

Chem. J. Chinese Universities ›› 2014, Vol. 35 ›› Issue (7): 1515.doi: 10.7503/cjcu20140108
• Physical Chemistry • Previous Articles Next Articles
Received:2014-02-14
Online:2014-07-10
Published:2014-04-29
Contact:
WANG Rui
E-mail:ree_wong@hotmail.com
Supported by:CLC Number:
TrendMD:
MA Yunqian, WANG Rui. H2S Absorption Capacity of Ionic Liquid-MDEA-H2O Combined Desulfurizers†[J]. Chem. J. Chinese Universities, 2014, 35(7): 1515.
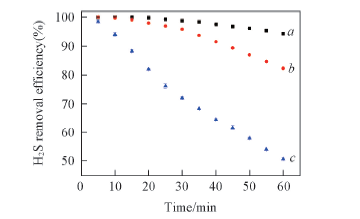
Fig.4 H2S absorption efficiency by [Bmim]Cl-MDEA-H2O combined deoxidizer as a function of time at 28 ℃(a), 60 ℃(b) and 80 ℃(c) Desulfurizer: 10 mL; [Bmim]Cl: 1 g; MDEA: 3 g.
| m(MDEA)/g | m([Bmim]Cl)/g | H2S removal efficiencya(%) | Capacity of sulfurc/(g·L-1) | m(MDEA)/g | m([Bmim]Cl)/g | H2S removal efficiencya(%) | Capacity of sulfurc/(g·L-1) |
|---|---|---|---|---|---|---|---|
| 0 | 0 | 17.6b | 3 | 0.5 | 93.0 | 3.36 | |
| 0 | 10 | 9.8b | 3 | 1 | 94.4 | 3.66 | |
| 0 | 1 | 14.9b | 3 | 1.5 | 95.0 | 3.74 | |
| 1 | 0 | 72.5 | 1.56 | 3 | 2 | 96.2 | 3.95 |
| 1 | 0.5 | 84.0 | 2.21 | 5 | 0 | 89.1 | 3.01 |
| 1 | 1 | 86.8 | 2.53 | 5 | 0.5 | 93.9 | 3.42 |
| 1 | 1.5 | 87.6 | 2.64 | 5 | 1 | 94.8 | 3.68 |
| 1 | 2 | 88.7 | 2.75 | 5 | 1.5 | 96.3 | 4.01 |
| 3 | 0 | 84.8 | 2.42 | 5 | 2 | 97.4 | 4.31 |
Table 1 H2S absorption capacity and sulfur capacity of [Bmim]Cl-MDEA-H2O combined eoxidizer with different component
| m(MDEA)/g | m([Bmim]Cl)/g | H2S removal efficiencya(%) | Capacity of sulfurc/(g·L-1) | m(MDEA)/g | m([Bmim]Cl)/g | H2S removal efficiencya(%) | Capacity of sulfurc/(g·L-1) |
|---|---|---|---|---|---|---|---|
| 0 | 0 | 17.6b | 3 | 0.5 | 93.0 | 3.36 | |
| 0 | 10 | 9.8b | 3 | 1 | 94.4 | 3.66 | |
| 0 | 1 | 14.9b | 3 | 1.5 | 95.0 | 3.74 | |
| 1 | 0 | 72.5 | 1.56 | 3 | 2 | 96.2 | 3.95 |
| 1 | 0.5 | 84.0 | 2.21 | 5 | 0 | 89.1 | 3.01 |
| 1 | 1 | 86.8 | 2.53 | 5 | 0.5 | 93.9 | 3.42 |
| 1 | 1.5 | 87.6 | 2.64 | 5 | 1 | 94.8 | 3.68 |
| 1 | 2 | 88.7 | 2.75 | 5 | 1.5 | 96.3 | 4.01 |
| 3 | 0 | 84.8 | 2.42 | 5 | 2 | 97.4 | 4.31 |
| Desulfurizer | m(MDEA)/g | m(IL)/g | Bubbling height/cm | Efficiency(%) |
|---|---|---|---|---|
| MDEA-H2O | 0.5 | 0 | 22 | |
| MDEA-[Bmim]Cl-H2O | 0.5 | 0.5 | 16.9 | 23.2 |
| MDEA-[Bmim]BF4-H2O | 0.5 | 0.5 | 15.2 | 30.9 |
| MDEA-[TMG]L-H2O | 0.5 | 0.5 | 22.1 | 0 |
| MDEA-[MEA]L-H2O | 0.5 | 0.5 | 20.5 | 6.8 |
| MDEA-[Bmim]HCO3-H2O | 0.5 | 0.5 | 11.8 | 46.4 |
| MDEA-[Bmim]HCO3-H2O | 0.5 | 1.5 | 7.8 | 64.5 |
Table 2 Capacity of ILs on eliminating bubbles
| Desulfurizer | m(MDEA)/g | m(IL)/g | Bubbling height/cm | Efficiency(%) |
|---|---|---|---|---|
| MDEA-H2O | 0.5 | 0 | 22 | |
| MDEA-[Bmim]Cl-H2O | 0.5 | 0.5 | 16.9 | 23.2 |
| MDEA-[Bmim]BF4-H2O | 0.5 | 0.5 | 15.2 | 30.9 |
| MDEA-[TMG]L-H2O | 0.5 | 0.5 | 22.1 | 0 |
| MDEA-[MEA]L-H2O | 0.5 | 0.5 | 20.5 | 6.8 |
| MDEA-[Bmim]HCO3-H2O | 0.5 | 0.5 | 11.8 | 46.4 |
| MDEA-[Bmim]HCO3-H2O | 0.5 | 1.5 | 7.8 | 64.5 |
| Compd. | MDEA | H2S | |||
|---|---|---|---|---|---|
| ΔHc/(kJ·mol-1) | Et/(kJ·mol-1) | ΔH/(kJ·mol-1) | |||
| 1 | -3702.3 | -5393.4 | -59.5 | -5375.8 | -32.3 |
| 2 | -3552.7 | -5243.8 | -30.6 | -5226.2 | -18.9 |
| 3 | -2881.9 | -4573.0 | -34.8 | -4555.4 | -32.7 |
| 4 | -2959.4 | -4650.5 | -14.2 | -4632.9 | -28.1 |
| 5 | -2651.0 | -4342.1 | -4.19 | -4324.5 | 13.0 |
| MDEA | -1691.1 | -3364.6 | -13.4 | ||
| H2O | -320.1 | -1993.6 | -14.7 | ||
Table 3 Energetics data on IL-MDEA , IL-H2S and MDEA-H2S
| Compd. | MDEA | H2S | |||
|---|---|---|---|---|---|
| ΔHc/(kJ·mol-1) | Et/(kJ·mol-1) | ΔH/(kJ·mol-1) | |||
| 1 | -3702.3 | -5393.4 | -59.5 | -5375.8 | -32.3 |
| 2 | -3552.7 | -5243.8 | -30.6 | -5226.2 | -18.9 |
| 3 | -2881.9 | -4573.0 | -34.8 | -4555.4 | -32.7 |
| 4 | -2959.4 | -4650.5 | -14.2 | -4632.9 | -28.1 |
| 5 | -2651.0 | -4342.1 | -4.19 | -4324.5 | 13.0 |
| MDEA | -1691.1 | -3364.6 | -13.4 | ||
| H2O | -320.1 | -1993.6 | -14.7 | ||
| Desulfurizer | c(S | Residue sulfurb/(g·L-1) | Sulfur capacity/(g·L-1) | Regeneration efficiencyc(%) |
|---|---|---|---|---|
| [Bmim]Cl-MDEA-H2O | 0.041 | 0.135 | 2.53 | 94.66 |
| [Bmim]BF4-MDEA-H2O | 0.027 | 0.090 | 1.76 | 94.89 |
| [Bmim]HCO3-MDEA-H2O | 0.036 | 0.120 | 2.28 | 94.74 |
Table 4 Regeneration analysis data of desulfurizer
| Desulfurizer | c(S | Residue sulfurb/(g·L-1) | Sulfur capacity/(g·L-1) | Regeneration efficiencyc(%) |
|---|---|---|---|---|
| [Bmim]Cl-MDEA-H2O | 0.041 | 0.135 | 2.53 | 94.66 |
| [Bmim]BF4-MDEA-H2O | 0.027 | 0.090 | 1.76 | 94.89 |
| [Bmim]HCO3-MDEA-H2O | 0.036 | 0.120 | 2.28 | 94.74 |
| [1] | Zhu G. Y., Dai J. X., Zhang S. C., Li J., Shi D., Wen Z. G., Nat. Gas Geoscience, 2004, 15(2), 166—170 |
| (朱光有, 戴金星, 张水昌, 李剑, 史斗, 文志刚. 天然气地球科学, 2004, 15(2), 166—170) | |
| [2] | Schmid R., Cross J. B., Latimer E. G., Energy Fuels, 2009, 23, 3612—3616 |
| [3] | Ma Y. Q., Yang F., Wang R., Chin. J. Inorg. Chem., 2012, 28(10), 2179—2185 |
| (马云倩, 杨烽, 王睿. 无机化学学报, 2012, 28(10), 2179—2185) | |
| [4] | Cadours R., Roquet D., Perdu G., Ind. Eng. Chem. Res., 2007, 46(1), 233—241 |
| [5] | Zhang J. J., Song H., Bai B., Wang L., Ind. Eng. Prog., 2012, 31(7), 1432—1436 |
| (张静娇, 宋华, 白冰, 王璐. 化工进展, 2012, 31(7), 1432—1436) | |
| [6] | Huttenhuis P. J. G., Agrawal N. J., Hogendoorn J. A., Versteeg G. F., J. Pet. Sci. & Eng., 2007, 55(1/2), 122—134 |
| [7] | Li C., Furst W., Chem. Eng. Sci., 2000, 55(15), 2975—2988 |
| [8] | Qian Z., Xu L. B., Li Z. H., Li H., Guo K., Ind. Eng. Chem. Res., 2010, 49(13), 6196—6203 |
| [9] | Mazloumi S. H., Haghtalab A., Jalili A. H., Shokouhi M., J. Chem. Eng. Data, 2012, 57(10), 2625—2631 |
| [10] | Fouad W. A., Berrouk A. S., Ind. Eng. Chem. Res., 2012, 51(18), 6591—6597 |
| [11] | Sakhaeinia H., Jalili A. H., Taghikhani V., Safekordi A. A., J. Chem. Eng. Data, 2010, 55(12), 5839—5845 |
| [12] | Jou F. Y., Mather A. E., Int. J. Thermophys., 2007, 28, 490—495 |
| [13] | Sakhaeinia H., Taghikhani V., Jalili A. H., Mehdizadeh A., Safekordi A. A., Fluid Phase Equilib., 2010, 298(2), 303—309 |
| [14] | Ghobadi A. F., Taghikhani V., Elliott J. R., J. Phys. Chem. B, 2011, 115(46), 13599—13607 |
| [15] | Tokuda H., Hayamizu K., Ishii K., Susan M. A. B. H., Watanabe M., J. Phys. Chem. B, 2004, 108(42), 16593—16600 |
| [16] | Shang Y., Li H., Zhang S., Xu H., Wang Z., Zhang L., Zhang J., J. Chem. Eng., 2011, 175, 324—329 |
| [17] | Huang J., Riisager A., Berg R. W., Fehrmann R., J. Mol. Catal. A: Chem., 2008, 279(2), 170—176 |
| [18] | Gutowski K. E., Maginn E. J., J. Am. Chem. Soc., 2008, 130(44), 14690—14704 |
| [19] | Guo L. Y., Shi T. J., Li Z., Duan Y. P., Wang Y. G., Chem. J. Chinese Universities, 2008, 29(9), 1901—1907 |
| (郭立颖, 史铁钧, 李忠, 段衍鹏, 王于刚. 高等学校化学学报, 2008, 29(9), 1901—1907) | |
| [20] | Liang D., Xin X., Duan H., Yin Y., Gao H., Lin Y., Xu J., Chem. Res. Chinese Universities, 2008, 24(1), 36—41 |
| [21] | Guo B., Duan E., Zhong Y., Gao L., Zhang X., Zhao D., Energy Fuels, 2011, 25, 159—161 |
| [22] | Heintz Y. J., Sehabiague L., Morsi B. I., Jones K. L., Luebke D. R., Pennline H. W., Energy Fuels, 2009, 23(10), 4822—4830 |
| [23] | Duan E., Guo B., Zhang M., Guan Y., Sun H., Han J., J. Hazard. Mater., 2011, 194, 48—52 |
| [24] | Yusoff R., Aroua M. K., Shamiri A., Ahmady A., Jusoh N. S., Asmuni N. F., Bong L. C., Thee S. H., J. Chem. Eng. Data, 2013, 58(2), 240—247 |
| [25] | Wang Z. L., Xu F., Xing X. L., Ren Z. B., Chem. Ind. & Eng. Prog., 2013, 32(2), 394—399 |
| (王占丽, 徐凡, 邢小林, 任增保. 化工进展, 2013, 32(2), 394—399) | |
| [26] | Pomelli C. S., Chiappe C., Vidis A., Laurenczy G., Dyson P. D., J. Phys. Chem. B, 2007, 111(45), 13014—13019 |
| [27] | Yue Q. F., Wang C. X., Zhang L. N., Ni Y., Jin Y. X., Polymer Degradation and Stability, 2011, 96(4), 399—403 |
| [28] | Wu W., Han B., Gao H., Liu Z., Jiang T., Huang J., Angew. Chem. Int . Ed., 2004, 43(18), 2415—2417 |
| [29] | Ji J. R., Yuan H. T., Wang J. Y., Fu L. L., Du Z. L., Hu Y. Q., Hebei Journal of Industrial Science and Technology, 2010, 27(5) 285—287(纪俊荣, 袁海涛, 王建英, 付林林, 杜振雷, 胡永琪. 河北工业科技, 2010, 27(5), 285—287) |
| [30] | Li R.X., Green Solvent-nthesis and Application of Ionic Liquid, Chemical Industry Press, Beijing, 2004, 20—25 |
| (李汝雄. 绿色溶剂-子液体的合成与应用, 北京: 化学工业出版社, 2004, 20—25) | |
| [31] | Wang X. D., Wu W. Y., Tu G. F., Jiang K. X., Chin. Sci. Bull., 2009, 54(1),21—26)(王晓丹, 吴文远, 涂赣峰, 蒋开喜. 科学通报,, 2009, 54(1), 21—26) |
| [32] | Yu G., Zhang S., Fluid Phase Equilib., 2007, 255, 86—92 |
| [33] | Zhai L. Z., Zhong Q., Du H. C., He C., Wang J., Journal of Chemical Industry and Engineering, 2009, 60(2),450—454(翟林智, 钟秦, 杜红彩, 何川, 王娟. , 2009, 60(2), 450—454) |
| [34] | Ma X. L., Wang X. X., Song C. S., J. Am. Chem. Soc., 2009, 131(16), 5777—5783 |
| [35] | Jin M., Hou Y., Wu W., Ren S., Tian S., Xiao L., Lei Z., J. Phys. Chem. B, 2011, 115(20), 6585—6591 |
| [36] | Tang D. Y., Hu J. P., Lü S. Z., Sun G. F., Zhang Y. Q., Acta Chim. Sinica, 2012, 70(8),943—948(唐典勇, 胡建平, 吕申壮, 孙国峰, 张元勤. 化学学报, 2012, 70(8), 943—948) |
| [37] | Wang Y., Li H., Han S., J. Phys. Chem. B, 2006, 110(48), 24646—24651 |
| (Ed.: V, Z) |
| [1] | HE Hongrui, XIA Wensheng, ZHANG Qinghong, WAN Huilin. Density-functional Theoretical Study on the Interaction of Indium Oxyhydroxide Clusters with Carbon Dioxide and Methane [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220196. |
| [2] | CUI Wei, ZHAO Deyin, BAI Wenxuan, ZHANG Xiaodong, YU Jiang. CO2 Absorption in Composite of Aprotic Solvent and Iron-based Ionic Liquid [J]. Chem. J. Chinese Universities, 2022, 43(8): 20220120. |
| [3] | PENG Kuilin, LI Guilin, JIANG Chongyang, ZENG Shaojuan, ZHANG Xiangping. Research Progress for the Role of Electrolytes in the CO2 Electrochemical Reduction [J]. Chem. J. Chinese Universities, 2022, 43(7): 20220238. |
| [4] | JI Shuangqi, JIN Zhao, GUAN Wenna, PAN Xiangyu, GUAN Tong. Preparation and Chromatographic Performance of Mixed-mode Silica Stationary Phase Modified by Double Cationic Ionic Liquid and Octadecyl Group [J]. Chem. J. Chinese Universities, 2022, 43(6): 20220008. |
| [5] | WONG Honho, LU Qiuyang, SUN Mingzi, HUANG Bolong. Rational Design of Graphdiyne-based Atomic Electrocatalysts: DFT and Self-validated Machine Learning [J]. Chem. J. Chinese Universities, 2022, 43(5): 20220042. |
| [6] | LIU Yang, LI Wangchang, ZHANG Zhuxia, WANG Fang, YANG Wenjing, GUO Zhen, CUI Peng. Theoretical Exploration of Noncovalent Interactions Between Sc3C2@C80 and [12]Cycloparaphenylene Nanoring [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220457. |
| [7] | CHANG Sihui, CHEN Tao, ZHAO Liming, QIU Yongjun. Thermal Degradation Mechanism of Bio-based Polybutylactam Plasticized by Ionic Liquids [J]. Chem. J. Chinese Universities, 2022, 43(11): 20220353. |
| [8] | WANG Yuanyue, AN Suosuo, ZHENG Xuming, ZHAO Yanying. Spectroscopic and Theoretical Studies on 5-Mercapto-1,3,4-thiadiazole-2-thione Microsolvation Clusters [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220354. |
| [9] | CHENG Yuanyuan, XI Biying. Theoretical Study on the Fragmentation Mechanism of CH3SSCH3 Radical Cation Initiated by OH Radical [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220271. |
| [10] | ZHOU Chengsi, ZHAO Yuanjin, HAN Meichen, YANG Xia, LIU Chenguang, HE Aihua. Regulation of Silanes as External Electron Donors on Propylene/butene Sequential Polymerization [J]. Chem. J. Chinese Universities, 2022, 43(10): 20220290. |
| [11] | HUANG Luoyi, WENG Yueyue, HUANG Xuhui, WANG Chaojie. Theoretical Study on the Structures and Properties of Flavonoids in Plantain [J]. Chem. J. Chinese Universities, 2021, 42(9): 2752. |
| [12] | MA Lijuan, GAO Shengqi, RONG Yifei, JIA Jianfeng, WU Haishun. Theoretical Investigation of Hydrogen Storage Properties of Sc, Ti, V-decorated and B/N-doped Monovacancy Graphene [J]. Chem. J. Chinese Universities, 2021, 42(9): 2842. |
| [13] | ZHONG Shengguang, XIA Wensheng, ZHANG Qinghong, WAN Huilin. Theoretical Study on Direct Conversion of CH4 and CO2 into Acetic Acid over MCu2Ox(M = Cu2+, Ce4+, Zr4+) Clusters [J]. Chem. J. Chinese Universities, 2021, 42(9): 2878. |
| [14] | ZHENG Ruoxin, ZHANG Igor Ying, XU Xin. Development and Benchmark of Lower Scaling Doubly Hybrid Density Functional XYG3 [J]. Chem. J. Chinese Universities, 2021, 42(7): 2210. |
| [15] | LIU Yang, LI Qingbo, SUN Jie, ZHAO Xian. Direct Synthesis of Graphene on AlN Substrates via Ga Remote Catalyzation [J]. Chem. J. Chinese Universities, 2021, 42(7): 2271. |
| Viewed | ||||||
|
Full text |
|
|||||
|
Abstract |
|
|||||
