

Chem. J. Chinese Universities ›› 2020, Vol. 41 ›› Issue (11): 2426.doi: 10.7503/cjcu20209236
Previous Articles Next Articles
WU Qian, CHENG Dan, LÜ Yun, YUAN Lin( ), ZHANG Xiaobing
), ZHANG Xiaobing
Received:2020-07-18
Online:2020-11-10
Published:2020-11-06
Contact:
YUAN Lin
E-mail:lyuan@hnu.edu.cn
Supported by:CLC Number:
TrendMD:
WU Qian, CHENG Dan, LÜ Yun, YUAN Lin, ZHANG Xiaobing. Monitoring of Peroxynitrite Variation During Liver Injury Adopting a Far Red to Near-infrared Fluorescent Probe with Large Stokes Shift[J]. Chem. J. Chinese Universities, 2020, 41(11): 2426.
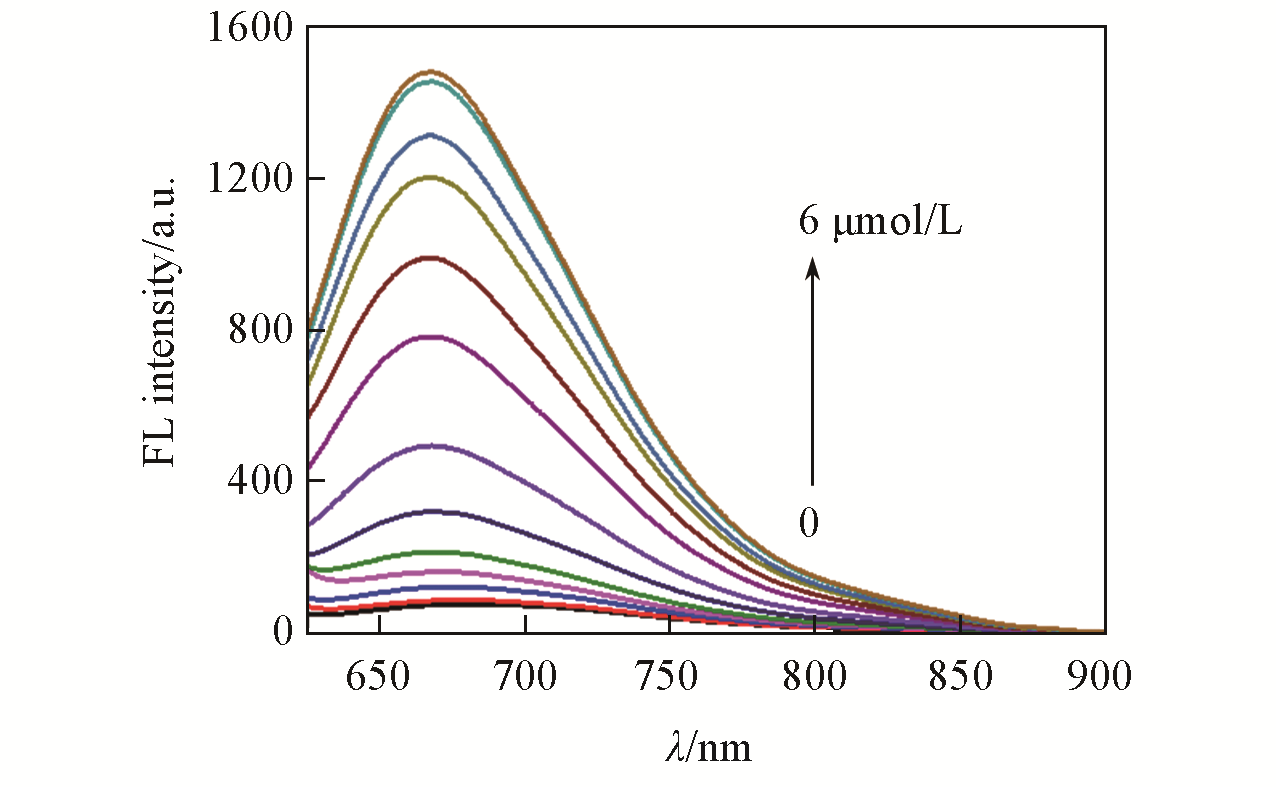
Fig.1 Fluorescence spectra of LSDQ?ONOO-(5 μmol/L) upon addition of ONOO- (0—6 μmol/L) in PBS/EtOH(volume ratio 8∶2, pH=7.4) buffer solutionnote:The mixture was kept for 20 min at 37 ℃ before the fluorescence intensity of the LSDQ?ONOO- solution was recorded. λex=580 nm.
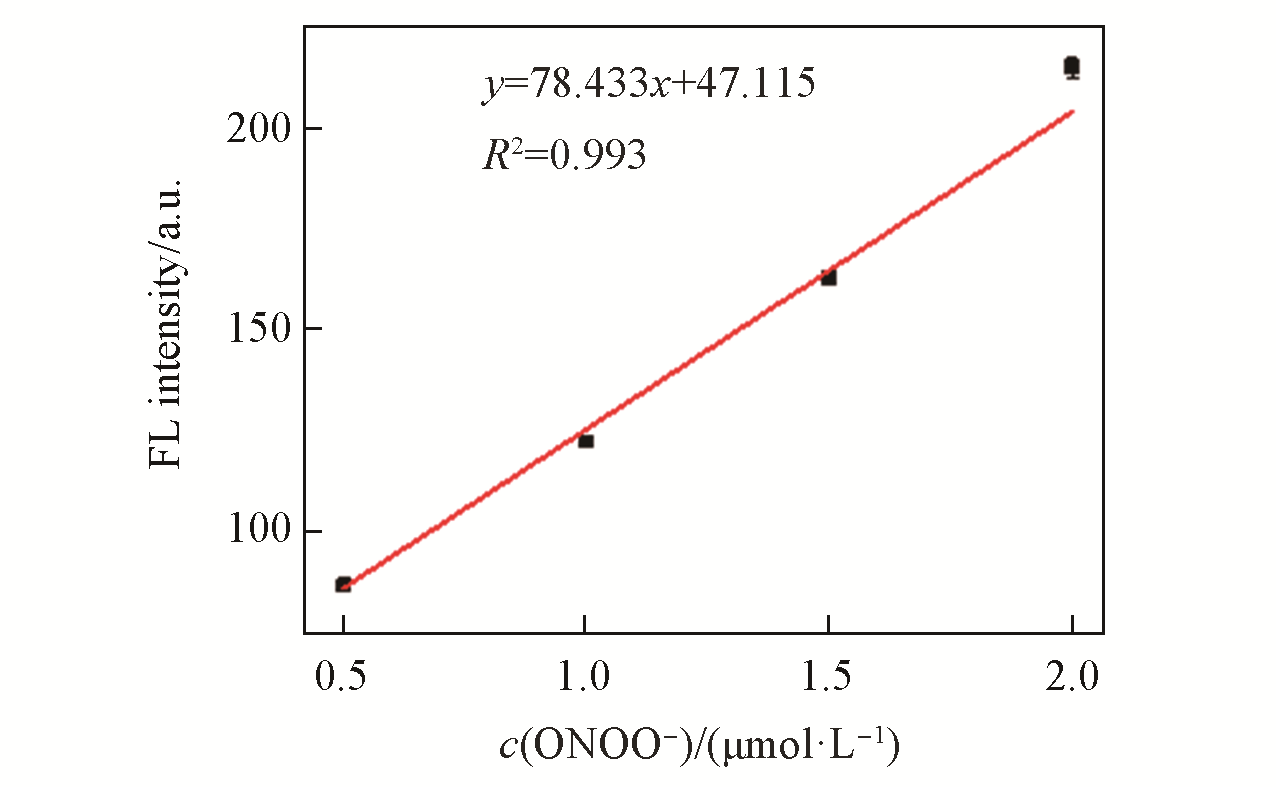
Fig.2 Linear correlation between the FL intensity of LSDQ?ONOO-(5 μmol/L) and ONOO- concentration in PBS/EtOH(volume ratio 8∶2, pH=7.4) buffer solutionnote:The mixture was kept for 20 min at 37 ℃ before the fluorescence intensity of the LSDQ?ONOO- solution was recorded. λex=580 nm.
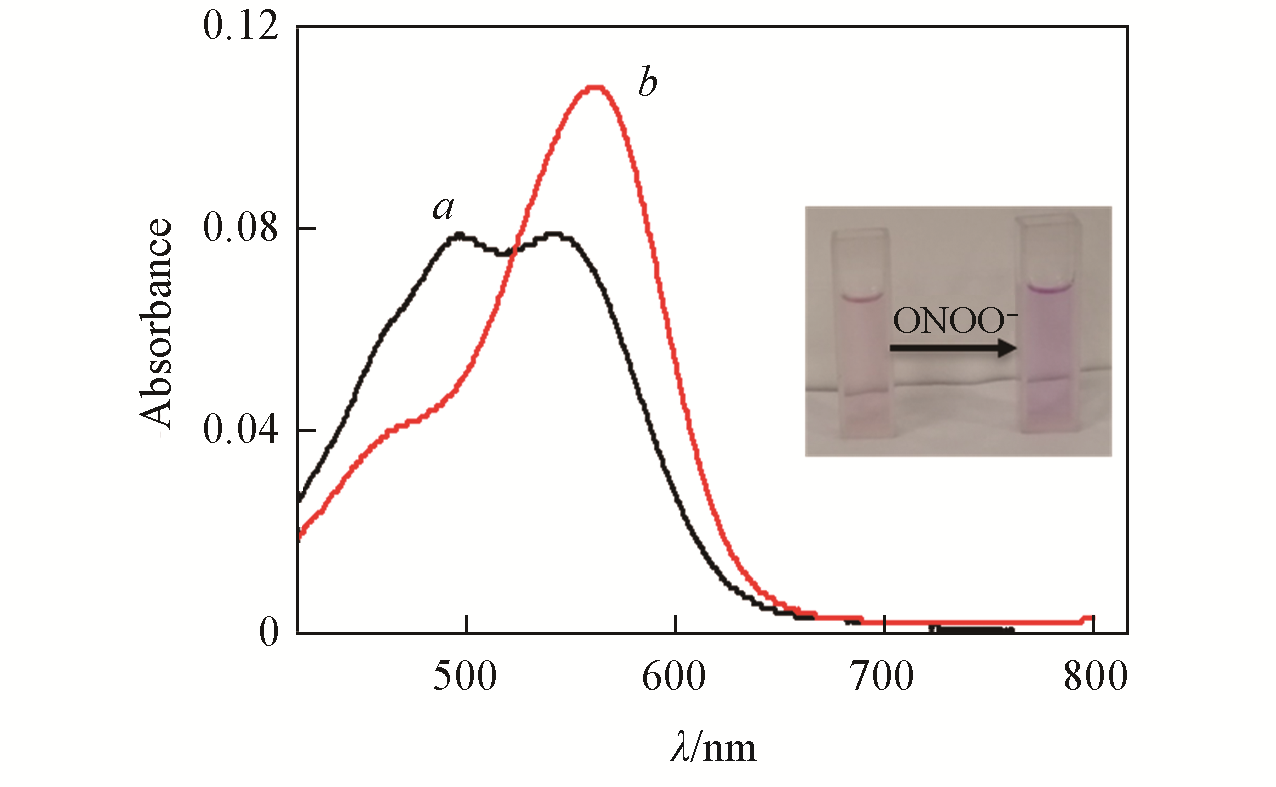
Fig.3 Absorption spectra of LSDQ?ONOO-(5 μmol/L) in the absence(a) and in the presence(b) of ONOO-(5 μmol/L) in PBS/EtOH(volume ratio 8∶2, pH=7.4) buffer solutionnote:The mixture was kept for 20 min at 37 ℃ before the absorption of the LSDQ?ONOO- solution was recorded. Inset: after reacting with ONOO?, the absorption peak of the probe shifted to 562 nm; due to the fluorescence recovery of the probe, the solution color changed from light red to dark red.
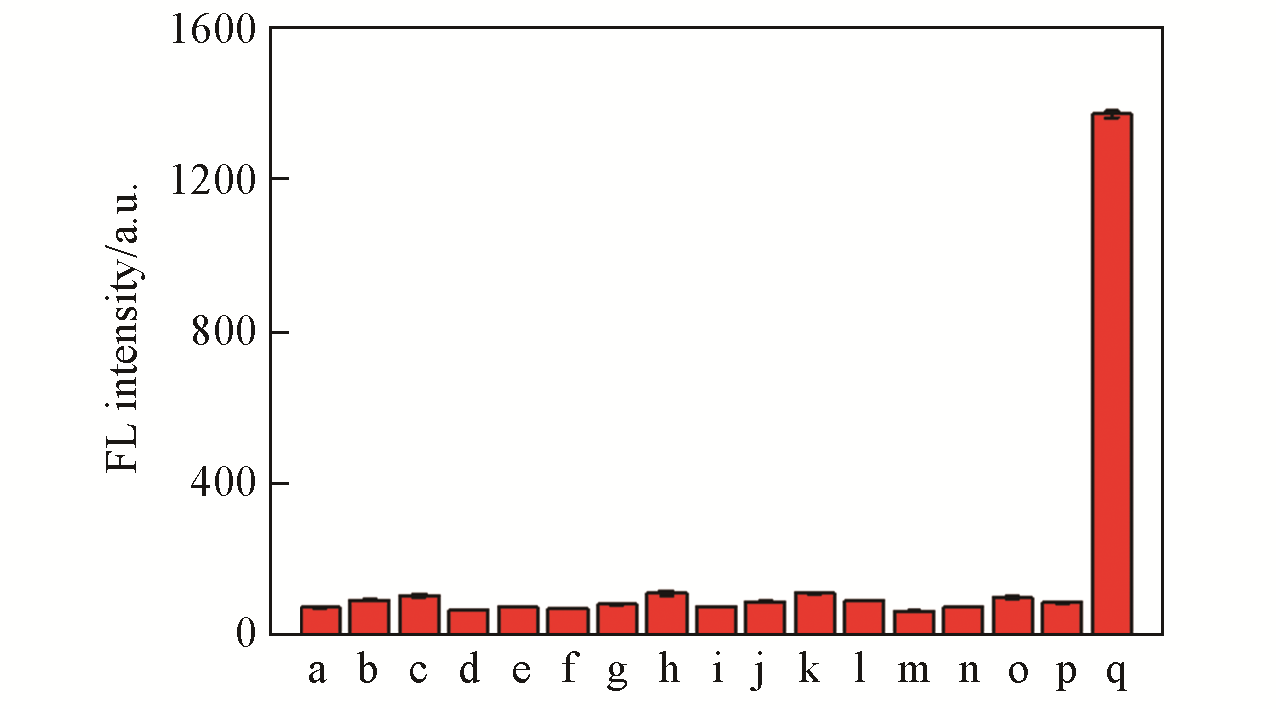
Fig.4 Fluorescence intensity of LSDQ?ONOO-(5 μmol/L) in PBS/EtOH(volume ratio 8∶2, pH=7.4) buffer solu? tion toward various analytes(100 μmol/L)note:1 mmol/L for GSH, 50 μmol/L for HOCl, 5 μmol/L for ONOO?. a. Blank; b. Cu2+; c. Fe2+; d. K+; e. Na+; f. Mg2+; g. SO42-; h. GSH; i. Cys; j. H2S; k. ·OH; l. NO2-; m. O2·-; n. H2O2; o. HOCl; p. t?BuOOH; q. ONOO-. The mixture was kept for 20 min at 37 ℃ before the fluorescence intensity of the LSDQ?ONOO- solution was recorded. λex=580 nm.
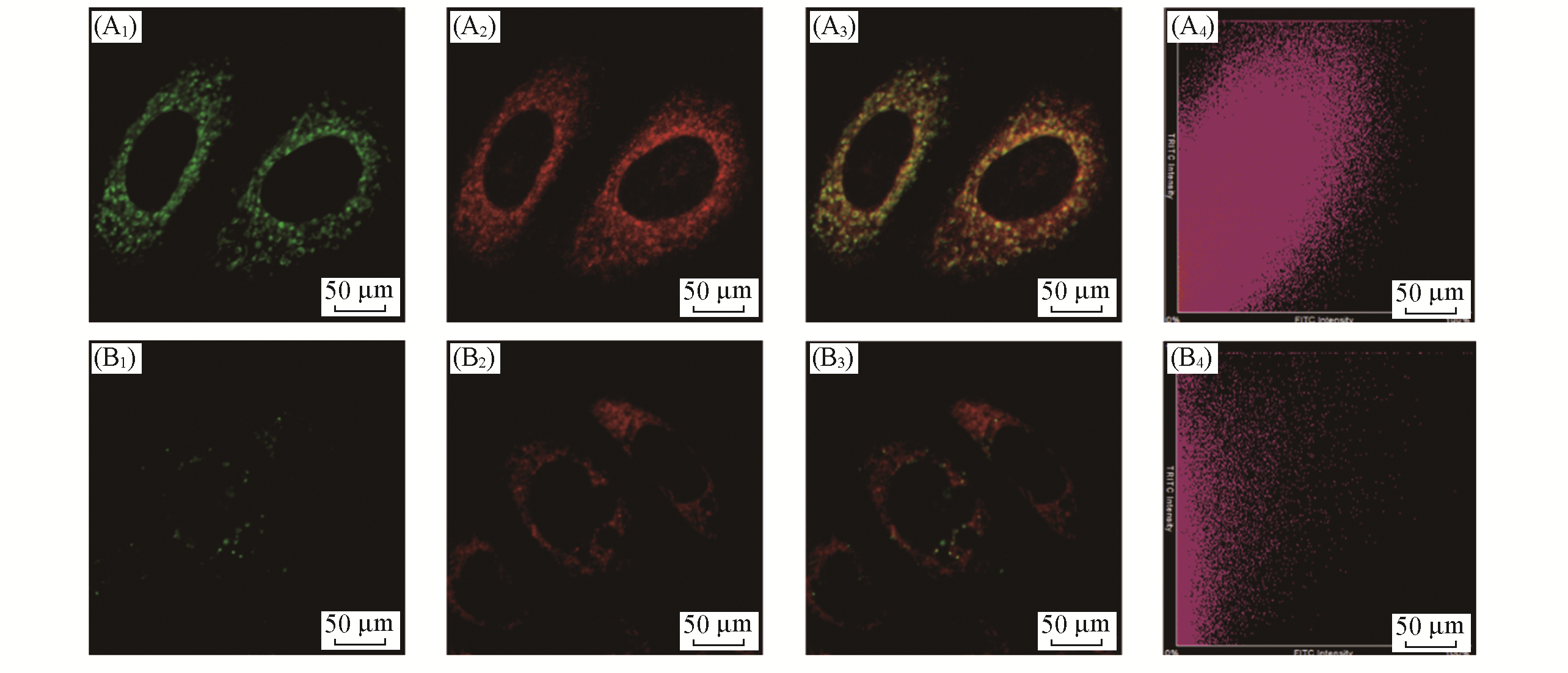
Fig.5 Distribution of LSDQ?ONOO- in HepG2 cellsnote:HepG2 cells were pretreated with LPS(1 μg/mL) and IFN?γ(50 ng/mL) for 12 h, and then incubated with 5 μmol/L LSDQ?ONOO- for 30 min and subsequently incubated with 1 μmol/L Mito?Tracker Green(or 1 μmol/L Lyso?Tracker Green) for 10 min. Green channel: Mito?Tracker Green(λex=488 nm, λem=500―550 nm) and Lyso?Tracker Green fluorescence(λex=488 nm, λem=500―550 nm); red channel: probe fluorescence(λex=561 nm, λem=640―750 nm); yellow: merged signal. (A1) Mito?tracker green; (B1) lyso?tracker green; (A2, B2) probe; (A3, B3) overlap; (A4, B4) scatter plot.
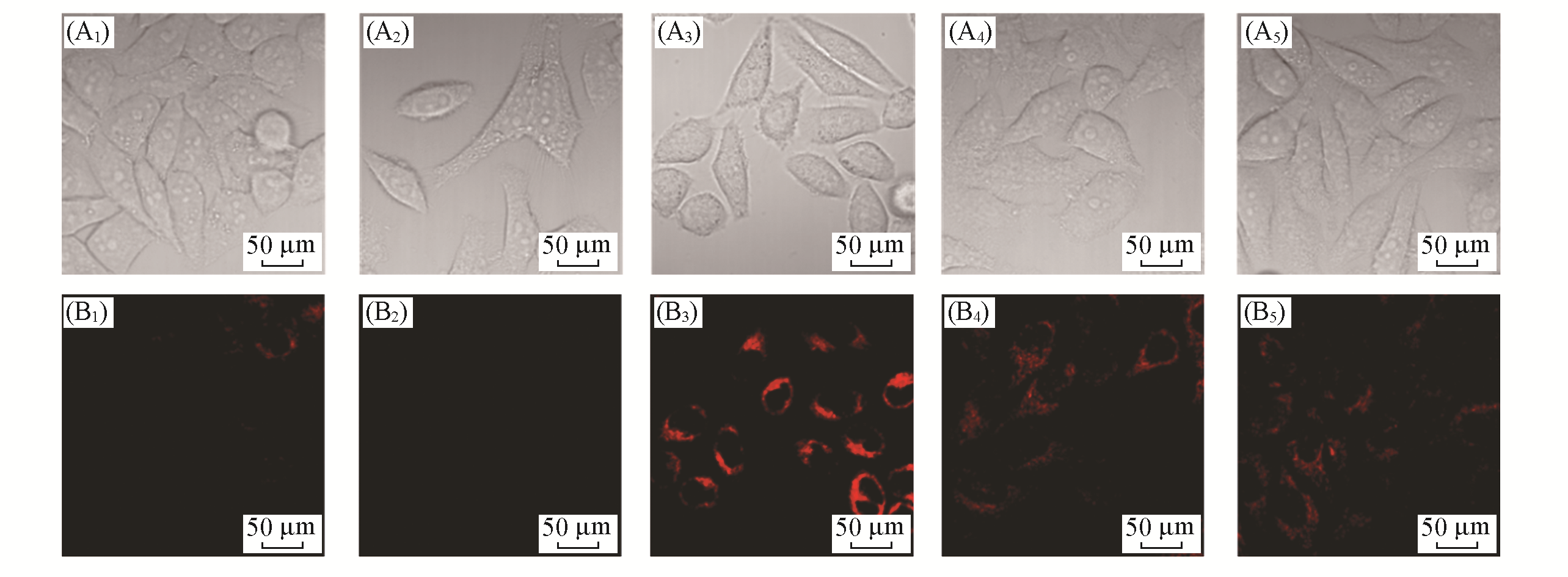
Fig.6 Fluorescent images of LSDQ?ONOO- in HepG2 cells under different conditionsnote:λex=561 nm, λem=640―750 nm. (A1―A5) Bright field; (B1―B5) red channel.(A1, B1) Cells were incubated with probe LSDQ?ONOO- (5 μmol/L, 30 min) and then imaged; (A2, B2) cells were pretreated with probe LSDQ?ONOO-(5 μmol/L, 30 min), subsequently incuba?ted with H2O2(100 μmol/L) for 30 min and then imaged; (A3, B3) cells were pre?stimulated with LPS(1 μg/mL) and IFN?γ (50 ng/mL) for 12 h, subsequently incubated with probe LSDQ?ONOO-(5 μmol/L, 30 min), and then imaged; (A4, A5, B4, B5) cells were pretreated with nitric oxide synthase inhibitor AG(5 mmol/L)(A4, B4) or O2·- scavenger TEMPO(300 μmol/L)(A5, B5) during stimulation with LPS (1 μg/mL) and IFN?γ(50 ng/mL) for 12 h, subsequently incubated with probe LSDQ?ONOO-(5 μmol/L, 30 min), and then imaged.
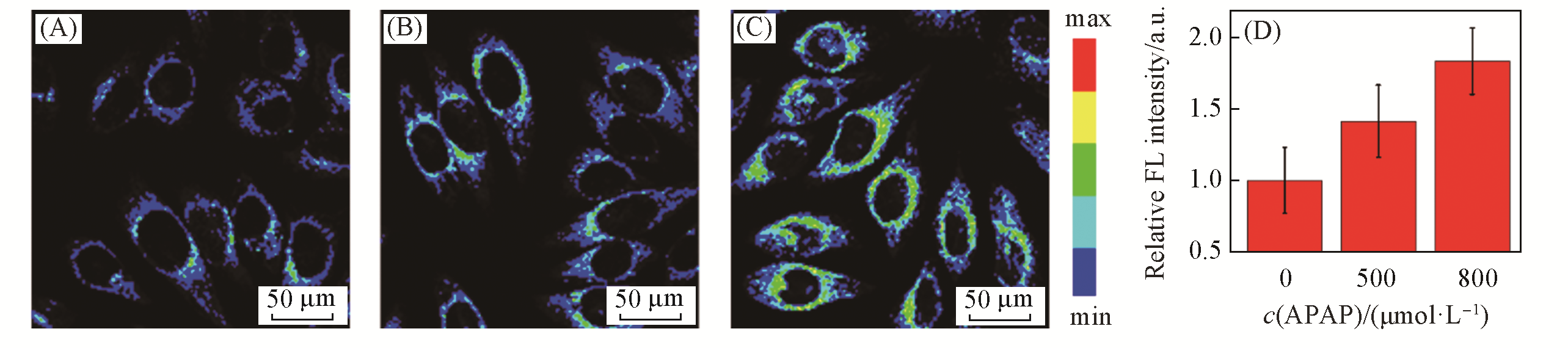
Fig.7 Fluorescent images of LSDQ?ONOO- in HepG2 cellsnote:(A—C) HepG2 cells were pretreated with different concentrations of APAP for 12 h and then incubated with 5 μmol/L LSDQ?ONOO- for 30 min. c(APAP)/(μmol·L?1): (A) 0; (B) 500; (C) 800. (D) Average intensity in panels(A—C). Data are expressed as mean ± standard deviation(SD) of three experiments. λex=561 nm, λem=640—750 nm.
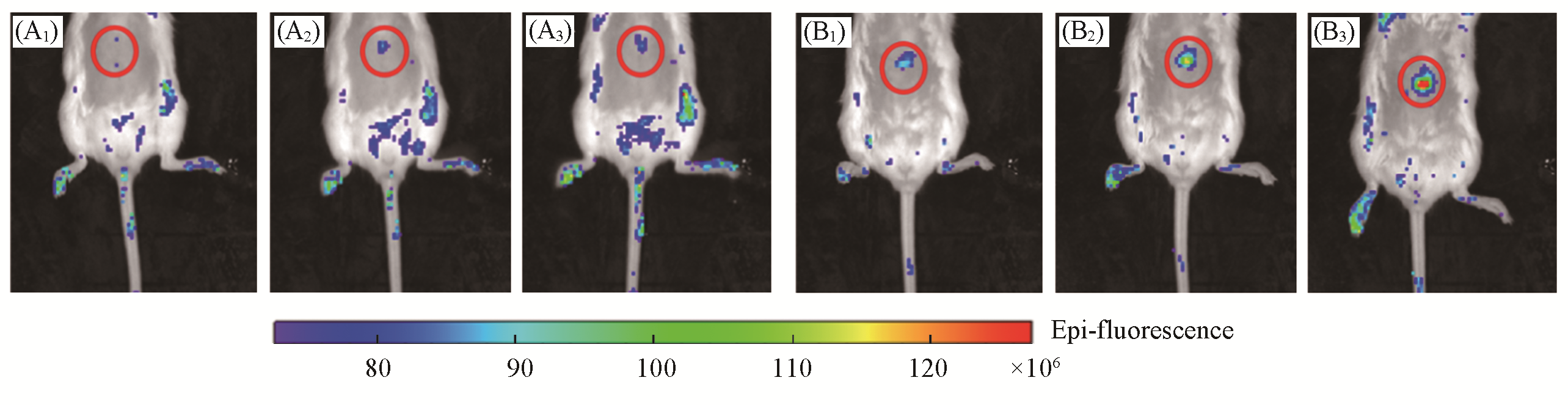
Fig.8 Representative images of BALB/c mice receiving saline(control, A1—A3), or APAP(300 mg/kg, intraperitoneally, B1—B3) followed by LSDQ?ONOO-(50 μL, 100 μmol/L, intravenously) in 30(A1, B1), 60(A2, B2) and 90 min(A3, B3)
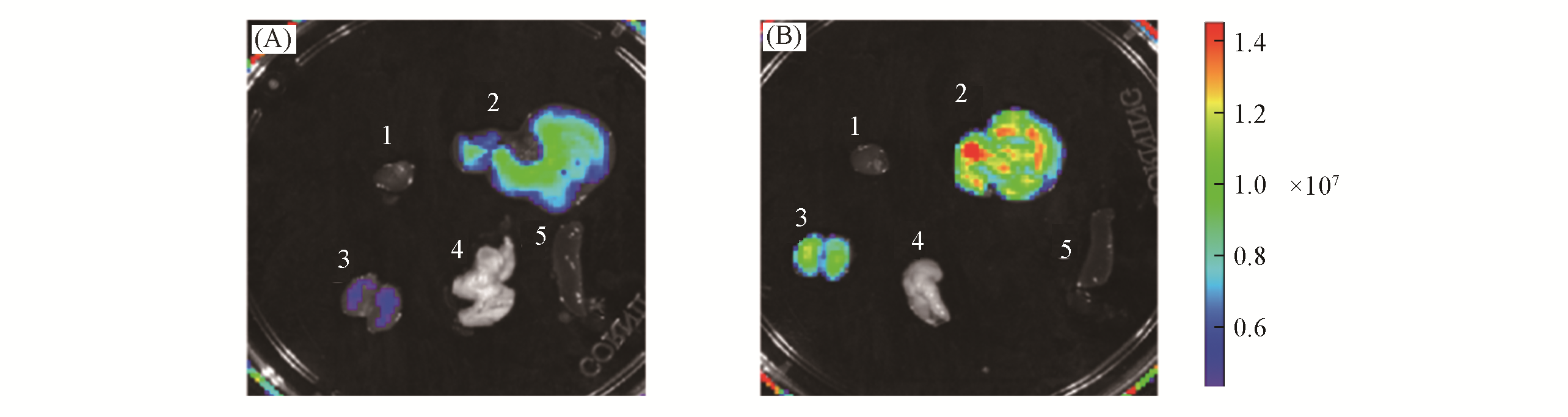
Fig.9 Fluorescence images of representative organs of BALB/c mice receiving saline(control, A) or APAP(300 mg/kg, intraperitoneally, B) followed by LSDQ?ONOO?(50 μL, 100 μmol/L, intravenously) after 90 minnote:Organs: 1. heart; 2. liver; 3. kidney; 4. lung; 5. spleen. λex=570 nm, λem=695―770 nm.
| 1 | Arnold D. T., Bentham L. M., Jacob R. P., Lilford R. J., Girling A. J., Bmc Fam. Pract., 2011, 12, 9 |
| 2 | Han X. Y., Wang R., Song X. Y., Yu F. B., Chen L. X., Anal. Chem., 2018, 90, 8108—8115 |
| 3 | Guo L. F., Tian M. G., Feng R. Q., Zhang G., Zhang R. Y., Li X. C., Liu Z. Q., He X. Q., Sun J. Z., Yu X. Q., ACS Appl. Mater.Interfaces, 2018, 10, 10706—10717 |
| 4 | Li W., Wang L., Yin S. L., Lai H. H., Yuan L., Zhang X. B., Chem. Sci., 2020, 11, 7991—7999 |
| 5 | Gao B., Li Z. T., Xue D.B ., Zhang W. H., Med. Hypotheses,2014, 82, 282—285 |
| 6 | Chen M., Vijay V., Shi Q., Liu Z., Fang H., Tong W., Drug Discov. Today, 2011, 16, 697—703 |
| 7 | Yuan L., Kaplowitz N., Clin. Liver Dis., 2013,17, 507—518 |
| 8 | Cheng D., Peng J., Lv Y., Su D., Liu D., Chen M., Yuan L., Zhang X., J. Am. Chem. Soc., 2019, 141, 6352—6361 |
| 9 | Cover C., Mansouri A., Knight T.R., Bajt M.L., Lemasters J.J., Pessayre D., Jaeschke H., J. Pharmacol. Exp. Ther., 2005, 315, 879—887 |
| 10 | Cheng D., Gong X. Y., Wu Q., Yuan J., Lv Y., Yuan L., Zhang X. B., Anal. Chem., 2020, 92, 11396—11404 |
| 11 | Li D., Wang S., Lei Z., Sun C., El⁃Toni A. M., Alhoshan M. S., Fan Y., Zhang F., Anal. Chem., 2019, 91, 4771—4779 |
| 12 | Li Y., Xie X., Yang X., Li M., Jiao X., Sun Y., Wang X., Tang B., Chem. Sci., 2017, 8, 4006—4011 |
| 13 | Zeng X. D., Chen Z. Y., Tang L., Yang H., Liu N., Zhou H., Li Y., Wu J. Z., Deng Z. X., Yu Y., Deng H., Hong X. C., Xiao Y. L., Chem. Commun., 2019, 55, 2541—2544 |
| 14 | Pacher P., Beckman J. S., Liaudet L., Physiol. Rev., 2007, 87, 315—424 |
| 15 | Ducrocq C., Blanchard B., Pignatelli B., Ohshima H., Cell. Mol. Life Sci., 1999, 55, 1068—1077 |
| 16 | Radi R., J. Biol. Chem., 2013, 288, 26464—26472 |
| 17 | Bezner B. J., Ryan L. S., Lippert A. R., Anal. Chem., 2020, 92, 309—326 |
| 18 | Masumoto H., Kissner R., Koppenol W. H., Sies H., FEBS Lett., 1996, 398, 179—182 |
| 19 | Chen X., Wang F., Hyun J. Y., Wei T., Qiang J., Ren X., Shin I., Yoon J., Chem. Soc. Rev., 2016, 45, 2976—3016 |
| 20 | Li X., Gao X., Shi W., Ma H., Chem. Rev., 2014, 114, 590—659 |
| 21 | Wang J., Guo J., Dou L., Wang R., Song Y., Yang Q., Du J., Li Y., Chem. Res. Chinese Universities, 2019, 35(4), 570—576 |
| 22 | Mi Z., Chen Y., Chen X., Yan L., Gu Q., Zhang H., Chen C., Zhang Y., Chem. Res. Chinese Universities, 2018, 34(3), 369—374 |
| 23 | Breen C., Pal R., Elsegood M. R. J., Teat S. J., Iza F., Wende K., Buckley B. R., Butler S. J., Chem. Sci., 2020, 11, 3164—3170 |
| 24 | Guo Y., Lu G., Zhuo J., Wang J., Li X., Zhang Z., J. Mater. Chem. B., 2018, 6, 2489—2496 |
| 25 | Hou J. T., Wang B., Zhang Y., Cui B., Cao X., Zhang M., Ye Y., Wang S., Chem. Commun., 2020, 56, 2759—2762 |
| 26 | Hu J. S., Shao C., Wang X., Di X., Xue X., Su Z., Zhao J., Zhu H. L., Liu H. K., Qian Y., Adv. Sci., 2019, 6, 1900341 |
| 27 | Li Y., Zhao Z., Xiao Y., Wang X., Jiao X., Xie X., Zhang J., Tang B., Anal. Chem., 2019, 91, 6097—6102 |
| 28 | Sedgwick A. C., Dou W. T., Jiao J. B., Wu L., Williams G. T., Jenkins A. T. A., Bull S. D., Sessler J. L., He X. P., James T. D., J. Am. Chem. Soc., 2018, 140, 14267—14271 |
| 29 | Sedgwick A. C., Han H. H., Gardiner J. E., Bull S. D., He X. P., James T. D., Chem. Sci., 2018, 9, 3672—3676 |
| 30 | Zhang J., Liang M., Wang X., Li Y., Jiao X., Xie X., Tang B., Chem. Commun., 2019, 55, 6719—6722 |
| 31 | Zhou D. Y., Li Y., Jiang W. L., Tian Y., Fei J., Li C. Y., Chem. Commun., 2018, 54, 11590—11593 |
| 32 | Xie X., Liu G., Su X., Li Y., Liu Y., Jiao X., Wang X., Tang B., Anal. Chem., 2019, 91, 6872—6879 |
| 33 | Cheng D., Xu W., Yuan L., Zhang X., Anal. Chem., 2017,89, 7693—7700 |
| 34 | Sedgwick A. C., Han H. H., Gardiner J. E., Bull S. D., He X. P., James T. D., Chem. Commun., 2017, 53, 12822—12825 |
| 35 | Pak Y. L., Park S. J., Wu D., Cheon B., Kim H. M., Bouffard J., Yoon J., Angew. Chem. Int. Ed., 2018, 57, 1567—1571 |
| 36 | Cheng P., Miao Q., Li J., Huang J., Xie C., Pu K., J. Am. Chem. Soc., 2019, 141, 10581—10584 |
| 37 | Kiang T. K. L., Teng X. W., Surendradoss J., Karagiozov S., Abbott F. S., Chang T. K. H., Toxicol. Appl. Pharmacol., 2011, 252, 318—324 |
| 38 | Ai X., Wang Z., Cheong H., Wang Y., Zhang R., Lin J., Zheng Y., Gao M., Xing B., Nat. Commun., 2019, 10, 1—11 |
| 39 | Ren T. B., Xu W., Zhang W., Zhang X. X., Wang Z. Y., Xiang Z., Yuan L., Zhang X. B., J. Am. Chem. Soc., 2018, 140, 7716—7722 |
| 40 | Xie X., Tang F., Liu G., Li Y., Su X., Jiao X., Wang X., Tang B., Anal. Chem., 2018, 90, 11629—11635 |
| 41 | Zhou R. H., Lu X. M., Yang Q., Wu P., Chin. Chem. Lett., 2019, 30, 1843—1848 |
| 42 | Lejeune D., Hasanuzzaman M., Pitcock A., Francis J., Sehgal I., Mol. Cancer, 2006, 5, 1—5 |
| 43 | Campbell I. L., J. Neuroimmunol., 1996, 71, 31—36 |
| [1] | ZHAO Yongmei, MU Yeshu, HONG Chen, LUO Wen, TIAN Zhiyong. Bis-naphthalimide Derivatives for Picronitric Acid Detection in Aqueous Solution [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210765. |
| [2] | TANG Qian, DAN Feijun, GUO Tao, LAN Haichuang. Synthesis and Application of Quinolinone-coumarin-based Colorimetric Fluorescent Probe for Recognition of Hg2+ [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210660. |
| [3] | WANG Di, ZHONG Keli, TANG Lijun, HOU Shuhua, LYU Chunxin. Synthesis of Schiff-based Covalent Organic Framework and Its Recognition of I ‒ [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220115. |
| [4] | HUANG Shan, YAO Jiandong, NING Gan, XIAO Qi, LIU Yi. Efficient Determination of Alkaline Phosphatase Activity Based on Graphene Quantum Dots Fluorescent Probes [J]. Chem. J. Chinese Universities, 2021, 42(8): 2412. |
| [5] | LI Anran, ZHAO Bing, KAN Wei, SONG Tianshu, KONG Xiangdong, BU Fanqiang, SUN Li, YIN Guangming, WANG Liyan. ON-OFF-ON Double Colorimetric and Fluorescent Probes Based on Phenanthro[9,10-d]imidazole Derivatives and Their Living Cells Imaging [J]. Chem. J. Chinese Universities, 2021, 42(8): 2403. |
| [6] | YANG Xinjie, LAI Yanqiong, LI Qiuyang, ZHANG Yanli, WANG Hongbin, PANG Pengfei, YANG Wenrong. An Enzyme-free and Label-free Fluorescent Probe for Detection of Microcystin-LR Based on Circular DNA-Silver Nanoclusters [J]. Chem. J. Chinese Universities, 2021, 42(12): 3600. |
| [7] | CHEN Weiju, CHEN Shiya, XUE Caoye, LIU Bo, ZHENG Jing. Fluorescent Probe for Hypoxia-triggered Imaging and Cancer Therapy [J]. Chem. J. Chinese Universities, 2021, 42(11): 3433. |
| [8] | HUANG Jialing,LIU Fengjiao,WANG Tingting,LIU Cuie,ZHENG Fengying,WANG Zhenhong,LI Shunxing. Nitrogen and Sulfur co-Doped Carbon Quantum Dots for Accurate Detection of pH in Gastric Juice† [J]. Chem. J. Chinese Universities, 2020, 41(7): 1513. |
| [9] |
WANG Jinjin,QI Shaolong,DU Jianshi,YANG Qingbiao,SONG Yan,LI Yaoxian.
Synthesis of Benzothiazole Fluorescent Probe for Detection of N2H4·H2O and HS |
| [10] | Yong ZHANG,Cheng SHEN,Zhirong XING,Guiqi CHEN,Zi LU,Zhibing HOU,Xuemei CHEN. Benzimidazole-Derived Fluorescence Enhancement Probe for Visual Detection of HClO † [J]. Chem. J. Chinese Universities, 2019, 40(12): 2480. |
| [11] | YANG Mei,LIU Qing,TANG Qing,WANG Chenghui,YANG Meixiang,SUN Tao,HUANG Ying,TAO Zhu. Water-soluble Supramolecular Fluorescent Probe for Sensing Carbendazim and Its Application in Living Cell Imaging† [J]. Chem. J. Chinese Universities, 2018, 39(12): 2665. |
| [12] | HE Xiaoqin, HE Junlin, XU Hua, GUO Lei, XIE Jianwei. Active Conformation of DNA Aptamer Against Recombinant Human Erythropoietin-α† [J]. Chem. J. Chinese Universities, 2018, 39(1): 48. |
| [13] | LIU Huiqiang, PENG Chao, CHEN Ning, LIU Yangping. Novel Fluorescent/EPR Difunctional Probe for Detecting Hypochlorite† [J]. Chem. J. Chinese Universities, 2017, 38(9): 1542. |
| [14] | WANG Xingxing, LI Panpan, HE Jinglin, OUYANG Wen, XIAO Hui, YANG Chan, CAO Zhong. Cytosine-rich Oligonucleotide-templated Fluorescent Silver Nanoclusters for Sensitive Assay of S1 Nuclease [J]. Chem. J. Chinese Universities, 2017, 38(8): 1334. |
| [15] | YANG Jingjing, YU Yuewen, WANG Bingxiang, JIANG Yuliang. A Sensitive Fluorescent Probe for Cysteine Based on 1,8-Naphthalimide Derivative and Application in Bioimaging† [J]. Chem. J. Chinese Universities, 2017, 38(7): 1198. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||