

高等学校化学学报 ›› 2026, Vol. 47 ›› Issue (5): 20260066.doi: 10.7503/cjcu20260066
收稿日期:2026-02-03
出版日期:2026-05-10
发布日期:2026-03-31
通讯作者:
秦毅,王东
E-mail:qinyi@szu.edu.cn;wangd@szu.edu.cn
基金资助:
ZENG Xianping, QIN Yi( ), WANG Dong(
), WANG Dong( )
)
Received:2026-02-03
Online:2026-05-10
Published:2026-03-31
Contact:
QIN Yi, WANG Dong
E-mail:qinyi@szu.edu.cn;wangd@szu.edu.cn
Supported by:摘要:
外科手术作为治愈重大疾病的主要手段, 在临床治疗中具有重要价值. 然而, 传统手术方式主要依赖外科医生的视觉观察与经验判断, 在病灶边界识别和切除过程中易出现偏差, 可能导致术后并发症. 因此, 开发具有高灵敏度与高特异性的手术导航工具已成为现代医学领域亟待解决的关键问题. 荧光成像技术因其非侵入性、 高灵敏度、 操作简便以及可实时在分子层面检测生物分析物等优势, 已成为手术导航强有力的工具. 荧光探针作为荧光成像的分子基础, 受到了广泛关注. 尽管已经有结构多样的荧光探针被开发, 但目前的荧光探针普遍存在聚集状态荧光易猝灭、 光稳定性差及波长较短等不足. 聚集诱导发光(AIE)探针凭借其明亮的聚集态发光、 优异的光稳定性和广泛可调的发射波长等特性, 正成为该领域新的研究热点. 本文综合评述了近年来AIE探针在手术导航方面的研究进展, 着重探讨其分子设计策略及具体实施案例, 并依据发射波长将其分为可见光/近红外一区与近红外二区两类进行阐述. 最后, 对AIE探针在手术导航领域的未来发展趋势与应用前景作出了展望.
中图分类号:
TrendMD:
曾宪平, 秦毅, 王东. 聚集诱导发光探针用于手术导航. 高等学校化学学报, 2026, 47(5): 20260066.
ZENG Xianping, QIN Yi, WANG Dong. Aggregation-induced Emission Probes for Imaging-guided Surgery. Chem. J. Chinese Universities, 2026, 47(5): 20260066.
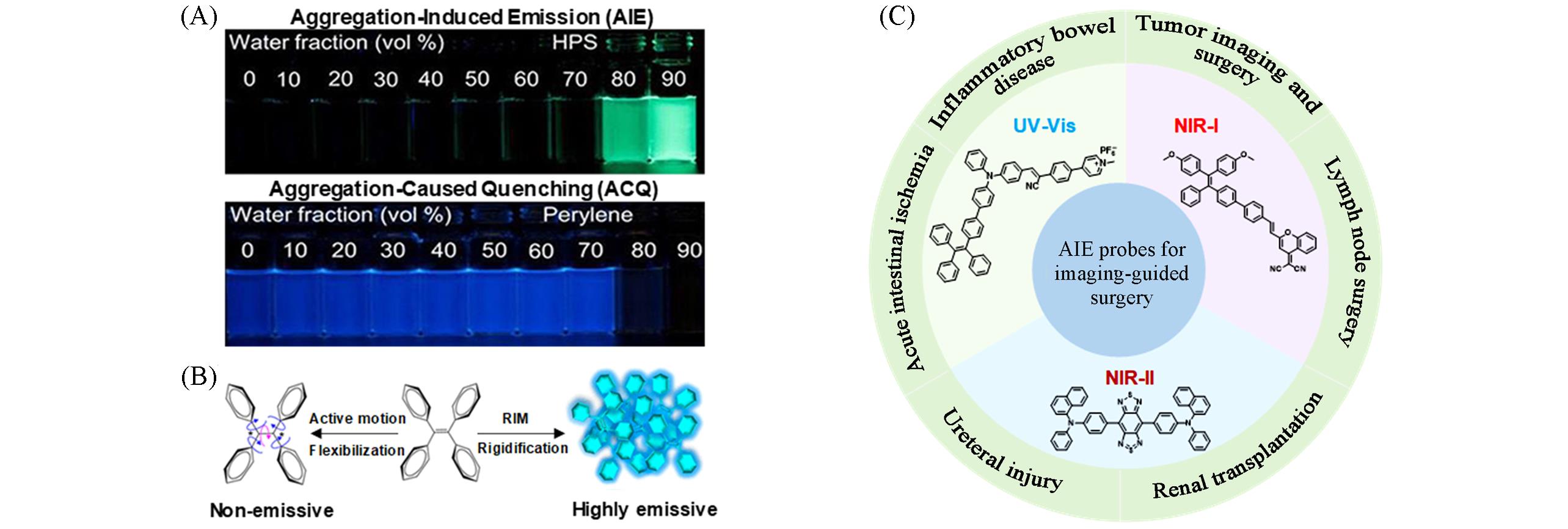
Fig.1 Phenomenon and mechanism of aggregation⁃induced emission(AIE) and AIE probes for imaging⁃guided surgery(A) The phenomenon of aggregation-induced emission; (B) the mechanism of aggregation-induced emission[8]; (C) illustration of the classification of AIE probes by emission wavelengths, the representative chemical structures of AIE probes at certain wavelength and practical surgical navigation applications.Copyright 2015, American Chemical Society
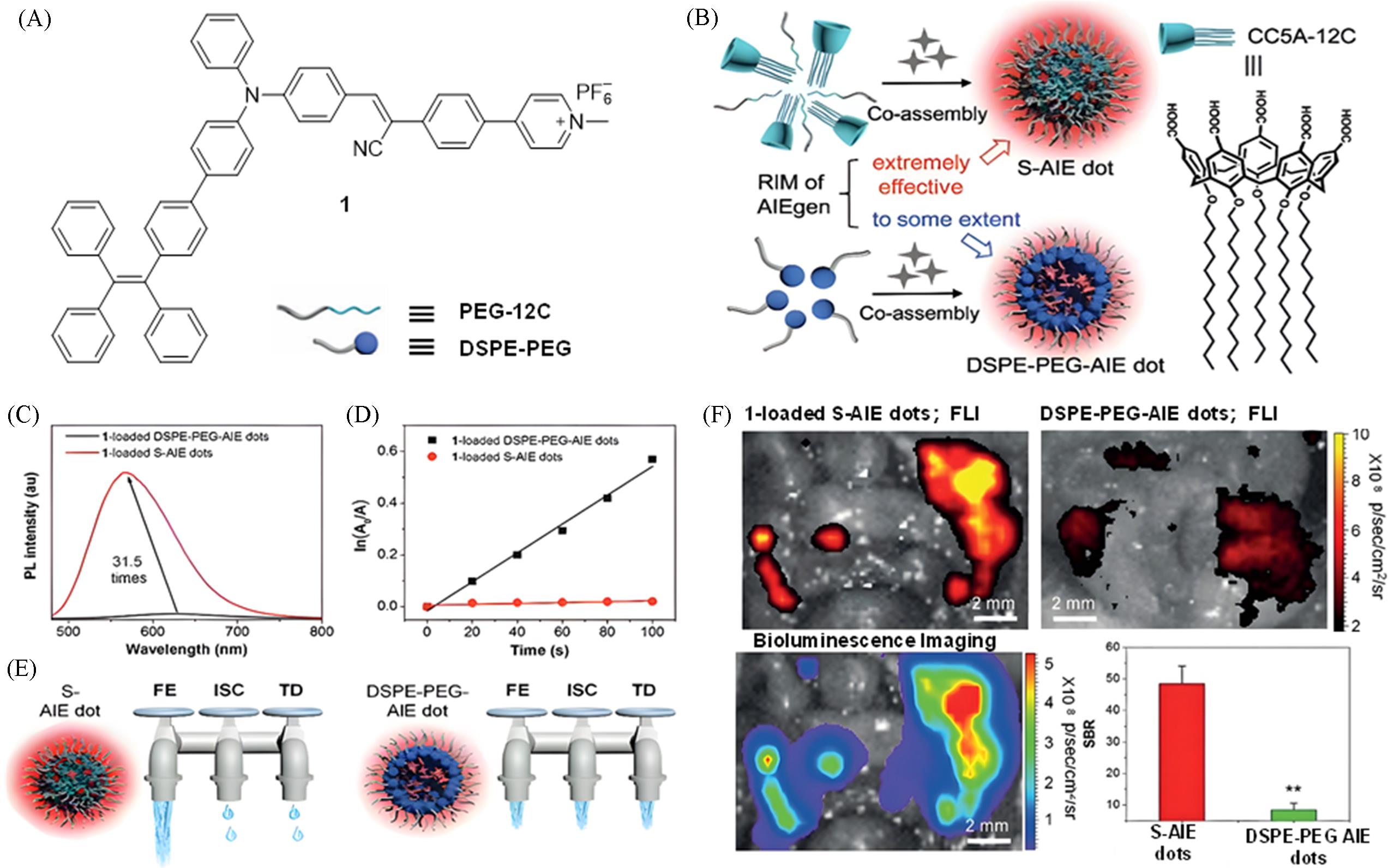
Fig.2 Construction of a highly bright visible⁃light fluorescent probe via host⁃guest complexation of calix[5]arene and AIEgen for peritoneal cancer surgical navigation[12](A) Chemical structure of AIE molecule 1; (B) illustration of the formation of S-AIE dots and DSPE-PEG-AIE dots, and chemical structure of CC5A-12C; (C) comparison of the fluorescence intensity of 1-loaded S-AIE dots and DSPE-PEG-AIE dots in water (λex=445 nm); (D) comparison of the singlet oxygen generation ability of S-AIE dots and DSPE-PEG-AIE dots; (E) illustration of the different dissipation pathways of the absorbed excitation energy for S-AIE dots and DSPE-PEG-AIE dots, which are represented as three water taps; FE: fluorescence emission; TD: thermal deactivation; ISC: intersystem crossing; (F) fluorescence imaging, SBR comparison, and in vivo tumor visualization of S-AIE dots and DSPE-PEG-AIE dots. Copyright 2020, Wiley-VCH GmbH.
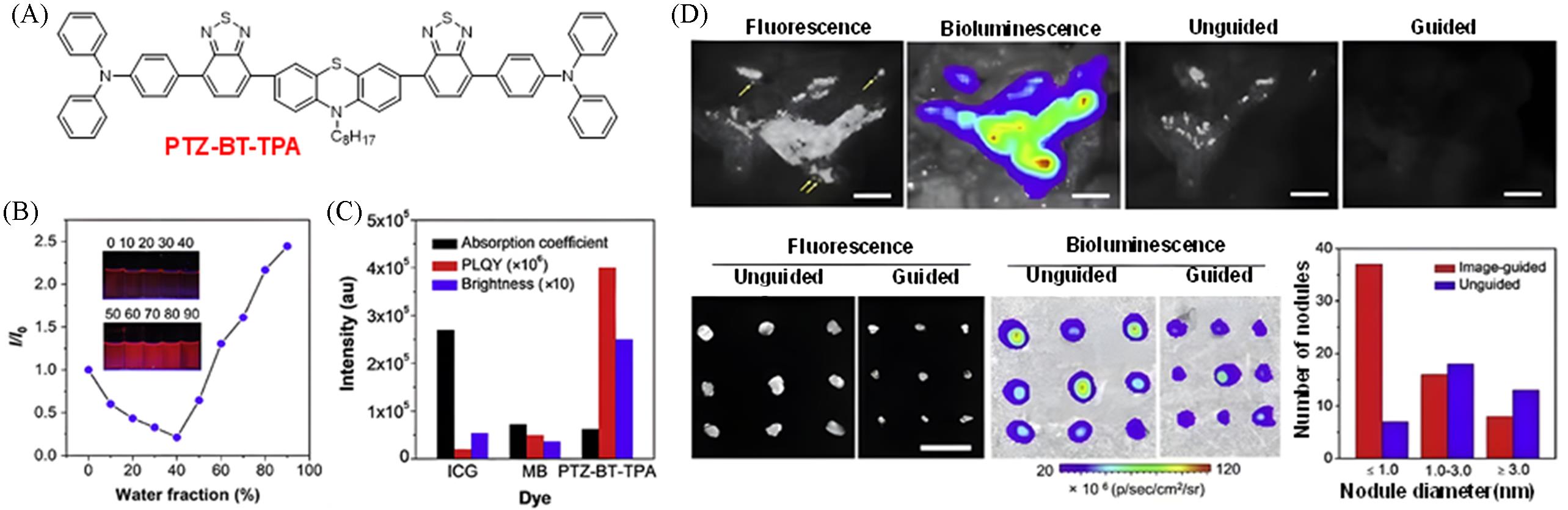
Fig.3 Construction of a highly bright NIR⁃I fluorescent probe via planar conjugated skeleton + vibrational substituents for peritoneal cancer surgical navigation[14](A) Chemical structure of PTZ-BT-TPA; (B) variation of photoluminescence intensity of PTZ-BT-TPA in THF/water mixed solvents with different water fractions(fw); the inset shows photographs of PTZ-BT-TPA in THF/water mixed solutions with different water volume fractions(%) under 365 nm UV light irradiation in the dark; (C) comparative analysis of absorption coefficient, PLQY and brightness of ICG, MB and PTZ-BT-TPA in water; (D) comparison of fluorescence/bioluminescence imaging results and postoperative outcomes between non-guided and fluorescence-guided surgical approaches. Copyright 2020, Elsevier B.V.
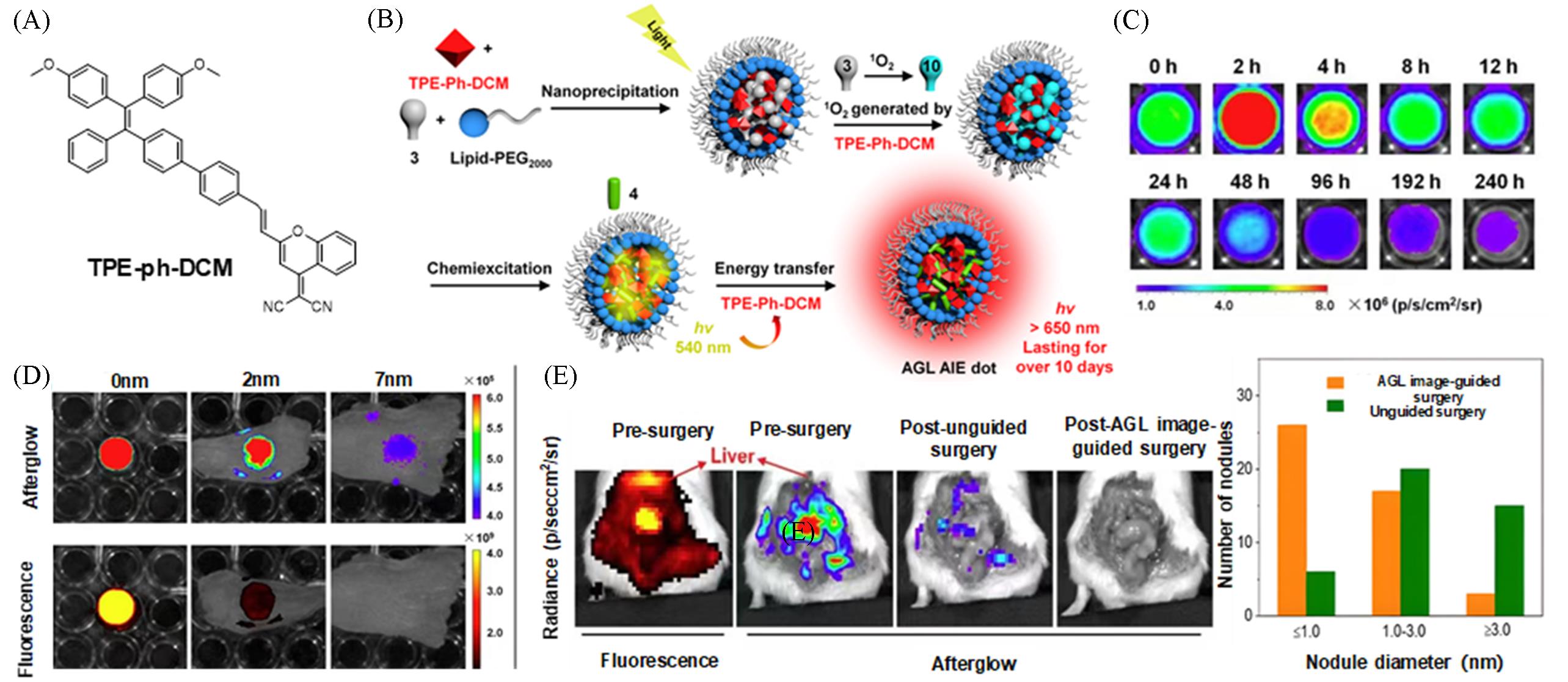
Fig.4 Construction of a highly bright NIR⁃I fluorescent probe via near⁃infrared afterglow luminescence for peritoneal cancer surgical navigation[18](A) Chemical structures of the TPE-Ph-DCM; (B) schematic of the mechanism underlying amplified NIR afterglow luminescence of AGL AIE dots; (C) NIR afterglow decay images of AGL AIE dots in PBS at 37 °C, captured at different time intervals after 2 min of pre-irradiation using white light at an intensity of 0.2 W/cm²; (D) NIR afterglow imaging and NIR fluorescence imaging(λex=465 nm) of AGL AIE dots covered by chicken tissues of varying thicknesses; [10-dot sample]=100 μmol/L(based on compound 3); [AGL AIE dots]=100 μmol/L(based on compound 3, with [compound 3]/[TPE-Ph-DCM]=2∶1); (E) comparison of afterglow imaging before and after surgeries and in the number of tumors resected. Copyright 2019, American Chemical Society.
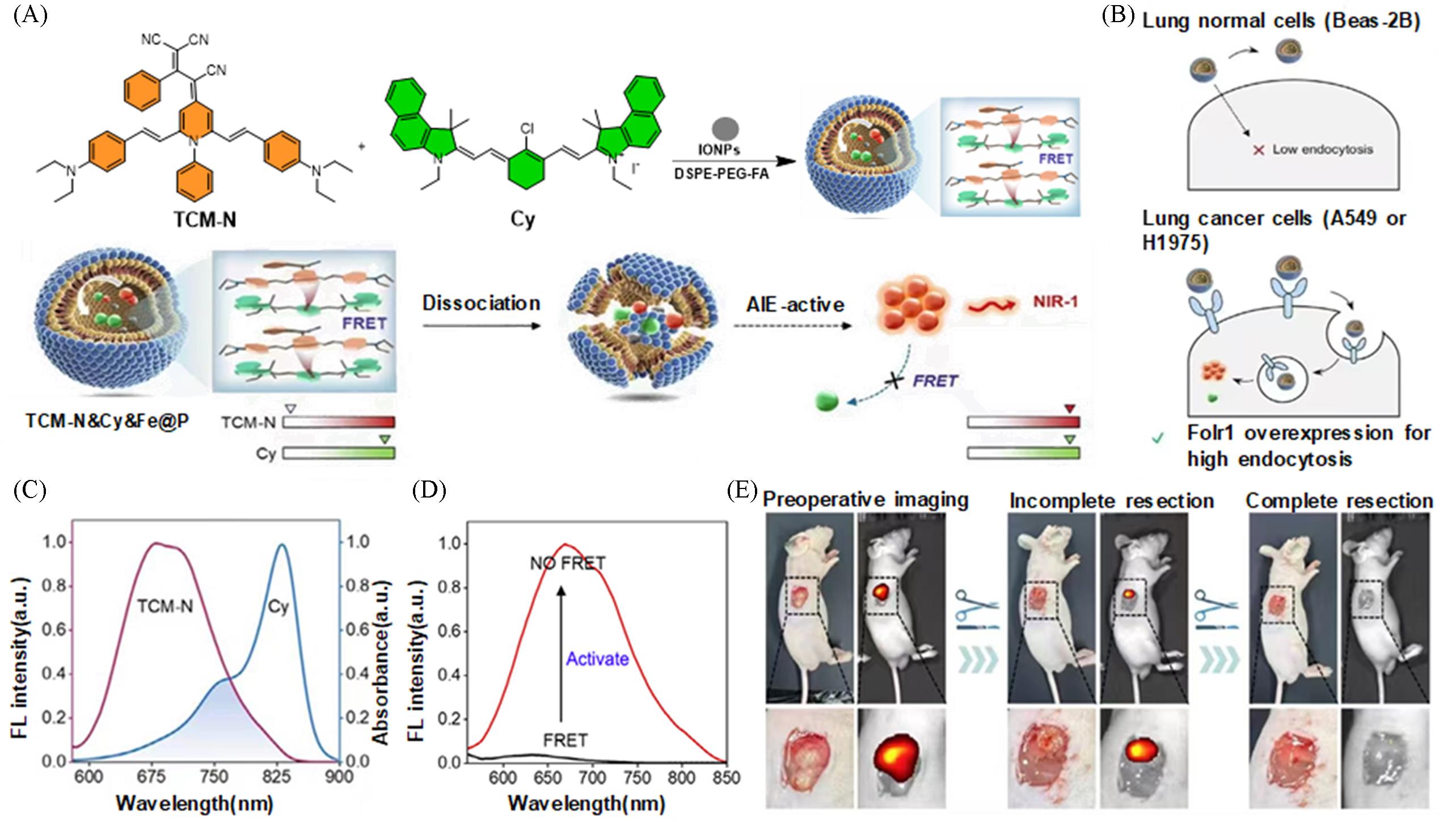
Fig.5 Highly bright NIR⁃I fluorescent probe for lung cancer surgical navigation[19](A) Representative molecules and mechanisms of AIE-active compounds; (B) dual-channel confocal images of Beas-2B cells and A549 cells; (C) overlap between the fluorescence spectrum of TCM-N and the absorption spectrum of Cy; (D) fluorescence activation of TCM-N from the assembled state(TCM-N&Cy&Fe@P) to the disassembled state; (E) near-infrared imaging-guided surgery in tumor-bearing mice. Copyright 2025, Wiley-VCH GmbH.
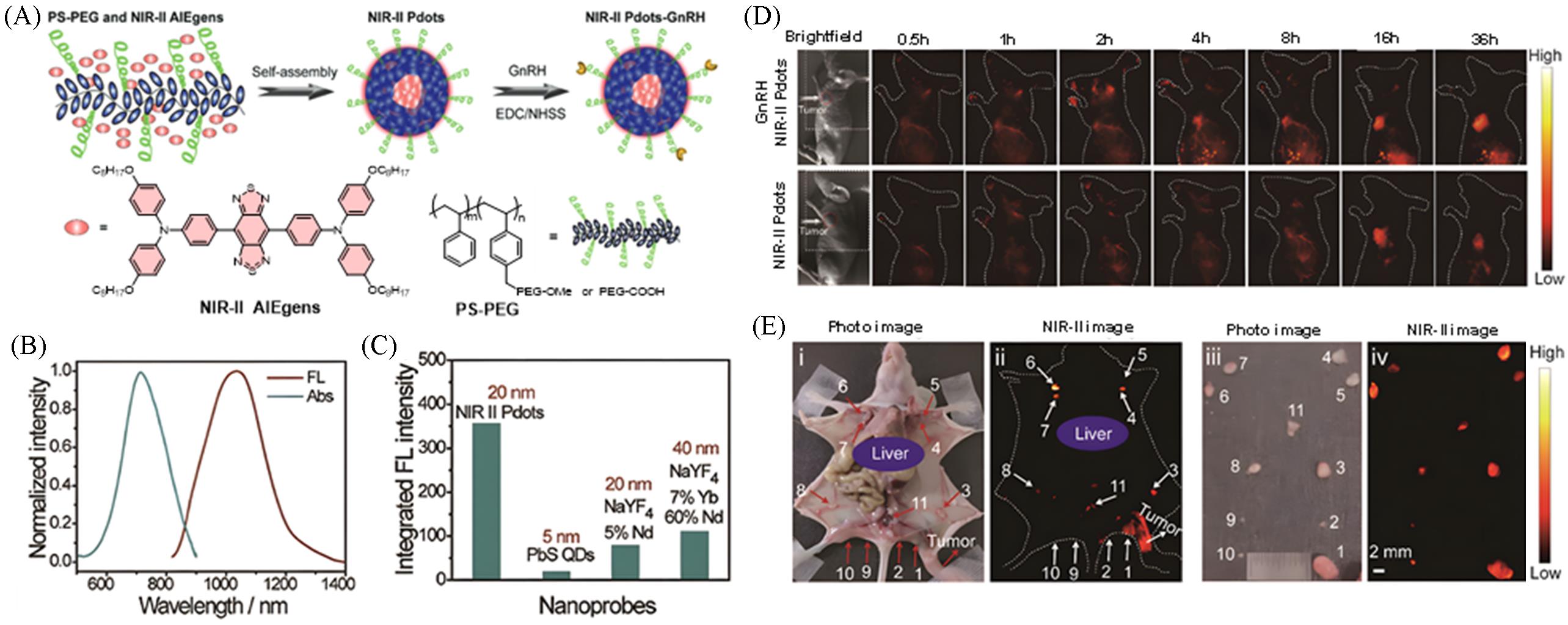
Fig.6 Ultra⁃bright NIR⁃II fluorescent probe for ovarian cancer surgical navigation[21](A) Preparation method of NIR-II Pdots-GnRH; (B) UV-Vis absorption and fluorescence spectra of NIR-II polymer dots(Pdots) in water; (C) comparison of NIR-II fluorescence intensities of equimolar nanoparticles; (D) in vivo fluorescence observation of NIR-II Pdots and targeted probes in tumor-bearing mice; (E) in vivo and ex vivo tracing of ovarian cancer lymphatic metastasis using NIR-II Pdots-GnRH. Copyright 2021, Wiley-VCH GmbH.
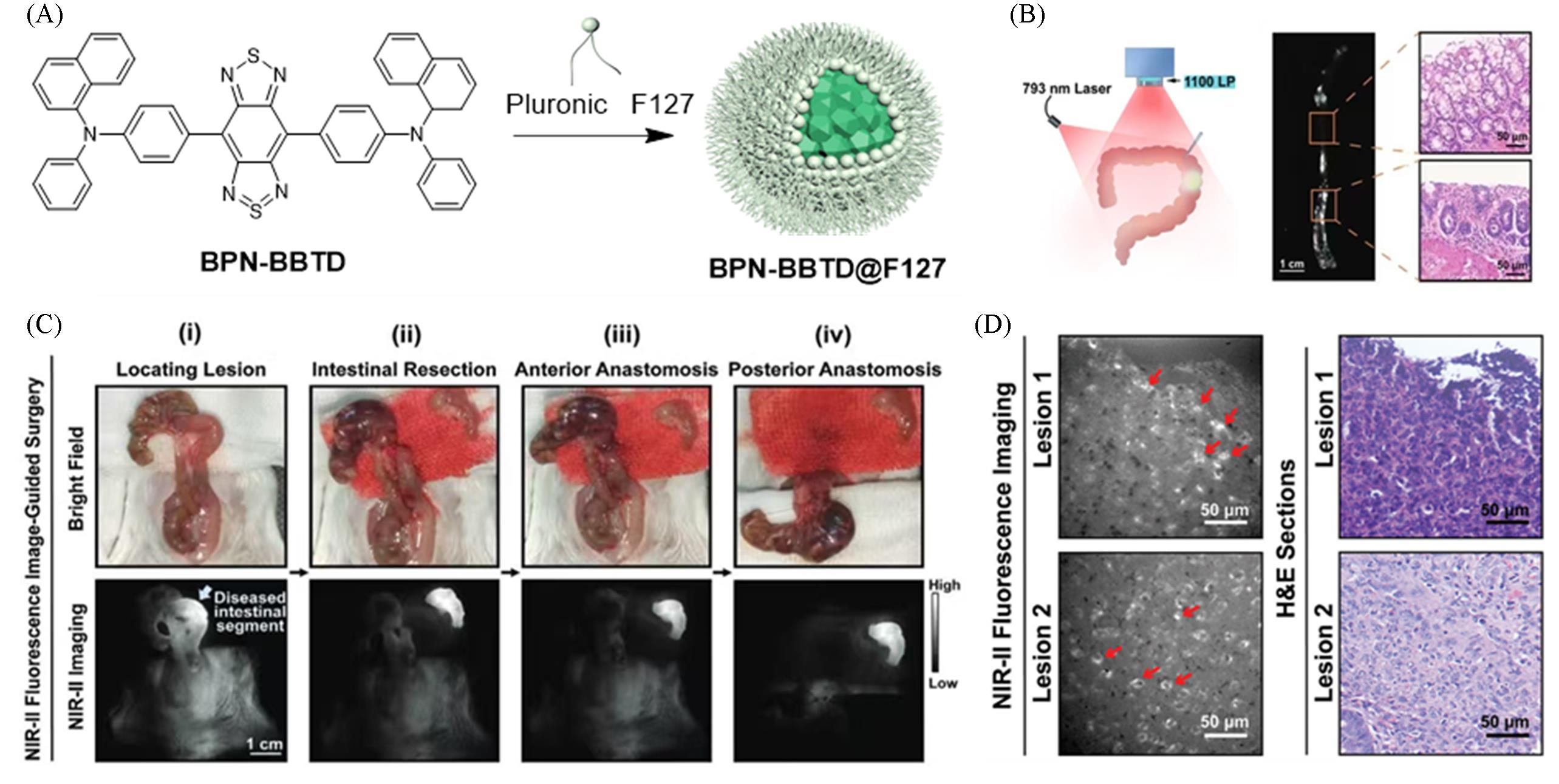
Fig.7 Highly bright NIR⁃II fluorescent probe for the treatment of inflammatory bowel disease[24](A) Chemical molecular formula of BPN-BBTD and preparation method of BPN-BBTD@F127; (B) precise guidance of intestinal surgery via near-infrared fluorescence imaging(scale bar: 50 μm); (C) (i) NIR-II intense fluorescence intestinal segment accurately localized under excitation with a 793 nm laser; (ii) intestinal resection; (iii) anterior anastomosis procedure; (iv) posterior anastomosis procedure(scale bar: 1 cm; light blue arrows: lesioned intestinal segments); laser power density of 793 nm during surgery: 25 mW/cm²; exposure time: 20 ms; (D) two selected lesions observed by high spatial-resolution NIR-II fluorescence wide-field microscopy and corresponding H&E staining results(scale bar: 50 μm). Copyright 2021, Wiley-VCH GmbH.
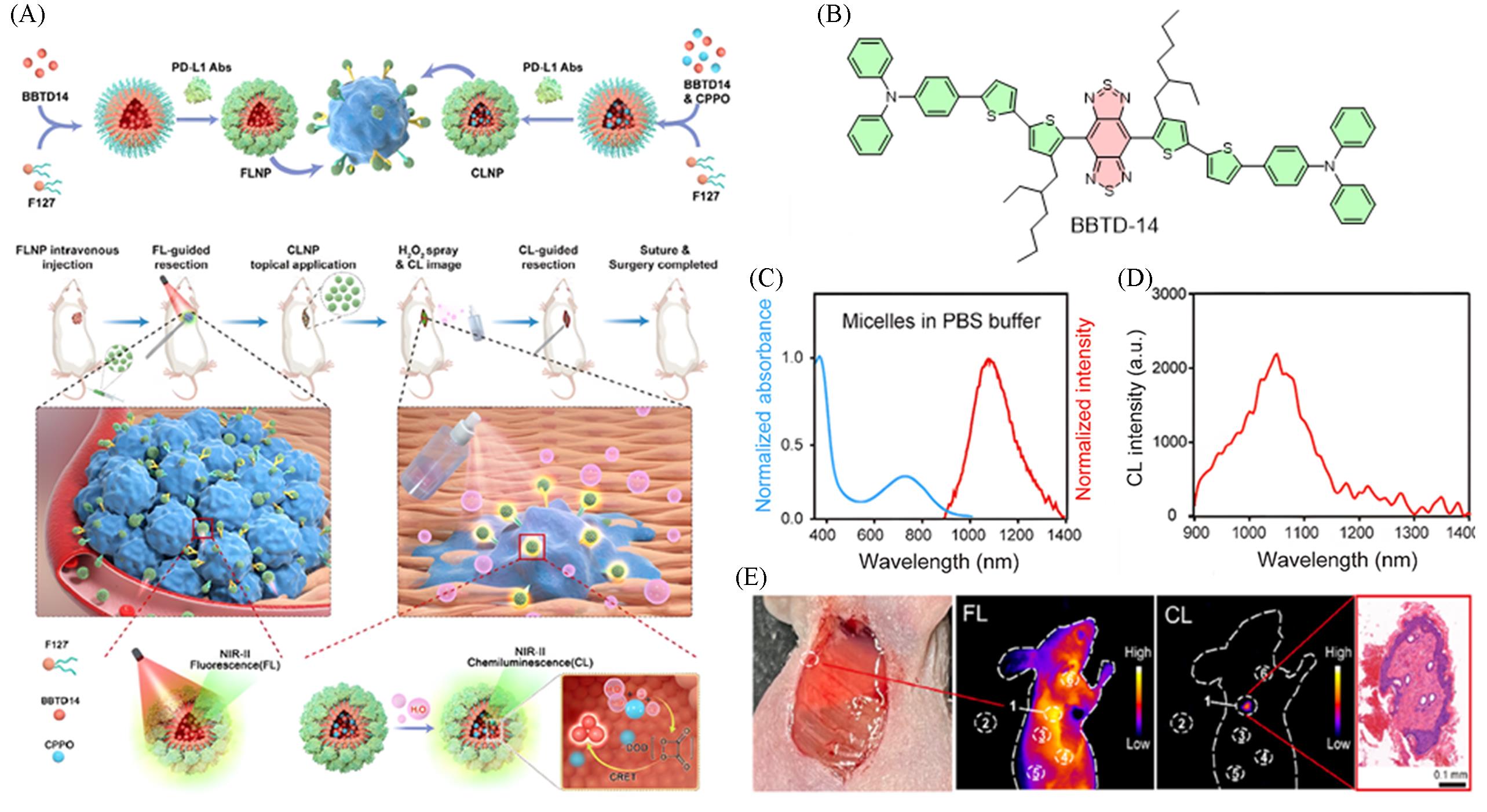
Fig.8 Achieving osteosarcoma surgical navigation via fluorescence⁃chemiluminescence synergistic imaging[25](A) Schematic illustration of the synthesis process of BBTD-14 nanoparticles and the nanosystem for synergistic imaging-guided osteosarcoma resection; (B) chemical structure of BBTD-14; (C) FLNP normalized absorption and emission spectra in PBS; (D) chemiluminescence spectrum of CLNP in the presence of H2O2; (E) synergistic fluorescence-chemiluminescence imaging for verifying complete tumor resection. Copyright 2024, Wiley-VCH GmbH.
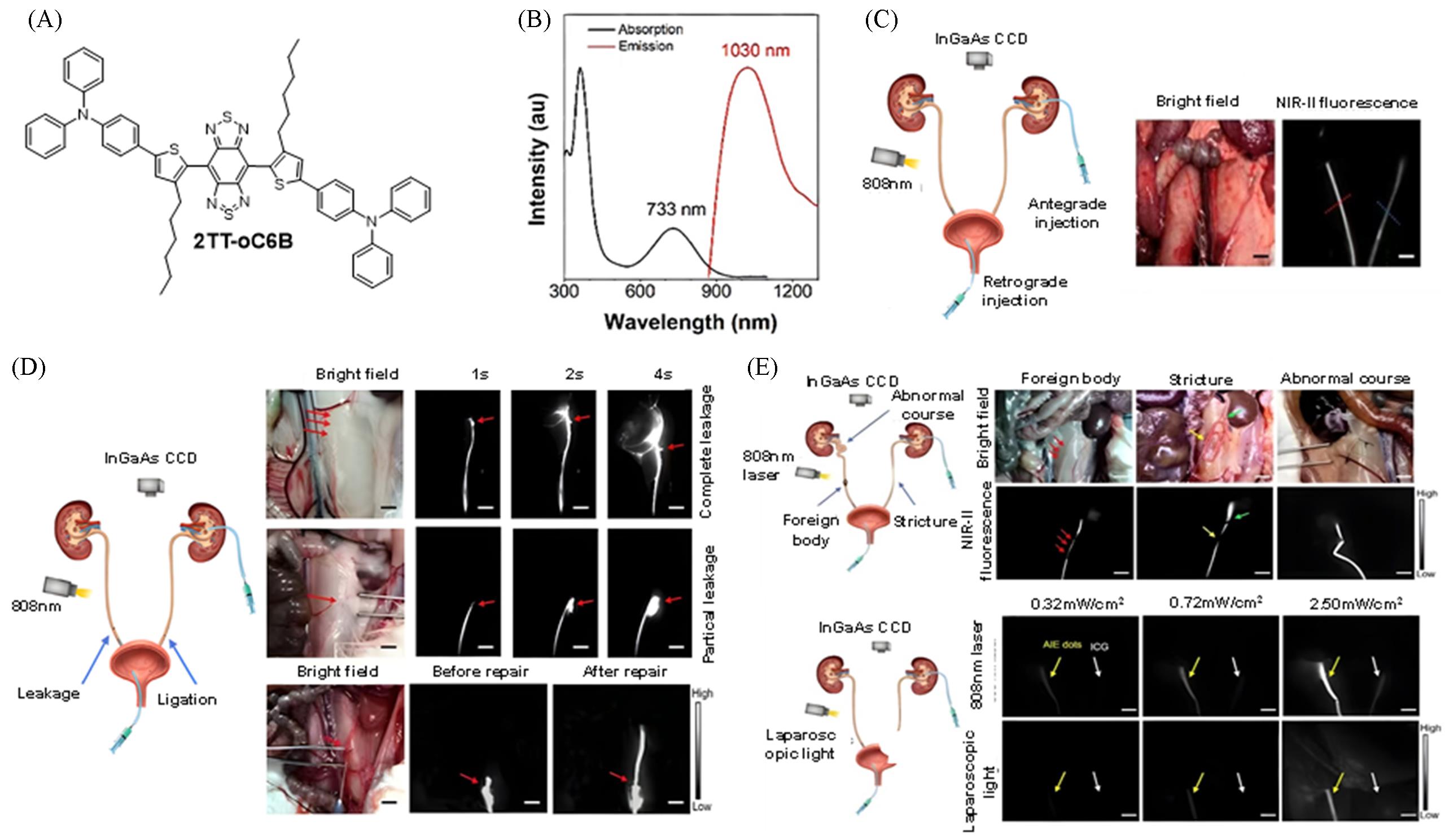
Fig.9 Highly bright nanoparticle probe for surgical navigation in the treatment of ureteral injury[27](A) Chemical structure of 2TT-oC6B; (B) absorption and emission curves of 2TT-oC6B dots in deionized water; (C) NIR imaging effect of rabbit ureter (left: 2TT-oC6B dots; right: ICG); (D) fluorescence imaging localization and efficacy evaluation of rabbit ureteral injury model; (E) application of NIR-II fluorescence imaging in the detection of common ureteral diseases and excitation under laparoscopic xenon light. Copyright 2020, American Chemical Society.
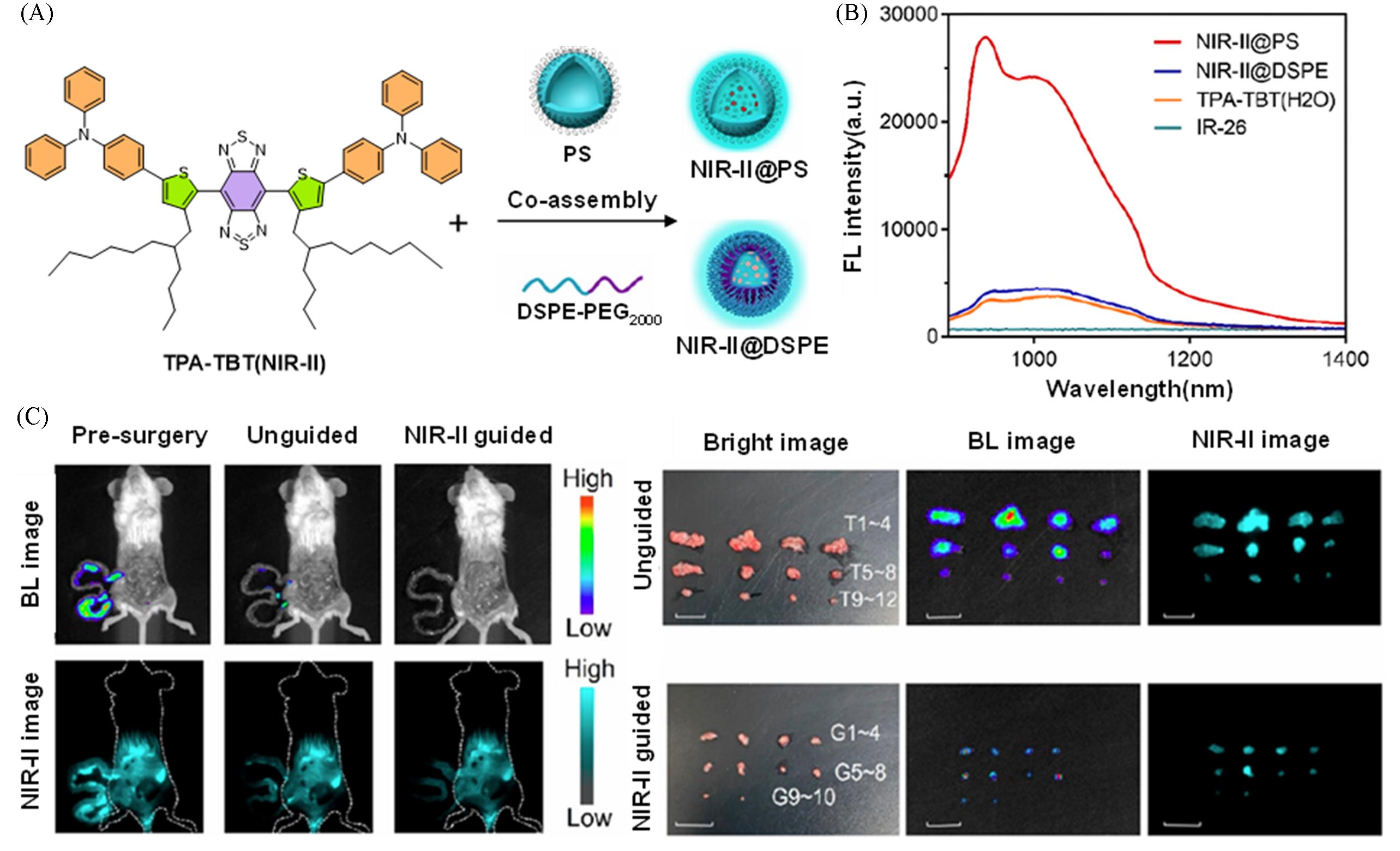
Fig.10 Construction of an ultra⁃bright NIR⁃II nanoprobe via rigid polymercarrier optimization for precise resection of micrometastases[30](A) Preparation methods of NIR-II@PS and NIR-II@DSPE; (B) NIR-I emission spectra of NIR-II@PS and NIR-II@DSPE in water under excitation at 808 nm; (C) precise surgery for micro-nodules guided by NIR-II@PS imaging. Copyright 2024, American Chemical Society.
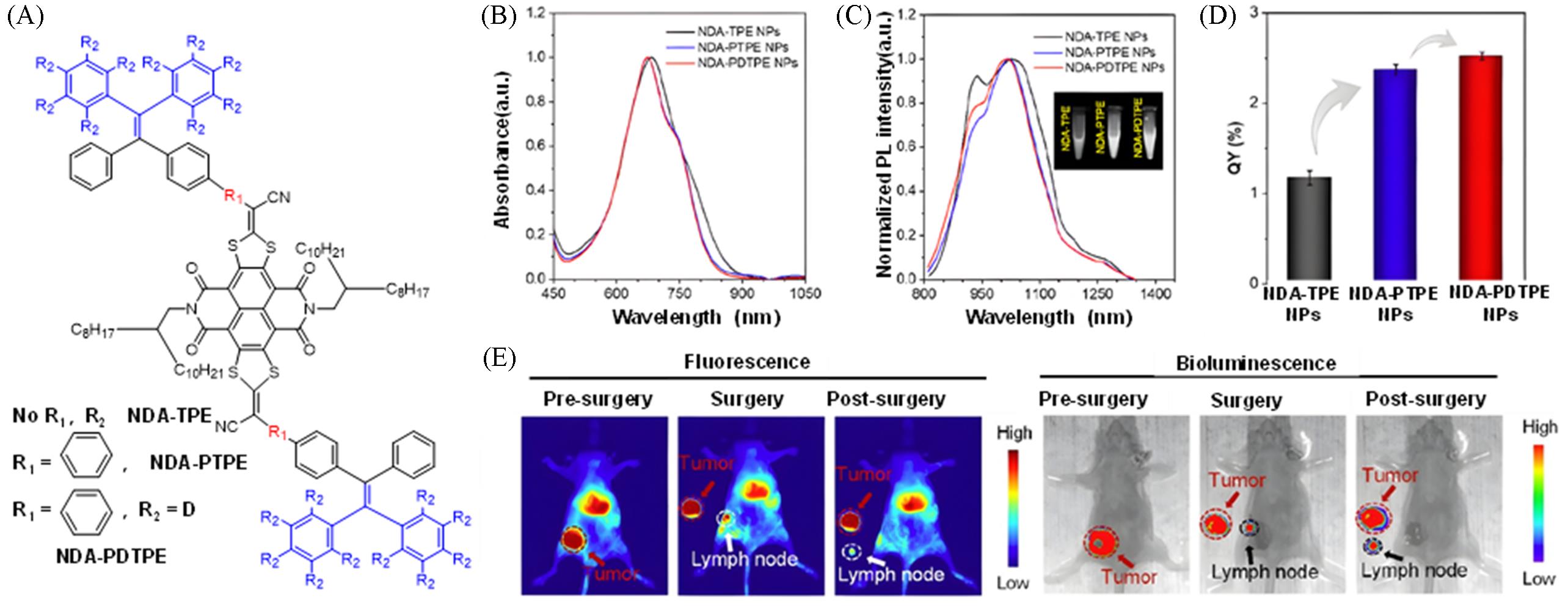
Fig.11 Construction of a high luminescent efficiency fluorescent probe via "π⁃conjugation extension + deuteration" for fluorescence⁃photoacoustic dual⁃modal guided sentinel lymph node surgery[32](A) Chemical structures of NDA-TPE, NDA-PTPE and NDA-PDTPE; (B) absorption spectra of NDA-TPE/PTPE/PDTPE nanoparticles; (C) emission spectra of NDA-TPE/PTPE/PDTPE nanoparticles; (D) photoluminescence quantum yields of NDA-TPE, NDA-PTPE and NDA-PDTPE nanoparticles; (E) precise surgical procedures and lesion localization guided by NIR-II and bioluminescence dual-modal imaging. Copyright 2024, American Chemical Society.
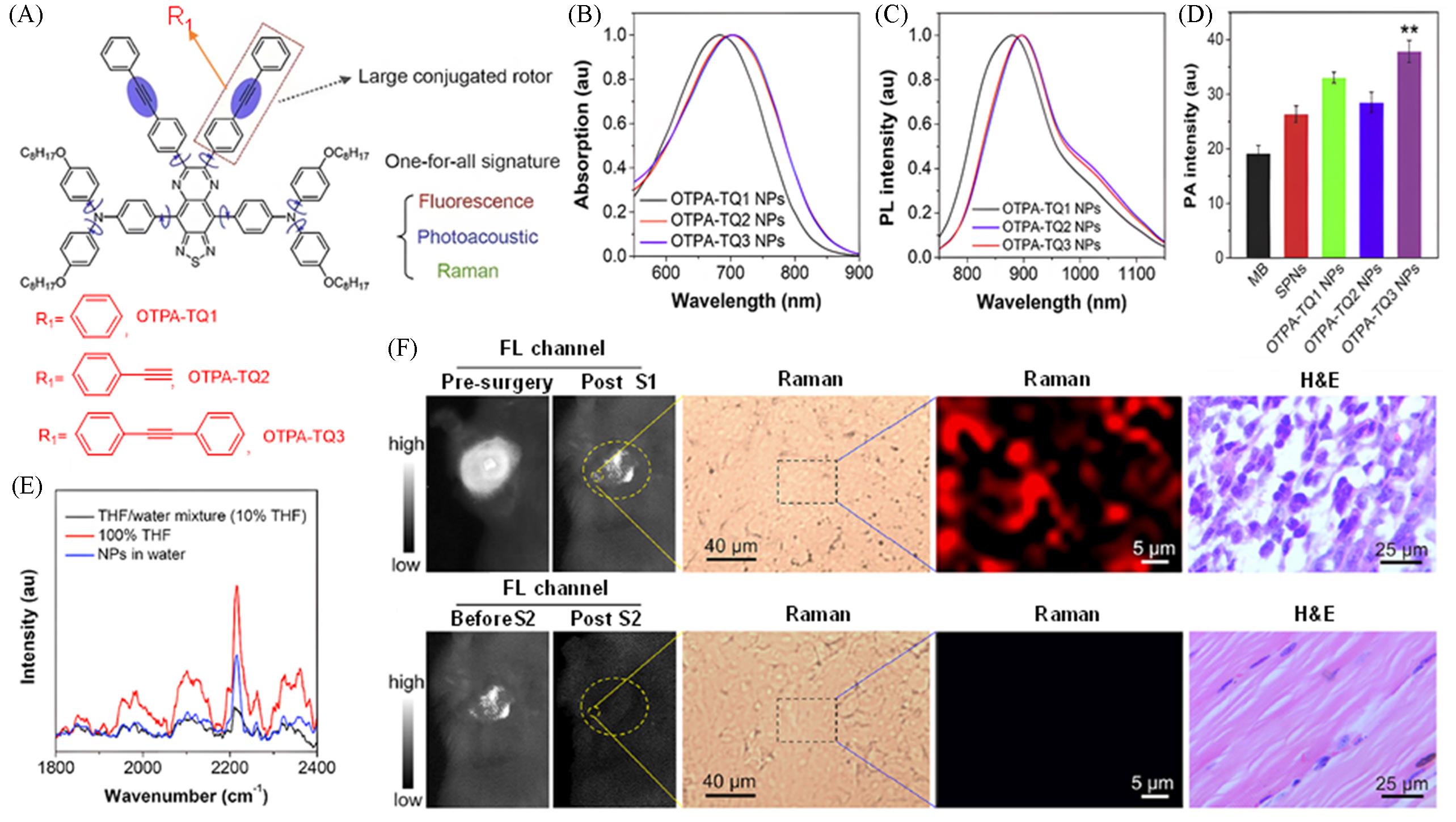
Fig.12 Fluorescence⁃photoacoustic⁃Raman trimodal synergistic imaging probe for surgical navigation of breast cancer[33](A) Chemical structures of OTPA-TQ1, OTPA-TQ2 and OTPA-TQ3; (B) OTPA-TQ1/2/3 nanoparticle solution absorption spectra; (C) emission spectra of OTPA-TQ1, OTPA-TQ2 and OTPA-TQ3 nanoparticle solutions; (D) comparison of PA intensities of OTPA-TQ1-3, SPNs and MB at 680 nm under equal concentration conditions; (E) Raman spectroscopy was performed on OTPA-TQ1-3 nanoparticles; (F) procedure and efficacy evaluation of precise tumor resection guided by intraoperative fluorescence-Raman dual-modal imaging. Copyright 2020, Wiley-VCH GmbH.
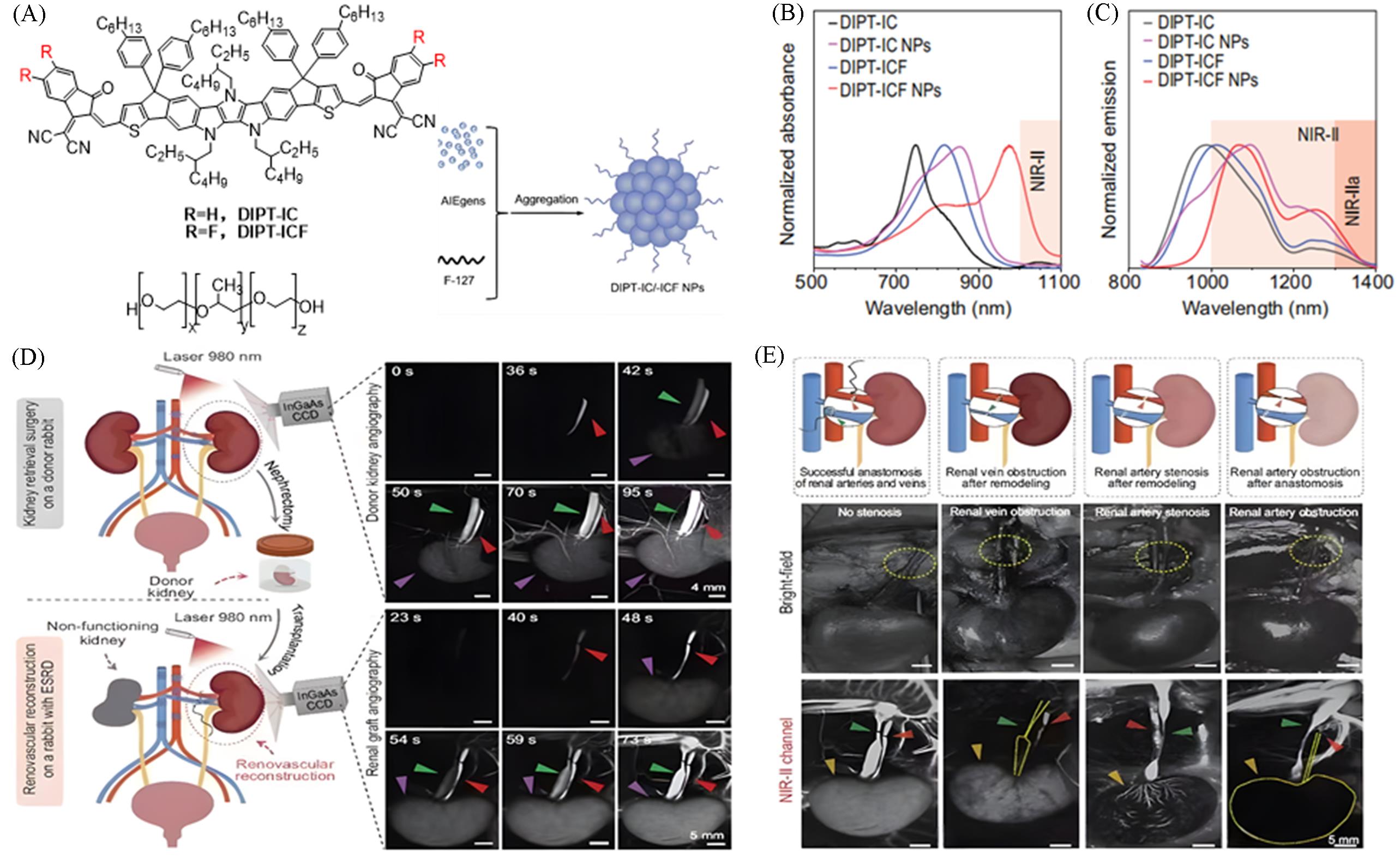
Fig.13 Achieving full⁃process monitoring, evaluation and surgical navigation of renal transplantation via a highly bright NIR⁃II AIE nanocontrast agent[37](A) Chemical structures of DIPT-IC and DIPT-ICF, and preparation methods of their nanoparticles(NPs); (B) absorption spectra of DIPT-IC/ICF and their NPs in THF solution; (C) emission spectra of DIPT-IC/ICF in THF and their NPs in deionized H2O; (D) real-time monitoring of the entire rabbit kidney transplantation process from renal vessel assessment to post-transplantation blood flow reperfusion via NIR-II fluorescence imaging based on DIPT-ICF NPs; (E) real-time monitoring of vascular anastomosis complications in a rabbit orthotopic kidney transplantation model using NIR-II angiography. Copyright 2023, Springer Nature.
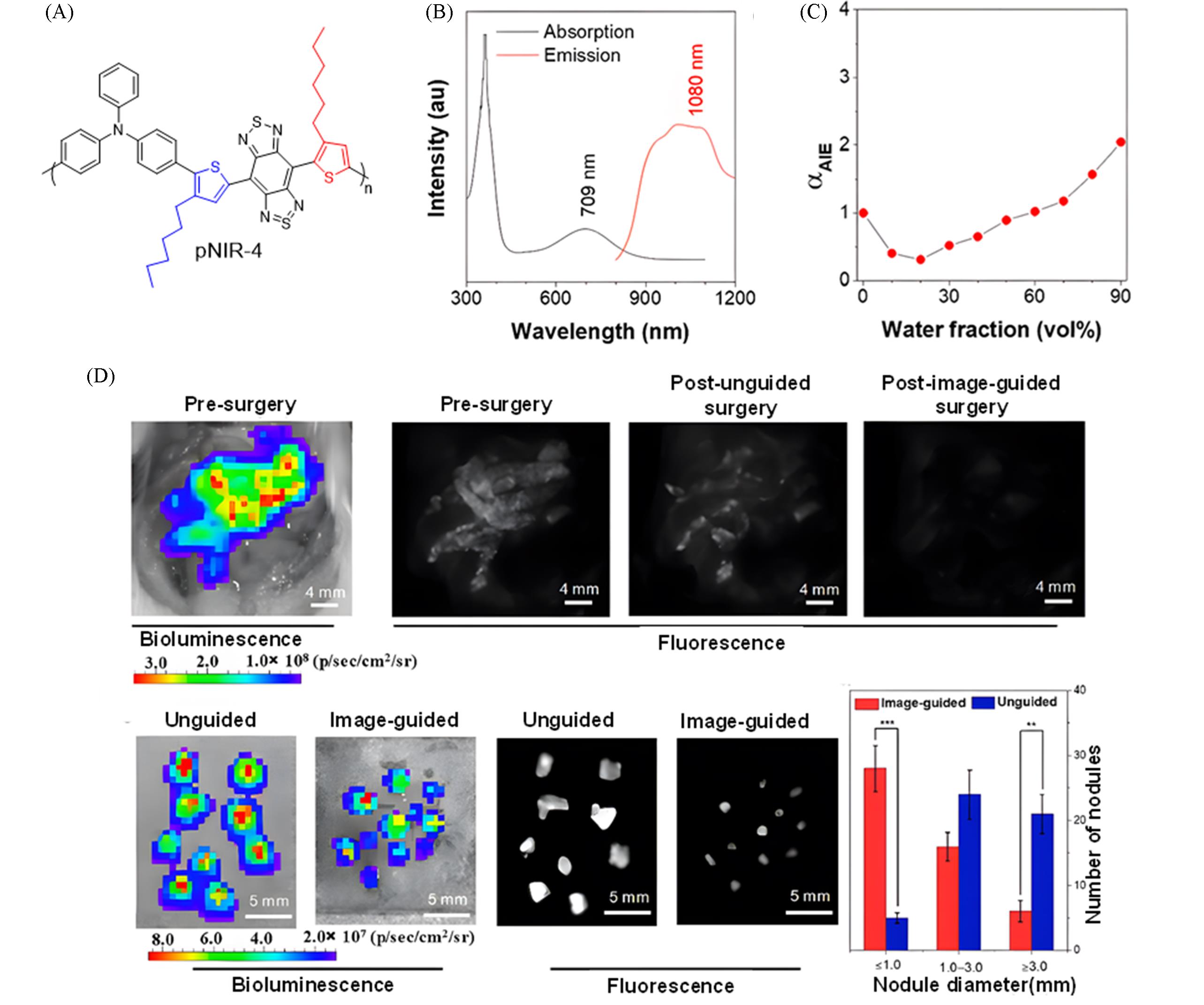
Fig.14 Construction of a highly bright NIR⁃II fluorescent probe via plane⁃twisted structure fusion design for surgical navigation of peritoneal metastatic carcinoma[38](A) Chemical structure of pNIR-4; (B) absorption and emission spectra of pNIR-4; (C) variation of photoluminescence intensity of pNIR-4 with water volume fraction in THF/H2O mixtures; (D) tumor resection guided by pNIR4-PAE NPs imaging. Copyright 2025, American Chemical Society.
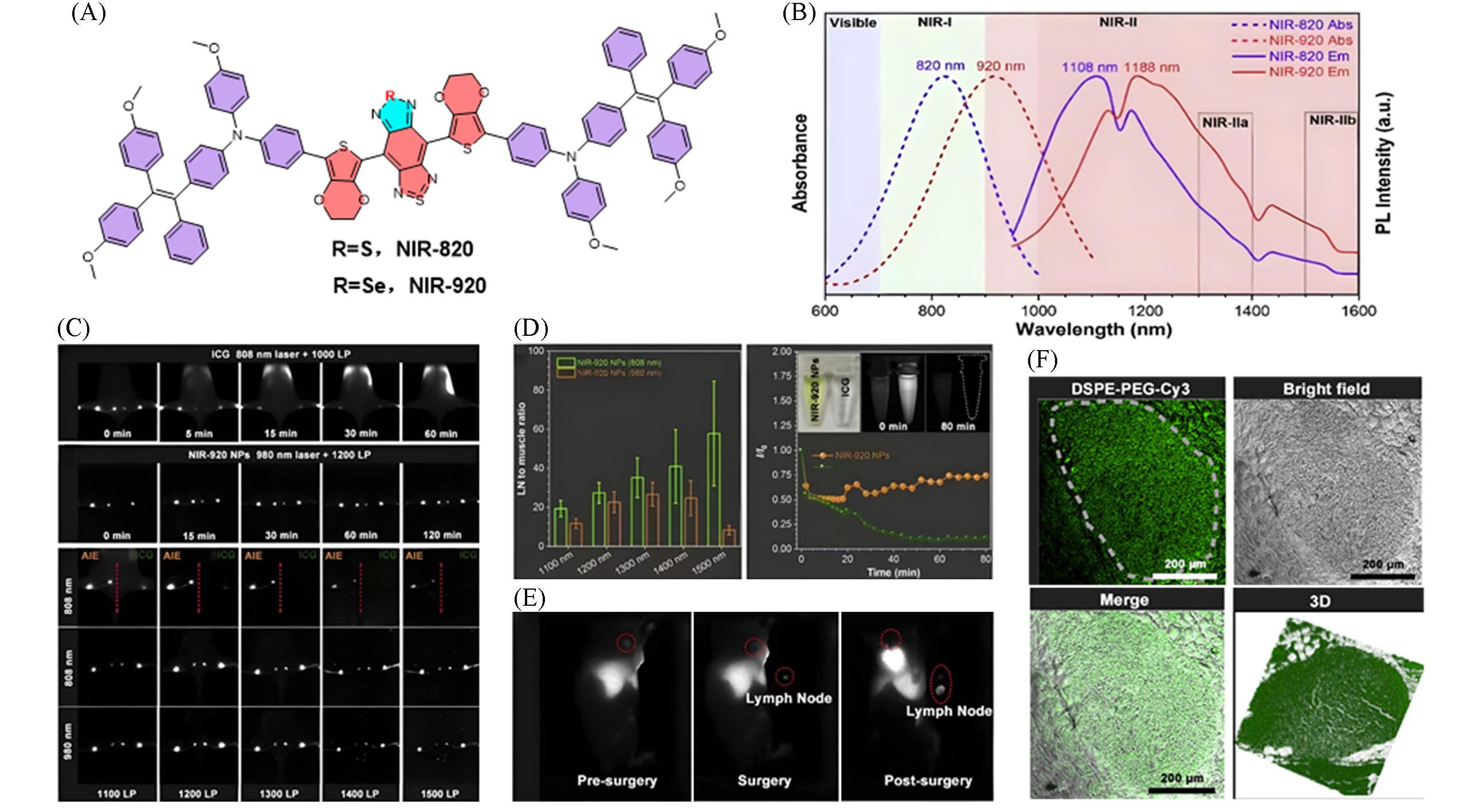
Fig.15 Construction of a highly bright NIR⁃II fluorescent probe via atomic substitution regulationfor lymph node surgical navigation[39](A) Chemical structures of NIR-820 and NIR-920; (B) normalized absorption and PL spectra of NIR-820 and NIR-920 dissolved in CHCl3; (C) lymph node imaging after injection of NIR-920 NPs and ICG; (D) comparison of lymph node imaging effects and signal-to-noise ratios(SNR) of NIR-920 and ICG under different lasers and filters; (E) before and after lymph node resection surgery; (F) paraffin sections of lymph nodes using Cy3-doped NIR-920 nanoparticles for confocal imaging. Copyright 2022, Elsevier B.V.
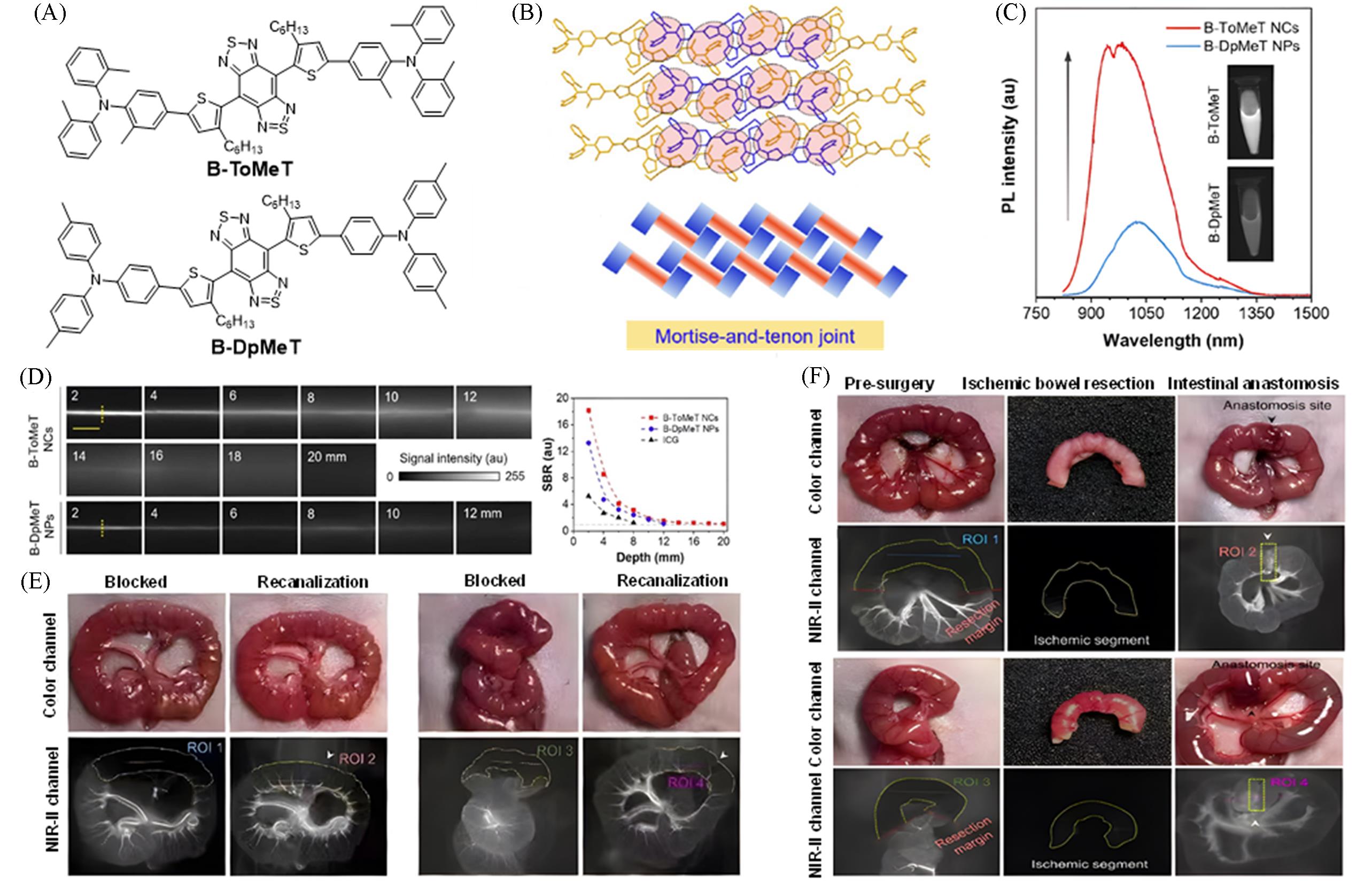
Fig.16 Construction of a highly bright NIR⁃II nanocrystal probe via a mortise⁃tenon interlocking structure for surgical navigation of acute intestinal ischemia[40](A) Chemical structures of B-ToMeT and B-DpMeT; (B) packing arrangement and schematic illustration of B-ToMeT; (C) photoluminescence(PL) spectra of B-DpMeT NPs and B-ToMeT NPs; (D) comparison of fluorescence imaging performance and signal-to-background ratios(SBR) of B-ToMeT NCs and B-DpMeT NPs in simulated tissue penetration depth experiments; (E) real-time tracking of intestinal ischemia and reperfusion recovery processes using B-ToMeT NCs; (F) NIR-II fluorescence-guided resection and anastomosis of acute intestinal ischemic necrosis segments. Copyright 2025, American Chemical Society.
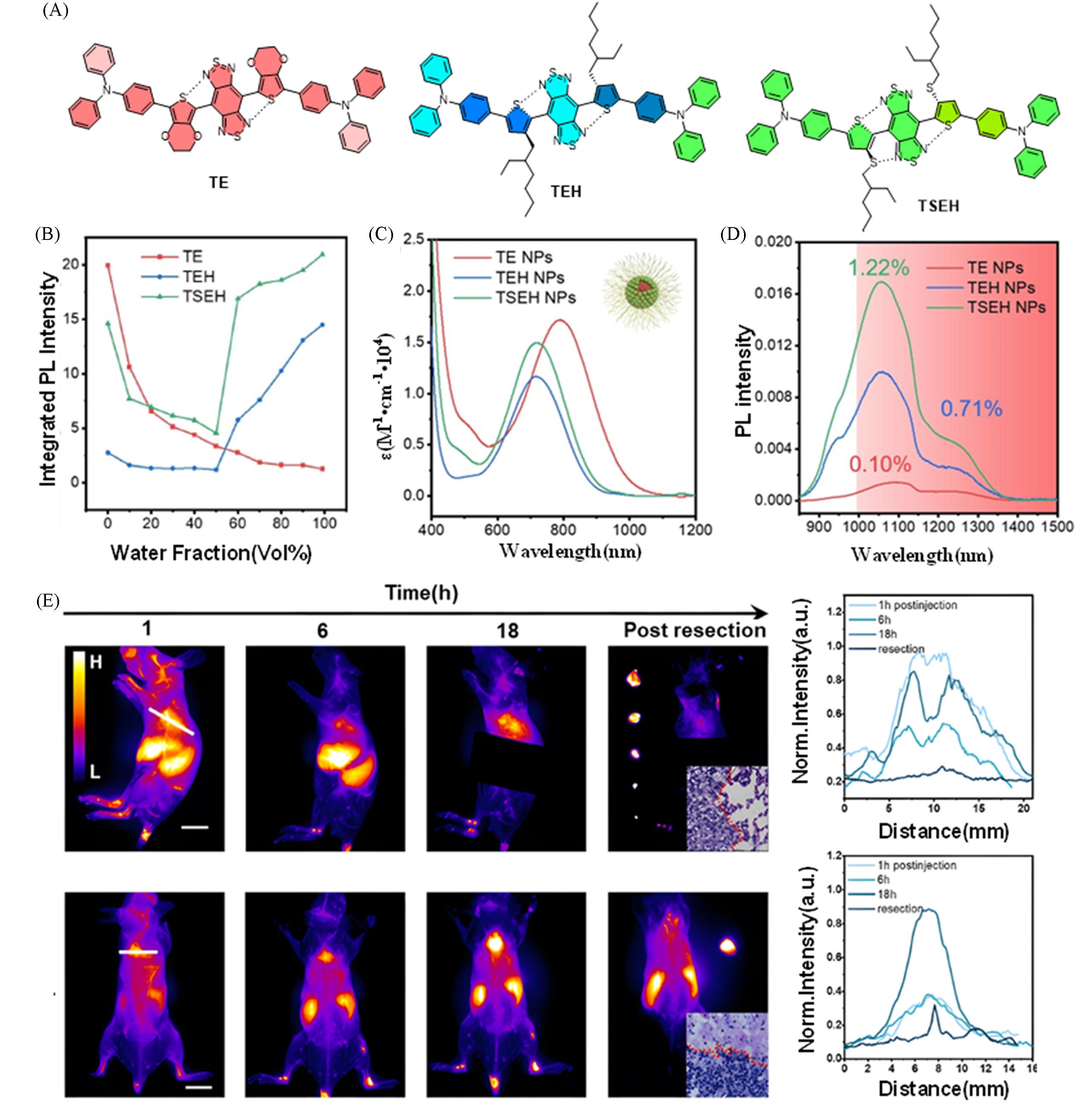
Fig.17 Construction of a bright NIR⁃II fluorescent probe via a noncovalent interlocking strategy forsurgical navigation of breast cancer and its pulmonary metastases[42](A) Chemical structures of TE, TEH and TSEH; (B) variation of fluorescence intensity of TE, TEH and TSEH with solvent water content; (C) absorption spectra in water of TE, TEH and TSEH nanoparticle solutions; (D) emission spectra of TE, TEH and TSEH nanoparticle solutions; (E) temporal dynamics of lung metastases in NIR-II imaging and tumor resection outcomes after injection of TSEH@RGD. Copyright 2024, Wiley-VCH GmbH.
| [1] | Shi Z. J., Chen S. F., Zhu H. W., Guo J. F., Xiao D. Y., Wang Z. X., Natl. Med. J. China, 2024, 104(31), 2957—2961 |
| 师忠杰, 陈四方, 朱宏伟, 郭剑峰, 肖德勇, 王占祥. 中华医学杂志, 2024, 104(31), 2957—2961 | |
| [2] | Betzig E., Patterson G. H., Sougrat R., Lindwasser O. W., Olenych S., Bonifacino J. S., Davidson M. W., Lippincott-Schwartz J., Hess H. F., Science, 2006, 313(5793), 1642—1645 |
| [3] | Blau R., Shelef O., Shabat D., Satchi⁃Fainaro R., Nat. Rev. Bioeng., 2023, 1(9), 648—664 |
| [4] | Wang F. F., Zhong Y. T., Bruns O., Liang Y. Y., Dai H. J., Nat. Photonics, 2024, 18(7), 535—547 |
| [5] | Vahrmeijer A. L., Hutteman M., van der Vorst J. R., van de Velde C. J. H., Frangioni J. V., Nat. Rev. Clin. Oncol., 2013, 10(9), 507—518 |
| [6] | Hong G., Antaris A. L., Dai H. J., Nat. Biomed. Eng., 2017, 1(1), 0010 |
| [7] | Luo J. D., Xie Z. L., Lam J. W. Y., Cheng L., Chen H. Y., Qiu C. F., Kwok H. S., Zhan X. W., Liu Y. Q., Zhu D. B., Tang B. Z., Chem. Commun., 2001, 18, 1740—1741 |
| [8] | Mei J., Leung N. L. C., Kwok R. T. K., Lam J. W. Y., Tang B. Z., Chem. Rev., 2015, 115(21), 11718—11940 |
| [9] | Yan D. Y., Wang D., Tang B. Z., Nat. Rev. Bioeng., 2025, 3(11), 976—991 |
| [10] | Dai J., Xue H. Y., Chen D. G., Lou X. D., Xia F., Wang S. X., Coord. Chem. Rev., 2022, 464, 214552 |
| [11] | He W., Zhang Z. C., Luo Y. M., Kwok R. T. K., Zhao Z., Tang B. Z., Biomaterials, 2022, 288, 121709 |
| [12] | Chen C., Ni X., Tian H. W., Liu Q., Guo D. S., Ding D., Angew. Chem. Int. Ed., 2020, 59(25), 10008—10012 |
| [13] | Qin Y., Niu N., Li X., Yan X. K., Lu S., Li Z. K., Gui Y. X., Zhu J. L., Xu L., Li X. P., Wang D. Tang B. Z., Aggregate, 2025, 6(3), e708 |
| [14] | Qi J., Duan X. C., Liu W. Y., Li Y., Cai Y. J., Lam J. W. Y., Kwok R. T. K., Ding D., Tang B. Z., Biomaterials, 2020, 248, 120036 |
| [15] | Liu S., Li Y., Zhang J., Zhang H., Wang Y., Chuah C., Tang Y., Lam J. W. Y., Kwok R. T. K., Ou H., Ding D., Tang B. Z., Mater. Today Bio., 2021, 10, 100087 |
| [16] | Li Y. Y., Zhang J. Q., Liu S. J., Zhang C., Chuah C., Tang Y. H., Kwok R. T. K., Lam J. W. Y., Ou H. L., Ding D., Tang B. Z., Adv. Funct. Mater., 2021, 31(29), 2102213 |
| [17] | Chen H. T., Bao P. P., Lv Y. H., Luo R., Deng J. Y., Yan Y. B., Ding D., Gao H. Q., ACS Appl. Mater. Interfaces, 2023, 15(49), 56895—56908 |
| [18] | Ni X., Zhang X. Y., Duan X. C., Zheng H. L., Xue X. S., Ding D., Nano Lett., 2019, 19(1), 318—330 |
| [19] | He Y. F., Yao Y. K., Yan C. X., Yu Z. W., Dai R. L., Li D., Zhu W. H., Xiao H. B., Guo Z. Q., Adv. Funct. Mater., 2025, 35(44), 2502087 |
| [20] | Ji S. L., Li S. G., Gao H. Q., Wang J. Y., Wang K. Y., Nan W. B., Chen H. L., Hao Y. W., Biomater. Sci., 2023, 11(6), 2221— 2229 |
| [21] | Zhou X. B., Liu Q. Y., Yuan W., Li Z. H., Xu Y. L., Feng W., Xu C. J., Li F. Y., Adv. Sci., 2021, 8(4), 2000441 |
| [22] | Dai J., Ouyang H. Z., Wei S. M., Chen B., Dong X. Y., Hu J. J., Wu M., Wang S. X., Xia F., Lou X. D., Anal. Chem., 2023, 95(40), 15068—15077 |
| [23] | Lu S. Y., Xue L. R., Yang M., Wang J. J., Li Y., Jiang Y. X., Hong X. C. A., Wu M. F., Xiao Y. L., Nano Res., 2022, 15(10), 9183—9191 |
| [24] | Fan X. X., Xia Q. M., Zhang Y. Y., Li Y. R., Feng Z., Zhou J., Qi J., Tang B. Z., Qian J., Lin H., Adv. Healthc. Mater., 2021, 10(24), 2101043 |
| [25] | Li R. T., Liu K. Y., Hu Q., Shen J. K., Zuo D. Q., Wang H. S., Zhu X. J., Sun W., Aggregate, 2025, 6(1), e658 |
| [26] | Wang Q., Chen B., Duan C., Wang T. T., Lou X. D., Dai J., Xia F., Anal. Chem., 2024, 96(8), 3609—3617 |
| [27] | Du J., Liu S., Zhang P. F., Liu H. X., Li Y. Y., He W., Li C. B., Chau J. H. C., Kwok R. T. K., Lam J. W. Y., Cai L. T., Huang Y. H., Zhang W. J., Hou J. Q., Tang B. Z., ACS Appl. Mater. Interfaces, 2020, 12(7), 8040—8049 |
| [28] | Chen D. D., Xiao T., Wang L. J., Chen S. J., Kam C., Zeng G. P., Peng L., Zhang J. X., Li M., Dong Y. P., Aggregate, 2024, 5(4), e550 |
| [29] | Zhong D. N., Chen W. Y., Xia Z. M., Hu R., Qi Y. C., Zhou B., Li W. L., He J., Wang Z. M., Zhao Z. J., Ding D., Tian M., Tang B. Z., Zhou M., Nat. Commun., 2021, 12(1), 6485 |
| [30] | Xu H. H., Yuan L. S., Shi Q. K., Tian Y., Hu F., Nano Lett., 2024, 24(4), 1367—1375 |
| [31] | Shi Q. K., Xu J. Y., Xu H. H., Wang Q., Huang S. H., Wang X. R., Wang P., Hu F., Small, 2024, 20(22), 2309589 |
| [32] | Ma F. L., Jia Q., Deng Z. W., Wang B. Z., Zhang S. W., Jiang J. H., Xing G. C., Wang Z. L., Qiu Z. J., Zhao Z., Tang B. Z., ACS Nano, 2024, 18(13), 9431—9442 |
| [33] | Qi J., Li J., Liu R. H., Li Q., Zhang H. K., Lam J. W. Y., Kwok R. T. K., Liu D. B., Ding D., Tang B. Z., Chem, 2019, 5(10), 2657—2677 |
| [34] | Xu Y. Q., Cui M. Y., Zhang W. C., Liu T. G., Ren X. Y., Gu Y. Q., Ran C. Z., Yang J., Wang P., Chem. Eng. J., 2022, 428, 132514 |
| [35] | Chen L. X., Xia B., Yan B., Liu J. H., Miao Z. H., Ma Y., Wang J. C., Peng H., He T., Zha Z. B., J. Mater. Chem. B, 2021, 9(15), 3317—3325 |
| [36] | Fan X. X., Xia Q. M., Liu S. J., Zheng Z., Zhang Y. Y., Wu T. X., Li Y. X., Tang G. P., Tang B. Z., Qian J., Lin H., Mater. Today Bio., 2022, 16, 100399 |
| [37] | Zhang R. Y., Shen P., Xiong Y., Wu T. J., Wang G., Wang Y. C., Zhang L. P., Yang H., He W., Du J., Wei X. D., Zhang S. W., Qiu Z. J., Zhang W. J., Zhao Z., Tang B. Z., Natl. Sci. Rev., 2023, 11(2), nwad286 |
| [38] | Liu S. J., Ou H. L., Li Y. Y., Zhang H. K., Liu J. K., Lu X. F., Kwok R. T. K., Lam J. W. Y., Ding D., Tang B. Z., J. Am. Chem. Soc., 2020, 142(35), 15146—15156 |
| [39] | Song S. L., Wang Y. J., Zhao Y., Huang W. B., Zhang F., Zhu S. J., Wu Q., Fu S., Tang B. Z., Wang D., Matter, 2022, 5(9), 2847—2863 |
| [40] | Ma F. L., Zhang R. Y., Wang B. Z., Liang Z. W., Zhang S. W., Jiang J. H., Tan H. Z., Xing G. C., Kwok R. T. K., Lam J. W. Y., Zhao Z., Tang B. Z., J. Am. Chem. Soc., 2025, 147(33), 29815—29828 |
| [41] | Xu P. F., Kang F., Yang W. D., Zhang M. R., Dang R. L., Jiang P., Wang J., Nanoscale, 2020, 12(8), 5084—5090 |
| [42] | Wang X. Y., Yang X. Q., Jiang G. Y., Hu Z. B., Liao T., Wang G. X., Zhang X., He X. Y., Zhang J. Y., Zhang J. Q., Cao W. K., Zhang K. Z., Lam J. W. Y., Sun J. W., Sun H. T., Liang Y. Y., Tang B. Z., Angew. Chem. Int. Ed., 2024, 63(29), e202404142 |
| [1] | 吴锐, 李政, 李琪, 石佳俊, 赵艳, 冯维旭, 颜红侠. 含Si, P, B的非传统超支化发光聚合物研究进展[J]. 高等学校化学学报, 2026, 47(5): 179. |
| [2] | 李伟健, 徐小琴, 王威, 杨海波. 聚集诱导发光树枝状分子的研究进展[J]. 高等学校化学学报, 2026, 47(5): 136. |
| [3] | 张凯为, 秦安军. 聚集诱导发光镧系配合物[J]. 高等学校化学学报, 2026, 47(5): 95. |
| [4] | 朱高桦, 舒菊, 耿江涛, 马夫龙, 熊玲红, 何学文. 原位激活型近红外二区聚集诱导发光探针应用于肿瘤中过氧化氢的高灵敏成像[J]. 高等学校化学学报, 2026, 47(5): 232. |
| [5] | 潘晶珂, 齐馨, 张露露, 王贝贝, 吕超. 主客体作用力构建的聚集诱导发光型超分子聚合物的合成与应用研究[J]. 高等学校化学学报, 2026, 47(5): 209. |
| [6] | 尹诗琪, 郑志刚, 何心桐, 王世敏, 顾星桂, 王二静. 从原生到功能: AIE天然产物的发光、 改性及应用[J]. 高等学校化学学报, 2026, 47(5): 75. |
| [7] | 焦海丽, 郑小燕. 限域环境诱导有机发光增强的理论研究进展[J]. 高等学校化学学报, 2026, 47(5): 20260060. |
| [8] | 翟哲, 刘雷静, 田文晶. 聚集诱导发光分子探针的设计及在细胞器成像中的应用[J]. 高等学校化学学报, 2026, 47(5): 20260078. |
| [9] | 张阳戴翼, 邵研, 姜世梅. 基于动态氢键网络协同调控AIE与力学性能的多响应水凝胶[J]. 高等学校化学学报, 2026, 47(4): 20250381. |
| [10] | 马欢, 董世龙, 杨均成, 祝海涛, 冯海涛. 基于杯[4]芳烃的手性AIEgen用于酸及氨基酸的对映选择性识别[J]. 高等学校化学学报, 2026, 47(4): 20260049. |
| [11] | 李雨婷, 罗亮. 拉曼成像技术的前沿进展及与聚集体科学的交叉研究[J]. 高等学校化学学报, 2026, 47(4): 20260008. |
| [12] | 方瑾钰, 黄瀚玮, 宋航, 吴谦, 赵征, 唐本忠. 智能响应, 精准点亮: 酶响应的聚集诱导发光材料在生物医学中的诊疗新策略[J]. 高等学校化学学报, 2026, 47(4): 20260009. |
| [13] | 孙妍, 朱东霞. 近红外激发有机发光材料的构筑及在疾病治疗中的应用[J]. 高等学校化学学报, 2026, 47(4): 20260002. |
| [14] | 杨湛, 邓皇俊, 池振国. 具有聚集诱导发光性质的氢键有机框架的研究进展[J]. 高等学校化学学报, 2026, 47(4): 20260012. |
| [15] | 李银, 汤睿霖, 瞿超, 程亮慧, 胡玉玺, 吴钰祥, 王志明. 离子化策略实现马来酸酐水溶性光敏探针的制备及其在高效抗菌中的应用[J]. 高等学校化学学报, 2026, 47(4): 20250413. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||