

Chem. J. Chinese Universities ›› 2016, Vol. 37 ›› Issue (10): 1882.doi: 10.7503/cjcu20160198
• Physical Chemistry • Previous Articles Next Articles
ZHANG Youfa*( ), AN Lijia, HAO Jianxia, YU Xinquan, CHEN Feng
), AN Lijia, HAO Jianxia, YU Xinquan, CHEN Feng
Received:2016-03-30
Online:2016-10-10
Published:2016-09-18
Contact:
ZHANG Youfa
E-mail:yfzhang@seu.edu.cn
Supported by:CLC Number:
TrendMD:
ZHANG Youfa, AN Lijia, HAO Jianxia, YU Xinquan, CHEN Feng. Fabrication of Transparent Superhydrophobic Nano-arrays and Self-ejecting Behavior of Dew Drops†[J]. Chem. J. Chinese Universities, 2016, 37(10): 1882.
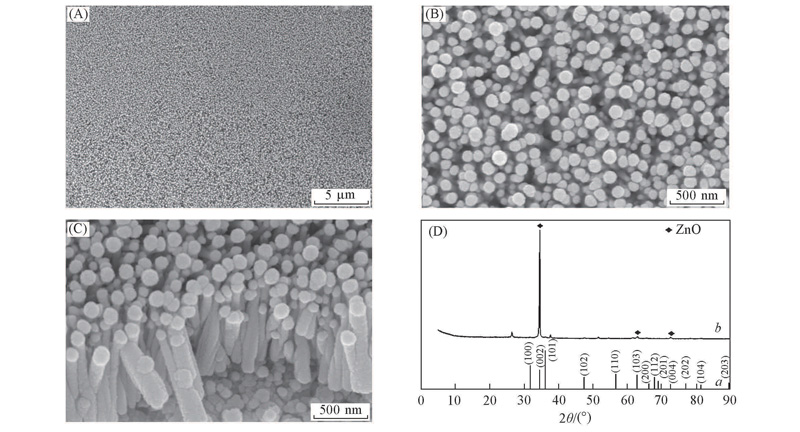
Fig.1 SEM images(A—C) and XRD patterns(D) of as-prepared ZnO nano-rod arrays Spin coating with 0.3 mol/L sol and subsequent 2 h growth in the solution. (A) Low magnification;(B) high magnification; (C) cross-section of nano-rods.
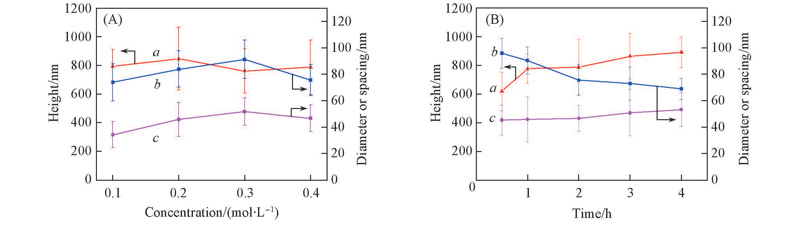
Fig.2 Size parameters of the ZnO nano-rod arrays on FTO(A) Different concentrations of seeds solution; (B) different immersion time in growth solution. a. Height; b. diameter; c. spacing.
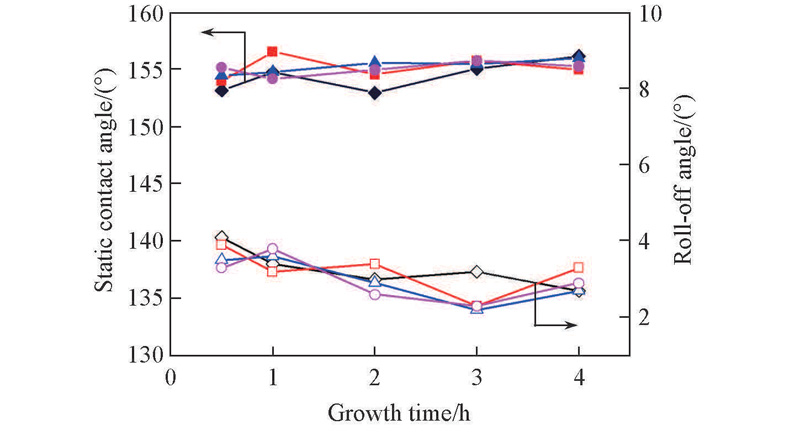
Fig.3 Effects of concentration of seeds solution and growth time on water wettability Concentration of seed solution/(mol·L-1): ◆◇ 0.1; ■□ 0.2; ▲△ 0.3; ●○ 0.4.
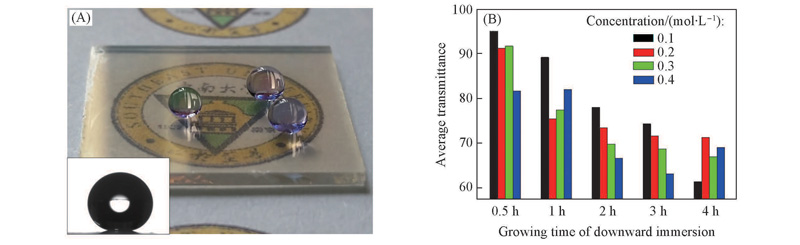
Fig.4 Effects of concentration of seeds solution and growth time on transmittance(A) Digital image of a transparent superhydrophobic FTO sample fabricated with concentration of seeds solution of 0.1 mol/L and growth time of 0.5 h, inset shows the spherical shape of a drop on the surface. (B) average transmittance of the samples.

Fig.5 Condensation behavior of the transparent superhydrophobic nano-arrays on a 2 ℃ cold stage under ambient conditions(temperature 17 ℃, relative humidity 42%)The sample is prepared with concentration of seeds solution of 0.1 mol/L and growth time of 0.5 h. Time-lapse optical images of dropwise condensation for 5 min(A), 10 min(B), 20 min(C) and 30 min(D), respectively. (E)—(H) Snap shots of self-jumping of a drop during condensation. Time/ms: (E) 0; (F) 1; (G) 5; (H) 48.
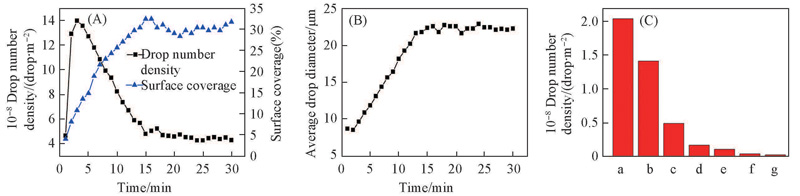
Fig.6 Quantification of the condensate drops on the transparent superhydrophobic nano-arrays (A) Measured drop number densities and drop surface coverage on the surface; (B) time evolution of average drop diameter; (C) histogram of drop diameter. a. 1—10 μm; b. 10—20 μm; c. 20—30 μm; d. 30—40 μm;e. 40—50 μm; f. 50—60 μm; g. >60 μm.
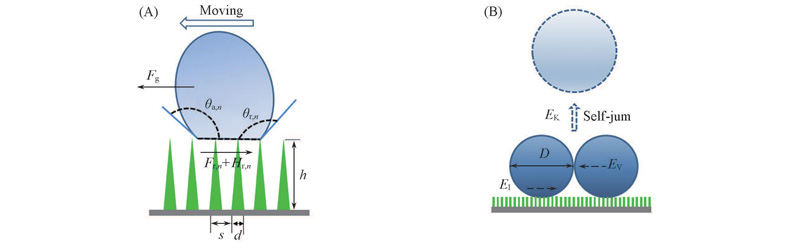
Fig.7 Schematic illustration of droplets motion on superhydrophobic nano-arrays(A) Forces of a droplet during moving; (B) self-jumping of a droplet resulted from coalesce.
| [1] | Barthlott W., Neinhuis C., Planta, 1997, 202,1—8 |
| [2] | Gao X. F., Jiang L., Nature, 2004, 432,36 |
| [3] | Gao X. F., Yan X., Yao X., Xu L., Zhang K., Zhang J. H., Yang B., Jiang L., Adv. Mater., 2007, 19,2213—2217 |
| [4] | Shi Y. L., Yang W., Feng X. J., Chem. J. Chinese Universities, 2015, 36(9), 1724—1729 |
| (石彦龙, 杨武, 冯晓娟.高等学校化学学报, 2015,36(9), 1724—1729) | |
| [5] | Wang S. T., Liu K. S., Yao X., Jiang L., Chem. Rev., 2015, 115(16), 8230—8293 |
| [6] | Kreder M. J., Alvarenga J., Kim P., Aizenberg J., Nat. Rev. Mater., 2016, 1,1—15 |
| [7] | Zhang Y. F., Wu J., Yu X. Q., Liang C. H., Wu J., Acta Phys. Chim. Sin., 2014, 30(10), 1970—1978 |
| (张友法, 吴洁, 余新泉, 梁彩华, 吴俊.物理化学学报, 2014,30(10), 1970—1978) | |
| [8] | Zhang Y. F., Yu X. Q., Wu H., Wu J. , Appl. Surf. Sci., 2012, 258,8253—8257 |
| [9] | Zhou Q. H., Yu X. Q., Zhang Y. F., Li K. N., Chen F., Gu Z. Z., Chem. J. Chinese Universities, 2010, 31(3), 456—462 |
| (周荃卉, 余新泉, 张友法, 李康宁, 陈锋, 顾忠泽.高等学校化学学报, 2010,31(3), 456—462) | |
| [10] | Xu L. B., Karunakaran R. G., Guo J., Yang S., ACS Appl. Mater. Inter., 2012, 4,1118—1125 |
| [11] | Ge D. T., Yang L. L., Zhang Y. F., Rahmawan Y., Yang S., Part. Part. Syst. Char., 2014, 31,763—770 |
| [12] | Deng X., Mammen L., Butt H. J., Vollmer D., Science, 2012, 335,67—70 |
| [13] | Chen C. H., Cai Q. J., Tsai C. L., Chen C. L., Xiong G. Y., Yu Y., Ren Z. F., Appl. Phys. Lett., 2007, 90(17), 173108 |
| [14] | Su B., Guo W., Jiang L., Small, 2015, 11,1072—1096 |
| [15] | Zhang Q., Sun D., Zhang Y., Zhu M., Langmuir, 2014, 30(42), 12559—12569 |
| [16] | Pang Y. C., Zhao Y., Feng J., Chem. J. Chinese Universities, 2013, 34(4), 919—924 |
| (庞艺川, 赵颖, 冯杰.高等学校化学学报, 2013,34(4), 919—924) | |
| [17] | Lau K. K. S., Bico J., Teo K. B. K., Chhowalla M., Amaratunga G. A. J., Milne W. I., McKinley G. H., Gleason K. K., Nano Lett., 2003, 3(12), 1701—1705 |
| [18] | Varanasi K. K., Hsu M., Bhate N., Yang W. S., Deng T., Appl. Phys. Lett., 2009, 95(9), 094101-1—3 |
| [19] | Varanasi K. K., Deng T., Smith J. D., Hsu M., Bhate N., Appl. Phys. Lett., 2010, 97(23), 234102-1—3 |
| [20] | Hou Y., Yu M., Chen X., Wang Z., Yao S., Acs Nano, 2014, 9(1), 71—81 |
| [21] | Miljkovic N., Enright R., Nam Y., Lopez K., Dou N., Sack J., Wang E. N., Nano Lett., 2013, 13,179—187 |
| [22] | Kim M. K., Cha H., Birbarah P., Chavan S., Zhong C., Xu Y. H., Miljkovic N., Langmuir, 2015, 31,13452—13466 |
| [23] | He M., Zhang Q. L., Zeng X. P., Cui D. P., Chen J., Li H. L., Wang J. J., Song Y. L., Adv. Mater., 2013, 25,2291—2295 |
| [24] | Tian J., Zhu J., Guo H.Y., Li J., Feng X. Q., Gao X. F., J. Phys. Chem. Lett., 2014, 5, 2084—2088 |
| [25] | Xia Y. M., Zhang Y. F., Yu X. Q., Chen F., J. Phys. Chem. B, 2014, 118,12002—12007 |
| [26] | Xia Y. M., Zhang W., Zhang Y. F., Yu X. Q., Chen F., Mater. Lett., 2014, 131,178—181 |
| [27] | Gao Y. Q., Gereige I., El Labban A., Cha D., Isimjan T. T., Beaujuge P. M., ACS Appl. Mater. Inter., 2014, 6,2219—2223 |
| [28] | Chen X. M., Wu J., Ma R. Y., Hua M., Koratkar N., Yao S. H., Wang Z. K., Adv. Funct. Mater., 2011, 21,4617—4623 |
| [29] | Feng J., Pang Y. C., Qin Z. Q., Ma R. Y., Yao S. H., ACS Appl. Mater. Inter., 2012, 4,6618—6625 |
| [30] | Hou Y. M., Yu M., Chen X. M., Wang Z. K., Yao S. H., ACS Nano, 2015, 9,71—81 |
| [31] | Liu T. Q., Sun W., Li X. Q., Sun X. Y., Ai H. R., Acta Phys. Chim. Sin., 2014, 63(8), 086801-1—11 |
| (刘天庆, 孙玮, 李香琴, 孙相彧, 艾宏儒.物理化学学报, 2014, 63(8), 086801-1—11) | |
| [32] | Luo Y. T., Li J., Zhu J., Zhao Y., Gao X. F., Angew. Chem. Int. Ed., 2015, 54(16), 4876—4879 |
| [33] | Lv C. J., Hao P. F., Zhang X. W., He F., ACS Nano, 2015, 9(8), 12311—12319 |
| [34] | Baxter A. C. S., Trans. Faraday Soc., 1944, 40,546—551 |
| [35] | Wenzel R. N., J. Phys. Colloid Chem., 1949, 53,1466—1467 |
| [36] | Jung S., Dorrestijn M., Raps D., Das A., Megaridis C. M., Poulikakos D., Langmuir, 2011, 27,3059—3066 |
| [37] | Rykaczewski K., Langmuir, 2012, 28,7720—7729 |
| [38] | Malvadkar N. A., Hancock M. J., Sekeroglu K., Dressick W. J., Demirel M. C., Nat. Mater., 2010, 9,1023—1028 |
| [1] | DONG Yanhong, LU Xinhuan, YANG Lu, SUN Fanqi, DUAN Jingui, GUO Haotian, ZHANG Qinjun, ZHOU Dan, XIA Qinghua. Preparation of Bifunctional Metal-organic Framework Materials and Application in Catalytic Olefins Epoxidation [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220458. |
| [2] | REN Wen, ZHANG Guoli, YAN Han, HU Xinghua, LI Kun, WANG Jingfeng, LI Ruiqi. Preparation of Superhydrophobic Polyaniline/Polytetrafluoroethylenethylene Composite Membrane and Its Separation Ability for Oil-Water Emulsion † [J]. Chem. J. Chinese Universities, 2020, 41(4): 846. |
| [3] | LIU Shuaizhuo,ZHANG Qian,LIU Ning,XIAO Wenyan,FAN Leiyi,ZHOU Ying. One-step Synergistic Hydrophobic Modification of Melamine Sponge and Its Application † [J]. Chem. J. Chinese Universities, 2020, 41(3): 521. |
| [4] | ZHAO Peng,ZHANG Jinteng,LIN Yanhong. Excellent Ultraviolet Photocatalytic Efficiency of Mg 2+ Doped ZnO and Analysis on Its Synergetic Effect † [J]. Chem. J. Chinese Universities, 2020, 41(3): 538. |
| [5] | WANG Wu, LAI Hua, CHENG Zhongjun, LIU Yuyan. Reversible Regulation of Droplet Directional/anti-directional Rolling on Superhydrophobic Shape Memory Microarray Surface [J]. Chem. J. Chinese Universities, 2020, 41(11): 2538. |
| [6] |
YANG Xiurong,ZHANG Chi,GAO Hongxu,ZHAO Fengqi,NIU Shiyao,GUO Zhaoqi,MA Haixia.
Density Functional Theory Study of NO, NO2 Adsorbed on ZnO(10 |
| [7] | WANG Fuxiang,CHEN Ziyu,YANG Weiting,LIU Lijuan,REN Guojian,LIU Yanfeng,PAN Qinhe. Preparation and Adsorption Performance for U(Ⅵ) of ZnO@ZIF-8 Core@Shell Microspheres† [J]. Chem. J. Chinese Universities, 2019, 40(1): 24. |
| [8] | HUANG Yuting,YING Zuping,ZHENG Jixing,ZHUANG Sigeng,LIU Lu,FENG Wei. Hierarchical Porous ZnO Nanomaterial Synthesized with Corn Straw as Biological Templates and Its Photocatalytic Performance† [J]. Chem. J. Chinese Universities, 2018, 39(9): 2031. |
| [9] | HAN Zhiying, LI Youji, LIN Xiao, WANG Ziyu, LI Ziqin, WANG Hao. Preparation and Photoelectrocatalytic Performance of Fe2O3/ZnO Composite Electrode Loading on Conductive Glass† [J]. Chem. J. Chinese Universities, 2018, 39(4): 771. |
| [10] | XUE Jiao, WANG Runwei, ZHANG Zongtao, QIU Shilun. Preparation and Photocatalytic Performance of Novel Zn-doped C/Nb2O5 Nanoparticles Catalyst† [J]. Chem. J. Chinese Universities, 2018, 39(2): 319. |
| [11] | KANG Hongjun,YU Xiaoyan,LAI Hua,CHENG Zhongjun,LIU Yuyan. Reversible Control of Under-oil Superhydrophobicity to Superhydrophilicity on TiO2 Nanostructured Thin Film† [J]. Chem. J. Chinese Universities, 2018, 39(12): 2621. |
| [12] | LIU Yu,CHEN Gang,ZHU Jiatian,CHEN Wenjin,HU Wen,LIU Yingyao,FANG Zhiqiang. Preparation, Structure and Properties of Strong, Transparent Cellulose Materials† [J]. Chem. J. Chinese Universities, 2018, 39(10): 2298. |
| [13] | HU Qian, DING Yadan, PAN Ying, HONG Xia. Effect of Substrate Surface Wettability on the Adsorption of Magnetic Carrier/Protein Nanocomposites† [J]. Chem. J. Chinese Universities, 2018, 39(1): 124. |
| [14] | SI Wenyan, ZHANG Hongdi, LIU Yanjie, ZHAO Aijing, ZHANG Zhiguang, GONG Maogang, ZHANG Juncheng, LONG Yunze. Fabrication and Pressure Sensing Analysis of ZnO/PVDF Composite Microfiber Arrays by Low-voltage Near-field Electrospinning [J]. Chem. J. Chinese Universities, 2017, 38(6): 997. |
| [15] | SUN Lianzhi, ZHAO Shengzhe, GAO Zhiling, CHENG Zhiqiang. Controllable Synthesis of Ag Decorated ZnO Nanofibers for Enhanced Photocatalysis [J]. Chem. J. Chinese Universities, 2017, 38(6): 907. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||