

高等学校化学学报 ›› 2021, Vol. 42 ›› Issue (11): 3421.doi: 10.7503/cjcu20210455
罗成1, 彭雅梅1, 沈宏1, 方群1,2, 潘建章1,2( )
)
收稿日期:2021-06-30
出版日期:2021-11-10
发布日期:2021-11-10
通讯作者:
潘建章
E-mail:kelvonpan@zju.edu.cn
基金资助:
LUO Cheng1, PENG Yamei1, SHEN Hong1, FANG Qun1,2, PAN Jianzhang1,2( )
)
Received:2021-06-30
Online:2021-11-10
Published:2021-11-10
Contact:
PAN Jianzhang
E-mail:kelvonpan@zju.edu.cn
Supported by:摘要:
免疫分析是临床上检测生理相关蛋白质指标的主要方法. 与单一指标的免疫分析相比, 临床体外诊断对多指标的生理相关蛋白质免疫分析有着更为广泛的需求. 通过在固相载体上完成免疫反应的非均相免疫分析具有灵敏度高的优点, 是当前多指标免疫分析的主流方法. 本文按照固相载体的不同, 对近年来的多指标免疫分析系统分别从技术原理、 实现方法、 各自特点等方面的研究进展进行综合阐述. 最后对比总结了不同系统的优势与不足, 并展望了微流控多指标免疫分析在即时检测领域的发展前景.
中图分类号:
TrendMD:
罗成, 彭雅梅, 沈宏, 方群, 潘建章. 多指标免疫分析的研究进展. 高等学校化学学报, 2021, 42(11): 3421.
LUO Cheng, PENG Yamei, SHEN Hong, FANG Qun, PAN Jianzhang. Research Progress of Multiplex Immunoassay. Chem. J. Chinese Universities, 2021, 42(11): 3421.
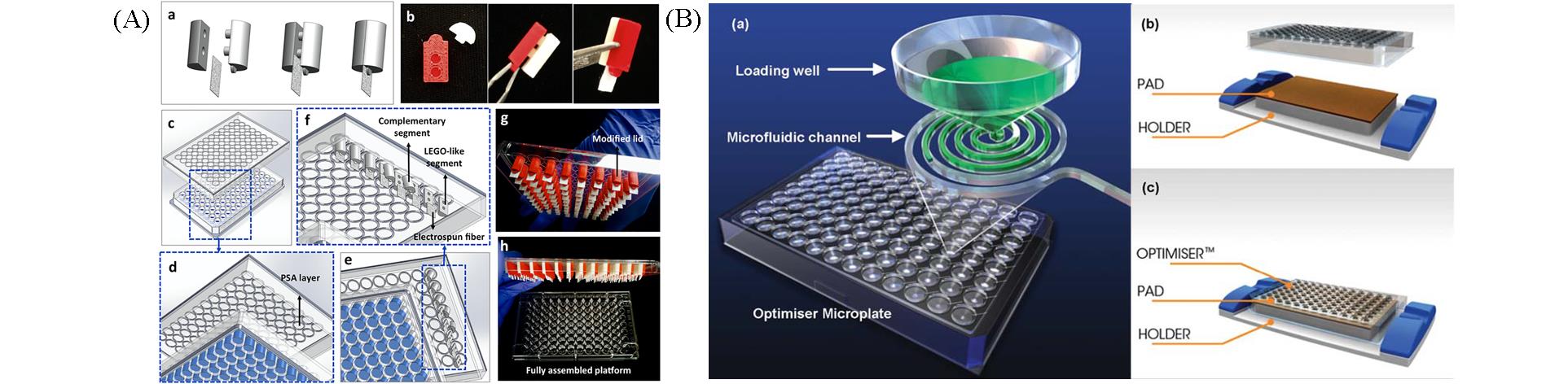
Fig.1 Immunoassay systems based on 96?well plates(A) An immunoassay system using electrospun fibers as protein carrier and 96?well plate as reaction tube[16].The complementary pieces of 3D printed cylinder probes clamp electrospun fibers(a, b). The lid of the conventional 96?well plate is modified to incorporate cylinder probes(c―f). Fully assembled platform(g, f). Copyright 2020, Elsevier. (B) The OptimizerTM microplate developed by Siloam Biosciences company[20]. A reaction unit is composed of a loading well and a microfluidic channel(a). The outlet of the microfluidic channel is connected to the absorbent pad under themicroplate(b, c).Copyright 2012, Royal Society of Chemistry.
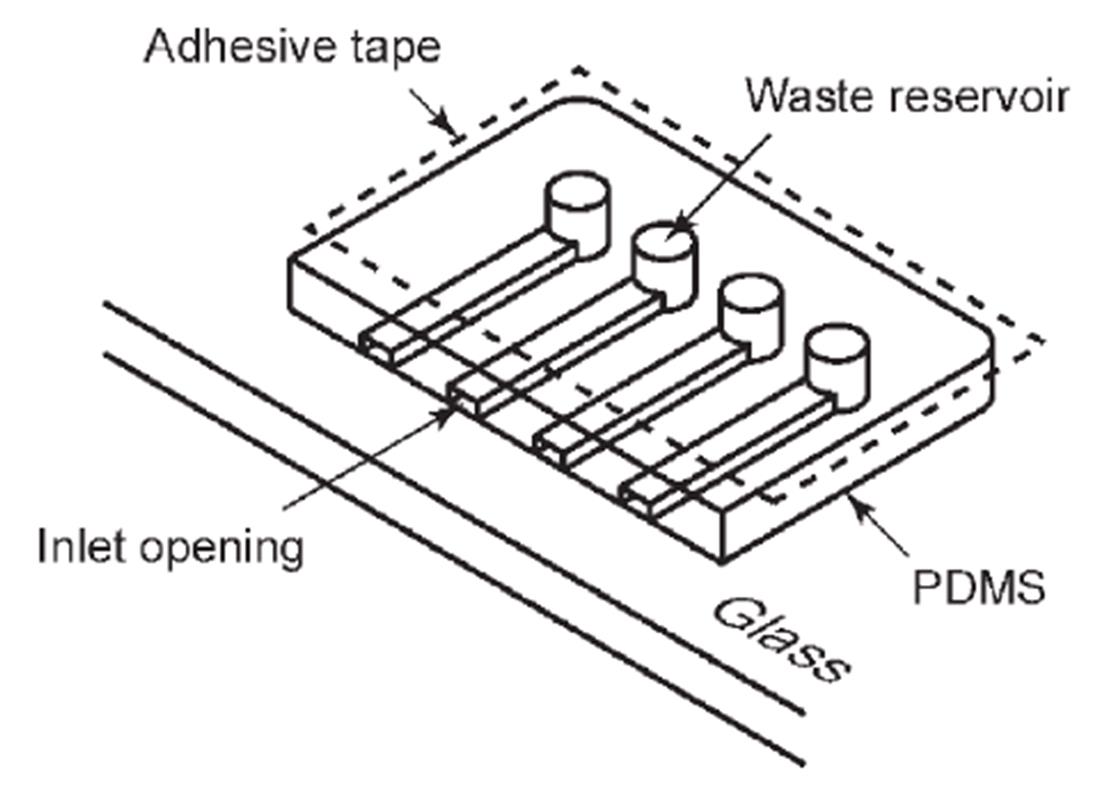
Fig.2 A microchip immunoassay with power?free sequential injection[28]The adhesive tape is attached after the degassing of the microchip. At atmospheric pressure, air dissolves into the PDMS again through all the exposed surfaces including walls of the microchannel and the reservoir. When the inlet opeaning is plugged with the reagent, the dissolution of air reduces air pressure in the channel-reservoir system, because air is no longer supplied from the outside. The pressure reduction leads the reagent into the microchannel.Copyright 2006, Royal Society of Chemistry.
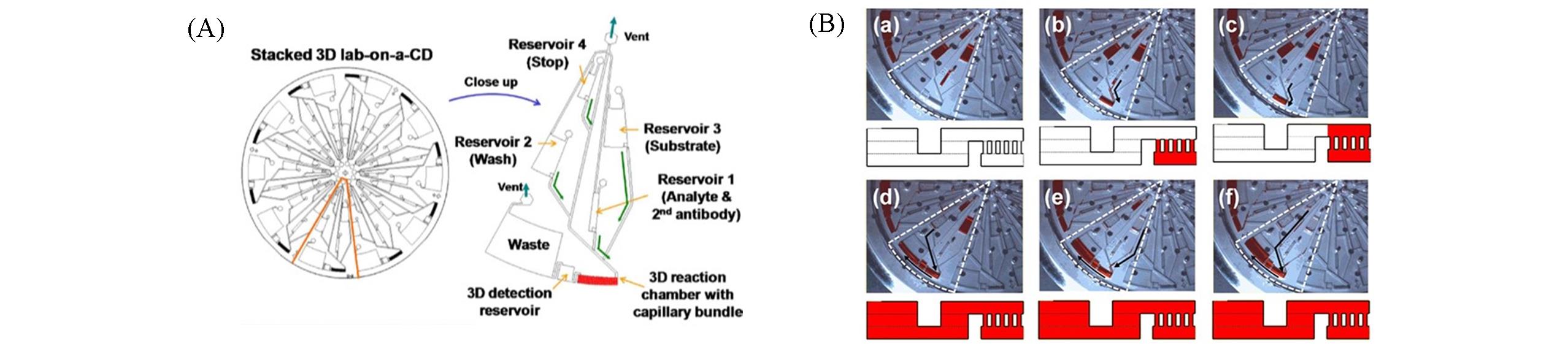
Fig.3 Stacked centrifugal microfluidic immunoassay chip with three?dimensional[30](A) Each reaction unit is loaded with stop reagent, substrate, washing buffer, and analyte from near to far from the center of the chip. (B) (a) All the regents are stored in the reservoir at 0 r/min. (b) Reservoir 1 releases the sample.(c) The sample fills the reaction reservoir. (d) Reservoir 2 releases the washing buffer. (e) Reservoir 3 releases the substrate. (f) Reservoir 4 releases the stopping reagent. Copyright 2012, Elsevier.
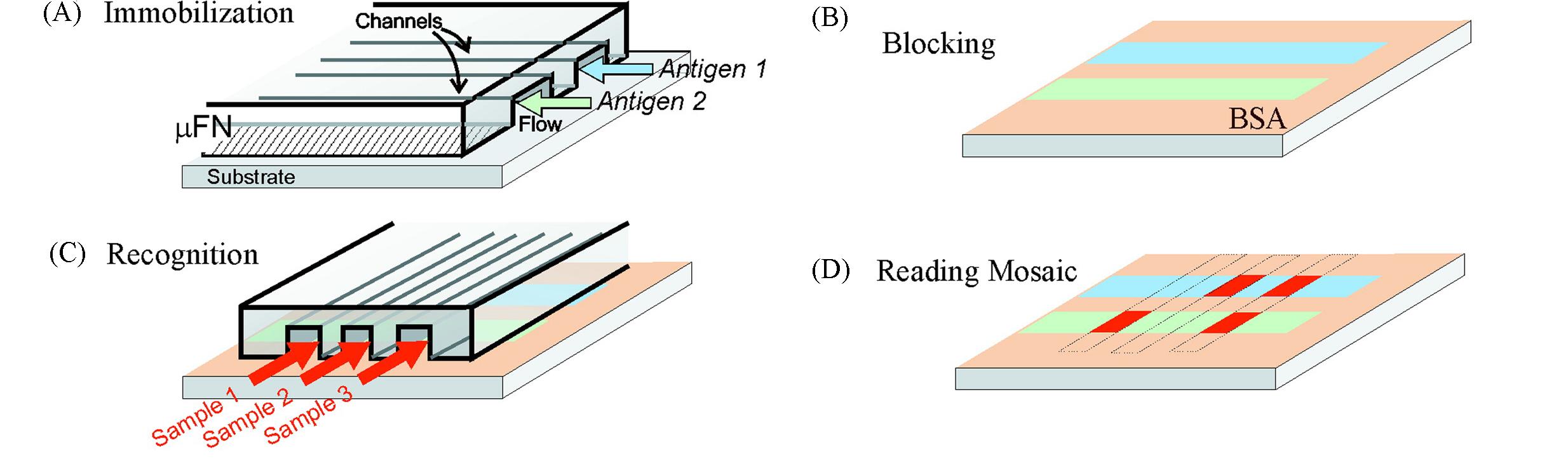
Fig.4 Principle for performing a μMIA on a surface with microfluidic network cross?delivery of a series of antigens and one of antibodies[32](A) A microfluidic network patterns different antigen molecules along single lines on a substrate. (B) The area of the substrate left unpatterned is blocked with BSA. (C) Sample flowing through the channels of a second microfluidic network. (D) Reading the binding mosaic reveals the amount of antibodies present in the samples. Copyright 2001, American Chemical Society.
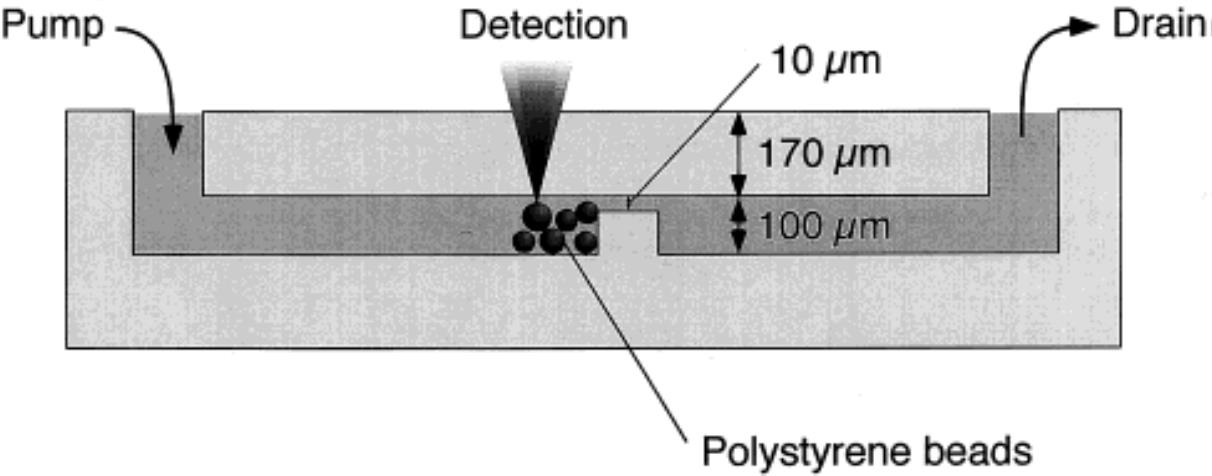
Fig.5 Layout of the glass microfluidic immunoassay chip with a dam structure[34]The channel depth is 100 μm, and a 90 μm dam is designed in the middle of the channel to prevent the microbeads from flowing out of the channel. The reaction reagents can flow through the 10 μm channel above the dam. Copyright 2000, American Chemical Society.

Fig.6 Microfluidic immunoassay chip with five chambers in series[39]The PDMS layer contains five reaction chambers (5 mm in diameter), the top layer contains two holes for inlets. Each chamber contains liquids with typical volumes of 7 μL. Between two chambers, four pillars(200 μm width, 200 μm length, and 300 μm height) are used as a robust interfacial barrier. Copyright 2015, Elsevier.
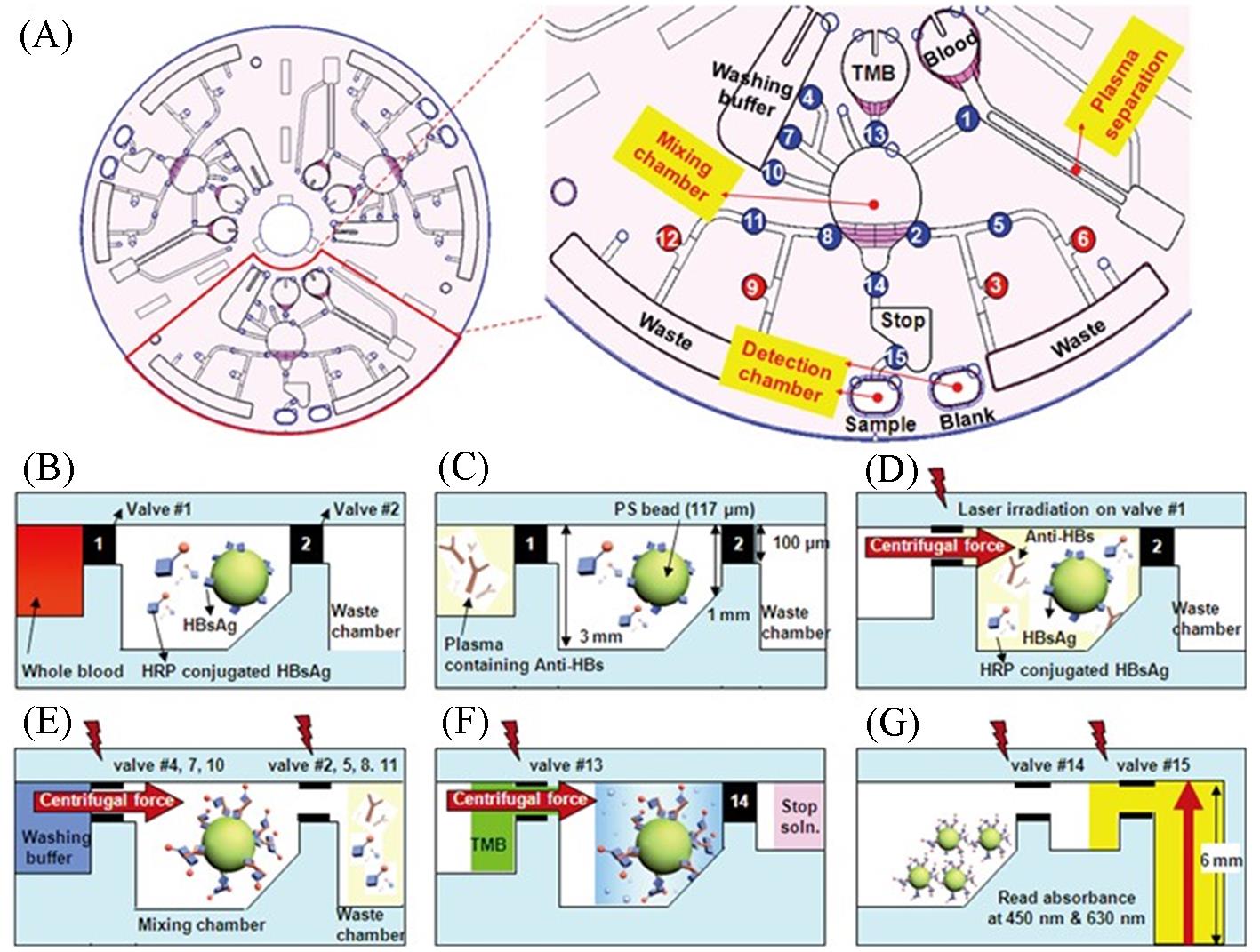
Fig.7 A centrifugal microfluidic chip for automated immunoassay of whole blood[40](A) The overall structure and unit composition of the centrifugal microfluidic chip. The number indicates the order of the laser irradiated ferrowax microvalve operation. (B)—(G) Schematic diagram of the reaction principle of the ELISA on a centrifugal microfluidic chip. The lighting symbols indicate the laser irradiation on the laser irradiated ferrowax microvalve. Copyright 2009, Royal Society of Chemistry.
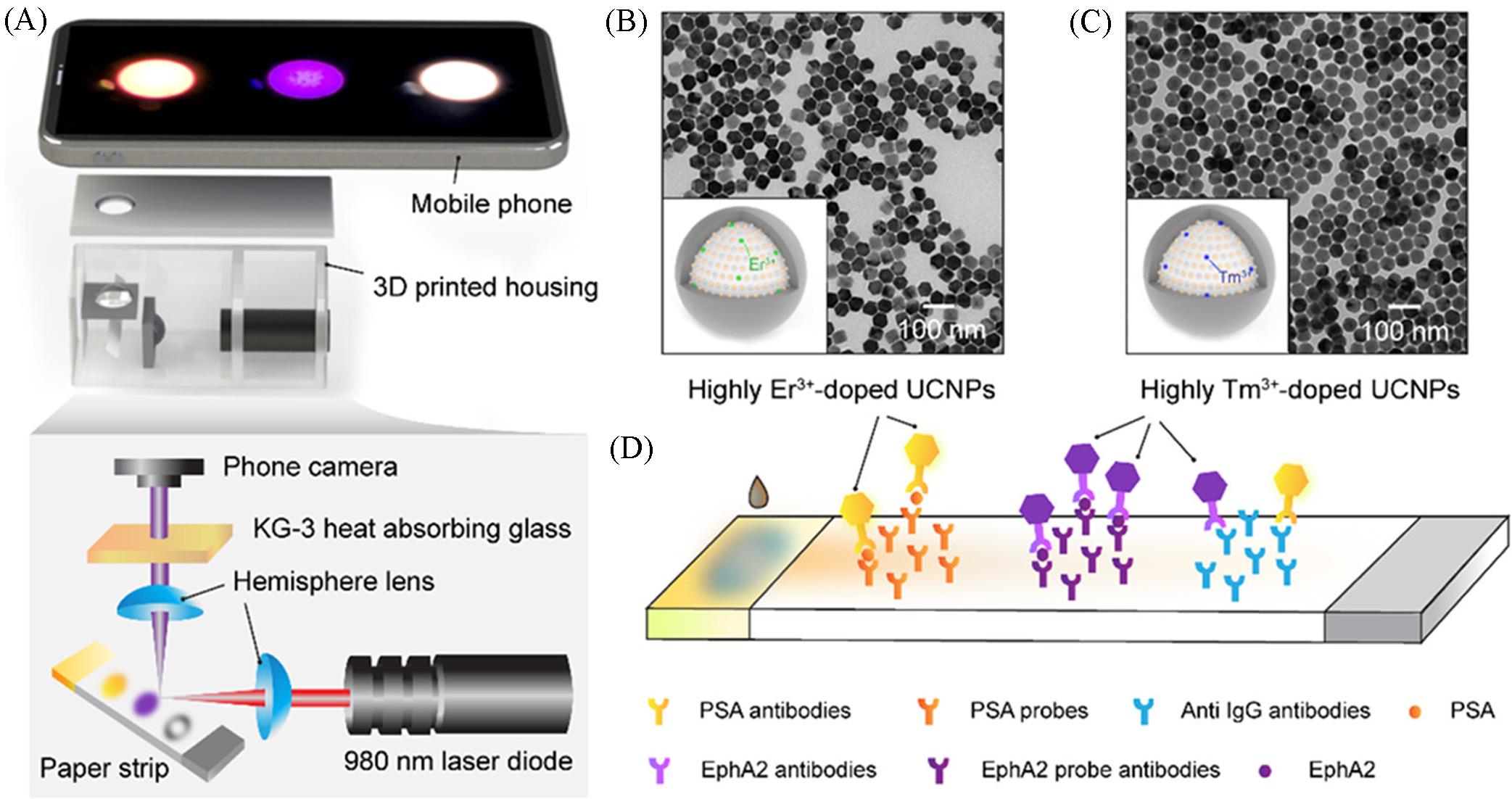
Fig.9 Quantitative lateral flow strip(LFS) sensor using highly doped upconversion nanoparticles as reporters and a phone camera as the readout element[54](A) A mobile phone?based reader and the optics layout; (B) structure of highly Er3+?doped core?shell upconversion nanoparticles(UCNPs) and the corresponding TEM image; (C) structure of highly Tm3+?doped core?shell UCPNs and the corresponding TEM image; (D) two?color LFS assays for PSA and EphA2 analytes. Copyright 2018, American Chemical Society.
| 1 | Yalow R. S., Berson S. A., J. Clin. Invest., 1960, 39(7), 1157—1175 |
| 2 | Takagi H., Hagiwara D., Handa T., Sugiyama M., Onoue T., Tsunekawa T., Ito Y., Iwama S., Goto M., Suga H., Banno R., Takahashi K., Matsui S., Arima H., Endocr. J., 2020, 67(3), 267—274 |
| 3 | Dlugash G., Schultheiss O. C., Psychoneuroendocrino., 2021, 127, 105175 |
| 4 | Rasmi R. R., Kadwad V. B., Sarnaik J., Shenoy K. B., Somashekarappa H. M., J. Radioanal. Nucl. Ch., 2021, 327(2), 923—928 |
| 5 | Yang F., Xu L., Dias A. C. P., Zhang X., Food Chem., 2021, 350, 129196 |
| 6 | Li Y., Lin Z., Hong C., Huang Z., Food Chem., 2021, 345, 128812 |
| 7 | Poturnayova A., Szabo K., Tatarko M., Hucker A., Kocsis R., Hianik T., Food Control, 2021, 123, 107774 |
| 8 | Soni D., Sahoo I., Mallya A. D., Kamthe P., Sahai A., Goel S. K., Kulkarni P. S., Dhere R. M., J. Immunol. Methods, 2021, 492, 112939 |
| 9 | He S., Yang M., Wu X., Cai G., Jiang K., Xie L., J. Microbiol. Meth., 2020, 173, 105921 |
| 10 | Li H., Zhao M., Liu W., Chu W., Guo Y., Talanta, 2016, 147, 430—436 |
| 11 | Zhao C., Niu L., Wang X., Sun W., Bioelectrochemistry, 2020, 135, 107585 |
| 12 | Sun Z., Zhang X., Xu D., Liu J., Yu R., Jing C., Han H., Ma W., Talanta, 2021, 225, 121963 |
| 13 | Zhang L., Zhou X., Dai Y., Lv C., Wu J., Wu Q., Li T., Wang Y., Xia P., Pei H., Huang B., J. Clin. Lab. Anal., 2021, 35(5), 1—8 |
| 14 | Gallian B., Zaeimian M. S., Hau D., Aucoin D., Zhu X., Frontiers in Physics, 2021, 8, 625424 |
| 15 | Manz A., Graber N., Widmer H. M., Sensor. Actuat. B: Chem., 1990, 1(1), 244—248 |
| 16 | Hosseini S., Azari P., Cardenas⁃Benitez B., Martínez⁃Guerra E., Aguirre⁃Tostado F. S., Vázquez⁃Villegas P., Pingguan⁃Murphy B., Madou M. J., Martinez⁃Chapa S. O., Materials Science and Engineering: C, 2020, 109, 110629 |
| 17 | Sun S., Matsui K., Tanabe S., Nguyen D., Kinoshita T., Yamamoto Y., Shiigi H., Anal. Sci., 2020, 36(7), 787—789 |
| 18 | Matsui K., Tanabe S., Sun S., Nguyen D., Kinoshita T., Yamamoto Y., Shiigi H., Anal. Sci., 2020, 36(12), 1461—1465 |
| 19 | Sapsford K. E., Francis J., Sun S., Kostov Y., Rasooly A., Anal. Bioanal. Chem., 2009, 394(2), 499—505 |
| 20 | Kai J., Puntambekar A., Santiago N., Lee S. H., Sehy D. W., Moore V., Han J., Ahn C. H., Lab Chip, 2012, 12(21), 4257—4262 |
| 21 | Zhang Z., Yu X., Zhao J., Shi X., Sun A., Jiao H., Xiao T., Li D., Chen J., Chemosphere, 2020, 246, 125622 |
| 22 | Renzi R. F., Stamps J., Horn B. A., Ferko S., Vandernoot V. A., West J. A. A., Crocker R., Wiedenman B., Yee D., Fruetel J. A., Anal. Chem., 2005, 77(2), 435—441 |
| 23 | Meagher R. J., Hatch A. V., Renzi R. F., Singh A. K., Lab Chip, 2008, 8(12), 2046—2053 |
| 24 | Xiao Q., Wu J., Dang P., Ju H., Anal. Chim. Acta, 2018, 1032, 130—137 |
| 25 | Guo W., Ding H., Gu C., Liu Y., Jiang X., Su B., Shao Y., J. Am. Chem. Soc., 2018, 140(46), 15904—15915 |
| 26 | Banaei N., Foley A., Houghton J. M., Sun Y., Kim B., Nanotechnology, 2017, 28(45), 455101 |
| 27 | Li P., Sherry A. J., Cortes J. A., Anagnostopoulos C., Faghri M., Biomed. Microdevices, 2011, 13(3), 475—483 |
| 28 | Hosokawa K., Omata M., Sato K., Maeda M., Lab Chip, 2006, 6(2), 236—241 |
| 29 | Gao X., Jiang L., Su X., Qin J., Lin B., Electrophoresis, 2009, 30(14), 2481—2487 |
| 30 | Ukita Y., Kondo S., Azeta T., Ishizawa M., Kataoka C., Takeo M., Utsumi Y., Sensors. Actuat. B: Chem., 2012, 166, 898—906 |
| 31 | He H., Yuan Y., Wang W., Chiou N., Epstein A. J., Lee L. J., Biomicrofluidics, 2009, 3(2), 22401 |
| 32 | Bernard A., Michel B., Delamarche E., Anal. Chem., 2001, 73(1), 8—12 |
| 33 | Yu W., Chen Y., Knauer M., Dietrich R., Märtlbauer E., Jiang X., Food Anal. Method, 2016, 9(11), 3163—3169 |
| 34 | Sato K., Tokeshi M., Odake T., Kimura H., Ooi T., Nakao M., Kitamori T., Anal. Chem., 2000, 72(6), 1144—1147 |
| 35 | Sato K., Yamanaka M., Takahashi H., Tokeshi M., Kimura H., Kitamori T., Electrophoresis, 2002, 23(5), 734—739 |
| 36 | Zhao M., Li X., Zhang Y., Wang Y., Wang B., Zheng L., Zhang D., Zhuang S., Food Chem., 2021, 339, 127857 |
| 37 | Viirlaid E., Ilisson M., Kopanchuk S., Mäeorg U., Rinken A., Rinken T., Environ. Monit. Assess., 2019, 191(8), 507 |
| 38 | Herrmann M., Veres T., Tabrizian M., Lab Chip, 2006, 6(4), 555—560 |
| 39 | Kim J. A., Kim M., Kang S. M., Lim K. T., Kim T. S., Kang J. Y., Biosensors and Bioelectronics, 2015, 67, 724—732 |
| 40 | Lee B. S., Lee J., Park J., Lee J., Kim S., Cho Y., Ko C., Lab Chip, 2009, 9(11), 1548 |
| 41 | Lou S. C., Patel C., Ching S. F., Gordon J., Clin. Chem., 1993, 39(4), 619—624 |
| 42 | Liu G., Mao X., Phillips J. A., Xu H., Tan W., Zeng L., Anal. Chem., 2009, 81(24), 10013—10018 |
| 43 | Rodríguez M. O., Covián L. B., García A. C., Blanco⁃López M. C., Talanta, 2016, 148, 272—278 |
| 44 | Deng J., Yang M., Wu J., Zhang W., Jiang X., Anal. Chem., 2018, 90(15), 9132—9137 |
| 45 | Bai Y., Tian C., Wei X., Wang Y., Wang D., Shi X., RSC Adv., 2012, 2(5), 1778 |
| 46 | Wang J., Cao F., He S., Xia Y., Liu X., Jiang W., Yu Y., Zhang H., Chen W., Talanta, 2018, 176, 444—449 |
| 47 | Gao X., Zheng P., Kasani S., Wu S., Yang F., Lewis S., Nayeem S., Engler⁃Chiurazzi E. B., Wigginton J. G., Simpkins J. W., Wu N., Anal. Chem., 2017, 89(18), 10104—10110 |
| 48 | Liu G., Lin Y., Wang J., Wu H., Wai C. M., Lin Y., Anal. Chem., 2007, 79(20), 7644—7653 |
| 49 | Ojaghi A., Pallapa M., Tabatabaei N., Rezai P., Sensor. Actuator. A: Phys., 2018, 273, 189—196 |
| 50 | Li M., Wang H., Sun J., Ji J., Ye Y., Lu X., Zhang Y., Sun X., Food Control, 2021, 121, 107616 |
| 51 | Chen Y., Sun J., Xianyu Y., Yin B., Niu Y., Wang S., Cao F., Zhang X., Wang Y., Jiang X., Nanoscale, 2016, 8(33), 15205—15212 |
| 52 | Wang C., Hou F., Ma Y., Biosensors and Bioelectronics, 2015, 68, 156—162 |
| 53 | Fang C., Chou C., Yang Y., Wei⁃Kai T., Wang Y., Chan Y., Anal. Chem., 2018, 90(3), 2134—2140 |
| 54 | He H., Liu B., Wen S., Liao J., Lin G., Zhou J., Jin D., Anal. Chem., 2018, 90(21), 12356—12360 |
| 55 | Rong Z., Wang Q., Sun N., Jia X., Wang K., Xiao R., Wang S., Anal. Chim. Acta, 2019, 1055, 140—147 |
| [1] | 王方圆, 张奋娴, 李毅, 高建华, 牛颜冰, 申少斐. 仿生树叶模型的制作及在琼脂糖微流控芯片中的应用[J]. 高等学校化学学报, 2022, 43(11): 20220445. |
| [2] | 刘园, 邓瑾琦, 赵帅, 田飞, 李轶, 孙佳姝, 刘超. 基于分子识别的免疫层析技术用于新冠肺炎感染的快速诊断[J]. 高等学校化学学报, 2021, 42(11): 3390. |
| [3] | 曹芷源, 孙慧, 苏彬. 量子点电化学发光研究进展及展望[J]. 高等学校化学学报, 2020, 41(9): 1945. |
| [4] | 彭伙, 高则航, 廖承悦, 王晓冬, 周洪波, 赵建龙. 一种用于核酸绝对定量检测的高鲁棒性液滴式数字PCR芯片[J]. 高等学校化学学报, 2020, 41(8): 1760. |
| [5] | 张作然, 张利, 张志凌. 基于侧向磁泳的微流控芯片对两种磁性纳米球的同时分选[J]. 高等学校化学学报, 2020, 41(6): 1243. |
| [6] | 李显明, 郑婷, 高露, 李峰, 侯贤灯, 吴鹏. 重组酶聚合酶扩增:扩增原理及性能分析[J]. 高等学校化学学报, 2020, 41(12): 2587. |
| [7] | 王洁,张宇,于敏,方瑾. 基于双向浓度梯度的微流控芯片系统对肿瘤细胞侵袭能力的多重分析[J]. 高等学校化学学报, 2019, 40(12): 2494. |
| [8] | 袁浩钧, 郜晚蕾, 景奉香, 刘松生, 周洪波, 贾春平, 金庆辉, 赵建龙. 一种用于核酸高灵敏检测的液滴式数字聚合酶链式反应芯片[J]. 高等学校化学学报, 2017, 38(7): 1140. |
| [9] | 姜健, 李盼盼, 马滢雪, 杨春光, 徐章润. 微流控液液萃取-液液波导集成化分析系统[J]. 高等学校化学学报, 2016, 37(4): 648. |
| [10] | 邬期望, 沈宏. 基于NUPACK预测设计的Toehold诱导链置换反应及其在DNA酶催化微流控化学发光单核苷酸多态性分析中的应用[J]. 高等学校化学学报, 2015, 36(12): 2386. |
| [11] | 朱丽娜, 祝莹, 方群. 基于微流控技术的蛋白质结晶及其筛选方法的研究进展[J]. 高等学校化学学报, 2014, 35(1): 1. |
| [12] | 解飞, 张莹, 金伟, 潘婷婷, 周超, 任昊, 金钦汉, 牟颖. 基于表面等离子体子共振成像技术研究苏拉明和顺铂对肝癌细胞生长的抑制作用[J]. 高等学校化学学报, 2014, 35(1): 30. |
| [13] | 霍丹群, 方可敬, 侯长军, 杨眉, 法焕宝, 黄承洪, 罗小刚. 水凝胶微流控芯片的快速加工及在细胞培养检测中的应用[J]. 高等学校化学学报, 2013, 34(6): 1327. |
| [14] | 朱强远, 杨文秀, 高一博, 于丙文, 邱琳, 周超, 金伟, 金钦汉, 牟颖. 一种可绝对定量核酸的数字PCR微流控芯片[J]. 高等学校化学学报, 2013, 34(3): 545. |
| [15] | 王强, 黄启欣, 孙远明, 刘英菊, 徐振林, 雷红涛. 三聚氰胺单试剂荧光偏振免疫分析法[J]. 高等学校化学学报, 2013, 34(1): 67. |
| 阅读次数 | ||||||
|
全文 |
|
|||||
|
摘要 |
|
|||||