

Chem. J. Chinese Universities ›› 2019, Vol. 40 ›› Issue (7): 1397.doi: 10.7503/cjcu20190159
• Organic Chemistry • Previous Articles Next Articles
WANG Jinjin1,2, QI Shaolong1,3, DU Jianshi3, YANG Qingbiao1,*( ), SONG Yan2, LI Yaoxian1
), SONG Yan2, LI Yaoxian1
Received:2019-03-18
Online:2019-05-24
Published:2019-07-09
Contact:
YANG Qingbiao
E-mail:yangqb@jlu.edu.cn
Supported by:CLC Number:
TrendMD:
WANG Jinjin,QI Shaolong,DU Jianshi,YANG Qingbiao,SONG Yan,LI Yaoxian. Synthesis of Benzothiazole Fluorescent Probe for Detection of N2H4·H2O and HS
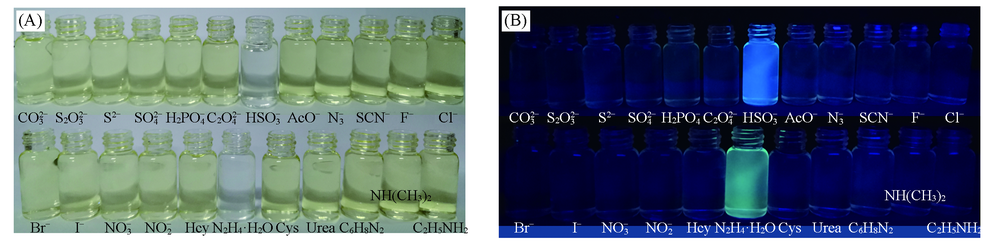
Fig.1 Photographs of color change(A) and fluorescence response(B) of HBT-CN(5.0×10-6 mol/L) after adding HSO3-(2.5×10-4 mol/L), N2H4·H2O(1×10-3 mol/L) and other analytes(2.5×10-3 mol/L)V(DMSO):V(PBS)=1:4.
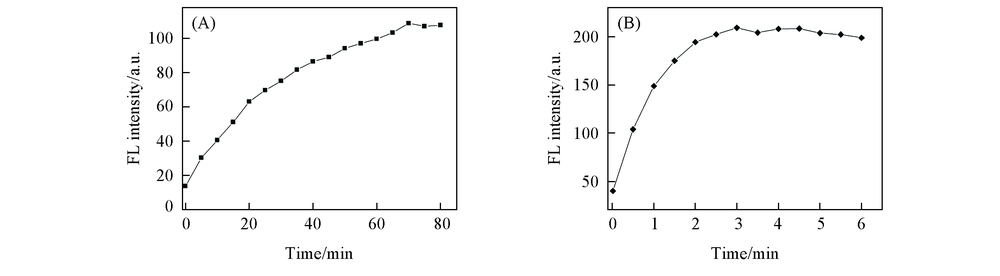
Fig.2 Time-dependent fluorescence intensity changes of HBT-CN(5×10-6 mol/L) in the presence of N2H4·H2O(1×10-3 mol/L, λex=399 nm, λem=500 nm, slits: 2.5 nm/5 nm)(A) and HSO3-(1×10-3 mol/L, λex=399 nm, λem=458 nm, slits: 2.5 nm/5 nm)(B)
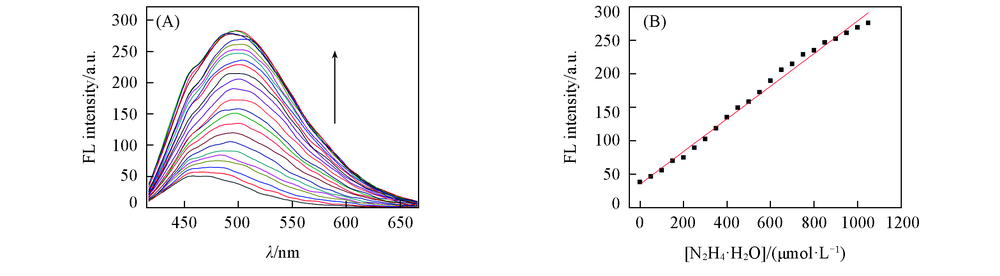
Fig.4 Fluorescence intensity of HBT-CN(5×10-6 mol/L) changes upon gradual addition of N2H4·H2O(0—1.2×10-3 mol/L)(A) and linear relationship between fluorescence intensity(500 nm) of HBT-CN and concentrations of N2H4·H2O(0—1.05×10-3 mol/L)[V(DMSO):V(PBS)=1:4; λex=399 nm, slit: 5 nm/5 nm](B)
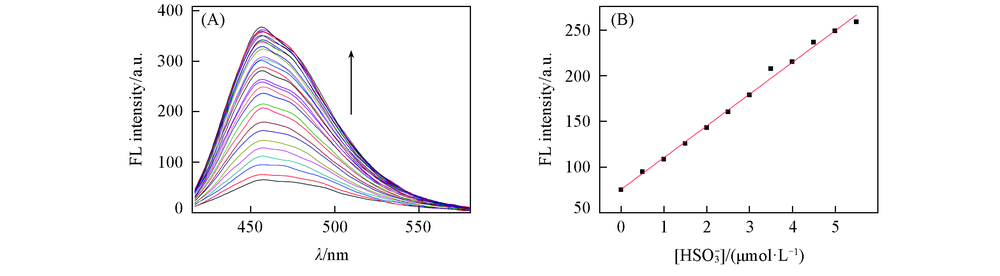
Fig.5 Fluorescence intensity of HBT-CN(5 μmol/L) changes upon gradual addition of HSO3-(0—3×10-4 mol/L)(A) and linear relationship between fluorescence intensity of HBT-CN and concentrations of HSO3-(0—1.2×10-4 mol/L) at 458 nm[V(DMSO):V(PBS)=1:4, λex=399 nm, slit: 5 nm/5 nm](B)

Fig.7 Fluorescence response of HBT-CN(5×10-6 mol/L) in the presence of HSO3-+anions(A) and N2H4·H2O+others(B)Black bars represent the fluorescent intensity(458 nm) of HBT-CN(5×10-6 mol/L) in the presence of anions after adding HSO3-; red bars represent the fluorescent intensity(458 nm) of HBT-CN(5×10-6 mol/L) in the presence of anions in the absence of HSO3-. (A) a. HSO3-; b. CO32-; c. S2O32-; d. S2-; e. SO42-; f. H2PO4-; g. C2O42-; h. AcO-; i. N3-; j. SCN-; k. F-; l. Cl-; m. Br-; n. I-; o. NO3-; p. NO2-. (B) a. N2H4·H2O; b.GSH; c. Cys; d. Hcy; e. H2NCONH2; f. C6H8N2; g. NH(CH3)2; h. CH3CH2NH2; i. K+; j. Na+; k. NH4+; l. Mg2+; m. Ba2+.
| [1] | Wang W.D., Hu Y., Li Q., Hu S. L., Inorg. Chim. Acta, 2018, 477, 206—211 |
| [2] | Xu W.Z., Liu W. Y., Zhou T. T., Yang Y. T., Li W., Spectrochim. Acta Part A, 2018, 193, 324—329 |
| [3] | Wu Q., Zheng J., Zhang W., Wang J., Liang W., Stadler F. J., Talanta, 2019, 195, 857—864 |
| [4] | Christopher A. R., Steven D. A., Chem. Res. Toxicol., 1997, 10, 328—334 |
| [5] | Zhang Y., Huang Y., Yue Y., Chao J., Huo F., Yin C., Sens. Actuators B: Chem., 2018, 273, 944—950 |
| [6] | Yang Y. H., Liu X. F., Yan D., Deng P.,Guo Z. Y., Zhan H. B., RSC Adv., 2018, 8, 17471—17474 |
| [7] | Mahmoud A. A. T., Hassan G. M., Hassan A. M. S., Abdel Latif A. K. M., Ramadan M. F., Eur. J. Integ. Med., 2015, 7, 234—242 |
| [8] | Wang H., Wu X., Yang S., Tian H., Liu Y., Sun B., Dyes and Pigments, 2019, 160, 757—764 |
| [9] | Ao H., Zhang Y., Plant Physiology Communication, 2007, 43(2), 259—263 |
| (敖红, 张羽. 植物生理学通讯, 2007, 43(2), 259—263) | |
| [10] | Li G., Sang N., Ecotox. Environ. Safe., 2009, 72, 236—241 |
| [11] | Chao J., Wang X., Liu Y., Zhang Y., Huo F., Yin C., Zhao M., Sun J., Xu M., Sens. Actuators B: Chem., 2018, 272, 195—202 |
| [12] | Dutta T., Chandra F., Koner A.L., Spectrochim. Acta Part A, 2018, 191, 217—220 |
| [13] | Zhao F., Sultanbawa Y., Feng H., Wang Y.L., Meng Q. T., Wang Y., Zhang Z. Q., Zhang R., J. Agric. Food Chem., 2019, 67, 4375—4383 |
| [14] | Michigami Y., Morooka M., Ueda K., J. Chromatogr. A, 1996, 732(2), 403—407 |
| [15] | Malone H. E., Anal. Chem., 1961, 33, 575—577 |
| [16] | Isaac A., Wain A.J., Compton R. G., Livingstone C., Davis J., The Analyst., 2005, 130, 1343—1344 |
| [17] | Barish R. F. M. A. A. O., J. Agric. Food Chem., 2003, 5, 1513—15717 |
| [18] | Elder D.P., Snodin D., Teasdale A., J. Phar. Biome. Analysis, 2011, 54, 900—910 |
| [19] | Lu Z., Shi X., Ma Y., Fan W., Lu Y., Wang Z., Fan C., Sens. Actuators B: Chem., 2018, 258, 42—49 |
| [20] | Ding Y. B., Zhu W. H.,Xie Y. S., Chem. Rev., 2017, 117, 2203—2256 |
| [21] | Ding Y. B., Tang Y. Y.,Zhu W. H., Xie Y. S., Chem. Soc. Rev., 2015, 44, 1101—1112 |
| [22] | Ding Y.B., Li X., Li T., Zhu W. H., Xie Y. S., J. Org. Chem., 2013, 78, 5328—5338 |
| [23] | Shi X., Yin C., Wen Y., Zhang Y., Huo F., Spectro. Acta Part A, 2018, 203, 106—111 |
| [24] | Lu Z., Fan W., Shi X., Lu Y., Fan C., Anal. Chem., 2017, 89, 9918—9925 |
| [25] | Xu H., Gu B., Li Y., Huang Z., Su W., Duan X., Yin P., Li H., Yao S., Talanta, 2018, 180, 199—205 |
| [26] | Yang C., Chen Y., Wu K., Wei T., Wang J., Zhang S., Han Y., Anal. Methods, 2015, 7, 3327—3330 |
| [27] | Zhang X., Liu J.Y., Dyes and Pigments, 2016, 125, 80—88 |
| [28] | Zhang H., Feng W., Feng G., Dyes and Pigments, 2017, 139, 73—78 |
| [29] | Gu B., Huang L., Su W., Duan X., Li H., Yao S., Anal. Chim. Acta, 2017, 954, 97—104 |
| [30] | Zhou Y., Zeng F., Liu Y., Li M., Xiao S., Yan J., Zhang N., Zheng K., Tetrah. Letters, 2018, 59, 3253—3257 |
| [31] | Wang S. J., Li J.,Gao Y., Guo Y., Asian J. Org. Chem., 2018, 7, 563—569 |
| [32] | Sun M., Du L., Yu H., Zhang K., Liu Y., Wang S., Talanta, 2017, 162, 180—186 |
| [33] | Wu J., Pan J., Ye Z., Zeng L., Su D., Sens. Actuators B: Chem., 2018, 274, 274—284 |
| [1] | ZHAO Yongmei, MU Yeshu, HONG Chen, LUO Wen, TIAN Zhiyong. Bis-naphthalimide Derivatives for Picronitric Acid Detection in Aqueous Solution [J]. Chem. J. Chinese Universities, 2022, 43(3): 20210765. |
| [2] | TANG Qian, DAN Feijun, GUO Tao, LAN Haichuang. Synthesis and Application of Quinolinone-coumarin-based Colorimetric Fluorescent Probe for Recognition of Hg2+ [J]. Chem. J. Chinese Universities, 2022, 43(2): 20210660. |
| [3] | WANG Di, ZHONG Keli, TANG Lijun, HOU Shuhua, LYU Chunxin. Synthesis of Schiff-based Covalent Organic Framework and Its Recognition of I ‒ [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220115. |
| [4] | HUANG Shan, YAO Jiandong, NING Gan, XIAO Qi, LIU Yi. Efficient Determination of Alkaline Phosphatase Activity Based on Graphene Quantum Dots Fluorescent Probes [J]. Chem. J. Chinese Universities, 2021, 42(8): 2412. |
| [5] | LI Anran, ZHAO Bing, KAN Wei, SONG Tianshu, KONG Xiangdong, BU Fanqiang, SUN Li, YIN Guangming, WANG Liyan. ON-OFF-ON Double Colorimetric and Fluorescent Probes Based on Phenanthro[9,10-d]imidazole Derivatives and Their Living Cells Imaging [J]. Chem. J. Chinese Universities, 2021, 42(8): 2403. |
| [6] | YANG Xinjie, LAI Yanqiong, LI Qiuyang, ZHANG Yanli, WANG Hongbin, PANG Pengfei, YANG Wenrong. An Enzyme-free and Label-free Fluorescent Probe for Detection of Microcystin-LR Based on Circular DNA-Silver Nanoclusters [J]. Chem. J. Chinese Universities, 2021, 42(12): 3600. |
| [7] | CHEN Weiju, CHEN Shiya, XUE Caoye, LIU Bo, ZHENG Jing. Fluorescent Probe for Hypoxia-triggered Imaging and Cancer Therapy [J]. Chem. J. Chinese Universities, 2021, 42(11): 3433. |
| [8] | HUANG Jialing,LIU Fengjiao,WANG Tingting,LIU Cuie,ZHENG Fengying,WANG Zhenhong,LI Shunxing. Nitrogen and Sulfur co-Doped Carbon Quantum Dots for Accurate Detection of pH in Gastric Juice† [J]. Chem. J. Chinese Universities, 2020, 41(7): 1513. |
| [9] | WU Qian, CHENG Dan, LÜ Yun, YUAN Lin, ZHANG Xiaobing. Monitoring of Peroxynitrite Variation During Liver Injury Adopting a Far Red to Near-infrared Fluorescent Probe with Large Stokes Shift [J]. Chem. J. Chinese Universities, 2020, 41(11): 2426. |
| [10] | CHEN Danyi, ZHANG Fumei, HE Dan, ZHANG Zimei, ZHONG Fen, WEN Simiaomiao, LIU Qixing, ZHOU Haifeng. Synthesis of Chiral Phenylbenzothiazole Methanol via Transfer Hydrogenation Catalyzed by Ruthenium Complexes† [J]. Chem. J. Chinese Universities, 2020, 41(10): 2264. |
| [11] | WEI Xin, DENG Yaoliang, ZHENG Xuming, ZHAO Yanying. Ground Structure and Excited State Proton Transfer Reaction of 2-Aminobenzothiazole [J]. Chem. J. Chinese Universities, 2019, 40(8): 1679. |
| [12] | Yong ZHANG,Cheng SHEN,Zhirong XING,Guiqi CHEN,Zi LU,Zhibing HOU,Xuemei CHEN. Benzimidazole-Derived Fluorescence Enhancement Probe for Visual Detection of HClO † [J]. Chem. J. Chinese Universities, 2019, 40(12): 2480. |
| [13] | YANG Mei,LIU Qing,TANG Qing,WANG Chenghui,YANG Meixiang,SUN Tao,HUANG Ying,TAO Zhu. Water-soluble Supramolecular Fluorescent Probe for Sensing Carbendazim and Its Application in Living Cell Imaging† [J]. Chem. J. Chinese Universities, 2018, 39(12): 2665. |
| [14] | HE Xiaoqin, HE Junlin, XU Hua, GUO Lei, XIE Jianwei. Active Conformation of DNA Aptamer Against Recombinant Human Erythropoietin-α† [J]. Chem. J. Chinese Universities, 2018, 39(1): 48. |
| [15] | DUAN Yongbin, YIN Yan, MENG Fanli, ZHAO Lianhua, LIU Yukun, YUAN Zhe, FENG Yangbo. Design, Synthesis and Biological Evaluation of Benzothiazoles as Highly Potent ROCK Inhibitors Through Molecular Docking and Free Energy Calculations† [J]. Chem. J. Chinese Universities, 2017, 38(9): 1568. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||