI Biying. Theoretical Study on the Fragmentation Mechanism of CH3SSCH3 Radical Cation Initiated by OH Radical [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220271. |
| [14] | MENG Fanwei, GAO Qi, YE Qing, LI Chenxi. Potassium Poisoning Mechanism of Cu-SAPO-18 Catalyst for Selective Catalytic Reduction of NOx by Ammonia [J]. Chem. J. Chinese Universities, 2021, 42(9): 2832. |
| [15] | YANG Yiying, ZHU Rongxiu, ZHANG Dongju, LIU Chengbu. Theoretical Study on Gold-catalyzed Cyclization of Alkynyl Benzodioxin to 8-Hydroxy-isocoumarin [J]. Chem. J. Chinese Universities, 2021, 42(7): 2299. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||